Abstract
Background and Aims
Necrotizing fasciitis (NF) and gas forming myonecrosis (GFM), both being subtypes of necrotizing soft tissue infection (NSTI), are life threatening conditions sharing certain similarities. Despite the necessity of early and radical surgical debridement in necrotizing infections, the distinction between these entities is of clinical relevance since gas forming myonecrosis in a number of cases results from an underlying abdominal cause and the focus of infection can be missed. This study was to evaluate the incidence and risk factors as well as the mortality rate in patients with NSTI and GFM.
Material and Methods
All patients with NSTI treated in the authors' hospital between January 2005 and December 2009 were enrolled in the study. Medical records, histological slides, microbiological and laboratory parameters as well as Computerized Tomography (CT) and magnetic resonance imaging (MRI) scans were reviewed for all patients. Differences between NF and GFM regarding hospital stay, number of surgical interventions and pre-existing comorbidities as well as mortality rate were analyzed. The laboratory risk factor for necrotizing fasciitis (LRINEC) score was calculated in all patients on admission.
Results and Conclusions
Thirty patients (17 female, 13 male) with necrotizing fasciitis with a mean age of 55 years (SD 15.5) were included in the study. There was no statistically significant difference between survivors and deceased patients comparing the LRINEC score (n.s.). Patients with necrotizing fasciitis secondarily involving the trunk had a significantly higher mortality rate (OR 11.2; 95% CI = 1.7–72.3). In the majority of cases (12 cases), minor skin lesions were identified as the site of origin. Amongst all necrotizing soft tissue infections six patients (female n = 3; male n = 3) with a mean age of 61.5 years (SD 12.2) with non-clostridial gas forming myonecrosis were identified. Three patients had a history of malignancy and in three patients the infection was secondary to major surgery. The mean LRINEC score was 8.5 (SD 1). Three patients (50%) died due to GFM.
Early diagnosis and appropriate intervention is critical to provide accurate treatment decisions. Eradicating the differing primary sources of infection in GFM and NF will have a positive impact on outcome.
Keywords
INTRODUCTION
Necrotizing fasciitis (NF) and gas forming myonecrosis (GFM) both represent subtypes of severe necrotizing soft tissue infections which share certain similarities in their clinical presentation. However, both life-threatening infections have to be differentiated with respect to the pathophysiology, clinical course and therapeutic consequences. NF initially presents with flue like symptoms, pain “out of proportion”, local swelling and erythema which normally exceeds the one found in gas gangrene. Local symptoms are rapidly progressing, pathognomonic signs are dusky and purplish patches with ill defined borders. Generalised features of septic shock like somnolence and hypotension may be present early after onset of the infection.
In 1979, Fisher described seven diagnostic parameters (1): Extensive fascial necrosis involving the overlaying skin, systemic septic involvement with reduced mental status, lack of a primary muscle infection, missing clostridium infection, lack of vascular occlusion, and finally a leukocyte infiltration with focal fascial necrosis and microthromboses as histological findings.
Purple, dark skin coloration and skin necrosis are signs of late stage of the disease. The infection primarily does not affect the musculature, muscular involvement is a sign of advanced stage of the infection associated with poorer prognosis. As in clostridial gas gangrene signs of systemic infection with reduction in mental status and deterioration of the general status of the patient are found early in the course of the disease. In contrast to clostridial infections minimal trauma or minor skin lesions (2) most of the time represent the origin of infection.
Early diagnosis, emergency surgical debridement, and wide-spectrum antibiotic therapy are the most effective treatment options to minimize the bacterial load and mortality rate associated with these conditions. Simple laboratory markers are helpful to detect life-threatening infection. The laboratory risk indicator for necrotizing fasciitis (LRINEC) score (3 –5), composed of the following six laboratory findings: glucose level, c reactive protein level, white blood cell count, sodium level, creatinin level and haemoglobin, a combination of rapidly available laboratory parameters has been designed to distinguish severe NSTI from other soft tissue infections such as cellulitis. A LRINEC score above six has been shown to be predictive for an increased mortality and higher rate of amputations.
In general, mortality rates vary from 30 to 70%, which may even increase significantly if the infection progresses to myonecrosis (6 –9). The aim of the present study is to demonstrate the diagnosis, the management and outcome of patients with NF and GFM.
MATERIAL AND METHODS
For the retrospective analysis of all patients with NSTI the database of the authors' hospital was searched for all patients admitted to our institution suffering from severe soft-tissue infections between January 2005 and December 2009. During this period 30 patients (17 female/13 male) with NF and six patients with non clostridial gas-forming myonecrosis (3 female/3 male) were treated at the BG-University Hospital Bergmannsheil (Table 1). Medical records, histological slides, microbiological specimens, Computed Tomography (CT) and Magnetic resonance imaging (MRI) scans were reviewed for all patients. In all patients, the event leading to the infection, the site of infection, comorbidities, the total number (including debridement and reconstructive surgery) and kind of surgical interventions as well as the duration of hospitalisation were recorded. The causative microorganisms isolated from tissue culture were determined and the mortality rate was estimated.
MeSh codes for patients with necrotizing soft tissue infection and non clostridial gas-forming myonecrosis.
The clinical diagnosis of necrotizing fasciitis was made by attending surgical physicians, whereas clinical suspicion was validated via histological examinations. Histologically, necrotizing fasciitis was diagnosed when necrosis of the superficial fascia, a polymorphonuclear infiltrate, and edema of the reticular dermis, subcutaneous fat and superficial fascia were present. A severe necrotizing infection affecting the superficial fascial layer and the surrounding soft tissue was confirmed histologically (10) in all but two cases, in which no histological specimens were obtained.
The clinical diagnosis of gas forming myonecrosis was also made clinically by the attending surgical physicians. Clinical suspicion was confirmed via histological examinations. The histological image of a severe myositis or myonecrosis in conjunction with the clinical evidence of gas entrapment with in the musculature confirmed the diagnosis of GFM. Gas forming myonecrosis was also evident on X-ray or CT scan. In all operated patients necrotizing myonecrosis with gas entrapments was confirmed on histological assessment.
Additionally, the laboratory risk indicator for necrotizing fasciitis (LRINEC-) score in the initial diagnosis of NF and GFM were calculated in all patients on admission. The LRINEC score is a calculation of a numerical score based on the rapidly available laboratory parameters glucose level, C-reactive protein level, leucocyte count, NA, creatinin level, hemoglobin. The maximum score is 13, while a score ≥ 6 is suspicious for NF and a score ≥ 8 strongly predictive for NF (3 –5) (Table 2).
LRINEC (laboratory risk indicator for necrotizing fasciitis) score.

Aggressive physiological support to correct fluid deficits, including electrolyte imbalances, extensive operative debridement and antiseptic wound dressing changes using sterile gauzes and wide-spectrum antibiotic therapy were the therapeutic strategy in all patients. Radical surgical debridement was required in almost every case prior to soft tissue closure.
STATISTICAL ANALYSIS
All variables were expressed in terms of mean and ± standard deviation (SD) of the mean. The t test was performed when the data were distributed normally; otherwise the Mann-Whitney test was used. The Shapiro-Wilk normality test was performed to test the null hypothesis that the data had a normal distribution. For all tests, P < 0.05 was considered statistically significant. The risk for mortality rate was compared between the infection involving and not involving the trunk using the odds ratio. Statistical analysis was carried out by means of a statistical software package (GraphPad Prism Version 4.1, GraphPad Software Inc., La Jolla, CA).
RESULTS
NECROTIZING FASCIITIS (NF)
Thirty patients (17 female, 13 male) with necrotizing fasciitis with a mean age of 55 years (SD 14.6) were included in the study. In fifteen cases (50%), the infection was localized at the lower extremity followed by the upper extremity with 13 cases (43.3%) and trunk with two cases (6.7%). In the majority of cases (12 cases), minor skin lesions (Fig. 1) were identified as the site of origin. In the remaining cases, NF resulted from postoperative wound infections (3 cases), pre-existing abscesses (3 cases), bursitis (one case) and cat bite (one case). No cause was identified in ten patients.
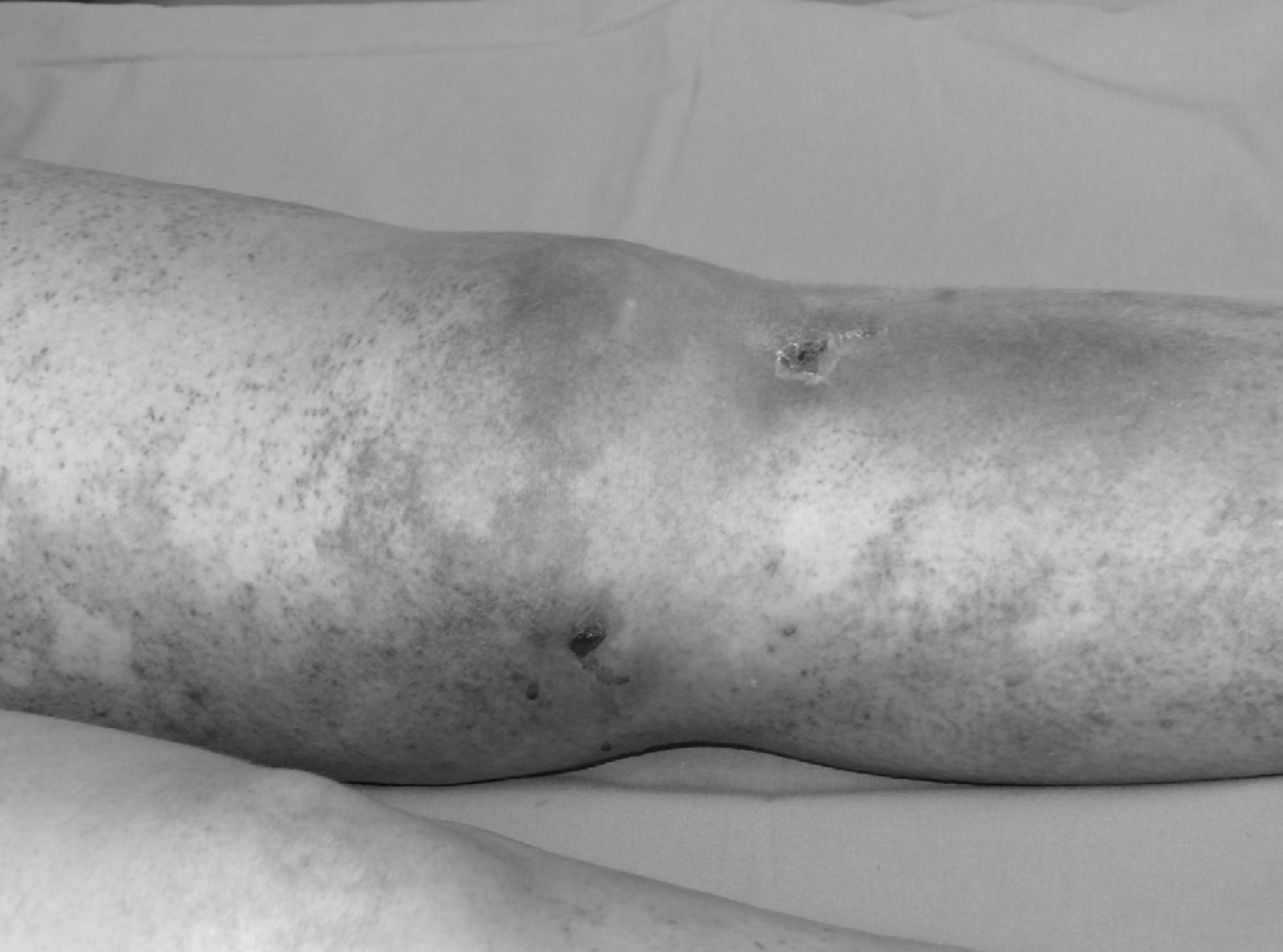
A r clinical presentation of a necrotizing soft tissue infection. The cause of NSTI was a minor skin lesion.
A total of 17 patients (57%) had at least two comorbidities. Diabetes mellitus was the most frequently observed co-morbidity present in 12 patients (40%) of all patients. A polymicrobial infection was found in eight patients (27%). No microorganism could be identified in 12 cases (40%). The most frequent isolated bacterium was Staphylococcus aureus with seven cases (23%) followed by Streptococcus pyogenes with five cases (17%).
The mean length of hospital stay was 28 days (SD 19.4) in all patients, while survivors stayed significantly longer in the hospital compared to deceased patients (p = 0.002). A mean of 4 (SD 2.7) surgical interventions were necessary during the treatment period (Table 3). Survivors underwent significantly more surgical interventions compared to deceased patients (p = 0.03). Elevated laboratory parameters of acute infection with leukocyte counts (15.4 mm3 SD 6.8) and C-reactive protein levels (28,2 mg/dl SD 16.9) were present in all patients. The mean LRINEC score was 7.3 (SD 2.2). There was no statistically significant difference between survivors and deceased patients comparing the LRINEC score (n.s.) (Fig. 2). The lowest LRINEC score in deceased patients was six and in survivors two. Twenty-five out of 30 patients (83.3%) had a LRINEC score ≥ 6. Only in five patients (16.7%), the LRINEC score was < 6. Five patients required major amputation of the affected extremity. Patients with necrotizing soft tissue infection secondarily involving the trunk had a significantly higher mortality rate (OR 11.2; 95% CI = 1.7–72.3).
Average age, gender ratio, LRINEC score, mean length of hospital stay, mean number of surgical interventions and mortality rate of all patients presenting between 2005 and 2009.
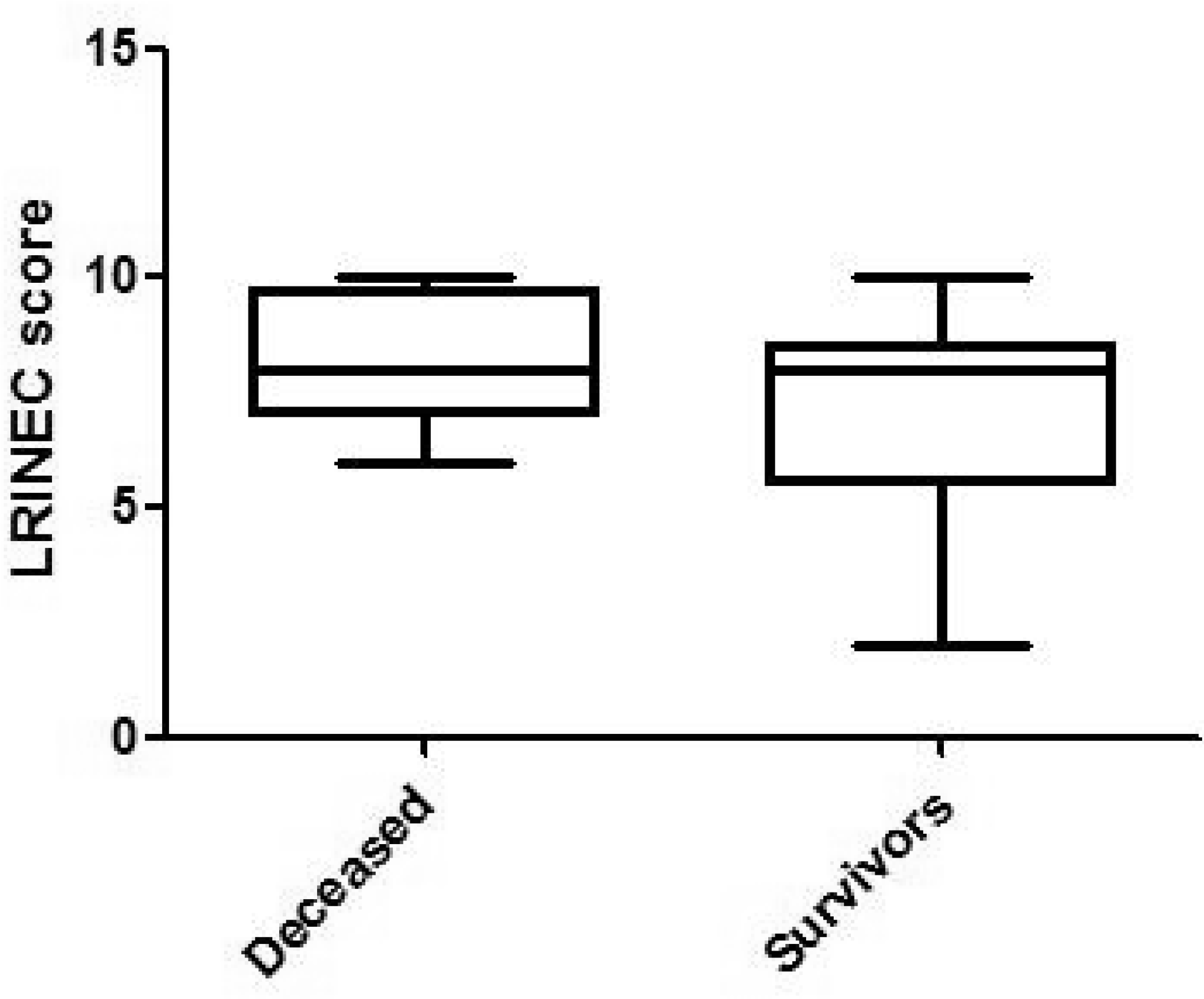
The LRINEC score for survivors and deceased patients with NSTI. There was no statistically significant difference between these groups.
GAS FORMING MYONECROSIS (GFM)
Six patients (female n = 3; male n = 3) with a mean age of 61.5 years (SD 12.2) with non-clostridial gas forming infection of the soft tissue were identified (Table 3). In five cases (83%) the infection was located in the lower extremity. None of the patients had diabetes mellitus. Three patients had a history of malignancy (rectal carcinoma, renal carcinoma, sarcoma). In two of these three patients metastatic disease was diagnosed after onset of infection. In three patients the infection was secondary to major surgery (one biaortofemoral vascular bypass, one peritonitis after a perforated gangrenous appendicitis, one infected hip arthroplasty). Three patients presented with intraabdominal lesions (one anastomosis dehision after rectal resection due to rectum carcinoma, one with a perforated sigmoid diverticulum and the before mentioned peritonitis following appendectomy). In the latter two patients the infection spread through the sciatic foramen into the thigh.
The patients underwent an average of 3.5 (SD 3) surgical interventions. High levels of C-reactive protein levels (29.8 mg/dl; SD 9.1) and leukocyte counts (21.8 mm3; SD 8.2) were also present on admission. The mean LRINEC score was 8.5 (SD 1). The infection was polymicrobial in two patients (33.3%), while the most frequent observed bacterium was Enterococcus faecium in two cases (33%). On average the patients were hospitalised for 29.3 days (SD 18). Three patients (50%) died due to GFM.
All patients were secondarily referred to our department with the tentative diagnosis of NSTI. Surgical debridement commenced 2.5 hours (SD 1.8) after admission to our department. Surgical debridement consisted of resection of the skin, subcutaneous tissue, the fascial layer and necrotic muscle tissue if affected.
DISCUSSION
The most important findings of this study are that in the majority of cases with NF, minor skin lesions were identified as the site of origin in contrast to patients with GFM. GFM was frequently associated with an intraabdominal pathology or secondary to major surgical trauma. Diabetes mellitus is the most common comorbidity in patients with NF whereas patients with GFM had a high incidence of malignancy. Clinically NF was a frequently observed to be an ascending infection in constrast to GFM which presented as a descending infection in all cases. Furthermore, patients who had secondary involvement of the trunk had a higher mortality rate.
In general, necrotizing fasciitis is an insidious rapidly progressing infection leading to necrosis of fascial layer. Due to anatomical borders the infection spreads along the fascial planes. During the initial phase NF maybe mistaken for cellulitis but prompt diagnosis is warranted (11).
Most of the time it is caused by a mixed aerobic/anaerobic bacterial infection. Only in a subset of cases a streptococcal group A infection is present. Mortality rates from 30 to 70% are described in the literature, which may even increase significantly if the infection progresses to myonecrosis (6 –9). However, in a recent study, Mills et al. reported a lower mortality rate with 12% in 688 patients with NSTI (12).
NSTI remains primarily a clinical diagnosis (7, 9). Several diagnostic measures such as the finger test, probe biopsy and frozen sections as well as ultrasound have been proposed as being useful adjuncts for early recognition of NSTI (2, 7, 13 –18). CT and MRI scans are considered dispensable and might delay the essential surgical intervention unnecessarily. With a fast and thorough surgical debridement in NF limb amputations can often times be avoided (19), whereas in gas gangrene with clostridial myonecrosis limb salvage is seldom feasible, rather the amputation in these cases has to be considered a possibly life saving treatment option.
Therapeutical strategies in patients with necrotizing soft tissue infections are early recognition followed by a through surgical debridement eliminating entire infected tissue (cutis/subcutaneous tissue, fascial layer and if affected musculature), an organism specific antibiotic regiment is the therapy of choice. Early enteral feeding, fluid resuscitation and an adequate intensive care treatment to overcome the symptoms of septic shock are corner stones of the treatment. Hyperbaric oxygen therapy has been described as beneficial but its value in the therapy remains controversial (20).
Previous studies have analyzed specific risk factors, e.g. diabetes mellitus, obesity, peripheral vascular disease, age and female gender in patients with necrotizing soft tissue infections (7, 21 –23), while the most common risk factor was diabetes mellitus, which was also confirmed in our study. In contrast to NF diabetes mellitus was not observed in patients with GFM.
In the presented study the LRINEC score proved to be a useful adjunct in the diagnosis of severe soft tissue infection. Su et al. reported that patients with a LRINEC score of ≥ 6 have higher rates of mortality and amputation (3). However, we did not find a statistical significant difference between deceased patients and survivors for patients with NSTI.
The incidence of GFM was very low and was encountered in only six patients over the five year interval. In contrast to NF, GFM originated either after major surgery or had an underlying intraabdominal source of infection. Four infections originated after bowel perforation and one further case after retroperitoneal surgery. None of these patients presented with clinical signs of acute abdomen or peritonitis.
Unlike NF in GFM the infection was observed to spread distally. The mean age of the patients was slightly above the age of NF patients. Three patients had a previous history of malignancy, out of these three patients two were found to suffer from metastatic disease, whereas none of the patients with NF presented with an intra-abdominal pathology or previous history of malignancy.
CONCLUSIONS
In conclusion, early diagnosis and extensive surgical debridement have been shown to improve the clinical outcome. In addition to a thorough clinical evaluation by experienced clinicians, simple laboratory parameters can be helpful in the early detection of NSTI and GFM. Hence in our opinion early diagnostic measures such as CT or MRI scan to rule out an intraabdominal process is warranted in these patients with descending gas forming infections to avoid a delay in eradication of the infection.
Footnotes
ACKNOWLEDGEMENT
The authors thank Dr. Ingo Stricker, M.D., Department of Pathology, BG University Hospital Bergmannsheil, Ruhr University Bochum, Bochum, Germany, for his support with the histological assessment.
