Abstract
Aims:
Leisure-time physical activity (LTPA) is known to be hereditary for two generations, but its transmission beyond that is less studied. This study examined LTPA aggregation in three-generation families.
Methods:
Data on self-reported LTPA were extracted from the Young Finns Study in 2018–2020, including three generational groups: offspring (G2; aged 7–38 years, n = 2499), parents (G1; aged 43–58 years, n = 1960) and grandparents (G0; aged 58–94 years, n = 2190). In the analysis, G2 was categorized into two age groups: youth offspring (aged 7–18 years) and adult offspring (aged 19–38 years). Correlations, chi-square test and binary logistic regressions were used to compare the differences in LTPA levels (low vs. high) between G1–G2, G0–G2 and G0–G1, stratified by gender and age.
Results:
In the youth offspring group, high-active G1 fathers were more likely to have highly active G2 sons, while high-active G1 mothers were more likely to have highly active G2 daughters when compared with their low-active counterparts. High-active G0 maternal grandmothers had a higher probability of having highly active G2 granddaughters than low-active maternal grandmothers. In the adult offspring group, high-active G0 maternal grandfathers were more likely to have highly active G2 grandsons compared with low-active maternal grandfathers.
Conclusions:
Background
Insufficient levels of leisure-time physical activity (LTPA) continue to be a significant issue among children and youth in Finland [1] and globally [2]. Fewer than 25% of Finnish adolescents aged 13–18 years meet the recommendation of the World Health Organization (WHO) of 60 min of moderate-to-vigorous physical activity (MVPA) daily [3]. This guideline includes activity across all domains (i.e. school, home, commuting and leisure). For adults, the WHO recommends 150–300 min of moderate or 75–150 min of vigorous aerobic activity weekly. LTPA, though just one domain of MVPA, is especially important as it is voluntary and modifiable, making it a key target for behavioural and public health intervention [3]. Promoting LTPA can support lifelong activity habits that adapt to changing social and environmental contexts.
Family dynamics strongly influence children’s LTPA, with parents playing a pivotal role in shaping active lifestyle [4–6]. The parent–child LTPA relationship varies by gender and age, as paternal and maternal role modelling might affect sons and daughters differently [7–10]. Since LTPA typically declines with age [2], and youth participate more in sports, they may be especially sensitive to familial influence [11]. These findings support early-life, gender-sensitive and family-based interventions to promote lifelong LTPA. Social learning theory [12] suggests that behaviours like LTPA are acquired through observation, imitation and reinforcement in social contexts. Within families, children are more likely to adopt behaviours modelled by close caregivers, making parents key role models for healthy habits [4–6]. This framework also helps explain multigenerational patterns of LTPA, where consistent modelling reinforces active behaviour across generations.
However, none has examined the impact of gender and age on familial LTPA patterns across three generations, primarily because of a limited sample size. In our ongoing Cardiovascular Risk in Young Finns Study (YFS), we have found that parental LTPA is not only linked to childhood and adolescent LTPA levels but also influences activity levels during the transition to adulthood [13–15]. Nevertheless, essential factors regarding the impact of parental modelling on children’s LTPA remain inadequately explored, potentially contributing to the observed inconsistencies in findings. A systematic review has highlighted a consistently positive association between parent–child LTPA, regardless of the child’s age, the gender composition of the parent–child pair, or the specific type of LTPA involved [16]. Similarly, no research has examined how age and gender affect the relationship between grandparental LTPA and their grandchildren’s LTPA.
Two previous studies found weak or no significant correlations in intergenerational LTPA across three generations [17,18]. Notably, dose–response relationships were observed between highly active mothers and daughters’ LTPA [17], and between mothers of active versus less active adolescents [18]. Overall, LTPA in one generation showed limited influence on the next. In the longitudinal YFS, participants transitioned from parents to grandparents (G0), their children became parents (G1) and they, in turn, had their own children (G2). In 2018–2020, G2 of the original cohort was enrolled, allowing us to assess intergenerational LTPA associations. To our knowledge, this is the first study to evaluate such associations within family samples, stratified by gender and age. The study aimed to investigate intergenerational LTPA associations across three generations using YFS data. This encompassed: (1) LTPA associations in G0 and G1 with G2, (2) associations between G0’s and G1’s LTPA and (3) whether these relationships were moderated by G2’s gender and age. We hypothesized that higher LTPA in G0 and G1 would correlate with increased LTPA in G2, and higher LTPA in G0 with G1, with potential gender and age differences.
Materials and methods
Data from the YFS included children and adolescents aged 3−18 years in 1980 (G1), representing six age-cohorts born between 1962 and 1977 [19]. Of the 4326 boys and girls invited, 3596 (83%) were consistently studied at 3−9-year intervals from baseline to 2018–2020. G1 was randomly selected in 1980 from five university cities with medical schools (Helsinki, Kuopio, Oulu, Tampere and Turku) and their surrounding rural communities. They were followed for 40 years (aged 43–58 years) and data for G2 offspring (aged 7–38 years) were collected in 2018–2020. The original G0 generation (aged 58–94 years) was also followed [13–15]. For this study, we used the 2018–2020 data as it marked the first time self-reported LTPA was collected across three generations, totalling 2499 responses for G2, 1960 for G1 and 2190 for G0. Sociodemographic and anthropometric characteristics were recorded for each generation in the YFS. The study protocol was approved by the ethics committees of participating universities (ETMK: 68/2017) and written informed consent was obtained from all participants in accordance with the Helsinki Declaration.
G2 offspring’s LTPA was assessed using a self-administered questionnaire, identical to that used for G1 and completed alongside clinical examinations (e.g. height, weight and blood sampling). Children aged 7–11 years were instructed to ask for parental assistance if needed. For youth offspring (ages 7–18), the questionnaire concerned LTPA frequency and intensity, sports club participation, involvement in competitions, and typical leisure activities. For adult offspring (ages 19–38), it included questions on activity intensity, frequency of vigorous LTPA, hours of vigorous activity, average session duration, and participation in organized LTPA. To account for developmental differences, G2 was categorized into youth offspring and adult offspring, reflecting slight age-based variations in LTPA measures [14]. G0 participants completed the same adult LTPA questionnaire as G1. Responses were based on average hours/times per week. Each item was scored from 1 to 3: 1 (inactive/very low), 2 (moderate) and 3 (frequent/vigorous). These were summed into an index ranging from 5 to 15, with higher scores indicating more activity. The indices were dichotomized into low (⩽8) and high (>8) categories for each generation, following a widely used approach [20]. The G1 index has demonstrated reliability and validity across the lifespan [15,21,22].
Age and residence (urban versus rural) were queried for all participants. Among G0, G1, and G2 adult offspring, educational attainment was self-reported as completed years of schooling and grouped into three levels: low (⩽ 9 years); intermediate (10−12 years) and high (>12 years). Occupation was divided into four categories based on the criteria of the Central Statistical Office of Finland: manual (builders, metal workers, nannies, etc.), lower non-manual (civil servants, specialized and skilled workers, etc.), upper non-manual (administrators, managers, academics, etc.) and student (adult offspring). Body mass index (BMI) was calculated as weight (kg)/height (m2). Screen time was self-reported as average daily minutes of TV viewing and computer use for weekdays and weekends. Mean daily screen time (min/day) was calculated as (5 × weekday + 2 × weekend)/7.
Descriptive characteristics are expressed as means (SD) for continuous variables and proportions (%) for categorical variables. Intergenerational correlations (Pearson’s r) were calculated to assess relationships between LTPA across generations. Chi-square tests were employed to examine the differences in LTPA levels between different generations. Binary logistic regressions were used to estimate odds ratios (ORs) and 95% confidence intervals (CIs) for LTPA differences, both unadjusted and after adjusting for parental or grandparental age, residence, education, occupation, BMI and screen time. Analyses were stratified by gender and age within each G2 group. Data analysis was performed using the R environment [23] and Mplus Version 7.0 [24] via the R software package MplusAutomation [25]. Missing data were assumed to be missing at random, meaning they were not related to unobserved variables but were considered as a function of the observed covariates and outcomes [24].
Results
Characteristics of the participants, categorized by generation, are presented in Table I. The proportion of females exceeded that of males across all three generations, with over 60% residing in urban areas. LTPA and educational levels tended to be higher among younger generations. In occupational status, over 40% of G2 adult offspring were actively engaged in university studies, while manual work remained a significant occupation across all generations. BMI and screen time seemed to be relatively consistent across generations.
Characteristics of the study sample for all family members.
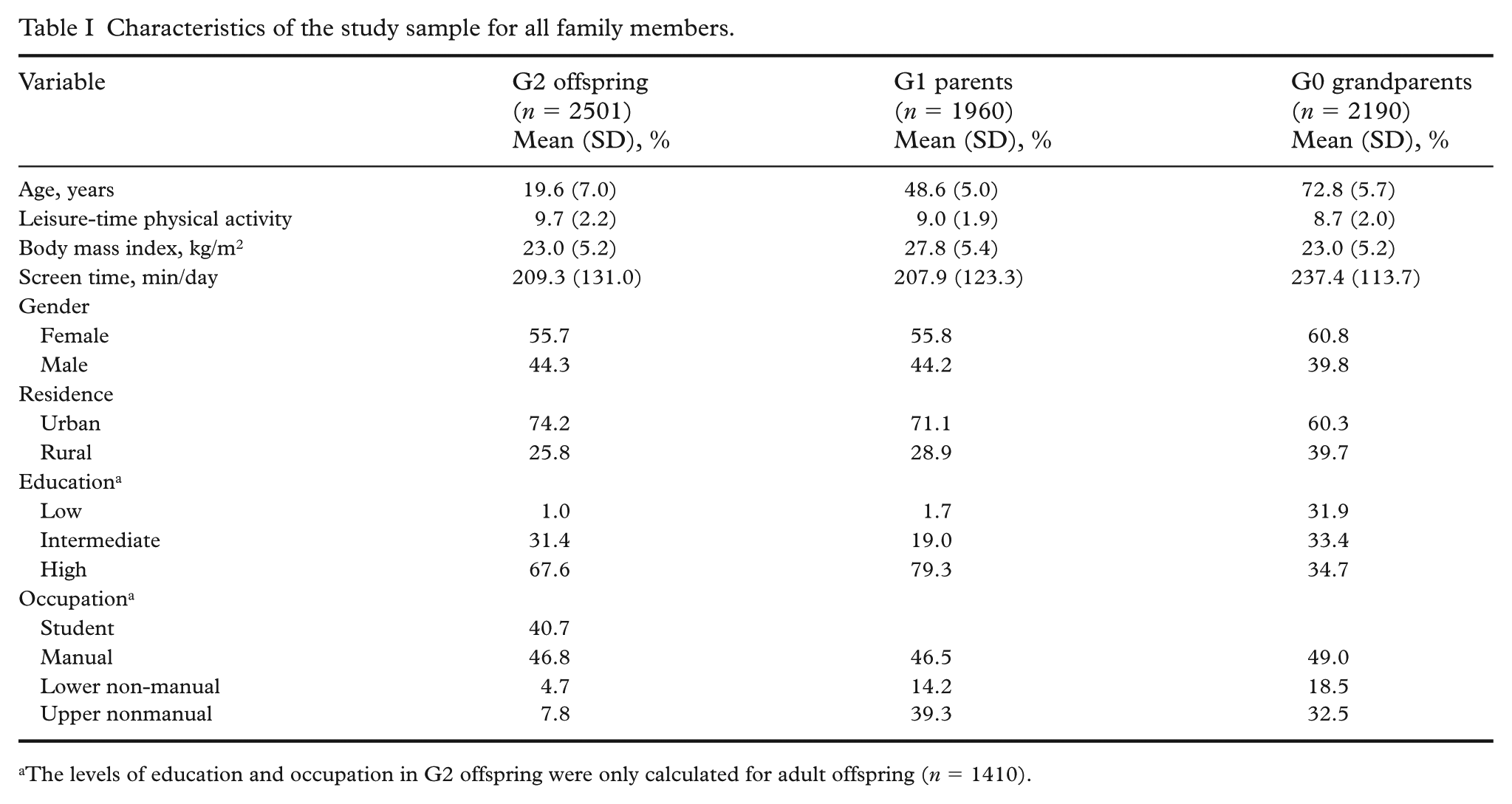
The levels of education and occupation in G2 offspring were only calculated for adult offspring (n = 1410).
In the youth offspring group, there were positive correlations between the LTPA of fathers and that of sons and daughters (p<0.05 for both)(Table II). Similar correlations were observed for mothers with sons (p<0.05) and daughters (p<0.01). Granddaughters’ LTPA was directly associated with the LTPA of maternal grandfathers and grandmothers (p<0.05 for both). In the adult offspring group, fathers’ LTPA was positively correlated with the LTPA of sons (p<0.01) and daughters (p<0.05), while mothers’ LTPA was directly associated with that of daughters (p<0.05). Maternal grandfathers’ LTPA was also directly correlated with the LTPA of grandsons (p<0.01). Only one significant correlation was found between mothers’ LTPA and that of their mothers (p<0.01).
Correlations (Pearson’s r) among measures of leisure-time physical activity in three generations stratified by sex and age group.
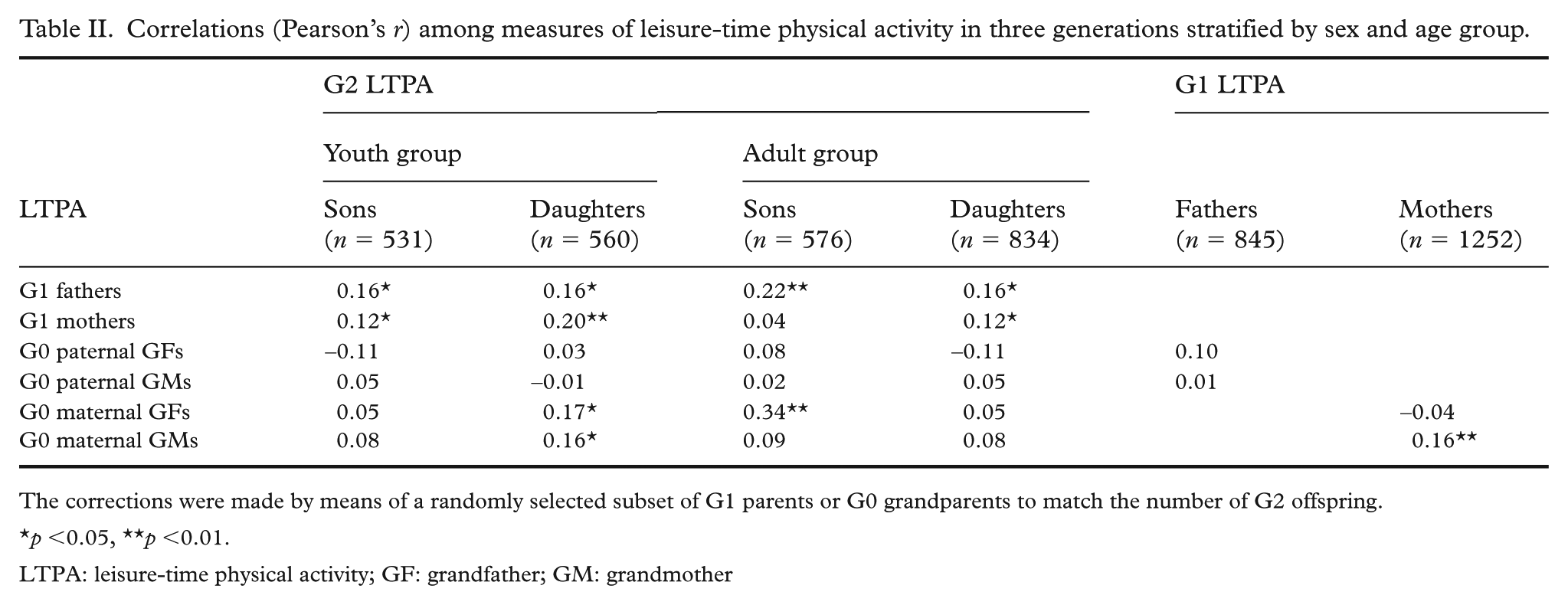
The corrections were made by means of a randomly selected subset of G1 parents or G0 grandparents to match the number of G2 offspring.
p <0.05, **p <0.01.
LTPA: leisure-time physical activity; GF: grandfather; GM: grandmother
In the youth offspring group, sons of high-active fathers had higher odds of being highly active compared with sons of low-active fathers (OR = 2.08; 95% CI: 1.02–4.26), after adjusting for potential covariates (Table III). Urban residence was positively associated with LTPA in sons, while non-manual occupation was associated with lower odds. Similarly, daughters of high-active mothers had more than twice the odds of being highly active compared with daughters of low-active mothers (OR = 2.69; 95% CI: 1.46–4.97), independent of the covariates. Among daughters, urban residence was positively associated with LTPA, whereas greater screen time was inversely associated.
Associations of parental leisure-time physical activity levels and covariates on offspring’s leisure-time physical activity levels. Odds ratios and 95% confidence intervals are from unadjusted (univariate) and adjusted (multiple) logistic regression models.
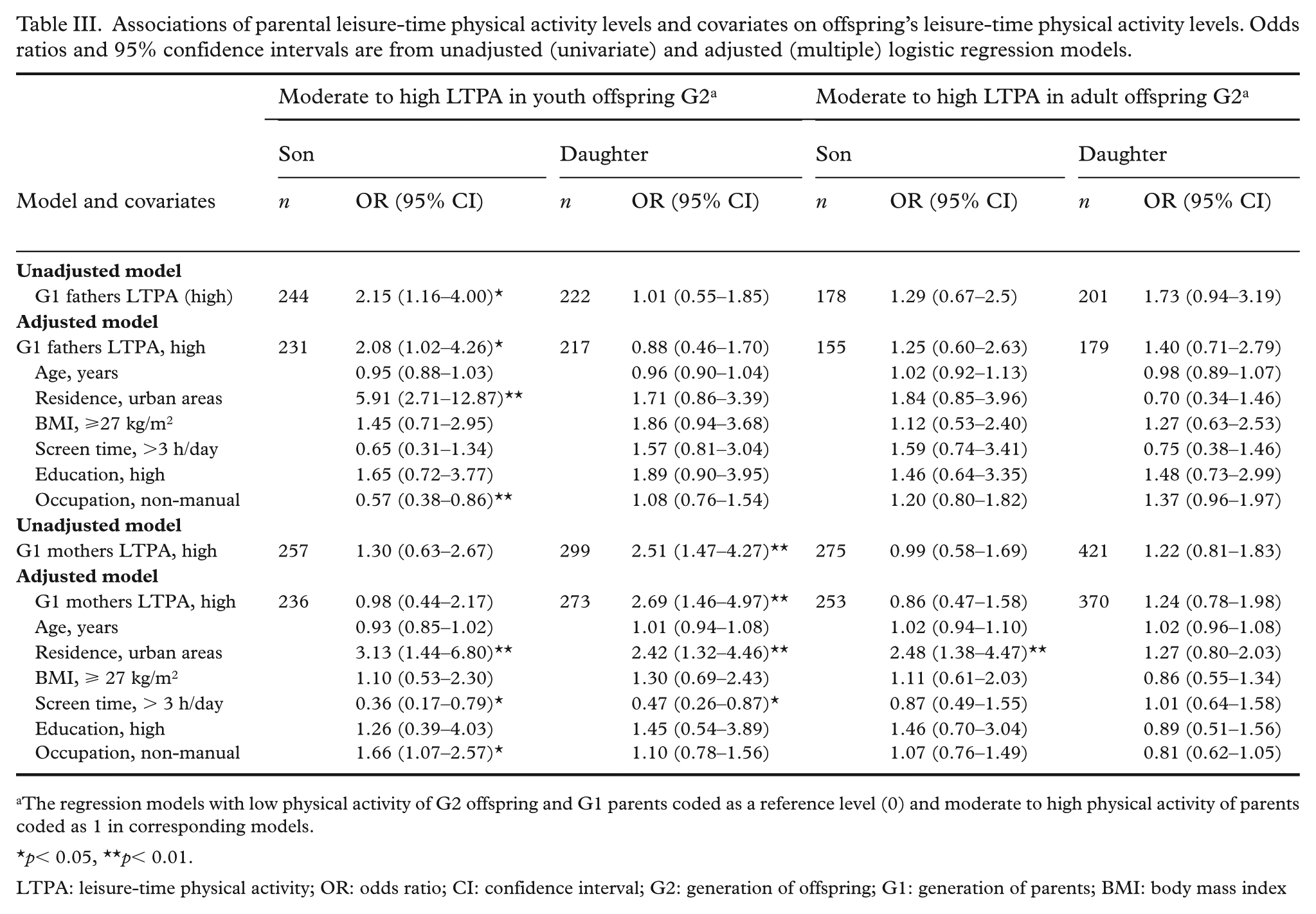
The regression models with low physical activity of G2 offspring and G1 parents coded as a reference level (0) and moderate to high physical activity of parents coded as 1 in corresponding models.
p< 0.05, **p< 0.01.
LTPA: leisure-time physical activity; OR: odds ratio; CI: confidence interval; G2: generation of offspring; G1: generation of parents; BMI: body mass index
In the adult offspring group, grandsons of high-active maternal grandfathers had markedly higher odds of being highly active compared with those of low-active grandfathers (OR = 12.23; 95% CI: 1.83–81.91) (Table IV). Similarly, in the youth offspring group, granddaughters of high-active maternal grandmothers were more likely to have highly active granddaughters compared with their low-active counterparts (OR = 5.21; 95% CI: 2.12–12.77). These associations remained significant after adjustment for covariates across both paternal and maternal lineages. Among adult granddaughters, greater screen time, higher educational attainment, and non-manual occupation were also associated with increased LTPA.
Associations of grandparents’ leisure-time physical activity levels and covariates on offspring’s leisure-time physical activity levels. Odds ratios and 95% confidence intervals are from unadjusted (univariate) and adjusted (multiple) logistic regression models.
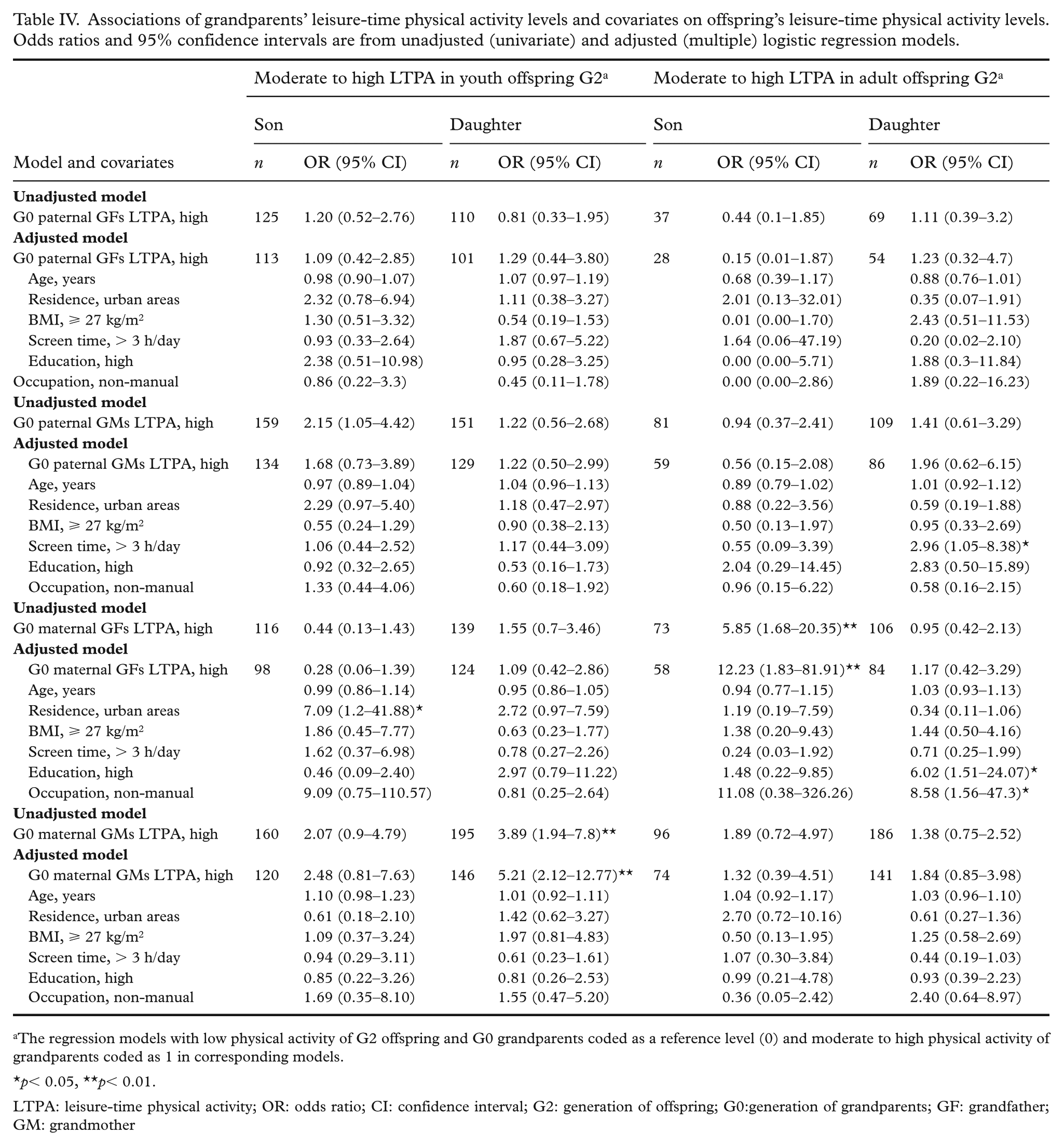
The regression models with low physical activity of G2 offspring and G0 grandparents coded as a reference level (0) and moderate to high physical activity of grandparents coded as 1 in corresponding models.
p< 0.05, **p< 0.01.
LTPA: leisure-time physical activity; OR: odds ratio; CI: confidence interval; G2: generation of offspring; G0:generation of grandparents; GF: grandfather; GM: grandmother
Discussion
The study examined the intergenerational pattern of LTPA within families, taking into account gender and age-related variations. We found significant relationships between the LTPA levels of G1−G2, G0−G2 and G0−G1 within specific gender and age subgroups. These findings partially support our hypotheses, indicating that the LTPA of both G0 and G1 is directly associated with that of G2 in certain groups. The results emphasize the effects of shared familial resemblance in shaping LTPA behaviours among younger generations, particularly G2, and highlight the role of gender- and age-specific factors on these intergenerational patterns.
The results provide compelling evidence for the links between the LTPA levels across three generations. Specifically, G1 fathers exhibited a stronger LTPA association with younger G2 sons, whereas G1 mothers displayed such association with younger G2 daughters. G0 grandmothers had a higher LTPA association with G1 daughters. We also examined a three-generational pattern of LTPA across these generations. Importantly, the maternal line indicated a clear transmission of LTPA across generations, influencing the LTPA levels of G2 in both the youth female and the adult male groups. In contrast, the paternal line seemed to exhibit a two-generational transmission of LTPA in G2. These results support and extend upon previous research [26,27], indicating that a substantial portion of LTPA variance is influenced by a combination of genetic and shared environmental factors. Additionally, these findings suggest that intergenerational associations in LTPA might be more persistent within the maternal line, reflecting the strong influence of heightened health awareness, lifestyle choices and cultural factors among Finnish women [28].
To date, only two previous studies extended beyond G1−G2 LTPA correlations to explore G0−G2 relationships. In a study of 3254 Finnish twins aged 16 years, G2 daughters of active G1 mothers had higher LTPA levels than those of inactive mothers [17]. However, relying on G1 reports for G0 LTPA might lead to inaccuracies. Another study of 248 Israel youth aged 12−16 years created composite LTPA measures using data from both G1 and G0, distinguishing between maternal and paternal lines [18]. The results revealed a direct correlation between G2 youths’ LTPA and their G1 mothers, with no significant associations for other family members. Our results are partially consistent, also indicating stronger maternal influences on daughters’ LTPA. However, unlike prior research that found no grandparental effects [17,18], we identified significant G0–G2 associations in specific subgroups, suggesting that intergenerational influence may be more persistent in certain family structures or cultural contexts.
The transmission of parental LTPA behaviour from one generation to the next serves as a mechanism to explain intergenerational transmission of the behaviour [29]. According to social learning theory [12], this transmission is facilitated through modelling, wherein children observe and replicate their parents’ behaviours. When parents or older family members prioritize LTPA, they set a positive example for the younger members of the family. This might serve as a potent source of inspiration for children to adopt a healthy and active lifestyle. Parental participation in LTPA has been associated with higher levels of LTPA in children, often through role modelling or shared activities [4–6]. Previous studies found that children engaged in higher LTPA levels when they had physically active parents to serve as role models [8,13]. Furthermore, active parents play a crucial role in establishing a foundation for fostering healthy behaviours in their offspring. Recent research has revealed that parents’ initial LTPA levels concurrently and prospectively predict their children’s LTPA levels [14,15]. We extended this theory regarding the transmission of behaviours from older to younger generations within families.
Our findings provided limited evidence of intergenerational transmission. Specifically, higher LTPA levels in G1 mothers during interactions with their G0 mothers were associated with increased LTPA in their G2 younger daughters. Similar associations were found between G0 maternal grandfathers and G2 adult grandsons. The relationship between LTPA levels of G0 paternal grandmothers and G2 youth grandsons disappeared when controlling for covariates. Cumulative positive outcomes were observed, particularly for G2 younger daughters with active mothers and active maternal grandmothers. The findings align with previous research demonstrating a matrilateral bias, where maternal grandparents, particularly grandmothers, are more involved in childcare and maintain stronger emotional bonds with their grandchildren than paternal grandparents [30]. This closer involvement might enhance their influence on granddaughters’ LTPA by providing more frequent opportunities for interactions and behavioural modelling, consistent with social learning theory. As primary caregivers in many families, maternal grandparents help shape daily routines and attitudes toward physical activity.
We also found that the association between G0 paternal grandmothers and G2 youth grandsons’ LTPA disappeared after adjusting for such factors as age, residence, education, occupation, BMI and screen time, suggesting that the observed intergenerational pattern, in part, reflects underlying socioeconomic and lifestyle factors rather than direct behavioural transmission.
Gender and age displayed distinct associations in intergenerational LTPA relationships across three generations. Significant differences in LTPA levels were observed within the maternal line, particularly pronounced between maternal grandparents and their grandchildren in both the G2 youth female and adult male groups. Understanding the underlying reasons for these diverse patterns among different gender–age groups poses a considerable challenge. One plausible explanation is that these differences might be attributed to increased interaction opportunities between the G2 generation and their G0 maternal grandparents, but further evidence is needed for validation. These findings contribute to the expanding body of literature that explores the relationships between gender, age and LTPA across generations. It is noteworthy that the large OR in the G2 adult male group might result from small sample size and coincidental factors, rather than a single outlier within the covariate pattern.
This study partially supports the hypothesis that G0 and G1 influence G2’s LTPA, particularly in younger generations. While not establishing causality, the findings suggest that LTPA behaviours in G0 and G1 might shape LTPA levels in younger G2, though this influence weakens as G2 reach adulthood. As they mature, individual and environmental factors increasingly drive their LTPA choices, underscoring a shift from intergenerational influence on personal agency. Nonetheless, the role of social learning remains relevant, with behavioural modelling potentially shaped by broader factors such as education, health behaviours and access to supportive environments. Further research is needed to explore how individual, social and environmental dynamics impact LTPA across generations. These insights can inform targeted interventions aimed at promoting LTPA among G2 of varying ages, considering the contributions of both G1 and G0.
Our study has several key strengths. It employed a prospective study design spanning a substantial 40-year period and encompassing G0, G1 and G2. The most recent LTPA data collected for this study originated from three participant generations. This extended timeframe provided unique insights into the evolution of major potential covariates across the generations, enhancing the comprehensiveness of our analysis. The analyses conducted appropriate statistical methods to evaluate complex patterns of associations among variables across different generations.
However, we note some limitations of the study. First, self-reported LTPA might be affected by inaccurate recall and social desirability bias, potentially underestimating activity stability across generations. Second, although parent–child data were collected, co-parents were not included, which may introduce information bias. Further studies are warranted to confirm our findings, particularly in comparisons between biological and non-biological parents. Third, only LTPA was assessed, excluding other activity domains like school, work, household and commuting, possibly underestimating total physical activity. Fourth, environmental factors beyond residential location such as neighbourhood, school or community were not measured. These variables warrant attention in future intergenerational LTPA research. Fifth, although we found a positive dose–response relationship in LTPA across generations, possible non-differential misclassification might have weakened observed intergenerational associations. Finally, the sample consisted mainly of White Europeans with higher income, limiting generalizability to more diverse populations.
Conclusions
Grandparents’ and parents’ LTPA independently influence offspring’s LTPA, especially among younger individuals. Our findings highlight intergenerational dynamics within families and partially support the hypothesis of LTPA transmission across generations. This three-generational pattern offers a clear target for age- and gender-sensitive interventions. Future research should further explore multigenerational influences on LTPA. These patterns have important implications for promoting physical activity and well-being, regardless of biological relatedness.
Footnotes
Acknowledgements
The authors extend their gratitude to the study children and their parents and grandparents for their invaluable participation in the YFS over a 40-year follow-up period. The authors also wish to acknowledge the YFS staff who have contributed to the data collection process.
Declaration of conflicting interests
The authors have no conflicts of interest to declare.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Academy of Finland (grant numbers 356405, 322098, 286284, 134309 (EYE), 126925, 121584, 124282, 129378 (SALVE), 117797 (GENDI) and 141071 (SKIDI); the Finnish Ministry of Education and Culture; the Social Insurance Institution of Finland; Competitive State Research Financing of the Expert Responsibility area of Kuopio, Tampere and Turku University Hospitals (grant number X51001); Juho Vainio Foundation; Paavo Nurmi Foundation; Finnish Foundation for Cardiovascular Research; Finnish Cultural Foundation; the Sigrid Juselius Foundation; Tampere Tuberculosis Foundation; Emil Aaltonen Foundation; Yrjö Jahnsson Foundation; Signe and Ane Gyllenberg Foundation; Diabetes Research Foundation of Finnish Diabetes Association; EU Horizon 2020 (grant number 755320 for TAXINOMISIS and grant number 848146 for To Aition); European Research Council (grant number 742927 for MULTIEPIGEN project); Tampere University Hospital Supporting Foundation; Finnish Society of Clinical Chemistry; the Cancer Foundation Finland; pBETTER4U_EU (Preventing obesity through Biologically and bEhaviorally Tailored inTERventions for you; project 101080117); CVDLink (EU grant number 101137278), the Jane and Aatos Erkko Foundation, and Academy of Finland (research fellowship number 322112).
