Abstract
Background
Resistin is a circulating protein linked to systemic metabolic disorders and inflammation, both of which can contribute to the pathogenesis of Alzheimer's disease (AD). However, the role of resistin during the preclinical stage of AD and its associations with amyloid-β and tau pathology have not been established.
Objective
To measure plasma resistin concentrations in cognitively normal older adults and examine its association with cerebrospinal fluid (CSF) AD biomarkers and neuropsychological measures.
Methods
155 (64 men and 91 women) cognitively normal (Clinical Dementia Rating 0) volunteers met all study criteria with 55 (29 men and 26 women) categorized as preclinical AD based on established CSF criteria. Plasma resistin concentrations were measured by immunoassay.
Results
Since plasma resistin concentrations were higher in men compared to women, all analyses were sex stratified. In men, plasma resistin concentrations were significantly higher in preclinical AD compared to biomarker negative controls and were associated with CSF concentrations of tau and p-tau181; however, after correcting for multiple comparisons, there were no significant associations with any AD biomarkers. Furthermore, plasma resistin concentrations were significantly associated with semantic fluency but not with episodic memory or executive function. In women, plasma resistin concentrations were similar between preclinical AD and controls, and there were no significant associations with CSF AD biomarkers and cognitive measures.
Conclusions
These findings raise the possibility that, in men, alterations in peripheral resistin signaling occur during the earliest stages of AD and could represent an early link between systemic metabolic and inflammation dysregulation in AD.
Keywords
Introduction
Systemic metabolic and immune dysregulation are increasingly recognized as significant contributors to the pathogenesis of Alzheimer's disease (AD). In epidemiological studies, both cardiometabolic disorders (e.g., obesity, diabetes, and hypertension) and higher levels of peripheral inflammatory markers (e.g., C-reactive protein and cytokines like IL-6) are associated with increased risk of developing AD and age-related dementias.1–4 Furthermore, there is a strong link between systemic metabolism and inflammation with high levels of inflammation contributing to the dysregulation of systemic metabolism and serving as important mediators of the pathological effects of cardiometabolic disorders. 5 Supporting the synergistic effects of systemic metabolism and immune dysregulation on the brain, in the Health, Aging, and Body Composition Study, older adults with metabolic syndrome and high inflammation had an increased likelihood of developing cognitive impairment compared to those with metabolic syndrome and low inflammation. 6 However, the exact mechanisms underlying how systemic metabolic and immune dysregulation contribute to the development of AD and age-related dementias have not been fully elucidated.
Resistin was originally identified in rodents as an adipocyte-derived signaling molecule that contributes to insulin resistance. 7 In humans, resistin is produced primarily by immune cells, especially macrophages, and can act as a proinflammatory signal by inducing cytokine signaling, enhancing expression of cell adhesion molecules, and directly activating complement signaling. 8 Due to the pleiotropic effects of circulating resistin on systemic metabolism and inflammation, several studies have implicated resistin as an important mediator of several chronic and age-related diseases including cardiometabolic disorders, rheumatological disorders, and cancer.8,9 While the potential role for resistin in AD has been investigated in prior studies, the results have been conflicting and inconclusive.10–17 Additionally, how resistin signaling may be altered early in AD and the associations of circulating resistin with amyloid-β (Aβ) and tau pathology during the preclinical stage of AD are not known. Therefore, in this study, we measured the concentrations of circulating resistin in a well-characterized cohort of cognitively normal older adults with cerebrospinal fluid (CSF) biomarker-defined preclinical AD and CSF biomarker-negative controls and examined the associations with CSF AD biomarkers and cognitive measures.
Methods
Study participants
Research volunteers were recruited and enrolled in longitudinal studies by the Charles F. and Joanne Knight Alzheimer's Disease Research Center at Washington University in St Louis as previously described.18,19 All participants were community-dwelling older adults in good general health. Study inclusion criteria were age of 50 years and older, cognitively normal with a Clinical Dementia Rating (CDR) of 0, and availability of fasting blood and CSF samples for analysis. Study exclusion criteria were any known medical illness that could contribute to cognitive impairment or dementia and body mass index (BMI) greater than or equal to 30 as obesity could be a possible confounding factor.
This study was approved by the Human Research Protection Office at Washington University School of Medicine in St Louis (Institutional Review Board Protocol Number 201109100) and the Institutional Review Board at Weill Cornell Medicine (IRB Protocol Number 1804019148). Written informed consent was obtained from all study participants and their informants.
Classification of preclinical Alzheimer's disease
CSF concentrations of Aβ40, Aβ42, total tau, and tau phosphorylated at threonine 181 (p-tau181) were measured previously as described using a chemiluminescent enzyme linked immunoassay (Lumipulse G1200, Fujirebio). 20 Using published criteria, preclinical AD was defined as cognitively normal with CDR 0 and CSF tau/Aβ42 ratio > 0.54.19,20
Neuropsychological tests
All participants underwent testing for global cognitive function by Mini-Mental State Examination (MMSE). 21 A subset of participants underwent additional neuropsychological testing including for semantic processing with categorical fluency for animals (100 controls and 54 preclinical AD) and vegetables (62 controls and 51 preclinical AD), 22 immediate and delayed memory with Wechsler Memory Scale-Revised, Logical Memory (WMS-R-LM I & II; 62 controls and 51 preclinical AD), 23 and executive function with Trail Making Test Part A (100 preclinical AD and 54 preclinical AD) and Trail Making Test Part B (64 controls and 52 preclinical AD). 24
Measurement of plasma resistin concentrations
Concentrations of resistin were measured in de-identified plasma samples (1:400 dilution) using a commercial immunoassay (Catalog Number HADK1MAG-61 K; MilliporeSigma) with the Luminex MAGPIX system. A standard curve was constructed using known recombinant resistin protein concentrations ranging from 6.4 to 100,000 pg/mL. The mean minimum detectable dose of resistin for this immunoassay was 2.2 pg/mL. All samples were run in duplicates, and mean values were used. The mean %CV of replicates was 4.5%. Only values that were found within the standard curve were included in the analysis.
Statistical analysis
To normalize the data, plasma resistin concentrations were natural logarithmic transformed. Variables were summarized as mean (standard deviation). For continuous measures, t-tests were used to compare between groups. For categorical measures, Fisher's exact tests were used. For associations between plasma resistin concentrations and age (years) or BMI, Spearman's correlations were used. For associations between plasma resistin concentrations and the CSF AD biomarkers, linear regression adjusting for age and APOE genotype (E4 or non-E4 carriers) were used. For associations between plasma resistin concentrations and cognitive measures, linear regression adjusting for age, APOE genotype (E4 or non-E4 carriers), and education in years were used. Both unadjusted and adjusted models are reported. For all tests, the threshold of statistical significance was set at p < 0.05. To correct for multiple comparisons for the analyses between plasma resistin levels and the CSF AD biomarkers and the cognitive measures, Bonferroni correction was applied resulting in a corrected threshold of statistical significance of p < 0.00833 for comparing 6 CSF biomarkers (i.e., Aβ42, Aβ40, Aβ42/Aβ40 ratio, t-tau, p-tau181, tau/Aβ42 ratio) or p < 0.007 for the 7 cognitive domain testing (i.e., MMSE, animal fluency, vegetable fluency, Trail Making A, Trail Making B, WMS-R-LM I and II). All statistical analyses were performed using Stata version 13.1 (StataCorp, TX), and graphs were made using Prism version 10.3.1 (GraphPad, MA).
Results
Demographic and clinical characteristics
A total of 155 (91 women and 64 men) participants met all study criteria with 55 (29 men and 26 women) participants classified as preclinical AD and 100 (35 men and 65 women) participants classified as biomarker-negative controls (Table 1). Compared to controls, preclinical AD participants were older and had higher proportion of APOE E4 carriers (Table 1). As we previously reported for this cohort, in men but not women, BMI was lower in preclinical AD compared to control participants as previously described (Table 1). 19
Study demographics and characteristics.
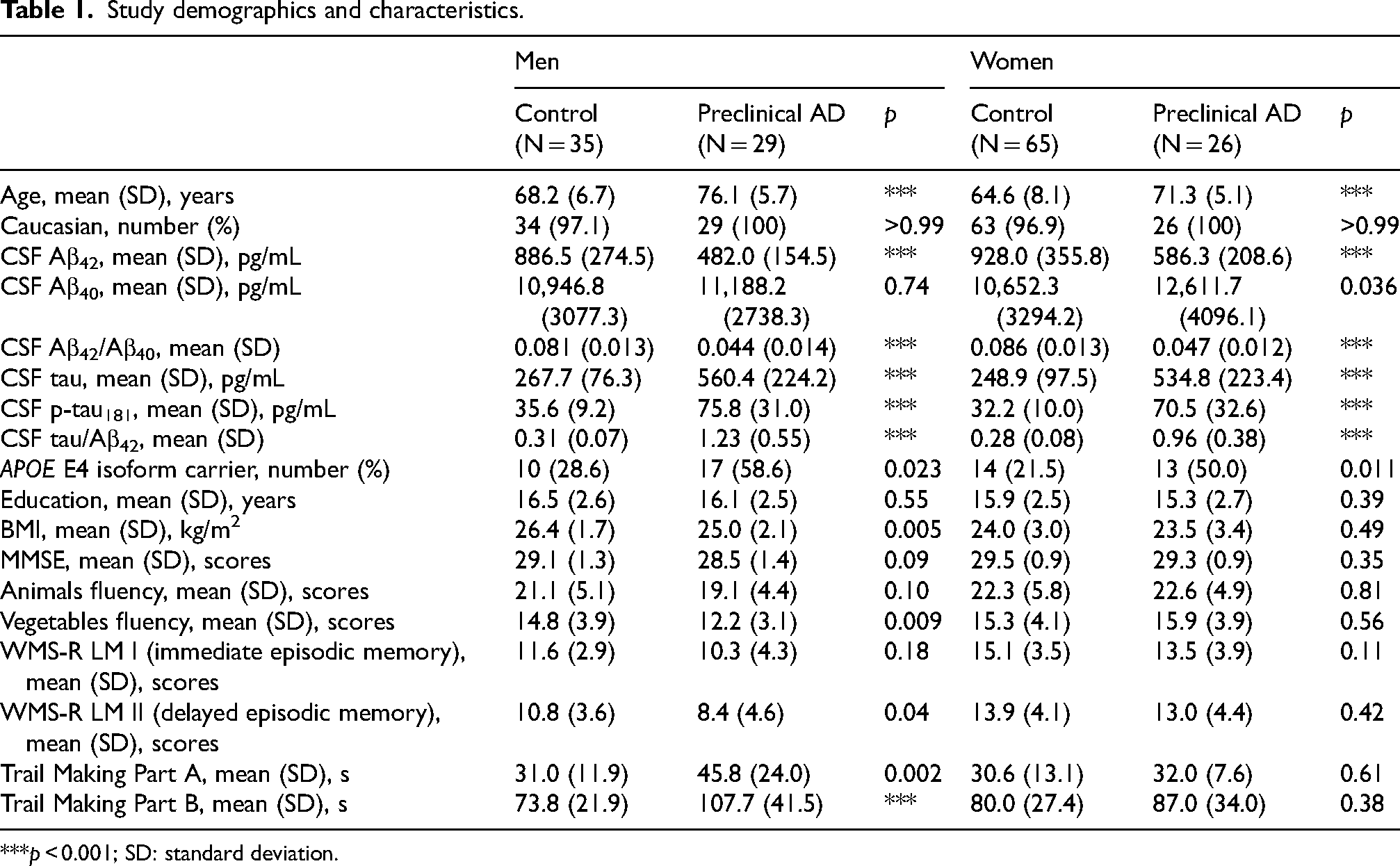
***p < 0.001; SD: standard deviation.
Plasma resistin concentrations are associated with age but not BMI
As resistin has been implicated in age-related and cardiometabolic disorders, 8 we examined the associations between plasma resistin concentrations and age and BMI. In men but not women, plasma resistin concentrations were significantly associated with age (Figure 1A, B). For both men and women, there were no significant associations between plasma resistin concentrations and BMI (Figure 1C, D).
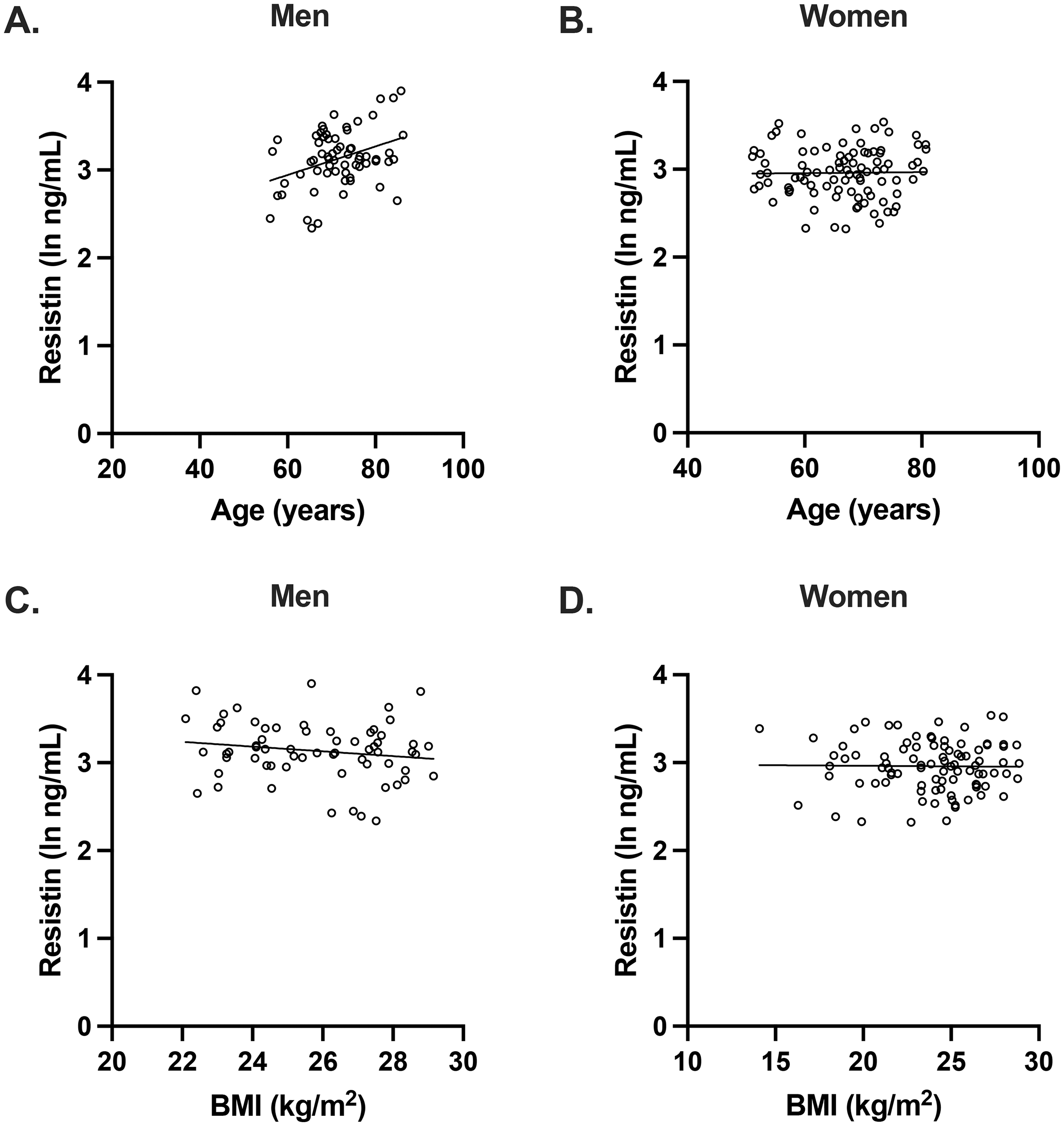
Plasma resistin concentrations were associated with age but not BMI. (A, B). Plasma resistin concentrations were associated with age in men but not women (men: r = 0.363, p = 0.003; women: r = 0.0154, p = 0.885). (C, D). There were no significant associations between plasma resistin concentrations in BMI in both men and women (men: r = -0.166, p = 0.189; women: r = -0.0133, p = 0.900).
Plasma resistin concentrations are higher in men than women
In this cohort of older adults with preclinical AD, we previously identified significant sex differences in measures of systemic metabolism. 19 Therefore, we examined whether there were differences in plasma resistin concentrations between men and women. As plasma resistin concentrations were found to be significantly higher in men than women (Figure 2A), all further analyses were sex stratified.
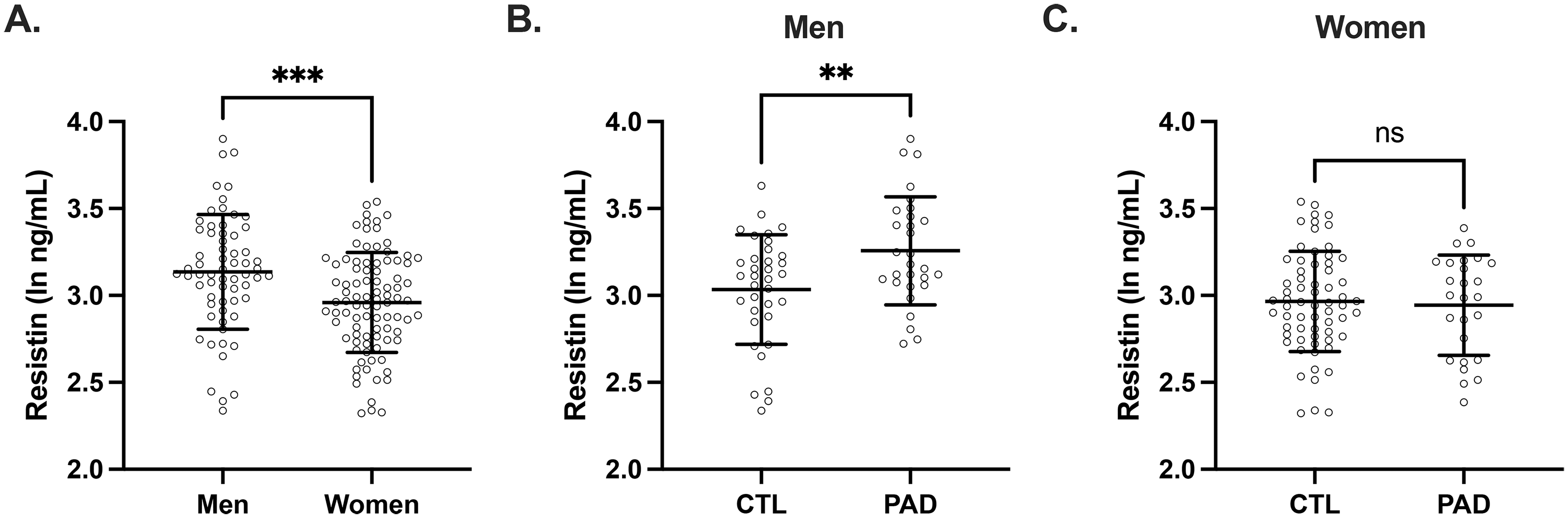
Plasma resistin concentrations in preclinical AD group were higher than the CSF biomarker-negative control group in men but not women. (A) When stratified by sex, plasma resistin concentrations were significantly higher in men compared to women (ln resistin ng/mL: men 3.14 ± 0.33 women AD 2.96 ± 0.29, p = 0.0006, n = 91 men and 64 women). (B) In men, plasma resistin concentrations in preclinical AD group were significantly higher than the control group (ln resistin ng/mL: control 3.03 ± 0.31, preclinical AD 3.26 ± 0.31, p = 0.0061, n = 35 control and 29 preclinical AD study participants). (C) In women, plasma resistin concentrations were not different between preclinical AD and control groups (ln resistin ng/mL: control 2.97 ± 0.289, preclinical AD 2.94 ± 0.288, p = 0.744, n = 65 control and 26 preclinical AD study participants). Individual values are shown with bars depicting means and standard deviations. CTL: control study participants; PAD: preclinical AD study participants; ns: not significant; *p < 0.05, and.
Plasma resistin concentrations are higher in preclinical AD in men but not women
We then examined if there were differences in plasma resistin concentrations between preclinical AD and control participants and if there were any associations between plasma resistin and CSF AD biomarkers. In men, plasma resistin concentrations were significantly higher in preclinical AD compared to controls (Figure 2B). However, in women, there were no significant differences in plasma resistin concentrations between preclinical AD and control participants (Figure 2C). Additionally, in men, after adjusting for age and APOE E4 carrier status, plasma resistin concentrations were associated with CSF concentrations of total tau and p-tau181 but not with Aβ40, Aβ42, Aβ42/Aβ40, or tau/Aβ42; however, none of these associations reached significance after correcting for multiple comparisons (Figure 3). In women, after adjusting for age and APOE E4 carrier status, plasma resistin concentrations were not significantly associated with any of the CSF AD biomarkers (Figure 4).
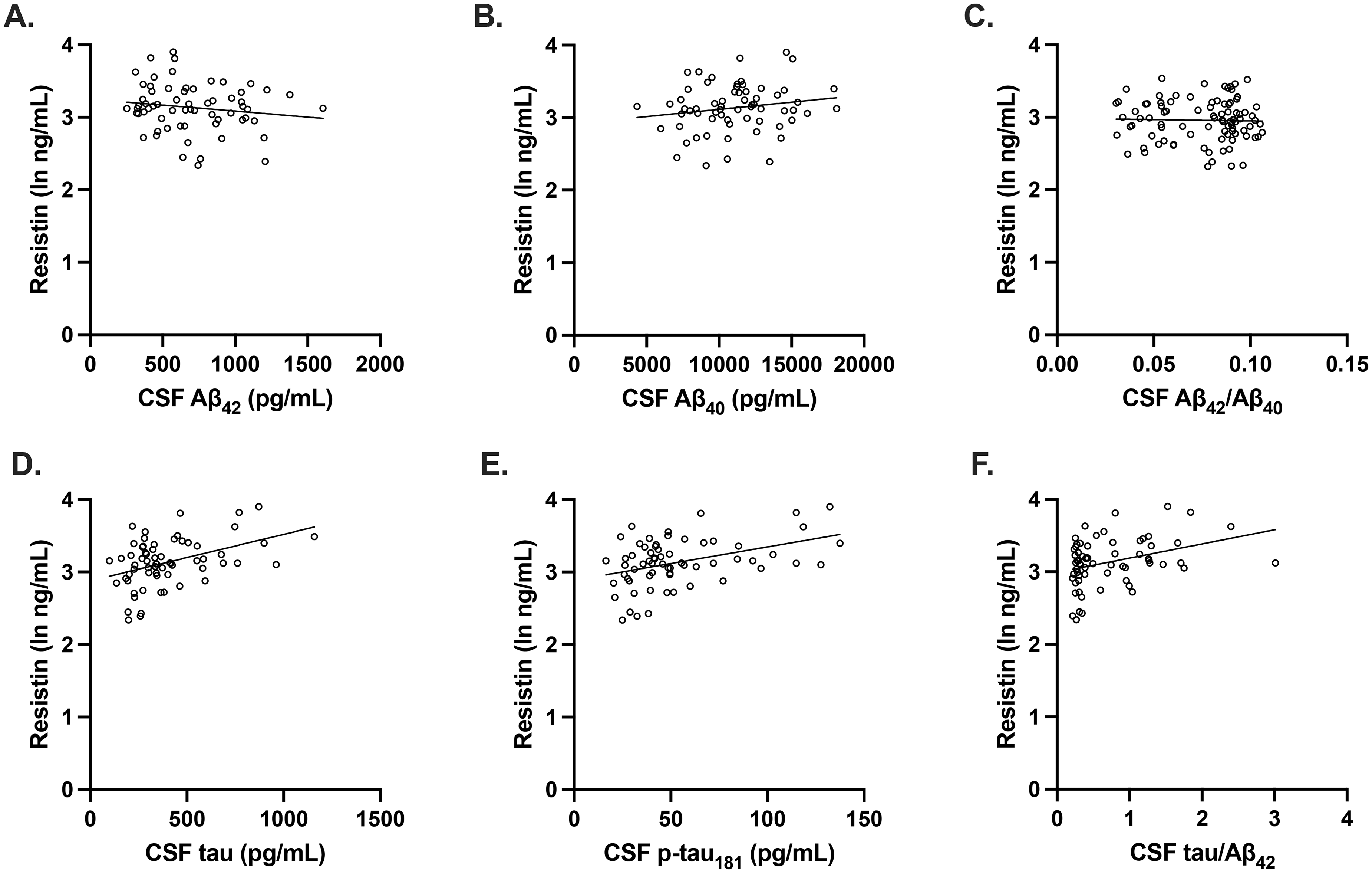
Plasma resistin concentrations were associated with tau pathology in men. (A- F) After adjusting for age and APOE E4 carrier status and correcting for multiple comparisons, there were no significant associations between plasma resistin concentrations and CSF concentrations of Aβ42 (unadjusted beta coefficient −0.152 p = 0.231; adjusted beta coefficient 0.00274, p = 0.985,), Aβ42/Aβ40 (unadjusted beta coefficient −0.299 p = 0.016; adjusted beta coefficient −0.193, p = 0.214), Aβ40 (unadjusted beta coefficient 0.175 p = 0.167; adjusted beta coefficient 0.152, p = 0.205), tau (unadjusted beta coefficient 0.421, p < 0.001; adjusted beta coefficient 0.325, p = 0.015), p-tau181 (unadjusted beta coefficient 0.413, p < 0.001; adjusted beta coefficient 0.317, p = 0.020), and tau/Aβ42 (unadjusted beta coefficient 0.350, p = 0.005; adjusted beta coefficient 0.228, p = 0.108). Scatterplots show individual values with unadjusted regression lines.
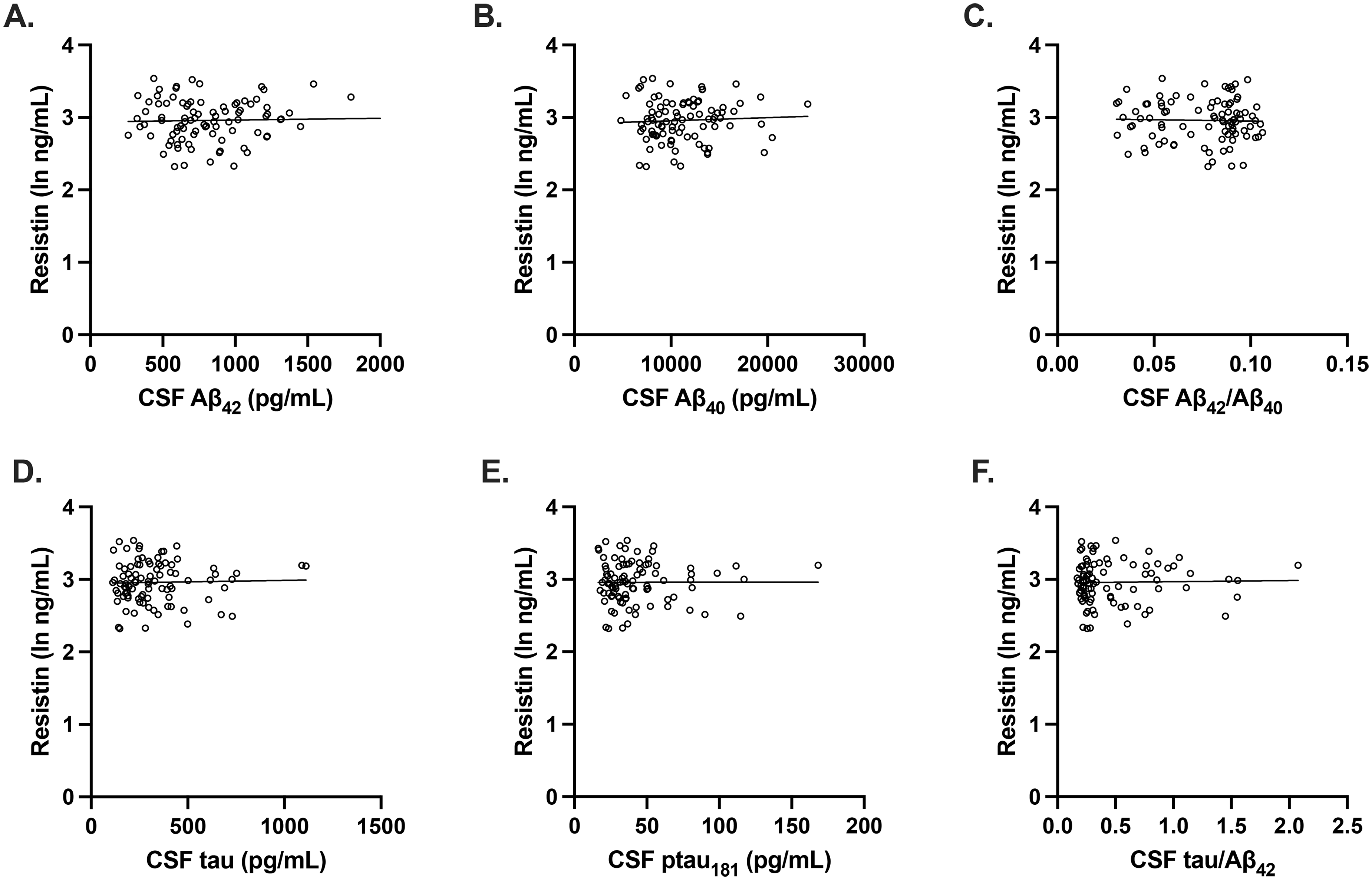
Plasma resistin concentrations were not associated with Aβ or tau pathology in women. (A-F) After adjusting for age and APOE E4 carrier status, there were no significant associations between plasma resistin levels and CSF concentrations of Aβ42 (unadjusted beta coefficient, 0.0309 p = 0.772; adjusted beta coefficient 0.0436, p = 0.704), Aβ42/Aβ40 (unadjusted beta coefficient −0.0269, p = 0.801; adjusted beta coefficient −0.0169, p = 0.901), Aβ40 (unadjusted beta coefficient 0.0544, p = 0.609; adjusted beta coefficient 0.0587, p = 0.617), tau (unadjusted beta coefficient 0.0275, p = 0.796; adjusted beta coefficient 0.0212, p = 0.863), p-tau181 (unadjusted beta coefficient 0.000915, p = 0.993; adjusted beta coefficient −0.0142, p = 0.908), and tau/Aβ42 (unadjusted beta coefficient 0.0203, p = 0.849; adjusted beta coefficient 0.00883, p = 0.944). Scatterplots show individual values with unadjusted regression lines.
Plasma resistin concentrations are associated with semantic fluency in men but not women
Finally, we investigated whether plasma resistin concentrations were associated with neuropsychological measures, particularly those that may be affected during the preclinical stage of AD including global cognitive function (MMSE), semantic processing (categorical fluency for animals and vegetables), immediate and delayed episodic memory (WMS-R-LM I & II), and executive function (Trail Making Test Part A and Part B).25,26 There were no significant differences in MMSE scores between preclinical AD and controls for both men and women (Table 1). Furthermore, after adjusting for age, APOE E4 carrier status, and education, plasma resistin concentrations were not significantly associated with MMSE scores in both men and women (men: adjusted beta coefficient 0.196, p = 0.097; women: adjusted beta coefficient −0.115, p = 0.274).
For the other neuropsychological measures, compared to controls, men with preclinical AD had significantly lower performance scores on vegetable fluency, delayed episodic memory (WMS-R-LM II), and Trail Making Test Parts A and B (Table 1). After adjusting for age, APOE E4 carrier status, and education and multiple comparisons, plasma resistin concentrations were significantly associated with semantic processing (categorical fluency for vegetables) but not with the other neuropsychological measures (Figure 5). In women, there were no significant differences between preclinical AD and control participants in any of the neuropsychological measures (Table 1). After adjusting for age, APOE E4 carrier status, and education, plasma resistin concentrations were not significantly associated with any of the neuropsychological measures (Figure 6).
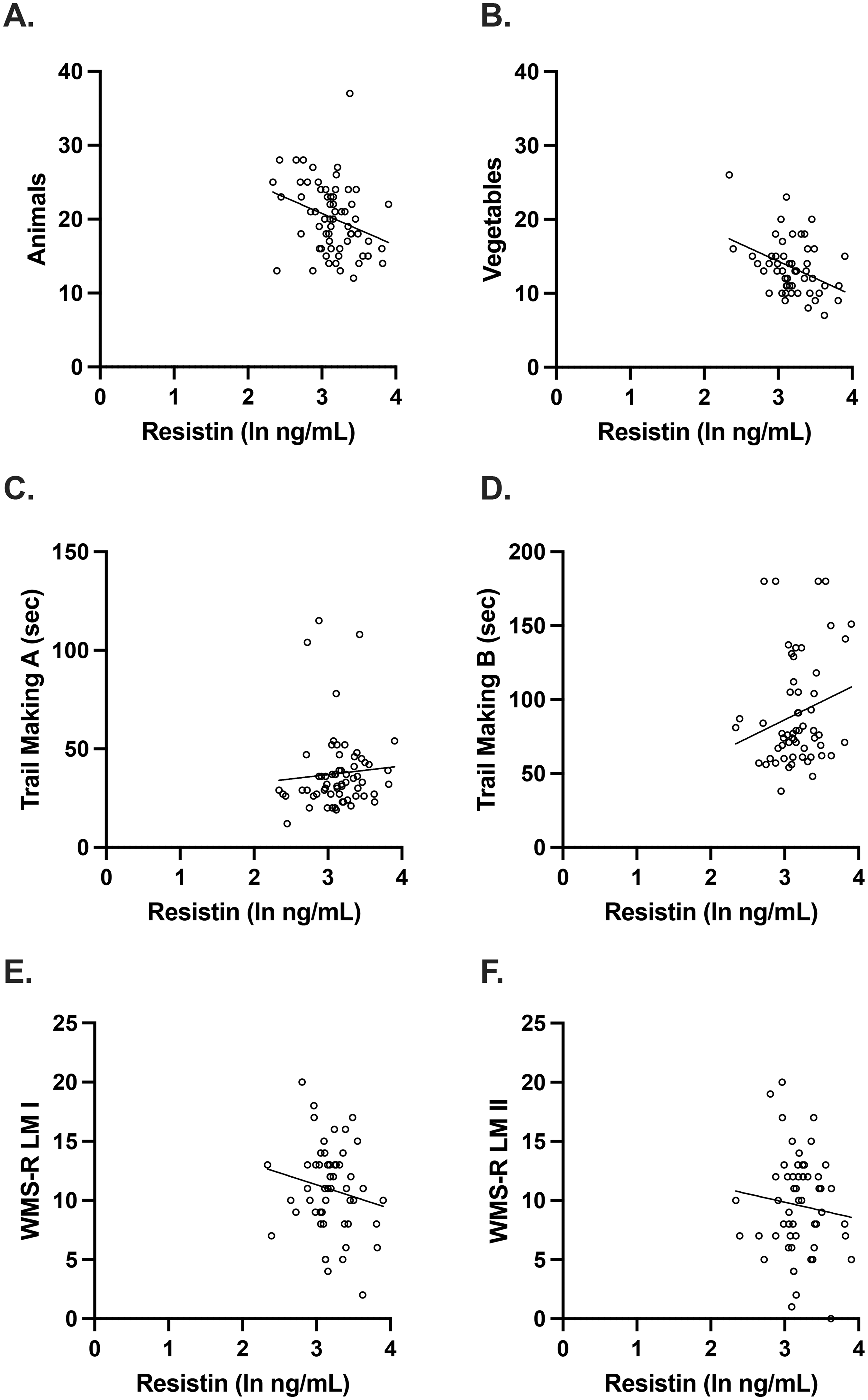
Plasma resistin concentrations were associated with category fluency in men. (A, B) After adjusting for age, APOE E4 carrier status, and education and correcting for multiple comparisons, there were significant associations between plasma resistin levels and semantic fluency (Vegetables: unadjusted beta coefficient −0.383, p = 0.004; adjusted beta coefficient −0.403, p = 0.005). (C-F) However, there were no significant associations with the other cognitive measures (Animals: unadjusted beta coefficient −0.300, p = 0.017; adjusted beta coefficient −0.318, p = 0.019, Trail Making A: unadjusted beta coefficient 0.0741, p = 0.564; adjusted beta coefficient 0.0758, p = 0.584, Trail Making B: unadjusted beta coefficient 0.209, p = 0.118; adjusted beta coefficient 0.197, p = 0.142, WMS-R LM I: unadjusted beta coefficient −0.168, p = 0.224; adjusted beta coefficient −0.193, p = 0.171, and WMS-R LM II: unadjusted beta coefficient −0.103, p = 0.458; adjusted beta coefficient −0.119, p = 0.400). Scatterplots show individual values with unadjusted regression lines.
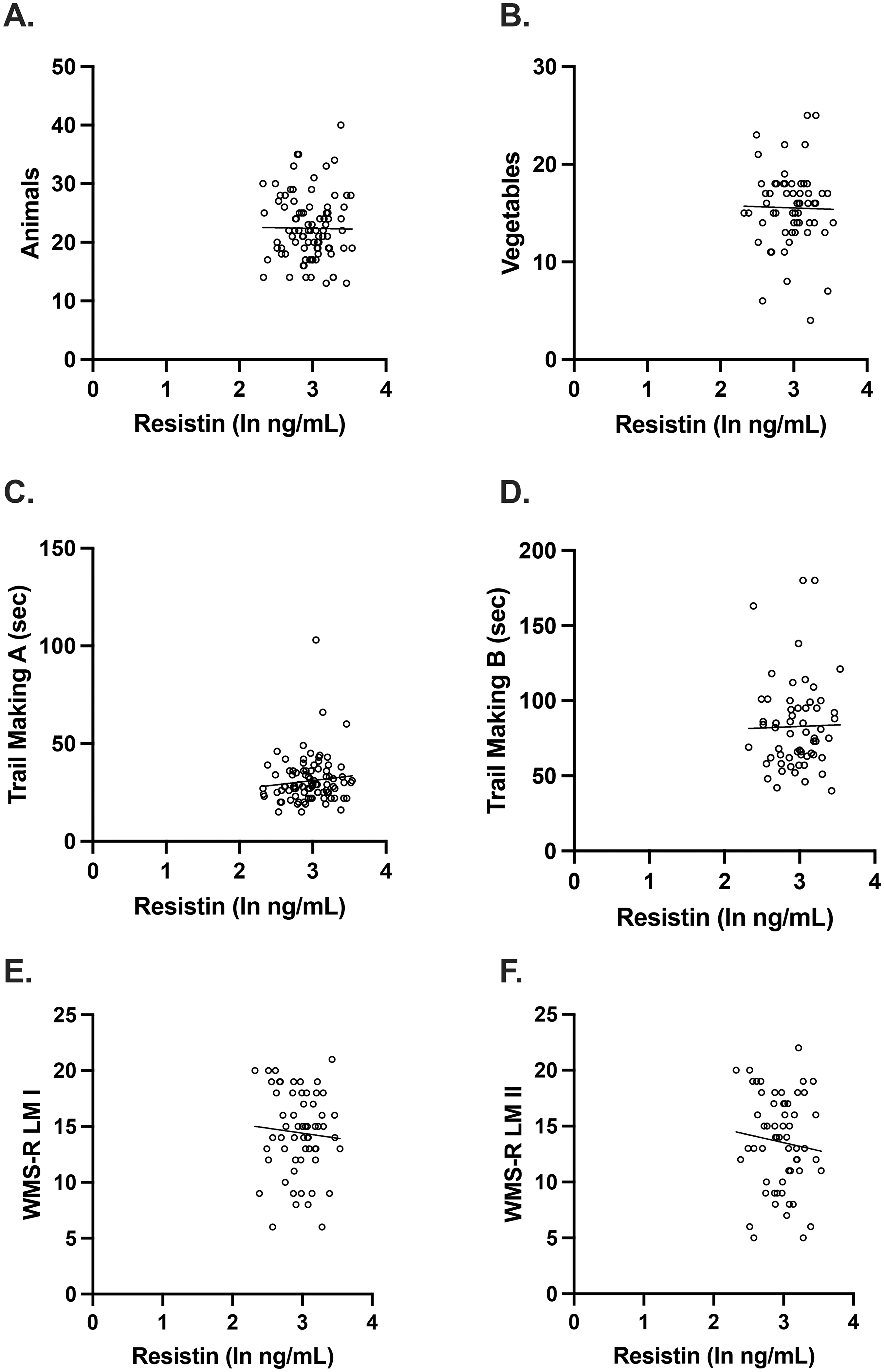
Plasma resistin concentrations were not associated with any of the neuropsychological measures in women. (A-F) After adjusting for age, APOE E4 carrier status, and education, there were no significant associations between plasma resistin levels and any of the neuropsychological measures (Animals: unadjusted beta coefficient −0.0115, p = 0.913; adjusted beta coefficient −0.0139, p = 0.890; Vegetables: unadjusted beta coefficient −0.0189, p = 0.887; adjusted beta coefficient 0.0176, p = 0.896; Trail Making A: unadjusted beta coefficient 0.108, p = 0.308; adjusted beta coefficient 0.105, p = 0.298, and Trail Making B: unadjusted beta coefficient 0.0193, p = 0.885; adjusted beta coefficient −0.0282, p = 0.828; WMS-R LM I: unadjusted beta coefficient −0.0694, p = 0.602; adjusted beta coefficient −0.0462, p = 0.727 and WMS-R LM II: unadjusted beta coefficient −0.0962, p = 0.469; adjusted beta coefficient −0.0630, p = 0.630). Scatterplots show individual values with unadjusted regression lines.
Discussion
To the best of our knowledge, this is the first study to examine plasma resistin concentrations in older adults with CSF biomarker defined preclinical AD. Like our previous study in this cohort, 19 there was sexual dimorphism with plasma resistin concentrations significantly higher in preclinical AD compared to CSF biomarker negative control participants in men but no significant differences in women. Furthermore, in men, plasma resistin concentrations were associated with tau pathology (i.e., CSF concentrations of tau and p-tau181) but not with Aβ pathology (i.e., CSF concentrations of Aβ40, Aβ42, Aβ42/Aβ40, or tau/ Aβ42); however, after correcting for multiple comparisons, there were no significant associations between plasma resistin concentrations and any of the CSF AD biomarkers. In women, plasma resistin concentrations were not associated with any of the CSF AD biomarkers. Finally, in exploratory analyses, plasma resistin concentrations in men were significantly associated with semantic fluency (categorical fluency with vegetables) but not with measures of executive function (Trail Making Test Parts A and B), episodic memory (WMS-R-LM I & II), or global cognition (MMSE). In women, there were no significant associations between plasma resistin concentrations and any of the neuropsychological measures. Collectively, the results from our study would be consistent with circulating resistin having a role early in AD pathogenesis in a sexually dimorphic manner and support circulating resistin as a possible biomarker of cognitive dysfunction in AD.
Our finding of higher plasma resistin concentrations in preclinical AD compared to CSF biomarker negative controls is consistent with prior studies that found higher circulating resistin concentrations in AD.10,11,14,27 However, other cross-sectional studies have reported lower or no significant changes in circulating resistin concentrations in AD.12,15 The conflicting findings from these studies could be from demographic differences among the study populations, relatively small sample sizes, and not fully considering possible confounding factors (e.g., sex differences, changes in inflammatory or metabolic conditions, etc.). As the conflicting studies relied solely on clinical diagnostic criteria to classify AD without the use of CSF or positron emission tomography (PET) AD biomarkers, it is also possible that there were some misclassifications. 28 While we did not examine resistin in the CNS, CSF resistin concentrations have been previously reported to be higher in autopsy-confirmed AD compared to cognitively normal subjects, 29 which would provide further evidence supporting high resistin signaling in AD.
The marked sex differences seen in this cohort is consistent with our prior study, where we found that men but not women in the preclinical stage of AD had lower BMI and alterations in concentrations of circulating cardiometabolic factors. 19 Earlier studies have found sex differences that support men may be more susceptible to systemic metabolic changes early in AD pathogenesis including greater body weight loss in men but not women who developed incident MCI compared to those that did not. 30 Additional studies have also found associations between lower BMI and greater AD pathology and conversion from MCI to AD in men but not women.31,32 However, other studies have found no sex differences in the association between body weight loss and dementia risk. 33 It should also be noted that it remains possible that circulating resistin concentrations are altered in women at later stages of AD such as those with MCI or dementia.
The role of sex differences in resistin has not been fully established with published studies showing conflicting results. Contrary to our findings, some studies have reported higher circulating resistin concentration in women compared to men.34–36 In a study of non-diabetic obese patients, circulating resistin concentrations were significantly associated with cardiovascular risk factors in women but not in men. 34 Another study found modestly elevated plasma resistin concentrations in women compared to men, but there were no significant interactions of sex with inflammatory or metabolic factors in the relationship with plasma resistin. 36 However, several other studies have found no significant sex differences in circulating resistin concentrations.37–39 Additional studies clarifying the potential sex differences that resistin signaling has on inflammation and cardiometabolic factors during the early stages of AD are clearly needed.
There have been limited studies investigating the relation between circulating resistin and Aβ and tau pathology. In a dementia cohort from Italy consisting of both AD and frontotemporal dementia (FTD), circulating resistin concentrations were found to be positively associated with CSF Aβ42 concentrations. 12 In contrast, we found that circulating resistin concentrations were associated with tau but not Aβ pathology; however, these associations were not significant after correcting for multiple comparisons. A possible reason for the discrepancy between the studies could be the use of more advanced clinical stages (i.e., dementia compared to preclinical disease). Nonetheless, our findings suggest that circulating resistin may be a relatively non-specific marker of neurodegeneration rather than a marker specific for AD pathology. Supporting this possibility is a study that found higher circulating resistin concentrations in patients with FTD, particularly those carrying null mutations in the progranulin gene. 40
There are also relatively few studies examining the link between circulating resistin and neuropsychological measures. In one cross-sectional study, despite higher serum resistin concentrations in patients with AD compared with cognitively normal controls, there was no significant association between serum resistin concentrations and MMSE scores. 14 Another study found an inverse relation with plasma resistin concentrations negatively associated with CDR scores, although the magnitude of the association was relatively modest. 15 Notably, studies examining the association between circulating resistin and specific cognitive domains are lacking. In our exploratory analyses, circulating resistin concentrations were significantly associated with category fluency measures. Our findings suggest that alterations in circulating resistin may reflect early changes in the structure and function of brain regions important for semantic processing such as the left temporal lobe.41–43 Consistent with this possibility, a recent examination of longitudinal data from the Alzheimer's Disease Neuroimaging Initiative (ADNI) found that higher resistin levels were associated with decreased hippocampal and entorhinal cortex volumes. 16 However, our findings should be interpreted with caution as all participants were cognitively normal with neuropsychological measures within the normal limits, which could make it difficult to assess cognitive domains that are minimally affected. Future studies examining the associations between circulating resistin and structural and functional changes in brain regions and related neuropsychological measures across the various stages of AD would further elucidate the connections between resistin and cognition.
There are some conflicting data regarding whether circulating resistin has a protective role or contributes to the pathogenesis of AD. In a case-cohort study, higher plasma concentrations of resistin were associated with a decreased risk of dementia, suggesting that resistin could have a beneficial and protective role against AD and related dementias. 13 In contrast, a recent longitudinal study of the UK Biobank cohort found that plasma resistin levels were positively associated with dementia risk. 17 Furthermore, in the ADNI cohort, higher plasma resistin levels were associated with greater impairment in functional activities of daily living. 16 Additionally, several studies in neurologic disorders ranging from intracerebral hemorrhage, atherothrombotic ischemic stroke, and traumatic brain injury have found that higher circulating concentrations of resistin were associated with poorer outcomes.44,45 Finally, in an animal model of Aβ pathology with metabolic syndrome, exogenous administration of resistin exacerbated Aβ pathology, increased oxidative stress, and reduced glucose metabolism. 46 Together, these studies largely support resistin as a pathologic factor in AD by potentially promoting insulin resistance and inflammation. However, as this study was cross-sectional in nature, reverse causality cannot be excluded, where the neurodegeneration contributes to the systemic inflammatory response and elevated circulating resistin levels. Additional studies including longitudinal studies in humans with AD and detailed mechanistic studies in animal and cellular models are needed to further clarify the role of resistin in AD and related neurodegenerative disorders.
Our study has several strengths including the use of well-characterized study cohort with neuropsychological measures and CSF measurements of AD pathology to categorize cognitively normal individuals in the preclinical stages of AD. However, there are several limitations. As discussed earlier, the study was cross-sectional in nature. Therefore, the temporal relation between plasma resistin concentrations and CSF AD biomarkers or changes in neuropsychological function is not known. Furthermore, we did not measure resistin concentrations in the CSF and therefore any central role that resistin has is not known. Finally, the findings from our study need to be verified in additional larger and more diverse cohorts.
In conclusion, in a cohort of cognitively normal older adults, circulating resistin concentrations were higher in the preclinical stage of AD and associated with worsening semantic fluency in a sexually dimorphic manner. These sex differences were similar to our previous study in this cohort, where men, but not women, with preclinical AD had evidence of early systemic metabolic changes as reflected by low BMI and plasma plasminogen-activator inhibitor-1 levels. 19 The underlying etiology and the significance of these sex differences remain unclear. Collectively, the results from our study suggest that increased peripheral resistin signaling could serve as a link between systemic metabolic and immune dysregulation in the early stages of AD. Future studies will be needed in additional human cohorts to verify and validate these findings and in cellular and animal models to explore the mechanistic implications of increased resistin signaling in early stages of AD.
Footnotes
Acknowledgements
The authors would like to first thank all study volunteers. We would also like to thank Drs. Anne Fagan, Krista Moulder, Betsy Grant, David Holtzman, the Knight Alzheimer's Disease Research Center (P50AG05681), Adult Children Study (P01AG026276), and Healthy Aging and Senile Dementia (P01AG03991) for providing plasma samples and associated data used in this study.
Ethical considerations
This study was conducted in accordance with the Declaration of Helsinki and was approved by the Human Research Protection Office at Washington University School of Medicine in St Louis (IRB Protocol Number 201109100) and the Institutional Review Board at Weill Cornell Medicine (IRB Protocol Number 1804019148).
Consent to participate
Written informed consent was obtained from all study participants and their informants.
Consent for publication
Not applicable.
Author contribution(s)
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Institutes of Health (K08AG051179, R01AG070868, and UL1TR001384) and the BrightFocus Foundation (A2015485S and A2020363S). The Knight Alzheimer's Disease Research Center (P50AG05681), Adult Children Study (P01AG026276), and Healthy Aging and Senile Dementia (P01AG03991) provided plasma samples and associated data. The sponsors did not have any role in the study design and conduct, data collection, analysis, and interpretation, and preparation, review, or approval of the article.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Makoto Ishii is an Editorial Board Member of this journal but was not involved in the peer-review process of this article nor had access to any information regarding its peer-review.
Data availability statement
The data supporting the findings of this study are available upon request from the corresponding author.
