Abstract
Keywords
Opioid dependence (OD) is a chronic relapsing disorder with risk of major adverse health outcomes. According to European Monitoring Centre for Drugs and Drug Addiction (EMCDDA) definitions (EMCDDA, 2022), high-risk opioid use is defined as injecting use or long duration/regular use of opioids. Currently in Europe, there are about one million high-risk opioid users (EMCDDA, 2022), and in Finland, the number of high-risk opioid users has been estimated to have doubled from 12,700–15,000 in 2012 to 31,100 in 2017 (Rönkä et al., 2020). Alongside this, opioid-related fatal overdoses in Finland have also increased, especially in the younger population (Kriikku et al., 2022).
Opioid agonist treatment (OAT) has been shown to reduce all-cause mortality due to OD (Heikkinen et al., 2022; Santo et al., 2021), and it is recommended as standard of care for OD by the World Health Organization (WHO, 2009). In Europe, 514,000 opioid users received OAT in 2020. Approximately 20% of them were new to treatment while approximately 75% had previous treatment experience (EMCDDA, 2022). OAT, when provided as oral treatment, very often includes daily supervision of medicine intake to reduce misuse and diversion, as well as urine toxicology testing to control for the illicit use of opioids as well as other drugs (THL, 2022a, 2023; Wright et al., 2015).
Although the extent of illicit drug use is regularly estimated, the untreated population of high-risk opioid users remains poorly understood. Willingness to receive given medication has been shown to be complex and differ among opioid users (Tompkins et al., 2019). Further, the reasons why many high-risk opioid users are not currently entering treatment or why patients decide to leave OAT are little known. In addition, the reasons behind continuous opioid use have not been well studied.
In Finland, the number of patients receiving OAT increased by 44% between 2015 and 2019 (Partanen et al., 2022). On the other hand, it has been estimated that only up to 20% of high-risk opioid users are currently in treatment in Finland (Partanen et al., 2022; Selin et al., 2015). According to a recent discussion paper, the number of patients receiving OAT has further increased by 50% in Finland between 2019 and 2021, and 20% of all OAT patients in 2021 were new to treatment (THL, 2023).
The overarching objective of this study was to investigate the current situation of the untreated population of high-risk opioid users in Finland. The primary aim was to determine the reasons why high-risk opioid users do not enter treatment and to better understand their unwillingness to seek treatment. Furthermore, a secondary aim was to characterise the population of untreated high-risk opioid users in Finland, and the reasons behind their opioid use.
Methods
Study design and participants
This study was conducted as a survey in Finland between July 2021 and January 2022. An electronic questionnaire was available on iPads in needle exchange units. Participation was anonymous, voluntary and based on informed consent. The inclusion criterion was self-reported OD, and the exclusion criteria were (1) age under 16 years and (2) current OAT. The aim was to recruit 100–200 respondents.
The survey was designed in collaboration with Finnish addiction psychiatrists. To ensure that the questions and response options were understandable and relevant for the target population, the questionnaire was reviewed by nurses working in needle exchange units before launching the survey. The final electronic questionnaire consisted of 24 structured multiple-choice questions (without open fields) on the following topics: (1) demographic characteristics; (2) history of drug use, current drug use and possible treatment history; (3) opinions about OAT; and (4) burden of disease. Respondents were informed that they were allowed to leave questions unanswered or stop answering at any point, if they so wished. Since no identifiers were registered, the right to correct or delete data specified in the EU General Data Protection Regulation (GDPR) was not applicable to this survey or the resulting dataset.
To ensure nationwide coverage, data were collected in 10 cities in Finland (capital and urban areas), in a total of 16 needle exchange units that offer health counselling. Permission to collect data was gathered from all service providers, communal or private. The survey was conducted according to the guidelines of the Finnish National Advisory Board on Research Integrity (TENK, 2019). According to these guidelines, ethics committee approval is not required in surveys targeted to respondents aged 15 years or older.
Statistical analysis
Due to the nature of data collection (the right to leave questions unanswered), some values were missing, and the number of received answers varied between questions. Missing values were excluded from the analyses, and the results were based on the actual number of responses received per question.
Continuous variables were reported as medians and means, and categorial variables as total number and percentages of received responses. If the number of respondents was <5 in any certain category, the results were reported as not available (“NA”) on the pooled group level to protect the privacy of the singular respondents. Statistical software R version 3.6.1 (for Windows) was used in the analyses.
Results
Population characteristics
In total, 167 participants were eligible and responded to the questionnaire. Of the respondents, a majority (62%) were men, and approximately half (53%) were aged 34 years or younger (Table 1). For the majority (90%), the highest level of education was secondary education, and two-thirds (68%) were unemployed. A majority of the respondents were not in a relationship (72%) and almost one-third (28%) of them had underaged children. The most common form of accommodation was living in a rental apartment (73%).
Characteristics of the respondents

Completed or unfinished. bEmployee / subsidised employee / rehabilitative employment activity / work trial. cPart-time or full disability pension / another pension type. dIf the sum of n under certain variable differs from N of that variable categories have been masked to protect anonymity.
Drug use history
The median age for commencing illegal drug use was 17 years (mean age = 19.27 years, N = 161). At the time of the survey, two-thirds (66%) of the respondents had used opioids longer than 6 years (Table 2). The median age for starting intravenous (IV) drug use was 20 years (mean age = 21.3 years, N = 149).
Drug use and treatment history.
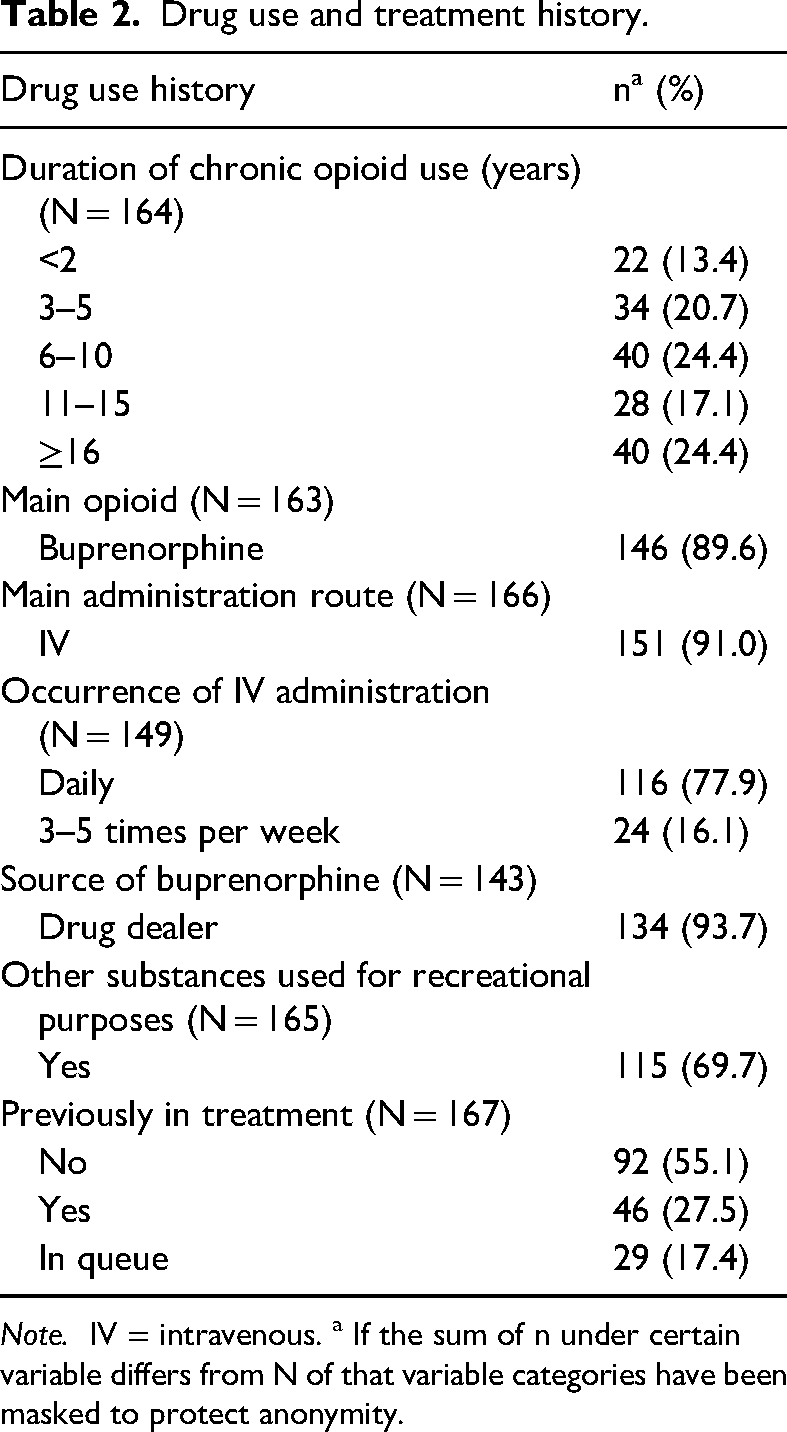
Note. IV = intravenous. a If the sum of n under certain variable differs from N of that variable categories have been masked to protect anonymity.
The most commonly used opioids were buprenorphine (95%), oxycodone (24%) and tramadol (24%) (Figure 1). Of all respondents, 60% used only one opioid. The majority (91%) administrated opioids mainly IV and for most respondents (78%) IV administration occurred on a daily basis (Table 2). Approximately 90% of the respondents named buprenorphine as their main opioid in use, and most of them (94%) got their buprenorphine (with or without naloxone) from a dealer (Table 2).
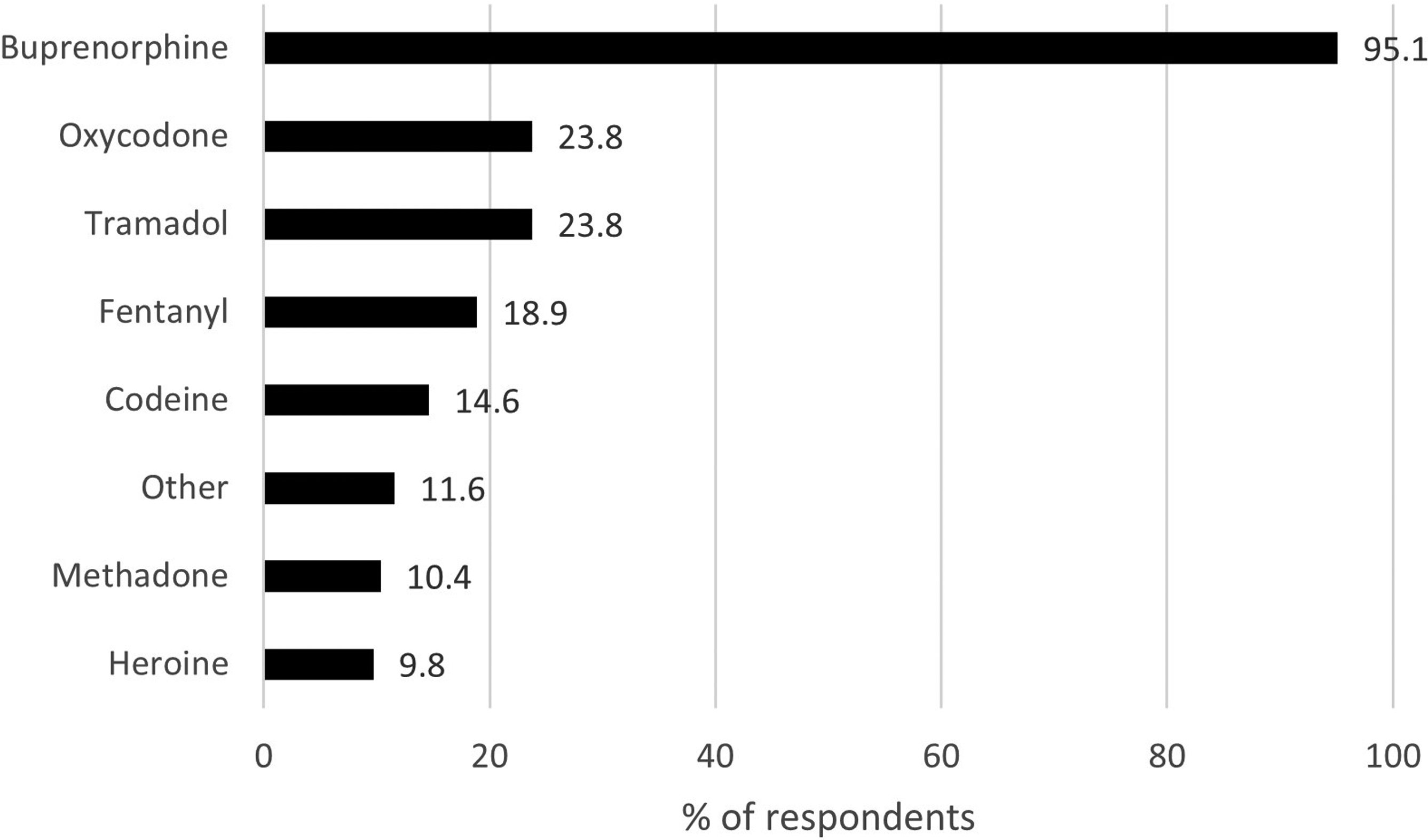
Currently used opioids (N = 164) presented by % of respondents (with the possibility to select several options in the multiple-choice questionnaire).
A majority of the respondents used opioids as self-medication for withdrawal symptoms (75%), to treat psychological symptoms such as anxiety or depression (59%), or for pain management (43%). Less than one-fifth (19%) used opioids to get high or for recreational purposes. More than two-thirds (70%) of the respondents used other substances for recreational purposes (Table 2). Of them, 80% used more than one other substance besides opioids. The most commonly used substances other than opioids were stimulants (82%), sedatives or anxiolytics (67%), and cannabis (61%) (Figure 2).
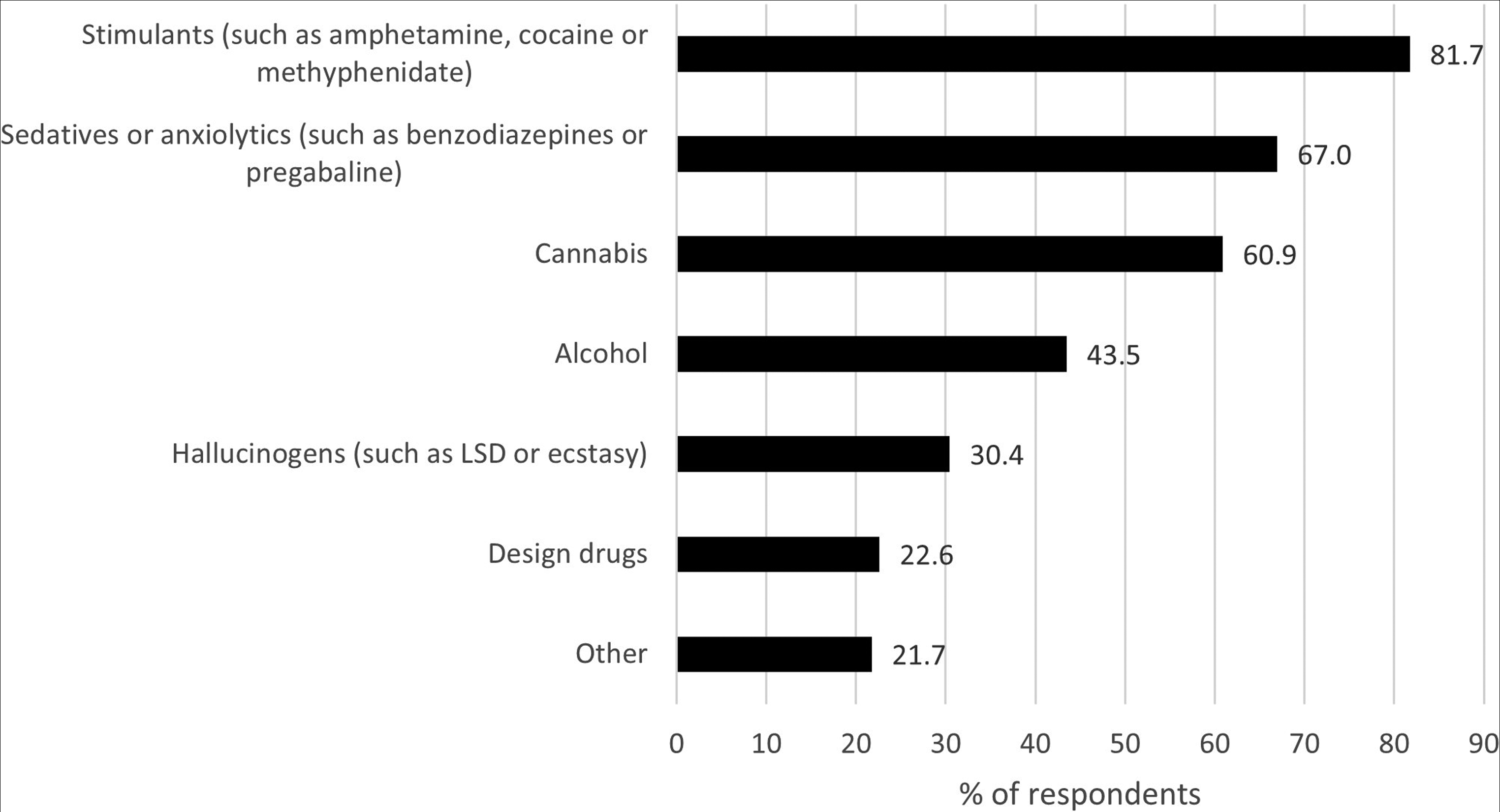
Other substances used for recreational purposes (N = 115) presented by % of respondents (possibility to select several options in the multiple-choice questionnaire). LSD = lysergic acid diethylamide.
Of the respondents, 28% had previous experience with OAT (of which 61% with sublingual buprenorphine-naloxone) but the treatment had been discontinued, 17% were currently on a waiting list for treatment, and 55% had no previous history of OAT (Table 2). Of those that had previous experience of OAT, 67% described that the discontinuation was their own decision, 22% that the decision was made by the clinic alone, and the remaining 11% that the decision was made together with the clinic.
The respondents who were waiting for OAT belonged primarily to the age group 25–34 years (52%) and had been using opioids for more than 6 years (70%). Most of these respondents (86%) were not in a relationship (single, separated, divorced or widowed). Among these people who were waiting for OAT, self-treatment of psychological symptoms as well as recreational use of opioids was lower than among other opioid users (Figure 3).
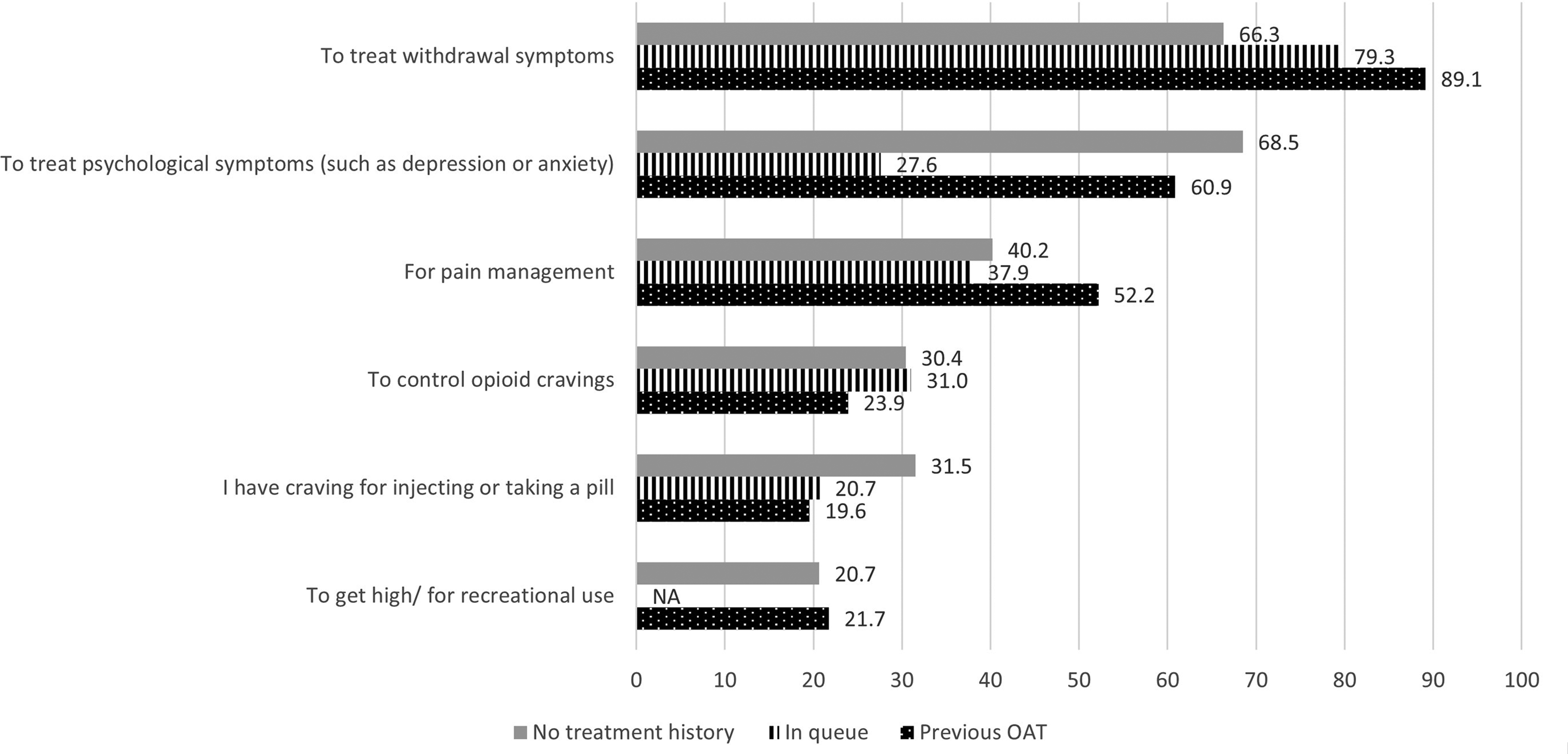
Reasons why opioids are used (N = 167) presented by % of respondents (with the possibility to select several options in the multiple-choice questionnaire) divided by treatment history. NA = masked (N < 5). OAT = opioid agonist treatment.
Opinions about opioid agonist treatment
The most common selected reasons why respondents were currently not in OAT were as follows (Figure 4): (1) seeking treatment is too difficult (37%); (2) treatment is too binding (36%); and (3) fear of actions from authorities (23%). The proportion of respondents that chose option “Other reason” was 30%. Those who were waiting for OAT felt less often that the treatment was too binding (Figure 4). Those with no treatment history did not want to speak about their drug use or were afraid of actions from authorities more often than the other respondents. In the youngest age group (<25 years), being afraid of actions from authorities was the most common reason to stay out of OAT (50% of the respondents in this age group).
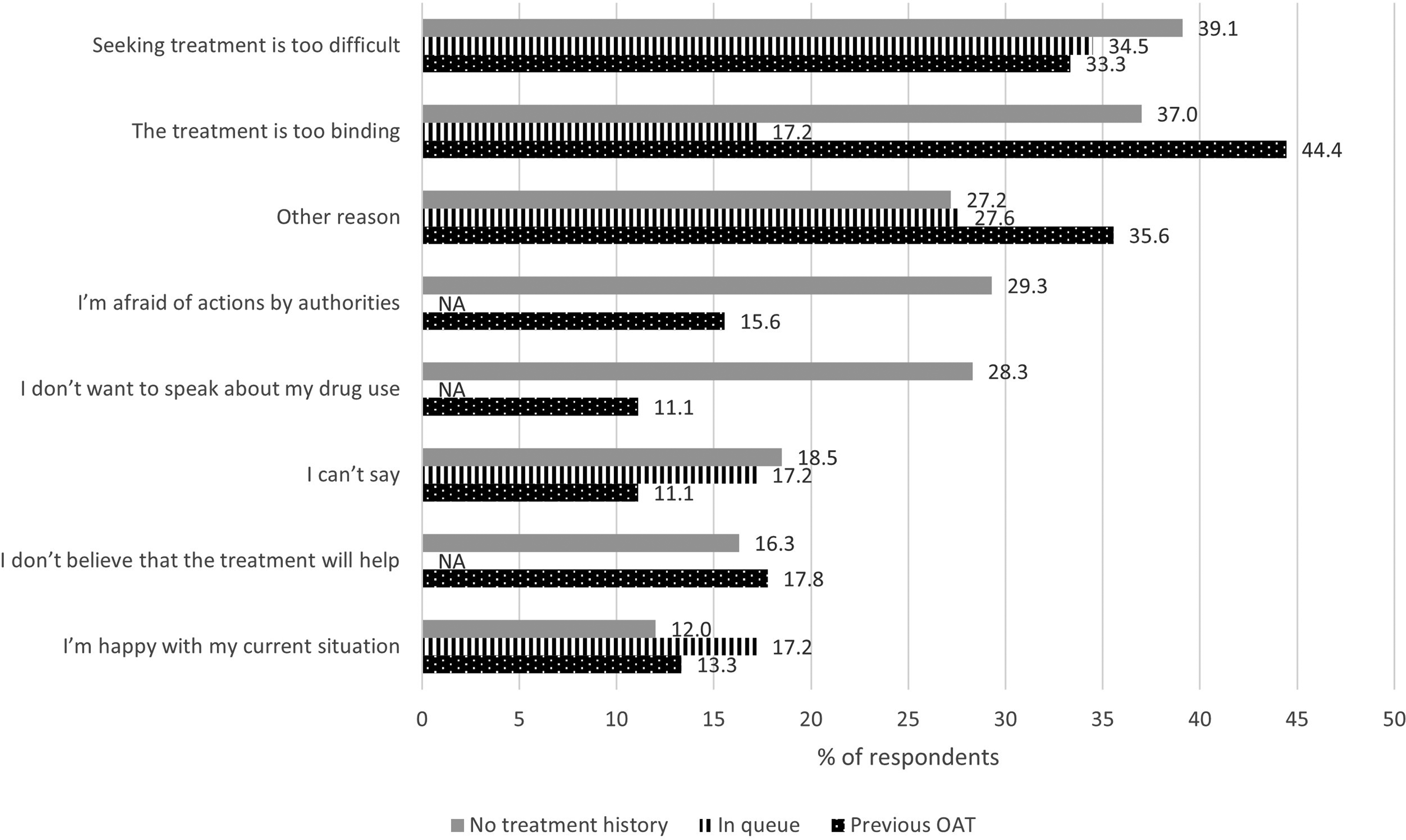
Reasons why not currently under treatment (N = 166) presented by % of respondents (with the possibility to select several options in the multiple-choice questionnaire) divided by treatment history. NA = masked (N < 5). OAT = opioid agonist treatment.
A large proportion (44%) of the respondents with previous experience of OAT believed that the treatment was too binding and named this as the most common reason for not currently being in OAT (Figure 4). This was also the most common reason (40%) for discontinuation from their previous OAT treatment (Figure 5). The two following reasons for discontinuing the OAT were equally common (33%): rules related to the treatment were too strict; and they wished to try to discontinue opioid use without medication.
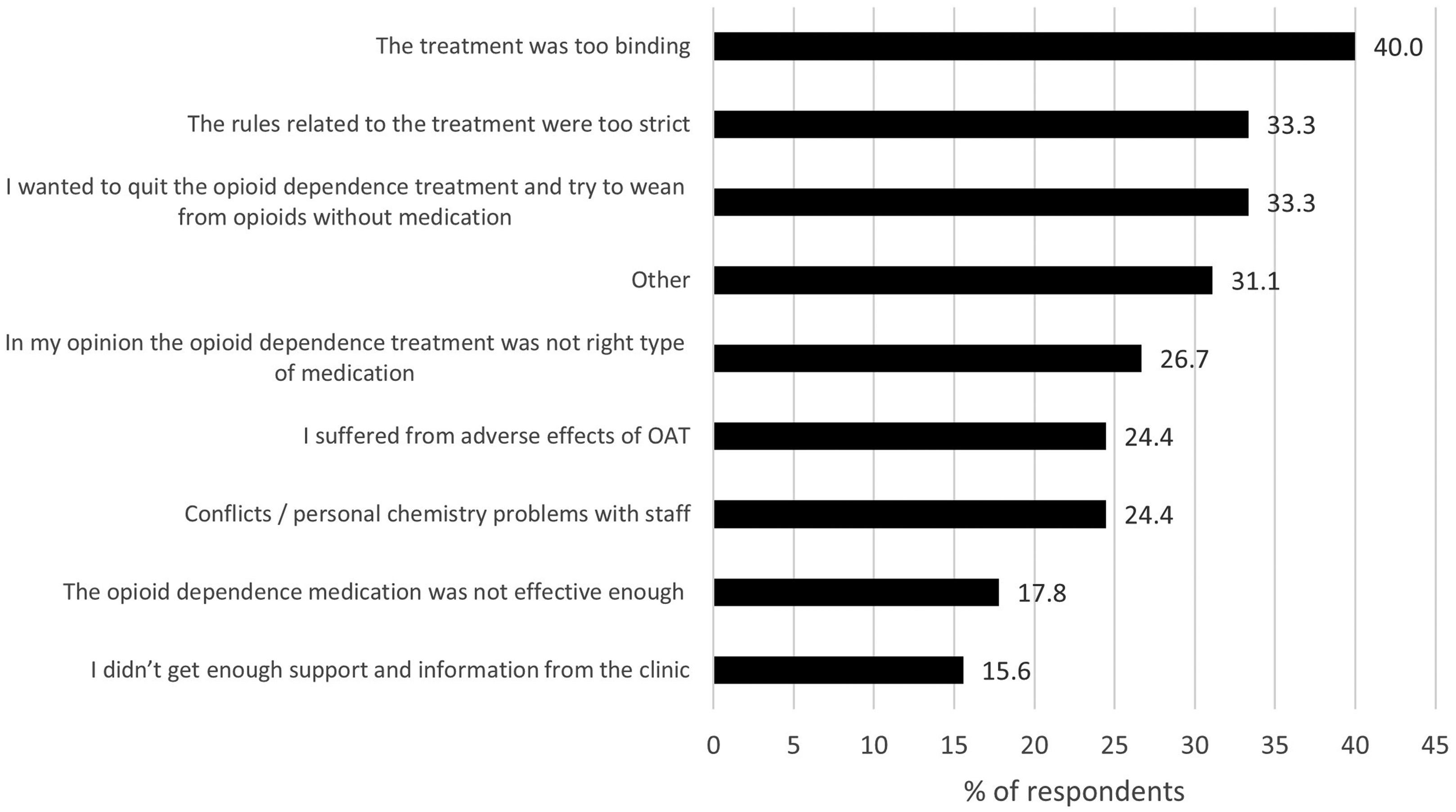
Reasons why previous treatment had discontinued (N = 45) presented by % of respondents (with the possibility to select several options in the multiple-choice questionnaire). OAT = opioid agonist treatment.
When asked about their thoughts on the availability of information regarding OAT and where to seek treatment, 35% of the respondents felt that they did not have enough information about OAT and 18% did not know where to seek help for their OD. Out of the youngest respondents (<25 years), 50% felt that they did not have enough information about OAT. A majority of the respondents (84%) were aware that long-acting injectable buprenorphine (LAIB) treatment was available, administered subcutaneously in a clinic once a week or once a month. Of the respondents, 42% felt that information about LAIB treatment increased their willingness to seek treatment.
Burden of disease
Most of the participants (81%) responded that they use public healthcare services. However, only 41% were tested regularly for hepatitis C infection. Of all respondents, about half always used clean needles for IV use (52%); this was also the case for most of the remaining respondents if clean needles were available (43%).
Of the respondents, 62% (N = 165) answered that they had or had previously had a hepatitis C infection (Supplementary Figure 1a). Out of these, 60% (N = 104) answered that the infection had not been treated (Supplementary Figure 1b).
The prevalence of hepatitis C infections increased in relation to longer use of opioids. It ranged from 32% in respondents with less than 2 years of opioid use to 83% in respondents with more than 15 years of opioid use. Despite this, hepatitis C infections were common in relatively young respondents: 59% in the 25–34-year age group and 75% in the 35–44-year age group.
Discussion
This study showed that the most common reason for high-risk opioid users to stay out of OAT was difficulty to seek treatment. This suggests that there is a need for increasing information about available treatments targeted to high-risk users, together with uncomplicated information on how to contact the appropriate treatment providers.
In addition, importantly, in several highly regulated countries, such as Finland and Sweden, accessibility of OAT may be difficult due to strict rules, time-consuming evaluation processes and further lack of resources (Gedeon et al., 2019; Johnson et al., 2022; Rönkä & Niemelä, 2020). In Finland, the evaluation processes and queues for treatment are long, as long as several months, and there are regional differences in accessibility to OAT (Rönkä & Niemelä, 2020). Depending on the treatment provider, a high-risk opioid user may need to go through a two-phase assessment to enter treatment: first an initial referral in an assessment proceeding unit and only then the actual OAT assessment. In addition, rules related to required follow-up periods, discontinuation attempts and use of other substances may differ between units, and these may further prolong the assessment process. After the treatment need has finally been determined, the actual OAT initiation may be delayed, and in the worst case, the patient may need to be referred to yet another unit for treatment initiation. In this survey, 17% of the respondents were on the waiting list for initiating treatment, but waiting times were unknown. Legislation related to treatment for patients with OD has recently been updated in Finland, which from one's part enables reduction of the complexity of the processes in the future (Finlex 642/2023).
This study was targeted to respondents aged 16 years and older. The youngest participants were, however, 18 years old. This raises a question whether underaged people had a barrier for participating in the study or whether they overstated their age. In Finland, the legislation obliges healthcare professionals to give child welfare notifications with a low threshold (Finlex 417/2007), which might have affected the lack of underaged respondents even in an anonymous study. In this study, the respondents’ age at starting using illegal drugs began at 10 years and age at starting IV opioid use at 12 years. In further studies, it would be important to try to reach the youngest opioid users. This would be especially important in Finland, where fatal opioid overdoses have recently become more common among people aged under 25 years (Kriikku et al., 2022).
Fear of actions from authorities was also the most common reason to stay out of OAT in the youngest population (aged 18–24 years) in our study. Since no open text boxes were used in this survey, particular reasons remain unclear, but the above-mentioned Child Welfare Act is a possible reason. Fear of actions from authorities as a reason to stay out of OAT was also more common among women than among men (43% vs. 18%, respectively). In addition to child protection issues, one reason might be the Driving Licence Act (Finlex 386/2011), which obliges healthcare professionals to inform the police if a driver's health conditions are not met.
High levels of control may lead to mistrust between patients and staff and may prevent patients from entering treatment or adhering to treatment programs (Richert and Johnson, 2015). Shared negative peer experiences may influence the opinions of treatment-naïve individuals as well. Even if more than 70% of the respondents in this study had not been on OAT before, 35% thought that the treatment would be too binding. This was also the most common reason for discontinuing previous OAT. These results are in line with findings from the EQUATOR project from 2012, where 34% of the respondents (patients on medication-assisted treatment for OD) stated that greater flexibility in the rules would have made them start OAT earlier, and 38% stated that fewer rules would have made it easier for them to stay in treatment (Benyamina, 2014).
OAT is globally recommended as a first-line treatment for OD maintenance treatment (WHO, 2009). OAT in Finland, as in many other western countries, should be given in conjunction with psychosocial interventions. A systematic review from 2016 concluded that study results generally support psychosocial interventions when they are given in addition to OAT, although the additional effectiveness varies across studies, outcomes, medications and interventions (Dugosh et al., 2016). This survey did not specifically investigate the interest in non-pharmacological versus opioid agonist treatment.
According to the present study, knowledge about the availability of LAIB formulations increases willingness to seek treatment. This result was expected since it has previously been described that the main benefits of depot injection, compared to oral treatment, are increased freedom in everyday life, greater possibilities to decide over their own time, increased opportunities to travel, increased sense of freedom and “normality” and reduced stigma, as well as a shift in self-perception (Johnson et al., 2022). The results regarding LAIB increasing willingness to treatment are also in line with another study where most opioid users, currently not in treatment, were willing to try LAIB if offered it (Tompkins et al., 2019). Several participants in the Tompkins study also expressed that they wanted more information to be able to decide whether to try LAIB or not. Along the same line, over 35% did not feel that they had enough information about OAT in general. This fits very well with our findings that more than one-third of the respondents thought they did not have enough information about OAT, and almost one-fifth did not know where to seek treatment.
Despite COVID-19, when needle exchange rooms were closed and service was occasionally arranged outdoors, the target population could be reached, and the aimed number of responses was collected. Participation in the survey was high, with a response rate over 99% (among those who started answering), even though it was possible to skip answers. However, a survey setting is prone to biases. One type of limitation is that this study only targeted people visiting needle exchange units, possibly causing selection bias. However, our sample appears to reflect the general population of high-risk opioid users in Finland. Unlike other European countries, the most misused IV drug in Finland is buprenorphine. This has been shown by looking at residues in used syringes (EMCDDA, 2021) and was also verified in our study as more than 90% of the respondents named buprenorphine as their main opioid. The number of IV users was high (over 90%), and almost all respondents used IV opioids daily. More than half were long-term users with opioid use longer than 6 years. This indicates that we reached opioid-dependent respondents who would be eligible for OAT in Finland (Finlex 642/2023) and the results well represented their opinions. On the other hand, other substances than opioids were commonly used in our study (70% used at least one other substance), which did not differ much from opioid users in treatment in Finland (Mäkelä et al., 2019).
Among the respondents, almost everyone acquired their buprenorphine from a drug dealer. Even if the opioids were named to originate from a drug dealer, it does not exclude the possibility that they were, originally, legally prescribed and purchased from Finnish pharmacies or received as OAT. In 2022, approximately 23% of the buprenorphine found in opioid traces in syringes originated from the buprenorphine-naloxone combination (Suboxone) commonly used in OAT in Finland, and the remaining portion most likely originated from illegally imported mono-buprenorphine (Subutex) (THL, 2022b).
In our study, opioids were used mainly for withdrawal symptoms but also for psychological symptoms and pain management. For respondents who were queuing for OAT, the control of opioid cravings was the most common reason for opioid use. These people thought less often that the treatment protocol was too binding, and they were happier with their current situation compared to other respondents, indicating the importance of easy access to treatment.
In conclusion, the results of this anonymous survey among high-risk opioid users highlight difficulties in seeking treatment and that treatment is considered too binding. There is a need for increase in general as well as targeted information about, accessibility to, acceptance for and individualisation of OAT.
Supplemental Material
sj-docx-1-nad-10.1177_14550725231204723 - Supplemental material for Reasons for not entering opioid agonist treatment: A survey among high-risk opioid users in Finland
Supplemental material, sj-docx-1-nad-10.1177_14550725231204723 for Reasons for not entering opioid agonist treatment: A survey among high-risk opioid users in Finland by Tuire Prami, Mari Pölkki, Jarno Ruotsalainen, Elin Banke Nordbeck, Susanna Meyner, Ari Kaski in Nordic Studies on Alcohol and Drugs
Footnotes
Acknowledgements
We are deeply grateful to all respondents as well as the personnel in the 16 needle exchange units for their collaboration in data collection. BSc Pharm Mia Nevalainen, BSc Pharm Iiro Mytty and study nurse Merja Pihlaja are acknowledged for their excellent study coordination work.
Declaration of conflicting interests
The authors declare the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: SM and EBN are employees of Camurus; AK has received consultation fees from Camurus, Indivior, AbbVie, and Orion Pharma; TP, MP, and JR report no conflicts of interest.
Funding
Financial support for the research, authorship, and/or publication of this article was given by Camurus AB, Lund, Sweden.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
