Abstract
Cellular adaptation to external signals is essential for biological functions, and it is an important field of interest in systems biology. This study examines the impact of cooperativity on the adaptation response of the Incoherent Feedforward Loop (IFFL) network motif to various signal profiles. Through comprehensive simulations, we studied how the IFFL motif responds to constant and pulse-type signals under varying levels of cooperativity. The results of our study demonstrate that positive cooperativity generally enhances the system’s ability to adapt to different signal profiles. Nevertheless, given specific signal profiles, higher levels of cooperativity may decrease the system’s adaptability. On the other hand, the adaptive response breaks down for negative cooperativity. For constant signals, increased positive cooperativity leads to a response with higher amplitude, and it accelerates the response time but delays the return time required to settle back down to the pre-stimulus state. Upon signal cessation, high positive cooperativity not only slows the system’s response and return times but, in some cases, can lead to a complete temporary halt in response. For the pulse-like signal, cooperativity increases the maximum amplitude of the oscillatory response. These insights highlight the delicate balance between cooperativity and signal profile in cellular adaptation mechanisms involving the IFFL network motif.
Introduction
Cells are subjected to many external stimuli and have developed intricate adaption mechanisms to maintain a state of equilibrium. This adaptive capacity is more than a mere cellular response; it is essential for the survival and efficient functioning of cells. It enables cells to precisely react to the continuously shifting external stimuli (Bleris et al., 2011; Ma et al., 2009). This complex regulatory relationship between stimulus and response is partly governed by small, evolutionarily conserved interaction networks, referred to as network motifs. The network motifs that are capable of displaying adaptive dynamics include negative feedback and incoherent feedforward loops (IFFLs). These motifs, characterized by unique structural designs, have a key role in the complex dynamics of adaptation. They determine how cells perceive and respond to different environmental signals (Alon, 2007; Mangan & Alon, 2003).
The IFFL motif, particularly its Type I form, is crucial in gene regulatory networks for processing environmental signals through two opposing regulatory pathways on a specific protein. It initially triggers an increase in the abundance of a response protein via an activatory pathway. Then, an inhibitory pathway counteracts this effect, leading to a precise regulation of the protein’s level. This results in an initial rapid increase in the protein levels, followed by a gradual return to its basal level, a process known as perfect adaptation. It has been shown that an IFFL motif with adaptive dynamics must have at least three proteins (Ma et al., 2009).
The proteins regulated by IFFL motif are involved in a wide range of biological functions, including specific cellular tasks and interacting with other transcription factors (Mangan et al., 2006; Mangan & Alon, 2003; Novák & Tyson, 2008). The IFFL adaptive dynamics are observed in biological processes, such as light detection in plants, homeostatic adaptation, neuronal regeneration, and eukaryotic chemotaxis, highlighting the importance of the motif’s role in different cellular contexts (Frick et al., 2017; Lee et al., 2014; MacGillavry et al., 2009; Shi & Iglesias, 2013; Takeda et al., 2012; Tendler et al., 2018). Besides its widespread occurrence in many biological systems, the IFFL motif is a major focus in synthetic biology research. Biologists have constructed the IFFLs in many systems to carry out a variety of tasks, including mitigation of resource competition, and signal filtering (Bleris et al., 2011; Frei & Khammash, 2021; Jones et al., 2019). To effectively utilize this network in the regulation of biological systems, a better understanding of its mechanics is crucial.
Cooperativity among proteins, such as transcription factors, is vital in controlling biological processes. Cooperative molecule binding is identified as a key mechanism for ultra-sensitive responses in biological systems (Goldbeter & Koshland Jr, 1984). Within transcriptional networks, cooperativity can either enhance or diminish the reaction to external stimuli, highlighting its dual role in biological regulation (Berg et al., 2004; Forsén & Linse, 1995). Furthermore, cooperativity tends to increase in cascading systems, which are prevalent in developmental networks (Blake et al., 2003). Such cascading effects contribute significantly to the complex behavior of these networks (Chickarmane et al., 2006; Demarez et al., 2018; Pimanda & Göttgens, 2010; Yoshida et al., 2020). The incoherent feedforward loop can still exist with cascading proteins (Ferrell, 2016). Specifically, in IFFLs, cooperativity significantly impacts these motifs’ signal-processing abilities, leading to significant changes in the system’s adaptability and response characteristics (Benary et al., 2013; Sun et al., 2022).
Positive cooperativity is commonly observed in biological systems, where the binding of one molecule increases the likelihood of subsequent molecule’s binding. However, negative cooperativity also plays a significant role, where the binding of one molecule decreases the affinity for the next molecule to bind (Koshland Jr, 1996). Specifically, negative cooperativity is instrumental in the application of Weber’s Law within transcription systems (Ferrell, 2009). Weber’s Law describes how the perception of a change in a stimulus (e.g., light intensity) is proportional to the magnitude of the stimulus itself, suggesting that organisms are sensitive to relative changes rather than absolute ones. This principle is crucial for fold-change detection in biological systems, a process that requires perfect adaptation to accurately measure relative changes in stimulus intensity (Shoval et al., 2010).
Our study investigates the role of cooperativity within the Type-I IFFL motif and focuses on how cooperativity influences the motif’s capacity for cellular dynamics and adaptation. To unravel the impact of varying levels of cooperativity on IFFL dynamics, we employ computational models and analyze these effects across different signal profiles. We utilize adaptation metrics to quantify response dynamics such as response level, response time, return time, and adaptation error to comprehensively study and gain a better understanding of the adaptive dynamics and its regulation in the IFFL motif under different cooperativity conditions (Frei & Khammash, 2021; Shi et al., 2017; Yildirim, 2024). The results of this study are expected to provide a novel understanding regarding the dynamic control of proteins, a critical component in cellular adaptation and function.
The Mathematical Modeling and Analysis of the IFFL Network
This section presents the mathematical model for the Type-I IFFL motif and derives the necessary and sufficient conditions for the adaptive dynamics. An IFFL network with fewer than three proteins is unable to generate adaptive dynamics (Ma et al., 2009; Shi et al., 2017). A cartoon showing the interactions of three proteins, A, B, and C, in the Type-I IFFL network is displayed in Figure 1(a). Here we name protein A as “a signal protein” on which environmental input signal S increases its abundance. The signal protein A positively regulates two proteins downstream, which are “intermediate protein” B and “response protein” C. Protein B also negatively regulates the response protein C.

(a): A cartoon showing IFFL network involving three proteins, which is capable of producing adaptive dynamics. In this cartoon, environmental signal S increases the abundance of the signal protein A, which positively interacts with both intermediate protein B and response protein C, while the intermediate protein B negatively regulates the abundance of the response protein C. (b): A plot illustrating the response metrics
The model equation governing IFFL motif is given by Equations (1) to (3) (Shi et al., 2017). Equation (1) describes the dynamics of the signal protein A, where 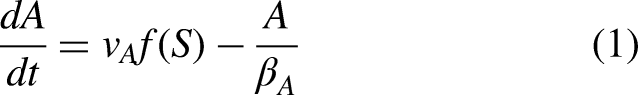

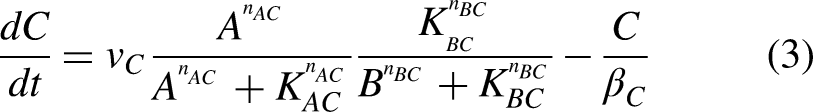
Steady State Analysis and Adaptive Dynamics
To carry out analytical work and study model dynamics, the model is written in dimensionless form to reduce the number of parameters while retaining its dynamic properties. By defining new variables as 
The adaptive dynamic response requires the steady states of the response protein 


When 
These state levels could further be simplified. For some large 
Local Stability Analysis of the Adaptive Model
To study the local dynamics of the IFFL model in response to a small increase in the input signal S while the system is at resting state, the model equations are linearized around the steady state to obtain:




Numerical Simulations
We conducted a series of numerical simulations to further study sensitivity of the adaptive dynamics of the Type I IFFL model to the changes in the cooperativity parameters
Different metrics were used to quantify the IFFL’s adaptation response to different signal profiles. For analyzing the response to a signal when it is activated, three specific metrics were employed: (i)
To explore the adaptive dynamics after a signal is turned off, we modify these metrics slightly: (i)
We also utilized the amplitude metric to analyze pulse-type signals. Amplitude is defined as the difference between the maximum and minimum responses, expressed as
Section “Adaptation Dynamics Under Varying Cooperativity Levels and Constant Signal Inputs” examines how the Type I IFFL network adapts under conditions of both positive and negative cooperativity using the adaptation error metric. Subsequent sections then concentrate solely on the effects of positive cooperativity. Section “System Response to Constant Signal Inputs” focuses on the system’s response to constant weak and strong signals and looks at the effect of different levels of cooperativity on the adaptation metrics. Section “System Response to Varying Signal Inputs” presents a similar analysis of the system to the signals of changing strengths as the cooperativity levels vary. Section “Adaptation Dynamics for Post-signal Deactivation Under Constant Signal Inputs” examines the system dynamics after the signal is turned off, investigating how the cooperativity regulates the adaptive system’s behavior once the input signal is ceased. Section “System Response to Varying Signal Inputs Post Signal Deactivation” extends this analysis to the influence of signals of differing strengths after the signal has been turned off as signal cooperativity levels vary. Finally, Section “Adaptation Dynamics Under Repetitive Pulse Type Signals” analyzes this adaptive system’s response to pulse-type signals. We assess how the system processes varying pulse strengths and profiles under different cooperativity levels.
Adaptation Dynamics Under Varying Cooperativity Levels and Constant Signal Inputs
To elucidate the model’s response dynamics to constant external stimuli under varying cooperativity levels, we conducted a detailed examination of system behavior under three distinct signal strengths: (i) a weak signal of
As illustrated in Figure 2, our findings show a discernible difference in the response dynamics to the weak (Figure 2(a)), medium (Figure 2(b)), and strong (Figure 2(c)) signals. The system demonstrates more refined adaptability to the weaker signal compared to the stronger signal. Furthermore, it displays a better adaptive response under positive cooperativity as opposed to negative cooperativity. Our results show that negative cooperativity eliminates the system’s ability to adapt, especially as the signal becomes stronger (Figure 2(a) to (d)). Due to negative cooperativity at large hindering the system’s ability to adapt, we decided to base our further analysis on positive cooperativity. The time series plot in ((Figure 2(d))) shows three representative dynamics when

Heat maps illustrating the system’s dynamics and adaptation for different cooperativity levels of
System Response to Constant Signal Inputs
To elucidate the model’s response dynamics to constant external input signals, we conducted a detailed examination of system dynamics under two distinct signal strengths (i) a weak signal, which corresponds to a 2-fold increase in the basal level of the signal protein
As illustrated in Figure 3, our findings show a noticeable difference in the system’s response dynamics to the weak and strong input signals. The system demonstrates better adaptability to the weaker signal than the stronger signal. For example, adaptation fails when

Heat maps illustrating the system’s response to different cooperativity levels (
The adaptation metrics exhibit sensitivity to the variations in the Hill coefficients. Increases in either
System Response to Varying Signal Inputs
To study how the adaptation dynamics change under varying signal levels in our system, we have simulated the model under a broad spectrum of signal strengths ranging from

Heat maps showing changes in
Our simulations unveiled a critical relationship between the model parameters
Adaptation Dynamics for Post-signal Deactivation Under Constant Signal Inputs
To study adaptation dynamics for post-signal deactivation, we sustained the signal for an extended period,
Similar to prior analyses, we observed signal-dependent changes in response metrics (Figure 5). Specifically, the system exhibited a reduced minimum response

Heat maps illustrating the system’s response to different cooperativity levels
System Response to Varying Signal Inputs Post Signal Deactivation
We extended our analysis to examine the system’s adaptive dynamics after signal termination, spanning a comprehensive range of signal strengths and cooperativity levels. Similar to the previous section, we sustained the signal for an extended period,
Our results delineated a discernible pattern: as signal strength and cooperativity coefficients increase, there is an observable decrease in the minimum response
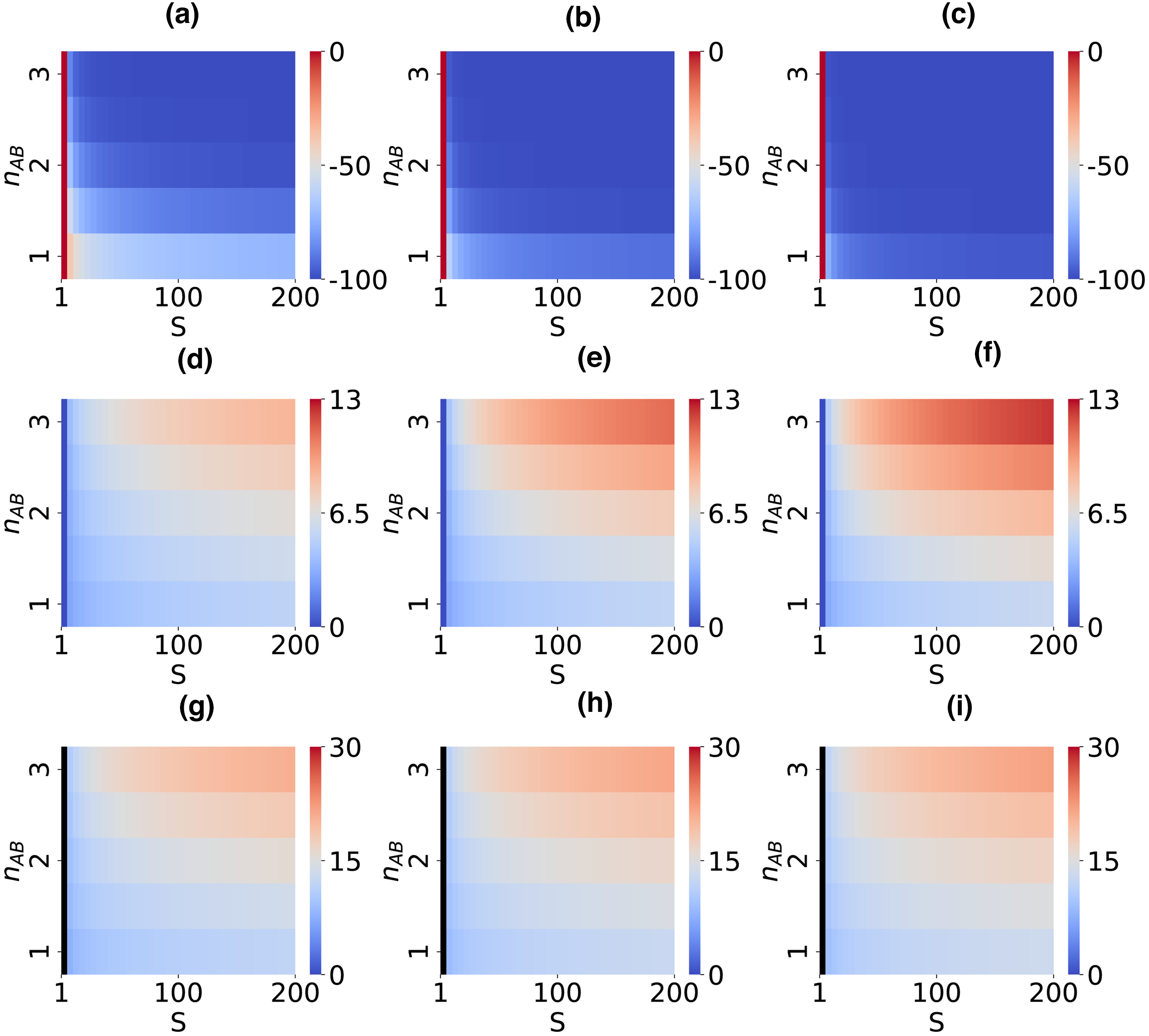
Heat maps showing changes in
Adaptation Dynamics Under Repetitive Pulse Type Signals
Here, we concentrated on evaluating the amplitude of the response to discrete signal pulses across a gradient of cooperative interaction strengths for weak (

Heat maps showing response amplitude changes to pulse type signals as the cooperativity levels (
It was observed that longer periods of signal activation led to significantly higher system responses compared to shorter periods of signal activation. This trend was consistent across both weak and strong signal strengths. Interestingly, the response dynamics varied with signal strength. The most substantial responses were elicited for weaker signals when both the activation and deactivation periods were prolonged. Conversely, stronger signals achieved the response with maximum amplitude when the activation period was brief, followed by an extended deactivation phase. This phenomenon underscores the intricate relationship between signal duration, system adaptability, and cooperative interactions.
Discussion
In this study, we have investigated the role of cooperativity and signal profiles in the adaptation dynamics of transcriptional type I IFFLs. The adaptation dynamics are assessed through metrics such as maximum/minimum response level, response, and return times as cooperativity level and signal profile are varied. Our analysis reveals that the system’s adaptability and response dynamics strongly depend on these parameters. Specifically, the Type I IFFL motif exhibits better adaptation under conditions of positive cooperativity compared to negative cooperativity. Our analysis of positive cooperativity shows that increased cooperativity and signal strength led to higher maximum response levels, shorter response time
It has been shown that positive cooperativity could prevent the system’s adaptability in IFFLs for enzymatic reactions (Ma et al., 2009). In another study, it was suggested that adaptation in transcriptional IFFLs can be attained under specific cooperativity conditions (Shi et al., 2017). However, we extend these findings by showing that altering both signal and cooperativity levels simultaneously can disrupt the IFFL’s adaptive capability beyond known parameter regimes. By simultaneously altering signal and cooperativity levels beyond the parameter regimes considered in Shi et al. (2017), we observed disruptions in the IFFL’s adaptive capability. This discrepancy underscores the nuanced influence of cooperativity on network dynamics, particularly in challenging the previously established prerequisite of
Building upon the foundational work of Mangan and Alon (2003), our study extends the understanding of type I IFFL response dynamics. This study suggests that the response time
We have shown that cooperativity alters the function of the IFFL. One of the other major functions of the IFFL is fold change detection in which the behavior of the function depends on the relative change in the signal rather than the overall level of the signal (Goentoro et al., 2009). Perfect adaptation is a key attribute of fold change detection in the IFFL (Shoval et al., 2010). In our simulations, we have shown that with varying cooperativity, the IFFL may not always adapt, especially with stronger signals. Thus, our results suggest that high levels of negative and positive cooperativity can impair the fold change detection capability of the IFFL motif.
Another function of the IFFL motif is to act as a high-pass filter, allowing high-frequency signals to pass through (De Ronde et al., 2012). However, our findings reveal that the response level of the IFFL is influenced not only by the signal’s frequency but also by its duration and the level of cooperativity. Specifically, short pulses elicited the weakest response for weak signals and a slightly more robust response for strong signals. Conversely, longer-duration signals at lower frequencies resulted in the strongest response for weak signals and a comparably high response for strong signals, indicating that signal strength remains robust despite less frequent pulses. This suggests a new perspective on the high-pass filtering capability of the IFFL, showing that signal duration plays a critical role in the system’s response, beyond just signal frequency. This nuanced view of IFFL signal processing capabilities, particularly in handling short pulses and longer signals, adds depth to our understanding of network motif functionalities.
IFFL systems are shown to be widely unaffected by retroactivity (Wang et al., 2020). Retroactivity refers to the phenomenon where downstream binding sites alter the dynamics of an upstream system by sequestering transcription factors, making them unavailable for other molecular activities such as degradation, protein-protein interactions, or regulation of other genes (Wang et al., 2020). Wang and Belta (2019) analyzed the IFFL in the context of a pulse generator and response accelerator and also performed an analysis of cooperativity. They fluctuated the value of
In synthetic biology, it has been of interest to make a more precise adaptive device (Tyson et al., 2003). There is an interest in fine-tuning the IFFL’s adaptive ability. One study performed by Hong et al. determined that adjusting the binding affinity of the genes would alter the system’s ability to adapt (Hong et al., 2018), and another study by Reeves suggested that incorporating negative feedback to the IFFL would help facilitate adaptation (Reeves, 2019). Our results provide a complementary perspective by showing that cooperativity and signal characteristics can be crucial in fine-tuning the IFFL’s adaptive responses, suggesting a potential avenue for engineering more robust adaptive motifs. This comprehensive understanding enables the fine-tuning of networks for enhanced functionality in biological systems. Even though our study involves type I IFFLs, cooperative effects and signal properties could also have an impact on other networks, like enzymatic IFFL networks and multi-IFFLs. Future studies could extend this research to explore cooperativity’s influence in broader contexts, such as enzymatic IFFLs and multi-IFFL systems, further enhancing our understanding of network behavior. These insights pave the way for future research to explore the effects of cooperativity and signal characteristics across a broader range of networks, aiming to deepen our understanding of their dynamic interactions. In essence, this study adds to the knowledge of network motifs’ complex behaviors, underscoring cooperativity and signal characteristics’ vital roles and setting the stage for further exploration into the mechanisms underlying cellular information processing.
Footnotes
Funding
This research was supported by a generous grant from the Colgate University Research Council (Grant #824586) to Ahmet Ay.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
