Abstract
Background
Bringing features of nature indoors can positively influence indicators of human stress. Since wood is a natural material, it may produce similar benefits. The objective of the review was to (1) examine the influence of visual (real or virtual) contact with either real or imitated indoor wooden surfaces on certain stress indicators, that is affective, physiological or cognitive performance outcomes (compared to non-wooden surfaces) and to (2) assess the methodological quality of the reviewed studies.
Method
We conducted a systematic literature search for English articles on Scopus, PubMed, Web of Science, Cochrane Central and Google Scholar on 6 August 2019. The results of the eligible studies were synthesized narratively in light of the identified methodological shortcomings.
Results
We reviewed nine studies with 386 participants in total. Studies with longer exposure times to wood generally observed improved affective states and decreased physiological arousal in wooden settings, but the results are not entirely clear-cut. We discuss several methodological issues uncovered in the reviewed studies and provide guidelines for future robust research.
Conclusions
Current evidence suggests that visual wood exposure may improve certain indicators of human stress, but additional research is needed to confirm the existing findings.
Introduction
Currently, 55% of the world’s population lives in urban areas; by 2050, this percentage is expected to increase to 68%. 1 As the population further shifts to urban environments, the effects of urban living must be carefully considered. People living in cities have an increased risk of suffering from mental disorders, 2 which could be related to their heightened sensitivity to stress 3 and repeated exposure to environmental (e.g. noise), social (e.g. greater social disparities) and behavioural stressors (e.g. increased competition).4,5 Constantly adapting to these stressors may, in turn, lead to attentional fatigue, which can increase stress levels even further. 6 The experience of stress does not have to depend on adverse external factors. Stress can be induced by neutral or seemingly harmless occurrences that are cognitively appraised as negative 7 and the stress response can be sustained with persistent cognitive representation of stress-related content. 8 Accordingly, stress can be widespread even in the absence of obvious stress-inducing occurrences.
While acute – short and infrequent – stress responses typically do not represent a risk, chronic – persistent and long-term – stress responses may damage health by altering nervous, cardiovascular, endocrine and immune systems.9,10 Chronic stress can directly or indirectly lead to debilitating mental illnesses, including anxiety and depression and other threatening conditions, such as cardiovascular disease and perhaps even cancer.9,11,12 Reduced attentional capacity presents additional issues beyond exacerbating stress, including impaired problem-solving capabilities and inhibition of inappropriate behaviours.13–15
Various interventions are helpful in reducing stress and the subsequent harm stress may cause; for instance, physical exercise, 16 meditation 17 and cognitive-behavioural therapy 4 have received wide attention. Unfortunately, these interventions call for energy, time and commitment, and may not be attractive for stressed and fatigued individuals. Complementing these active approaches with passive interventions, introduced to places where people spend most of their time, could bring stress reduction to more people.
Exposure to nature or natural elements could be such an intervention, as it may lead to enhanced affective states (referring to subjective experience of feelings, emotions and moods), reduced physiological arousal and improved attentional capacities.18–20 The restorative qualities of nature are usually interpreted through either stress reduction theory (SRT), 21 attention restoration theory (ART)6,22 or both.
According to SRT, stress is the culprit which leaves the individual in need of psychophysiological restoration. The theory proposes that contact with nature (or natural elements) results in favourable changes in (physiological) arousal and affective states. 21
ART, on the other hand, is centred on the exhaustion of attentional resources. It proposes that we often operate on voluntary (or ‘directed’) effortful attention which is susceptible to depletion and must be periodically allowed to rest by activating involuntary attention, which often occurs in natural environments. 6
The theoretical underpinnings of SRT and ART need further development.23–29 Among other issues, neither theory convincingly unifies affective and physiological states (advocated by SRT) and attention (advocated by ART), even though these constructs overlap substantially and rarely operate independently of each other.27,28 Additional issues arise when ART and SRT are recruited to account for human responses to single elements of nature (e.g. to plants), considering that both theories primarily explain human response to rich natural environments.
Despite the theoretical shortcomings, a growing body of evidence shows that being in nature improves several indicators of human stress. However, urban dwellers spend most of their time indoors 30 and might have limited access to nature, which encourages bringing nature to interior spaces. The presence of nature indoors can be increased simply by introducing photos of landscapes, potted plants or the scent of fresh flowers. Such interventions can bring nature to nearly any indoor environment, regardless of its pre-existing characteristics. Importantly, similar positive effects on human well-being that are observed in outdoor natural environments are detected when nature is brought into indoor settings. 18
Wood is of particular interest in bringing nature in interior spaces, because it is a versatile and renewable natural material that can be used structurally, decoratively and for other functional elements in buildings. 31 It is perceived as more natural than other common building materials32,33; correspondingly, interiors containing more wood are rated as more natural than the interiors with lower wood coverage.34–37 As such, wood allows us to embed naturalness in the foundations of the built environment while supporting sustainable construction practices. 38 However, does the presence of wood in indoor spaces lead to favourable physiological, affective and cognitive performance outcomes?
Few experimental studies addressed this question and those that attempted have employed diverse methodological approaches. Our objectives were to review the existing randomized controlled trials in order (a) to assess the effects of visual contact with wooden surfaces in the indoor environment on at least one physiological, affective or cognitive performance outcome (compared to visual contact with any other surface) in the entire population; (b) to identify positive and negative aspects of study designs and (c) to develop recommendations for future robust studies. Compared to the existing reviews,39,40 this review is the first to address the methodological issues in-depth and use the resulting insights to critically evaluate the reviewed research. In addition, this article examines several recent studies that were not included in the previous reviews.
Method
The process of conducting and reporting this review followed the general principles recommended by the Cochrane guidelines for systematic reviews of interventions 41 and the PRISMA statement for reporting systematic reviews. 42 Cochrane guidelines were developed to provide a consistent and reliable framework for systematic, informed and explicit reviews. The guidelines advise all stages of review preparation, from preparing questions and designing the initial search strategy to collecting and analysing data and drawing conclusions. The PRISMA statement encourages the process of review preparation to be fully and transparently reported, allowing readers to assess the strengths and weaknesses of the investigation. It provides reporting guidelines concerning the entire article, starting with the manuscript title and ending with conclusions.
We searched for published randomized controlled trials assessing at least one physiological, affective or cognitive performance outcome in response to visual (real or virtual, of any duration) wood exposure in indoor environments (to both real wood and wood imitations). Eligible studies had to include at least one control intervention, that is visual exposure to a non-wood material. Studies with visual wood exposure interventions that allowed participants tactile or olfactory contact with materials were not excluded. Eligible primary outcomes were any indicator of autonomic nervous system functioning for ‘physiological outcomes’; any measure capturing either core affect (i.e. simplest consciously accessible feelings, such as pleasantness), emotions or mood of participants for ‘affective outcomes’, and any measure capturing any facet of executive functions for ‘cognitive performance outcomes’. Secondary outcomes included measures of central nervous system functioning and non-affective self-report measures (e.g. fatigue, vigilance) that might provide additional insight into human responses in wooden indoor environments. Only full texts reported in English were included. We did not impose any additional restrictions related to the year of publication, publication type, study design, intervention duration, type of implemented wood or characteristics of participants.
Studies were identified by searching electronic databases (on 6 August 2019) and scanning reference lists of articles. Specifically, we searched in Scopus, PubMed, Web of Science, Cochrane Central and Google Scholar for all article titles containing the word ‘wood’ or ‘wooden’ along with any of the following expressions or their derivatives: psychology, emotion, affect, mood, physiology, arousal, human stress, stress response, attention, cognition. The search was developed and conducted by the first author and checked by the second author; a detailed search strategy is available in Table S1. The same search phrases were used in all databases. Screening and eligibility assessments were performed independently by both reviewers and disagreements were planned to be resolved by consensus. Additional articles were identified by scanning reference lists and manual searching.
To ascertain the risk for bias in individual studies, both authors screened each study with the assistance of the revised Cochrane risk of bias tool for randomized trials. The tool includes questions that capture several domains of potential bias (e.g. different aspects of trial design) and algorithms that aid in judging the risk of bias according to the answers on these questions. 43 To extract data, the lead author developed a data extraction form based on the Cochrane Consumers and Communication Review Group’s data extraction template, which is designed to help authors capture all relevant information about the included studies. 44 The author then extracted the data from the studies that were later checked by the second author. When a single study was reported in multiple reports, the data from all reports were extracted directly into one data collection form. Any disagreements were resolved by discussion between the authors. From each study, the information was extracted on (1) study design; (2) location; (3) participants (number, age, gender, sociodemographic information, inclusion and exclusion criteria); (4) intervention setting(s); (5) control setting; (6) duration of the exposure and (7) physiological, affective and cognitive performance outcomes. The primary outcome measure was the difference in any physiological, affective or cognitive performance outcome between the intervention (i.e. wooden) and control (i.e. non-wooden) for both within- and between-subject studies. Meta-analyses (or other forms of quantitative synthesis) were not conducted due to incomplete reporting of results and considerable methodological diversity across studies, including differences in measured outcomes and certain studies not controlling for olfactory stimulation. Results were summarized and synthesized narratively. Methods of the analysis and inclusion criteria were not documented in a registered protocol.
Results
Study selection
The search produced 3267 unique articles which were individually examined by title, and, when needed, by abstract and full text. A considerable number of studies was excluded due to their focus on biological (e.g. investigating physiology of wood) or mechanical wood properties (e.g. mechanical stress in wood), while some studies were excluded due to assessing responses to wood that were not relevant for this review (e.g. tactile perception of wood). The full texts of 11 articles (reporting nine studies) were assessed against eligibility criteria, and of those, all were included in the review. The detailed study selection procedure is presented in Figure 1. There were no disagreements on inclusion between the reviewers.
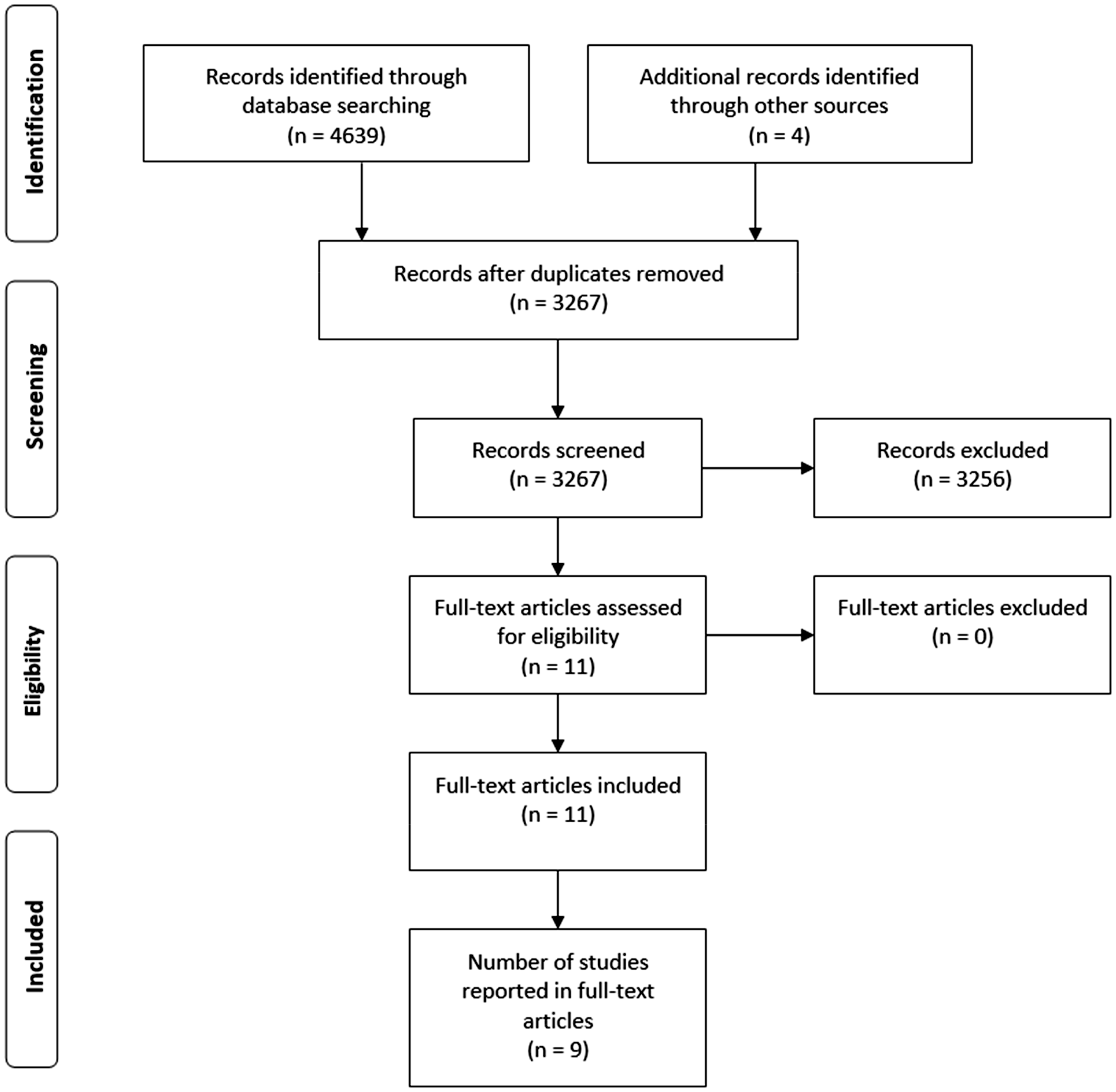
Flowchart of the study selection process.
Characteristics of included studies
Characteristics of included studies are summarized in Table 1 and presented in more detail in Table S2. With the exception of Fell’s between-subject experiment, 36 all studies employed a within-subject design. Four experiments are characterized by short exposure times to wood (90 s) and low numbers of participants (between 14 and 28),34,47–50 while the other five studies employed longer exposure times (10–75 min) and typically larger number of participants (between 12 and 119).35,36,45,46,51,52 Five studies created wooden settings of only light colour wood,35,36,45,47,48 two studies used only dark wood,34,49,50 and two studies employed both light and dark wood, either separately in different settings or combined in the same room.46,51,52 Five studies included solid wood,35,48–52 three used wood composites34,36,46 and one used images of wood. 47
Summary of the characteristics of the reviewed studies.

bs: between-subject design; RCT: randomized controlled trial; RH: relative humidity; ws: within-subject design.
Four of the reviewed studies investigated both physiological and affective responses,34,47,48,50 two studies examined a combination of physiological and cognitive performance outcomes,36,46 one study investigated physiological outcomes, 45 one study assessed affective states 35 and one study inspected all three domains – physiological arousal, affective states and cognitive performance (although the results of cognitive performance tasks will not be reviewed here, since they have not yet been reported in a peer-reviewed article).51,52
Risk of bias within studies
Risk of bias assessment based on the Cochrane revised risk of bias tool is summarized in Figure 2 and presented in detail in Table S3. Overall risk of bias was low in four studies,35,45–47 high in three studies (due to the insufficient randomization process 36 or due to reporting only a selection of results48,51,52) and presents some concerns in two studies (due to missing outcome data34,49,50).
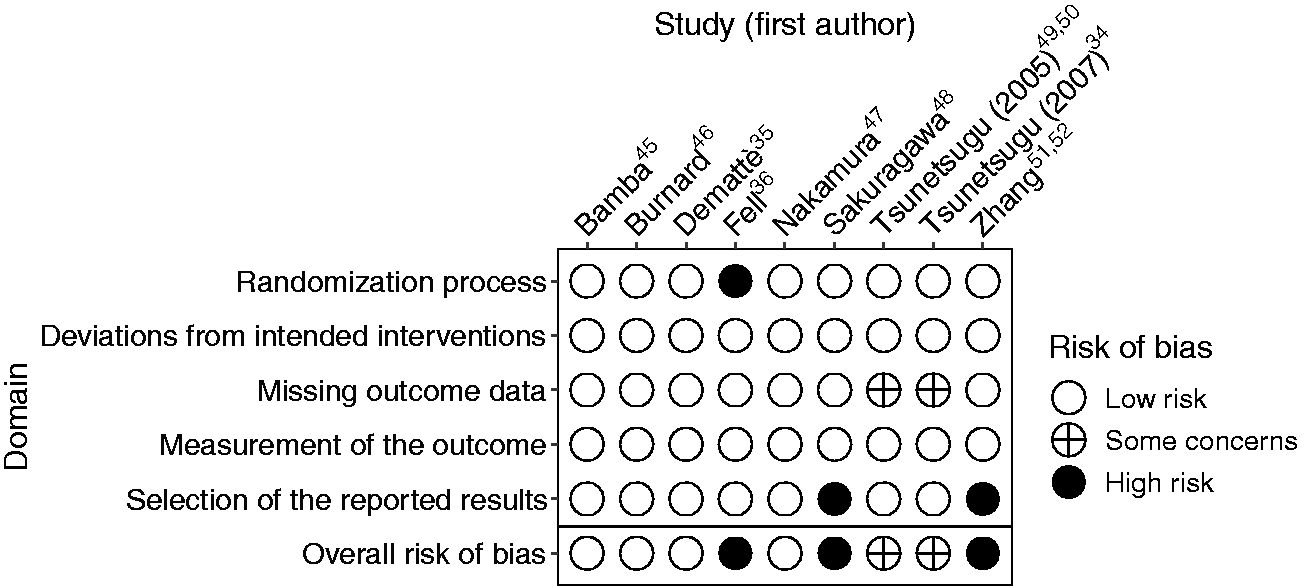
Risk of bias in the reviewed studies.
Methodological issues in the reviewed studies
Several methodological issues were identified in measuring physiological arousal, affective states and cognitive performance. Below we discuss methodological issues separately for each group of these measures.
Assessing physiological arousal
With the exception of the study conducted by Demattè et al., 35 all reviewed studies included at least one physiological measure. Although such measures seemingly provide robust and objective results, they are often difficult to interpret if not accompanied by a suitable study design. The aspects of the reviewed studies that make interpreting physiological outcomes difficult are discussed below.
Physiological measures not sufficiently corroborated
Physiological response cannot be easily interpreted on its own, especially if the physiological data are derived from few sources. It may be tempting to conclude that lower arousal levels denote lower stress response and thus a favourable outcome (and vice versa), but this is not necessarily the case. First, according to SRT, exposure to pleasant natural environments can either increase, decrease or not influence arousal, depending on the initial arousal level. 21 Second, autonomic nervous system activation corresponds to a variety of functions, including homeostasis, attention, effort and digestion. 53 Third, both positive (e.g. excitement, relaxation) and negative outcomes (e.g. nervousness, sadness) can be reflected in either increased or decreased physiological activation, depending on the context and the specific physiological measure used.54,55
Short assessment period
Three of the reviewed studies34,48–50 measured autonomic activation in each tested environment for 90 s. By offering only a short glimpse into physiological responses, such an approach further complicates the differentiation between positive and negative (affective) outcomes, which are often manifested in overlapping patterns.54,55 Additionally, a certain stimulus can produce fleeting states that may dissipate soon after the initial stimulation 54 and thus the subsequent effects, that might be a better target when assessing restoration, are not captured.
Including few physiological measures
Most of the reviewed studies included few physiological measures, which are likely to provide inconclusive results. Different autonomic arousal measures can function independently or even in opposition to each other in response to affective states.54,56 For these reasons, only one or few measures of physiological activation can fail to detect important changes in arousal. Alternatively, they may detect only a subset of differences (e.g. an increase in heart rate) while failing to detect others (e.g. an accompanying decrease in blood pressure), which may, in turn, lead to misleading results. Including a wider array of physiological measures (e.g. heart rate, heart rate variability, blood pressure, electrodermal variability, skin temperature, salivary cortisol) is essential to strengthen the study design. However, incorporating too many measures may prove intrusive and obstruct the potential restorative effects of an environment.
Not including a stress-inducing activity
Five out of nine reviewed studies did not incorporate a stress-inducing activity34,35,47–50 that can help interpret changes in arousal levels. Researchers can presume that heightened arousal levels after certain stressors (e.g. giving a public presentation) are likelier accompanied by an unpleasant (e.g. fear) rather than a pleasant (e.g. excitement) state. 57
Another important reason to include a stressor is that it provides the possibility of assessing physiological (stress) recovery. Increased physiological activation is not necessarily a detrimental outcome; large stress-induced increases in arousal are associated with several favourable outcomes, including improved cognitive performance, lower day-to-day stress levels and lower levels of psychosomatic symptoms. 58 Thus, a healthy reaction can be associated with a rapid physiological response to a stressor; however, it is also associated with a quick dissipation of the physiological arousal once the stressor is removed.8,59
In contrast, the unhealthy response pattern is characterized by physiological arousal that either persists long after the stressor is removed or is repeatedly activated.10,60,61 To be able to capture this response pattern, studies should include a stressor and increase the length of the assessment period. When selecting stressors, advantage should be given to those that can reliably elicit intense stress responses. 62 Note that the mentioned considerations are compatible with SRT, which does not predict unequivocal changes in arousal without first considering other factors. 21
Despite the importance of including a stress-inducing activity for experimental purposes, the experience of stress should not be viewed as a dichotomous event, either fully expressed or not existing at all. Instead, stress can be considered as existing on a continuum with relaxation on the opposite end, where one side is characterized by extreme feelings of distress together with high physiological arousal and the other by considerable feelings of calm and low levels of physiological arousal.63–65 In the absence of a distinct stress-inducing occurrence, one should not be presumed completely relaxed. Accordingly, even studies not including a stress-activity can bring useful findings, because we cannot assume that in such studies a person is already in an optimal state and that environmental interventions cannot further improve it. In such situations, however, it is important to be especially careful in interpreting physiological data before drawing conclusions, as the data can reflect states other than stress or relaxation (e.g. specific affective states, such as feelings of interest).
Assessing affective states
Six out of nine reviewed studies included a measure of affective states.34,35,47–51 While the incorporation of such a measure is valuable, choosing the one most fitting to the study design is important.
Not including a measure of affective states
Measuring affective states is critical when investigating responses to stress in indoor environments, as this both clarifies and complements physiological measures.
On top of illuminating often ambiguous physiological data, assessing affective states enables capturing changes that are too subtle to be detected by physiological measures alone. Subjects may experience changes in their feelings without the concomitant changes in autonomic arousal 66 and these changes are important in the aetiology of stress. For example, pleasant affective states are thought to both restore coping resources and sustain coping with stressful situations.67,68
Selecting an unsuitable measure of affective states
Five out of six reviewed studies34,47–51 that used a measure of affective states employed the Profile of Mood States (POMS; e.g. Yokoyama et al. 69 ), but none of the studies provided a rationale for using this questionnaire. POMS, originally named the Psychiatric Outpatients Mood Scale, measures six specific states which were deemed important by psychiatrists assessing the effects of various drugs on patients, particularly on war veterans showing symptoms of post-traumatic stress disorder. ‘POMS’ primary targets were depression and anxiety (reflected in the scales ‘depression’ and ‘tension’), while the scales ‘anger’, ‘vigour’ and ‘fatigue’ were of interest due to being related to common side effects of medication. ‘Confusion’ scale was added to assess potential disruptive effects of drugs on mental functioning. 70 Why these specific affective phenomena are expected to vary in indoor environments is not clear. Consequently, the relevance of POMS in restoration studies is unclear, despite its popularity. The reviewed studies using POMS may have missed changes in affective states by failing to measure more relevant constructs (or they measured relevant affective states that were then diluted by the presence of irrelevant ones).
Another issue with using POMS in such contexts is related to its length and its repeated administrations. The questionnaire consists of many items (65 in its original form), which are often responded to multiple times in a short period of time. Multiple mood ratings in quick succession may lead to misleading similarities in results between measurements. 71
To select more appropriate measures, researchers should first decide which affective phenomena to study (i.e. core affect, emotion or mood), then select the most relevant theoretical framework conceptualizing the chosen phenomena, and finally select the psychometrically most robust measure that is based on the chosen theoretical framework. 70 Moods (e.g. irritability) are an appropriate target for studies seeking to examine longer-lasting affective changes, for example when investigating long-term effects of exposure to indoor environments. Other studies might be interested in assessing a specific emotion, such as social anxiety following a public speech task. When a specific affective response cannot be anticipated in advance, as is the case in many restoration studies, it is reasonable to capture core affect70,72 (e.g. with the Affect Grid that targets the broad states of pleasure and arousal 73 ).
Assessing cognitive performance
Only two out of nine reviewed studies incorporated a measure of executive functions.36,46 Considering the lack of studies including a cognitive task, we would like to emphasize the importance of examining cognitive performance, while at the same time taking into account the roles of affective states and physiological arousal.
Necessity of assessing cognitive performance
Assessing cognitive performance is important for at least two reasons. First, directed (voluntary) attention, an important facet of cognitive performance, 74 may play an important role in the aetiology of human stress. 6
Second, directed attention may be a common resource in executive functioning and self-regulation. 75 Recent findings show, for example, that exposure to nature delays gratification, 14 inhibits aggressive urges 15 and boosts persistence and results on logical reasoning tasks. 13 Thus, a natural environment could not only enhance performance on a variety of cognitive tasks, but also lead to other health-related improvements that are associated with higher self-regulation ability, such as improved coping with stress and healthier food choices.
As natural environments may influence executive functioning without significantly changing affective or physiological states, important discoveries can be overlooked if cognitive tasks are not incorporated (see Parsons, 27 for a brief overview).
Considering methodological caveats when assessing cognitive performance
There are many methodological caveats when investigating changes in cognitive performance in response to natural environments. Some of the important considerations include (1) measuring cognitive performance both before and after exposure to natural stimuli, (2) employing a cognitive task (before the exposure) that is demanding enough to sufficiently deplete cognitive capabilities and (3) selecting the duration of the rest period that will be long enough to allow restorative qualities of an environment to take effect but short enough that cognitive capabilities will not recover regardless of the environment. 76 Specific properties of cognitive tasks should also be considered, as some tasks may be better suited to capture potential restorative qualities of the environment.26,29 Due to the scope and significance of important considerations, we refer the reader to the work of Neilson et al., 25 Ohly et al., 26 Stevenson et al. 29 and Hartig and Jahncke 76 for an in-depth discussion on these issues.
Considering affective states and physiological arousal when assessing cognitive performance
Additionally, we would like to emphasize that affective and physiological states have an important role in executive functions77,78 that has been widely debated.75,79–81 In restoration studies, it is not clear if improvements in cognitive performance are observed due to recovered cognitive capabilities or instead result from changes in affective states and physiological arousal.27,28 To ascertain the mechanisms behind potential improvements in cognitive performance, affective and physiological states must be considered.
Results of individual studies and discussion
After addressing general methodological issues uncovered in the reviewed studies, we will now separately examine the findings of each study. Results of individual studies are summarized in Table 2 and presented in more detail in Table S4.
Summary of the results of the reviewed studies.

We will first address four studies that employed shorter contact times with wood (90 s to each condition),34,48–50 before continuing with an overview of studies with longer exposure durations.
In the study from Tsunetsugu et al.49,50 15 subjects first spent time in a practice room, which consisted of intermediate amounts of wood. After that, they were exposed for 90 s to both a ‘standard’ and a ‘designed’ room, in random order. The standard room was prepared to resemble a typical Japanese living room, where wood was applied mainly in flooring; the designed room was identical but also included exposed wooden elements on the walls and ceiling. In each room, heart rate, blood pressure and blood flow in the prefrontal cortex were measured; in addition, subjects completed a self-report of affective states (i.e. POMS). The results showed that the heart rate tended to increase in the designed room and decrease in the standard room. Diastolic and systolic blood pressure tended to slightly decrease in both rooms, while blood flow in prefrontal cortex increased in both rooms. Differences in POMS among these rooms were not observed.49,50
The interpretation of these results is challenging. First, because of the short assessment time and absence of a stress-inducing activity, what the physiological data represents is unclear. In this case, the detected increase in heart rate may have reflected either pleasant or unpleasant changes in affective states 54 that could go undetected by POMS. SRT cannot illuminate the detected pattern of physiological responses, since the theory predicts regulation of arousal; depending on the initial arousal level, both decrease and increase could be considered a positive outcome. However, even if the data would be more revealing, attributing physiological changes to any particular aspect of the tested environments would be difficult, as the test settings did not differ only by the quantity of wood, but also by the degree of room novelty and specific design features.
The same authors later conducted a similar study using the same physiological and psychological measures. 34 This time, they created three test rooms with clearer differences in the amount of wood; these rooms were treated with either 0%, 45% or 90% wood coverage. After spending time in the practice room, 15 subjects were exposed for 90 s to each of the three rooms in random order. In all rooms, diastolic blood pressure decreased significantly, while systolic blood pressure followed a similar pattern. Subjects in all rooms also exhibited a tendency towards increased blood flow to the prefrontal cortex. Heart rate tended to increase in the two rooms with the largest amount of wood coverage, while it did not change in the non-wood room. The two wooden rooms were also rated as more natural than the room without wood. There were no differences in reported affective states on POMS among rooms.
As in the previous study, the meaning of the physiological data is unclear. Perhaps the observation that the two wooden rooms showed both an increase in heart rate and a higher rating of naturalness hints to the possibility that an increase in heart rate reflected a certain pleasant affective state, which has been shown to occur in natural environments. 18 However, this is just one possible explanation; the implication of the observed outcomes is uncertain. Tsunetsugu et al.34,50 are appropriately modest in interpreting physiological data, which they see as an indication that changes among the environments occurred, but their explanation does not go beyond that.
Another similar study conducted by Sakuragawa et al. 48 compared the effects of being exposed to a wall panel made of either wood or steel. Fourteen subjects were exposed for 90 s to each of the following three conditions in random order: facing either a wooden wall panel, a white steel wall panel or a white curtain (control). Their blood pressure was monitored constantly throughout this process. In each condition, subjects also completed a semantic differential scale and POMS.
Yet again the results are inconclusive. Systolic blood pressure slightly increased in the first seconds of exposure to hinoki wood panels and then quickly returned to pre-exposure levels. In contrast, no significant changes in systolic blood pressure were detected when participants were exposed to the white steel panel. The results were then separated into three groups based on participants’ preference of the respective wall panels (‘like’, ‘neither like nor dislike’ and ‘dislike’ group; based on the answers provided on one item) and blood pressure data were analysed for each group separately. The analysis showed that the subjects’ blood pressure tended to be lower when they spent time in the settings they liked, and vice versa, suggesting the decreases in blood pressure reflected a positive outcome, but the evidence behind this explanation is weak. The authors equate increased blood pressure with ‘stress’, but we argue that this proposed relationship is not strongly supported by the observed data.
POMS results speak in favour of the wooden setting. Compared to the control setting, participants who were exposed to wood panels had lower scores on the Depression scale (with items such as ‘unhappy’ and ‘discouraged’). In contrast, when exposed to white steel panels, subjects tended to have higher scores on the Depression scale in addition to lower scores on the Vigour scale (with items such as ‘energetic’ and ‘active’). Taken together, results from the study suggest that even a brief exposure to a wooden wall panel may be enough to produce favourable physiological and affective changes. This response pattern, noted by decreased physiological arousal accompanied with improved affective states, could be consistent with certain aspects of SRT, which predict that decreased arousal is linked with mild and moderate levels of interest accompanied by preference of an environment and feelings of calm. However, the results should be interpreted cautiously. Subjects’ brief exposures to different wall panels were followed by relatively long self-reports of affective states. This might have inadvertently created conditions encouraging the good-subject effect, where participants are able to discern the hypothesis of a study and start to behave in ways that will confirm the hypothesis. 82
In Nakamura et al.’s study, 47 28 participants viewed three image projections (1 m × 1 m; 1 m away from the subject) for 90 s (in random order). The images consisted of grey colour (control), vertical wood grain and horizontal wood grain. Heart rate, heart rate variability and prefrontal cortex oxygenation were measured throughout, while affective states were captured with the POMS (2nd edition) following each exposure.
Differences in heart rate or heart rate variability between the image viewings were not detected. In contrast, blood oxygenation in subjects’ prefrontal cortices was lower when they observed the two wood images (compared to the grey image viewing). The authors interpret this observation as ‘physiological relaxation’ but we argue that the interpretation is not that straightforward, as the left and right prefrontal cortices and even specific regions within each of these cortices can respond differently to the same stimulus.83,84 This is further complicated due to the variety of cognitive and affective processes prefrontal cortex is implicated in.85,86 The overall activity in the prefrontal cortex should not be equated with arousal or relaxation.
Compared to the control image, participants reported their affective states as more favourable after viewing the wood images; these ratings were even more favourable for vertical wood grain image compared to the horizontal wood grain image. This finding suggests that the rotation of the grain pattern may have an important role and should be considered. Explaining this observation with SRT is especially difficult, as the theory discusses human response to the natural environment as a whole. Such findings demonstrate the need for theories to delve deeper in explaining human response to natural stimuli (e.g. Joye et al. 87 ). As in the study from Sakuragawa et al., 48 the results on affective states should be interpreted cautiously, as they may suffer from the good-subject effect, with participants acting in ways to confirm the experiment’s hypothesis.
The interpretability of the results reviewed so far is limited due to several methodological approaches: short exposure time to wood, no stress-reducing activity, few physiological measures, unsuitable measurement of affective states, conditions possibly encouraging the good-subject effect and small sample sizes. In contrast, the following five studies are characterized by longer exposure times to wood and typically include a stress-inducing activity and larger sample sizes.
One of these studies was conducted by Fell, 36 where each quarter of the total 119 subjects spent approximately 40 min in one of the four settings – a room with a wooden interior with plants, a room with a wooden interior without plants, a room with a non-wooden interior with plants or a room with a non-wooden interior without plants. After spending 10 min in the room (baseline period), subjects performed a cognitive task (Paced Auditory Serial Addition Test (PASAT) 88 ) for approximately 15–20 min which was primarily employed to induce stress. After completing the task, subjects spent additional 10 min in the room (recovery period). Their electrodermal activity and heart rate were constantly monitored.
Physiological outcomes did not differ between the conditions with and without plants, while differences between the wooden and non-wooden settings were detected. Specifically, exposure to wood was associated with lower skin conductance levels and frequency of non-specific skin conductance responses. As the ‘anticipation and performance of practically any task will increase both skin conductance levels and the frequency of NS-SCRs [non-specific skin conductance responses]’, 89 we have a reason to presume that the exposure to wood and the decreased physiological response are linked. However, as discussed in the previous sections, it is not certain that such an outcome must be considered positive. Importantly, even though the tested settings differed in the average arousal levels, they did not differ in the degree of recovery following stress induction. This observation is not in line with SRT, which suggests that, when the initial arousal level is high, exposure to natural stimuli will be more effective in decreasing it compared to non-natural environments. One possibility is that the wooden environment led to a decrease in arousal already at the beginning of exposure and that the arousal remained lower throughout the entire experimental session. Overall, the findings of the study provide some evidence that visual exposure to wood leads to lower levels of physiological arousal, but the difference between these environments was found only in average arousal levels, not in the degree of recovery after stress was induced. Additional information would be valuable to corroborate these results (e.g. self-reports of affective states).
The study did not find any differences in cognitive performance among subjects. By taking a strict ART perspective, we could argue that there were no differences because no stress-inducing or attention-depleting activity took place prior to the cognitive task, so it is reasonable to expect that subjects were able to perform near their best regardless of the environment. Put differently, even if wooden environments possessed attention restoring qualities, they were not given the opportunity to demonstrate them. It is far from clear, however, if the improved attentional capabilities in natural environments indeed result from restoration of attentional resources or if other mechanisms are central. 27 For instance, if enhanced attention capacity is mediated by affective states, differences in PASAT scores had the opportunity to emerge between tested environments.
We would like to draw attention to additional reservations about the results of this study. First, since only one room was available for the experiment, all non-wood sessions were completed before moving on to wood sessions, which might have influenced the results (as the author recognized). The second issue is that the study did not follow a proper randomization process which might have brought on baseline differences between participants that were not accounted for. A proper randomization process and taking baseline measures of participants before they were exposed to the experimental setting would have strengthened the study’s findings.
In Bamba and Azuma’s study, 45 12 subjects started each of the three experimental sessions in the baseline room, where they spent approximately 45 min while completing a 30 min long cognitive task used as a stressor. After this, they spent 10 min in one of the three test rooms; a different test room was used every session (the order was randomized). In the first experimental room, one wall was almost entirely covered by a solid wood panel in which slits were cut to increase the room odour (volatile organic compounds). The odour also reached the adjacent second experimental room that did not include a wood panel. The control room did not include either the wood panel or the wood odour. Measures of heart rate, heart rate variability and salivary alpha-amylase activity (higher levels of salivary alpha-amylase activity indicate activation of the sympathetic nervous system) 90 were taken both in the baseline room and in the test settings.
There were no detected differences in any of the physiological outcomes between the three test rooms, even though the subjects reported a more pleasant odour and lower levels of fatigue in the room with the wood panel, compared to the other two rooms. The results do not match the findings from Sakuragawa et al., 46 who observed differences in physiological responding in similar experimental circumstances. The findings also go against several studies observing decreases in the same physiological indicators following stimulation with wood odour91–94; however, the olfactory stimulation in these studies was likely more intense. While the intervention employed by Nakamura and colleagues might not enhance recovery after stress induction, confident conclusions cannot be drawn mainly due to the small sample size of the study.
In Zhang et al. study,51,52 20 subjects were exposed to four rooms in random order. One of the rooms was painted white (control); the other three rooms consisted of either 100% of dark wood coverage, 100% of light wood coverage, or 50% of light wood coverage and 50% of walls painted white. Before entering each room, subjects spent 30 min in the preparation room and then another 60 min in each of the experimental rooms. Several physiological measures were taken throughout the whole procedure, together with the measures of cognitive performance, affective states (POMS) and fatigue.
Physiological responses during exposure to wooden environments were conflicting. Some markers were associated with increased levels of autonomic arousal (i.e. skin conductance level, peripheral oxygen saturation), while others corresponded to decreased autonomic activation (i.e. heart rate, heart rate variability, blood pressure), or did not differ between settings (i.e. skin temperature). 56 The most prominent physiological changes were observed in systolic blood pressure, which was generally lower in the wooden environments. Due to diverging findings, the authors’ claim that ‘the participants were in a more relaxed and comfortable state in a wooden indoor environment’ is not sufficiently grounded in the data. 52
POMS produced clearer results. After spending time in the preparation room, subjects exhibited higher mood improvement in all three wooden rooms compared to the room without wood. However, as the (wood) odour differed between the test rooms, it is not clear if the improved affective states resulted from visual or olfactory stimulation. SRT notes that certain natural smells (and sounds) can influence humans positively but future studies are needed to distinguish between the respective effects of visual and olfactory exposure to wood.
In a study from Demattè et al., 35 102 participants spent 15 min in each of these two test rooms in random order. Both rooms featured one wooden and one non-wooden desk. Floor and walls were heavily treated with wood in one room, while they remained untreated (i.e. white) in the other. In each setting, subjects completed a Positive and Negative Affect Schedule (PANAS) 95 once when they entered and once directly before they left the room.
There were no differences between the first and second PANAS administration within the two settings; a brief experience of either of the two indoor environments did not significantly influence affective states tapped by PANAS. In contrast, when the PANAS scores were compared between the rooms, the results indicated that the participants experienced higher levels of pleasant and lower levels of unpleasant affective states in the wooden environment. However, due to solid wood use, the odour differed between the two test settings and it is not clear whether visual or olfactory stimulation is responsible for the observed effects. However, the authors were not concerned with differentiating between the effects of visual and olfactory stimulation, as they intended to test the effects of wood in ‘immersive everyday life conditions’.
In Burnard and Kutnar’s study, 46 61 subjects spent 75 min in each of the two office-like rooms in random order. All subjects spent time in a control room with white furniture, and a room with either oak veneered or walnut veneered furniture. In each setting, participants were exposed to a stress-inducing video and completed a cognitive task, while their salivary cortisol and heart rate were monitored (neither heart rate nor cognitive task results were reported). Cortisol responses in the walnut room did not significantly differ from the control room responses, while the subjects in the oak room exhibited lower average cortisol levels. However, the study did not find any differences in the magnitude of cortisol response and recovery after stress induction. As was the case in the study from Fell, 36 this finding is not consistent with SRT that predicts more effective recovery in natural environments when the initial arousal levels are high. Assuming that the wood office affected stress response and recovery, the differences in magnitude could have been missed due to non-continuous cortisol measurement or because peak cortisol concentrations may have occurred between the cortisol readings. Along similar lines, the experiment may not have lasted long enough to allow the cortisol levels to return to the baseline. Although cortisol responses typically return to baseline after 60 min following the stress induction, 62 the last stage of the experiment consisted of a cognitive task that might have delayed the recovery.
As cortisol is generally a more reliable indicator of distress than typically deployed physiological measures (e.g. electrodermal activity), 58 lower average cortisol readings in the oak office represent a promising finding. Still, cortisol concentrations can be expected to only moderately correlate with perceived stress.96,97 In addition, the cortisol readings in the oak office were lower than in the control room even when compared only for the period before the stress induction. Because peak cortisol concentrations appear in blood and saliva between approximately 21 and 30 min after stress induction, 62 the first three readings were likely affected by the time before the experiment, suggesting a possible difference in baseline cortisol levels of participants. Alternatively, and similar to the study by Fell, 36 participants could have been influenced by wood exposure immediately upon arriving in the test office and remained in a more relaxed state throughout the entire experiment.
In summary, from the eight studies assessing physiological arousal, four studies provide inconclusive results,34,47–50 two studies offer some evidence that physiological arousal is decreased in wooden environments,36,46 one study presents mixed findings51,52 and one study reports no detected differences in physiological responding between the tested environments. 45 From the six studies examining affective states, two studies observed no differences in response to higher amount of wood coverage,34,49,50 while the other four studies found evidence that affective states are improved in wooden environments35,47,48,51,52; however, in the two out of four studies, olfactory stimulation could have been the main cause of improved affective states. From the two studies investigating cognitive performance, one study did not report the results 46 and the other did not observe any differences between wooden and non-wooden settings. 36
Summary of evidence
We reviewed nine studies assessing either affective, physiological or cognitive performance outcomes to visual wood exposure. The results of four studies with shorter exposure durations to wood provide relatively little information regarding the influence of wood exposure on indicators of stress.34,47–50 Four out of five studies with longer exposure durations detected at least some favourable (or seemingly favourable) outcomes in wooden environments.35,36,46,51,52 The results from Fell 36 and Burnard and Kutnar 46 are promising since both studies found that the physiological arousal of participants is lower in the wooden environments. However, neither study detected any differences between the settings regarding the degree of stress recovery, and in both cases the findings were not corroborated by additional measures of affective states, physiological arousal or cognitive performance. Studies from Zhang et al.51,52 and Demattè et al. 35 observed more favourable affective states in the wooden environment, but in neither case it is clear if this was influenced by visual or olfactory properties of the experimental room(s). Only Fell’s 36 study reported cognitive performance outcome and it did not find any differences between the wooden and non-wooden environments. Overall, current research suggests that visual wood exposure may lead to certain favourable outcomes, but the evidence is limited. Future studies are needed to clarify and confirm the current findings before confident conclusions can be drawn.
Limitations
The present review has several limitations. It includes only nine studies and in those the overall risk of bias is high in three experiments, concerning in two investigations and low in only four studies. Additional limitations are found in specific methodological approaches observed in the reviewed studies. Most studies measured only one or few outcomes, which is generally not sufficient to arrive at robust conclusions when examining the effects of indoor environments on humans. On top of that, it is not clear if several positive findings of the reviewed studies should be attributed to olfactory or visual stimulation of wood (or both). More generally, methodology in restoration studies may suffer from demand characteristics, 25 where participants anticipate what researchers are predicting and (unconsciously) respond in a way that fits the research hypothesis. 82 Additional limitations of the review result from the small number of included studies; even few additional studies could have influenced the conclusions of the review. Along similar lines, publication bias might have had a major effect on the review’s findings. The number of studies may be limited due to including only studies reported in English and using a search strategy that restricts the topic to human response to wood in indoor environments. A review of the stress-related outcomes following exposure to wood in outdoor environments may be warranted as well.
Conclusions
Our review addresses how using wood in indoor environments influences affective states, physiological arousal and cognitive performance of the room occupants. We reviewed nine studies reported in 10 scientific articles and one doctoral dissertation. Our inspection assessed the methodology and the results through the lens of the multi-dimensional examination of human stress. Current research suggests that visual wood exposure could lead to beneficial outcomes, but the evidence is limited. In general, studies are limited by not examining multiple dimensions of stress indicators simultaneously, which limits the interpretability of their findings. Taken together, the studies reveal a potential for the benefits of wood use in buildings, but it is critical that future studies confirm and expand current findings to ensure any recommendations for building design can be supported by evidence.
Recommendations for future studies
When examining the effects of wood exposure in built environments, future studies should simultaneously investigate affective, physiological and cognitive performance outcomes. By considering the interplay among these concepts, we can better understand human responses to different indoor settings. In addition, each of the incorporated measures should be chosen carefully to fit with each other as well as with the general study design. In general, studies should (1) incorporate a variety of physiological measures to better encompass variable changes in physiological arousal levels; (2) include a suitable measure of affective states (e.g. a measure of core affect) that will both help explain physiological data and provide additional information about the subjects’ response to environments and (3) incorporate an appropriate task assessing executive functions, ideally combined with an intervention that will lead to attention fatigue in participants. Researchers should primarily focus on assessing stress recovery, that is capturing subjects’ physiological, affective and cognitive performance outcomes following the induction of stress. Studies aiming to test the effects of visual wood exposure should be designed carefully in order to control for the effects of tactile and olfactory contact with wood.
The findings of this review reflect a field in its infancy. However, with (1) the minimal risk of side effects, (2) relative affordability, (3) high potential for large scale and long-term implementation and (4) minimal demands on human effort, visual wood exposure is a potential environmental intervention against stress that remains worthy of future investigation.
Supplemental Material
sj-pdf-1-ibe-10.1177_1420326X20927437 - Supplemental material for Effects of visual exposure to wood on human affective states, physiological arousal and cognitive performance: A systematic review of randomized trials
Supplemental material, sj-pdf-1-ibe-10.1177_1420326X20927437 for Effects of visual exposure to wood on human affective states, physiological arousal and cognitive performance: A systematic review of randomized trials by Dean Lipovac and Michael D. Burnard in Indoor and Built Environment
Supplemental Material
sj-pdf-2-ibe-10.1177_1420326X20927437 - Supplemental material for Effects of visual exposure to wood on human affective states, physiological arousal and cognitive performance: A systematic review of randomized trials
Supplemental material, sj-pdf-2-ibe-10.1177_1420326X20927437 for Effects of visual exposure to wood on human affective states, physiological arousal and cognitive performance: A systematic review of randomized trials by Dean Lipovac and Michael D. Burnard in Indoor and Built Environment
Supplemental Material
sj-pdf-3-ibe-10.1177_1420326X20927437 - Supplemental material for Effects of visual exposure to wood on human affective states, physiological arousal and cognitive performance: A systematic review of randomized trials
Supplemental material, sj-pdf-3-ibe-10.1177_1420326X20927437 for Effects of visual exposure to wood on human affective states, physiological arousal and cognitive performance: A systematic review of randomized trials by Dean Lipovac and Michael D. Burnard in Indoor and Built Environment
Supplemental Material
sj-pdf-4-ibe-10.1177_1420326X20927437 - Supplemental material for Effects of visual exposure to wood on human affective states, physiological arousal and cognitive performance: A systematic review of randomized trials
Supplemental material, sj-pdf-4-ibe-10.1177_1420326X20927437 for Effects of visual exposure to wood on human affective states, physiological arousal and cognitive performance: A systematic review of randomized trials by Dean Lipovac and Michael D. Burnard in Indoor and Built Environment
Footnotes
Authors' contribution
All authors contributed equally in the preparation of this manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors acknowledge the InnoRenew CoE project (Grant Agreement 739574) funded by the European Commission under the Horizon2020 Widespread-Teaming program and the Republic of Slovenia (Investment funding of the Republic of Slovenia and the European Union of the European Regional Development Fund).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
