Abstract
Background and aims:
The relationship between childhood tobacco smoke exposure and cardiac structure and function in midlife is unclear. We investigated the association between parental smoking with cardiac structure and function in adulthood.
Methods:
1250 participants (56.5% female) from the Cardiovascular Risk in Young Finns Study who had data on parental smoking and/or serum cotinine, a biomarker of exposure to tobacco smoke, at baseline 1980 (age 3–18 years) and echocardiography performed in 2011. Parental smoking hygiene (i.e., smoking in the vicinity of children) was categorized by parental smoking and serum cotinine levels in offspring. Dimensions of the left ventricle, diastolic and systolic function, and cardiac remodeling were used as outcomes. Analyses were adjusted for sex, age, and covariates (blood pressure (BP), serum lipids, body mass index, socioeconomic status, smoking (only in adulthood)) in childhood and adulthood.
Results:
Parental smoking was not associated with systolic or diastolic function in adulthood. Participants exposed to parental smoking (odds ratio (OR) 1.90, 95%CI 1.23–2.92), hygienic parental smoking (OR 1.74, 95%CI 1.12–2.71), and non-hygienic parental smoking (OR 1.88, 95%CI 1.02–3.45) had higher odds of concentric remodeling (relative wall thickness >85th sex-specific percentile without left ventricular hypertrophy). These associations were attenuated after adjustment for child and adult covariates in the non-hygienic parental smoking group.
Conclusions:
Keywords
Introduction
Cardiovascular disease (CVD) is the main cause of death globally and the majority of these deaths are preventable [1]. Tobacco smoke is an important modifiable risk factor for CVD and atherosclerosis. Both active smoking and exposure to environmental tobacco smoke (or passive smoking) are associated with CVD [2] and with adverse changes in cardiac structure and left ventricle (LV) diastolic function, which have been shown to be associated with an increased incidence of heart failure [3].
Although implementation of strict tobacco policies has reduced smoking and exposure to secondhand tobacco smoke, there is increasing evidence that children exposed to parental smoking suffer long-term detrimental effects to their vascular health, independent of individual smoking habits and exposure to passive smoking later in life [4,5]. Exposure to parental smoking in childhood might increase the harmful effects of subsequent active smoking [6]. Furthermore, non-hygienic parental smoking (i.e. smoking in the presence of children) in childhood has been shown to increase the risk of carotid atherosclerotic plaque in adulthood [7]. Moreover, acute and chronic exposure to passive smoking is known to impair LV systolic and diastolic function [3]. These dysfunctions may result from cardiac remodeling, which is defined as molecular, cellular and/or interstitial changes that appear clinically as alterations in the size, mass, geometry, and function of the heart after injury [8]. Incidentally, four-tiered (normal geometry, concentric remodeling, eccentric hypertrophy, and concentric hypertrophy) classification of LV remodeling by relative LV wall thickness and LV mass can be used to assess risk for cardiovascular events in high-risk patients [9]. However, the relationship between exposure to tobacco smoke and the structure and function of the heart is not well understood.
The main aim of this study was to determine the association between parental smoking in childhood and cardiac structure and function in adulthood, and whether hygienic smoking modifies this effect. We used data from the Cardiovascular Risk in Young Finns Study, a population-based sample of individuals followed from childhood to adulthood for up to 31 years. We hypothesized that those exposed to parental smoking in childhood/adolescence, especially to poor parental smoking hygiene, have worse cardiac structure and function in adulthood than those not exposed to parental smoking.
Methods
The Cardiovascular Risk in Young Finns Study is an ongoing longitudinal population-based study of cardiovascular risk factors from childhood to adulthood, conducted in five university hospitals in Finland (Helsinki, Kuopio, Oulu, Tampere, and Turku) and their rural surrounds. The baseline study was conducted in 1980 when 3596 randomly selected children and adolescents aged 3, 6, 9, 12, 15, and 18 years participated. Since 1980 the cohort has been regularly followed up in 3- to 9-year intervals. A detailed description of the cohort has been published previously [10]. Participants or their parents provided written informed consent and the study was approved by local ethics committees. Participants who had a) full data on parental smoking from 1980 or 1983 and serum cotinine levels from 1980 (n = 1678), and b) echocardiography performed in 2011 (n = 1491) were included in this study (n = 1250). Supplementary Figure 1 provides a flow chart of the participants and non-participants.
Information on parental smoking was collected from self-report questionnaires in 1980 and 1983. Parents who indicated that they or their partner had ever smoked daily for at least 1 year were classified as “at least one smoking parent” in 1980 or 1983 as this has been shown to associate with measured serum cotinine in child offspring [7].
Fasting serum samples of the participants were collected in 1980 and stored at −20°C without thawing and analyzed in 2014. A total of 1999 participants had cotinine analyzed from childhood/adolescence. Serum cotinine concentration was quantified using two methods (described in Supplemental material). For statistical analyses, participants were divided into (a) low (0–0.99 ng/mL, n = 1448) and (b) elevated (⩾1 to <3 ng/mL, n = 232) serum cotinine groups [11].
We generated a variable of parental smoking hygiene that combined self-reports of parental smoking data from the baseline survey in 1980 with serum cotinine measurements [10]. Parental smoking hygiene was categorized as (a) no parental smoking: children with non-smoking parents and low serum cotinine levels (29.4%); (b) hygienic parental smoking: children with at least one smoking parent and low serum cotinine levels (58.2%); and (c) non-hygienic parental smoking: children with at least one smoking parent and a serum cotinine level ⩾1 and <3 ng/mL (12.4%) [11]. Twenty-seven participants were excluded from analyses because they had measurable cotinine levels without self-reported parental smoking and three participants were excluded because they were missing information on parental smoking (Supplementary Figure 1). Participants with cotinine levels of ⩾3 ng/ml were excluded from the analyses as they were assumed to be active smokers [12].
Covariates included questionnaire and anthropometric measures. Questionnaire measures gathered at baseline included childhood physical activity index, fruit and vegetable consumption, and parent self-report of family annual income, which was considered to be an indicator of socioeconomic status (SES) and categorized as (a) very low (<18,000 euros/year), (b) low (18,000–28,000 euros/year), (c) intermediate (28,001–38,000 euros/year), and (d) high (>38,000 euros/year) income groups [11]. In the case of missing information in 1980, data from the first follow-up in 1983 was used. Adolescent smoking status was defined from baseline (1980) or the first follow-up (1983). Participants aged under 12 years were considered non-smokers. The physical activity index was calculated at baseline and, due to separate questionnaires being used for children (3–6 years of age) and older children (9–18 years of age), the values were standardized as previously described [13,14]. Participants’ annual household income in the year 2011 was considered to be an indicator of SES in adulthood and was categorized as (a) low (<21,680 euros/year), (b) intermediate (21,680–48,770 euros/year), and (c) high (>48,770 euros/year) income groups. In the case of missing information in 2011, data from the previous follow-up in 2007 was used. Adult participants’ current daily smoking status was gathered at the year 2011 follow-up.
At baseline and all follow-up visits, weight was measured without shoes in light clothes with a digital Seca weighing scale to nearest kilogram. A Seca stadiometer was used for height measurements, and body mass index (BMI) was calculated as weight (kg) divided by height in meters squared. Baseline (1980) measurement was used as the primary indicator of childhood/adolescent BMI. In the case of missing information, data from year 1983 follow-up was used. Adulthood BMI was derived from the latest follow-up study (2011). In the case of missing information, data from the 2007 follow-up was used.
Fasting lipids were measured in the same laboratory at each follow-up with standard methods for serum total cholesterol, high-density lipoprotein (HDL) cholesterol, and triglycerides. Low-density lipoprotein (LDL) cholesterol was calculated using the Friedewald equation.
Brachial artery blood pressure was measured at baseline using an ultrasound device (Arteriosonde 1020, Roche) for participants aged 3 years, and using a standard mercury sphygmomanometer for participants aged ⩾6 years at baseline. In the case of missing information, data from the 1983 follow-up was used. Adult blood pressure measurements were collected in the 2011 follow-up using a random zero sphygmomanometer (Hawksley & Sons Ltd, Lancin, UK). All measurements were taken using a standardized method repeated three times on the right arm after the participant had been seated for 5 min with the average of the three measurements used.
Echocardiographic examinations were performed in 2011 according to American and European guidelines [15,16]. Transthoracic echocardiography was performed using a 3.5 MHz scanning frequency phased-array transducer (Sequoia 512, Acuson, CA, USA). Studies were saved in digital images, which were all analyzed using the ComPACS 10.7.8 (MediMatic Solutions, Genova, Italy) analysis program by one reader blinded to subjects’ details [17].
LV mass was calculated as previously described [18] and indexed LV mass was attained according to participant height using the allometric power of 2.7 (indexed LV mass = LV mass/height2.7) since this indexation has been shown to perform better among those with obesity [19]. Relative wall thickness was calculated and LV geometry groups were defined using sex-specific cut-off points [18]. Concentric remodeling was defined as high relative wall thickness (>85 percentile points) without LV hypertrophy. Eccentric hypertrophy was defined as LV hypertrophy without high relative wall thickness. Concentric hypertrophy was defined as LV hypertrophy with relative wall thickness. Intraclass correlation coefficients at the 5th and 95th percentile confidence intervals and coefficient of variance were reported earlier along with the complete methodology for cardiac imaging and image analysis in the young Finns study [17]. Interventricular septal wall thickness and LV posterior wall thickness were measured from the parasternal long-axis view in M-mode at end-diastole. LV diameter was measured from the parasternal long-axis view in M-mode at the end-diastole. LV ejection fraction and ratios of E/e’ and E/A were calculated according to American and European guidelines [15,16]. Left atrium volume index was calculated in four-chamber apical view at end-systole and divided by body surface area using the Du Bois formula (BSA = 0.007184 × weight0.425 × height0.725).
Baseline characteristics of the study population are reported as mean (SD) or median (25th and 75th percentiles, if skewed distributions) for continuous variables or as proportions for categorical variables.
For confirmation of participants’ parental smoking exposure, we combined cotinine levels with questionnaire data for the parental smoking hygiene variable. The relationship between parental smoking and serum cotinine measurements with continuous outcome variables was assessed using least squares means in a generalized linear model adjusted with Tukey–Kramer approximation, and for categorical outcome variables using logistic regression. Two models were created for the analyses: Model 1 was adjusted for sex and age; Model 2 included Model 1 covariates and was additionally adjusted for childhood risk factors (systolic blood pressure, HDL cholesterol, LDL cholesterol, triglycerides, BMI, and family SES) and adult risk factors (BMI, systolic and diastolic blood pressure, HDL cholesterol, LDL cholesterol and triglycerides, participants’ own SES and own smoking status). We performed sensitivity analyses that excluded participants who reported they were current smokers in adolescence and that additionally adjusted for daily fruit and vegetable consumption and physical activity index in addition to those covariates included in Model 2. We also conducted sensitivity analyses categorizing subgroups by age: (a) ⩽6 years of age at baseline; (b) 15–18 years of age at baseline. Moreover, we analyzed the data adjusted for age, sex, and adulthood alcohol intake (the results of which are shown in Supplemental material). To assess the degree of multicollinearity in the multivariable analyses, we investigated variance inflation factors and found no highly collinear relationships (variance inflation factor always <2.9). All statistical analyses were performed using SAS version 9.4 and statistical significance was inferred at a two-tailed p-value <0.05.
Results
The baseline characteristics of participants are shown in Table I. The total number of participants who had serum cotinine levels and parental smoking data available in childhood was 1987 (54.2% female). Of these, 1491 had at least one echocardiography measurement available. The mean age of participants was 42.2 years at the 2011 follow-up.
Baseline (1980) and follow-up (2011) characteristics of study participants.
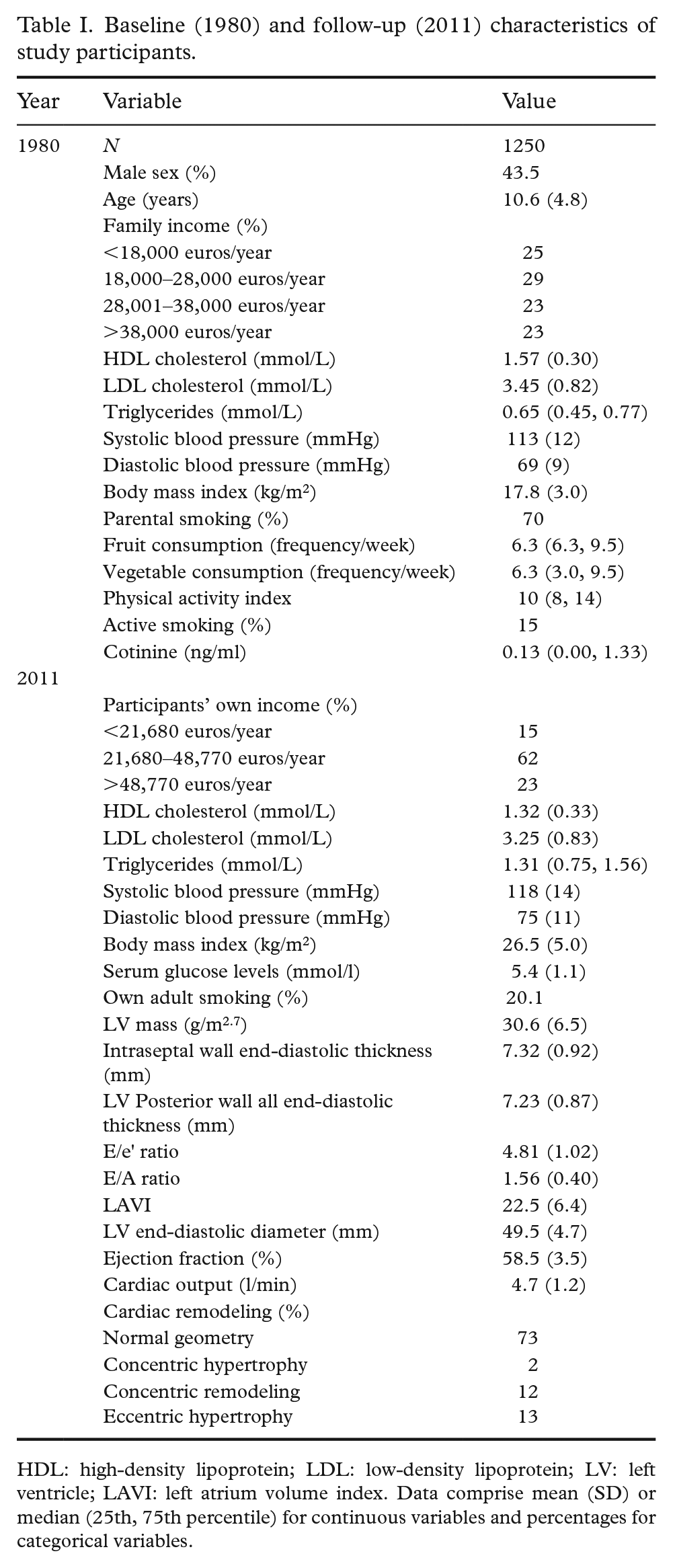
HDL: high-density lipoprotein; LDL: low-density lipoprotein; LV: left ventricle; LAVI: left atrium volume index. Data comprise mean (SD) or median (25th, 75th percentile) for continuous variables and percentages for categorical variables.
Associations between parental smoking with cardiac outcomes are shown in Table II. We observed associations between parental smoking status with interventricular septal wall thickness (adjusted means, 95% confidence interval (CI): 7.29 (7.20–7.37) among participants with non-smoking parents vs. 7.39 (7.34–7.45) among participants with smoking parents, p = 0.04); and with LV posterior wall thickness (adjusted means + 95%CI: 7.22 (7.15–7.30) vs. 7.32 (7.26–7.37), p = 0.045), but these were attenuated when further adjusted for childhood and adulthood covariates (adjusted means + 95%CI: 7.40 (7.29–7.51) vs. 7.48 (7.39–7.57), p = 0.13; and 7.32 (7.22–7.42) vs. 7.37 (7.29–7.45), p = 0.27, respectively). We observed no significant differences in the other echocardiography outcomes examined.
Effect estimates and their 95% confidence intervals (CIs) for adult outcome variables according to parental smoking status.
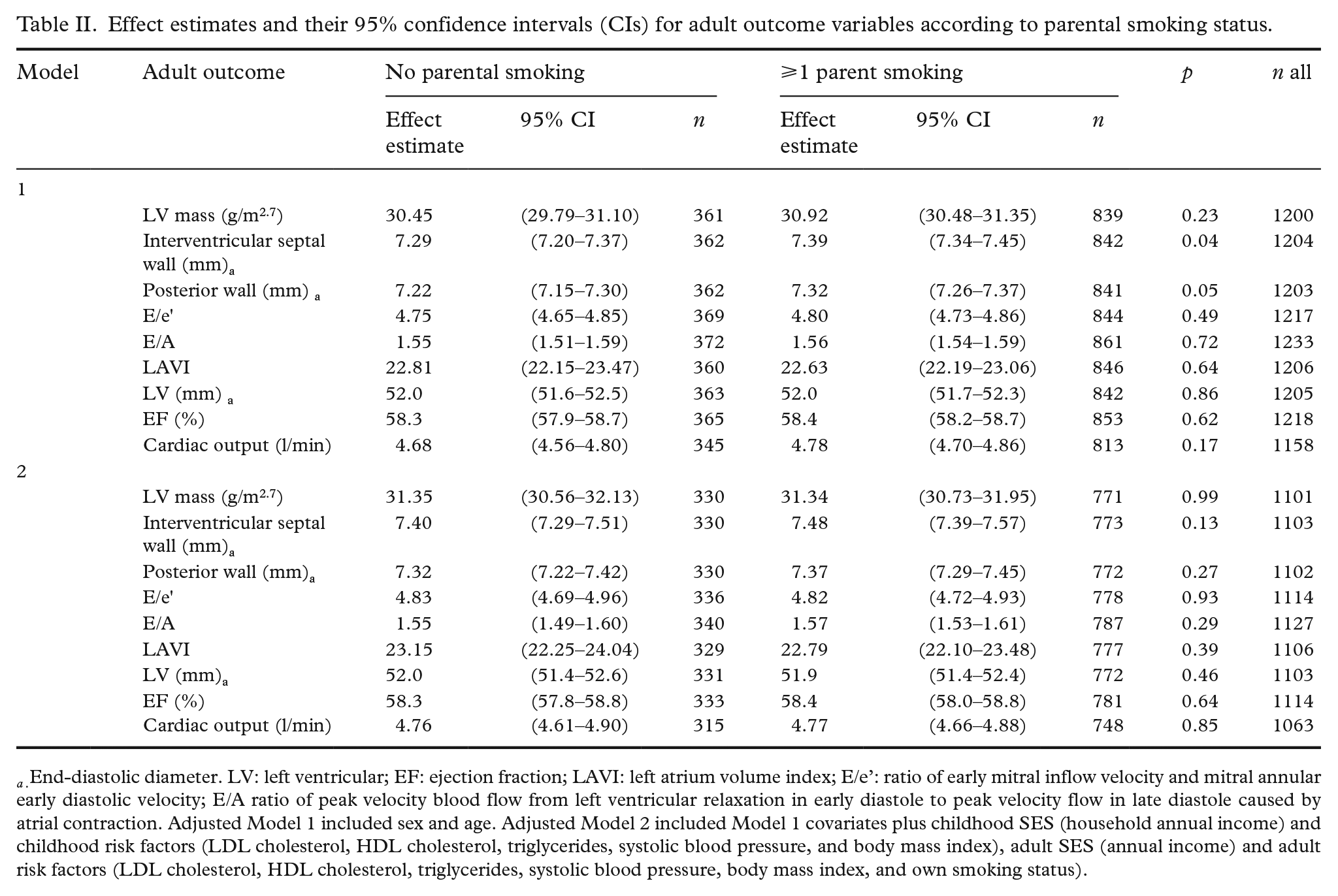
a . End-diastolic diameter. LV: left ventricular; EF: ejection fraction; LAVI: left atrium volume index; E/e’: ratio of early mitral inflow velocity and mitral annular early diastolic velocity; E/A ratio of peak velocity blood flow from left ventricular relaxation in early diastole to peak velocity flow in late diastole caused by atrial contraction. Adjusted Model 1 included sex and age. Adjusted Model 2 included Model 1 covariates plus childhood SES (household annual income) and childhood risk factors (LDL cholesterol, HDL cholesterol, triglycerides, systolic blood pressure, and body mass index), adult SES (annual income) and adult risk factors (LDL cholesterol, HDL cholesterol, triglycerides, systolic blood pressure, body mass index, and own smoking status).
Associations between parental smoking hygiene and cardiac outcomes are shown in Table III. We observed no significant differences in adult echocardiography variables according to parental smoking hygiene (ps > 0.10).
Effect estimates and their 95% confidence intervals (CIs) for adult outcome variables according to parental smoking hygiene.
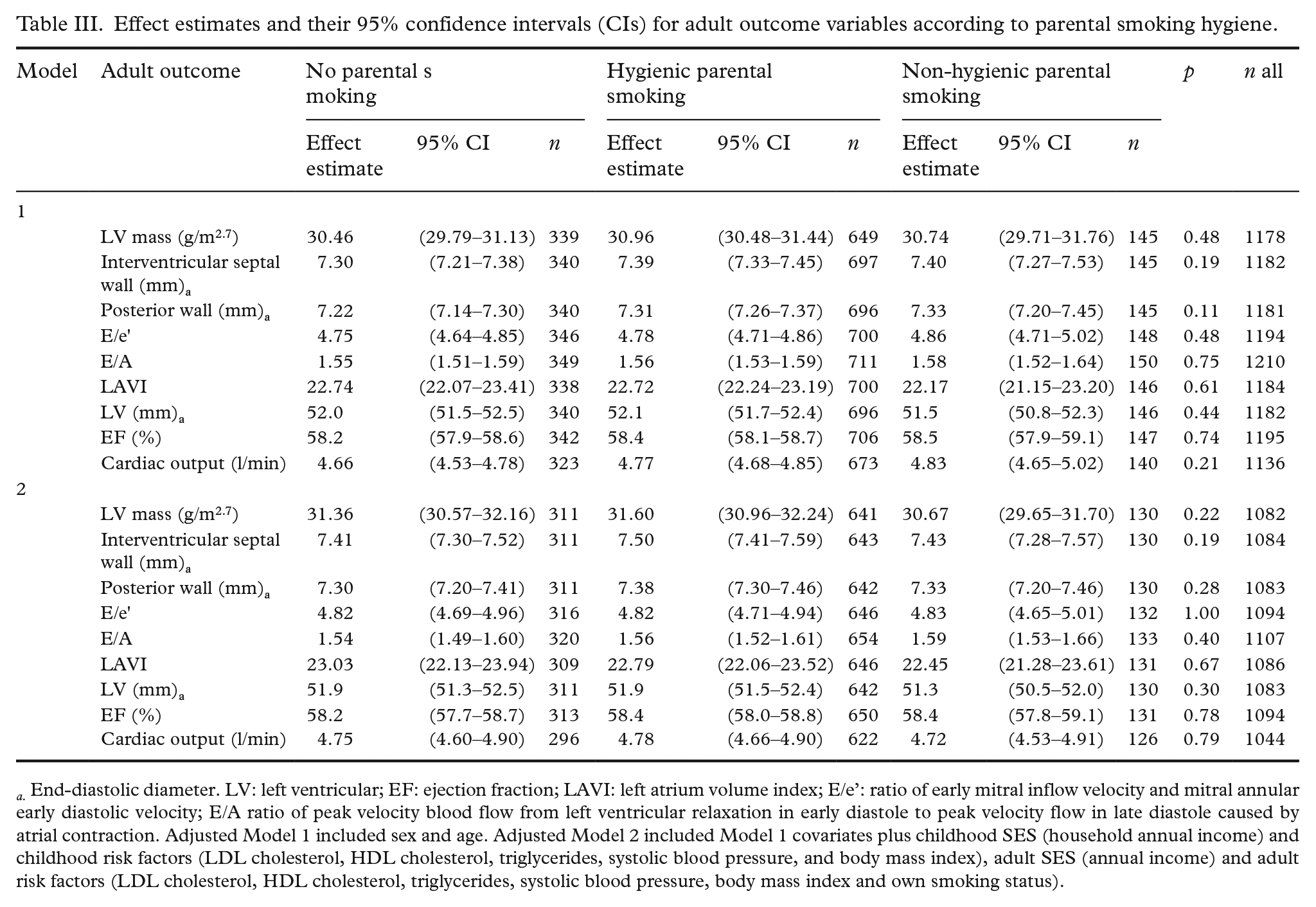
a. End-diastolic diameter. LV: left ventricular; EF: ejection fraction; LAVI: left atrium volume index; E/e’: ratio of early mitral inflow velocity and mitral annular early diastolic velocity; E/A ratio of peak velocity blood flow from left ventricular relaxation in early diastole to peak velocity flow in late diastole caused by atrial contraction. Adjusted Model 1 included sex and age. Adjusted Model 2 included Model 1 covariates plus childhood SES (household annual income) and childhood risk factors (LDL cholesterol, HDL cholesterol, triglycerides, systolic blood pressure, and body mass index), adult SES (annual income) and adult risk factors (LDL cholesterol, HDL cholesterol, triglycerides, systolic blood pressure, body mass index and own smoking status).
ORs between parental smoking, childhood serum cotinine levels, and parental smoking hygiene with cardiac remodeling are shown in Table IV. In analyses adjusted for age and sex, participants exposed to parental smoking (OR 1.90, 95%CI 1.23–2.92), hygienic parental smoking (OR 1.74, 95%CI 1.12–2.71), and non-hygienic parental smoking (OR 1.88, 95%CI 1.02–3.45) had higher odds of concentric remodeling. The association between parental smoking and concentric remodeling persisted with adjustment for child and adult covariates (OR 1.72, 95%CI 1.09–2.70).
Adjusted odds ratio (OR) and 95% confidence intervals (CIs) for adult cardiac remodeling according to different measures of child exposure to passive smoking.
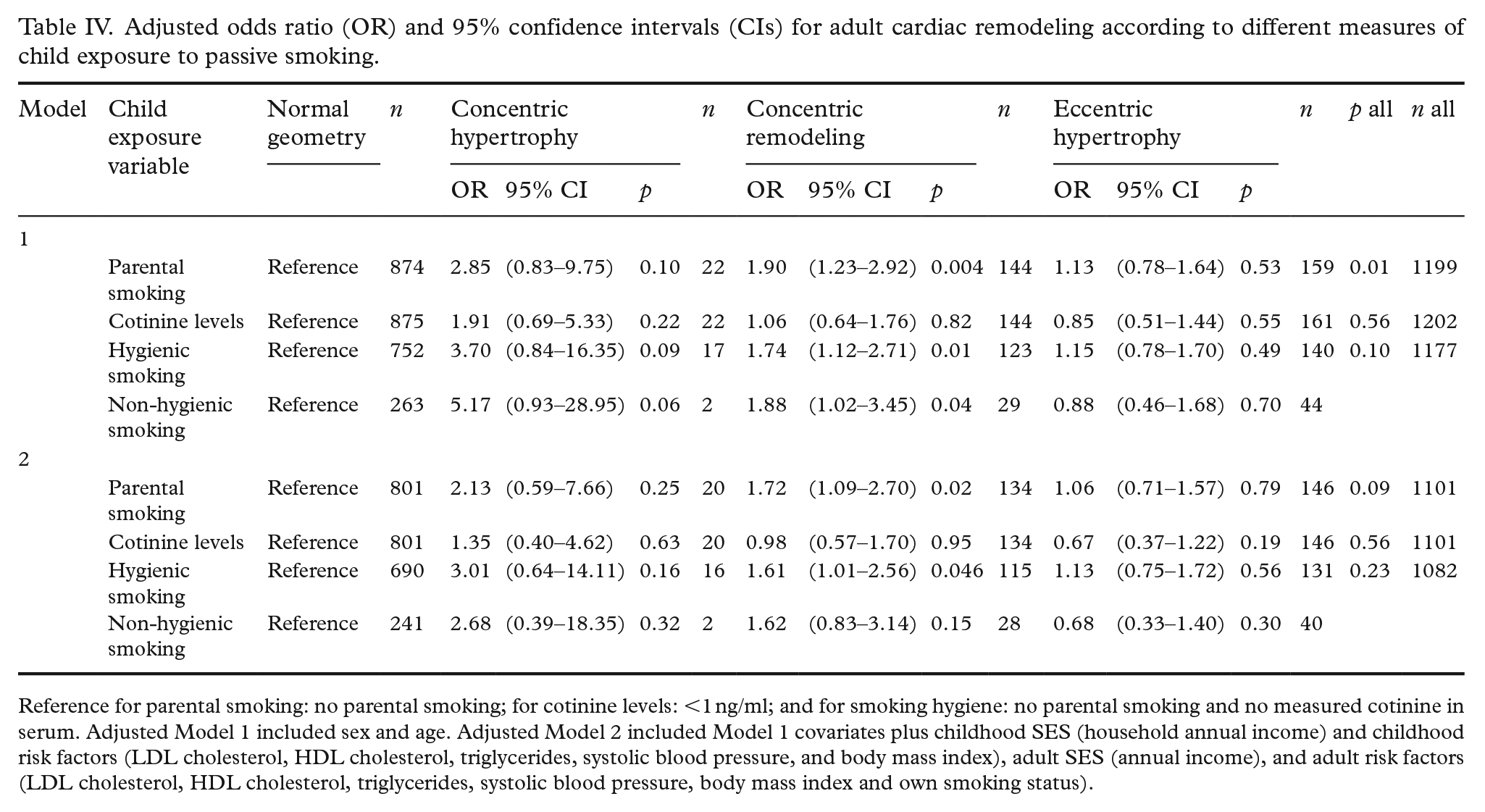
Reference for parental smoking: no parental smoking; for cotinine levels: <1 ng/ml; and for smoking hygiene: no parental smoking and no measured cotinine in serum. Adjusted Model 1 included sex and age. Adjusted Model 2 included Model 1 covariates plus childhood SES (household annual income) and childhood risk factors (LDL cholesterol, HDL cholesterol, triglycerides, systolic blood pressure, and body mass index), adult SES (annual income), and adult risk factors (LDL cholesterol, HDL cholesterol, triglycerides, systolic blood pressure, body mass index and own smoking status).
The association between hygienic parental smoking and concentric remodeling remained following adjustment for childhood and adulthood risk factors (OR 1.61, 95%CI 1.01–2.56) whereas the association between non-hygienic parental smoking and concentric remodeling became non-significant when adjusted for child- and adult covariates.
The association between hygienic parental smoking and concentric hypertrophy was not statistically significant.
The evidence for associations between non-hygienic parental smoking and concentric hypertrophy were limited (n = 2) in analyses adjusted for sex and age (OR 5.17, 95%CI 0.93–28.95) but additional adjustments for risk factors attenuated the association.
We observed no other associations between parental smoking or parental smoking hygiene and concentric or eccentric hypertrophy.
The association between childhood serum cotinine levels and cardiac outcomes are shown in Supplementary Table I. We observed no significant differences in adult echocardiography variables according to childhood serum cotinine level.
In sensitivity analyses, active smokers in adolescence were excluded and additional adjustments were made for daily fruit and vegetable consumption and for childhood physical activity index with the results remaining essentially similar except the association between non-hygienic parental smoking and concentric remodeling (OR 1.78, 95%CI 0.92–3.45) and concentric hypertrophy was diluted. We also conducted sensitivity analyses categorizing subgroups by age: (a) ⩽6 years of age at baseline; and (b) 15–18 years of age at baseline. The overall results in the fully adjusted models were consistent with those shown for the main results. Finally, we analyzed the data adjusted for age, sex, and adulthood risk factors and these were in line with our reported findings and did not change the results.
Discussion
We hypothesized that parental smoking would be associated with adverse structural and functional cardiac changes, but we found no strong evidence of this. However, we observed an association between parental smoking in childhood with interventricular septal and LV posterior wall thickness in adulthood. Furthermore, parental smoking and smoking hygiene status were associated with concentric remodeling in adults.
To our knowledge this is the only longitudinal prospective study examining the association between exposure to parental smoking in childhood with the use of an objectively measured biomarker (serum cotinine) and echocardiography measurements performed in adulthood.
While increased LV mass, a predecessor to cardiac remodeling, has been shown to associate with smoking in midlife [20] and to exposure to secondhand smoke in rabbits [21], our results did not confirm this association. However, parental smoking was associated with increased thickness of the interventricular septal and LV posterior walls, which may be predictive of future LV hypertrophy. As our participants were relatively young, the association with LV hypertrophy and function might become evident as they age. In line with previous studies, after adjusting for SES the association was diluted [22]. In addition, it is known that exposure to secondhand smoking in childhood can result in higher blood pressure [23], increased arterial stiffness [24], flow-mediated dilation impairment [4], and endothelial dysfunction, among other conditions [25]. Combined, these factors might cause cardiac remodeling later in life by pressure overload [26].
Previous studies have found LV remodeling and higher age to associate with worse LV diastolic function, which is often the first stage of heart failure [27]. Also, cardiovascular morbidity is higher among patients with concentric remodeling than in patients with normal geometry [28]. In our study, concentric remodeling (high relative wall thickness without LV hypertrophy) was associated with exposure to parental smoking and parental smoking hygiene.
Although both active smoking and exposure to chronic secondhand smoking in childhood have been linked to decreased LV diastolic function in Hispanics/Latinos [3], we found no association regarding parental smoking, serum cotinine levels, or parental smoking hygiene status in our cohort. Potentially, this difference could be due to the younger age of our participants (42 vs. 56 years of age).
While smoking is associated with a higher risk of heart failure, a recent study found chronic exposure to secondhand smoking in childhood paradoxically increased LV ejection fraction [3]. Nevertheless, in our relatively young cohort, we found no association between parental smoking and LV ejection fraction or cardiac output.
The main strength of this study is its large study population and comprehensive data comprising lifestyle, biochemistry, and anthropometric measurements as well as socioeconomic information starting in childhood with over 30 years of follow-up. An apparent limitation of observational studies is that they are not able to establish causality; however, it would be impossible to achieve a lifelong trial on CVD progression in humans. Additional limitations include the lack of data on prenatal parental smoking exposure, which has been shown to have long-lasting adverse effects on cardiovascular health. Furthermore, we were not able to evaluate changes in smoking hygiene over time and some of the parents stopped regular smoking during their offspring’s childhood which could have diluted our findings. Moreover, young adults in the 1980s might have been exposed to secondhand smoke outside of their family unit. However, we tried to minimize this effect by using cotinine levels in our study and excluding participants who had inconsistencies between questionnaire data and cotinine levels. Therefore, the contrast between the results for serum cotinine and queried parental smoking suggests an underestimation of smoking among the parents. In addition, we were not able to consider snus (smokeless tobacco product) use in childhood as a potential confounder, as data was not collected. Nevertheless, use of snus in youth in the 1980s was rare in Finland and became more common among males only during the 1990s – though the rates remained relatively low (daily use of snus ~3% in boys aged 16–18 years and <1% in girls and women) [29]. Thus, use of snus is not likely to affect our results significantly. In addition, individual variability in the rate of elimination of nicotine and cotinine could not be evaluated from our data, which should be borne in mind when interpreting our results. Loss to follow-up through attrition at follow-up is inevitable in longitudinal studies. However, participation rates were reasonably high, and participation has been dynamic, so that many participants lost to follow-up early in the study returned at subsequent follow-ups [30]. Thus, the study population is likely to be representative of the original population [10,31]. Moreover, we understand that additional adjustments for Model 2 might have included factors that were perhaps mediating at least part of the association between exposure and outcomes. We have previously reported baseline risk factor levels to be essentially similar among participants and non-participants at subsequent follow-ups [32]. Furthermore, it is not possible to study the association of exposure to parental smoking with CVD as it develops gradually over decades and participants of the young Finns study were in their early midlife. Hence, none of the participants met the clinical criteria of concentric hypertrophy. However, the cut-offs used allowed us to investigate associations between parental smoking and cardiac remodeling. We acknowledge that strict measures have been implemented on tobacco control in most developed countries since our cohort commenced in 1980, which has led to a substantial decline in daily smokers that might limit the generalizability of our findings. Public awareness of the adverse effects of secondhand smoking has subsequently increased but remains a problem in disadvantaged groups and among those in low- and middle-income countries. Finally, our study was composed of an ethnically homogenous group of white participants, which limits the generalizability of our results.
Conclusions
We found that exposure to parental smoking in childhood was associated with higher odds of concentric remodeling in later life and may also influence left ventricular wall diameter. Although these findings need to be replicated in other independent cohorts, we found exposure to parental smoking was not associated with a higher likelihood of systolic or diastolic dysfunction in this relatively young study population.
Supplemental Material
sj-docx-1-sjp-10.1177_14034948221119611 – Supplemental material for Exposure to parental smoking and cardiac structure and function in adulthood: the Cardiovascular Risk in Young Finns Study
Supplemental material, sj-docx-1-sjp-10.1177_14034948221119611 for Exposure to parental smoking and cardiac structure and function in adulthood: the Cardiovascular Risk in Young Finns Study by Jukka Pihlman, Joel Nuotio, Suvi Rovio, Katja Pahkala, Saku Ruohonen, Eero Jokinen, Tomi P. Laitinen, David P. Burgner, Nina Hutri-Kähönen, Päivi Tossavainen, Leena Taittonen, Mika Kähönen, Jorma S.A. Viikari, Olli T. Raitakari, Costan G. Magnussen and Markus Juonala in Scandinavian Journal of Public Health
Supplemental Material
sj-docx-2-sjp-10.1177_14034948221119611 – Supplemental material for Exposure to parental smoking and cardiac structure and function in adulthood: the Cardiovascular Risk in Young Finns Study
Supplemental material, sj-docx-2-sjp-10.1177_14034948221119611 for Exposure to parental smoking and cardiac structure and function in adulthood: the Cardiovascular Risk in Young Finns Study by Jukka Pihlman, Joel Nuotio, Suvi Rovio, Katja Pahkala, Saku Ruohonen, Eero Jokinen, Tomi P. Laitinen, David P. Burgner, Nina Hutri-Kähönen, Päivi Tossavainen, Leena Taittonen, Mika Kähönen, Jorma S.A. Viikari, Olli T. Raitakari, Costan G. Magnussen and Markus Juonala in Scandinavian Journal of Public Health
Supplemental Material
sj-docx-3-sjp-10.1177_14034948221119611 – Supplemental material for Exposure to parental smoking and cardiac structure and function in adulthood: the Cardiovascular Risk in Young Finns Study
Supplemental material, sj-docx-3-sjp-10.1177_14034948221119611 for Exposure to parental smoking and cardiac structure and function in adulthood: the Cardiovascular Risk in Young Finns Study by Jukka Pihlman, Joel Nuotio, Suvi Rovio, Katja Pahkala, Saku Ruohonen, Eero Jokinen, Tomi P. Laitinen, David P. Burgner, Nina Hutri-Kähönen, Päivi Tossavainen, Leena Taittonen, Mika Kähönen, Jorma S.A. Viikari, Olli T. Raitakari, Costan G. Magnussen and Markus Juonala in Scandinavian Journal of Public Health
Supplemental Material
sj-docx-4-sjp-10.1177_14034948221119611 – Supplemental material for Exposure to parental smoking and cardiac structure and function in adulthood: the Cardiovascular Risk in Young Finns Study
Supplemental material, sj-docx-4-sjp-10.1177_14034948221119611 for Exposure to parental smoking and cardiac structure and function in adulthood: the Cardiovascular Risk in Young Finns Study by Jukka Pihlman, Joel Nuotio, Suvi Rovio, Katja Pahkala, Saku Ruohonen, Eero Jokinen, Tomi P. Laitinen, David P. Burgner, Nina Hutri-Kähönen, Päivi Tossavainen, Leena Taittonen, Mika Kähönen, Jorma S.A. Viikari, Olli T. Raitakari, Costan G. Magnussen and Markus Juonala in Scandinavian Journal of Public Health
Footnotes
Acknowledgements
The authors wish to thank Noora Kartiosuo from the Research Centre of Applied and Preventive Cardiovascular Medicine, University of Turku, for statistical advice.
Author contributions
The authors confirm contribution to the paper as follows: JP had full access to all the data in the study and takes responsibility for its integrity and the accuracy of the data analysis; project conceptualization: all authors; investigation: JP; analysis: JP; software: JP; data curation: JP; validation: JP, JSAV, OTR, MJ; methodology: JP, MJ; writing, review, and editing: all authors; funding acquisition: KP, EJ, TPL, NH-K, PT, LT, MK, JSAV, OTR, MJ; supervision: JN, CGM, MJ; project administration: KP, EJ, TPL, NH-K, PT, LT, MK, JSAV, OTR, MJ.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The Young Finns Study has been financially supported by the Academy of Finland: grants 286284, 134309 (Eye), 126925, 121584, 124282, 129378 (Salve), 117787 (Gondi), and 41071 (Skidi); the Social Insurance Institution of Finland; Competitive State Research Financing of the Expert Responsibility area of Kuopio, Tampere and Turku University Hospitals (grant X51001); Juho Vainion Foundation; Paavo Nurmi Foundation; Finnish Foundation for Cardiovascular Research; Finnish Cultural Foundation; The Sigrid Julius Foundation; Tampere Tuberculosis Foundation; Emil Aaltonen Foundation; Yrjö Jansson Foundation; Signe and Ane Gyllenberg Foundation; Diabetes Research Foundation of Finnish Diabetes Association; and EU Horizon 2020 (grant 755320 for TAXINOMISIS); and European Research Council (grant 742927 for MULTIEPIGEN project); Tampere University Hospital Supporting Foundation. CGM is supported by a National Heart Foundation of Australia Future Leader Fellowship (100849).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
