Abstract
The use of nasoenteric tubes (NETs) is ubiquitous, and clinicians often take their placement, function, and maintenance for granted. NETs are used for gastrointestinal decompression, enteral feeding, medication administration, naso-biliary drainage, and specialized indications such as upper gastrointestinal bleeding. Morbidity associated with NETETs is common, but frequently subtle, mandating high index of suspicion, clinical vigilance, and patient safety protocols. Common complications include sinusitis, sore throat and epistaxis. More serious complications include luminal perforation, pulmonary injury, aspiration, and intracranial placement. Frequent monitoring and continual re-review of the indications for continued use of any NETET is prudent, including consideration of changing goals of care. This manuscript reviews NET-related complications and associated topics.
Keywords
INTRODUCTION
Nasoenteric tubes (NETs) are used for a variety of indications, including gastrointestinal decompression, enteral feeding, medication administration, naso-biliary drainage, and specialized indications such as upper gastrointestinal bleeding (from simple lavage to variceal bleeding tamponade with balloon tubes). NET-associated morbidity is rare but can be severe. The risk of complications (i.e., perforation, misplacement) generally increases with difficulty of NET placement. Common complications include sinusitis, sore throat and epistaxis. More serious complications include luminal perforation, pulmonary injury, aspiration, and intracranial placement 1). This is a comphrehensive, problem-focused review of NET complications and related topics.
GENERAL OVERVIEW OF NASOENTERIC TUBES
NETs are usually categorized by their most “distal” anatomic location (i.e., N-G or gastric, N-D or duodenal, or N-J or jejunal). Some tubes offer both gastric and small intestinal access (i.e, “G-J” tubes). NETs are usually made of silicone or polyurethane, are between 3.5–16 French in diameter and up to 170 cm long (2). Silicone tubes are thinner, pliable and non-reactive, but their walls are weaker and more prone to usage-induced damage. Polyurethane tubes are non-reactive and durable. A stylet may aid in NET passage, but it increases rigidity and injury risk. Some NETs contain non-toxic weights for easier placement. Although the nasoenteric route of insertion is usually indicated, certain situations demand oral insertion. For the purposes of this review, the two are used interchangeably, unless specifically stated.
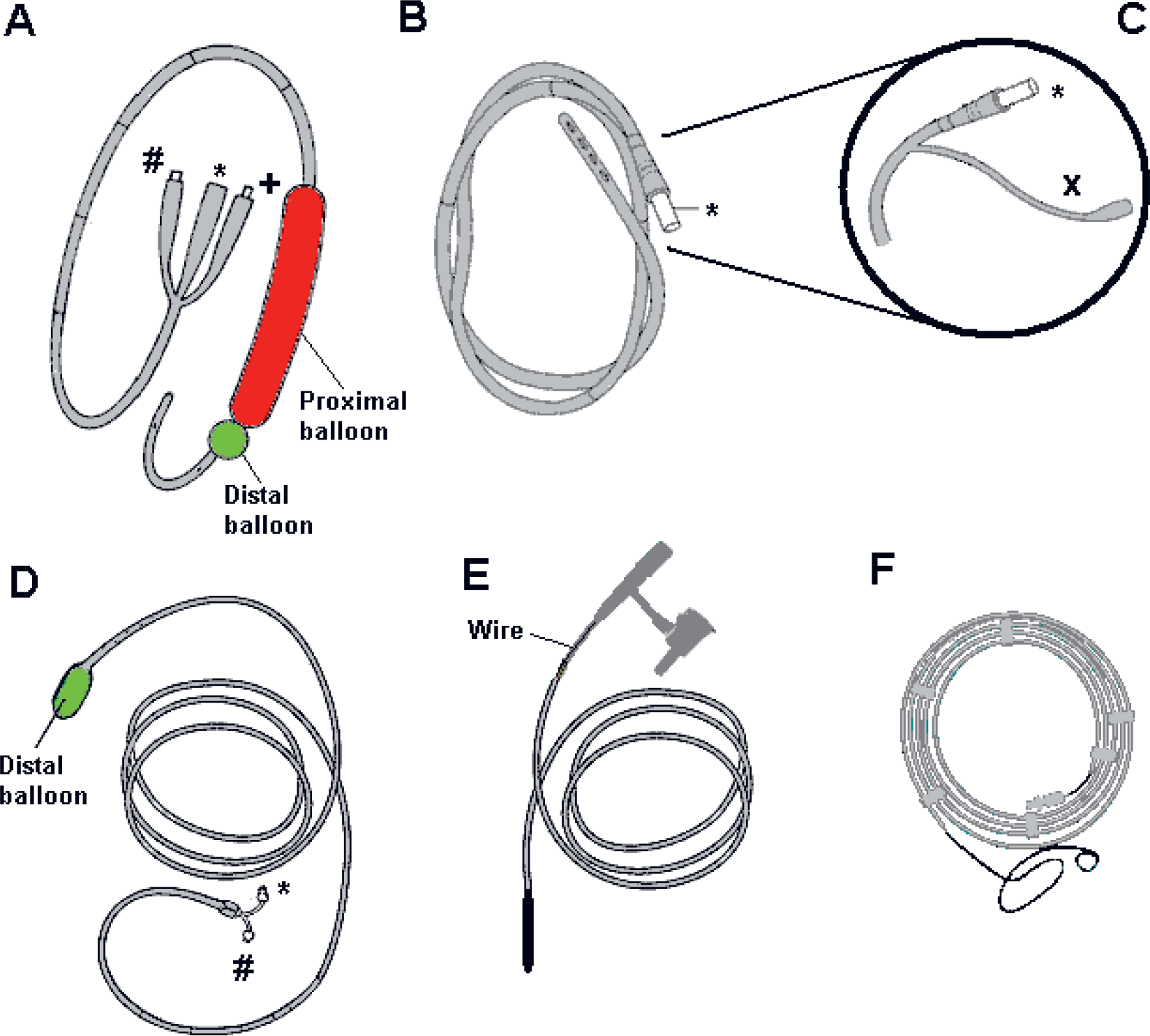
Naso-/oroenteric tube types discussed in this manuscript.
Nasoenteric feeding is the most widely used modality of enteral nutrition (EN). Early and aggressive EN maintains gastrointestinal integrity, prevents mucosal atrophy, attenuates injury-related stress, preserves immunocompetence and normal gut flora (3). Conventional nasogastric (N-G) and nasojejunal (N-J) tubes are not suitable for long-term usage in enteral feeding as they may be damaged by enteric contents and become increasingly rigid (4). By contrast, flexible polyurethane tubes suitable for prolonged use do not stiffen, become brittle, or biodegrade in vivo. Small diameter long polyurethane NETs (8–10 French diameter, 105–145 cm length) are more comfortable and less erosive, but they clog more easily. They also come with a wire stylet to facilitate nasal insertion with/without fluoroscopy. Different NET types can be seen in Fig. 1.
METHODS OF NET PLACEMENT
NETs can be placed using a number of techniques. The most common is blind placement, where the NET is inserted transnasally and advanced to the cricopharyngeus (15–20 cm in adults). The head is then flexed “chin-to-chest” to help narrow the trachea and open the esophagus. With solicited swallowing, the tube is then advanced slowly and steadily another 30–40 cm to a distance of approximately 50–60 cm in adults, leaving the tip ∼10 cm intragastric. Alternatively, the total distance from the nose to the pinna and from the pinna to the xiphoid process, plus another 5 cm, will place NET tip around gastric fundus. Intragastric placement must be confirmed by auscultation of insufflated air in the epigastrium. If air cannot be auscultated, the tube is withdrawn into the nasopharynx and replaced. Aspiration of light-colored secretions with minimal bile suggests intragastric position. Postpyloric placement is suggested by minimal aspiration of air after insufflation and return of darker bilious secretions. NET coiling is suggested by increased resistance or difficulty removing a guidewire. Tube positioning must be confirmed prior to feeding, especially when post-pyloric placement is sought in patients at risk of aspiration.
Difficult insertions may occur in patients who are endotracheally intubated (due to impaired swallowing and/or endotracheal tube cuff), have depressed mental status, or impaired cough reflex. Intraoral NET coiling is common. Placement methods in intubated patients include forward displacement of the larynx, use of a nasopharyngeal airway as obturator, immersion of the NET in ice-water, forward neck flexion and oropharyngeal air insufflation (opens upper esophageal sphincter), and occasionally direct laryngoscopy. Filling the NET with water increases success rates (93.5% vs 65.7%) (5). Fluoroscopy is a useful adjunct during N-J tube placement (6). Endoscopy facilitates the use of grasper/snare to manipulate the tube or transnasal NET placement over guidewire. Fluoroscopic and endoscopic placement of feeding tubes is highly effective (success rate ∼90%, typical time ∼15 min) (6). Endoscopy is often successful after fluoroscopic failure.
When postpyloric placement is desired, electromagnetic sensing devices (Corpak MedSystems, Wheeling, IL, USA) are helpful (7). Prokinetic agents can be useful adjuncts. Erythromycin 500 mg given prior to N-J tube placement improved post-pyloric placement rates from 55% to 93% (7, 8). Erythromycin (200 mg ∼30 minutes pre-placement) along with 500 mL air insufflation resulted in 88% post-pyloric placements (9). Metoclopramide is often used to aid in postpyloric placement, despite uncertain effectiveness (10, 11).
MEDICATION ADMINISTRATION AND MECHANICAL OCCLUSION
Crushed tablet and opened capsule administration through NETs is commonly performed but may lead to altered absorption, interactions with enteral feeds (physical binding) and tube occlusion (12, 13). These practices may alter bioavailability, potentially causing morbidity. For some medications, such as enteral-coated tablets, crushing may result in reduced efficacy (12). Tubes usually occlude due to some combination of coagulation of feeding formula, tube kinking, medication fragments, and incompatible infusate precipitation (14). Other factors associated with occlusions include tube length, tube diameter, and usage-related factors (14, 15). NETs should be regularly flushed to prevent occlusion, including before and after each medication administration, bolus feeds, and at regular intervals with continuous feedings (12, 15). Most obstructions can be resolved using warm water with alternating gentle pressure and suction (13, 14). Acidic agents (i.e., carbonated soda, cranberry juice) are often used but may exacerbate the problem if occlusion is caused by feed coagulation or acid-base precipitation (12, 13). Pancreatic enzymes may be useful but are pH-dependent (16). Blunt guidewire/stylet use may be considered under special circumstances (17) but is discouraged due to perforation risks. Persistent NET blockage requires tube replacement.
SENGSTAKEN-BLAKEMORE TUBES AND POTENTIAL COMPLICATIONS
Sengstaken-Blakemore (S-B) tubes (Fig. 1A) provide balloon tamponade and temporary hemorrhage control of esophageal variceal hemorrhage while resuscitation, correction of coagulopathy, and preparation for definitive endoscopic, interventional, or surgical treatment take place. Hemorrhage is controlled by the inflated gastric balloon traction against the coronary-esophageal venous junction, with temporary control success in ∼85% cases (18). An esophageal balloon may supplement hemostasis. S-B tube use carries a 10–20% aspiration risk (19), which can be reduced by supplemental N-E drainage (i.e., multi-lumen “Minnesota tube”) (20). Endobronchial placement (21) and external tracheal compression and esophageal rupture have been described with S-B tube malpositioning (esophageal inflation of gastric balloon) (22, 23), mandating placement confirmation before balloon inflation. When gastric balloon becomes impacted due to outflow channel malfunction, endoscopic puncture helps facilitate tube removal. Dental, mucosal injuries (including necrosis) and parotitis can occur with S-B tubes (24).
NASOENTERIC TUBE BREAKAGE AND DISLODGMENT
Intra- and/or extra-corporeal NET breakage/cracking happen in 11–20% cases (17, 25,–27). Besides manufacturing defects, the most important contributory factor to structural damage of the distal tube is gastric acid exposure (17, 26). Breakage/cracking also occurs with excessive flushing for food/medication impactions (27). Fractured tube fragments can be removed endoscopically (26, 27). However if the fragment is small and not endoscopically retrievable, it can be allowed to pass through the gastrointestinal tract (26). Unintentional enteric tube breakage (and entrapment) can occur intraoperatively in the setting of gastric and esophageal surgery, where the tube is used to intraluminally pass stapling device parts and facilitate anastomosis creation. If tube fragment is caught in a staple/suture line, the involved anastomosis needs to be taken down, the fragment removed, and the anastomosis re-done. Unplanned dislodgment occurs in 25–50% of NETs, usually secondary to altered mental status, patient repositioning, physical therapy, patient transport, or tube entrapment on extraneous devices (28). Nasal bridles in patients with repeated dislodgements are is successful in 85% cases (29).
LARYNGOSPASM
Laryngeal NET malpositioning may cause an exaggerated superior laryngeal nerve response (30) and overstimulation of distal afferent esophageal fibers (31). During anesthesia, the overall incidence of laryngospasm is 0.87%, increasing to 4.85% with NET presence (32). While infrequent, intense laryngospasm (∼0.5% of cases) can result in laryngeal obstruction, aspiration, cardiac arrest, and even death (30, 32). Therapeutic maneuvers include NET removal and ventilatory support by continuous positive airway pressure with 100% oxygen via tight-fitting facemask. Additional airway maneuvers include digital pressure to the “laryngospasm notch” (between the mastoid process and the ear lobule) and pressing inward on the styloid process, which induces periosteal pain, causing autonomic system reflex and vocal cord relaxation. Another option is a vigorous forward pull of the mandible, which causes a painful stimulus and stretches the geniohyoid muscle to partially open the larynx (33). Endotracheal intubation may be necessary. Laryngospasm can be minimized postoperatively by intravenous lidocaine (2 mg/kg) given ∼1 minute pre-extubation. Lidocaine spray/nebulization may offer prevention in awake patients. Trans-tracheal lidocaine injection and cricothyrotomy may be required for refractory cases (30).
ASPIRATION
Aspiration, or unintended inhalation of saliva, food, or secretions should be distinguished from simple regurgitation of gastric contents that remain above the true vocal cords. Tracheal aspiration can be divided into two broad categories: (a) aspiration of “inert” fluids and (b) aspiration of acid (or alkaline) contents. NET presence is considered a risk factor for aspiration because the tube can mechanically interfere with the lower esophageal sphincter, altering the natural gastro-esophageal alignment, thus increasing gastro-esophageal reflux (34). Aspiration risk increases with decreasing levels of consciousness, impaired gastric emptying, and depressed cough/gag reflexes (34). Clinical presentation of aspiration may be “silent” or acutely symptomatic (i.e., coughing, choking, respiratory distress).
The incidence of aspiration in tube fed patients varies (4% to 95% depending on study) (35), and depends on: (a) how aspiration is defined (i.e., silent vs symptomatic); (b) method of diagnosis (radionuclide tagged feeds, pepsin analysis); (c) distal position of the feeding tube; and (d) the type of feeding tube (N-G vs. gastrostomy). The most important agent responsible for pneumonitis is gastric acid. Duodenal/jejual feeding may carry lower aspiration risk than gastric feeding, especially in neurologically impaired patients. Although aspiration may occur without significant gastric residuals, high residuals (i.e., > 500 mL) increase risk (36). Promotility agents may help reduce gastric residuals. Administration rate and total volume of feeds can both impact intragastric pressures and risk of reflux (34). Gastroparesis may also predispose to aspiration. In a prospective ICU study, at least 1 aspiration event was identified in 89% tube fed patients (37).
Pneumonia risk increases following aspiration, especially among those with impaired consciousness, altered airway defenses, and depressed immunity. Post-aspiration pneumonia carries mortality risk of 17–62% (37, 38). In mechanically ventilated patients, sustained supine position (with head of bed flat) increases gastroesophageal reflux and aspiration risk (39). This risk may be reduced by 30–45 degree semirecumbent positioning, continuous subglottic suctioning, and oral decontamination (40, 41). Selective digestive decontamination to reduce potentially virulent bacterial colonization has been suggested as beneficial (41), as well as chlorhexidine gluconate use (42).
INTRACRANIAL INSERTION
One of the most dreaded NET complications is intracranial misplacement (Fig. 2). This most commonly occurs in the setting of facial trauma or skull base surgery (43, 44). Trauma-related risks include basilar skull fractures involving the cribiform plate or comminuted skull base fractures involving anterior cranial fossa (45). Cerebrospinal fluid (CSF) rhinorrhea or presence of periorbital ecchymosis (“raccoon eyes”) should warn of significant risk. Unusually thin cribiform plate (i.e., secondary to sinusitis) also increases risk (45). Intracranial NET insertion can be devastating, causing symptoms corresponding to the area(s) injured by the tube, and reported mortality > 60% (43). After confirming misplacement with plain radiography, emergent CT imaging and antibiotic coverage should follow. Treatment includes removal of the tube and treatment of any associated complications. Craniotomy with direct visualization may be required (43). Prevention includes oropharyngeal placement or NET placement and advancement perpendicularly to the face. Fiberoptic bronchoscopy or laryngoscopy during placement may be helpful for patients with basal skull fractures without midface or nasal trauma. Larger NETs may be safer than small-caliber ones.
PNEUMOTHORAX
Thoracic complications of NETs occur in up to 8% cases, and include: (a) bronchial placement leading to atelectasis, pneumonia, lung abscess; (b) bronchial perforation and pleural cavity penetration; (c) pneumothorax; (d) isocalothorax (enteral feed hydrothorax); (e) empyema/sepsis; (f) pleural knotted tube; (g) pulmonary hemorrhage; and (h) bronchial suture line entrapment following lobectomy (46). Pneumothorax accounts for ∼60% of tracheopulmonary NET complications, requiring thoracostomy in ∼50% of instances (46).
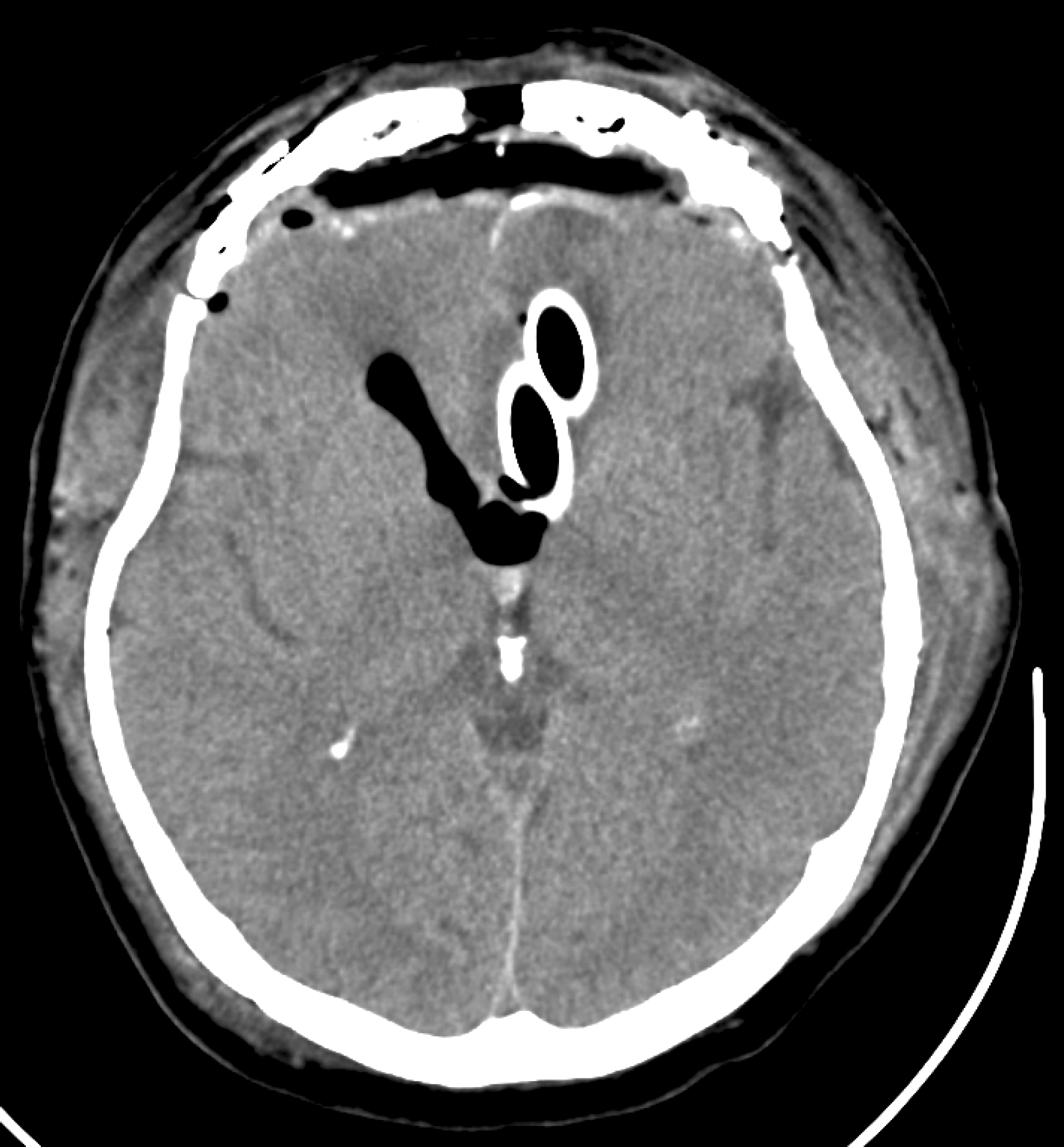
Intracranial mis-placement of soft, flexible, large-bore nasal tube following skull base surgery. Immediate operative removal was performed.
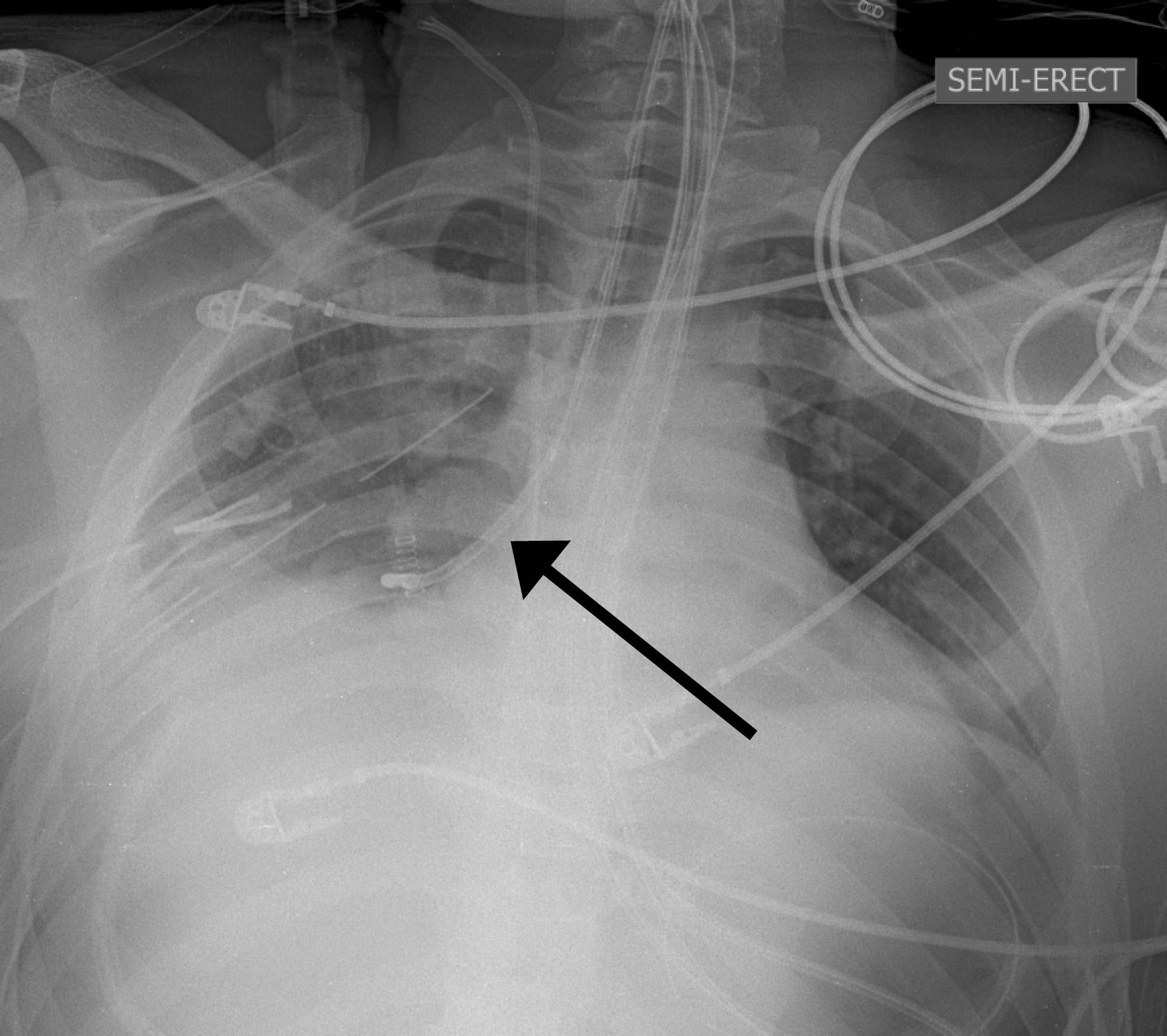
Endobronchial placement of wire-bearing naso-duodenal tube (arrow). This image was obtained as a part of routine safety protocol that empolys imaging evaluation during long enteral tube placement procedures after 30–40 cm initial NET excursion (to rule out airway mis-placement) followed by an abdominal x-ray to document the position of the distal NET terminus. In this case, the NET was withdrawn and successfully re-inserted, repeating the safety protocol outlined above.
NET incursion into the tracheobronchial tree is relatively common, is usually recognized promptly, and followed by tube repositioning (Fig. 3). In a study of 4,190 patients NET placement resulted in 87 intrabronchial malpositions and 9 pneumothoraces (47). Subsequent safety measures significantly decreased procedure-related pneumothorax (0.09% vs 0.38%), and pneumothorax risk after intrabronchial placement (3% vs 27%). Over two-thirds of patients with misplaced tube had an endotracheal tube (ETT) or tracheostomy. Pneumothorax risk was associated with misplacement occurring at night (16% vs 4.7%) and with > 1 misplacement (47).
Cuffed ETTs do not offer significant protection from tracheal NET placement. In fact, ETTs may actually increase the risk of pulmonary entry by preventing glottic closure and impairing swallowing. Stylet-stiffened fine bore tube can easily “squeeze” past low-pressure ETT cuffs (47). Intra-tracheal misplacement should be considered if NET placement induces coughing or is associated with inability to phonate normally. It is possible to auscultate air insufflation into tracheally placed NETs during abdominal auscultation, or to mistake aspirated fluid for gastric contents. Abdominal radiography should therefore routinely be used to confirm NET position.
ESOPHAGEAL PERFORATION
Esophageal perforation during NET placement usually occurs at the level of cricopharyngeus, mainly due to esophageal narrowing and lack of longitudinal esophageal muscle fibers at this level. Cervical esophageal or pharyngeal perforation is rare during NET insertion. Factors that may predispose to esophageal perforation include repeated placement attempts, preexisting anatomic esophageal abnormalities, altered mental status, cardiomegaly, tracheal intubation, and the presence of cervical osteophytes (48, 49).
Diagnosis of esophageal perforation requires high index of suspicion, and the location of clinical findings often suggest the location of perforation. For example, cervical crepitus is seen in ∼60% of cervical esophageal perforations, one-third of mid-esophageal perforations, and is uncommon in distal esophageal perforation. While most esophageal perforations are associated with pneumothorax, pneumomediastinum, and pleural effusion(s), as many as 12% have no associated acute radiographic findings (48). Posterior nasopharyngeal perforations may produce left pleural effusion and pneumomediastinum (49). Roentgenognaphic findings which suggest the diagnosis include location of the NET at the gastroesophageal junction (but not in the stomach), rapidly accumulating left pleural effusion, and pneumomediastinum. Effusion fluid may contain elevated levels of salivary amylase. Contrast esophagogram is the diagnostic procedure of choice, and diagnostic delay > 24 hours is associated with increased morbidity and mortality.
MECHANICAL EROSION, ULCERATION, AND BLEEDING
Clinically significant complications related to direct interaction between NETs and the mucosa are rare (50). This is likely due to the use of double lumen sump tubes causing fewer suction injuries. Nonetheless, mucosal erosion can occur and contribute to ulceration and bleeding. In a study of mucosal biopsies taken >24 hours after N-G tube placement, hyperemic mucosa, submucosal hemorrhage/clots, and erosions/ulcers near the esophago-gastric junction were common (51). Endoscopic and histologic changes were related to duration of mucosal contact (51). Acute upper gastrointestinal bleeding secondary to submucosal passage can occur (52). Perforations of the hypopharynx, esophagus, and stomach constitute more serious forms of injury. NET-specific risk factors for erosion/perforation include tube size, rigidity, and tube degradation in digestive juices (i.e., stiffening from acid hydrolysis) (50, 53, 54).
SUBMUCOSAL PASSAGE
Mucosal injury with submucosal tunnel or “bridge” formation is an uncommon complication of NET placement (52, 55 –58). Risk factors for this complication include septal deviation, presence of artificial airway, comatose patient, esophageal tumor, traumatic injury, and use of rigid stylets (55, 56). In the presence of these risk factors, any difficult placement should raise the suspicion of a mucosal injury, and blind placement should be aborted. Clinical indicators of submucosal passage include blood return from the tube, inability to advance, or lack of expected content returning upon initiation of suctioning (55, 58). When these signs are present, tube removal with another placement attempt can be considered if there is minimal or no bleeding and a low degree of suspicion (55). Significant bleeding combined with a moderate or high degree of suspicion for injury should prompt direct laryngoscopy, esophagoscopy, or gastroscopy (52, 55, 56, 58). Once injury is recognized/confirmed, further diagnostic and therapeutic steps may be warranted due to the risk of associated esophageal perforation (55 –58).
AORTO-ESOPHAGEAL FISTULA (AEF)
Vascular rings may produce airway- and swallowing-related symptoms (59). In the setting of double aortic arch, trachea/esophagus are spatially constricted, and any indwelling tubes (i.e., ETT, NET) can produce pressure necrosis and fistulization. Therefore, prolonged NET use (> 1 week) should be avoided in these patients (60). A clinical diagnosis must be made with confidence as imaging is not universally reliable. Early cardiothoracic consultation is essential (59). Sengstaken-Blakemore tubes may have a temporizing role by tamponading even massive hemorrhage from AEF (59).
STRICTURE FORMATION
Esophageal stricture is a rare complication of long term N-G intubation (50, 61, 62). There are only a few reported cases in the literature, and ≤ 1% of strictures are associated with NET use (62). Of cases related to NET use, most have been associated with the use of large diameter tubes, although stricture has been reported with tubes as small as 12F (58). Although the etiology is not completely known, suggested causes include injury secondary to intubation, gastric reflux, impaired esophageal clearance, and steroid use (58, 62).
Most patients with NGTs for nutritional support spend most of their time in a supine position, resulting in decreased esophageal acid clearance (i.e.,. impaired gravity-dependent emptying) (63). The esophagus is thus exposed to acid, resulting in esophagitis, which may lead to stricturing. Persistent postoperative emesis, hiatal hernia, duodenal ulcer, uremia, immunosuppression (i.e., chemotherapy or steroid use), previous gastrectomy or gastrojejunostomy can lead to esophageal mucosal injury, which can in turn lead to esophagitis with stricture formation (62). The mid- and lower segments of the esophagus are particularly vulnerable, and mucosal ulceration is usually seen on endoscopy (64).
INTRAPULMONARY FEEDING
Inadvertent airway intubation is relatively common during NET placement (65). Specialized “magnet tracking” of NET position in healthy subjects showed accuracy ∼100% both above/below the diaphragm (66). Although N-G tubes are used principally for gastric decompression, they are routinely used to provide EN through smaller diameter tubes. Such tubes may be associated with increased risk for intrapulmonary placement and/or displacement during retching or regurgitation (68). Intrabronchial/pulmonary feeding can result in pneumonia, asphyxiation, pneumothorax, and death.
EMPYEMA
Empyema in the context of NET may be associated with intrapleural feeding in the presence of oropharyngeal flora (69). It can also occur secondary to esophageal perforation and mediastinitis (70). Prevention involves placement verification before utilizing the tube for feeding. If enteral feeding or medications are infused into the pleural cavity via the misplaced tube, tube thoracostomy should be placed and the N-G tube removed (4). Intrapleural charcoal instillation will likely require continuous irrigation and can result in severe pulmonary parenchymal injury and potentially death (71).
NASOENTERIC TUBE SYNDROME AND VOCAL CORD DYSFUNCTION
The NET-syndrome is described by the triad of nasoenteric intubation, throat pain, and vocal cord paralysis (usually bilateral). Symptoms include throat pain (62%), stridor (43%), dysphagia (29%), hoarseness (24%), and less commonly dyspnea, otalgia and fever. NET-syndrome pathophysiology involves: (a) the mobile larynx, sliding up and down against semi-rigid NGT during coughing and swallowing; (b) normal tonic contractile state of the cricopharyngeus muscle pressing the tube against posterior cricoid cartilage; and (c) in supine patients, gravity pulling the larynx posteriorly, pinching the NET between the cricoid cartilage and cervical spine (72, 73). These forces generate local irritation, edema and tissue ulceration, causing impaired vocal cord function. NET use may also be associated with direct mechanical injury resulting in arytenoid edema.
Stridor and vocal cord paralysis can appear anytime between 12 hours post-placement to 2 weeks post-removal. Pain, an important early symptom, should not be ignored and should prompt thorough clinical assessment. NET-syndrome is less likely with small-bore tubes. In cases of suspected vocal cord paralysis comprehensive examination is required to rule out other causes, including nasolaryngoscopy to evaluate vocal cord function/ulceration (73). No consensus exists on management of NET-syndrome. NET removal, nutritional support, corticosteroids, and antibiotic treatment are all important. Although tracheotomy may be needed in severely affected patients, most patients regain vocal cord function within 2 weeks (reported recovery times 1 day to 2 months) (73).
COMPLICATIONS OF NASOBILIARY TUBES
Complications associated with endoscopic nasobiliary drainage tubes (ENBT, Fig. 1F) include: (a) Cholangitis (∼10%) and contralateral cholangitis (∼10%); (b) Tube occlusion (∼10%); (c) Need for additional drainage (i.e., conversion to endoscopic biliary stent or percutaneous transhepatic catheter (PTC) 22% to 26%) (74); (d) Tube dislocation (15%); (e) Post-ERCP pancreatitis (3%); (f) Liver/intra-abdominal abscess; (g) Sepsis; (h) Hepatic insufficiency; (i) Bleeding (5.9%) (75); and (j) Retroperitoneal perforation.
Tube occlusion usually presents with biliary obstruction, elevated liver function tests, and fevers. Segmental cholangitis is diagnosed when there is no improvement despite proven ENBT patency. Contralateral cholangitis is defined as infection affecting the undrained hepatic segments. Cholangitis secondary to tube occlusion occurs in ∼10% of cases. ENBTs carry higher risk of cholangitis than PTCs. Cholangitis due to tube occlusion can be treated by recanulation, tube exchange or placement of additional tube(s) into affected segment(s) based on diagnostic imaging.
Contralateral cholangitis is seen in up to 10% of cases. Additional PTC biliary drainage is commonly utilized to treat it but placement of a second ENBT is an option. If cholangitis recurs after two nasobiliary tubes, PTC becomes the best alternative. Inability to place the ENBT occurs in as many as 10% cases (76), and is often due to inability to canulate the bile duct or negotiate a guide wire past the obstructing lesion. Gallbladder perforation with hepatic subcapsular hematoma has also been reported after nasobiliary drainage (77).
RHINITIS AND SINUSITIS
One relatively common finding in critically ill hospitalized patients is rhinitis and sinusitis associated with upper aerodigestive tubes. Despite its common occurrence, NET-related sinusitis has been relatively neglected in the world literature. In one study, use of N-G tubes was significantly associated with nasal and maxillary sinus inflammation (78). Radiographic evidence of sinus inflammation was much more prevalent than clinical signs/symptoms of sinusitis (78). Reducing the duration of NET use and utilizing traditional symptomatic therapies (i.e., nasal sprays, head of bed elevation) are usually effective when clinical manifestations are limited to congestive symptoms. Antibiotic therapy may be required in the setting of active sinus infection with local and/or systemic signs and symptoms. Minimizing the indwelling time for NETs can also reduce the risk of any pressure-related injuries (i.e., erosion) of the nasal area.
NASOENTERIC TUBES: MISCELLANEOUS TOPICS
Refeeding syndrome (RFS) occurs in severely malnourished individuals or starvation victims (>10% weight loss over 2–3 months) undergoing (re)initiation of caloric support. It is commonly seen in hospitalized patients receiving enteral nutrition and practitioners should be familiar with its diagnostic and therapeutic characteristics (79).
Another largely historical topic that warrants brief mention is the use of long naso-jejunal tubes for feeding or management of bowel obstruction (80). These tubes are rarely utilized today, and certain types featuring mercury-weighed tip are no longer considered safe for clinical use. In addition, there are anecdotal reports of intussusception associated with these devices.
Using NETs for decompression of malignant bowel obstruction (MBO) produces unnecessary distress, increases the volume of air swallowed, and may lead to morbidity. After excluding surgery as therapeutic alternative for irreversible MBO, practitioners should focus on developing a comprehensive plan for end-of-life care and deciding what to do with the NET. It is important to note that most patients wth inoperable MBO can be managed medically (81) with combination therapy until death, without an NGT (81). Individualized treatment plans should involve collaboration between surgeons, oncologists, and palliative care physicians. (81) For terminally ill patients the issue of futile feeding commonly arises, often during discussions to clarify that goals of care are no longer curative but rather palliative. Physicians may be faced with a patient or family request to insert a NET for feeding a terminally ill patient, or to maintain an existing temporary NET for feeding. Provision of nutritional support is a life support measure with benefits and burdens that deserve re-evaluation when the clinical context changes. Clinicians and families may struggle more with withdrawing existing artificial feedings than withholding artificial feedings that were never started. Complications of enteral feedings in such settings include discomfort, aspiration, and prolonged suffering. At end of life, based upon a risk-benefit analysis, artificial hydration may be used selectively for symptom control and enteral artificial nutrition applied infrequently (82).
CONCLUSIONS
The use of NET's is ubiquitous, and clinicians often take their placement, function, and maintenance for granted. Morbidity associated with NETs is common, but frequently subtle, necessitating a high index of suspicion and clinical vigilance. An approach to emphasize quality and patient safety in the use of these universal devices is crucial, being particularly diligent to prevent harm to a patient's upper aerodigestive tract. Frequent monitoring and continual re-review of the indications for continued use of a NET is prudent, including consideration of changing goals of care.
Footnotes
ACKNOWLEDGMENTS
OPUS 12 Foundation review group members attest that all individuals credited with co-authorship participated in idea/concept creation, co-writing of the manuscript text, critically reviewing the content, and approval of the final version.
