Abstract
Background and Aims:
To characterize predictors of failure when treating critical limb ischemia (CLI) patients with an endovascular intervention as the first-line strategy.
Patients and Methods:
This retrospective, registry-based study included 217 consecutive patients with 240 chronic critically ischemic limbs treated with infrainguinal percutaneous transluminal angioplasty (PTA) during 2006–2007 at Helsinki University Central Hospital, Finland.
The primary outcome measures were death, major (above-ankle) amputation, and the need for surgical re-intervention within 6 months after the primary procedure. The secondary outcome measures were overall major amputation and survival rates as well as the overall need for surgical or any other (surgical or endovascular) type of re-intervention.
Predictors of outcome endpoints were identified with a univariate screen, and a Cox regression model was used in the multivariate analysis.
Results:
Compared to ulcer, gangrene was significantly more strongly associated with amputation within 6 months post-procedurally as well as during the whole follow-up period (p ≤ 0.028). The patient's inability to walk upon hospital arrival was a significant predictor of death, amputation and surgical re-intervention.
Mediasclerotic ankle-brachial index (ABI) was an independent predictor of amputation as well as endovascular re-interventions.
Conclusions:
The strong predictors of poor outcome after endovascular revascularization for patients with CLI are cardiac morbidity, the inability to ambulate upon hospital arrival, and gangrene as a manifestation of CLI. The risk of amputation seems to be significantly higher for gangrene than for ulcer and this matter should be taken into account in the clinical classifications for CLI.
INTRODUCTION
Patients suffering from critical limb ischemia (CLI) have traditionally been treated with surgical bypass to avoid major amputation. However, there are increasing data on the efficacy of endovascular revascularization procedures in achieving good leg salvage rates (1,–3). Indeed, the use of endovascular interventions has rapidly increased during the last decade due to, for example, their mini-invasiveness and the reduced hospital stay in comparison to bypass surgery (1, 4). Furthermore, as balloon angioplasty does not usually preclude later bypass surgery, it can be attempted rather liberally as a first-line revascularization strategy (5,–7). This study sought to define factors predicting the failure of endovascular interventions when treating patients with CLI.
PATIENTS AND METHODS
A total of 620 patients with CLI underwent lower limb revascularization at the Department of Vascular Surgery, Helsinki University Central Hospital, between January 2006 and December 2007.
Of these, 404 patients underwent bypass surgery, and 217 patients with 240 chronic critically ischemic limbs were treated with infrainguinal endovascular revascularization. All cases and angiographies were reviewed and discussed for decision making at the daily integrated vascular meetings of vascular surgeons and interventional radiologists.
The majority of the endovascular revascularizations were primary interventions; in only 18 legs the same arterial segment had been treated at least 1 year earlier with PTA.
The clinical characteristics as well as the operative and immediate postoperative outcome data of these patients were collected prospectively in our institutional database (HUSVasc) and scrutinized retrospectively. Dates of death were retrieved from the Finnish national registry (Statistics Finland).
CLI was defined as Fontaine stage III and IV disease (8). Fontaine IV was scrutinized as two groups; patients with an ulcer and those with gangrene.
Arterial lesions were classified according to the TransAtlanticInterSociety Consensus (TASC II) document (9). A run-off score according to SVS/ISCVS (10) was calculated from the completion angiogram.
Patients with acute lower limb ischemia or a patent bypass-graft in the ipsilateral leg and those who underwent hybrid revascularization were excluded from the study. Complete immediate technical failures defined as failure of crossing the arterial lesion with a guidewire were also excluded.
Chronically bedridden patients were not revascularized. The ambulatory status of the patient was recorded when the patient first was evaluated by a vascular surgeon due to the ischemic leg symptoms leading to the endovascular procedure studied. The patients walking independently or with the aid of a cane or a walker were considered as ambulatory, whereas wheelchair dependent or bed-bound patients were regarded as nonambulatory.
Dyslipidemia was defined as a total serum cholesterol level of >5 mmol/L, serum high density lipoprotein cholesterol level of < 1 mmol/L, or serum triglyceride level of > 2 mmol/L. Dyslipidemia was also assumed if a patient was on lipid-lowering medication.
Renal insufficiency was defined as serum creatinine levels of ≥ 140 μmol/L.
Patients included in the cardiac morbidity class suffered from coronary artery disease (former coronary bypass operation, a suffered myocardial infarction, angina pectoris), cardiac heart failure (admission to hospital because of heart insufficiency or medication for the treatment of cardiac heart failure), or atrial fibrillation (chronic or paroxysmal).
Vessel-related complications refer to arterial perforation, a dissection obstructing blood flow, arterial thrombosis, or distal arterial embolization.
Urgent procedure means that the intervention needs to be performed within 1–2 days.
All endovascular procedures were performed in accordance with general guidelines. Contralateral femoral access was preferred, with ipsilateral antegrade femoral access reserved for patients found to have a distal SFA, or popliteal or infrapopliteal artery occlusive disease. Intra-arterial heparin (5000 IU) was routinely administered. Lesions were preferentially treated with balloon angioplasty. Stents were placed selectively in cases where there was a flow-limiting dissection or significant residual stenosis after balloon angioplasty. One stent was applied after the recanalization of a several-centimeter-long SFA occlusion. All stents were placed at the SFA level; popliteal or infrapopliteal arteries were not stented. Completion angiography was performed to assess technical success.
After a successful endovascular intervention, patients were administered 300 mg of clopidogrel orally and maintained on 75 mg/day for at least 1 month, except for those who were already on continued anticoagulation therapy since before the revascularization. In addition, the patients were indefinitely put on 100 mg aspirin orally. Routine follow-up included one clinical examination and ankle-brachial index (ABI) measurement at one month after the procedure. ABI-measurements are routinely made by a vascular nurse using laser Doppler device (Perimed, Stockholm, Sweden). However, all patients were followed until their symptoms subsided and/or the wound healed. A duplex Doppler ultrasound scan was performed when clinically necessary.
All secondary revascularizations (including all open and endovascular re-interventions performed to maintain the original revascularization, and redo-procedures carried out due to the occlusion of the original revascularization) performed within 12 months after the primary intervention were regarded as re-interventions.
OUTCOME ENDPOINTS
The primary outcome measures were death, major (above-ankle) amputation, or the need for secondary bypass surgery within 6 months after the primary endovascular procedure.
The secondary outcome measures were the overall survival and major amputation rate, the overall need for secondary bypass surgery, the overall need for any re-intervention, and the need for endovascular re-intervention.
STATISTICAL ANALYSES
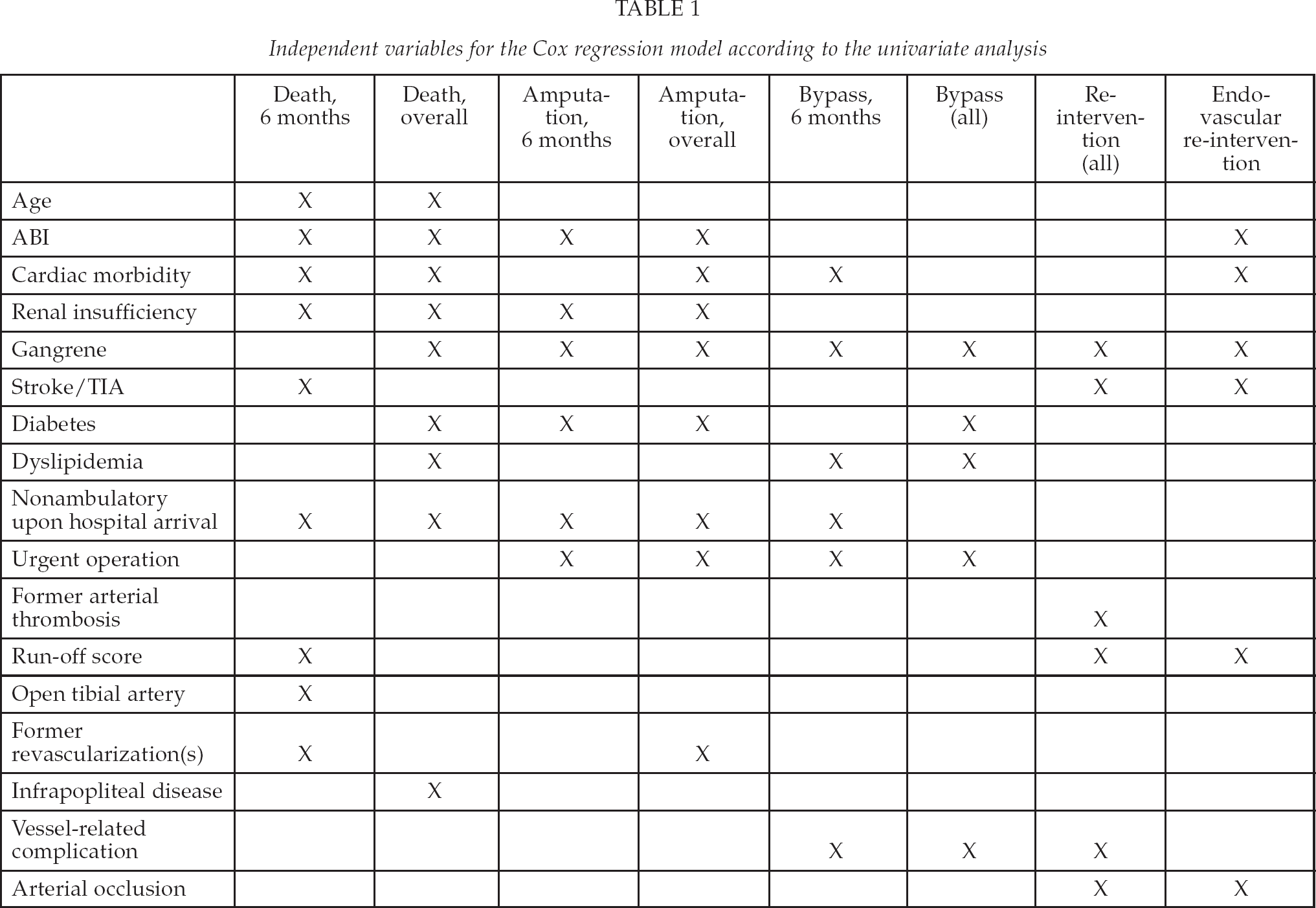
For statistical analyses SPSS 17.0 software (SPSS Inc.; Chicago, IL, USA) was used. Predictors of primary and secondary endpoints were identified with a univariate screen using p < 0.2 as a threshold for inclusion in the multivariate analyses. The independent factors considered in these are presented in Table 1. A Cox regression model with enter selection was used in the multivariate analysis for the outcome endpoint measures. Kaplan-Meier survival curves were utilized to describe the differences in survival with associated factors.
Independent variables for the Cox regression model according to the univariate analysis
RESULTS
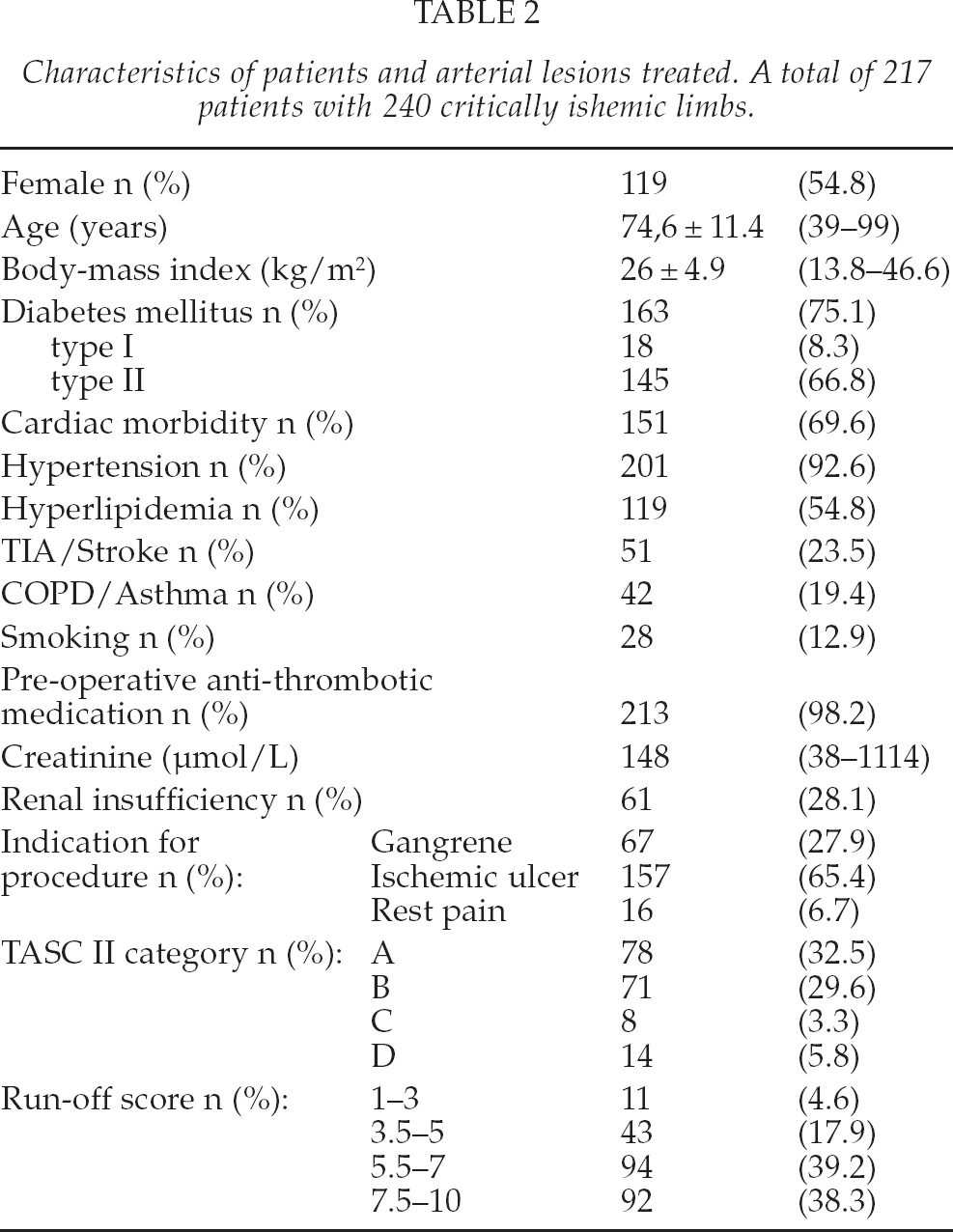
The mean follow-up was 17 months (range 1 day–36.4 months). The patient characteristics, the indications for the procedure as well as the TASC class and runoff-scores are depicted in Table 2.
Characteristics of patients and arterial lesions treated. A total of 217 patients with 240 critically ishemic limbs.
The most distal level treated was the femoropopliteal segment in 132 (55%) legs and interventions extending to infrapopliteal arteries were performed on 108 (45%) legs. Interventions and re-interventions are depicted in Fig. 1.
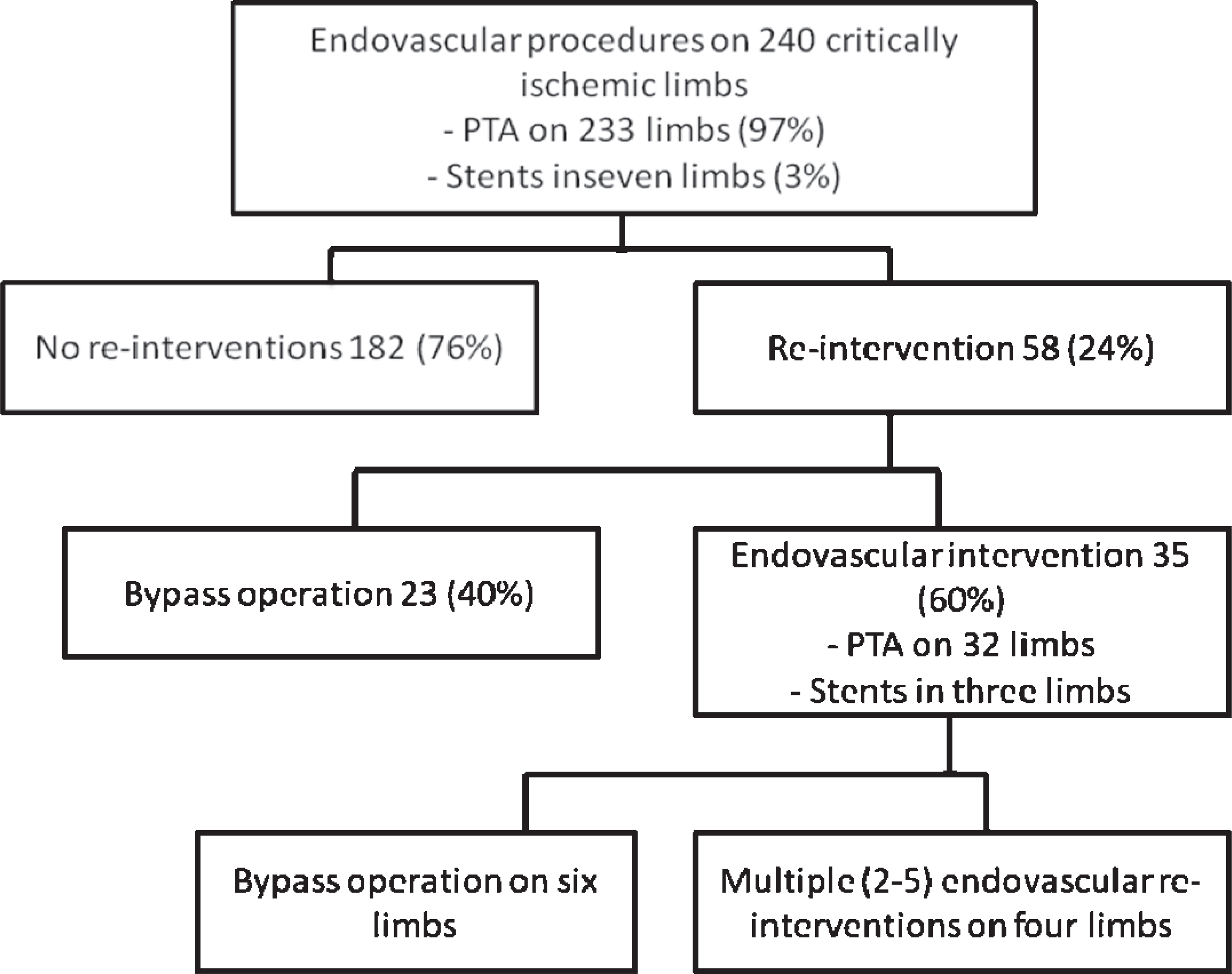
A flow-chart summarizing the need for re-interventions after endovascular intervention for critical leg ischemia.
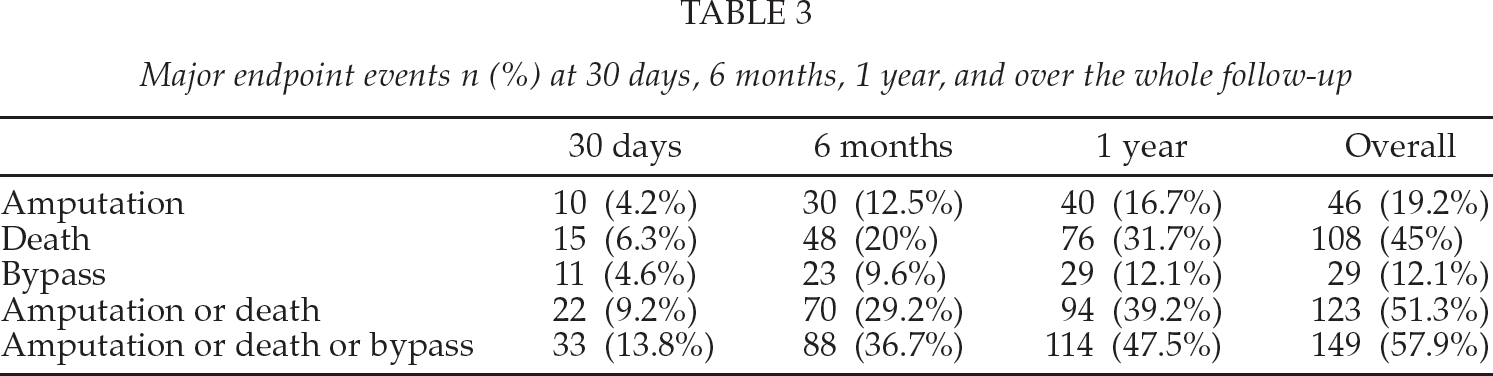
Amputation free-survival was 71%, 61%, and 49%, at 6 months, 1 year and during the whole follow-up period, respectively. Limb salvage was 88%, 83% and 80% at 6 months, 1 year and during the whole follow-up period, respectively. 37% of the patients had died, lost their limb or had a surgical re-intervention at 6 months post-procedurally and this number was 48% and 58% at 1 year and during the whole follow-up period, respectively.
Table 3 summarizes the mortality, amputation, and bypass surgery rates.
Major endpoint events n (%) at 30 days, 6 months, 1 year, and over the whole follow-up
MULTIVARIATE ANALYSIS OF THE PRIMARY OUTCOME MEASURES
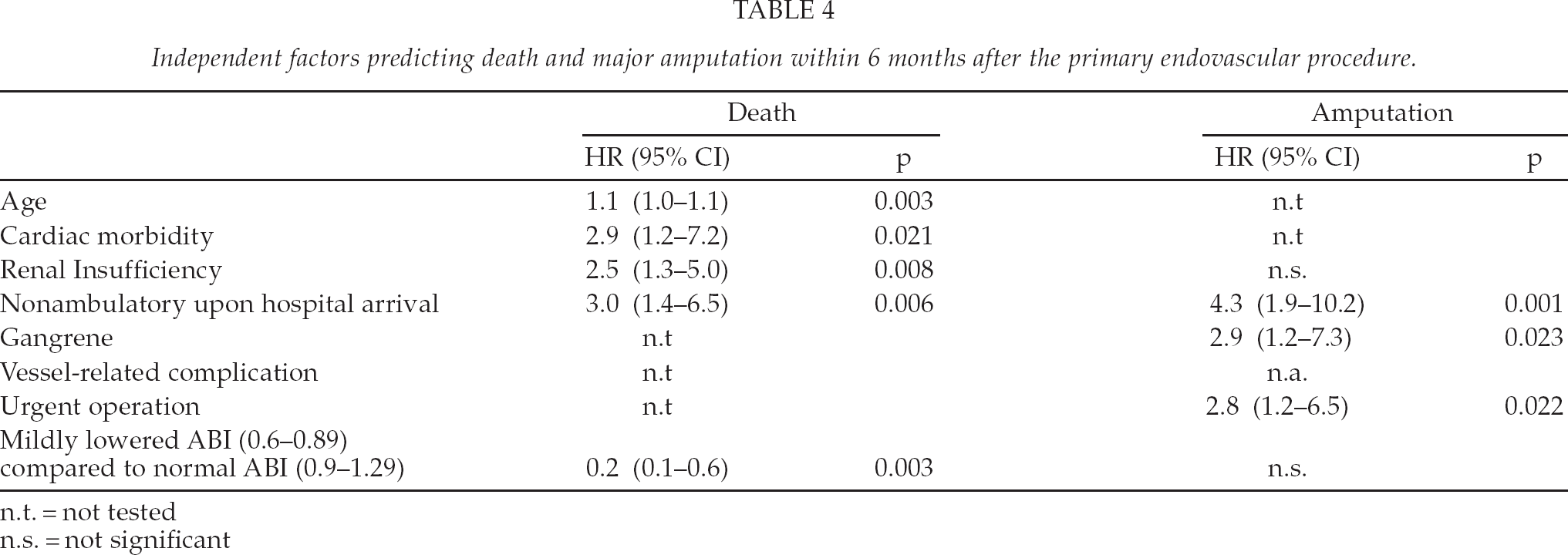
In the multivariate analysis, the independent risk factors associated with death within 6 months after the primary procedure were age, cardiac morbidity, inability to ambulate upon hospital arrival, and renal insufficiency.
Surprisingly, patients with a normal or mediasclerotic pre-procedural ABI (ABI ≥ 0.9) had the highest 6-month mortality when compared to those with a lowered (≤ 0.89) pre-procedural ABI. For instance, the patients whose pre-procedural ABI was in the range of 0.6–0.89 had a significantly lower mortality rate at 6 months when compared to those with a normal (0.9–1.29) pre-procedural ABI.
Table 4 summarizes the respective hazard ratios, confidence intervals, and p values.
Independent factors predicting death and major amputation within 6 months after the primary endovascular procedure.
n.t. = not tested
n.s. = not significant
The significant predictors of amputation within 6 months after the primary intervention were an urgent procedure, the inability to ambulate upon hospital arrival, and gangrene as the indication for the procedure. Table 4 summarizes the respective hazard ratios, confidence intervals, and p values.
Compared to ulcer, gangrene was significantly more strongly associated with amputation at 6 months (p = 0.023, HR = 3.0; 95% CI 95% 1.2–7.8) (Fig. 2).
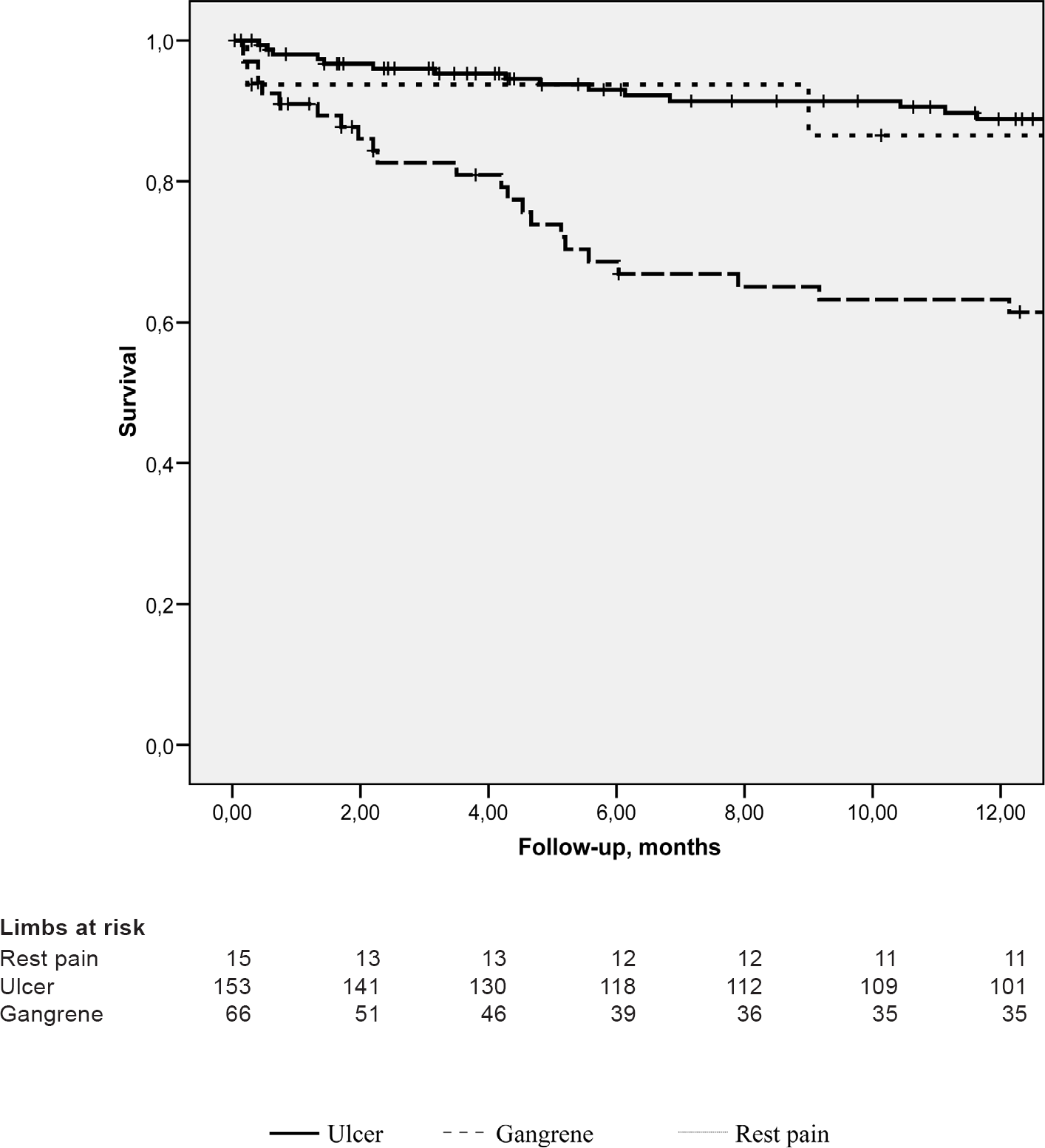
A Kaplan-Meier survival curve depicting amputation-free survival in patient groups with different indications for endovascular revascularization.
The only independent risk factor associated with the need for secondary bypass surgery within 6 months after the primary procedure was the inability to ambulate upon hospital arrival (p = 0.045, HR = 2.7; 95% CI 1.02–7.0).
MULTIVARIATE ANALYSIS OF THE SECONDARY OUTCOME MEASURES
In the multivariate analysis, the independent risk factors associated with overall mortality were age (p = 0.005, HR = 1.03; 95% CI 1.01–1.1), cardiac morbidity (p = 0.003, HR = 2.3; 95% CI 1.3–4.1), the inability to ambulate upon hospital arrival (p = 0.002, HR = 2.3; 95% CI 1.4–4.0), and renal insufficiency (p = 0.000, HR = 2.6; 95% CI 1.6–4.4). Dyslipidemia had a protecting effect in regard to overall mortality (p = 0.006, HR = 0.5; 95% CI 0.3–0.8).
The significant predictors of amputation during the whole follow-up period were an urgent procedure (p = 0.007, HR = 2.7; 95% CI 1.3–5.6), the inability to ambulate upon hospital arrival (p = 0.000, HR = 3.8; 95% CI 1.9–7.7), gangrene as the indication for the procedure (p = 0.012, HR = 2.5; 95% CI 1.2–5.2), and a mediasclerotic pre-procedural ABI (≥ 1.3) (p = 0.023, HR = 2.2; 95% CI 1.1–4.5).
Compared to ulcer, gangrene was significantly more strongly associated with amputation during the whole follow-up period (p = 0.014, HR = 2.6; 95% CI 1.2–5.4) (Fig. 2).
The independent risk factors associated with the overall need for secondary bypass surgery were an urgent procedure (p = 0.024, HR = 2.7; 95% CI 1.1–6.6) and a vessel-related complication during the primary procedure (p = 0.044, HR = 3.1; 95% CI 1.03–9.6).
The significant predictors of any re-intervention were gangrene (p = 0.000, HR = 2.6; 95% CI 1.6–4.5), a vessel-related complication during the primary procedure (p = 0.004, HR = 3.7; 95% CI 1.5–9.0), and former arterial thrombosis (p = 0.000, HR = 3.5; 95% CI 1.9–6.5).
The independent risk factors for the need for endovascular re-interventions were a mediasclerotic pre-procedural ABI (≥ 1.3) (p = 0.003, HR = 4.0; 95% CI 1.6–10.3) and gangrene (p = 0.032, HR = 2.4; 95% CI 1.1–5.2).
DISCUSSION
Patients with critical limb ischemia have severe co-morbidities -usually, coronary artery disease- and therefore high mortality. Indeed, in the current study, 9% of the patients underwent an amputation or died within 30 days and 40% within one year of the procedure. The risk of amputation is evident and revascularization is indicated to avoid it. Endovascular treatment is usually less invasive and suitable also for patients who are unfit for surgery. The number of patients is increasing as the population ages. This will inevitably lead to a situation where the resources in health care must be targeted to those who benefit from the treatments. The aim of the present study was to identify the predictors of poor outcome, i.e., those who require an amputation or die within 6 months.
The most significant risk factor for both death and amputation within 6 months after the procedure was a nonambulatory patient upon hospital arrival. Here, chronically bedridden patients were not revascularized; the nonambulatory study patients could have been able to walk before and became nonambulatory due to a more or less acute impairment in their condition. The inability to ambulate is often a sign of the patient's worsening overall condition rather than a reflection of local ischemic leg symptoms, which indicates that no benefits in terms of prognosis are to be gained even through a successful revascularization, rendering the mortality and amputation rates high in this patient group. Moreover, in old age, the general condition deteriorates quickly when patients lose their ability to walk.
Most commonly used clinical classifications for CLI (8, 11) include ischemic ulcer and focal gangrene in the same class, and these two manifestations of limb ischemia have often been discussed as one entity. Also for example, the risk scoring method PIII includes tissue loss but does not make a distinction between gangrene and ulcer (12). In the current study, however, gangrene was an independent risk factor for amputation within 6 months post-procedurally and during the whole follow-up period. Furthermore, gangrene was significantly more strongly associated with amputation than ulceration, which did not differ from rest pain as a risk factor for amputation in the present series. Biancari et al. found gangrene to be an independent risk factor for 30-day amputation and amputation-free survival, and gangrene was also a significantly stronger predictor than ulcer or rest pain (13). Gangrene is a clear consequence of ischemia and poor circulation, while an ulcer, in turn, may also have a neuropathic etiologic component, and when offloading is achieved together with revascularization, healing is more probable than in the case of gangrene. When these two significant factors were combined – i.e., nonambulatory patients with gangrene – the outcome was extremely poor: after 6 months, 67% had died or undergone major amputation, and after one year the figure was 83%.
ABI has been shown to be associated with mortality and amputation in patients with peripheral arterial occlusive disease: both lowered (< 0.9) and mediasclerotic (≥ 1.3) ABI values have been associated with increased cardiovascular mortality(14,15) and, additionally, mediasclerotic ABI values have been shown to predict major amputation in CLI patients (16).
Surprisingly, in our study population, patients with a normal pre-procedural ABI (0.9–1.29) had the highest mortality at 6 months after the primary intervention, and those with an ABI≥1.3 had the second highest 6-month mortality. However, the majority (79%) of the patients with a normal ABI had a pre-procedural toe pressure of ≤ 50, and all these patients also had clinically established CLI and angiographically ascertained infrainguinal arterial lesion(s). Additionally, 75% of them were diabetics. Hence, it seems likely that the patients with normal ABI values had, in actual fact, mediasclerotic arteries, giving falsely high ABI measurements and thus explaining the connection with higher mortality when compared to patients with an ABI< 0.9. In concordance with previous studies (15, 16), the current patients with a pre-procedural mediasclerotic ABI had a poorer overall survival and a higher overall amputation rate than those with lowered pre-procedural ABI values. Additionally, mediasclerotic pre-procedural ABI values were predictors of endovascular re-interventions, which is also in concordance with previous studies (17).
In our series, patients with dyslipidemia had a significantly lower overall mortality rate. At the end of follow-up, 39% of the patients with dyslipidemia and 50% of those without dyslipidemia had died. The difference is clear. This is probably due to the fact that practically all of these patients were on statin treatment, which has been demonstrated to be efficient in reducing death and major cardiovascular events (18, 19).
The present study was limited by the relatively small numbers of patients, especially in subgroups, and a relatively short follow-up. In addition, the retrospective nature of the study and the absence of a control group may cause bias. Concern on data validity and completeness is typically associated with registry-based studies and we have made an effort to minimize these limitations. Our vascular registry includes all patients that underwent an endovascular revascularization at our institution. The completeness of the registry data has been checked against hospital registries and missing data have been completed into the registry afterwards. The key outcome end-points were double checked against official national registries.
In conclusion, strong predictors of poor outcome after endovascular revascularization for patients with critical limb ischemia are cardiac morbidity, the inability to ambulate upon hospital arrival, and gangrene as a manifestation of critical limb ischemia. There are increasing data that the risk of amputation seems to be significantly higher for gangrene than for ulcer and this was demonstrated also in this study. This prognostic difference should be better recognized in the most commonly used clinical classifications for critical limb ischemia by dividing these two types of tissue loss into different subgroups.
Footnotes
ACKNOWLEDGEMENTS
This study was supported by a grant from the special governmental subsidy for health sciences research (TYH2011222) and a grant from the Finnish Association of Interventional Radiologists.
