Abstract
The angiosome concept delineates the human body into three-dimensional blocks of tissue fed by specific arterial and venous sources named “angiosomes.” Adjacent angiosomes are connected by a vast compensatory collateral web, or “choke vessels.” This concept may provide new information applicable to improving targeted revascularization of ischemic tissue lesions.
A few dedicated studies available seem to favor this strategy, as encouraging ulcer healing and limb preservation are reported in connection with both bypass and endovascular techniques based on these principles.
The theory on the angiosome model of revascularization (AMV) may help the clinician to better refine vessel selection, vascular access, and specific strategies in the revascularization of critically ischemic legs with tissue lesions. Specific applications of angiosome-guided revascularization were recently suggested for patients with diabetes or renal insufficiency, with ischemic tissue lesions of the lower limb, and extended large- and medium-size collateral network decay. For these cases, the concept may allow deliberate arterial reconstruction following individual wound topographies in specific ischemic areas, although deprived from “rescue-vessel” supply. The AMV theory may contribute to a shift in common reperfusion options.
However, the data available is suggestive and does not provide strong evidence as factors such as case mix and the severity of ischemia are unsatisfactorily controlled.
At present, the evidence is scarce as to the effect of the severity of the arterial disease. In all comparisons, the groups treated are likely to be dissimilar and mismatched. The angiosome concept is postulated to be valid especially in diabetics, whose ischemic lesions tend to heal worse than those of non-diabetics.
Keywords
INTRODUCTION
It has been estimated that at least 15% of diabetics will develop a foot ulcer during their lifetime, 14%–43% of whom will require amputation (1, 2). The rate of major amputations among diabetic patients has decreased with the development of revascularization techniques over the last two decades (2, 3), yet the risk of amputation remains more than seven times higher in the diabetic population than among non-diabetics (3).
In diabetics, atherosclerotic lesions are mostly located in the crural arteries (7). Neuroischemic foot ulcers seem to carry a particular risk of major tissue loss enhanced by microvascular dysfunction (8) and foot arterial collateral depletion (8), requiring appropriate vascular and multidisciplinary assessment (1, 8, 10, 11).
Indeed, contemporary revascularization series report a 10%–18% rate of unhealed ischemic wounds and frustrating major amputations despite good bypass patency (11) or successful endovascular treatment (5, 6, 13). Furthermore, wound healing after successful revascularization is extremely slow – median ulcer healing times of up to 6 months have been reported (14). Therefore, there is a need to identify methods to optimize the arterial supply to the ulcer area. Angiosome research has mainly aimed at identifying complicated flaps that would not develop necrosis due to a lack of arterial supply.
THE CONCEPT
The angiosome concept was first described within the field of reconstructive plastic surgery by Taylor and Palmer in 1987 (15). In their anatomical studies, the authors pioneered in preferential strategies for surgical access, tissue reconstruction, and amputation in specific 3D sectors of the body delineated by specific arterial and venous supply, named “angiosomes” (15, 16).
Following this concept, each “angiosome” encompasses a topographically specific “arteriosome” and correspondent “venosome” supply, blended in unitary block systems of perfusion (15 –17). Adjacent angiosomes are linked by numerous communicants, i.e. “choke vessels”(15 –17). These interconnections between neighboring angiosomes have been described to create an effective compensatory system against ischemic conditions (15 –17) in non-atherosclerotic and non-diabetic limbs (9, 10, 17). Huge collateral depletions, such as those typically accompanying diabetic arterial disease below the knee (9, 10, 13, 17) and end-stage renal disease (ESRD) (10, 13) may jeopardize this natural “rescue system” between adjacent angiosomes (9, 10, 17).
ANATOMICAL CONSIDERATIONS
Schematically, the distribution of the main foot and lower ankle angiosomes (15 –17) is as follows (Fig. 1): a) the medial calcaneal artery (MCA), the medial plantar artery (MPA), and the lateral plantar (LP) artery angiosomes derived from the posterior tibial artery and supplying the entire plantar heel, the medial and lateral plantar surface of the toes; b) the dorsalis pedis artery (DPA) angiosome which prolongs the anterior tibial artery, nourishing the dorsum of the foot and toes as well as the upper anterior perimalleolar vascularization; and c) the lateral calcaneal artery (LCA) angiosome from the peroneal artery supplying the lateral and plantar aspects of the heel. In this enumeration, one may optionally add the anterior perforating branch and the postero-lateral malleolar arteries' angiosomes, derived from the peroneal artery. The peroneal flow generally covers the lateral and plantar heel, and via its anterior peroneal perforating branch, it connects to the anterior tibial territory (Fig. 1).
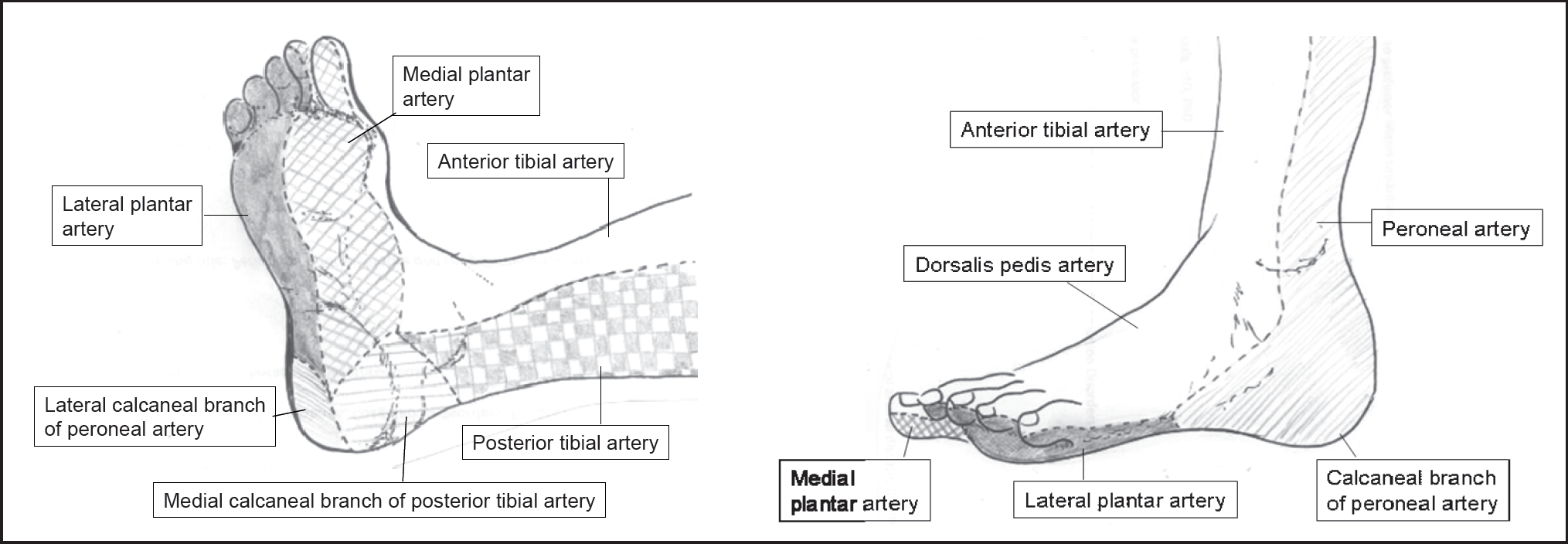
Distribution of the main foot and lower ankle angiosomes
Going up to the superior ankle and distal calf zones, other angiosomes have also been suggested/introduced, such as the lateral/medial anterior malleolar artery angiosomes (from the anterior tibial artery) and the postero-medial malleolar artery angiosomes (from the posterior tibial artery) (15 –17).
A high number of arterial anastomoses have been equally described to supply neighboring foot and ankle angiosomes (16–17).
Although it falls beyond the purpose of this article to thoroughly detail these extended connections, they can be summarized as follows: 1) the connections between the posterior tibial and peroneal artery (via the medial and lateral calcaneal branches), playing an important role in the etiology of ischemic heel ulcers; 2) the communications between the anterior (dorsalis pedis) and the posterior tibial artery (plantar arteries) either directly, at the level of the first metatarsal interspace, or via the metatarsal irrigation (through paired anterior and posterior arch origin), playing a substantial role in tarsal/metatarsal reperfusion; 3) the lateral perimalleolar anastomoses linking the peroneal (via the anterior perforating branch) and the anterior tibial (via the anterolateral malleolar branch), together with the medial perimalleolar network (via corresponding medial malleolar branches), representing distinct perimalleolar derivations for local blood supply; and 4) the communicants of both plantar arteries (arising from the posterior tibial trunk) that link the lateral and medial tarsal arteries (via the anterior tibial artery) and constitute notable compensatory collaterals to the sole.
In vascular surgery, the potential application of the presented angiosome model is to target the revascularization to the artery supplying the area of the tissue lesion to improve healing (18, 19).
CLINICAL EXPERIENCE
CURRENT APPLICATIONS OF THE ANGIOSOME CONCEPT
There are already existing applications of the AMV strategy in other fields of medicine, such as specific myocardial revascularizations, selective visceral embolizations, and the planning of different characteristic types of covering flaps, incisions, or amputations (15, 16, 17, 18). Only over the last decade, however, a small number of reports have analyzed its feasibility in limb salvage interventions for CLI in conjunction to either bypass (17, 19, 22) or endovascular techniques (10, 20 –22), with promising preliminary results (19 –24). Ulcer healing is the most important endpoint of these studies.
Ulcer healing
In their initial series on 52 distal bypasses, Attinger and co-workers (17) reported a 9% healing failure rate when ulcers were treated by their angiosome in contrast to a 38% lack of success in wounds that were revascularized indirectly (17). Accordingly, the same group documented (19) a 9% amputation rate in the angiosome versus a 38% amputation rate in the non-angiosome-oriented subgroup. In a larger series of 203 consecutive limbs with ischemic ulcerations undergoing endovascular reconstructions, Iida et al.(21) observed an 86% limb preservation rate in the angiosome-related group versus 69% in the nonspecific contingent (21). In line with these reports, Varela et al.(22), with a similar series of 76 ischemic ulcers treated by either bypass or endovascular therapy, noted significantly better results in terms of wound healing (92% versus 73%) and limb salvage (93% versus 72%) in the AMV-guided cohort of patients. In a recent and larger report, Iida et al. included 369 limbs with Rutherford class 5–6 ischemia and isolated below-the-knee arterial lesions; 200 direct and 169 indirect successful endovascular revascularizations were compared using a propensity score analysis. After this adjustment, the estimated rates of amputation-free survival (49% versus 29%) as well as freedom from major adverse limb events (51% versus 28%) and from major amputation (82% versus 68%) were significantly higher in the direct than the indirect group up to 4 years after the index procedure (23). These data suggest that the AMV strategy may be beneficial in treating CLI with tissue lesions, irrespective of the revascularization strategy chosen (11, 18 –22). However, in a retrospective Japanese analysis on 228 patients undergoing distal bypass for CLI with tissue loss, the outcome was not as clear. In this highly diabetic sample (81%), the healing rate was significantly faster if direct revascularization was possible, but after adjusting the risk factors with propensity score analysis, there was no difference in wound healing between the IR and DR groups (49%) (25). Similarly, Blanes et al.(22) did not observe any statistical differences among 32 retrospectively reviewed Rutherford category 5 and 6 patients when comparing the angiosome-targeted (direct) versus non-targeted (indirect) percutaneous revascularizations (24).
Specific features of diabetic neuroischemic foot ulcers and the potential advantages of the angiosome model
The neuroischemic diabetic foot may be connected to particular barriers in distal flow redistribution when compared to parallel “purely atherosclerotic” CLI subjects (17, 21, 25). Patients with long-lasting diabetes and distal neuroischemic infected wounds appear to express severe depletion in the foot's collateral reserve (9). Novel studies on selected CLI groups of populations suggest that the more proximal and unspecific the revascularization, the greater the dependency on the remnant collateral reserve may be expected (9, 17, 18, 22). Although this collateral network provides a remarkable “rescue system” in non-atherosclerotic patients, it may be dramatically hindered in CLI patients suffering from miscellaneous systemic arterial disease (such as uremic or diabetic atherosclerosis, obliterating arteritis, etc.) (9, 18). It has also been suggested that the neuroischemic “diabetic foot syndrome” associates a more distal and aggressive form of “atherosclerotic macro-angiopathy” to the “functional microcirculatory impairment” induced by both neuropathy and local sepsis (9, 17, 28).
Against this specific background, O'Neal et al. have emphasized the concept of “diabetic end-artery occlusive disease” (EAOD) (9). This theory focuses on the residual irrigation to the foot, since medium-sized “patchy” atherosclerosis associates with the acute septic thrombosis of “small collaterals” surrounding the wound inflammation (19). The EAOD model (9) helps the clinician to better understand why irrigation from “a few millimeters of skin” to the “entire diabetic foot or leg”(9) relies on precise nourishing vessels. These “vital” blood sources seem specifically hinged to their dominant angiosome-dependent arteries (17 –20). It may then be justified to note that for those particular subjects with long-lasting diabetes who exhibit a compromised collateral network (“choke-vessels”) (9, 18), the probability of enhancing adequate tissue healing may seem to increase with a more distally and topographically oriented revascularization approach (9, 17 –22). Novel indications in the revascularization of diabetic neuroischemic foot wounds, connecting the EAOD theory (9) to the wider angiosome concept (17 –22), may effectively produce promising results in diabetic wound regeneration and limb salvage for endovascular (10, 20 –22)and bypass surgery (17, 19, 22) alike.
PRINCESS PAOLA HOSPITAL EXPERIENCE
A preliminary report that shares this strategy demonstrated equally encouraging results for limb preservation (91%, 88%, and 84% at 12, 24, and 36 months, respectively) and wound healing rates (85%, 81%, and 73%, respectively, at the same time intervals) in comparison to available equivalent non-angiosome-oriented results (10). A more recent retrospective analysis on similar cohorts of CLI patients, diabetics, and Rutherford category 5 (1) patients was conducted with and without applying the AMV concept (18). Although there was no statistical significance for primary (p = 0.813) and secondary patency (p = 0.511), there was a significant difference in wound healing (HR 2.19, p = 0.025) and limb preservation (HR 2.32, p = 0.035), with better results in the angiosome-guided subgroup of revascularizations (18). Despite a marginally higher number of initial technical failures among the primary angiosome-oriented angioplasties (21% versus 18%), this study revealed no significant difference (p < 0.05) in reinterventions, perioperative morbidity, or mortality between angiosome-oriented versus angiosome-unspecific angioplasties (18).
HELSINKI UNIVERSITY HOSPITAL EXPERIENCE
The special interest of research in our hospital has been in ulcer healing, especially in diabetics (14). We have found the location of the wound to be one of the most important factors that have an impact on wound healing. We have now (also) paid attention to the impact of angiosome-directed revascularization on wound healing. We found that in half of the cases over the last 3 years, the target of the revascularization was the ulcer angiosome, the other half comprising indirect revascularizations. If the revascularization of the ulcer angiosome was successful, 74% of the wounds healed in one year as compared to the 46% rate of the indirect approach (p = 0.002). In 18 months, the respective healing rates were 87% and 75% (p = 0.002), suggesting that wound healing was significantly slower in the indirect approach group.
An example of current angiosome-guided endovascular interventions for diabetic neuro-ischemic foot wounds is depicted in Fig. 2.
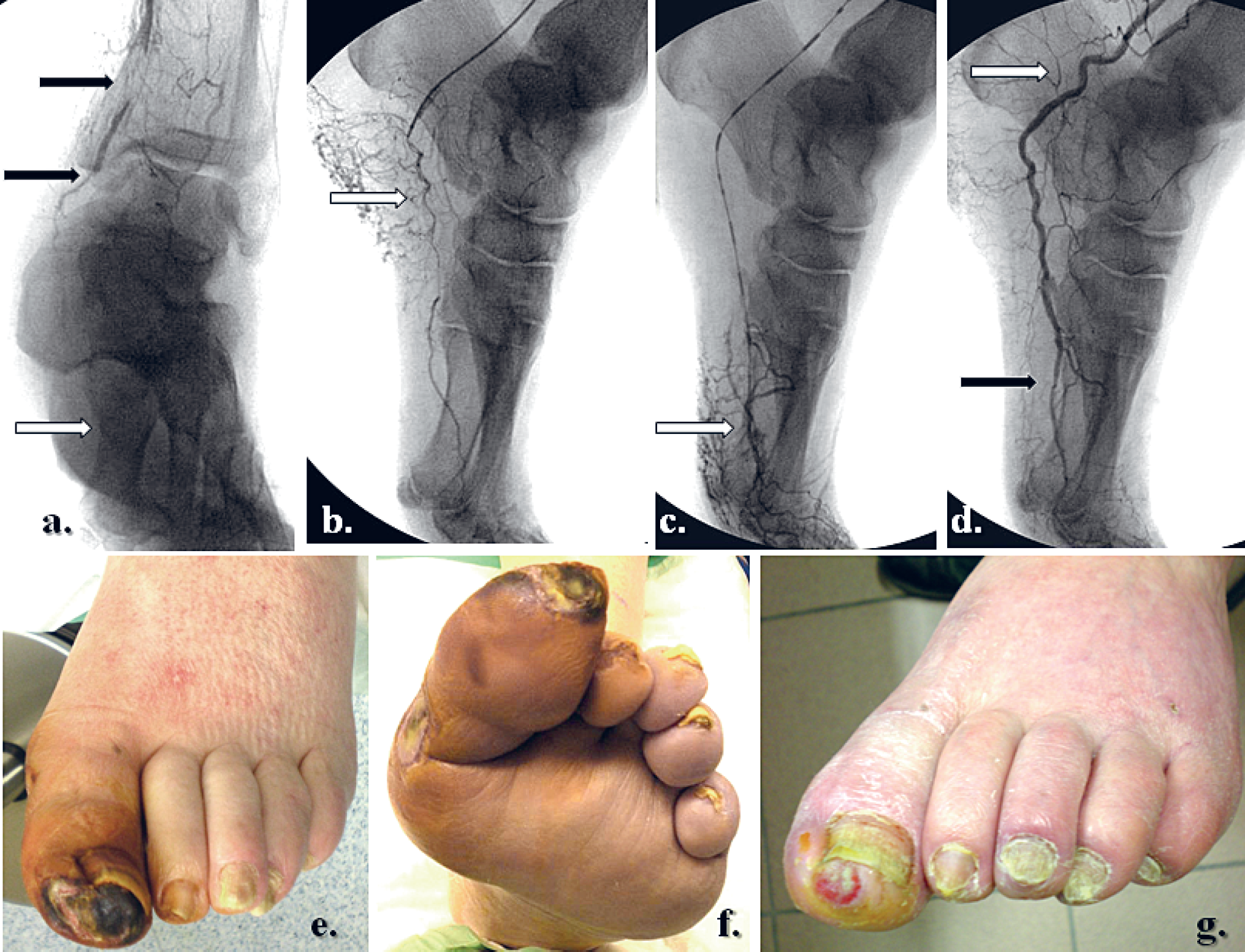
Selective revascularization of the posterior tibial and medial plantar artery and consequent flow reperfusion in the medial plantar angiosome. a. The initial staged occlusions of the distal posterior tibial artery and left plantar artery. Both peroneal and anterior tibial arteries were occluded at different levels in the calf. b. – c. Selective reopening of the posterior tibial and medial plantar artery. d. Angiographic control after staged angioplasties with correct medial plantar reperfusion (arrows). e. – f. Related clinical “diabetic foot syndrome”, revealing severe toe ischemia and tissue defects on the plantar aspect of the left hallux (topographic correspondence of the medial plantar angiosome). g. Clinical result at 14 weeks, after selective posterior tibial and plantar artery revascularization.
DISCUSSION
In current surgical bypass or endovascular practice, it is generally accepted that the outflow vessel and appended run-off (based on angiographic or duplex findings) represent the major decision-making factor in arterial reconstruction. The distal ischemic wound territory is eligible for revascularization either directly (angiosome-dependent) or, quite often, indirectly (without angiosome orientation) via the surrounding collaterals of the available target vessel (18, 22). Indications for the type of revascularization to be performed are currently deduced from an iconographical point of view depending on the most “suitable” artery to be reopened, or “patent” distal vessel to anchor the bypass (17, 18, 22).
Not surprisingly, following the AMV orientation, the main infragenicular arterial axis addressing the distal wound territory commonly appears to be affected by vast atherosclerotic disease (10, 17 –20). A further shift in the commonly accepted strategy for revascularization from “which vessel is the most accessible for reconstruction” to a multidisciplinary clinical perspective could be anticipated based on “which region of perfusion governed by which artery should be treated?”(20).
While original bypass still holds an important role in diabetic CLI revascularization due to higher local pressure and physiological pulsatile flow (1, 16), prime endovascular approaches seem to offer the alternative of reopening two or several tibial and foot arteries simultaneously with a single intervention (9, 17, 19, 20). These strategies therefore seem to have complementary rather than competitive roles in topographical, angiosome-guided approaches to revascularization (17, 21).
An accurate assessment of the remnant collateral framework in every CLI presentation carries a pivotal role in any type of revascularization (8, 12, 13, 18, 2). This statement, however, concerns both the main angiosome of the targeted tissue defect zone and its appended “choke vessels” (17, 18).
It has been shown that, beyond rare (4%–6%) individual anatomical variations, the main angiosomes of the foot and ankle are constant among the general population (15 –17). However, their boundaries may be subject to changes, in direct correlation with the local collateral network (18, 21). Seemingly, the main arterial axes, the “choke vessels” or small collateral ramifications, are equally subject to local variations regarding their amount and size (9, 17, 18).
This global collateral reserve with notable influences on either the principal angiosome or its appended “choke vessels,” seems to be strongly influenced by three main factors: 1) ageing, 2) the genuine pathology that generates CLI, and 3) the location of each angiosome itself (18, 20, 22).
While the first two conditions, including the EAOD phenomenon in the “diabetic foot” or “renal” presentations (9), were emphasized early in the text, the third factor should also be weighted carefully in the preoperative assessment. It has been demonstrated that angiosomes of the sole and forefoot possess a larger collateral network arising from both foot arches (15 –18). This original anatomical feature strongly differentiates them from the heel and hindfoot angiosomes, natively supplied by a scarce and low-volume mesh of collaterals (15 –22).
In the light of these observations, one can picture two extreme “poles” of clinical CLI presentations: at one end, there is the atherosclerotic, non-diabetic, middle-aged patient with forefoot trophic lesions, and at the other, the aged, long-lasting diabetic or renal patient, exhibiting neuroischemic heel tissue defects. The vascular surgeon should be aware of the crucial importance of collaterals in different patterns of CLI in order to influence the fate of any revascularization, with or without appended angiosome orientation.
Therefore, appropriate assessment of collaterals seems to be of outstanding significance in planning an AMV for surgical or endovascular reperfusion (18, 22). Perioperative collateral reserve evaluation by duplex, angio-CT or angio-MRI, TCpO2, the transcutaneous sensi-laser system, or intra-operative angiographic scoring (18, 20, 21), could be extremely useful in assessing peculiar foot arch perfusion, eventual anatomical variations, and compulsory alternatives for indirect revascularization (10, 20).
WHAT DO WE NEED TO UNDERSTAND ABOUT THE ANGIOSOME PRINCIPLE BEFORE ADOPTING IT IN CURRENT CLI REVASCULARIZATION?
Deliberate angiosome-supported revascularization needs, like any other novel therapeutic concept, a solid ground of evidence to stand on before it is applied. At present, its usefulness in arterial reconstruction for CLI remains without practical validation. This issue may be explained by several concurrent factors:
It represents quite a novel application in lower limb ulcer healing therapy, with only preliminary support in the literature, all from retrospective approaches. Any possible data comparison with pre-existing wound healing or limb salvage series are invariably biased by heterogeneity in patient selection, associated comorbidities, types of CLI, as well as subjacent arterial pathologies, types of ulcers, differences in clinical endpoints, and follow-up features, etc. Its position between the defenders of endovascular versus bypass surgery in the CLI revascularization algorithm remains controversial. The concrete role of arterial collaterals, in addition to their precise quantification before and after surgical versus endovascular limb revascularization – and, moreover, the succeeding direct (angiosome-oriented) or indirect (angiosome-free) approach – remains to be determined. There is a lack of uniform contemporary stratification in patient selection, type of intervention, clinical presentation, risk factors, and underlying CLI pathologies to be able to analyze outcomes among patients with or without the AMV model. A realistic inventory of the current therapeutic arsenal, with rational outcome expectations, should be conceived. Acknowledging that in AMV strategies more laborious procedures are commonly anticipated (18), “the most suitable angiosome-dependent artery” to treat is not necessarily the simplest vessel to recanalize (10, 18
–21). This added complexity in the operation (open or endovascular) may affect the technical success rate. Further prospective and randomized studies in larger cohorts of patients are needed to reach agreement on the potential advantages or drawbacks of this strategy in CLI revascularization. It is probably of key importance to allow further statistical analysis on equivalent cohorts of patients, with homogeneous demographic and pathological risk factors in similar multi-center studies, to address the entire multifaceted clinical background of critical limb ischemia.
Future angiosome-oriented practice does imply inherent technological progress. Beyond refined training for below-the-knee or below-the-ankle surgical and trans-catheter interventions, new technologies and devices for recanalization, adapted for long and calcified chronic arterial occlusions, are warranted (18, 20).
CONCLUSION
Angiosome-based strategies in CLI infragenicular revascularizations seem to provide encouraging wound healing and limb preservation rates for both bypass and endovascular techniques.
The affiliation of the angiosome concept with contemporary infra-popliteal strategies in revascularization may be valuable; however, wider-scale clinical experience in randomized and prospective cohort studies are mandatory before making any relevant assertions in favor or against this concept.
