Abstract
Background
Tau pathology begins to accumulate in the medial temporal lobe in many individuals in middle age. Some individuals develop amyloid pathology, more advanced tau pathology (Braak stage), and higher levels of CA1 hippocampal tau pathology in the context of Alzheimer's disease. Others never develop significant amyloid pathology; in these cases, hippocampal tau pathology can be CA2-predominant and tends not to accumulate past intermediate stages. But factors associated with the early formation of these tau patterns is unclear.
Objective
The objective of this study is to examine demographic, genetic (APOE), and pathologic factors associated with the emergence of hippocampal tau pathology patterns in middle age.
Methods
We identified 89 individuals with hippocampal tau pathology ages 65 and younger. These cases were assessed for tau stage, amyloid plaques, and APOE4 status. Hippocampal tau pathology in CA1 and CA2 was scored semi-quantitatively.
Results
In a younger aging population, tau pathology initially deposits at low levels in CA1 and CA2. In the presence of amyloid, tau pathology is increased in both CA1 and CA2, but with a shift toward a greater proportion of cases with higher tau pathology in CA1.
Conclusions
Patterns of tau pathology in middle age are strongly associated with amyloid plaques, including increased CA1 and CA2 pathology and a greater proportion of CA1-predominant cases. In the absence of amyloid, tau pathology shows a normal (Gaussian) distribution between CA1 and CA2. These tau pathology distributions at younger ages are similar to the patterns seen at older ages in AD and PART.
Introduction
The accumulation of tau pathology is universal in aging, with all individuals over age 80 showing tau pathology at autopsy. 1 However, many individuals accumulate tau in the brain at much younger ages, with more than half of individuals showing early mesial temporal lobe tau pathology in their 30s and 40s. 2 At some point during the aging process, individuals diverge in their patterns of pathology in the brain. Many develop amyloid plaques over time which, in combination with tau pathology, are diagnostic of Alzheimer's disease (AD) neuropathologic changes. Others age in the absence of significant amyloid plaques and the brain shows primary age-related tauopathy (PART) at autopsy.
The relationship between the tau pathology in AD and PART remains ambiguous. The two conditions share many features, including the same tau aggregate structure and composition,1,3 the same transcriptional signature associated with tau aggregates, 4 evidence of synaptic loss with high tau pathology,5,6 and cognitive decline associated with higher levels of tau pathology.7–11 However, the tau pathology differs in two primary ways between AD and PART. While AD tau pathology frequently extends across the entire neocortex, tau pathology in PART in many cases remains more localized to the temporal lobe. 1 AD and PART also may show differential preferences for hippocampal subregions, though this remains debated in the field.12,13 While AD tau pathology tends to be CA1 predominant, PART tau pathology may show a predilection for CA2.11,14–16 These patterns have largely been established at older age groups, which is necessary to define their association with cognitive decline. But how these tau pathology patterns emerge and what is associated with differential tau accumulation in pre-symptomatic middle age is less clear.
To examine patterns of tau pathology during early tau aggregation in the mesial temporal lobe, we identified 89 cases of individuals who had some evidence of hippocampal tau pathology at ages 65 and younger. Using semi-quantitative scores for tau pathology in hippocampal CA1 and CA2, we quantified the early distribution of tau pathology in the hippocampus and examined the effects of demographic and AD-associated factors (amyloid, APOE4) on the absolute and relative distributions of tau pathology in middle age.
Methods
Human tissue
We reviewed records from autopsy cases age 65 years and younger with no known clinical neurodegenerative diagnosis from the Johns Hopkins autopsy service and Johns Hopkins Brain Resource Center, a subset of which have been published previously. 17 Cases in all cohorts were sampled according to standard protocols with similar tissue sections obtained from each. We reviewed records from 704 cases of individuals 65 years of age and younger, from which we identified 208 cases which had some note of mesial temporal lobe tau deposition and slides available for review. Cases noted to be entirely negative for tau were not selected for review. Of these 208 cases, 89 cases had tau immunostaining, good anatomic representation of CA1 and CA2 on the section, and had accumulation of tau pathology in at least one neuronal cell body of either CA1 or CA2 (Table 1). Co-pathology rates in this cohort were low, with only 2 of 89 cases (2.2%) showing non-Alzheimer's and non-aging related pathology. There was one case with Lewy body disease in the brainstem and one case of an early non AD-tauopathy of uncertain diagnosis. Notably, neither of the two cases with co-pathology were CA2-predominant.
Early hippocampal pathology cohort.

Values are either mean (range) or N (percentage).
Amyloid data available for 86 cases.
Braak 0 cases show pre-tangles in the hippocampus, corresponding to modified stage Braak 1b. 2 Complete Braak staging available for 74 cases.
F: female; M: male.
The Mai-Paxinos atlas 18 used to established regional boundaries. For previously published cases, immunostains for amyloid and tau, and modified Bielschowsky silver stains were reviewed. 17 Braak staging was performed according to established protocols19,20 using sections of medial temporal lobe (hippocampus, entorhinal cortex), frontal cortex, temporal cortex, and occipital cortex. Amyloid assessment was done several ways: first using amyloid immunostaining and silver stains to assess for presence or absence of amyloid plaques and for Thal staging per established protocols 21 using cortex (frontal, temporal, and/or occipital), entorhinal cortex/hippocampus, basal ganglia, midbrain, and cerebellum. If staging could not be determined from the slides and tissue available, the case was not staged for the analysis. Second, the density of diffuse amyloid plaques was scored in the hippocampus as none, sparse, moderate, or frequent. Finally, silver or tau stains in the medial temporal lobe (entorhinal cortex, hippocampus, temporo-occipital gyrus) were used to determine whether neuritic plaques were present and, if present, neuritic plaques were also scored as none, sparse, moderate, or frequent using CERAD scoring19,22 (Supplemental Table 1). Additional immunostains were performed as necessary for amyloid and tau evaluation as below. All quantitative and scoring was performed by a single reviewer who was blinded to other pathologic and demographic information. All work in this study was acknowledged by the Johns Hopkins Institutional Review Board as not human subjects research.
Immunohistochemistry
Formalin-fixed paraffin-embedded tissue was sectioned at 10 µm, incubated at 60°C for 30 min, then immunostained using an autostainer or manually depending on the number of sections processed. Automated staining was performed using the EprediaTM Lab Vision™ Autostainer 360-2D (ThermoFisher Scientific, Waltham, MA, USA) according to manufacturer protocols. Antigen retrieval and tissue rehydration were performed using the Lab Vision™ PT Module (ThermoFisher Scientific, Waltham, MA, USA) with Epredia™ Dewax and HIER Buffer M (ThermoFisher Scientific, Waltham, MA, USA). For amyloid staining, the slides were briefly removed, incubated for 5 min in 88% formic acid, rinsed in water, then returned to the autostainer to complete the protocol. For manual immunostaining, slides were deparaffinized with xylene, then rehydrated using 100% and 95% ethanol. For phospho-tau immunostaining, antigen retrieval was performed with citrate buffer (ab93678, Abcam, Waltham, MA, USA) in a pressure-based heat retrieval system (TintoRetriever, Bio SB Inc, Goleta, CA, USA) at 100˚C at low pressure for 10 min. For amyloid-stained slides, antigen retrieval was performed instead with 88% formic acid (F0507, Sigma Aldrich, St Louis, MO, USA) for 5 min. All slides underwent blocking for endogenous peroxidases using 3% hydrogen peroxide and protein blocking with 3% normal goat serum. Both automated and manual staining protocols used primary antibody AT8 for phospho-tau (automated 1:200, manual 1:1000; MN1020, ThermoFisher Scientific, Waltham, MA, USA) and 6E10 for amyloid (1:500, #803001, BioLegend, San Diego, CA, USA). Secondary labeling was done with biotinylated anti-mouse IgG (1:200, BA-2000-1.5, VectorLabs, Newark, CA, USA), then slides were incubated with ABC reagent (PK-4002, Vector Labs, Newark, CA, USA) and developed with 3,3'-diaminobenzidine (DAB; SK-4100, Vector Labs, Newark, CA, USA). After immunostaining, slides were rinsed with water, counterstained with hematoxylin 7221 (Richard-Allan Scientific, San Diego, CA, UAS), and coverslipped.
APOE genotyping
If not available from published cases or prior brain bank records, APOE genotyping was performed based on a modification of prior protocols. 23 If frozen tissue was available, DNA was extracted using the QIAamp DNA Mini Kit (#51304, Qiagen, Germantown, MD, USA). For cases in which only paraffin-embedded tissue was available, DNA was extracted from 10 micron scrolls using a truXTRC FFPE DNA microtube Kit (Covaris, Waltham, MA, USA) according to manufacturer protocols by the Johns Hopkins Genetic Resources Core Facility. DNA concentration was measured using NanoDropTM One (ThermoFisher Scientific, Waltham, MA, USA). PCR was performed using 100 ng of DNA, 0.2 µM of primers 1 and 2 (primer 1: 5′-ACAGAATTCGCCCCGGCCTGGTACACTGCCA-3′, primer 2: 5′-TCCAAGGAGCTGCAGGCGGCGCA-3′) and 2× Rapid Taq Master Mix (#P222, Vazyme, Nanjing, PRC). After denaturation at 95°C for 3 min, PCR was performed for 36 cycles of 95°C for 15 s, 69°C for 15 s and 72°C for 15 s, and a final extension at 72°C for 15 min. From each PCR reaction, approximately half the products were incubated with 5 units of AflIII in r3.1 buffer (#R0541L, New England Biolabs, Ipswich, MA, USA) and the other half with HaeIII in rCutSmart buffer (#R0107L, New England Biolabs, Ipswich, MA, USA) for at least 3 h at 37°C. The products were run on a 2.5% agarose gel in TBE buffer (#351-001-131, Quality Biological, Gaithersburg, MD, USA) with GelRed Nucleic Acid Stain (# SCT121, MilliporeSigma, Burlington, MA, USA). The presence of an APOE4 allele was confirmed by allele-specific PCR of 100 ng of genomic DNA using primer 1 (above) and primer 3 (5’-CGCGGACATGGAGGACGTTC-3’) in Taq Plus Master Mix II (#P212, Vazyme, Nanjing, PRC). PCR was performed as above, with the exception that the extension was performed at 63-67°C. A positive control was included in all runs of allele-specific PCR.
Semi-quantitative tau score
Tau pathology in CA1 and CA2 was scored on sections immunostained for phospho-tau using the Mai-Paxinos human brain atlas for hippocampal regional boundaries. 18 To quantify tau pathology at very low levels in a younger age range, a semi-quantitative scoring system was developed with more detailed scoring of low levels of pathology. Tau pathology was scored in the most affected field in each region at 100× magnification (10× objective), similar to the strategy implemented in the CERAD study 22 but with an expanded scale for assessment of lower levels of pathology. Cases were scored by the number of neurons positive for tau pathology (Supplemental Figure 1) as follows: 0, no tau-positive neurons; 1: 1 neuron; 2: 2–3 neurons; 3: 4–6 neurons; 4: 7–10 neurons; 5:11–15 neurons; 6: >15 neurons. All regions were scored by a single reviewer blinded to the pathologic and demographic information for each case.
Statistics
Data were analyzed in R (version 4.4.2) / RStudio (Posit, Boston, MA), using glm2 (version 1.2.1) and MASS (version 7.3–65).24,25 Braak stage was treated as an ordered factor variable. Amyloid was divided into three groups: no plaques, diffuse plaques only, neuritic and diffuse plaques. Amyloid was initially treated as an ordered factor variable, with neuritic plaques considered most severe. However, as plaques showed a linear relationship with Braak stage and CA1/CA2 tau scores, it was coded as a linear variable for all analyses as follows: no plaques- 0, diffuse plaques-1, neuritic plaques-2. Similarly, amyloid plaque density in the hippocampus was initially treated as an ordered factor variable, then treated as a linear variable when the ordered factor variable showed a linear relationship with Braak stage and CA1/CA2 tau scores (none = 0, sparse = 1, moderate = 2, frequent = 3). Tau score data were analyzed with Poisson generalized linear modeling, as appropriate for non-continuous positive integer data. Tau score models were initially performed with all variables in the model: Age, sex, race, and an interaction term with amyloid and APOE4. The models were then simplified to include only significant terms. Braak stage was analyzed using ordinal logistic regression with amyloid plaques alone, as appropriate for an ordered factor variable. Chi-Square testing was done on the expected versus observed tau pathology distributions and included all groups (CA1 > CA2, CA2 > CA1, CA1 = CA2). Race in the Chi-square analysis compared white and non-white groups. GraphPad Prism 9.4.1 (GraphPad Software, San Diego, CA) was used for all graphs, and for Chi-square, Fisher's exact, and Mann-Whitney tests, and for simple linear regression.
Results
To investigate early patterns of tau pathology in the hippocampus, we identified 89 cases with hippocampal tau pathology, either pre-tangles or mature neurofibrillary tangles (Table 1). The average age of cases with hippocampal tau pathology was 55.8 years (range: 29–65 years), and the cases were male-predominant (73% male, 65/89 cases). The group was 62.9% white and 34.8% black. APOE4 allele carriers represented 31.4% (28/89 cases) of the cohort and 39.5% (34/86 cases) were amyloid positive by immunohistochemistry. Consistent with prior studies, APOE4 carriers showed a higher rate of amyloid positivity (APOE4: 70.4% amyloid positive, No APOE4: 25.4% amyloid positive, p = 0.0001, Fisher's exact test). Plaques were generally low level (mean Thal stage 0.88, range 0–5). In this age group, only 10 cases (11%) had neuritic amyloid plaques which were sparse in most cases (Supplemental Table 1). Braak staging ranged from 0 (pre-tangles, modified Braak stage 1b) 2 to V, with a median score of II. Amyloid deposition was associated with higher Braak stages (odds ratio: 2.42, p = 0.048, ordinal logistic regression), consistent with prior reports. 2
Hippocampal tau pathology in CA1 and CA2 were scored using a semi-quantitative scoring system (Supplemental Figure 1). The average tau pathology scores in this young cohort were similar in CA1 and CA2 at 2.4 and 2.1, respectively, implying an average of less than 4 tau-positive neurons in a microscopic field for each region (Supplemental Figure 2). There was a wide range of tau pathology in this younger cohort, including 33 cases (37%) with a score of 0 in one of the two regions and 25 cases with a score of 5 or 6 in at least one of the two regions (28%) (Supplemental Figure 2). Using Poisson regression, we quantified the effects of demographic (race, sex, age) and AD-associated (amyloid, APOE4) variables on the severity of tau pathology in CA1 and CA2.
The effects of amyloid on hippocampal tau pathology were assessed first using the presence or absence of amyloid, then by assessing the correlation with the severity of amyloid pathology in two ways. First, cases were divided into categories based on the type of amyloid plaques in the medial temporal lobe: no amyloid deposition, diffuse plaques only, and both neuritic and diffuse plaques, with the neuritic plaque group considered most severe. Second, since diffuse plaque severity has been shown to correlate with tau pathology burden in the hippocampus in older individuals, we used a semi-quantitative score to assess the hippocampal diffuse plaque density. These two measures of amyloid deposition were highly correlated (amyloid density versus type of plaque, rate ratio 5.71, p < 1.06e-15, Poisson modeling), implying that individuals with more dense amyloid accumulation in the hippocampus at younger ages were also more likely to have neuritic amyloid plaque pathology. Both measures of amyloid pathology also correlated with Braak stage (density odds ratio: 1.56, p = 0.038; plaque type odds ratio: 2.15, p = 0.024, ordinal logistic regression), though plaque type was more highly correlated with Braak stage in this age group.
Amyloid pathology was consistently associated with increased tau pathology scores in both CA1 and CA2 (Table 2, Figure 1D-I). This association remained when the models were simplified to include only significant terms (Figure 1D-I) or, for the relationship between amyloid plaque density on CA1 tau scores, became significant when only amyloid was included in the final model (CA1 amyloid density model: rate ratio 1.16, p = 0.011). When looking at plaque severity, the model of plaque type (diffuse versus neuritic) was more closely related to tau pathology in CA1 and CA2 compared to plaque density at this age. Interestingly, in APOE4 carriers the association of amyloid plaques with tau pathology accumulation was less pronounced (Table 2, Supplemental Figure 3). However, this relationship was lost in models which included plaque severity.
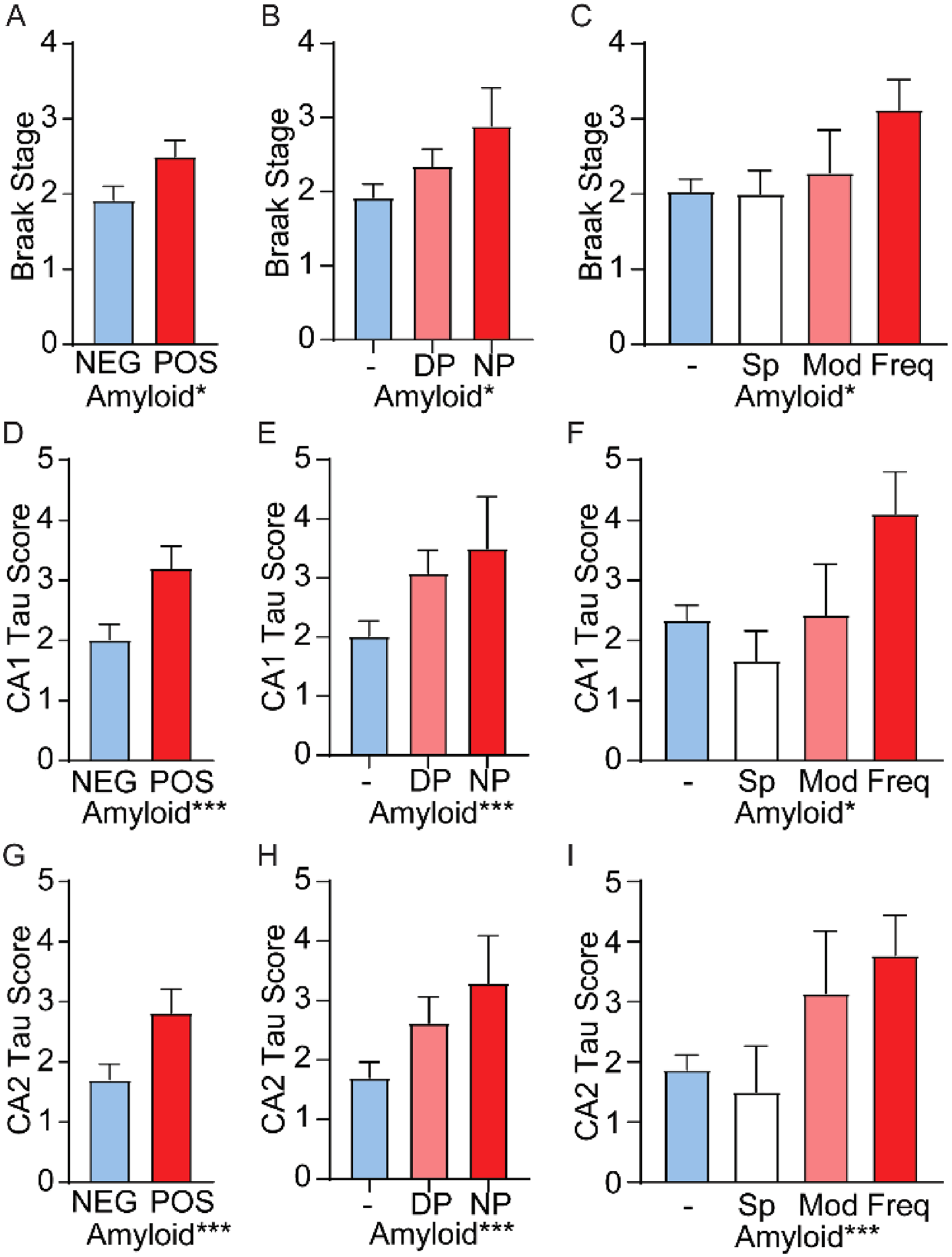
Amyloid severity is associated with increased tau pathology at younger ages. To determine the association of amyloid and tau pathology in younger individuals, amyloid was quantified three ways. Cases were first categorized by the presence or absence of amyloid plaques (A,D,G), next by the type of plaque (B, E, H), and finally by hippocampal plaque density (C, F, I). At younger ages, amyloid plaques were associated with increased tau Braak stage (A, *p = 0.048), with increasing severity of plaque pathology also correlating with increased Braak stage (B,*p = 0.024 plaque type; C,*p = 0.038 plaque density). CA1 tau pathology scores also correlate with amyloid pathology (D, *p = 2.1e-5) and severity (E,*p = 7.4e-4 plaque type; F,*p = 0.011 plaque density). CA2 tau pathology scores were similarly increased with amyloid (G, *p = 1.5e-6) and were correlated with amyloid severity (H,*p = 3.1e-4 plaque type; I,*p = 1.3e-4 plaque density). Significance determined by ordinal logistic regression (A-C), or Poisson regression (D-I) using simplified models with only amyloid (A-C,E-F,H-I) or the interaction model of amyloid and APOE4 only (D,G). Freq, frequent plaques; DP, diffuse plaques; Mod, moderate plaques; NEG, negative; NP, neuritic and diffuse plaques; POS, positive; Sp, sparse plaques. Error bars are SEM.
Associations with early CA1 and CA2 hippocampal tau pathology.
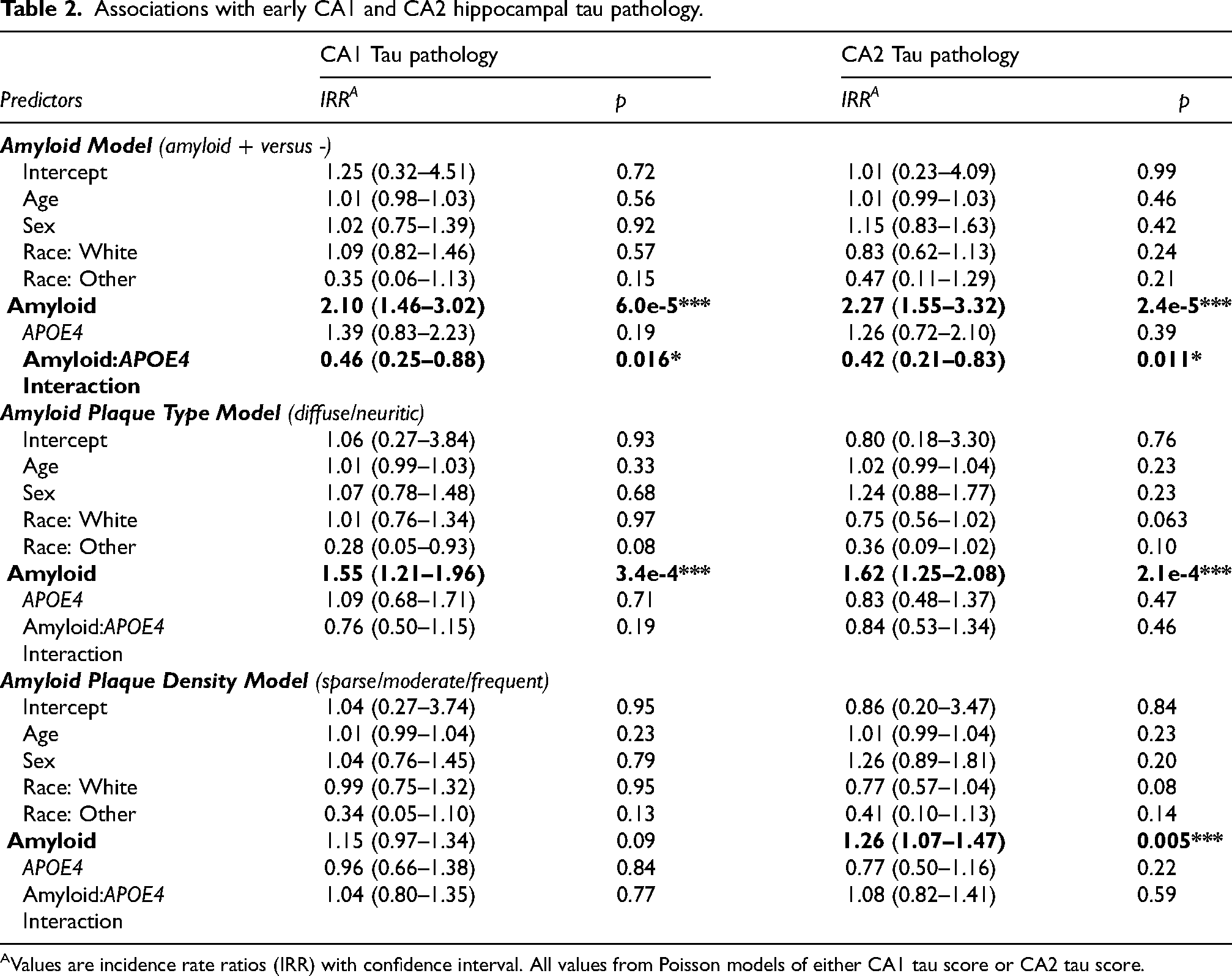
Values are incidence rate ratios (IRR) with confidence interval. All values from Poisson models of either CA1 tau score or CA2 tau score.
Prior studies comparing AD and PART have measured the relative distribution of tau pathology between CA1 and CA2. We therefore divided the cohort into groups based on the relative distribution of tau pathology. The first, and largest group, with 56 cases (62.9%) had similar levels of tau pathology in CA1 and CA2. The second group showed tau pathology predominantly in CA1, scoring higher than CA2 by 2 or more points. The third group showed tau pathology predominantly in CA2, scoring higher than CA1 by 2 or more points.
Amyloid was associated with a significant shift toward CA1-predominant tau pathology compared to amyloid negative cases (Figure 2A, Table 3). In amyloid negative cases, there was a similar proportion of CA1 predominant (20%) and CA2 predominant (16%) cases. However, amyloid positive cases showed a distribution shift toward more CA1-predominance (34%) and fewer CA2-predominant cases (8.6%) (p = 0.0063, Chi-square test). If the amyloid positive cases were restricted to low level amyloid only (Thal 1-2, CERAD 0-1, 74% of amyloid-positive cases), the distribution of cases was similar to amyloid cases overall, with 30.8% of cases showing CA1-predominance and only 7.7% of cases with CA2-predominance. Notably there were no CA2-predominant cases with neuritic plaques. Though the number of neuritic plaque cases were too few for robust statistical comparison (n = 10), this trend is consistent with prior findings in older cohorts. 14 Other demographic and pathologic variables were not associated with a shift in the distribution of hippocampal tau (Table 3).
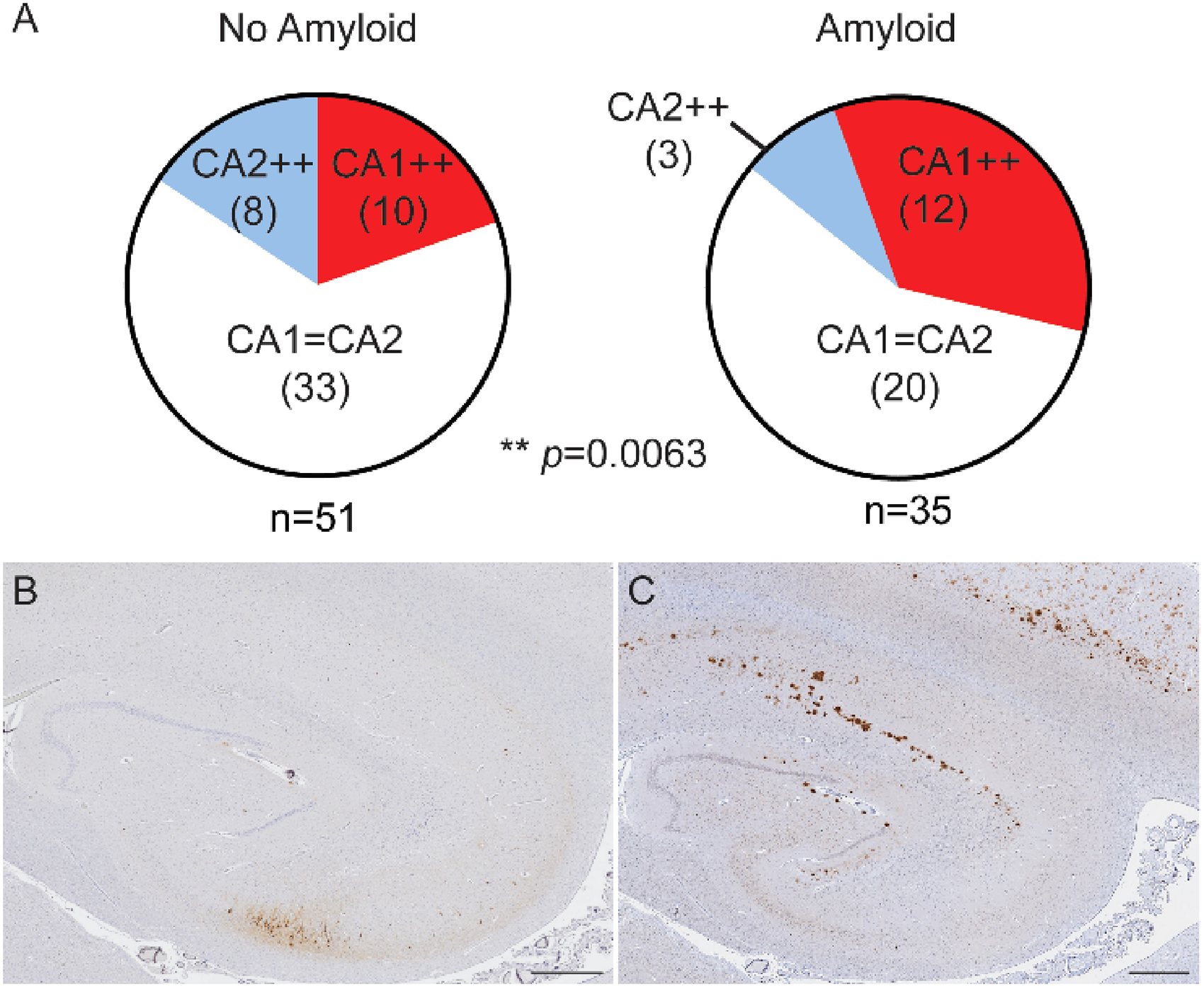
Amyloid associated with CA1-predominant tau pathology. (A) Relative distribution of hippocampal tau pathology in the presence and absence of amyloid plaques. Significance determined by Chi-Square test. Parentheses are number of cases in each category. (B-C) Representative case of CA2-predominant hippocampal tau pathology (B) with amyloid deposition (C). CA1++, CA1-predominant tau pathology; CA2++, CA2-predominant tau pathology; CA1 = CA2, even tau pathology distribution. Scale bar is 1000 µm (B-C).
Factors associated with CA1-predominant Tau pathology.
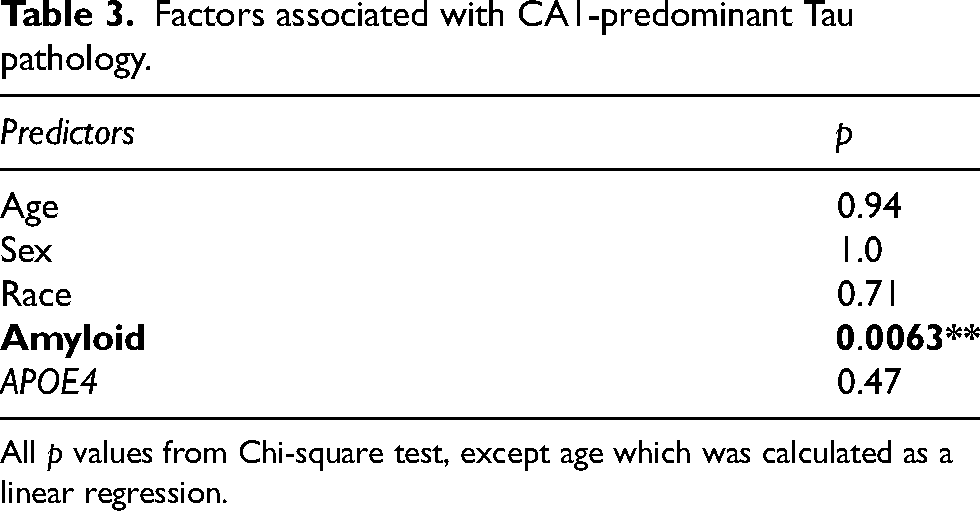
All p values from Chi-square test, except age which was calculated as a linear regression.
To attempt a more direct comparison with prior literature, we calculated the ratio of CA2 to CA1 pathology after excluding cases with a zero value in either region. The CA2/CA1 tau pathology ratio tended to be higher in cases without amyloid, though this was not significant (amyloid 1.10 versus no amyloid 1.26, p = 0.41, Mann Whitney test).
It is interesting to note, however, that there was a wide range of tau pathology distributions in this younger age group. There were a few cases of dramatic CA2 predominant tau pathology with deposition of amyloid (Figure 2B, C) with a Thal stage 2–3, though no cases showed neuritic plaques. There were also cases of marked CA1 predominant tau pathology in cases without amyloid. Thus, hippocampal tau pathology shows a wide regional variability in middle age.
Discussion
Tau pathology is universal in aging, though the patterns of tau pathology have been reported to differ between AD and PART in older individuals. Here we examined patterns of early hippocampal tau pathology in middle age and how they change in association with amyloid plaques (Figure 3). Even in middle age (average age 55.8), amyloid plaques were associated with increased Braak stage and increased tau pathology in both CA1 and CA2. Increasing severity of plaque pathology, either by the presence of neuritic plaques or with frequent plaques in the hippocampus, was also associated with increased severity of tau pathology in CA1 and CA2. When examining the overall distribution of tau pathology, amyloid was associated with a shift toward a greater proportion of CA1-predominant tau pathology.
These early associations of amyloid with changes in hippocampal tau pathology mimic the more established patterns of tau pathology seen at older age groups in AD and PART. Given the younger age of this cohort, pathology was generally low level. The median Braak score was II, the average Thal score was 0.88, and only 11% of cases showed neuritic plaques, which were typically sparse. Despite these low levels of pathology, amyloid was associated with higher tau pathology, which has been documented at older ages,6,14 and was associated with more CA1 predominant cases, mimicking the tau pathology pattern seen in more established older cases of AD.14,26 While older adults can show low levels of amyloid deposition and still be considered possible PART, we have avoided this classification here as the presence of amyloid pathology at younger ages is likely pathologic and more closely biologically related to early AD cases. It remains hotly debated as to whether tau pathology alone in middle age represents early AD-type tau pathology or PART.1,2,12,13,27 While our study cannot answer this important question, the findings in this younger cohort do suggest that amyloid deposition represents a critical pathologic divergence point which is associated with distinct patterns of tau deposition even in middle age.
Increased severity of amyloid pathology was associated with increased tau pathology by Braak stage and in hippocampal regions CA1 and CA2. In this younger cohort, measures of diffuse plaque density and the presence of neuritic plaques were correlated. However, the presence of neuritic plaques in the medial temporal lobe was a stronger predictor of tau pathology in the hippocampus than diffuse plaque density, consistent with the concept that neuritic plaques are thought to be more pathogenic compared to diffuse plaques. 28
The patterns of hippocampal tau pathology in PART remain debated in the field.12,13 Some prior studies of older age groups have found a predilection for CA2 pathology in PART compared to AD,11,14,26 while others have not identified this predilection, 27 and yet others have noted that the CA2-predominant cases represent a smaller group of PART cases. 16 In this study, tau pathology showed an even Gaussian distribution between CA1 and CA2 in the absence of amyloid. 16% of these middle-aged cases without amyloid showed CA2 predominant tau pathology, which is similar to prior to the prior reported rates of 10.8 27 –12%. 16 Interestingly, if a ratio of CA2/CA1 tau pathology is calculated (excluding cases with a no pathology in one region), this middle-aged cohort shows a ratio of 1.26 in cases without amyloid compared to 1.10 for cases with amyloid. While not significant, as many cases had to be excluded for tau scores of 0 in this younger group, the ratios are similar to the 1.20 (PART) versus 0.94 (AD) ratios previously reported. 14 From these comparisons, we can infer that tau pathology patterns established in the absence of amyloid are similar in middle- and older age groups. However, in this younger cohort at least, it appears that tau pathology shows no predilection for CA1 or CA2. Rather, it appears that amyloid is associated with a shift away from CA2-predominant tau pathology. It is tempting to speculate that the absence of amyloid allows CA2-predominant tau pathology to persist into older age, resulting in more frequent CA2-predominant tau pathology in PART when compared to AD.
This early association of amyloid with increased tau pathology in CA2 is contrary to the published concept that CA2 may be relatively spared early in AD.14,19 However, the association of amyloid with increased tau pathology in both regions may, in part, explain the absence of CA2 predominant tau pathology in AD. As amyloid at younger ages is associated with similar rates of increased tau pathology in both CA1 and CA2, the ratio of tau pathology between the two regions would inevitably shift toward 1 as both regions accumulate tau pathology over time. This ratio near 1 is consistent with both the findings in this young cohort and with published literature in older individuals.14,26 What remains less clear is why amyloid may be associated with a greater proportion of CA1-predominant cases. The increased rates of tau pathology in the presence of amyloid were similar in CA1 and CA2 in statistical modeling (Table 2), so it remains unclear why amyloid deposition would tend to become CA1 predominant.
The effects of amyloid on tau accumulation in CA1 and CA2 may be attenuated in this age group in the presence of APOE4. In amyloid negative individuals, tau scores tended to be higher in APOE4 carriers in CA1 and CA2; however, the association of increased tau pathology with increasing severity of amyloid appears to be attenuated. The significance of this finding is uncertain, particularly given that the APOE4 carriers are a smaller subset in the group (n = 28) and statistical models which included measures of amyloid severity did not show the same relationship. Large imaging studies show accelerated tau pathology deposition in APOE4 carriers at low levels of amyloid deposition, 29 potentially correlating with the higher hippocampal tau scores in APOE4 carriers at baseline. However, larger pathologic studies would be needed to determine whether this finding is consistent across cohorts.
In summary, patterns of tau pathology in middle age are strongly associated with amyloid plaque deposition (Figure 3). Amyloid is associated with increased Braak stages, increased tau pathology in both hippocampal CA1 and CA2 regions, and increased CA1-predominant tau pathology cases. In the absence of amyloid, tau pathology shows a normal (Gaussian) distribution of pathology between CA1 and CA2. These distributions of tau pathology in early stages in middle age are similar to the more established patterns of pathology seen in older ages for AD and PART.
Model of relationship between amyloid and hippocampal tau pathology. In a younger aging population, tau pathology initially deposits at low levels in both CA1 and CA2. In the presence of amyloid, tau pathology is increased in both CA1 and CA2 but with a shift toward a greater proportion of cases with higher tau pathology in CA1.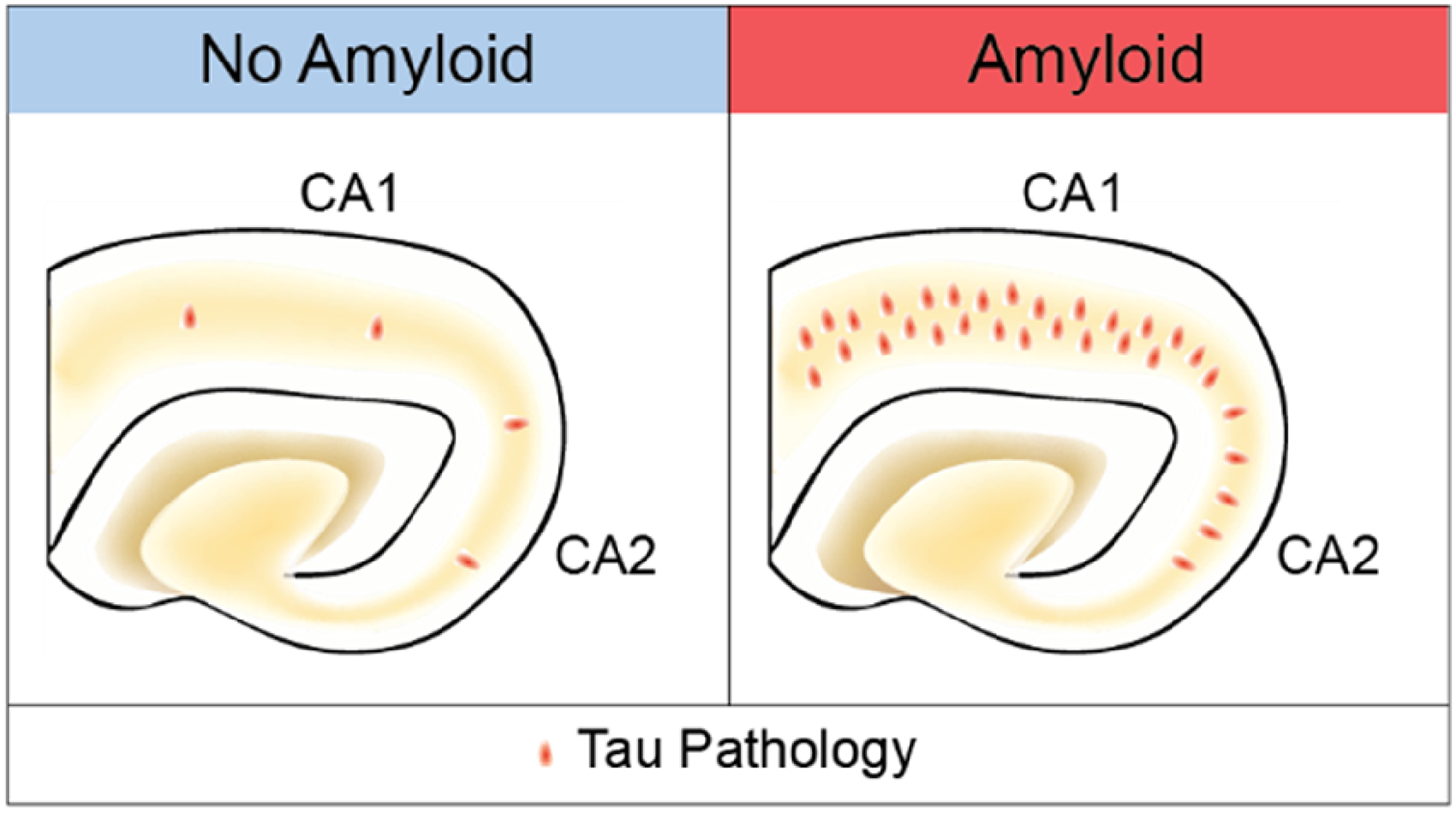
Supplemental Material
sj-docx-1-alz-10.1177_13872877261443643 - Supplemental material for Amyloid plaques associated with shift in patterns of hippocampal tau pathology in middle age
Supplemental material, sj-docx-1-alz-10.1177_13872877261443643 for Amyloid plaques associated with shift in patterns of hippocampal tau pathology in middle age by Rocio Fernanda Rodriguez-Reyes, Haidan Guo, Maria Luongo, Laura Gomez-Isaza, Olga Pletnikova, Juan C. Troncoso and Meaghan Morris in Journal of Alzheimer's Disease
Footnotes
Acknowledgements
The authors have no acknowledgments to report.
ORCID iDs
Ethical considerations
All work in this study was acknowledged by the Johns Hopkins Institutional Review Board as not human subjects research.
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Author contribution(s)
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by awards from the BrightFocus Foundation (A2023004S) and the Johns Hopkins Armstrong Family Human Aging Project/Center for Innovative Medicine Scholar Award. This worked used the Neuropathology histology and immunology lab, which is supported in part by the Johns Hopkins Alzheimer's Disease Research Center (P30 AG066507).
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
The data supporting the findings of this study are available on request from the corresponding author. The data are not publicly available due to privacy or ethical restrictions.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
