Abstract
The use of screening and brief interventions (SBI) for hazardous and harmful alcohol use in primary healthcare (PHC) and other settings has been recognised as an evidence-based and effective strategy in addressing the needs of the many patients presenting in these settings and helping them to reduce or stop their drinking altogether before health and social consequences become pronounced (Bertholet et al., 2005; Kaner et al., 2009; World Health Organization, 2001). Facilitating access to SBI, treatment and care for alcohol use disorders (AUDs), and co-morbid conditions has been included into the WHO-led initiative and technical package called “SAFER”, which outlines five high-impact strategies that can help governments in reducing alcohol use and related health, social and economic consequences at the population level (World Health Organization, 2020).
As part of this approach, the use of structured and tested tools, such as the Alcohol Use Disorders Identification Test (AUDIT), is recommended to routinely assess the level of risk for AUDs in all patients presenting at PHC settings and to identify those who might benefit from reducing their alcohol consumption (World Health Organization, 2019b).
The AUDIT was developed as a standard assessment instrument by a WHO Collaborative Project on Identification and Treatment of Persons with Harmful Alcohol Consumption, which aimed to build the scientific basis for the use of SBI in primary care settings (Saunders et al., 1987). The AUDIT is a short test that consists of 10 questions divided into three domains: (1) three items on alcohol consumption to detect hazardous use patterns; (2) four items on dependence symptoms; and (3) three items on problems related to alcohol use. It can be applied with a minimum amount of time and resources in PHC facilities, and the first three of its consumption items from the first domain form an abbreviated version known as the AUDIT-C (Bradley et al., 2003, 2007; Bush et al., 1998).
Although the AUDIT was developed in six very different countries (Australia, Bulgaria, Kenya, Mexico, Norway and the United States), and a total of 13 countries, including the Russian Federation, have participated in Phase 4 of the larger WHO project mentioned above, which aimed to implement SBI in the PHC systems in these 13 countries, it was never formally adapted and validated for use in the Russian Federation (World Health Organization, 2006). This was quickly recognised as one of many existing barriers to implementing SBI in Russia by an international expert group, which was involved in tailoring the existing WHO materials on SBI to the local context. Following the development of the WHO training manual on alcohol brief interventions for primary care and its translation into Russian (World Health Organization, 2017), the group was soon confronted with inconsistent versions of the existing Russian translations of the AUDIT. Systematic reviews of the different versions of the AUDIT in the Russian language as well any validation exercises specifically in the Russian Federation have confirmed that, so far, the formal process of test translation and validation has not been carried out for any of the existing Russian versions of the AUDIT (Bunova et al., 2021; Neufeld et al., 2021a).
However, there is a series of studies, which have examined the performance of the AUDIT, i.e., various translated versions of the tool with undocumented translation procedures, in various patient samples, comparing its performance with the outcomes of drinking diaries and exploring correlations with different instruments, such as the Composite International Diagnostic Interview (CIDI), CAGE or Beck's Depression Inventory (Barinova et al., 2013b; Plavinski et al., 2012b, 2013a; Yanov et al., 2009). For instance, a study involving a relatively large sample (n = 1538), comprising subsamples of medical students (n = 529), patients of general practitioners (n = 411), patients of dermatovenerologists (n = 581) and patients seeking obstetric/gynaecological care (n = 17), compared the AUDIT scores with alcohol consumption diaries, revealing a weak correlation between the two (Plavinski et al., 2012b). Another study comparing AUDIT and CAGE outcomes with drinking diaries in a sample of men from detention facilities (n = 100, aged 19–47 years) and age-matched controls (n = 100) who were patients of general practitioners (n = 50) and patients of dermatovenerologists (n = 191) suggested a high alcohol intake in the prisoners group before detention as measured through drinking diaries (average alcohol intake of about 450 g of ethanol per week); however, this could not be adequately measured through the first three AUDIT items because the first item assesses current consumption status (Barinova et al., 2013b). Various other studies from the same research group have investigated the performance of the AUDIT in older adults and various patient groups as well as its correlation with biomarkers and skin symptoms that might predict higher alcohol intake (Barinova et al., 2013a; Plavinski et al., 2013b). However, the group seemed to have used at least two different Russian translations of the AUDIT, both of which featured modified second and third test items with a different count of standard drinks, and no documented explanations for these modifications could be identified (e.g., the second test item featured up to 13 standard drinks and more in the final answer option instead of ≥10 standard drinks) (Bunova et al., 2021).
Besides the need to follow established translation rules for instruments, such as the formal rules suggested by the WHO (World Health Organization, 2009), local adaptation procedures are sometimes required to ensure that the test is measuring the concept that it is supposed to measure (Li et al., 2011). Thanks to its international popularity, the AUDIT is available in many different languages, sometimes with region-specific translations, as is seen for various Chinese versions, and at least some of these versions are based on local validation studies and/or are informed by other empirical evidence (Leung & Arthur, 2000; Li et al., 2011). For Russia, however, such studies were never carried out for the PHC setting and the only identified validation study had a small sample size (n = 252) and was limited to the specialised setting of a tuberculosis treatment facility in Siberia, reporting a sensitivity of 91.7% and specificity of 44.6% for cut-off scores ≥8 (Neufeld et al., 2021a; Yanov et al., 2009).
Recognising the need to formally translate and validate the RUS-AUDIT in order to ensure the successful implementation of the SBI strategy in the country, the Russian Ministry of Health has initiated the RUS-AUDIT validation project and, with the support of the WHO Regional Office for Europe, formed a Project Advisory Board (PAB) involving experts from various disciplines (for an overview of stakeholders represented on the Board, please see Appendix A). Considering the issues with the different translations of the AUDIT in Russian and the existing literature on the distinct impact of Russian drinking patterns on health and the need to account for the known specificities as part of the screening process, the PAB experts initiated a set of formal translation and adaptation procedures. The goal was to construct a Russian version of the test to be used in a validation study in PHC facilities in different regions of Russia, the results of which have been recently published (Rehm et al., 2020; World Health Organization, 2021a, 2021b).
According to the current regulations of the Ministry of Health of the Russian Federation, the prevention, treatment and rehabilitation of substance use disorders, including detoxification and rehabilitation, are carried out in the specialised field of narcology (Ministry of Health of the Russian Federation, 2015). Since 2011, the provision of narcological care has been governed by a national law and before that it was subject to a number of regional regulations (Koshkina & Babicheva, 2014). Government-run narcological services are provided at no cost, but the mandatory narcological registration that is required to use these services entails mandated monitoring processes that restrict job opportunities and have other consequences, such as the potential loss of one's driver's license (Bobrova et al., 2008). As part of the diagnostic procedures, narcologists asses the ICD-10 diagnostic criteria for AUDs in the form of a consultation, following an established clinical protocol for the detection and management of AUDs (Ministry of Health of the Russian Federation, 2021b). Screening procedures for potential AUDs are currently conducted as part of large-scale population screenings of “dispanserisation” in PHC facilities, which comprise a wide range of regular preventive check-ups (Ministry of Health of the Russian Federation, 2021a). However, it is documented that the dispanserisation programme does not cover the entire population (Ministry of Health of the Russian Federation, 2021a) and systematically omits population groups at highest risk of substance use disorders, such as, for example, young men, and that there are specific issues with the used AUDIT versions as part of dispanserisation (Starodubov et al., 2019).
The aim of the present study was to inform on the different steps taken to construct the RUS-AUDIT and document the process of translating, adapting and testing the RUS-AUDIT in patients and healthcare professionals in PHC as well as specialised alcohol and drug treatment services in Russia, known as narcology services (a special subdiscipline of psychiatry in Russia and some other countries of the former Soviet Union).
Material and methods
Translation and adaptation procedures
The overall process of developing the RUS-AUDIT followed the formal WHO procedures for test translation and adaptation, involving translators, bilingual experts and an expert panel to discuss the new version of the test (World Health Organization, 2009).
A forward translation from English into Russian was done by a group of specialists as part of the Russian translation of the WHO alcohol brief intervention training manual for primary care, which provides a “train-the-trainer” SBI toolkit (World Health Organization, 2017). The version was discussed in a RUS-AUDIT PAB meeting, which was the first time the expert panel was convened. Different versions of the identified translations as informed by the outcomes of the systematic searches (Bunova et al., 2021; Neufeld et al., 2021a) were presented to the panel and discussed.
A second expert panel was formed with professional translators, communication specialists and linguists who were consulted separately during the process. The different versions of possible translations were then considered and discussed by a smaller group of experts who had experience in instrument development, translation and validation. As a result of this multi-step consultation process, and also informed by the results of qualitative expert interviews with PHC and narcology patients, as well as health professionals on their perceptions of the AUDIT and its ability to assess Russia-specific drinking patterns (Neufeld et al., 2022), an adapted Russian version of the AUDIT was constructed to be tested in a first pilot study at the Center for Therapy and Preventive Medicine (Appendix B, Table 1). Since most of the challenges in the use of the AUDIT in Russia related to the first three consumption items of the AUDIT-C, and, more specifically, to the representation of standard drinks (SDs), and the understanding of what constituted a heavy drinking occasion, the first RUS-AUDIT pilot version used pictorial material as part of the second test item to represent volumes of different alcoholic beverages and to convert them into SDs. In addition, an alternative representation of the third item on heavy episodic drinking was chosen, asking for the maximum drinking intake, not in terms of SDs but rather in specific volumes of the most common beverages—beer, wine and vodka. An additional open-ended question on heavy drinking occasions was added, asking for the maximum amount consumed on one occasion in the past three months. A back-translation of this penultimate version was then made by an independent expert, whose mother tongue was English and who had no previous knowledge of the questionnaire. Some issues and discrepancies were discussed again in the smaller expert group and a version was prepared for a quantitative pilot study with cognitive debriefing elements for the consumption items.
Pilot testing of the translated and adapted version
The RUS-AUDIT was then test-piloted in a total of four different pilot phases in patients from different institutions in Moscow, namely the Center for Therapy and Preventive Medicine and a city polyclinic in Moscow, which are both providers of PHC services as per Russian law, as well as patients from the Moscow Research and Practical Centre for Narcology and the National Research Centre on Addictions – branch, V. Serbsky National Medical Research Centre for Psychiatry and Narcology (for more details on the different pilot phases and results, see Appendix B). The sampling frame included all patients who visited the respective facilities on the day of the interviews and provided consent to participate in the interview. All interviews were carried out by trained interviewers between 25 March 25 and 2 July 2019, and followed a standardised script. Only patients who were aged at least 18 years, provided verbal consent and were current drinkers (had consumed alcohol in the past 12 months) were included in the sampling frame.
Next, we describe the different phases involved in piloting the adapted RUS-AUDIT, the most important changes made to the test, and present its final version as it was suggested for a large-scale validation study in different Russian regions, the protocol and results of which are reported elsewhere (Neufeld et al., 2021b; Rehm et al., 2020; World Health Organization, 2021a, 2021b).
Results
First pilot phase in the Center for Therapy and Preventive Medicine and changes to the instrument
The first version of the RUS-AUDIT was piloted on a sample of 79 patients, all recruited from the Center for Therapy and Preventive Medicine. The main objective of this first pilot phase was to empirically test if the constructed version of the RUS-AUDIT (Table 1 of the Appendix) was understandable for the respondents and whether it was comprehensive enough to detect hazardous alcohol consumption and potential AUDs.
The sample included patients from cardiology, surgery and internal medicine inpatient units, as well as the health centre of the facility, a separate unit that specialises in the promotion of healthy lifestyles (Consultant.ru, 2009). The participants of an ongoing nutrition study that was carried out in the health centre were also included in the sample as the researchers for that study had asked the preventive services unit of the PHC facility to be included. Patients were approached in the facility by two interviewers and asked for informed verbal consent to participate in the study in accordance with a standardised script (see Appendix B). Interviews were conducted face to face, either in a separate room or in the hallway of the medical unit involved, and were audiotaped for further analysis. All patients were administered the first three test items of the adapted version of the AUDIT and randomly assigned to answer one-third of the remaining items in order to save time, as these items were not found to be problematic by the pre-studies conducted (Bunova et al., 2021; Neufeld et al., 2021a). A cognitive debriefing was carried out after each test item by means of several questions, and general observations were documented by the interviewers (Ryan et al., 2012).
The majority of the interviewed patients from this first pilot phase were women (n = 52), and it was therefore decided that a quota of at least 50% men should be introduced in future to ensure that the RUS-AUDIT was sufficiently tested in men.
Overall, patients did not have any major issues in replying to the questions, and the analysis of the cognitive debriefing elements did not yield any additional value as almost all patients said they did not have any difficulties in understanding or replying to the questions. However, since almost half of the sample (n = 38 patients) reported their drinking volume on a typical drinking day in litres, millilitres or even grams when asked in an open-ended question format, a conversion table of beverage volumes was added to help interviewers quantify standard drinks for the second test item (see Table 2 in Appendix B for the conversion table and for further changes which were introduced). A series of changes in wording and grammar was made after an internal discussion with the small expert panel and a revised version was piloted for use in the next phase.
Second pilot phase in the Center for Therapy and Preventive Medicine and panel discussion
The second pilot phase of the RUS-AUDIT also took place in the Center for Therapy and Preventive Medicine, this time involving a sample of 32 patients (18 of whom were male, thanks to the newly established sampling quota). The second pilot was dedicated to fine-tuning the RUS-AUDIT questionnaire and, specifically, the newly developed conversion tables, which were tested on several patients using a black-and-white pictogram form (for more details, see Tables 3 and 4 in Appendix B). It was quickly found that a coloured show card with realistic (photographic) images of alcoholic beverages worked better for assessment and quantification because it considerably reduced the reaction times of patients navigating the table and attempting to recall typical drinking volumes. An additional test item assessing heavy episodic drinking was added to the test and reformulated in the course of the pilot. Moreover, the wording of all test items that used the time window of “last year” as a reference point was changed to “12 months” to ensure that patients referred to the time window of the prior 12 months instead of the previous calendar year. The revised version as based on this second pilot phase (Table 5, Appendix B) was then presented to the RUS-AUDIT PAB, along with a short report on the first pilot phase that documented all the changes and decisions made so far. In addition to the PAB expert group, two separate consultations, restricted to preventive medicine and narcology specialists, were carried out.
A series of further amendments was introduced as a result of this discussion and a decision was made to extend the piloting of the newly revised form (Table 6, Appendix B) to include patients from the polyclinic and the narcological settings. The main changes concerned the conversion table for the second test item and the wording of several test items. The newly added test items on heavy episodic drinking were reformulated and then moved to the end of the test to ensure that they did not disturb the initial structure and flow of the AUDIT and therefore did not introduce any bias. Detailed changes are documented in Appendix B.
Results of the third pilot phase in polyclinical and narcological settings and construction of the final RUS-AUDIT form for the validation study
Based on the discussions of the RUS-AUDIT PAB and the resulting changes to the RUS-AUDIT, a third pilot phase was initiated, this time expanding to polyclinics (large outpatient facilities that are the main providers of PHC services in Russia) and the narcology setting to explore how the test performed in people with confirmed AUDs. As part of this phase, a total of 40 patients were interviewed, 12 of whom were patients from a city polyclinic in Moscow, 14 from the Moscow Research and Practical Centre for Narcology and 14 from the National Research Centre on Addictions – branch, V. Serbsky National Medical Research Centre for Psychiatry and Narcology.
As part of this pilot phase, some minor changes to the third test item were introduced and a separate show card with a frequency–volume conversion table was developed as a hand-out aid for the patient. It also served as an assistive device for the interviewer, to help with standard drink quantification and score calculation for the second test item. The show card (Table 1) was received by patients and interviewers alike and was found to be useful in dealing with the already known and documented issues related to the lack of a concept for standard drinks in Russia.
The RUS-AUDIT show card and conversion table that assists the interviewers with the quantification of standard drinks (SDs) in the second test item.

The show card serves to simultaneously clarify different concepts: (1) it provides visual examples and familiar volumes of the different alcoholic beverages typically consumed in Russia that are known to the patients; (2) it provides a conversion table of drinking volumes and standard drinks that is easy to navigate for the interviewer; and (3) it still encompasses the concept of the standard drinks, but no longer makes it the focus of the second test item as the patient does not need to understand it in order to give a meaningful and valid response.
Both the revised RUS-AUDIT (Table 6) and the show card (Table 1) performed well as part of the pilot and were found to be feasible and comprehensible by the patients. Some issues were reported specifically for the narcological subsamples as the patients did not know how to respond to the first question on drinking frequency because they had stopped drinking as part of their treatment. Therefore, the show card with the conversion table did not reflect the volume and number of standard drinks they consumed on a typical drinking day.
The final RUS-AUDIT version (Table 2 and Table 7 in Appendix B) was then tested on a total of 16 patients, 11 of whom were recruited in a city polyclinic and five from the National Research Centre on Addictions – branch, V. Serbsky National Medical Research Centre for Psychiatry and Narcology.
Final RUS-AUDIT version as prepared for the main validation study.

The RUS-AUDIT was administered together with the show card and other instruments that were field-tested for the RUS-AUDIT validation study, such as the Russian version of the Kessler Psychological Distress Scale (K10) (Kessler et al., 2003) and the AUD module of the CIDI (Neufeld et al., 2021b; Rehm et al., 2015, 2020). This final pilot test suggested that the materials are well understood by patients and that there was no need for further adaptation.
General findings from the entire pilot of the RUS-AUDIT instrument
Characteristics of the different sub-samples of PHC and narcology patients as well as the characteristics of the total sample as recruited during all the described pilot phases can be found in Table 3.
Sample characteristics of the different subsamples
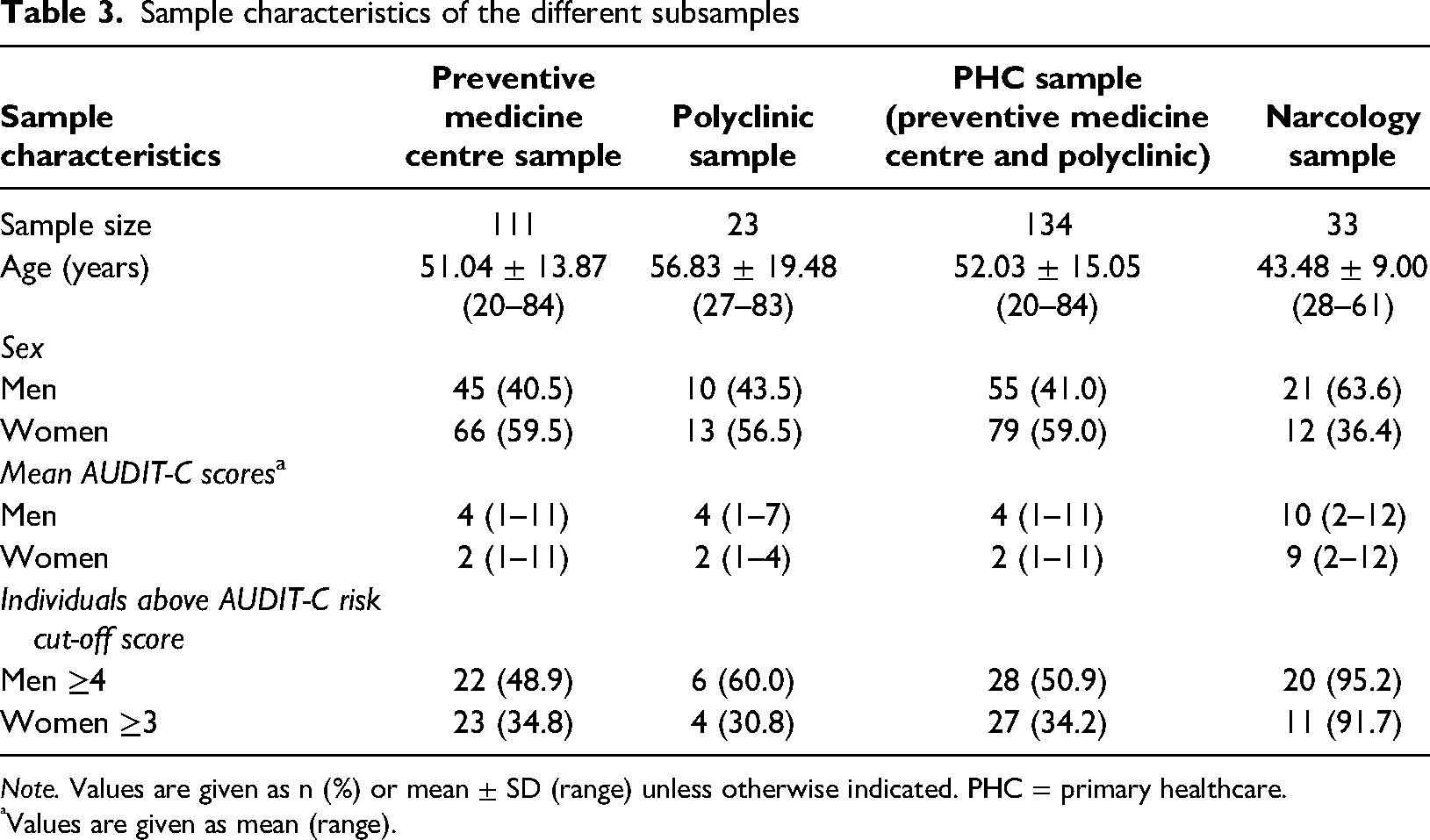
Note. Values are given as n (%) or mean ± SD (range) unless otherwise indicated. PHC = primary healthcare.
Values are given as mean (range).
The PHC sample that comprised patients recruited in the Preventive Medicine Center and in the city polyclinic included a total of 134 patients, the majority of whom were women (59.0%; mean age = 52 years). The mean AUDIT-C score in women was 2 (range = 1–11), and 4 in men (range = 1–11). When looking at the proportion of those individuals who exceeded the international risk cut-offs for the AUDIT-C (≥3 for women and ≥4 for men), then the results suggest that more than one-third (34.2%) of the screened women and about half (50.9%) of the screened men exceeded this risk threshold. A closer look at the age and sex distribution of the sample (Table 4) reveals that the majority of surveyed women were aged 45–59 years, while the majority of surveyed men were aged 60–74 years. Unsurprisingly, the youngest and oldest age groups for both sexes had the lowest number of participants. The total number of screened participants who exceeded the AUDIT-C risk thresholds is too low to calculate any meaningful proportions by age group, but it is worth noting that the highest proportions are generally observed for the youngest age groups except for women aged 30–44 years.
Age and sex distribution of the PHC sample, including the total number and proportion of individuals above AUDIT-C risk cut-off score (≥3 for women and ≥4 for men).
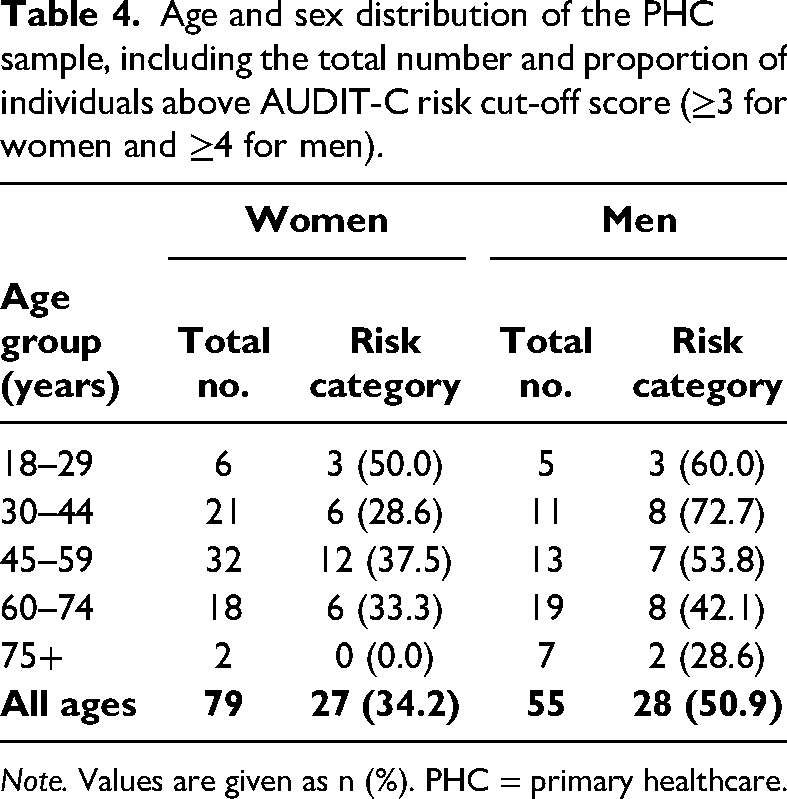
Note. Values are given as n (%). PHC = primary healthcare.
The narcology sample that comprised patients from two narcological facilities, included a total of 33 patients, the majority of whom were men (63.6%; mean age = 43 years). The mean AUDIT-C score in women was 9 (range = 2–12) and 10 in men (range = 2–12). Almost all men (95.2%) and the vast majority of women (91.7%) exceeded the international AUDIT-C risk thresholds.
It was found that, compared to PHC patients, narcology patients had greater difficulty understanding the concepts of a “typical drinking day” as it was used in the second test item and the concept of an “occasion of drinking” as it was used in the third item on heavy episodic drinking. They also struggled to find a suitable frame in which to recall their drinking behaviours over the past 12 months as various patients reported prolonged periods of zapoi (usually defined as heavy use and withdrawing from social life for at least two days), followed by periods of abstention, which is typical drinking behaviour for this clinical population (Bunova et al., 2021; Saburova et al., 2011; Tomkins et al., 2007).
Most of our discussions in all patient groups revolved around the topic of alcohol consumption in general, which elicited emotional reactions. Participants often shared personal beliefs, stories and experiences regarding alcohol consumption, often expressing how alcohol is an integral part of celebrations and leisure activities. The discussions in the expert panel have also centred mainly on the issue of Russia-specific drinking patterns, the difficulty of adequately capturing them with the AUDIT, the specific differences in translation and their meaning, and how alcohol is engrained in many aspects of daily life in Russia.
Discussion
The present study describes the construction of the RUS-AUDIT as based on an iterative process of expert discussion, field testing and revisions. It is provides a final version of the RUS-AUDIT, which was developed as a paper-and-pencil interview version for the main validation study of the instrument in the Russian Federation (Neufeld et al., 2021b; Rehm et al., 2020). The documented steps can be useful for conducting other country adaptations of the AUDIT. Although the AUDIT has been translated into more than 40 languages and research on its properties has increased considerably, especially in providing new insights for its use in low- and middle-income countries, the translations were rarely found to be adapted to suit cultural or national conditions and very few validation studies of the AUDIT in different settings exist (Babor & Robaina, 2016; Lange et al., 2019; Nadkarni et al., 2019).
The present study has some limitations. First, the sampling method used was a convenience sampling, approaching not only all patients that were present in the respective institutions at the time of the interviews, but also recruiting some PHC patients of an already ongoing nutrition study, thereby introducing potential bias. Thus, our sample is not representative of Russian PHC and narcology patients. Second, the usual bias of self-reported data cannot be excluded, such as recall bias or social desirability bias, which in this case is aggravated by the fact that substance use is being assessed. Moreover, PHC patients in Russian facilities tend to underreport their alcohol intake because they fear certain repercussions and the involvement of the narcology services and possible narcological registration once they are identified as being above a certain risk threshold. Following current regulations, narcological registration excludes patients from holding certain professional designations or positions and has been documented as being associated with the experience of stigma, which explains why patients in PHC facilities might not wish to fully disclose their alcohol use, let alone any associated problems (Bunova et al., 2017; World Health Organization, 2019a). Finally, the comparison of the AUDIT-C scores across the different samples is a heuristic that should be interpreted with great caution because the scores are based on different versions of the test items—specifically the second test item on typical drinking occasions—which have evolved over the course of the study.
Nevertheless, the provided comparison of the AUDIT-C scores and the resulting finding that approximately one-third of women and half of men who are current drinkers and patients of Russian PHC facilities drink at a level that poses potential risks to their health is in line with previous findings. For instance, previous studies using the full 10-item version of the AUDIT found that 76% of male and 47% of female dermatology patients and 55% of male and 45% of female general practice patients exceeded the non-sex-specific cut-off score of ≥8 for hazardous drinking, although these studies have been using slightly different translations and modifications of the tool (Plavinski et al., 2012a). The same research group found even higher proportions in male patients of dermatologists (57%) and general practitioners (80%) when using the same AUDIT version and the same cut-off score of ≥8 (Barinova et al., 2013b). The same study found that every 10th patient who presented to a general practitioner potentially had an AUD because they had an AUDIT score of ≥20.
From a health organisation perspective, the outlined proportions, including the discussed proportions of the present study, seem too high to inform brief intervention procedures and allocate resources in a balanced and sensible manner as they would imply that more than half of all patients who have consumed alcohol in the past year would require a brief intervention and some follow-up procedures. In the current circumstances, this would mean a too high strain on the health system and would likely result in unnecessary interventions, including potential referrals to the narcology system, which are associated with stigma, as described above. The large-scale validation study that was informed by the results of the present article and that has been recently completed in over 20 PHC facilities from eight different Russian regions has now established new Russia-specific thresholds for the different risk levels, which can inform appropriate interventions within the health system, but specific implementation studies and evaluation of the newly proposed RUS-AUDIT are yet to be carried out (Neufeld et al., 2021b; World Health Organization, 2021a).
The main results of the present study confirm the overall importance of the AUDIT-C items and again highlight the already-documented difficulties in operating with the SD concept as part of the second test item. However, the results also demonstrate that these difficulties can be overcome by using assistive devices, such as the show card, developed for this study. The show card was found to be a compromise between the many approaches and ideas documented in the different pre-studies (Bunova et al., 2021; Neufeld et al., 2021a). As described above, one of the advantages of this show card is that while it still uses the concept of an SD, this does not affect the results of screening procedure. Since this unknown term appears rather technical to patients and not as familiar as the typically used measurement units, such as glasses, bottles, millilitres and grams, it may provide an opening for a more honest conversation with healthcare workers because it may be less associated with stigma. The conversation about what a SD is could also serve as an entry point to a brief intervention, and provide an educational function by raising the awareness in drinkers that the consumption of low-alcohol beverages can also lead to a high SD count if consumed in large enough volumes—a situation that was emphasised by PHC professionals during a pre-study (Bunova et al., 2021). Besides this health literacy function, the show card is also aligned with changing drinking patterns in the Russian Federation, where overall drinking levels have declined and drinking occasions have shifted from the home environment to public settings, such as restaurants and bars, and where counting one's drinks is now much more commonplace.
The problems encountered in some narcology patients when responding to the RUS-AUDIT were not surprising, considering that this test was developed as a screening tool and not as a diagnostic instrument. It is therefore to be expected that it will behave differently when used in clinical populations, especially narcology patients in Russia. At the same time, the results of this pilot phase corroborated the findings of the qualitative interviews and the systematic reviews (Bunova et al., 2021; Neufeld et al., 2021a), suggesting that particular attention needs to be paid to heavy drinking occasions and unique Russian drinking patterns. Specific items on zapoi and unrecorded alcohol use were included in the RUS-AUDIT validation study to explore whether they might contribute to the psychometric properties of the tool.
As the results of the final validation study indicated, the RUS-AUDIT, as constructed using the described multi-step exercise, performed well as a scale, showing high internal consistency and yielding a Cronbach alpha score of >0.8 (Neufeld et al., 2021b; World Health Organization, 2021a). The validated version accurately predicted AUDs and other outcomes, such as hazardous drinking (area under the curve >75%). The high specificity and sensitivity of the tool has practical implications for the Russian healthcare system in a sense that brief interventions and referral to the narcology will be offered only to those who are in need, thereby relieving the narcology system from the burden of patients who are not likely to meet the diagnostic criteria of AUDs and who should not fall under narcological monitoring and its restrictions. The newly added test items on heavy episodic drinking, which were constructed using the described adaptation procedures, correlated highly with the full scale, but replacing the initial items or adding the new items to the full test did not yield any significant improvement to the test's psychometric properties. Thus, the test version as presented in Table 2 constitutes the final version of the RUS-AUDIT, which will be implemented as a standard screening tool into the Russian healthcare system. Implementation of this newly validated tool and brief interventions in general is a promising approach to identify the still relatively large population of heavy drinkers in Russia, who do not yet meet the criteria of AUDs, but who would greatly benefit from alcohol reduction and cessation to reduce their risks of non-communicable diseases, first and foremost cardiovascular diseases (World Health Organization, 2019b).
Conclusion
The RUS-AUDIT, as developed in the course of the study, was found to be a feasible and comprehensible instrument by interviewers and patients alike. Although some issues were documented on its use with narcology patients, these were expected due to the fact that this is a screening tool and not a diagnostic instrument and that narcology patients are not the target group for the AUDIT. Although the proportions of PHC patients presented here who exceeded the risk level as per the first three consumption items of the RUS-AUDIT must be interpreted with extreme caution since they are based on different versions of the instrument, they are nevertheless in line with previous findings that the different Russian version of the AUDIT classifies a large number of Russian PHC patients as at-risk drinkers (Neufeld et al., 2021a). The discussed findings can be used in the further implementation of SBI programmes in PHC and other settings and the described procedures can be used as an algorithm for further adaptation and validation studies of the AUDIT in other countries with distinct drinking patterns.
Supplemental Material
sj-docx-1-nad-10.1177_14550725231183236 - Supplemental material for Translating and adapting the Alcohol Use Disorders Identification Test (AUDIT) for use in the Russian Federation: A multicentre pilot study to inform validation procedures
Supplemental material, sj-docx-1-nad-10.1177_14550725231183236 for Translating and adapting the Alcohol Use Disorders Identification Test (AUDIT) for use in the Russian Federation: A multicentre pilot study to inform validation procedures by Maria Neufeld, Anna Bunova, Eugenia Fadeeva, Alexey Nadezhdin, Elena Tetenova, Konstantin Vyshinsky, Carina Ferreira-Borges, Elena Yurasova, Andrey Allenov and Boris Gornyi in Nordic Studies on Alcohol and Drugs
Supplemental Material
sj-docx-2-nad-10.1177_14550725231183236 - Supplemental material for Translating and adapting the Alcohol Use Disorders Identification Test (AUDIT) for use in the Russian Federation: A multicentre pilot study to inform validation procedures
Supplemental material, sj-docx-2-nad-10.1177_14550725231183236 for Translating and adapting the Alcohol Use Disorders Identification Test (AUDIT) for use in the Russian Federation: A multicentre pilot study to inform validation procedures by Maria Neufeld, Anna Bunova, Eugenia Fadeeva, Alexey Nadezhdin, Elena Tetenova, Konstantin Vyshinsky, Carina Ferreira-Borges, Elena Yurasova, Andrey Allenov and Boris Gornyi in Nordic Studies on Alcohol and Drugs
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported through a grant of the Russian Government to the WHO European Office for the Prevention and Control of Noncommunicable Diseases. This funding source had no role in the design of the study, its execution, data collection and analyses, interpretation of the data, writing of the manuscript or decision to submit it.
Ethics approval and informed consent
The interviews that were conducted with patients as part of this pilot study were considered to be part of routine care by participating primary healthcare institutions since they were aimed at quality improvement of screening procedures without collecting any identifying patient information. The participating institutions obtained formal approval from local authorities and the overall RUS-AUDIT study was approved by the Ethics Committee of the V. Serbsky Federal Medical Research Centre of Psychiatry and Narcology of the Ministry of Health of the Russian Federation, Protocol No 25/6 from 21 January 2019. Informed consent was obtained verbally from all participants involved in the study, which is in line with local regulations. All parts of the study were fully compliant with ethical principles, including the provisions of the World Medical Association Declaration of Helsinki, as amended by the 59th General Assembly, Seoul, Republic of Korea.
Data availability
The data that support the findings of this study are available from the corresponding author, upon reasonable request.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
