Abstract
Background
Alzheimer's disease (AD) affects over 10% of adults aged 65 and older. Black and Hispanic/Latino individuals experience 1.5–2.0 times higher prevalence than White individuals, yet AD remains underdiagnosed, particularly in non-White populations. Common screening tools such as the Mini-Mental State Examination and Montreal Cognitive Assessment are limited by time burden and cultural sensitivity.
Objective
To evaluate the impact of integrating a brief dementia screening tool (DST) into the electronic health record (EHR) on diagnosis, evaluation, and treatment in primary care.
Methods
The DST, developed by the University of California Alzheimer's Disease Centers and the California Department of Public Health, takes under five minutes and includes a three-question patient form, optional informant questionnaire, and Mini-Cog. It was translated and culturally adapted for Spanish-speaking patients and embedded in the EHR. Patients aged ≥60 in a diverse Los Angeles County family medicine clinic completed the DST before annual wellness visits. We conducted a pre-post study comparing patients without prior dementia diagnoses during pre-intervention (February 2021–August 2022) and post-intervention (January 2023–June 2024) periods. Primary outcome was new dementia diagnosis; secondary outcomes included medications, referrals, labs, and imaging.
Results
Among 1515 eligible patients post-intervention, 1249 completed screening. New dementia diagnoses increased from 0.75% pre-DST to 2.45% among those screening positive (adjusted OR 2.99; p = 0.01). Dementia medications, laboratory orders, and specialty referrals significantly increased; imaging did not.
Conclusions
A brief, culturally adapted DST integrated into primary care improved dementia diagnosis, evaluation, and treatment.
Introduction
Based predominantly on longitudinal cohort studies, it is estimated that Alzheimer's disease (AD) affects approximately 10.9% of individuals over the age of 65. 1 Furthermore, certain demographic groups, such as Black and Hispanic/Latino individuals, face an increased risk of AD, with relative risks ranging from 1.5 to 2.0 compared to their non-Hispanic White individuals.2,3
However, when examining real-world clinical data, such as electronic health records (EHR), there is significant underdiagnosis. For instance, the documented diagnoses of dementia in Medicare claims or EHR is 50–65% when compared to clinical research criteria.4–9 This indicates a considerable number of individuals with AD may be undiagnosed, missing crucial chances for early dementia intervention. Delays in diagnosis limit opportunities for care planning, participation in clinical trials, and support services that may improve patient and caregiver quality of life.4,10 The underdiagnosis of AD in real-world clinical settings, particularly among Black and Hispanic/Latino patients, 11 underscores the need for improved dementia screening methods. Racial and ethnic disparities in dementia diagnosis have been linked to factors such as lower access to specialty care, cultural stigma, provider bias, and limited use of validated screening tools in primary care.3,12,13 Addressing these disparities requires tools that are not only brief and practical for clinical use but also accessible and acceptable to historically underserved populations.
Several screening tools for dementia have been developed, including the Mini-Mental State Examination (MMSE), Montreal Cognitive Assessment (MoCA), Saint Louis University Mental Status (SLUMS), TabCAT, Mini-Cog, and Rowland Universal Dementia Assessment Scale (RUDAS). The MMSE is a frequently used screening tools, offering a broad evaluation of cognitive abilities. It tests cognitive domains such as orientation, memory, attention, and language proficiency. 14 The MoCA serves as another instrument designed to identify mild cognitive impairment (MCI), evaluating cognitive domains including attention, executive functions, memory, language aptitude, conceptual thinking, mathematical capabilities, and orientation. 15 However, the MMSE and MoCA take 10–15 min to complete, which can be too long as a primary care screening tool especially when the total visit time is 20–30 min. 16 The SLUMS examination may offer advantages over the MMSE in detecting mild neurocognitive disorder (MNCD) among older adults, particularly across various educational backgrounds and demographic groups. 17 SLUMS takes less time to administer than the MMSE or MoCA, 18 but would benefit from further validation across diverse populations. The UCSF TabCAT is a tablet-based cognitive assessment tool designed to evaluate cognitive function in various domains such as memory, attention, and language. 19 Furthermore, the RUDAS is a screening tool designed to mitigate the influence of educational and cultural biases in dementia assessment. 20 However, the RUDAS has been shown to have a lower sensitivity or specificity when assessing individuals with milder cognitive impairments. 21 The Mini-Cog is a brief cognitive screening tool that combines a three-item recall test with a clock-drawing task. It was designed to be quickly administered making it useful for primary care,22,23 but does not include patient subjective questions.
By incorporating more accessible screening measures within routine clinical care, healthcare providers can improve the detection of AD and facilitate timely interventions. Such efforts are crucial not only for ensuring equitable access to diagnosis and treatment but also for addressing the growing public health challenge posed by AD, especially within underrepresented populations. 24 Addressing these disparities entails specific strategies such as leveraging EHR data 25 and adapting neuro-cognitive assessments based on site-specific needs. 26 Thus, prioritizing dementia screening initiatives27–29 represents a proactive step towards reducing health disparities and enhancing quality of care for dementia.
Our primary objective of this quality improvement study was to assess the impact of implementing a brief dementia screening tool integrated into the EHR on dementia diagnoses, workup, and treatment. We conducted a pre-post intervention study in one academically affiliated primary care clinic that serves a multiracial, multiethnic population in Los Angeles County.
Methods
Dementia screening tool
Prior work by University of California Alzheimer's Disease Centers in 2018 in collaboration with the California Department of Public Health led to the develop a concise and efficient dementia screening tool, the Assessment of Cognitive Complaints Toolkit for Alzheimer’s Disease (CDPH DST), 30 that could be completed in less than five minutes. This tool was developed to improve early dementia detection by incorporating a brief three-question patient questionnaire and three-question informant questionnaire if available. The Mini-Cog is to be performed if no informant was available. A positive screening result consists of any questions answered affirmatively by either the patient or informant, or if the Mini-Cog assessment yielded a score below 3 out of 5 points, which is the standardized scoring for the Mini-Cog (Figure 1).
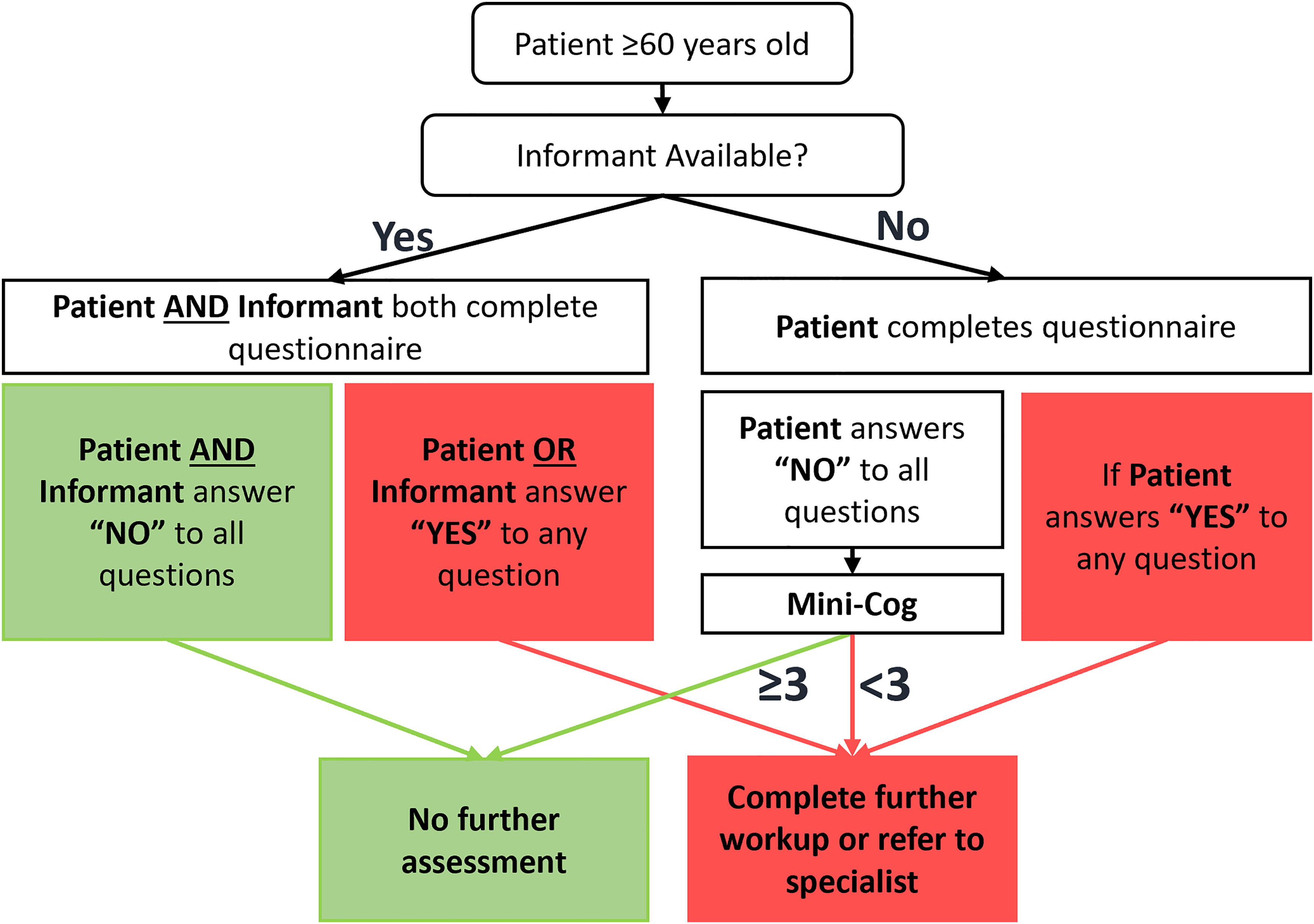
Dementia Screening Toolkit (DST) workflow.
The patient questionnaire (Supplemental Figure 1) asks the patient, “Do you think your memory or thinking has changed within the last 5–10 years?”, “Have you noticed changes in your language within the last 5–10 years?”, and “Have you noticed changes in your mood or personality within the last 5–10 years?” with examples for each question. We included additional examples to the questions from the CDPH DST. 30 For instance, “Have you had trouble recalling important events, planned activities, or conversations?” is an example under “Do you think your memory or thinking has changed within the last 5–10 years?” and “Do you feel that you have trouble following/understanding/comprehending what you are hearing on the TV, radio, or when you are reading?” is an example under “Have you noticed changes in your language within the last 5–10 years?”. We chose to ask about experiences within the last 5–10 years because Alzheimer's disease and related dementias often develop gradually, with changes in function or behavior emerging well before a formal diagnosis.
Additionally, we adapted the CDPH DST questionnaire for informants (Supplemental Figure 2). The informant is asked the same three questions about the patient. The informant examples vary slightly from the patient examples, as the informant might notice changes about the patient that the patient would not notice about themselves. For instance, “Have you noticed [patient's name] getting lost or confused in familiar places (public transportation, in the neighborhood)?” is an example under “Do you think their memory or thinking has changed within the last 5–10 years?” and “Have you noticed that they talk less in social settings?” is an example under “Have you noticed changes in their language within the last 5–10 years?”
Spanish cultural and linguistic adaptation
A Spanish-language version of the questionnaire (Supplemental Figures 3 and 4) was developed, which was adapted to align with the cultural nuances and linguistic preferences commonly observed within the Hispanic/Latino community. To achieve this, an informal review of instrument content was conducted by native speakers and other stakeholders, including a face validity assessment, as well as forward and backward translations. Pilot testing with a sample of native speakers was completed to evaluate content through cognitive testing of the adapted measure and cultural priming using language, storytelling, and narrative methods. In addition to linguistic translation, adaptations were made to reflect culturally relevant communication styles, such as warm and respectful tone (personalismo and respeto), and to incorporate examples that reflect common values, health beliefs, and family-centered decision-making practices. The translation process was finalized with role-playing intervention sessions involving research team members who share the cultural background of many anticipated participants. These sessions also helped ensure that the screener would be perceived as trustworthy and approachable, which is especially important in cultures where stigma around memory loss may delay help-seeking. With these edits, the questionnaires and examples became more conversational after the translations.
Primary care clinic implementation
The questionnaire was administered to patients 60 years and older before their annual wellness visit (AWV) or long visit at one family medicine clinic, the Family Health Center (FHC) clinic in Santa Monica, California. The Family Health Center is an academically affiliated primary care clinic that trains residents and serves a multiracial, multiethnic primary care population in Los Angeles County. The questionnaire was only available once a calendar year per patient. Prior to the study's implementation, there was no dementia screening tool or program in this clinic.
Electronic health record integration
We uploaded the patient and informant questionnaires in both English and Spanish versions to the UCLA Health EHR forms portal. The forms portal upload was a necessary EHR compliance step to enabling pre-visit and day-of visit questionnaire completion. Pre-visit, the questionnaire was sent to patients with an EHR (MyChart) account, along with other standard check-in questionnaires. The questionnaire was sent in the patient's preferred language, English or Spanish. If the questionnaire was not completed via MyChart ahead of time, the rooming staff asked the patients the three questions. Within the EHR interface, the questionnaires were available on the rooming staff's “Rooming” tab, facilitating integration into existing workflows. If an informant was present for the visit, the rooming staff asked the patient if they could ask the informant the DST questions. The questionnaires, for patient and informant, could be completed in-person in English and Spanish.
The Mini-Cog assessment was embedded into the EHR. Primary care physicians completed the Mini-Cog, and it was displayed in the “Dx/Orders” tab within the EHR. Based on feedback from providers using the tool, the Mini-Cog was adapted to always appear on the Dx/Orders tab, even when it was not required based on the DST criteria.
With the questionnaires and the Mini-Cog available in the EHR, we built a dotphrase to pull the results of both the questionnaires and Mini-Cog into the provider's visit notes (Supplemental Figure 5). The dotphrase pulled the results into a table, the DST result, and potential next steps. The two sentences are, “The dementia screening toolkit is Positive and recommends further workup or referral” or “The dementia screening toolkit is Negative and recommends no further assessment”.
Staff and provider training
After the DST went live in September 2022, we deployed in-person and virtual training sessions for the providers and staff asking the questionnaires. Additionally, we had a dedicated staff member in the clinic 50% of the time to answer any questions and collect feedback (Supplemental Figures 6 and 7). For instance, we facilitated the option for completing the Mini-Cog even outside of the DST workflow. In addition, we received feedback to include the Mini-Cog in the printed patient packet for providers. Our clinic staff began attaching a Mini-Cog questionnaire (double sided with one side in English or Spanish and on the other side a picture of a circle drawn for the clock), to all patient packets applicable for DST evaluation. This adjustment allowed providers to improve incorporation of the Mini-Cog into their standardized workflow.
Pre-post intervention study design
We included people 60 years and older, scheduled for an in-person new/long visit at the FHC, who had a unique medical record number (MRN), and did not have a dementia diagnosis prior to the visit. In the pre-period, if a patient had multiple new/long visits, the earliest visit was used. For example, if someone was seen in the FHC on February 7, 2023, and again on February 10, 2024, we evaluated their February 7, 2023, visit. In the post-period, the first screening visit was used if available; otherwise, the first recorded visit was used (Figure 2).
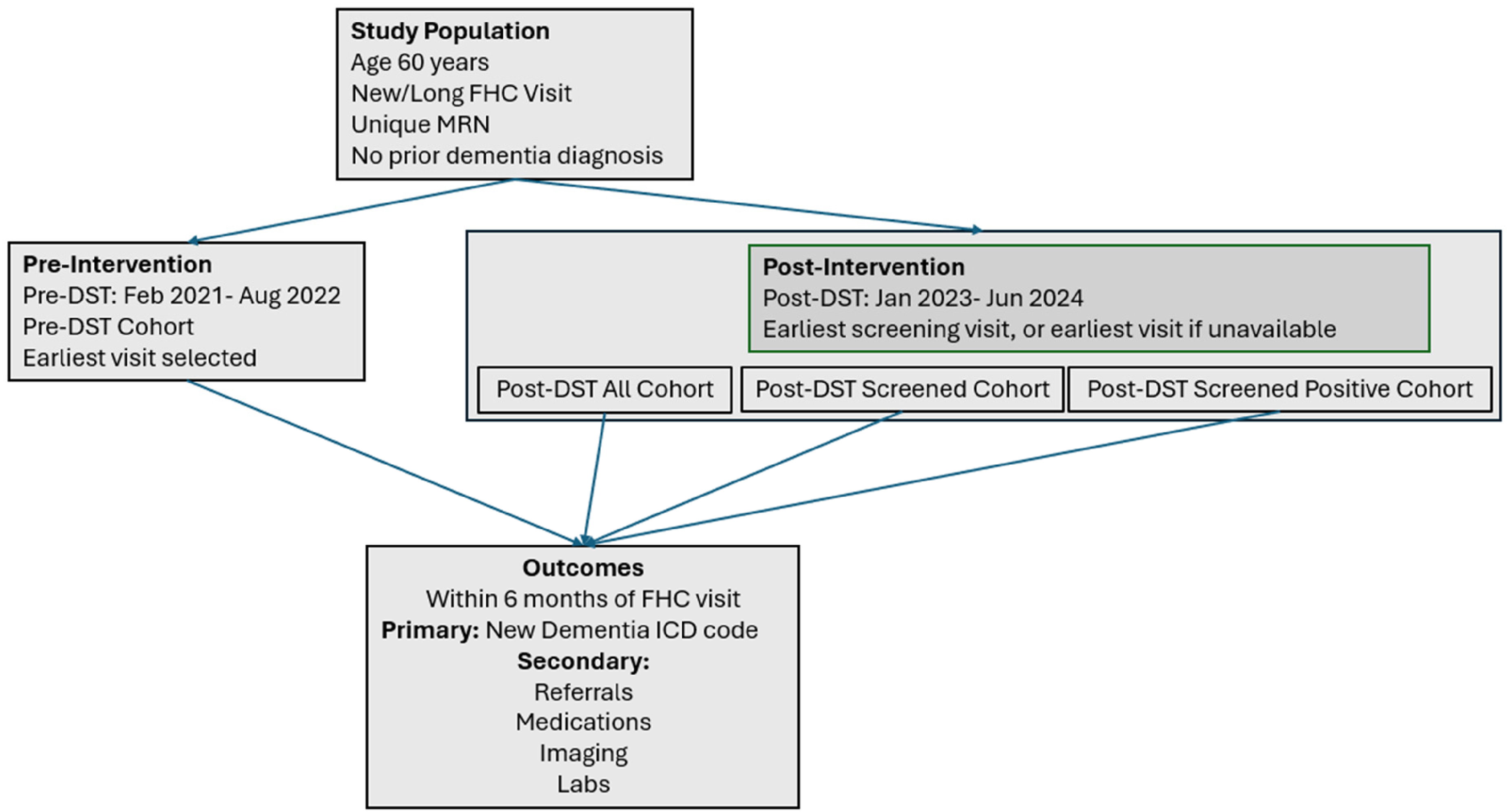
Pre-post intervention study design for dementia screening at the FHC. FHC: Family Health Center; MRN: medical record number; DST: Dementia Screening Toolkit; ICD: International Classification of Diseases.
For the pre-intervention period, “pre-DST” refers to patient data collected before the project implementation, covering the period from February 2021 to August 2022. “Post-DST all” refers to all patient data collected four months after the project implementation (January 2023 to June 2024), if they did or did not undergo screening. We chose a 4-month interval after the implementation to allow sufficient adjustment time for providers to become familiar with the DST workflow and ensure the test was being administered consistently and appropriately. “Post-DST screened” refers to the data of patients who were screened during the same period from January 2023 to June 2024. For this cohort, we included only patients with at least one recorded questionnaire response or Mini-Cog score. Patients with all items missing were classified under “post-DST all”. “Post-DST screened positive” refers to the data of patients who screened positive within the period from January 2023 to June 2024. For patients who did not have a prior International Classification of Diseases (ICD) code of dementia or cognitive change (F02.80, F02.81, F03.9, F03.90, F03.91, G30, G30.0, G30.1, G30.8, G30.9, G31.0, G31.01, G31.09, G31.1, G31.83, F03, F01, F01.5, F01.50, F01.51, G31.84, G31.85, G23.1, and F41.3), we evaluated if patients had one of the following outcomes within six months of a FHC new/long visit. The primary outcome was a new dementia/cognitive change ICD code (F02.80, F02.81, F03.9, F03.90, F03.91, G30, G30.0, G30.1, G30.8, G30.9, G31.0, G31.01, G31.09, G31.1, G31.83, F03, F01, F01.5, F01.50, F01.51, G31.84, G31.85, G23.1, and F41.3). ICD codes are assigned by clinicians as part of routine clinical care for each encounter. Our secondary outcomes included: specialty referrals (Alzheimer's Disease, Behavioral, Geriatric Medicine, Alzheimer's and Dementia Care Program, and Neuropsychological Testing), medication prescribed (donepezil, galantamine, rivastigmine, memantine, and lecanemab), imaging orders (MRI brain without contract, MRI brain with and without contrast, PET CT Brain, and CT head without contrast), and labs ordered (vitamin B12).
Statistical analysis
We compared primary and secondary outcomes of the three post-DST cohorts (post-DST all, post-DST screen, post-screened positive) to the pre-DST period using Firth penalized logistic regression to account for sparse events in dementia diagnosis and medication prescription outcomes. Covariates included age at visit, sex, ethnicity (Hispanic/Latino), and race (White, Black/African American, Asian, and Others). Unknown or missing race was categorized as “Others” and unknown or missing ethnicity was categorized as “Not Hispanic/Latino”. We additionally adjusted for Annual Wellness Visits, a preventive care framework that requires cognitive impairment assessment and has been associated with increased identification of MCI and earlier dementia diagnoses, even in the absence of additional screening interventions.29,31 An AWV was indicated by the CPT codes G0438, G0439, G0402, 99386, 88387, 99396, 99397. We stratified our results by sex. The likelihood ratio test was used to compare post-DST to pre-DST.
We calculated the sensitivity and specificity of patients who were screened compared to patients with or without a new ICD code dementia diagnosis within 6 months of screening. To evaluate reliability, we stratified results by the provider who performed the most screenings compared to others. Although there were only 15 patients with a new dementia diagnosis, we evaluated content validity by comparing patients who had an MCI diagnosis compared to other dementia diagnoses. We reported statistical significance for sensitivity using Fisher's exact test and for specificity using a two-sample z-test for proportions.
Ethical considerations
This project was evaluated and considered a quality improvement study by the UCLA Institutional Review Board, which did not require informed consent.
Results
Table 1 compares patient demographics and outcomes across four groups: pre-DST, post-DST all, post-DST screened, and post-DST screened positive. Among patients aged 60 years or older who were scheduled for an in-person new or long visit at the FHC and had a unique MRN, the sample sizes were 1387 (pre-DST), 1561 (post-DST all), 1283 (post-DST screened), and 517 (post-DST screened positive). After excluding patients with a dementia diagnosis prior to their earliest visit or screening visit, the sample sizes were 1336 (pre-DST), 1515 (post-DST all), 1249 (post-DST screened), and 489 (post-DST screened positive). Table 1 therefore presents two cohorts: (1) all eligible patients meeting age and visit criteria, and (2) a restricted cohort excluding those with preexisting dementia. The restricted cohort was used for all pre–post intervention analyses.
Data distribution of demographics and clinical outcomes pre- and post-Dementia Screening Toolkit (DST). N (Demographics) represents all patients aged 60 years or older who were scheduled for an in-person new or long visit at the FHC and had a unique MRN. N (Outcomes) represents the subset of these patients after excluding those with a dementia diagnosis prior to their earliest visit or screening visit.
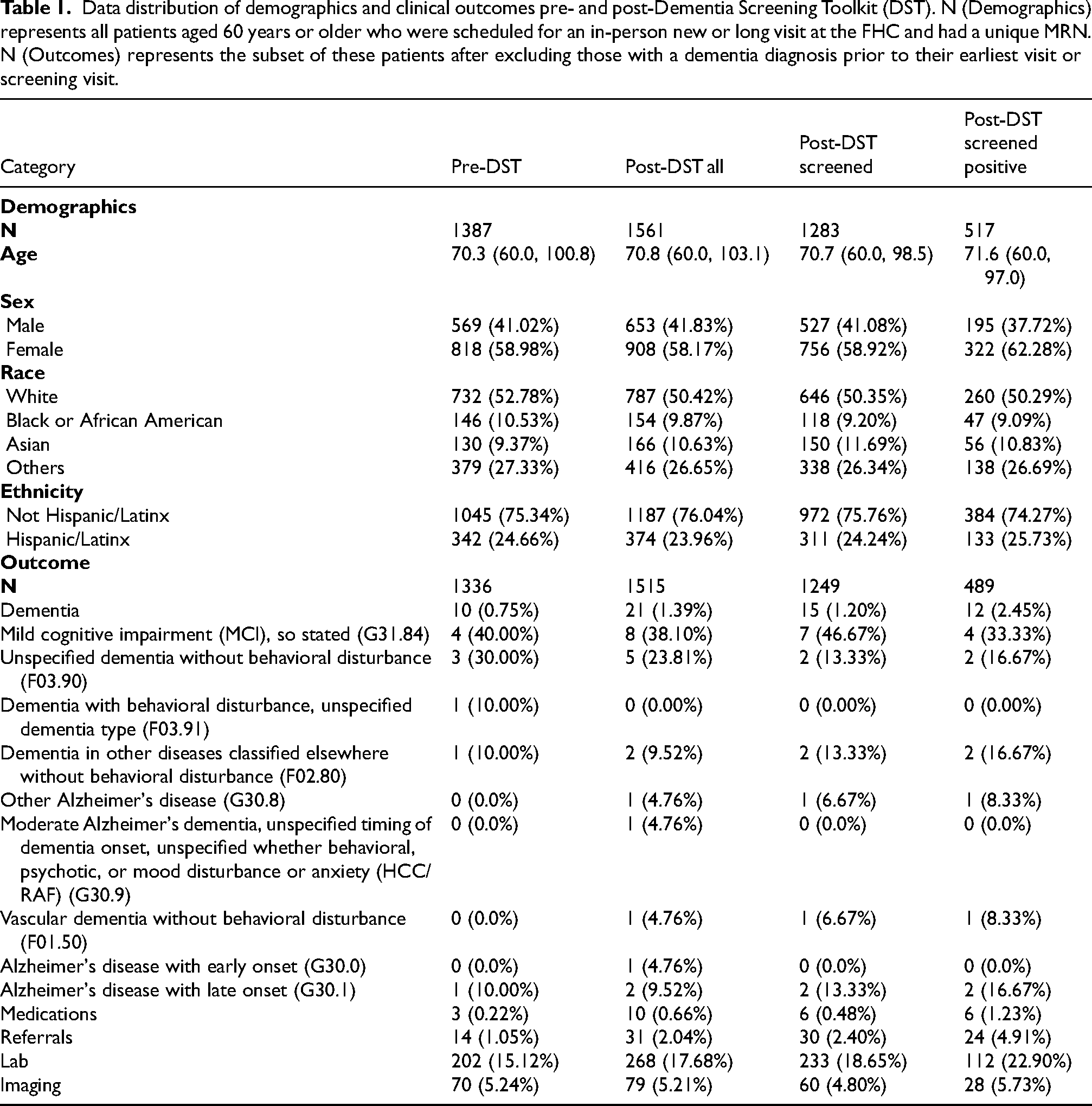
The mean age of patients slightly increased from 70.3 years in the pre-DST group to 70.8, 70.7, and 71.6 years in the post-DST all group, the post-DST screened group, and the post-DST screened positive group, respectively. Females consistently made up a larger proportion of the groups where they constituted 58.2% to 62.3% of the post-DST groups. The proportion of Hispanic/Latino patients was similar across groups: 24.7% pre-DST, 24.0% post-DST all, 24.2% post-DST screened, and 25.7% post-DST screened positive.
Figure 3 (left panel) shows the racial and ethnic distribution of patients who were screened versus not screened patients during the post-DST period from January 2023 to June 2024. Among the patients screened, 11.6% were Asian, 9.2% were Black, 15.4% were Hispanic/Latino, and 50.4% were White.
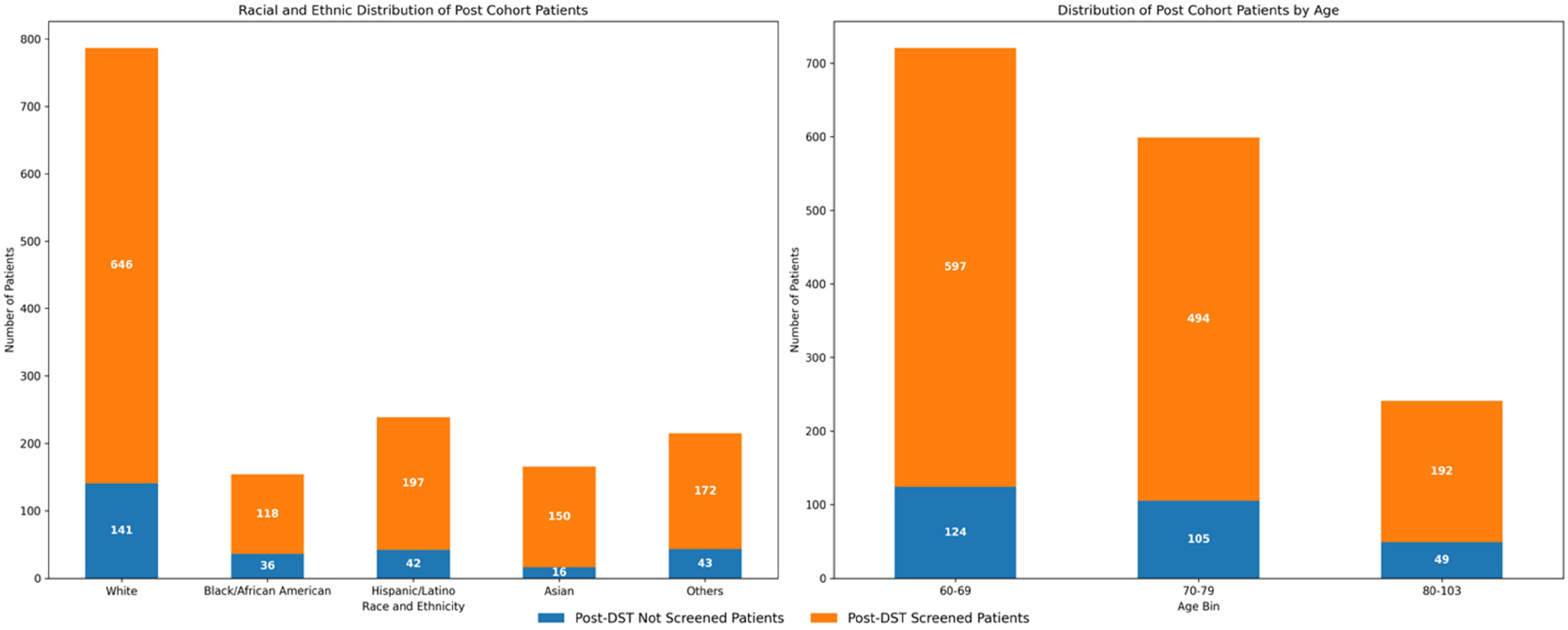
Post-Dementia Screening Toolkit (DST) analysis: racial, ethnic, and age distribution of screened and non-screened patients.
Figure 3 (right panel) shows the age distribution of patients who were screened or not screened during the post-DST period. Younger age groups represented a larger portion of the screened population due to their higher overall representation in the dataset. The percentage of screened patients was 82.8%, 82.5% and 79.7% for patients 60–69, 70–79 and 80 and over respectively.
Outcomes
New dementia diagnoses increased from 0.8% in the pre-DST group to 1.4% in the post-DST all group and to 1.2% in the post-DST screened group, with a marked increase to 2.5% in the post-DST screened positive group (Table 1). The most common dementia and cognitive diagnoses included MCI (G31.84), unspecified dementia without behavioral disturbance (F03.90), and dementia in other diseases classified elsewhere without behavioral disturbance (F02.80).
Dementia medication prescription also increased from 0.2% pre-DST to 0.7% post-DST all, 0.5% post-DST screened, and 1.2% post-DST screened positive. Referrals were more frequent post-DST, increasing to 2.0% in the post-DST all group, 2.4% in the post-DST screened group, and 4.9% in the post-DST screened positive group. Lab orders were commonly utilized, increasing from 15.1% pre-DST to 22.9% post-DST screened positive.
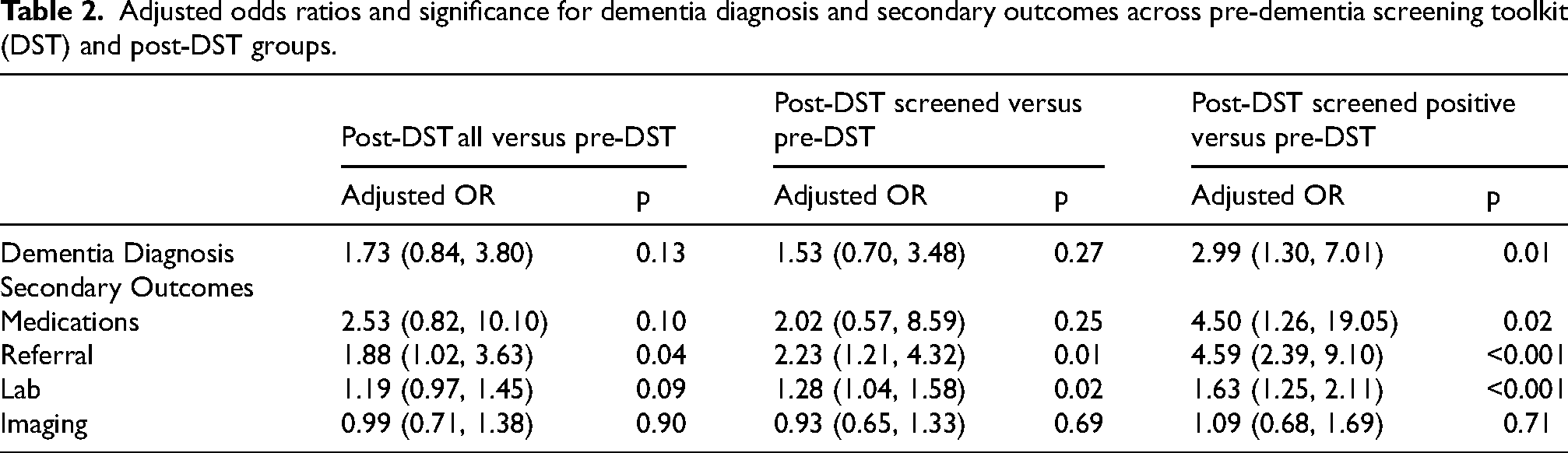
We conducted logistic regression modeling to evaluate the effectiveness of the DST in a pre-post intervention study design. We evaluated three comparisons: post-DST all versus pre-DST, post-DST screened versus pre-DST, and post-DST screened positive versus pre-DST.
The adjusted odds ratio of new dementia diagnoses significantly increased in the post-DST screened positive group (adjusted OR 2.99; 95% CI 1.30, 7.01; p = 0.01). Significant increases were also observed in multiple secondary outcomes (Table 2). Medication prescription was notably higher in the post-DST screened positive group relative to the pre-DST group (adjusted OR 4.50; 95% CI 1.26, 19.05; p = 0.02). Referral rates likewise rose significantly in the post-DST screened positive group compared to the pre-DST group (adjusted OR 4.59; 95% CI 2.39, 9.10; p < 0.001), as did lab tests (adjusted OR 1.63; 95% CI 1.25, 2.11; p < 0.001). There was no significant increase in imaging orders.
Adjusted odds ratios and significance for dementia diagnosis and secondary outcomes across pre-dementia screening toolkit (DST) and post-DST groups.
To account for the effects of temporal factors independent of the DST intervention, we additionally examined outcome prevalence over time by plotting the proportion of patients with each primary and secondary outcome in six-month intervals (Supplemental Figure 6). During the pre-DST period, we observed minimal changes in the prevalence of new dementia diagnoses, medication orders, and referral orders, along with a downward trend in laboratory and imaging orders. In contrast, we observed a pronounced upward trend following the third pre-DST time interval (after DST administration), especially among the post-DST screened positive group. Together, these findings suggest that secular temporal trends alone are unlikely to explain the observed increases in outcomes, which are more consistent with an association with DST screening.
We further stratified analyses by sex in the post- versus pre-DST comparisons. In males, new dementia diagnoses (adjusted OR 5.20; 95% CI 1.42, 22.71; p = 0.01) were significantly increased in post-screened positive individuals. Referrals and laboratory orders were significantly increased in both post-screened positive males (referrals: adjusted OR 6.42; 95% CI 1.88, 26.67; p = 0.002; lab orders: adjusted OR 1.55; 95% CI 1.02, 2.32; p = 0.04) and females (referrals: adjusted OR 3.96; 95% CI 1.87, 8.71; p < 0.001; lab orders: adjusted OR 1.68; 95% CI 1.20, 2.35; p = 0.003). Stratified analyses by sex were not significant for other post-DST to pre-DST comparisons.
Diagnostic performance
We next evaluated the diagnostic performance of the DST. Our test achieved a sensitivity of 0.8 (N = 15 patients with a dementia diagnosis within 6 months of screening) and a specificity of 0.61 (N = 1234 patients without a dementia diagnosis within 6 months of screening). To account for potential provider bias, we stratified results by the provider performing the most screenings versus others. Sensitivities were similar between groups (provider performing most screening: 0.75, other providers: 0.82, p = 1), whereas specificities differed (provider performing most screening: 0.54, other providers: 0.63, p = 0.04). We further evaluated performance by dementia diagnosis, comparing patients with MCI to those with other dementia diagnoses, as MCI patients present milder symptoms that can reduce screening sensitivity. Sensitivity was lower among patients with MCI (0.69) compared to those with other dementia diagnoses (0.93), although this difference was not statistically significant (p = 0.16), likely due to limited sample size. Specificity was the same for both groups (0.61 versus 0.61; p = 0.88). Across all stratifications, the DST had high sensitivity and moderate specificity.
Discussion
The implementation of the culturally and linguistically responsive DST integrated in the EHR improved new dementia diagnoses and workup within a diverse patient population, particularly in individuals who tested positive during screening. The quality improvement study results showed a higher odds of new dementia diagnosis post-screening. The post-DST screened positive group represented a higher dementia risk group. Additionally, the screening intervention was associated with higher adjusted odds of secondary outcomes such as dementia medication prescriptions, referrals, lab tests, and imaging, highlighting its broader impact on patient management and care. Our findings underscore the value of dementia screening in this primary care clinic.
Successful integration of the DST into the EHR streamlined workflow for healthcare providers and enabled data collection and analysis. The success of the DST at the UCLA FHC presents an opportunity for its adoption in other primary care clinics at UCLA and other healthcare systems, with potential benefits for improving dementia screening and diagnosis across diverse communities.
Methodological considerations
Our bilingual and bicultural DST represented a culturally competent health intervention. The increasing diversity of patient populations presents both challenges and opportunities in delivering effective healthcare. Language and cultural differences can lead to significant disparities in health outcomes, especially in complex areas like dementia care.13,32–34 Our screening tool addressed these disparities by prioritizing accessibility and inclusivity. While our intention was to offer screening to all patients meeting inclusion criteria, real world clinical care including onboarding, training, patient preference and provider preference precluded screening of all patients when the screening tool was first released.
Implementing a dementia screening toolkit within the EHR of a diverse clinic presented challenges, such as technological barriers and patient engagement. Technological barriers posed limitations to DST adoption, as providers had varying levels of proficiency with the EHR system, requiring comprehensive training and ongoing support. Patient and provider hesitations to discuss changes in memory may deter usage, underscoring the need for more provider and patient education about dementia and potential treatment options. Overcoming these challenges requires ongoing commitment to training, technical support, and resource allocation, strategic planning and collaboration.
The initial focus on English and Spanish languages acknowledges the linguistic needs of a large segment of the primary care clinic. Unfortunately, in our EHR we were unable to track which language the DST is provided in. In future work, we are developing plans to incorporate additional languages.
Limitations
The limited sample size in this study precluded an analysis of the effectiveness of the DST within specific racial and ethnic groups. Additionally, a limitation of this study is the use of ICD codes from the EHR as an outcome. Clinicians may or may not have new standardized diagnostic criteria when assigning ICD codes in a given encounter. We did not perform manual chart review for patients who did or did not have dementia ICD code diagnosis or include Alzheimer's disease biomarkers as diagnostic criteria. As these biomarkers become increasingly used in clinical practice, we will be able to incorporate these as outcomes. Furthermore, the training sessions provided to providers and staff may have indirectly influenced them to order more labs and imaging, introducing a potential confounding factor. However, this also underscores the strength of the study, as it highlights the potential for training to enhance diagnostic practices.
Conclusion
In conclusion, our efforts to integrate a dementia screening toolkit into the EPIC EHR system at a UCLA Health primary care clinic highlights an innovative approach to patient care. Our commitment to expanding language options and continuously refining our tools reflects a dedication to minimizing healthcare disparities and improving the quality of life for individuals facing cognitive health challenges.
Supplemental Material
sj-docx-1-alz-10.1177_13872877261441776 - Supplemental material for Increased dementia diagnosis and workup in primary care with a bilingual screening tool integrated in the electronic health record
Supplemental material, sj-docx-1-alz-10.1177_13872877261441776 for Increased dementia diagnosis and workup in primary care with a bilingual screening tool integrated in the electronic health record by Samantha Shah, Thai Tran, Satpal S. Wadhwa, Gabriela Islas Huerta, Stephanie Ovalle-Eliseo, Ariadna Martinez, Keith Vossel, Blanca Campos, Michelle Bholat, Gerado Moreno, Mirella Diaz-Santos and Timothy S. Chang in Journal of Alzheimer's Disease
Footnotes
Acknowledgements
We thank all staff, providers and patients who participated at the UCLA Health Family Health Center.
Ethical considerations
This project was evaluated and considered a quality improvement study by the UCLA Institutional Review Board, which did not require informed consent.
Consent to participate
This study was conducted in accordance with the Declaration of Helsinki. It was reviewed by UCLA Institutional Review Board, IRB# 22-000456, which did not require informed consent.
Consent for publication
Not applicable
Author contribution(s)
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research made possible by the California Department of Public Health grants (23-10648, 22-10904) (SS, SSW, GIH, KV, MB, MDS, TSC), Fineberg and Bloomberg family philanthropy gifts (SS, SSW, AM), National Institutes of Health (NIH) National Institute of Aging (NIA) grant K08AG065519-01A1 (TSC), NIH/NIA UH2AG083254 (SS, KV, TSC), and residual class settlement funds in the matter of April Krueger v. Wyeth, Inc., Case No. 03-cv-2496 (US District Court, SD of Calif.) (SS, SSW, GIH, MDS, TSC).
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
This study was not pre-registered and is not publicly available as protected health information was used for analysis.
Supplemental material
Supplemental material for this article is available online.
