Abstract
Aims:
The autonomic nervous system includes parasympathetic and sympathetic components that monitor and regulate most of the bodily functions and play a central role in the physiology and homeostasis of the human body. Heart rate variability is a non-invasive tool for quantification of rhythmic fluctuations in heart rate that reflects the function of the autonomic nervous system. The study aims to describe the heart rate variability distribution in the general population, stratified in sex and age groups, which is currently insufficiently described.
Methods:
A cross-sectional population-based study recruited participants in 10 municipalities in the western part of the greater Copenhagen area in Denmark, including 6891 men and women aged 18–72 years (participation rate was 29.5%). Short-term heart rate variability measures were obtained and related to age and gender.
Results:
Both time and frequency domain measures showed a huge variation in the different sex and age groups. Women had a higher median heart rate than men, and the association with age was U-shaped. Measures indicating a predominance of the parasympathetic component in relation to the sympathetic component were more frequent in women and younger age groups.
Conclusions:
Keywords
Background
The autonomic nervous system (ANS) plays a central role in the physiology and homeostasis of the human body. Both subsystems of ANS, the parasympathetic nervous system (PNS) and the sympathetic nervous system (SNS), monitor and regulate most of the bodily functions – often in an antagonistic interplay. ANS is centrally located in the reflex arcs and has negative feedback mechanisms that maintain an optimal internal environment [1]. Heart rate variability (HRV) is a non-invasive and inexpensive tool for the quantification of rhythmic fluctuations in heart rate (HR) reflecting the influence of SNS and PNS on the sinus node, and HRV is thereby an approximate measure of the ANS function [2].
The association between variations in heart rate, health and disease states have been recognized for more than 2000 years [2], but it is only during the last 50 years that the distinct physiological rhythms embedded in the oscillations of HR have been described [2].
In 1996, the European Society of Cardiology and the North American Society of Pacing and Electrophysiology established a task force concerning HRV methodology [3]. Since then, thousands of papers on HRV have been published, and the importance of HRV as a measure of ANS activity in an array of diseases has been recognized. Reduced HRV can predict the risk of arrhythmia and mortality after acute myocardial infarction [4] and is associated with a poor prognosis in a wide range of other diseases, including diabetes [5]. Psychophysiological research has in recent years taken an interest in HRV, driven by the association between the parasympathetic nervous system and self-regulation mechanisms [6].
Female sex is associated with higher overall variability in HR – with a tendency for this sex-dependency to decline with age [7]. Furthermore, there is a well-documented association between increasing age and decreasing HRV, most notably with regard to measures reflecting PNS activity and reactivity [3,8].
The demonstrated relationship between altered HRV and both diagnosis and prognosis of an array of conditions has highlighted the value of altered HRV to predict conditions that profoundly influence public health and has placed HRV as a measure of interest in public health research. In 1996, the task force emphasized the need for large prospective population-based studies to establish normative values for HRV, as the implementation of HRV measures in clinical work as well as public health research has been hampered by large inter-individual variation in HRV, the lack of relevant reference values across sex and age subsets, and reliance on cross-sectional studies collecting data from different selected materials [3,9]. In recent years, studies have emerged presenting normative values of relevant HRV indices and demonstrating the influence of age and sex on HRV. However, most of these studies have been based on small or selected populations and a narrow age span and have employed different methodological approaches [10–16].
With the present study, we aim to present normative values for the most commonly reported HRV parameters in the time and frequency domain in age and sex subsets in a large Danish population-based study, the Danish Study of Functional Disorders (DanFunD) (N=6891) [17].
Methods
Study population
Details of the DanFunD study have been reported previously [17]. The DanFunD-II cohort is a random sample of the general population obtained from the Danish Central Personal Register. A total of 25,368 men and women aged 18–72 years living in the western part of the greater Copenhagen area were invited, of whom 7493 participated (29.5%). The only exclusion criteria were not being born in Denmark, not being a Danish citizen, or being pregnant. Written informed consent was obtained from all participants, and the study was approved by the Ethical Committee of Copenhagen County (Ethics Committee: H-3-2012–0015) and the Danish Data Protection Agency.
Participants were invited for a health examination, including questionnaires, physical tests and biological samples [17]. All examinations were performed between 08:00 h and 15:00 h and lasted about 90 min. Participants were examined after at least 6 h of fasting and were asked to abstain from smoking at least one hour prior to the examination. Participants were resting in the supine position for 5 min before the measurement of continuous heart rate using the ‘E-motion’ heart rate monitor device (eMotion HRV, Bittium, Kuopio, Finland). The analysis is based on the normal-to-normal (NN) intervals between successive sinus node derived heart beats, and the files were selected and prepared accordingly. Measurements were performed during 7 min of supine rest and normal breathing. Both staff and subjects were instructed to refrain from talking apart from necessary commands.
Patient and public involvement
There was no patient or public involvement in the design of the study.
Data preparation
In concordance with current guidelines [3] the last 5 min of the 7 min supine rest and a sampling rate of 250 Hz were chosen. Analysis of HRV was performed using a standardized analysis programme (Kubios, v. 2.0, http://kubios.uku.fi) [18]. In 270 participants, RR intervals were not recorded due to technical errors or known pacemaker-implant. Participants with atrial fibrillation or excessive extrasystoles, defined as more than 20 ectopic beats during the 5-min sampling period, were excluded from further analysis (n=100). Files with technical artefacts due to poorly attached electrodes or equipment failure were also excluded (n=232) leaving 6891 for analysis. Ectopic beats were corrected using a threshold-based artefact correction algorithm [19]. Detrending was performed using smoothness priors with a lambda value of 500 [20].
Data analysis
From the NN intervals, the following statistical indices were calculated: average heart rate (mean HR), RR interval length based on NN intervals (meanNN), the standard deviation of instantaneous of HR values (STDHR) and NN (STDNN), and the root mean square of successive difference (RMSSD) as the square root of the mean squared differences between adjacent NN intervals. pNN50 was calculated as the percentage of successive NN intervals in the recording that differ by more than 50 ms [3].
Power spectral density analysis allows for the estimation of power as a function of frequency. In a short-term recording of instantaneous heart rate, three main spectral components are differentiated by non-parametric Fast Fourier Transformation using Welch’s periodogram with a 300-s window with 50% overlap [21]: very low frequency (VLF; <0.04 Hz), low frequency (LF; 0.04–0.15 Hz) and high frequency (HF; 0.15–0.4 Hz) components. In short-term recordings (5 min or less), total power and VLF variation have ill-defined physiological meaning and are, therefore, not reported [3]. The LF and HF variations are measured and reported both as total power (LFtotal, HFtotal), normalized units representing the relative values of each power component proportional to the total power (minus the VLF component) (LFnu, HFnu) and as the ratio between LF and HF variation (LF/HF).
Statistical analysis
The 5th, 25th, 50th, 75th and 95th percentiles were calculated for all age groups separately for men and women. By means of the Kolmogorov–Smirnov test, it was confirmed that data were not normally distributed, and consequently, non-parametric tests were used to examine differences in various HRV measures, that is, the Mann–Whitney U test was used to examine differences between men and women and the Kruskal–Wallis test was used to examine age-dependent differences for men and women, separately. A p-value of 0.05 or below was considered significant, and all calculations were performed using SPSS statistics version 22 (IBM Corp, Armonk, NY, USA).
To avoid the influence of heart diseases that could possibly affect HRV, participants answering yes to the question ‘has a doctor ever told you that you have had a heart attack or other heart disease?’ (n=345) were omitted in a sensitivity analysis, where the analyses were repeated in the remaining 6546 participants.
Results
The number of cases categorized according to sex, age and median and interquartile range of body mass index (BMI), and systolic and diastolic blood pressure is shown in Table 1.
Number of persons according to sex, age, BMI, and systolic and diastolic blood pressure in the Danish Study of Functional Disorders (DanFunD). The DanFunD study was conducted between 2012 and 2015 and included 3152 men and 3739 women aged 18–72 years living in the western part of the greater Copenhagen area, Denmark.
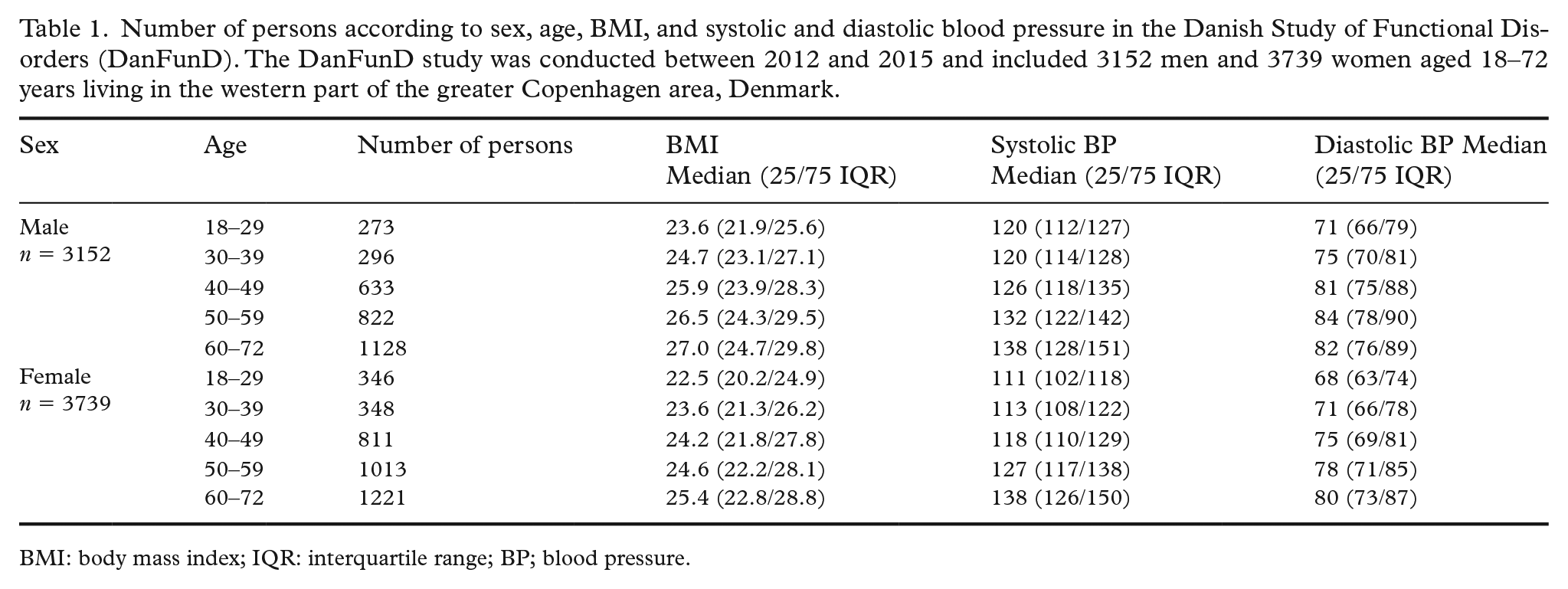
BMI: body mass index; IQR: interquartile range; BP; blood pressure.
Normative values are given as the 5th, 25th, 50th, 75th and 95th percentiles for HRV parameters according to sex and five age groups (Tables 2 and 3).
The Danish Study of Functional Disorders (DanFunD) was conducted between 2012 and 2015 and included 3152 men and 3739 women aged 18–72 years living in the western part of the greater Copenhagen area, Denmark. Percentiles for time-domain heart rate variability measures are categorized according to age group and each sex. N=6891.
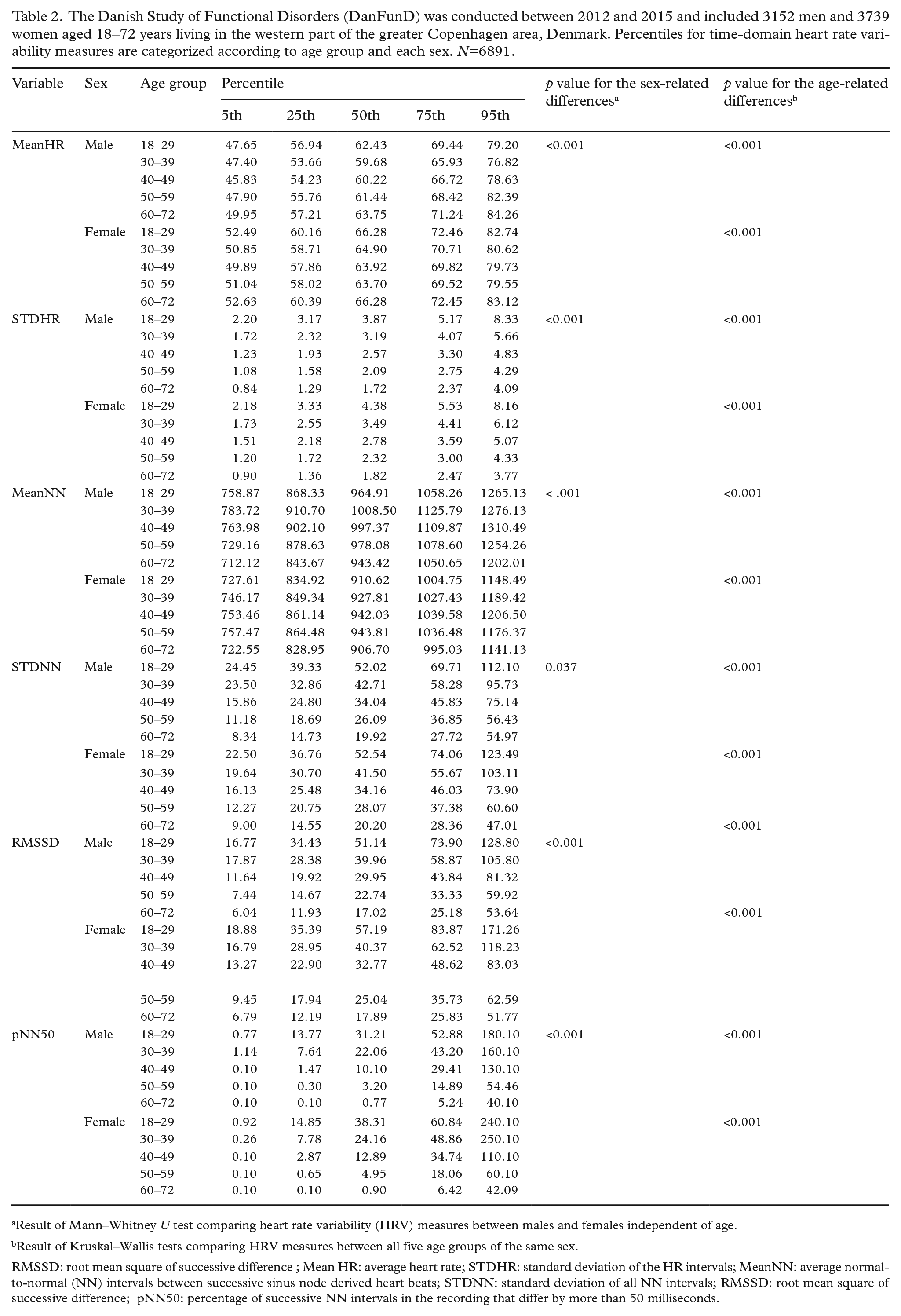
Result of Mann–Whitney U test comparing heart rate variability (HRV) measures between males and females independent of age.
Result of Kruskal–Wallis tests comparing HRV measures between all five age groups of the same sex.
RMSSD: root mean square of successive difference ; Mean HR: average heart rate; STDHR: standard deviation of the HR intervals; MeanNN: average normal-to-normal (NN) intervals between successive sinus node derived heart beats; STDNN: standard deviation of all NN intervals; RMSSD: root mean square of successive difference; pNN50: percentage of successive NN intervals in the recording that differ by more than 50 milliseconds.
The Danish Study of Functional Disorders (DanFunD) was conducted between the years 2012 and 2015 and included 3152 men and 3739 women aged 18–72 years living in the western part of the greater Copenhagen area, Denmark. Percentiles for frequency-domain heart rate variability measures are categorized according to age groups and each sex. N=6891.
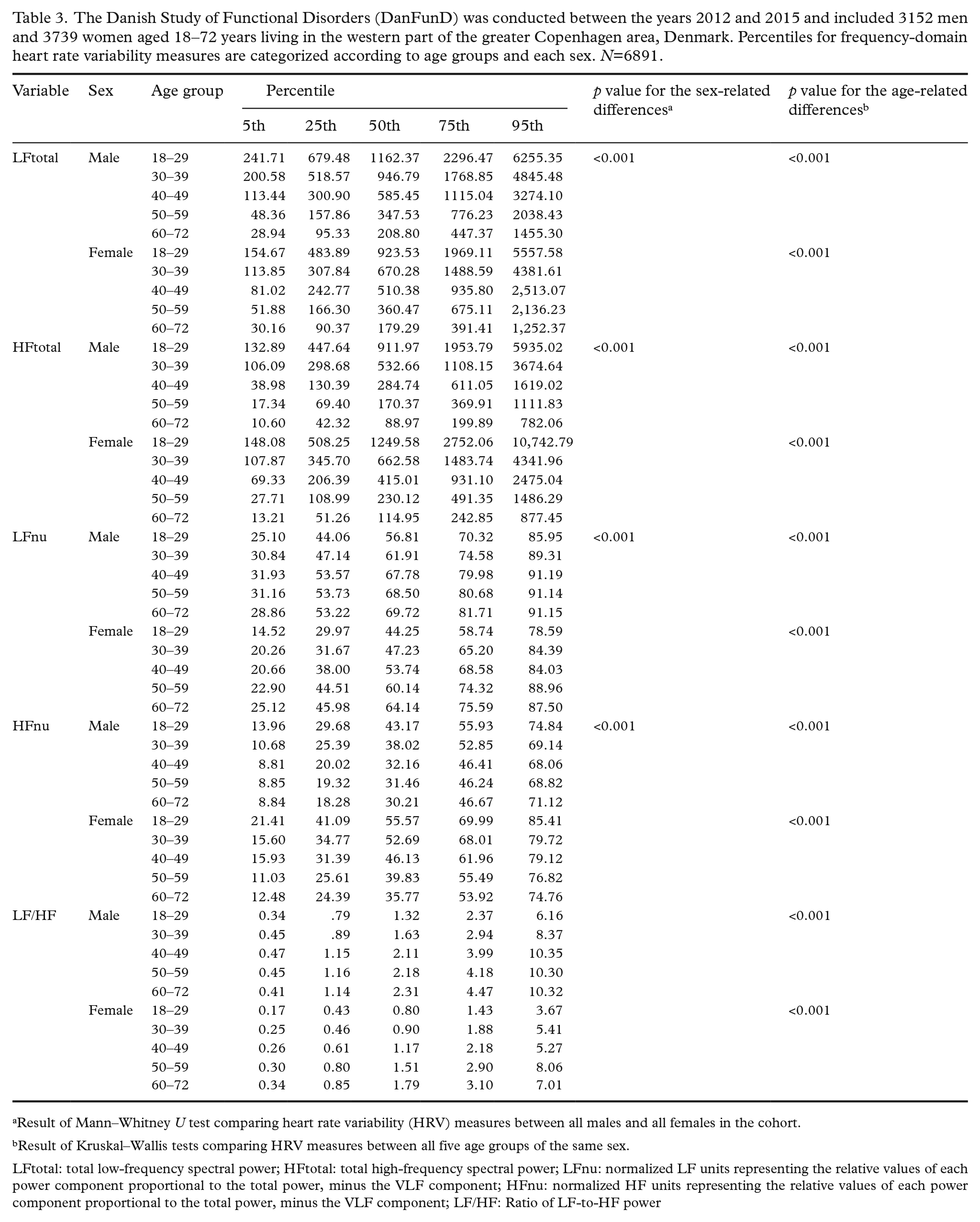
Result of Mann–Whitney U test comparing heart rate variability (HRV) measures between all males and all females in the cohort.
Result of Kruskal–Wallis tests comparing HRV measures between all five age groups of the same sex.
LFtotal: total low-frequency spectral power; HFtotal: total high-frequency spectral power; LFnu: normalized LF units representing the relative values of each power component proportional to the total power, minus the VLF component; HFnu: normalized HF units representing the relative values of each power component proportional to the total power, minus the VLF component; LF/HF: Ratio of LF-to-HF power
Both mean HR and STDHR were higher in women than in men. The association with age was U-shaped for mean HR in both sexes, whereas the association of STDHR with age declined with increasing age (Table 2; Figure 1).
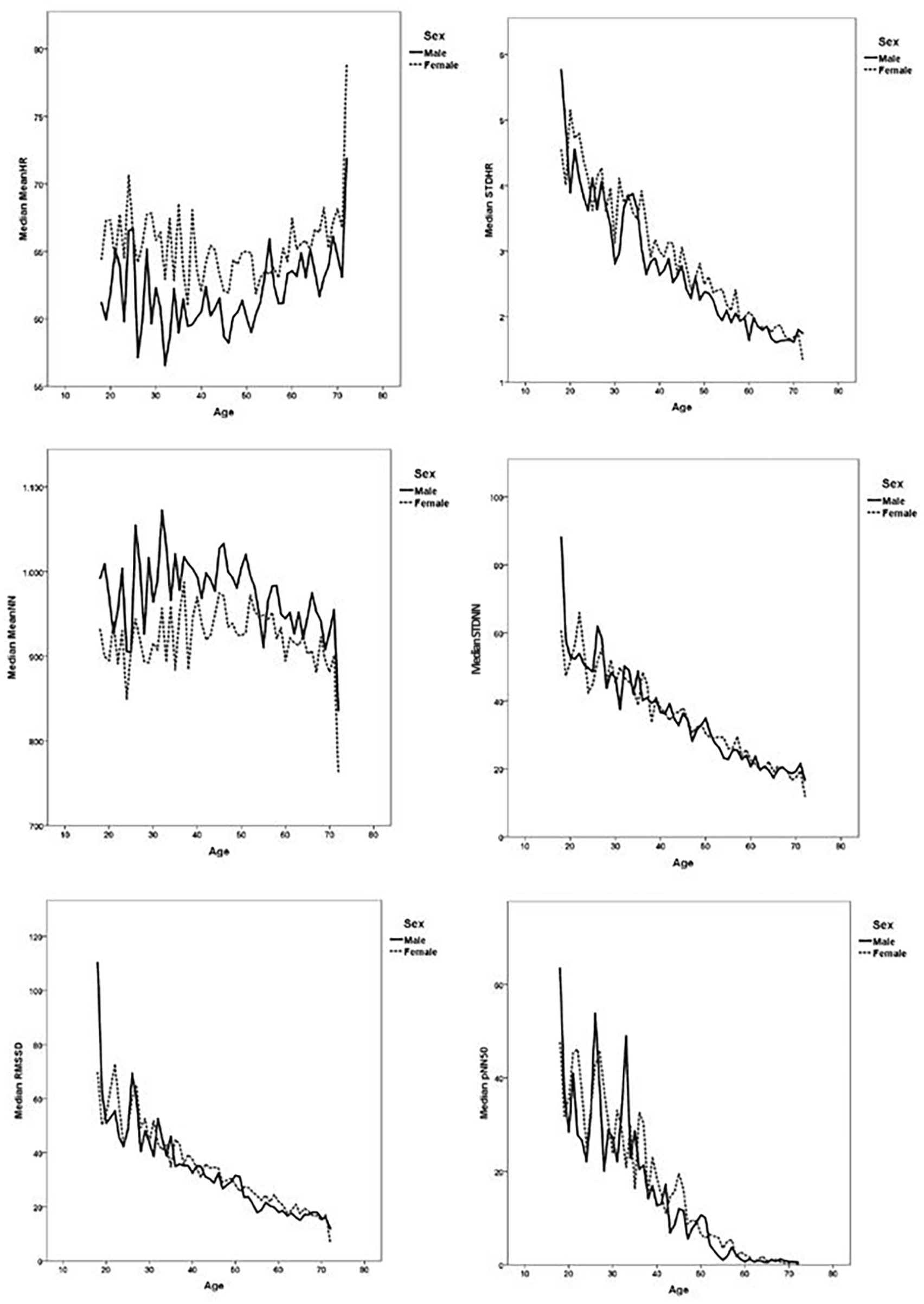
The Danish Study of Functional Disorders (DanFunD) was conducted between 2012 and 2015 and included 3152 men and 3739 women aged 18–72 years living in the western part of the greater Copenhagen area, Denmark. Time-domain heart rate variability measures are shown as median values according to age and sex.
MeanNN was higher in men than in women, and the age relation showed an inverse U-shaped curve in both sexes. STDNN was higher in women than men (Table 2), but curves were very close (Figure 1). STDNN declined with increasing age in both sexes. RMSSD and pNN50 values were higher in women than in men and declined with increasing age in both sexes.
The absolute measures of variability in the LF and HF domains (LFtotal, HFtotal) were negatively correlated with age in both sexes. Men had higher LF variation and lower HF variation compared with women (Table 3; Figure 2).
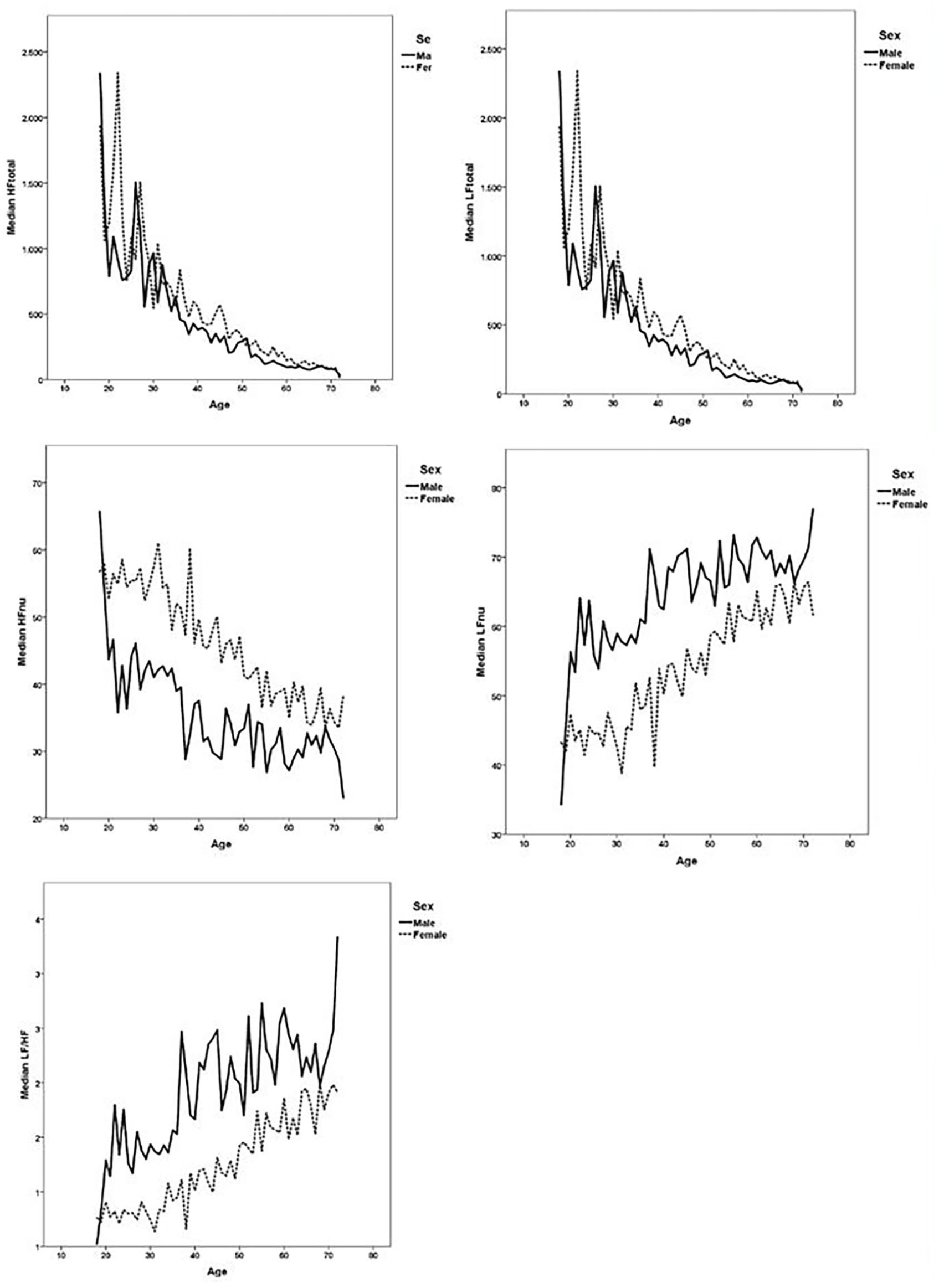
The Danish Study of Functional Disorders (DanFunD) was conducted between 2012 and 2015 and included 3152 men and 3739 women aged 18–72 years living in the western part of the greater Copenhagen area, Denmark. Frequency-domain heart rate variability measures are shown as median values according to age and sex.
The normative and relative measures in the frequency domain (LFnu, HFnu and LF/HF) showed that men had higher LFnu- and lower HFnu-variation compared with women. Women had a relative increase in LFnu and a relative decrease in HFnu with advancing age. Men had a more complex pattern with a tendency for LFnu to increase with age, whereas the slight decrease in HFnu turned into an increase in the 95th percentile. The increase in LF and a concomitant decrease in HF with age resulted in a marked increase in the LF/HF ratio in both sexes but was most pronounced in women, where the LF/HF ratio doubled when comparing the youngest and oldest age groups. Men had a higher LF/HF ratio than women (Table 3; Figure 2).
The sensitivity analysis excluding participants with a previous heart attack or other heart disease did not cause any substantial differences in the described results (data not shown).
Discussion
This manuscript presents age- and sex-stratified reference values for selected HRV measures in the time and frequency domains based on a population-based cohort of 6891 adult Danes by using percentile values for 5-min recordings.
In general, we find age- and sex-related trends similar to those observed in other studies [8,9,11–13,15,16], but the reference values are different, which could be due to differences in size and selection of material and different methodological approaches.
In the healthy heart at rest, the sinus node is under tonic PNS inhibitory control, and basal HR is, therefore, predominantly determined by the activity of the PNS [1]. PNS control on the sinus node is transmitted through large myelinated fibres exerting their effect faster than sympathetic nerve fibres. Thus, high and frequent changes in HR quantified by the time domain parameters RMSSD and pNN50 and the HF parameters of frequency domain analysis will predominantly reflect PNS control on the sinus node. The physiological explanation of the LF component of HRV is less clear. Most likely, the LF HRV predominantly mirrors a baroreflex-mediated response to the SNS mediated rhythmic oscillation of the blood vessels – the so-called ‘Mayer waves’ [22]. Thus, these slower oscillations in HR, quantified by the LF parameters in the frequency domain (LF, LFnu), are thought to reflect the activity in the SNS. However, it is important to recognize that the LF HRV is also dependent on parasympathetic activity and therefore mirrors both sympathetic and parasympathetic activity [22].
The myelin covering the vagal nerve is subject to age-related degeneration [23], which may result in decreasing conduction velocity and ultimately in the well-established age-related decline in cardiovagal control [23,24], which is in accordance with the age-related decline in the present study. We find a U-shaped relationship between HR and age, whereas we find other HRV measures to decline linearly with age. In apparent contrast to our findings, other studies have found a U-shaped correlation between age and HRV parameters mirroring primarily cardiovagal regulation (RMSSD) [25]. However, the increase in parasympathetic indices seen with advancing age in other studies is seen in octo- and nonagenarians with trough values for parasympathetic indices around the age of 70 – which may explain the apparent discrepancy as we included few subjects above the age of 70. The apparent increase in indices of HRV in the octo- and nonagenarians found by others may be due to survival bias.
We confirm an overall tendency for higher values in HRV measures reflecting PNS activity (RMSSD, pNN50, HF, HFnu) in women compared with men. Furthermore, we confirm a tendency in young women (age 18–39 years) for the predominance of HF variation (HF, HFnu) over LF variation (LF, LFnu) and concomitant LF/HF ratios <1 in these younger decades, reflecting higher parasympathetic activity [7,10,16]. The sex-related difference subsides with age as men in all the included age groups (18–72 years) and women from the fourth decade (40–72 years) have a predominance of LF variation –more accentuated in men, which is reflected in higher LF/HF ratios. In both sexes, LF/HF ratios increase with age, mirroring a decrease in cardiovagal regulation and an increase or stationary sympathetic activity with advancing age [7,16]. As an apparent and well-known paradox, we find that women have a higher mean heart rate compared with men [7], which is thought to be mediated not by higher SNS activity but rather by the smaller size of the female heart [26].
Our findings confirm previous findings in both human and animal studies – that (young) females tend to show greater parasympathetic activity compared with men, which may explain some of the health disparity between sexes – one example being the higher cardiovascular morbidity in men as the higher cardiovagal control seen in females is cardioprotective [27,28]. The higher parasympathetic activity in women may be related to the female sex hormone profile as oestrogen and oxytocin increase vagal tone [28,29].
Strength and limitations
The physiological processes generating HRV are mediated in the brain and transferred through the parasympathetic (vagal) and sympathetic nerves to the sinus node, thereby regulating the sinus node’s pacemaker function. Thus, it is important to keep in mind that HRV reflects autonomic regulation of the heart and not generalized autonomic control and activity. Under many circumstances, the autonomic regulation of HR will be in concordance with autonomic outflow to the rest of the body – but in some circumstances, the autonomic outflow is differentiated. One example is postprandially, where vagal activity is suppressed in the heart but increased in the gastrointestinal tract [30].
A major strength, besides the size of the study (N=6891), is the use of a random selection of persons from the background population, which we believe will be valuable in both the research and clinical settings even though the design may introduce healthy user bias. However, omitting participants with known heart disease did not significantly change the overall pattern described or the normative values calculated. Furthermore, data on BMI and blood pressure (Table 1) support the generalizability of the material. A limitation is the age span of 18–72 years, and it would be valuable for future studies to include the whole lifespan.
Analysis of HRV is an inexpensive, non-invasive method that is based on something as readily available as a continuous measurement of heart rate, and data analysis has been made increasingly accessible in the last decade through better analysis algorithms making almost instant test results accessible. HRV assesses activity in the ANS and reflects the ability of the ANS to respond to a variety of physiological and psychological stimuli. HRV has proven to be a promising marker for both physiological and psychological processes and disease states at the group level.
Robust data on normative values for HRV, as we present in this study, are crucial when measures of HRV in both clinical and research setting must be interpreted.
Conclusion
Both sex and age influence HRV in an adult Danish population. Measures indicating a predominance of the parasympathetic component in relation to the sympathetic component were more frequent in women and in the younger age groups. Therefore, age- and sex-related reference values of HRV in the time and frequency domain should be used in further epidemiological and clinical research on HRV.
Footnotes
Acknowledgements
The DanFunD steering committee consists of Professor MD DMSc Torben Jørgensen (PI), Professor MD DMSc Per Fink, senior consultant MD PhD Lene Falgaard Eplov, Professor MD PhD Allan Linneberg, Professor MSc PhD Susanne Brix Pedersen and Professor MD PhD Michael Eriksen Benros.
Data availability
Data cannot be made publicly available for ethical and legal reasons. Public availability may compromise participant privacy, and this would not comply with Danish legislation. Access to the subset of data included in this study can be gained through submitting a request to The Capital Region Knowledge Centre for Data Compliance, The Capital Region Denmark: cru-fp-vfd@regionh.dk. Acquisition of data is allowed only after permission to handle data has been obtained in accordance with the guidelines stated by the Danish Data Protection Agency: ![]() .
.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
