Abstract
Alzheimer's disease (AD) affects not only memory and cognition but also the body's automatic functions, such as heart rate and blood pressure. These changes reflect the early disruption of the central autonomic network, the system that links the brain and the heart to maintain physiological balance. Electroencephalography (EEG) measures the brain's electrical activity and reveals patterns of cortical slowing and desynchronization, while heart-rate variability (HRV) reflects how flexibly the heart responds to internal and external demands. This narrative review brings together evidence published between 2000 and 2025 on the combined use of EEG and HRV as a single, integrated biomarker for AD. Studies consistently show that EEG-HRV coupling, which reflects how well the brain and heart communicate, provides better accuracy in distinguishing mild cognitive impairment and early AD than either measure alone. The findings reveal a shared loss of co-ordination between neural and autonomic systems, which is a hallmark of neurovisceral decline. The review also identifies key methodological gaps, including inconsistent recording conditions and lack of standardized analytic methods, which currently limit reproducibility. To bridge this gap, a translational roadmap is proposed to outline short-, mid-, and long-term goals for clinical validation, wearable integration, and digital health applications. Together, EEG–HRV coupling represents a scalable, non-invasive, and physiologically grounded tool that could support earlier and personalized monitoring of AD, helping to connect laboratory discovery with real-world clinical care.
Keywords
Introduction
Alzheimer's disease (AD) is a progressive, neurodegenerative disorder and the most common cause of dementia, accounting for 60–70% of cases globally. 1 It is clinically characterized by memory loss, impaired executive function, language difficulties, motor deficits, and behavioral changes, which progressively interfere with daily functioning. Pathologically, AD is marked by two core hallmarks: extracellular amyloid-β (Aβ) plaques and intracellular neurofibrillary tangles composed of hyperphosphorylated tau protein. 2 These features are accompanied by widespread synaptic dysfunction, neuronal loss, and neuroinflammation, particularly affecting the hippocampus and associative cortical areas early in the disease course.
The pathogenesis of late-onset AD is multifactorial, influenced by genetic and environmental factors. The ε4 allele of apolipoprotein E (APOE) is the strongest known genetic risk factor for sporadic AD, 3 while factors such as cardiovascular health, metabolic disease, and education level also modulate disease risk and progression. 4 Other genes identified via genome-wide association studies play smaller roles but contribute to understanding AD biology, particularly in neuroinflammation, lipid metabolism, and tau pathology. 5 While the amyloid cascade hypothesis suggests Aβ peptide accumulation drives tau pathology and neurodegeneration, 6 recent evidence highlights neuroinflammation, vascular dysfunction, and impaired clearance mechanisms as critical contributors. 7 Notably, tau pathology and neuroinflammation may be more closely correlated with cognitive decline and disease progression than amyloid load alone,8,9 broadening perspectives on AD pathophysiology. Clinical studies have further revealed that patients with AD frequently exhibit cardiac abnormalities, including diastolic dysfunction, increased arterial stiffness, prolonged atrial conduction, and enlarged left ventricular diameter, features that have been associated with increased brain Aβ deposition. 10
Recent evidence highlights an important distinction in how AD develops. Although the accumulation of Aβ is thought to initiate the disease process, amyloid burden on its own shows a surprisingly weak relationship with memory loss or cognitive decline. Many older adults with completely normal cognition still show high levels of amyloid on positron emission tomography (PET) imaging, a phenomenon often referred to as the “amyloid paradox”. 11 In contrast, tau pathology and neuroinflammation show a far closer alignment with clinical progression. The spread of abnormal tau tangles follows a predictable pattern through the brain that mirrors worsening cognitive symptoms.12,13 At the same time, chronic activation of microglia, the brain's immune cells, drives persistent inflammation, synaptic injury, and neuronal loss, making these processes more immediate contributors to cognitive decline.8,14,15 Together, these findings support a more integrated model of AD pathogenesis. Rather than being driven by a single pathological event, the disease reflects interacting processes involving amyloid accumulation, tau-mediated neurodegeneration, immune activation, vascular dysfunction, and synaptic loss. In this framework, amyloid may set the stage, but tau pathology and sustained neuroinflammation appear to be the more direct drivers of the symptoms that eventually emerge. Despite decades of research, effective disease-modifying treatments for AD remain elusive due to the biological complexity and clinical heterogeneity of the disorder. The heterogeneous etiology of AD, encompassing genetic, inflammatory, and neurodegenerative pathways, limits the efficacy of therapeutics that target a single mechanism, such as amyloid removal, especially in later stages of the disease. Additionally, the long preclinical phase, where pathological changes occur 10 to 20 years before symptoms, results in diagnoses following substantial neuronal damage. 16 While traditional approaches focus on neuropathological markers, such as amyloid and tau, evidence points to a multifaceted disease process involving neuroinflammation, metabolic dysregulation, and autonomic dysfunction. Combining circulating biomarkers, such as inflammatory cytokines, neurofilament light chain, and metabolic indicators, with physiological signals like electroencephalography (EEG) and electrocardiogram (ECG), offers a holistic and more accessible framework for early detection and disease monitoring (see Figure 1).
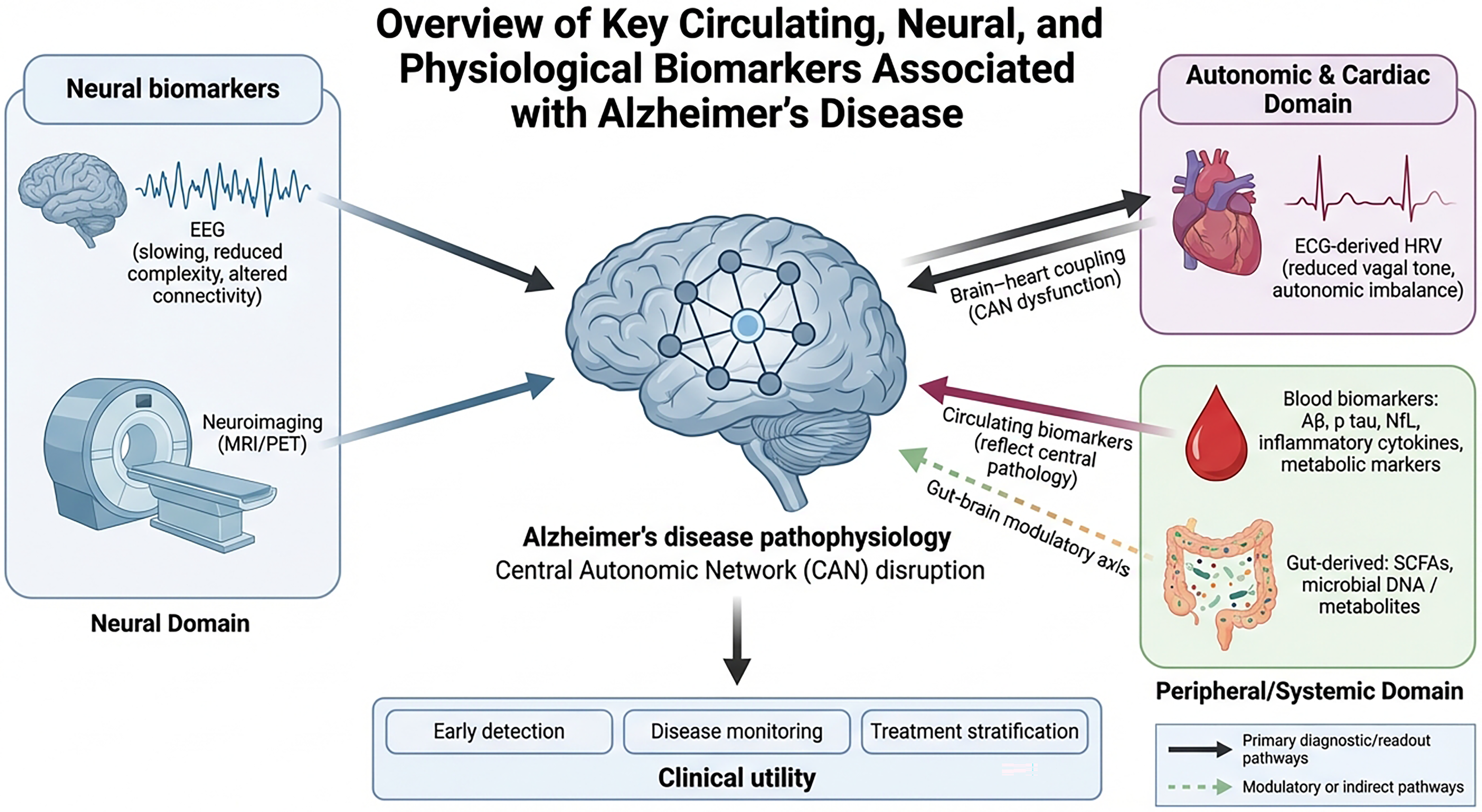
The central schematic illustrates Alzheimer's disease pathophysiology and disruption of the central autonomic network, which links brain activity to autonomic control of the heart. Surrounding panels show three biomarker domains: (i) neural markers, including electroencephalography and MRI/PET neuroimaging, indexing cortical slowing, reduced network complexity, and neurodegeneration; (ii) autonomic-cardiac markers, represented by ECG-derived heart rate variability, reflecting reduced vagal tone and autonomic imbalance; and (iii) peripheral/systemic markers, including blood biomarkers (amyloid-β, phosphorylated tau, neurofilament light chain, inflammatory cytokines, and metabolic indicators) and gut-derived metabolites or microbial signatures (e.g., short-chain fatty acids, microbial DNA/metabolites) that interact with the brain via a gut-brain modulatory axis. Solid arrows denote primary diagnostic or readout pathways, whereas dashed arrows indicate modulatory or indirect influences, and the lower panel highlights how integrating these signals may support early detection, longitudinal monitoring, and treatment stratification in AD. (Initial form of Figure 1 created with ChatGPT-4o, OpenAI).
The blood-brain barrier (BBB) restricts drug delivery to the central nervous system (CNS), posing significant challenges for AD treatment.17,18 Additionally, disease heterogeneity, including genetic, comorbid, and environmental variability, further complicates clinical presentations and treatment responses. 1 These factors underscore the difficulty of developing effective and generalizable therapies, highlighting the need for early detection strategies and multimodal approaches. Recent advances in blood-based biomarkers, such as Aβ and p-tau217 ratios, alongside established cerebrospinal fluid (CSF) markers, are improving diagnostic precision and clinical trial stratification. This progress addresses the historical lack of reliable biomarkers, which has long hindered early detection and therapeutic development.
Pharmacological interventions for AD primarily manage symptoms without halting or reversing disease progression. The most widely used drugs are cholinesterase inhibitors, such as donepezil, rivastigmine, and galantamine, which aim to enhance cholinergic transmission by inhibiting the breakdown of acetylcholine, a neurotransmitter typically depleted early in AD pathology, offering modest cognitive improvements in early to moderate stages, but with limited efficacy and side effects such as nausea and bradycardia. 19 Another class of drugs includes N-methyl-D-aspartate (NMDA) receptor antagonists, such as memantine, which modulate glutamatergic activity to reduce excitotoxic neuronal damage. Memantine is approved for moderate to severe AD and has shown some benefit in preserving cognitive and functional abilities. 20 However, neither drug class alters disease progression, with effects on cognition and overall outcomes remaining limited.
Recent therapeutic strategies focus on clearance or neutralization of Aβ plaques, utilizing anti-amyloid monoclonal antibodies, such as aducanumab, lecanemab, and donanemab. These drugs aim to slow cognitive decline by targeting aggregated forms of Aβ, thus promoting clearance of amyloid deposits, with PET imaging showing plaque reduction. Despite their controversial clinical efficacy, some of these drugs received conditional approvals by the FDA. Lecanemab has shown statistically significant slowing of cognitive decline in early AD (e.g., CLARITY-AD study); however, cognitive benefits remain modest and inconsistent, and safety concerns over amyloid-related imaging abnormalities (ARIA) and brain swelling were raised.21,22 Nevertheless, lecanemab has received approval from the U.S. Food and Drug Administration and European Medicines Agency and is expected to enter clinical use in several European countries. In contrast, the commercial discontinuation of aducanumab by Biogen was ultimately a strategic business decision, rather than a regulatory action based on safety concerns. Although the drug generated significant debate due to its modest clinical benefit and the need for careful monitoring of ARIA, including brain swelling and microhemorrhages, the company's choice to halt development and commercialization reflected broader commercial and logistical challenges rather than evidence of new safety risks. 23 In the case of donanemab, gradual up-titration of the dose has been shown to significantly reduce the risk of ARIA-E, while maintaining comparable pharmacokinetic and pharmacodynamic profiles to standard dosing. 24 Despite progress, these treatments are costly, require intravenous administration, and carry logistical challenges for widespread use.
Taken together, these therapies highlight the limitations of current approaches that primarily address downstream effects or singular pathological targets. The modest efficacy, high cost, and risk profile of new drugs illustrate the need for alternative therapeutic strategies that go beyond amyloid and cholinergic pathways. There is a need for comprehensive strategies that integrate early diagnosis, multifactorial interventions, and system-level biomarkers, to better address the complexity of AD.
Recent research has highlighted the importance of the central autonomic network (CAN), a distributed system of cortical and subcortical brain regions, in coordinating cognitive, emotional, and autonomic functions. Core CAN structures, including the anterior cingulate cortex, insula, amygdala, and brainstem nuclei, mediate bidirectional communication between the brain and heart.25,26 In neurodegenerative diseases such as AD, dysfunction within this network is thought to underlie both cognitive decline and autonomic dysregulation.27,28 Importantly, CAN function can be non-invasively probed through measures of cortical activity and autonomic output, making it highly relevant to research on neurodegenerative diseases. EEG provides real-time insights into cortical neuronal activity, offering valuable information on cognitive function and neurophysiological alterations. Autonomic nervous system function and cardiovascular dynamics, both of which are increasingly recognized as integral to brain health, are reflected in heart-rate variability (HRV), estimated from ECG activity. Both EEG and ECG modalities are under CAN control.26,29
While several recent reviews have explored aspects of brain–heart interactions, most have done so within broader or single-modality frameworks. For example, some have examined brain–heart coupling across multiple neurological disorders, 10 others have focused on HRV and cognitive performance in general populations,30–32 and a few have centered exclusively on electroencephalography as an early diagnostic tool for AD.33,34 However, these studies typically treat EEG and HRV as separate physiological measures or as part of generalized models of neurodegeneration.
The present review takes a more integrative and disease-specific perspective by examining EEG-HRV coupling as a unified biomarker framework for AD. It highlights four key dimensions: (1) the mechanistic foundation of this interaction through the CAN; (2) evidence from studies that have directly quantified brain–heart coupling metrics in mild cognitive impairment (MCI) and AD; (3) the translational pathway linking these findings to emerging digital and clinical applications; and (4) novel therapeutic approaches aimed at restoring CAN function. Together, these elements position EEG-HRV coupling as a physiologically grounded, scalable, and clinically relevant tool for early diagnosis and personalized monitoring of AD.
Review approach
This review followed a narrative synthesis framework, meaning that evidence was carefully collected, interpreted, and discussed to build a clear, integrated understanding of how the brain and heart interact in AD. Although not a full systematic review, it was guided by systematic principles to ensure transparency and balance.
Peer-reviewed studies were identified through major scientific databases such as PubMed, Scopus, and Google Scholar using combinations of the keywords “Alzheimer's disease,” “EEG,” “ECG,” “heart rate variability,” “brain–heart coupling,” and “central autonomic network.” The search covered the years 2000 to 2025, focusing on both human studies and translational research that link brain activity (EEG) and autonomic function (HRV or ECG) in AD. Reference lists of important papers were also screened to capture any additional relevant work.
Studies were included if they examined EEG, HRV, or both in people diagnosed with AD, MCI, or healthy age-matched controls. This approach provided a broad yet focused overview, emphasizing methodological differences, sample sizes, and clinical implications rather than aiming to list every single study. In doing so, it highlights not only what is known, but also where the field still needs clearer standards and deeper integration across disciplines.
A narrative approach was chosen because it allows integration of diverse study designs and analytical methods, capturing the conceptual and translational landscape of EEG-HRV research in AD, rather than restricting the discussion to narrow inclusion criteria typical of systematic reviews.
Electroencephalogram in AD
EEG captures electrical neural signals originating from the cerebral cortex 35 and is a valuable, non-invasive and cost-effective method in the clinical setting for diagnosing neurological conditions, such as epilepsy and sleep disorders.35,36 EEG recordings are characterized by distinct frequency bands, including delta (0.5–4 Hz), theta (4–7 Hz), alpha (8–12 Hz), sigma (12–16 Hz), and beta (13–30 Hz), each associated with specific physiological and cognitive states. 37 One of EEG's notable strengths lies in its ability to track real-time neurophysiological changes. This makes it particularly useful in studying dynamic brain function and behavioral responses. 38 However, it has limited spatial resolution, which can complicate spatial interpretation during clinical assessments. 39
EEG as an early diagnostic and biomarker tool
In the context of AD, EEG has gained increasing attention not only for monitoring neural dysfunction but also for its potential as an early diagnostic tool, particularly considering the relationship between specific EEG markers and cognition (for example, see Alba et al. 40 ). Several studies have explored EEG features that may reflect AD-related pathology, especially in the preclinical and MCI stages. This has prompted ongoing efforts to validate EEG as a cost-effective alternative to more invasive or expensive diagnostic methods such as PET imaging or CSF analysis. Several studies to date have identified AD-specific changes in EEG features that support the development of EEG-based biomarkers that can set AD apart from healthy aging as well as other types of neurodegeneration.
In a study by Lin et al. it was examined how large-scale brain networks during relaxed wakefulness are altered in subjective cognitive decline (SCD, n = 30), MCI (n = 51) and AD (n = 28) patients, compared to healthy controls (n = 168), using the debiased weighted phase lag index (dwPLI), a measure of functional connectivity in EEG recordings. 41 Their findings revealed two distinct patterns of disruption. First, AD was associated with global reductions in connectivity across the alpha and lower-beta (13–20 Hz) bands, indicating a widespread breakdown in the brain's ability to coordinate activity across distant regions. Second, more focal, region-specific reductions were observed in the higher-beta (20–30 Hz) and lower-gamma (30–40 Hz) bands, particularly affecting the temporal and occipital lobes. The regions are critical for memory, visual processing, and multimodal integration. Together, these global and regional disturbances reflect advanced disintegration of the brain's communication networks. In contrast, individuals with MCI and SCD showed more localized and distinct connectivity profiles. SCD was characterized by increased theta-band connectivity across posterior regions, with only subtle alterations in alpha and gamma activity. MCI displayed fewer overall changes than AD but showed early reductions in theta connectivity and emerging disruptions in frontal and temporal pathways. Compared with healthy aging, this progression, from subtle reorganization in SCD to selective network vulnerability in MCI to widespread breakdown in AD, illustrates how EEG functional connectivity can map the gradual deterioration of large-scale brain systems. These evolving patterns underscore the potential of EEG as a sensitive biomarker for detecting early disease-related changes and for distinguishing AD from other neurodegenerative disorders.
In a large-scale study by Jiao and colleagues involving 890 participants, resting-state EEG data were analyzed to assess both spectral and connectivity parameters in individuals with MCI (n = 189) and AD (n = 330). 42 Their findings demonstrated that EEG features could accurately differentiate healthy controls (n = 246) from affected individuals, achieving a classification accuracy of approximately 70%. Key EEG markers in this study included Hjorth parameters (activity, complexity, mobility) and spectral power distributions. Notably, increased theta power, particularly in the occipital regions, was observed in individuals with MCI and AD, though this is more pronounced in dementia with Lewy bodies (patients with other dementias, n = 125), while relative decreases in beta activity were also reported. These shifts in oscillatory patterns appeared to correlate with core AD pathologies, such as elevated phosphorylated tau (p-tau) and reduced Aβ42. Further, the study found that Hjorth mobility in parietal and occipital regions (e.g., O1, O2, P4) positively correlated with cognitive scores, including the Montreal Cognitive Assessment (MoCA) and Mini-Mental State Examination (MMSE). This suggests that changes in EEG complexity may reflect underlying cognitive decline. 42 Supporting these results, Chetty et al. also found increased theta power during resting state across multiple brain regions in AD patients (n = 82), as well as in patients with prodromal AD (n = 30), compared to healthy controls (n = 105), which was inversely associated with MMSE performance. 43
To understand the mechanisms behind these EEG alterations, researchers have explored the role of hippocampal theta oscillations, which is critical for memory formation and cognitive processing.44,45 Normally, theta rhythms help organize neural activity via theta-gamma cross-frequency coupling, especially during memory encoding.46,47 However, in AD, soluble Aβ oligomers are known to disrupt this process. This disruption may occur through early hyperactivity and eventual degeneration of parvalbumin-positive interneurons, which play a central role in maintaining excitatory-inhibitory balance.47,48 As these interneurons degenerate, theta-gamma coupling is weakened, resulting in fragmented neural rhythms and impaired memory consolidation.46,47,49 Cholinergic deficits further compound this disruption by reducing theta power during cognitive tasks. 50 Resting-state EEG (rsEEG) studies have shown converging evidence of abnormalities in patients with amnestic MCI and AD, particularly within the delta, theta, and alpha frequency bands. 34 These disruptions have been demonstrated consistently across independent, multicenter investigations and various analytic approaches, at both group and individual levels. Furthermore, these rsEEG abnormalities correlate with established biomarkers of AD neuropathology and neurodegeneration and align with models of cortical overexcitability and hypersynchronization.
These observations suggest that early changes in EEG signals, especially increased theta activity and altered connectivity, may serve as early indicators of AD-related network dysfunction. Because these EEG patterns can emerge before overt clinical symptoms, they offer promising potential for early risk stratification and targeted intervention.
EEG for monitoring disease progression
Given the progressive nature of AD, monitoring changes in brain function over time is essential for informing treatment strategies, planning care, and potentially guiding early interventions. EEG, with its ability to capture real-time neural activity in a non-invasive and cost-effective manner, has been increasingly explored as a tool for tracking disease progression. One promising area is the use of quantitative EEG (qEEG), which involves analyzing spectral and connectivity patterns to identify electrophysiological changes associated with cognitive decline. Several studies have reported that increased theta/alpha power ratios, reduced peak frequency, and altered synchrony may serve as indicators of neurodegeneration in individuals at risk of AD. A particularly informative study by Engedal et al. employed a method known as statistical pattern recognition to track the neurophysiological progression in a large cohort of 213 participants. 51 The final cohort included healthy controls (n = 67), and individuals with MCI (n = 88), as well as individuals with SCD (n = 45), who are individuals reporting memory issues despite normal performance on standard cognitive tests. Over a follow-up period averaging 62.5 months, 35% of participants progressed to dementia, and 26% were diagnosed with AD. Using resting-state (eyes closed) EEG-derived features, the researchers constructed a Dementia Index (DI) which showed promising predictive utility, with an area under the curve (AUC) of 0.78. The DI achieved 71% sensitivity and 69% specificity in distinguishing converters from non-converters. Notably, the predictive accuracy improved when baseline cognitive assessments were combined with the EEG model, supporting the benefit of a multimodal approach for early detection. 52 The inclusion of SCD participants in this study is particularly important. These individuals often represent an early, overlooked stage in the disease continuum, and the ability of EEG to detect subtle abnormalities in this group reinforces its value in identifying preclinical AD risk.
Another study by Poil et al. investigated a similar question but with a smaller sample and shorter follow-up. 53 Among 86 MCI patients, approximately one-third converted to AD over two years. By incorporating six resting-state (eyes closed) EEG features into a logistic regression model, the researchers achieved 88% sensitivity and 82% specificity, significantly higher than any individual EEG marker alone. This improvement highlights the advantage of using combined biomarkers rather than relying on isolated EEG metrics. 53 The success of this integrated approach may reflect the complex and varied nature of AD-related neural changes. While a single EEG feature might capture only a limited aspect of brain dysfunction, such as reduced coherence or slowing in one frequency band, a combination of features can provide a more complete picture of the underlying pathology. Additionally, integrating multiple markers helps reduce the influence of confounding variables like age, medication use, or anatomical factors (e.g., skull thickness), which can otherwise introduce noise into EEG data. Importantly, these combined EEG features may capture complementary physiological processes. For example, some biomarkers may reflect disrupted synchrony in beta frequencies linked to network efficiency, while others may detect slowing of oscillations associated with synaptic loss or impaired neurotransmission. Together, they can reveal subtle, early-stage changes that precede overt clinical symptoms, offering valuable insights into disease trajectory.
In summary, EEG offers a valuable, non-invasive approach for both detecting early neural changes and monitoring the progression of AD. Its ability to capture alterations in brainwave patterns, such as increased theta power, reduced neural complexity, and disrupted theta-gamma coupling, provides important insights into the functional deterioration that underpins cognitive decline. Notably, EEG abnormalities often emerge before overt clinical symptoms, underscoring their potential as early biomarkers. Moreover, the integration of multiple EEG features has demonstrated superior predictive value over single metrics, reinforcing the utility of EEG in identifying individuals at risk and tracking disease evolution. When considered alongside physiological indicators like HRV, EEG may form part of a complementary, multidimensional strategy for AD diagnosis and management. Together, these tools have the potential to improve early detection, enhance prognostic accuracy, and guide timely interventions in vulnerable populations.
Autonomic nervous system function in AD
Emerging evidence from both clinical and preclinical studies highlights a significant bidirectional relationship between cardiovascular health and neurodegenerative disorders, particularly AD. Epidemiological data show that individuals with genetic risk factors for AD are more likely to develop cardiovascular conditions such as angina pectoris, coronary artery disease, and heart failure, all of which are associated with an increased risk of all-cause dementia, including AD. 10 Notably, earlier onset of cardiovascular disease correlates with more severe cognitive decline.
Heart rate variability
HRV refers to physiological variation in the time intervals between consecutive heartbeats. 54 The autonomic nervous system (ANS) regulates involuntary physiological functions such as blood pressure, digestion, and heart rate. It comprises three divisions: the enteric, sympathetic, and parasympathetic nervous systems. 55 The enteric nervous system primarily governs gastrointestinal functions. 56 Similarly, the sympathetic nervous system (SNS) facilitates the body's response to stress, while the parasympathetic nervous system (PNS) promotes restorative and relaxation processes. 57 Given that HRV reflects autonomic regulations, its close association with ANS activity is well established.
Notably, higher HRV is generally linked to better cardiovascular health and greater physiological adaptability to stress, contributing to overall well-being.58,59 Indeed, high HRV typically indicates predominance of PNS activity over SNS responses. For instance, in stressful situations marked by increased SNS arousal, a strong parasympathetic counter-regulation is essential for promoting recovery and restoring physiological homeostasis.29,54,60 In contrast, chronic sympathetic dominance has been associated with an elevated risk of cardiovascular disorders, including arrhythmia and hypertension.61–63
As noted earlier, the ANS governs numerous involuntary physiological processes. In addition to heart rate and blood pressure, the ANS regulates pupil diameter, digestion, sweating, and respiratory rate. Emerging evidence has linked dysregulation of these autonomic functions to pathophysiology of AD, highlighting the relevance of ANS alterations in neurodegenerative processes.
HRV and AD
AD is characterized by multiple intersecting pathophysiological mechanisms, including the accumulation of Aβ, the formation of neurofibrillary tangles, and the exacerbation of oxidative stress and neuroinflammation.64–66 These pathological features are closely linked to the progressive decline in cognitive abilities and executive functions observed in AD patients.64–66 In addition to these core pathological markers, several comorbid medical conditions as well as dysfunctions in specific brain regions have been implicated in increasing the risk of developing AD.64,67 Evidence suggests that HRV may be associated with several of these pathophysiological processes. HRV, a marker of ANS function and adaptability, has emerged as a potential non-invasive biomarker reflecting alterations in neurocardiac regulation, which may in turn be related to disease onset or progression in AD. Advanced ECG-based HRV analysis, particularly when combined with deep learning models, has revealed progressive autonomic dysregulation from normal aging through MCI to AD, with diminished parasympathetic activity and increased heart rate fragmentation, providing additional discriminatory power for early detection. 68
Additionally, a connection between HRV and cognition has been reported by Forte et al.. 31 In their systematic review, Forte et al. reported that higher resting HRV was associated with better cognitive performance, while lower resting HRV was linked to reduced prefrontal control over subcortical activity, resulting in impaired self-regulation and executive functioning. 31 Although the review highlighted evidence suggesting that increased sympathetic activity and decreased parasympathetic activity are associated with poorer cognitive outcomes, it is important to note that the included studies excluded individuals with dementia, severe cardiovascular disease, and other significant medical or psychiatric conditions.
A systematic review by Papadopoulou and colleagues found consistent evidence of dysautonomia in AD patients. 69 Across 30 eligible studies, AD patients were frequently reported to exhibit impaired autonomic regulation, including reduced heart rate variability, abnormal baroreflex sensitivity, and imbalances in sympathetic and parasympathetic tone. Although such symptoms are often underreported by AD patients, autonomic measures have, in some cases, differentiated AD patients from healthy controls and other neurodegenerative conditions, such as diffuse Lewy body disease.
Extending this line of evidence to earlier disease states, short segment ECG-derived HRV measures also differentiate individuals with MCI from cognitively healthy peers. In a cross-sectional study of 297 urban Indian adults, 19.2% of which with MCI, 10-s supine position ECGs showed significant group differences in time domain indices of HRV characterization, and simple classifiers trained on HRV features achieved about 81% accuracy when using root mean square of R-R intervals as input. 70 This data suggests that HR and HRV may provide accessible screening markers for cognitive decline before dementia is established, while underscoring the need longitudinal designs to clarify prognostic value and AD specificity.
These findings suggest that autonomic nervous system dysfunction may be an under-recognized, yet core, feature of AD, and support further investigation into the use of autonomic markers to distinguish between AD, other dementias, and healthy aging.
HRV and Aβ
The vagus nerve is a vital component of the PNS, facilitating bidirectional communication among the cardiovascular, (i.e., heart), neurological (i.e., brain), and digestive systems. It plays a regulatory role in various physiological processes, including heart rate, digestion, blood pressure, respiratory function, and mood regulation. 71 Evidence suggests that specific lifestyles practices, such as meditation, exercise, and slow, deep breathing, can activate the vagus nerve and promote physiological resilience and health benefits.72,73 In a study by Min et al., healthy adults (N = 108) were randomized into two groups (n = 54 per group). 74 One group (Osc+) performed slow-paced breathing techniques designed to increase heart rate oscillations (and thereby enhance HRV), while the control group (Osc-) engaged in breathing patterns that suppressed such oscillations. The two groups were also further stratified by age (mean ages were 22.7 and 65.9 in the younger and older group respectively). The intervention group (i.e., those with increased HRV) showed significantly lower levels of Aβ levels (Aβ42 and Aβ40). Interestingly, these reductions in Aβ coincided with downregulation of CREB (cAMP response element-binding) proteins, key transcription factors involved in neuronal survival, synaptic plasticity, and Aβ clearance. 74 This finding appears counterintuitive, as CREB is typically protective in AD pathology. However, several explanations may account for this apparent disparity. Firstly, CREB levels were measured in peripheral blood plasma, likely reflecting immune signaling rather than central neuronal activity, potentially indicating a neuroprotective anti-inflammatory response to chronic AD-related inflammation. Secondly, total CREB protein rather than its phosphorylated active form (pCREB) was measured, suggesting central pCREB expression in the brain may have been preserved or elevated, due to increased vagal tone driven by enhanced HRV. Lastly, the timing of the intervention and measurements may have captured transient immune shifts rather than long-term neuronal plasticity or gene expression alterations. These short-term shifts may not fully reflect the longer-term neurobiological consequences of sustained HRV modulation. Taken together, these interpretations underscore the importance of conducting further research that directly assesses central CREB activity, which may yield critical insights into how HRV influences Aβ dynamics and neuroplasticity in the context of AD.
Breathing exercises, slow-paced breathing in particular that enhances vagal activity and HRV, show promise in modulating autonomic function and even influencing AD-related biomarkers. However, direct evidence for symptom improvement in AD patients is limited, and future controlled clinical trials are needed to establish their efficacy in this population.
HRV and hippocampus
The hippocampus is key in memory formation and is among the earliest brain structures affected in AD. Hippocampal atrophy is a well-documented biomarker of AD progression and is closely correlated with cognitive decline. 64
In a recent study by Yoo and colleagues, the impact of daily HRV biofeedback, specifically through controlled heart rate oscillations, on hippocampal volume was examined across different age groups. 75 The researchers focused on hippocampal regions that receive projections from the LC. Participants engaged in a 5-week slow-paced breathing intervention, designed to enhance heart rate oscillations and thereby increase HRV. Results showed a significant increase in hippocampal structural volume in older adults, whereas no comparable changes were observed in younger adults. 75 The differential outcomes between age groups may be attributed to age-related changes in brain structure and function, such as reduced neuroplasticity and diminished LC activity in older individuals. These factors may render older adults more responsive to HRV-enhancing interventions.
Collectively, these findings suggest that HRV-targeted approaches, such as slow breathing techniques, may hold therapeutic potential for preserving or restoring hippocampal volume in aging populations, particularly those at risk for AD.
HRV and hypoxia
Hypoxia has a well-documented impact on autonomic functions as reflected by changes in HRV. Acute and chronic hypoxic exposure typically leads to a reduction in HRV, driven primarily by decreased parasympathetic (vagal) activity and heightened sympathetic activation. 76 These changes manifest as reductions in HRV indices, such as root mean square of successive differences (RMSSD) and high-frequency (HF) power, alongside an increase in the low-frequency/high-frequency (LF/HF) ratio, 32 reflecting autonomic imbalance. These effects have been consistently observed in both healthy individuals under experimental hypoxia and in clinical populations with compromised respiratory function.
One clinically relevant source of chronic intermittent hypoxia is obstructive sleep apnea, a condition that has been strongly associated with autonomic dysregulation and impaired HRV. Obstructive sleep apnea (OSA) is a sleep-related breathing disorder characterized by intermittent upper airway obstruction due to the temporal relaxation of the muscle walls of the tongue and soft palate. This obstruction leads to reduced airflow, resulting in episodic hypoxia and fragmented sleep architecture, thereby impairing overall sleep quality. 77 Emerging evidence suggests that OSA is a potential risk factor for AD. This association may be explained by converging mechanisms involving neurodegeneration, cognitive impairment, and compromised cerebrovascular and metabolic function, all of which are relevant to AD pathogenesis. Although the precise molecular pathways remain under investigation, OSA has been linked to abnormal protein changes including elevated Aβ and tau protein accumulation, both hallmark pathologies of AD. In a study by Bu et al., individuals with OSA syndrome were found to have significantly elevated serum levels of Aβ40, Aβ42, total Aβ, and phosphorylated tau (Ptau-181) compared to healthy snorers. 78 These changes were more pronounced in those with greater disease severity and were positively associated with the apnea-hypopnea index and oxygen desaturation index and negatively correlated with oxygen saturation. Notably, serum amyloid levels are also correlated with P-tau levels, suggesting a shared pathological pathway. These findings support the hypothesis that intermittent hypoxia and sleep disruption in OSA may contribute to AD-related protein changes, potentially accelerating disease onset. 78 Similarly, Yun et al. employed PET imaging to investigate brain amyloid deposition in OSA patients. Results showed significantly increased cortical amyloid accumulation, particularly in the right posterior cingulate gyrus and right temporal cortex, compared to matched controls. These differences remained after adjusting for major confounders, including APOE genotype and cardiometabolic factors. Interestingly, no differences in cortical thickness were observed, suggesting amyloid accumulation may precede structural atrophy. 79 Beyond the clinical findings linking OSA to increased Aβ levels, several experimental studies help explain the underlying mechanisms. In a study by Li et al., chronic hypoxia was shown to increase Aβ production by altering how amyloid-β protein precursor is processed. Specifically, hypoxia enhanced both β- and γ-secretase activity, leading to increased generation of Aβ40 and Aβ42, which are central to AD pathology. 80 Similarly, Zhang et al. found that hypoxia triggers the activation of hypoxia-inducible factor 1-alpha (HIF-1α). This transcription factor increases the expression of BACE1, the enzyme responsible for initiating the amyloidogenic pathway. As a result, more Aβ is produced under low-oxygen conditions. 81 These findings suggest that the repeated oxygen deprivation episodes seen in OSA may trigger molecular changes that promote Aβ formation. Taken together with human studies showing elevated Aβ in OSA patients, such as the studies by Bu et al. and Yun et al.,78,79 the evidence points to intermittent hypoxia as a key factor linking sleep apnea to AD pathology.
The complement system consists of a group of plasma proteins that, when activated, initiate an inflammatory response important for modulating infection. This activation is typically regulated by proteolytic cleavage, resulting in a cascade of molecular events that amplify immune signaling. 82 A recent study by Xue and colleagues investigated the potential relationship between complement activation, autonomic dysfunction, and cognitive performance in adults with OSA who had no formal diagnosis of dementia. The study found that reduced HRV, reflecting ANS dysfunction, was associated with poorer cognitive function. Interestingly, this relationship was linked to increased markers of complement activation, suggesting that excessive complement system activity may serve as a mediating pathway between autonomic dysregulation and cognitive decline in OSA patients. 83 Although the precise mechanism remains to be clarified, it is possible that complement cascade activation contributes to neuroinflammation and neuronal damage, which may further impair autonomic control, as shown in reduced HRV. This bidirectional interaction could accelerate cognitive deterioration in OSA patients, even before overt dementia manifests. These findings highlight a potentially important therapeutic window, where targeting the complement system, either through pharmacological or non-pharmacological strategies such as lifestyle interventions or vagal modulation, may improve cognitive outcomes in OSA and possibly delay or reduce the risk of AD progression, particularly in its early stages.
HRV and neuroinflammation
Autonomic dysfunction, and particularly reduced HRV, is not specific to AD. Individuals with cognitive decline, including AD, vascular dementia, and dementia with Lewy bodies, generally exhibit reduced HRV, reflecting dysregulation of the autonomic nervous system, 32 suggesting that neurodegenerative processes extend to the CAN. While direct investigations linking HRV to neuroinflammation in AD are limited, several studies have examined HRV's relationship with peripheral inflammatory markers known to play a role in AD pathophysiology, particularly C-reactive protein (CRP) and IL-6. Both markers are elevated in AD and are widely recognized as indicators of chronic inflammation with neurodegenerative consequences. In a study by Cooper et al., a significant inverse relationship was observed between HRV and multiple inflammatory markers, such as CRP, IL-6, fibrinogen, and intercellular adhesion molecule–1 (ICAM). After adjusting for a range of confounding variables (such as age, sex, medication use, race, body mass index, and comorbid conditions), CRP and IL-6 remained significantly associated with lower HRV, suggesting an independent link between ANS activity and inflammatory regulation. 84 These findings support the concept of the cholinergic anti-inflammatory pathway, wherein vagal activity, reflected in HRV, modulates immune responses and suppresses pro-inflammatory cytokine release. Such mechanisms may be particularly relevant in the context of AD, where chronic inflammation contributes to disease progression. Similar inverse associations between HRV and inflammatory markers have been replicated in other studies, further strengthening this connection. For example, lower HRV correlated with elevated levels of CRP and IL-6, even after extensive covariate adjustments.85,86 As a chief efferent pathway of the PNS, the vagus nerve plays a pivotal role in mediating anti-inflammatory effects via the cholinergic anti-inflammatory pathway. 87 This neuroimmunological pathway suppresses the production of pro-inflammatory cytokines by stimulating the release of acetylcholine, which acts on α7 nicotinic receptors expressed on immune cells.87,88 Consequently, reduced HRV, indicating diminished vagal tone, may compromise the anti-inflammatory pathway mechanism and foster a pro-inflammatory state. In the context of AD, this is especially relevant, as neuroinflammation is key contributor to disease pathogenesis. Chronic activation of microglia and astrocytes leads to sustained production of pro-inflammatory cytokines and reactive oxygen species, resulting in neuronal dysfunction, disrupted synaptic activity, and acceleration of Aβ and neurofibrillary tangle accumulation. 8 Additionally, perivascular macrophages in AD contribute to neuroinflammation and sympathetic overactivity, potentially linking neural immune responses to cardiac abnormalities. 10
Taken together, these findings imply that lower HRV in AD patients may signify impaired vagal regulation of inflammation, thereby sustaining a neuroinflammatory environment that exacerbates neurodegeneration. Insufficient anti-inflammatory response may reduce the brain's capacity to resolve inflammation, perpetuating a vicious cycle of neuronal injury and cognitive decline. These shared pathological processes (neuroinflammation, oxidative stress, vascular remodeling, and protein aggregation) underscore the systemic nature of aging and suggest overlapping mechanisms between heart and brain degeneration.
Future research is warranted to elucidate the direct links between HRV and neuroinflammation in AD. Longitudinal studies incorporating HRV monitoring, cognitive assessments, and inflammatory biomarkers from both plasma and CSF could provide key insights into this association. Furthermore, interventional approaches, such as vagus nerve stimulation, slow breathing, and mindfulness-based practices may offer promising strategies for modulating neuroinflammation and slowing cognitive deterioration in AD. In parallel, exploring the impact of modifiable lifestyle factors such as physical activity and diet on both HRV and inflammation could inform preventive strategies aimed at reducing AD risk.
Taken together, the evidence presented in this report underscores the significant role of HRV as a non-invasive indicator of autonomic nervous system function, particularly in the context of AD. HRV reflects the dynamic balance between sympathetic and parasympathetic activity and has shown associations with key processes implicated in AD pathophysiology, including inflammation, Aβ accumulation, hippocampal atrophy, and cognitive decline. Additionally, physiological markers such as sweating and pupil dilation, both governed by autonomic pathways, further reinforce the link between systemic autonomic dysfunction and neurodegenerative changes. The relevance of HRV is further highlighted in conditions like OSA, where autonomic imbalance may act as a mediator between intermittent hypoxia and AD-related protein changes. These findings suggest that HRV holds promise not only as a potential biomarker for early diagnosis and risk stratification but also as a therapeutic target. Future studies exploring its integration with other diagnostic tools, such as EEG, may provide deeper insights into the early detection and management of AD.
EEG-HRV complementarity and mechanistic overlap
Recent research shows that EEG and HRV capture two interconnected sides of the same physiological disturbance in AD. EEG reflects what is happening in the brain's electrical activity, revealing slowing of normal rhythms, particularly reduced alpha waves and increased theta activity, which indicates declining coordination among neurons. HRV, on the other hand, measures how flexibly the heart responds to changing demands. A reduction in HRV points to weakened vagal regulation and reduced adaptability of the ANS.
Although these signals originate from different parts of the body, they are tightly linked through a shared control network (i.e., the CAN), which includes regions such as the insula, anterior cingulate cortex, and amygdala. These structures act as a bridge between emotional, cognitive, and autonomic processes, allowing the brain and heart to work in synchrony. When this network begins to deteriorate in AD, both cortical rhythms and autonomic responses lose their normal flexibility.
The CAN is a key integrative system that coordinates autonomic, emotional, and cognitive processes through a distributed set of brain regions. It comprises the anterior cingulate cortex, insula, amygdala, hypothalamus, periaqueductal gray, nucleus tractus solitarius, and other brainstem nuclei.25,89 Through its influence on both sympathetic and parasympathetic output, the CAN maintains cardiovascular homeostasis while also modulating cognitive and affective responses.
This network plays a critical role in mediating brain-heart interactions, integrating top-down cortical control with bottom-up interoceptive signals. Importantly, CAN function can be non-invasively probed through measures of cortical activity, such as EEG, and autonomic output, such as HRV derived from ECG, making it highly relevant to research on neurodegenerative diseases.
While EEG and HRV each provide valuable information on different aspects of AD, interpreting them together offers a more complete picture of how the disorder affects both the brain and the body. EEG reveals the brain's electrical signature (i.e., showing how neural circuits lose their ability to maintain synchronized communication) whereas HRV captures the body's physiological response to stress and internal regulation. When examined side by side, these measures illustrate how changes in the brain's signaling are mirrored by changes in autonomic control, both governed by the same underlying network, which is the CAN.
This shared network links the cortex, limbic system, and brainstem to coordinate cognitive, emotional, and physiological states. As AD progresses, the integrity of this system declines. The result is a gradual breakdown in both the brain's ability to coordinate activity across regions (i.e., neural synchrony) and the heart's capacity to maintain flexible autonomic regulation (i.e., vagal tone).
Studying EEG and HRV together therefore allows researchers to move beyond single-system observations toward a systems-level understanding of disease. EEG-HRV coupling provides a window into how the brain and heart lose coordination over time, reflecting early disruption of neurovisceral integration. Recognizing this breakdown as a unified process rather than as separate symptoms shifts the focus from isolated brain pathology to a broader, network-based view of AD, one that connects cortical, autonomic, and behavioral changes within the same physiological framework.
Central-autonomic interplay: the brain-heart axis in AD
In the context of AD, and other forms of dementia, dysfunction of the CAN has been increasingly recognized as both a consequence and a possible contributor to disease progression. Structural and functional neuroimaging studies have shown that regions within the CAN, and in particular the insula, anterior cingulate cortex, and amygdala, are affected early in AD, leading to impaired autonomic regulation alongside cognitive decline.28,90 Parallel evidence from 24-h ECG-based studies demonstrates reductions in HRV among individuals with AD (n = 33) and MCI (n = 39) compared to cognitively healthy controls (n = 29), indicating autonomic imbalance. 91 Crucially, emerging studies highlight that it is not just individual EEG or ECG markers that are affected in neurodegeneration, but also their coupling. Despite this, attention has only recently shifted toward a more holistic view of AD that emphasizes the interplay between brain function, systemic physiology, and early biomarkers of cognitive decline.
Boudaya et al. use a multimodal approach combining EEG and HRV signals for early detection of MCI. 92 Despite such fusion not capturing heart-brain coupling per se, it simultaneously utilizes features that reflect both neural and cardiovascular activity. EEG and HRV data were recorded during a short and comprehensive battery of standardized tests used to assess cognitive impairments in AD (CERAD tests), with features from time, frequency, and non-linear domains extracted, preprocessed, and integrated using machine learning (ML) models. A hybrid ML model combining support vector machine, gradient boosting, and random forest algorithms with a voting mechanism was trained to classify MCI (n = 8) and healthy controls (n = 7). The classifier achieved 93.86% overall accuracy and 97.43% accuracy during the first CERAD task, surpassing previous EEG- and HRV-only approaches, which reported accuracies ranging from 73.4% to 91.6%. While the results highlight the potential of EEG-HRV fusion for early and non-invasive MCI detection, limitations include a small sample size (15 participants post-exclusion) and reliance on the hybrid model's voting system, which may lead to variability across datasets.
In a recent study, Bjerkan et al. explored disruptions in neurovascular phase coherence, defined as the temporal synchronization between neuronal activity and cerebral blood flow oscillations, in AD patients with MCI (n = 4), mild AD and moderate AD (n = 19), as measured by the MSSE scores. 93 Using simultaneous EEG and functional near-infrared spectroscopy (fNIRS), the authors examined phase relationships between EEG alpha rhythms and slow oscillations in cerebral hemodynamics during resting state. AD patients exhibited significantly reduced neurovascular coherence compared to healthy controls (n = 20), particularly in the prefrontal cortex, which correlated with cognitive impairment and disease severity. The findings suggest that neurovascular decoupling may reflect broader dysfunction within the central autonomic and neurovascular regulatory systems. Importantly, although the study did not directly analyze EEG-ECG coupling, it underscores the broader concept of disrupted systemic synchrony in AD, supporting the relevance of multimodal approaches that integrate electrophysiological and physiological signals to understand brain-body interaction in neurodegeneration.
In their study, Candia-Rivera et al. introduced a novel framework to quantify the coupling between brain connectivity and heartbeat dynamics to characterize the disrupted network physiology in Parkinson's disease (PD). 94 The researchers collected simultaneous EEG and ECG recordings from 15 PD patients, both on and off dopaminergic medication, and 16 healthy controls during resting-state conditions. They computed time-varying EEG connectivity in alpha and gamma frequency bands and derived cardiac sympathetic and parasympathetic indices from HRV analyses. To quantify the synchronization between brain network connectivity and cardiac dynamics, Pearson's correlations were estimated between time-varying EEG connectivity (in alpha or gamma bands), and the time-resolved HRV measures (LF, HF, and RMSSD). By assessing these correlations, it was found that PD patients off medication exhibited significantly reduced brain-heart correlations compared to healthy individuals. Importantly, dopaminergic treatment partially restored this coupling, with improvements correlating with reductions in motor symptoms as measured by the Unified Parkinson's Disease Rating Scale (UPDRS) Part III. These findings suggest that the integration of EEG and ECG analyses can provide valuable insights into the autonomic and neural dysfunctions in PD, highlighting the potential of brain-heart coupling metrics as biomarkers for disease progression and treatment efficacy. While this study provides a valuable framework for assessing brain-heart interactions in PD, the authors acknowledge that their method captures correlations between EEG connectivity and HRV measures, rather than true synchrony or causal relationships. The need for future studies that incorporate more advanced techniques, such as phase synchronization or causality analysis, is highlighted, to better characterize true neurocardiac coupling dynamics, as opposed to correlation.
Grässler et al. propose a multimodal measurement protocol along these lines that integrates fNIRS, EEG, and HRV derived from ECG to enhance early detection of MCI, a prodromal stage of dementia and AD. 95 This approach captures distinct but complementary aspects of neurophysiology, including cortical hemodynamics, brain electrical activity, and autonomic nervous system function, offering a more holistic view of cognitive decline. The protocol involves comparing neurophysiological responses between MCI patients and healthy controls during cognitive tasks such as the Stroop, N-back, and verbal fluency tests, and assessing correlations between signal features and cognitive performance. A central aim is to determine whether combining these modalities yields better classification accuracy than unimodal or bimodal methods. As a noninvasive, portable, and cost-effective alternative to traditional neuroimaging techniques like magnetic resonance imaging (MRI) or PET, this approach holds promise for scalable, biomarker-driven early detection strategies that could enable earlier and more effective interventions in at-risk populations.
Lin et al. (2014) investigated the relationship between cerebral and cardiac signal complexities in older adults with varying cognitive abilities. 96 The study involved 87 geriatric outpatients who underwent 19-channel EEG and 24-h ECG recordings. Multiscale entropy (MSE) analysis was applied to assess the complexity of EEG signals during different states (resting-awake, fast photic stimulation, and slow photic stimulation) and to RR interval (RRI) time series derived from ECG during awake and sleep states. The findings revealed significant inverse correlations between EEG and HRV complexities, suggesting a compensatory mechanism between brain and heart activities. Notably, reduced EEG complexity at the F8 electrode site correlated with lower cognitive test scores, supporting the cholinergic hypothesis that links cognitive decline to decreased cortical complexity and altered autonomic function. These results underscore the potential of EEG-ECG complexity analyses as biomarkers for cognitive impairment and highlight the importance of brain-heart interactions in neurodegenerative diseases.
These findings support the view that decoupling between central and autonomic systems may reflect CAN breakdown, offering a systems-level biomarker of neurodegeneration. Importantly, EEG-ECG coupling may capture aspects of dysfunction that are not evident when assessing either modality in isolation. Therefore, incorporating multimodal analyses of EEG and ECG signals provides a more comprehensive view of the physiological alterations in AD and related disorders, highlighting the CAN as a central mechanism linking neural and cardiovascular dysfunction.
EEG-HRV coupling therefore represents two outward expressions of the same underlying degenerative process: loss of cortical synchronization and loss of autonomic adaptability. Supporting this view, multimodal studies show that lower EEG complexity often parallels decreased HRV entropy, which are both indicators of reduced physiological flexibility. This shared decline may occur early in the disease, even before major cognitive symptoms appear, making EEG-HRV coupling a promising marker for detecting subtle neurophysiological changes that signal the onset of AD pathology.
Summary and critical synthesis of EEG-HRV evidence
Over the past decade, researchers have begun to look beyond the brain alone when studying AD. Increasingly, attention has turned to the brain-heart connection, which is how the body's autonomic rhythms mirror and even predict changes in cognition. HRV is emerging as a key indicator. In healthy adults, a higher HRV reflects strong parasympathetic or “rest-and-digest” activity and efficient physiological regulation. 30 In contrast, people with MCI or early AD often show a blunted HRV pattern, meaning the heart becomes less adaptable and more dominated by stress-related (sympathetic) tone.91,97 Recent multimodal designs, notably Boudaya et al., demonstrate that combining EEG and HRV can identify early cognitive changes with high accuracy, supporting their potential as accessible biomarkers for early detection. 92 Table 1 brings together some studies examining how the brain and heart change during healthy aging, MCI, and early AD. It illustrates the range of recording approaches, from brief resting sessions to simultaneous EEG-ECG during cognitive tasks, and a consistent pattern: reduced HRV, slowing of EEG rhythms, and weakened brain-heart coordination as cognition deteriorates.
Summary of key EEG-HRV and related multimodal studies in AD, MCI, and cognitive aging.
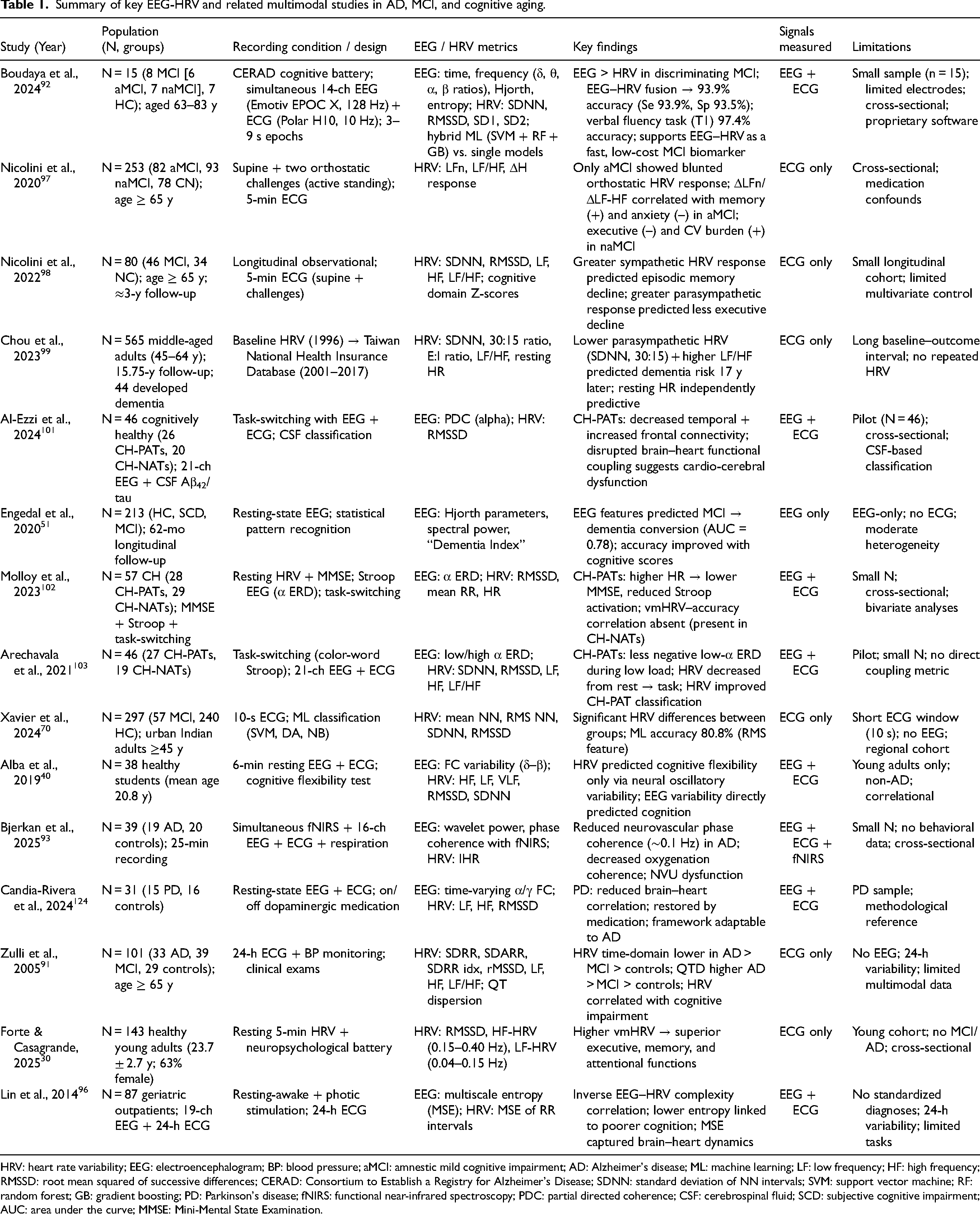
HRV: heart rate variability; EEG: electroencephalogram; BP: blood pressure; aMCI: amnestic mild cognitive impairment; AD: Alzheimer's disease; ML: machine learning; LF: low frequency; HF: high frequency; RMSSD: root mean squared of successive differences; CERAD: Consortium to Establish a Registry for Alzheimer's Disease; SDNN: standard deviation of NN intervals; SVM: support vector machine; RF: random forest; GB: gradient boosting; PD: Parkinson's disease; fNIRS: functional near-infrared spectroscopy; PDC: partial directed coherence; CSF: cerebrospinal fluid; SCD: subjective cognitive impairment; AUC: area under the curve; MMSE: Mini-Mental State Examination.
Studies that followed older adults over time, such as those by Nicolini and colleagues and Chou et al., found that reduced parasympathetic responsiveness could predict future memory and attention decline years before dementia was diagnosed.98,99 These findings suggest that subtle autonomic imbalance may be an early warning sign of neurodegeneration (i.e., one that appears before structural brain damage is visible on imaging).
EEG research complements this picture from the cortical side. In both MCI and early AD, EEG recordings show slowing of rhythmic brain activity, such as less alpha power, reduced connectivity between regions, and lower signal complexity.52,100 These changes reflect a gradual loss of coordination within the brain's communication networks. For example, Engedal et al. demonstrated that specific EEG signatures could forecast which individuals with MCI would later progress to dementia, supporting the idea that electrical rhythms provide a functional snapshot of brain health. 52
When EEG and HRV are measured together, a deeper insight emerges: the brain and heart start to lose their natural synchrony as cognitive impairment develops.101–104 Recent multimodal studies have shown that this loss of coordination can be detected during even short cognitive challenges. In a 2024 investigation, Boudaya et al. recorded EEG and HRV simultaneously while participants completed a brief memory task from the CERAD test battery. 92 By combining information from both signals (i.e., brain waves and heart-rate patterns), their machine-learning model could distinguish MCI (n = 8) from healthy aging (n = 7) with almost 94% accuracy, and even higher (97%) for the verbal-fluency task that engages executive function. 92 This result was achieved with recordings lasting only a few seconds and highlights the potential of EEG-HRV coupling as a rapid, low-cost biomarker of early cognitive decline.
Other multimodal studies101–104 echo this finding: weaker alpha desynchronization, lower vagally mediated HRV, and altered frontotemporal communication appear together as cognition declines. Collectively, these patterns suggest that AD affects not only cortical networks but also the broader brain-body regulatory axis. The heart and brain seem to “fall out of rhythm” long before dementia becomes clinically evident.
Despite growing recognition of brain-heart crosstalk in AD, several limitations hinder deeper mechanistic understanding. Much of the current evidence is based on observational studies or animal models, making it difficult to establish causality or generalize findings to diverse human populations. Confounding variables such as lifestyle, comorbidities, and medication use complicate interpretation. Moreover, the multifactorial nature of both cardiovascular and neurodegenerative diseases adds further complexity. Despite encouraging progress, most investigations still involve small samples, differing analytical pipelines, and limited methodological standardization.93,94 Yet the trend is clear: as AD begins to unfold, cortical, autonomic, and vascular systems drift out of sync, producing measurable changes in electrical and cardiac rhythms. Capturing these interactions through simultaneous EEG-HRV recording provides a promising, non-invasive window into early disease processes and may pave the way for preventive monitoring strategies.
Translational implications
The convergence of EEG and HRV research points toward a more holistic, physiology-based view of AD. Traditional diagnostics depend on neuroimaging or CSF biomarkers that, while informative, are expensive and invasive. EEG and HRV, by contrast, offer real-time insight into brain-body communication that can be captured with portable or wearable sensors in just a few minutes.
If validated in larger longitudinal studies, this neurocardiac framework could underpin a new generation of digital biomarkers, which are objective, affordable tools for identifying individuals at risk of cognitive decline years before symptoms become disabling. Such advances would shift dementia care from late recognition to proactive, continuous monitoring, enabling earlier lifestyle or therapeutic interventions when the brain remains most responsive to change.
Practical and clinical implications
The application of EEG, ECG, and brain-heart coupling measures in AD and related dementias is not only scientifically compelling but also practically advantageous. These modalities offer tangible benefits in terms of cost, accessibility, usability in aging populations, and integration into emerging digital health ecosystems. As the global burden of dementia continues to grow, with disproportionate impact in low-resource and aging communities, such tools may help bridge gaps in diagnosis, monitoring, and care.
Cost-effectiveness and accessibility
Traditional diagnostic tools for AD, such as MRI, PET, and CSF biomarker analysis, are often resource-intensive, costly, and logistically challenging to deploy at scale. For instance, amyloid PET scans can cost over $5000 per patient and are typically confined to specialized clinical research centers. 105 In contrast, EEG and ECG are relatively low-cost, portable, and widely available technologies that can be implemented in a variety of healthcare settings, including primary care and community clinics. In the USA, the average cost of an EEG test can range from $200 to $2500 (for uninsured patients), depending on factors such as facility type, duration, and whether video or ambulatory monitoring is included (https://newmedicare.com/eeg-test-cost/). In routine clinical practice within Europe, basic EEG examinations in private settings are commonly in the range of €230-€350 for standard recordings, with more comprehensive or specialized services (e.g., prolonged or video-EEG) potentially costing upwards of €1,000, depending on country and facility (https://us-uk.bookimed.com/clinics/procedure = eeg-electroencephalography/). Basic ECG tests are comparatively inexpensive in European cardiology clinics, often priced from about €60 for a standard resting ECG assessment (https://medicus-center.com/en/special-offers/cardiac-check-up), and in the range of $50-$200 in the USA (uninsured) (https://sophiarahmanmd.com/ekg-test-cost-without-insurance/).
Recent studies have shown that EEG can provide meaningful biomarkers of AD progression without the need for advanced imaging infrastructure. Similarly, ECG-derived HRV has been linked to cognitive function and neurodegenerative risk, offering an additional, non-invasive avenue for early detection. 91 The cost-effectiveness of these approaches becomes even more pronounced when used in combination, as multimodal systems can yield higher diagnostic accuracy while maintaining low per-test costs. Their simplicity and scalability make them particularly valuable in health systems struggling with capacity constraints or limited specialist access.
Use in aging and remote populations
Older adults and individuals in rural or underserved areas face unique barriers to timely dementia diagnosis and care, including transportation limitations, caregiver burden, and a shortage of neurologists or geriatricians. Wearable or mobile EEG and ECG technologies offer a practical solution by enabling in-home or community-based monitoring, thereby reducing dependence on centralized healthcare facilities.106,107
Several pilot programs have demonstrated the feasibility of deploying wearable neurophysiological sensors in aging populations to monitor cognitive changes over time. For example, Mathewson et al. reported that mobile EEG systems could reliably capture cortical signals relevant to memory and attention, even outside laboratory conditions. 108 When combined with autonomic markers such as HRV, these tools provide a more complete physiological profile of brain–body health, potentially enabling earlier identification of individuals at risk for MCI or dementia. Furthermore, continuous or periodic monitoring can facilitate longitudinal tracking, which is essential for evaluating disease progression, medication response, and non-pharmacological interventions.
Remote applicability also supports the growing shift toward aging-in-place models, in which elderly individuals receive support and diagnostics within their homes or communities. This is particularly crucial as the global population of adults over age 65 is expected to double by 2050, with the sharpest increases in low- and middle-income countries. 109
Potential for digital health integration
The integration of EEG, ECG, and brain-heart coupling metrics into digital health ecosystems represents one of the most promising advances in dementia care. These physiological signals can now be collected using wearable sensors, transmitted wirelessly to cloud-based platforms, and analyzed using machine learning algorithms to identify patterns indicative of cognitive decline or autonomic dysfunction. 110
Digital platforms enhance the interpretability and clinical relevance of EEG-ECG data by enabling real-time visualization, automated anomaly detection, and longitudinal trend analysis. For instance, AI-based models have been trained to distinguish dementia from healthy aging using EEG-based features. 111 Importantly, explainable AI frameworks allow clinicians to understand which features contribute most to predictions, enhancing clinical trust and adoption. 112
Digital health integration also opens the door to closed-loop systems, where neurophysiological inputs guide real-time therapeutic outputs. Examples include neurofeedback systems that adapt cognitive training intensity based on EEG activity, or HRV-guided breathing exercises aimed at improving vagal tone and emotional regulation. These approaches are especially useful in early-stage dementia or at-risk populations, where interventions may still slow or alter disease trajectories. 113
Moreover, integration with electronic health records, mobile apps, and telemedicine platforms facilitates communication among patients, caregivers, and multidisciplinary care teams. This connectivity enhances continuity of care, supports medication adherence, and empowers individuals with dementia to participate in their own health management.
The gut-brain-heart axis: an emerging triad in AD
The gut-brain axis is a bidirectional communication network linking the gastrointestinal microbiota and the CNS through neural, hormonal, metabolic, and immune pathways. This axis plays a critical role in maintaining homeostasis and regulating cognitive and emotional processes. Emerging evidence highlights the role of gut microorganisms influencing the CNS via the vagus nerve, immune signaling, and tryptophan metabolism. The vagus nerve represents a key neural pathway, directly connecting the gastrointestinal tract to the brain. It transmits signals from gut-derived metabolites, such as short-chain fatty acids, directly to the brain, thus impacting mood, cognition, and neuroinflammation. 114
Immune interactions are mediated by the gut-associated lymphoid tissue, where microbial dysbiosis can alter cytokine levels, promoting systemic inflammation and neuroinflammation. 115 Additionally, the gut microbiota can influence kynurenine pathway metabolism, altering the synthesis of neuroactive metabolites derived from tryptophan, a process implicated in neurodegenerative diseases, including AD. 116 Mounting evidence suggests that the gut microbiota can influence autonomic nervous system regulation, particularly through its effects on HRV, an index of vagal tone, and autonomic modulation. Vagus nerve activation, mediated by gut microbial activity, has been shown to alter HRV parameters, which are associated with stress resilience, emotional regulation, and cognitive outcomes. 117 For instance, microbial dysbiosis has been linked to a reduction in HRV by increasing systemic inflammation and decreasing vagal tone. 118 HRV profiles, in turn, have been connected to cognitive function, with lower measures of HRV correlating with poorer executive and memory performance in older adults, as well as higher dementia risk. 119
Altered gut microbiota composition has been consistently observed in AD, characterized by reduced beneficial taxa (e.g., Bacteroides and Lactobacillus) and an overrepresentation of pro-inflammatory species (e.g., Escherichia-Shigella). 120 These microbial imbalances are thought to contribute to AD pathogenesis via chronic low-grade inflammation, metabolic disruption, and neurotoxicity. Specifically, elevated systemic inflammation in AD can exacerbate Aβ plaque deposition and hyperphosphorylation of tau, two key neuropathological hallmarks of the disease. 121
Building on these mechanisms, vagus nerve stimulation (VNS) is being explored as a direct neuromodulatory intervention in early AD. VNS has been shown to increase the release of norepinephrine and acetylcholine, which are typically deficient in AD. It also enhances synaptic plasticity and reduces neuroinflammatory responses. Both invasive and non-invasive VNS methods have demonstrated safety and early evidence of cognitive benefit in individuals with MCI and early AD. 122 These findings suggest that VNS may not only serve as a therapeutic tool targeting central mechanisms of neurodegeneration but may also represent a physiological bridge between gut microbiota activity, autonomic regulation, and cognitive health.
To fully elucidate the gut-brain-heart axis in AD, research integrating gut microbiome profiling with EEG and HRV-based biomarkers is necessary. Such studies could provide a comprehensive understanding of how microbial dysbiosis influences neural and autonomic dysfunction in AD. For instance, profiling microbial metabolites and inflammatory markers alongside EEG and HRV measurements could identify early diagnostic biomarkers and inform preventative or therapeutic interventions, such as tailored probiotic or dietary regimens. This integrated approach lays the groundwork for developing personalized interventions targeting gut microbial health to promote cognitive resilience and mitigate the effects of neurodegenerative diseases.
Toward multimodal biomarker integration
As the pathophysiology of AD becomes increasingly understood as a multisystem, multifactorial process, the limitations of single-modality biomarkers have become more apparent. Traditionally, AD diagnostics have relied heavily on structural and molecular markers, such as MRI-based atrophy measures, PET imaging of amyloid and tau, and CSF levels of Aβ42 and phosphorylated tau. 13 While these have proven valuable for identifying disease pathology, they often fail to capture functional and physiological changes that evolve dynamically over time and across systems.
In contrast, multimodal biomarker integration, which combines data from neuroimaging, electrophysiology, autonomic measures, genetics and behavioral assessments, offers a more holistic view of brain function and disease progression (Figure 2). 123 For instance, EEG can provide real-time insights into cortical network activity and oscillatory dynamics, while ECG and HRV reflect autonomic regulation under CAN control. When integrated, these signals can help reveal breakdowns in brain-heart coupling that may precede overt cognitive symptoms.94,124 MSE analysis is one the techniques that can be used for such integration, by capturing the temporal complexity of both EEG and ECG signals across multiple time scales. Its utility in dementia research was demonstrated by Lin et al., who applied MSE to resting-state EEG and 24-h ECG recordings in older adults with varying cognitive status, revealing significant inverse correlations between EEG and heart rate complexity, particularly in frontopolar, central, and temporal regions, suggesting disrupted brain-heart dynamics associated with cognitive impairment. 96
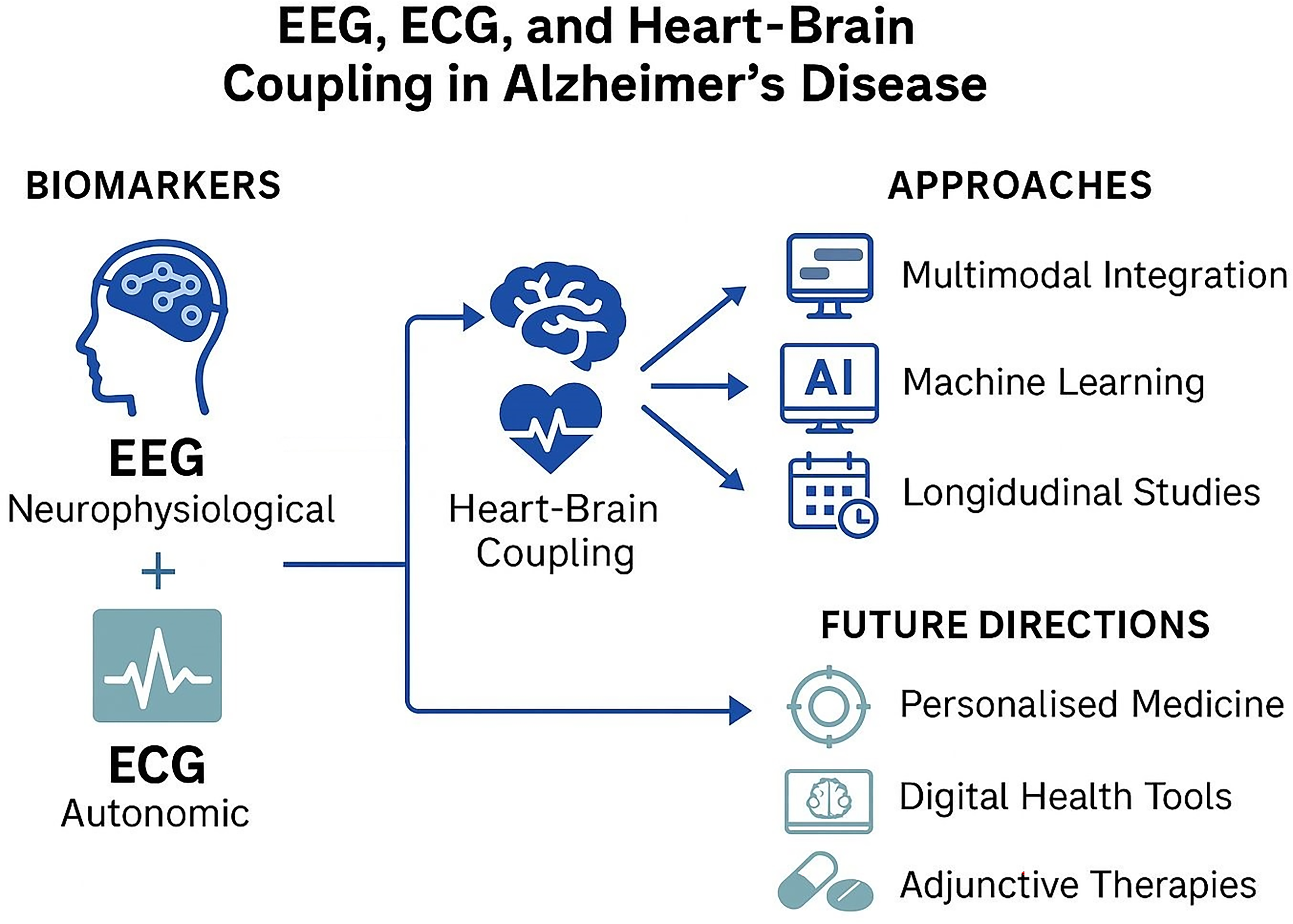
Multimodal neurophysiological assessment in Alzheimer's disease: conceptual framework and clinical relevance of integrating electroencephalography (EEG), electrocardiography (ECG), and brain-heart coupling metrics in the assessment of ad. EEG reflects cortical network dynamics, while ECG and heart rate variability (HRV) capture autonomic nervous system function. Disruptions in their coupling may signal early dysfunction in the central autonomic network (CAN), offering a sensitive and accessible biomarker for cognitive decline. Applications span early detection, real-world monitoring, and digital health integration in aging populations. (Initial form of Figure 2 created with ChatGPT-4o, OpenAI).
Moreover, wearable devices and remote sensing technologies now allow for continuous collection of EEG, ECG, and behavioral data in ecologically valid settings, enabling longitudinal and contextualized biomarker tracking. Combining such data with neuroimaging, genomics (e.g., APOE status), and digital cognitive assessments creates opportunities for machine learning-based risk stratification and early detection models. 110
Recent initiatives such as the Alzheimer's Disease Neuroimaging Initiative (ADNI) 125 and the European Prevention of Alzheimer's Dementia (EPAD) project 126 are beginning to incorporate multimodal platforms to improve both diagnostic specificity and trial stratification. Integrating central and peripheral physiological biomarkers will likely play a crucial role in the future of precision medicine for AD, offering a multidimensional approach to diagnosis, prognosis, and treatment response.
Niu et al. developed an explainable machine learning model that integrates EEG, eECG, and clinical features to predict neurological outcomes in comatose patients following cardiac arrest. 112 Utilizing data from the I-CARE database, the study employed various machine learning algorithms, including Logistic Regression, Support Vector Machine, Random Forest, and Gradient Boosting, to analyze the combined dataset. The integrated model outperformed single-modality models, achieving area under the curve (AUC) values ranging from 0.75 to 1.0, with peak accuracy observed within the 12–24-h post-arrest window. Notably, the study applied SHapley Additive exPlanations (SHAP) to interpret feature contributions, identifying “Shockable Rhythm” as the most influential predictor, followed by specific EEG and ECG-derived features. This approach underscores the potential of multi-modal data integration and explainable AI in enhancing early prognostication and supporting clinical decision-making in critical care settings.
Probiotics and neurophysiological modulation
Emerging research on the gut-brain axis has revealed that gut microbiota may influence neurodevelopment, mood, cognition, and even the progression of neurodegenerative diseases. In the context of AD, alterations in gut microbiota composition, known as dysbiosis, have been linked to systemic inflammation, BBB permeability, and aberrant neuroimmune responses,120,127 all of which may contribute to cognitive decline by disrupting CNS function.
Probiotics, which are live microorganisms conferring health benefits to the host, show promise as modulators of gut-brain-heart interactions. Certain strains of probiotics, such as Bifidobacterium and Lactobacillus, have been shown to reduce neuroinflammation, improve memory performance, and normalize brain activity in animal models of AD. 128 Dendritic spine maturation was suggested as the key mechanism by which probiotics, through the brain-gut axis, may attenuate AD pathogenesis. Supplementation with probiotics has also been associated with increased theta brain activity 129 and improved HRV, suggesting enhanced vagal tone and autonomic regulation. 130
Complementing these findings, clinical and preclinical studies suggest that specific probiotic strains can influence neuroinflammatory pathways, neurotransmitter metabolism, and oxidative stress, all of which are implicated in AD pathology.131,132 Their impact on neurophysiological markers has been the subject of recent investigations. For example, in a randomized, double-blind, placebo-controlled trial involving healthy adult participants, probiotic supplementation with B. Longum led to improvements in social stress, which correlated with changes in specific EEG rhythms. 129 Similarly, reported HRV improvements following probiotic suggest enhanced parasympathetic tone and reduced autonomic imbalance - both relevant to the integrity of brain-heart coupling mechanisms. 133
Together, these findings support the hypothesis that probiotics may benefit brain function through a combination of immune, endocrine, and autonomic pathways, as well as through modulation of neurophysiological rhythms. Despite these findings, direct evidence linking probiotics to concurrent changes in EEG patterns, HRV, and gut microbiota profiles, particularly in the AD population, remains limited. Future studies are needed to assess the long-term effects of probiotic interventions and how these might synergistically improve autonomic function, neural activity, and cognitive health.
Novel tools and technologies
A major limitation of current AD diagnostics is their static nature, relying on cross-sectional “snapshots” of cognition or neurobiology taken during brief clinical visits. However, AD is a dynamic, progressive disease that unfolds over years or decades. Longitudinal monitoring of physiological signals offers a more ecologically valid approach to capturing this progression, particularly in the early stages when interventions may be most effective.
The increasing availability of portable EEG and ECG technologies supports long-term, real-world data collection. Studies have shown that repeated, passive recordings in naturalistic environments, such as during sleep, daily activities, or stress, can reveal subtle patterns of decline in brain function and autonomic regulation not detectable through single-time-point assessments.106,107 For example, declining HRV over months may reflect increasing central autonomic dysregulation, 94 while changes in EEG complexity may signal incipient cognitive impairment. 134
Importantly, this approach allows for patient-centered data collection, reducing the need for frequent clinic visits while enabling adaptive monitoring. Data collected longitudinally can also serve as a foundation for machine learning models trained to detect deviations from an individual's baseline physiology, paving the way for proactive alerts and just-in-time interventions. This represents a shift from reactive to predictive care in neurodegenerative disease management.
Moreover, longitudinal neurophysiological tracking has significant implications for clinical trials, where physiological biomarkers can serve as dynamic endpoints to evaluate treatment response or disease-modifying effects. EEG and HRV measures are sensitive to both pharmacological and behavioral interventions and may help to detect early efficacy signals.
The future of AD diagnostics and monitoring lies increasingly in the integration of innovative, non-invasive technologies that complement existing neurophysiological and imaging modalities. Among the most promising are therapeutic and diagnostic ultrasound techniques, wearable biosensors, and AI for advanced signal analysis and decision support. These tools offer the potential to address key challenges in dementia care, including early detection, continuous monitoring, and accessibility of diagnostics in non-specialist settings.
Ultrasound. Therapeutic and diagnostic applications of ultrasound are gaining traction in neurology, including in AD. Recent studies have shown that FUS can transiently open the BBB, thereby enhancing the delivery of therapeutic agents to specific brain regions affected by AD pathology.135,136 In terms of clinical evidence, a recent phase 1 pilot study demonstrated that a portable, neuronavigation-guided focused ultrasound system could safely and feasibly open the BBB in patients with mild/moderate AD, with BBB opening associated with measurable changes in AD biomarkers and a slower regional increase in amyloid load. 137 Furthermore, transcranial Doppler ultrasound can be used to assess cerebral hemodynamics in real-time, offering a low-cost method to evaluate vascular contributions to cognitive impairment.138,139 In summary, there is preclinical and early clinical evidence that ultrasound, and in particular focused ultrasound for BBB opening and transcranial ultrasound neuromodulation, can influence AD pathology and cognitive function. While not yet standard practice in AD, ultrasound portability and low cost make it an appealing adjunct to EEG, ECG, and neuroimaging for real-time brain-body assessment, particularly in bedside or ambulatory settings. Larger, controlled clinical trials are needed to confirm efficacy, optimize protocols, and further clarify mechanisms of action.
Wearables. Wearable technologies are rapidly transforming the landscape of dementia research and care. Portable EEG and ECG devices, often integrated into headbands, smartwatches, or adhesive patches, enable continuous or ambulatory monitoring of brain and heart activity.106,108 Recent developments include multimodal wearables capable of measuring additional physiological signals such as skin conductance, respiration, movement, and sleep architecture, which are relevant to both cognitive and autonomic function. 140 The ability to track brain-heart coupling in everyday environments opens new possibilities for long-term, real-world monitoring of cognitive health trajectories. Importantly, these tools can be deployed in remote or aging-in-place scenarios, supporting early detection, behavioral monitoring, and intervention responsiveness with minimal clinical burden. Recent studies have demonstrated the feasibility and acceptability of wearable devices, including smartwatches, wearable EEG systems, and HRV monitors, for tracking biomarkers of cognitive decline, autonomic dysfunction, and daily living activities in individuals with subjective cognitive decline, MCI, Lewy-body dementia and AD.141,142 Wearable EEG devices, although still facing challenges related to signal quality and standardization, have shown potential for detecting early-stage network abnormalities and cortical slowing. 143 Similarly, multimodal wearables combining EEG, accelerometry, and other physiological metrics, such as sleep features, have been explored for AD screening with promising diagnostic accuracy in research settings, especially when enhanced by AI and machine learning algorithms. 144 Beyond detection, wearables may also support remote monitoring and personalized care in dementia management, although their direct impact on cognitive or symptomatic improvement remains to be established. Overall, while wearable technologies represent a valuable component of multimodal, patient-centered approaches in AD research and care, further clinical validation is needed to confirm their utility in improving symptoms or altering disease trajectories.
Artificial Intelligence (AI). AI and ML offer powerful tools to enhance the utility of EEG, ECG, and wearable data in AD. By extracting complex patterns from high-dimensional time-series data, AI can detect subtle physiological changes that may precede overt clinical symptoms, and recent work applying AI to EEG-based detection of AD shows promising gains in early diagnosis, particularly when studies integrate advanced signal-processing techniques, multimodal features, and carefully tuned machine-learning models. 145 For example, convolutional neural networks and recurrent neural networks have been trained to differentiate between healthy aging, MCI, and AD using EEG spectral features and ECG-derived heart rate variability. 110 Beyond classification, AI can also enable predictive modeling for disease progression and treatment outcomes, as well as explainable AI (XAI) frameworks that identify which signal features are most relevant for decision-making. 112 When integrated into digital health platforms, AI systems can support clinicians by generating automated alerts, guiding personalized treatment plans, and enabling scalable cognitive screening programs. However, challenges remain, such as limited dataset diversity, variability in acquisition methods, need for large labeled cohorts, model interpretability issues, and generalizability across populations, 145 underscoring the importance of rigorous methodology and XAI approaches in future research.
Together, ultrasound, wearable biosensors, and AI represent the next generation of tools that could complement and extend traditional assessments. These technologies align with a systems-level approach to neurodegeneration, one that recognizes the interdependence of neural, vascular, and autonomic domains, and offer a practical path toward earlier diagnosis, more personalized monitoring, and broader access to care.
Roadmap for clinical adoption
Translating EEG-HRV coupling into a practical clinical tool requires a clear and coordinated roadmap, one that moves step by step from laboratory innovation to real-world healthcare use. This process must align technological progress with clinical validation to ensure that findings are both scientifically robust and clinically meaningful.
In the short term, the focus should be on standardization. Researchers need to agree on how EEG and ECG data are collected, processed, and interpreted. This means developing shared acquisition protocols, ensuring that devices from different manufacturers can communicate seamlessly, and defining minimum quality standards for recordings. Achieving this level of consistency will make results comparable across studies and institutions.
In the medium term, large, multicenter studies should validate these standardized methods in diverse patient populations. Testing EEG-HRV coupling across individuals with MCI, early AD, or other forms of neurodegeneration, as well as healthy controls will clarify how well these measures distinguish different types of dementia and disease stages from healthy aging. At this stage, EEG-HRV metrics should also be compared with established biomarkers, such as CSF and blood tests, or PET imaging to confirm their diagnostic accuracy. Establishing normative reference ranges based on factors like age, sex, and ethnicity will be essential for clinical interpretation.
In the long term, the integration of EEG-HRV analysis into wearable devices and AI-based systems will allow continuous, real-world monitoring of brain and heart function. These technologies could enable clinicians to track cognitive and autonomic changes in everyday settings outside of the hospital through safe, low-cost, and portable systems. Ultimately, this would support early detection, remote monitoring, and personalized treatment planning.
The ultimate goal is to embed EEG-HRV assessment into everyday clinical workflows, from memory clinics to primary-care settings. By combining physiological signals with AI-driven decision tools and linking them to electronic health records, clinicians could receive real-time feedback about a patient's neurophysiological status. This would make dementia screening and monitoring more proactive, accessible, and tailored to each individual.
The roadmap (Table 2) outlines this stepwise transition, from experimental research to scalable clinical practice.
Roadmap for translating EEG-HRV integration into AD diagnosis and care.
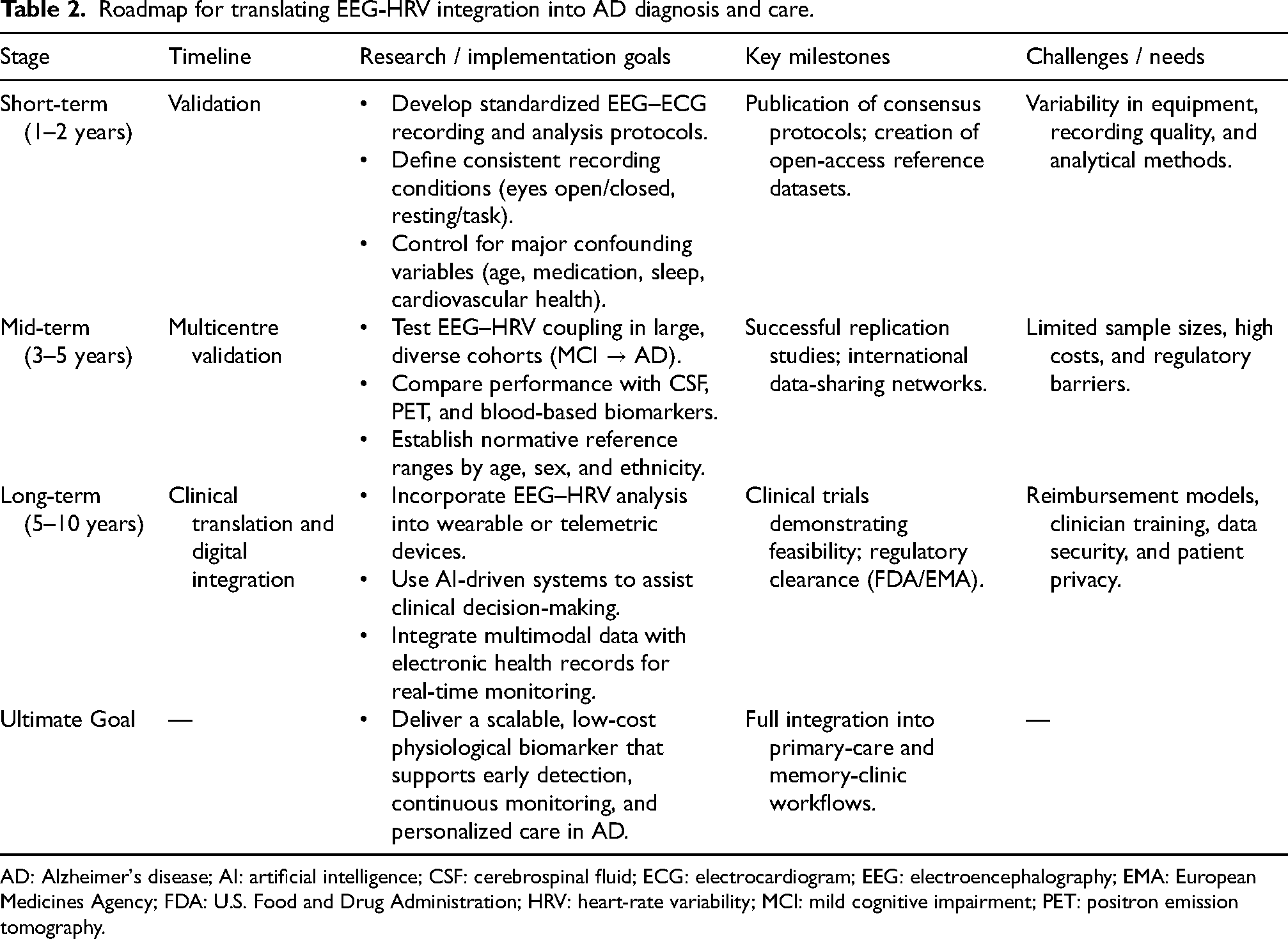
AD: Alzheimer's disease; AI: artificial intelligence; CSF: cerebrospinal fluid; ECG: electrocardiogram; EEG: electroencephalography; EMA: European Medicines Agency; FDA: U.S. Food and Drug Administration; HRV: heart-rate variability; MCI: mild cognitive impairment; PET: positron emission tomography.
Limitation and research gaps
Although combining EEG and HRV shows great promise for improving the detection and monitoring AD, several limitations still hinder its clinical adoption. Current research is fragmented, with differences in how data are collected, processed, and interpreted. Addressing these gaps is essential for transforming EEG-HRV coupling from an experimental concept into a reliable, standardized biomarker.
Methodological constraints
Current studies vary widely in their experimental design and technical settings. EEG recordings differ in the number of electrodes used, the type of filters applied, and the reference points chosen, while HRV analysis methods also range from simple time-domain measures to more complex non-linear models. Because of these inconsistencies, it is difficult to compare findings or perform meta-analyses across studies. Only a small number of investigations have used simultaneous EEG-ECG recordings, and even fewer have clearly described their recording conditions, such as whether participants had their eyes open or closed, were at rest, or engaged in cognitive tasks. Moreover, participants often differ in age, medication use, disease severity, and other health factors, making it challenging to isolate disease-specific effects.
Sample size and statistical power
Many studies are still based on small pilot samples, often fewer than thirty participants. This limited scale reduces the statistical power of the results and makes it harder to reproduce findings in other populations. In some cases, the differences between individuals within the same group are larger than the differences between groups themselves. Larger, multicenter studies that use harmonized protocols and share open-access datasets will be essential to confirm early findings and to calculate reliable effect sizes.
Standardization and reproducibility
There is currently no universal agreement on how EEG-HRV data should be pre-processed or analyzed. Researchers use different artifact-removal methods, frequency bands, and coupling metrics, such as phase-locking value, coherence, or entropy, to assess brain-heart relationships. This variability leads to mixed results that may reflect analytic choices rather than true biological differences. Developing shared methodological standards, benchmark datasets, and transparent code repositories will be crucial for building reproducible evidence.
Recommended guidelines could entail:
Defining core recording standards: at least 21 EEG electrodes, clear ECG lead placement for R-peak detection, a minimum of five minutes for reliable HRV estimation, and controlled environments that minimize noise and temperature fluctuations. Establishing pre-registered analysis pipelines that detail artifact detection, band-power definitions, coupling metrics, and statistical procedures. Such transparency will allow comparisons across studies and encourage collaboration.
Confounding factors and clinical variables
EEG and HRV signals are both sensitive to a wide range of physiological and environmental factors. Age, sex, cardiovascular disease, medications (such as beta-blockers or cholinesterase inhibitors), sleep quality, and even daily stress can significantly impact both EEG and HRV signals and, thus, influence results. The influence of such physiological and clinical confounders remains a major challenge in interpreting EEG-HRV coupling studies. While some studies adjust for these variables, others do not, which can obscure the interpretation of findings. Developing methodological guidelines and reporting standards will be essential for improving reproducibility and comparability in future EEG-HRV research in AD. Future research should consistently report and statistically control for these factors, using advanced approaches, such as multivariate modelling or propensity-score matching to isolate true disease effects.
Interpretation and causality
Most existing studies show that brain and heart activity change together in AD, but they do not clarify whether this is a cause or a consequence of the disease. Determining directionality requires longitudinal studies that track individuals over time and interventional research that tests whether improving brain-heart coupling through drugs, lifestyle, or neuromodulation can slow disease progression. These designs will be key to moving beyond correlation and toward understanding mechanisms.
Toward integrated multimodal frameworks
EEG-HRV coupling should be viewed as part of a broader, interconnected biomarker system rather than a standalone signal. Combining electrophysiological data with imaging, genetic, metabolic, and fluid markers could create a more complete picture of disease processes at multiple levels, from molecular changes to system-wide physiological disruption. Advances in machine learning and AI can help integrate these complex datasets, translating them into clinically meaningful insights for diagnosis, prognosis, and treatment monitoring.
Summary of research gaps
In summary, while current evidence clearly shows that EEG-HRV integration offers valuable insight into the loss of brain-body coordination in AD, the field remains in its early stages. Progress will depend on larger, collaborative, and standardized studies that unite neuroscience, cardiology, data science, and clinical medicine. Establishing consensus protocols, open data sharing, and clear causal frameworks will determine whether EEG-HRV coupling evolves from a promising research tool into a validated, scalable diagnostic platform for AD.
Conclusion
AD is increasingly recognized as a multisystem disorder that extends beyond isolated cortical pathology to encompass widespread autonomic dysregulation long before dementia becomes clinically apparent. The convergence of EEG and HRV research underscores this dual disturbance: cortical desynchronization mirrors a progressive decline in vagal tone, together reflecting the breakdown of the CAN. Examining these modalities together provides a dynamic lens into brain-heart coordination, an integrative physiological dialogue that gradually deteriorates as cognitive decline advances.
Synthesizing evidence from spectral, connectivity, and coupling studies reveals that simultaneous EEG-HRV analysis yields greater diagnostic sensitivity than either modality alone. EEG captures neural oscillatory slowing and disrupted network synchrony, while HRV quantifies the adaptive capacity of autonomic output. Their coupling represents the integrity of central–peripheral communication, a physiological synchrony whose disruption may precede overt neurodegeneration. Consequently, EEG-HRV coupling emerges as a promising biomarker for the preclinical and MCI stages of AD, offering early insight into disease trajectory.
From a translational standpoint, the joint assessment of EEG and HRV is low-cost, portable, and amenable to longitudinal monitoring across diverse settings, from specialized laboratories to community clinics. Establishing standardized acquisition protocols, artifact-correction pipelines, and normative reference ranges will be essential to ensure reproducibility and clinical reliability. Integration with artificial intelligence analytics and wearable sensor technologies could further enable continuous, home-based tracking, and thus, advance precision diagnostics even in primary-care or resource-limited contexts.
Looking ahead, the field must progress from correlation to causation. Longitudinal and interventional studies are needed to determine whether restoring brain-heart synchrony can modify disease progression. Embedding EEG-HRV coupling within multimodal frameworks that include neuroimaging, fluid, and genetic biomarkers may improve both diagnostic precision and prognostic value, paving the way for individualized disease monitoring.
Ultimately, uniting brain and heart physiology does more than enhance our understanding of AD as it opens a pathway toward personalized, accessible, and preventive neurology. By linking neural oscillations with autonomic rhythms, EEG-HRV coupling bridges the gap between the laboratory and the clinic, transforming two routine physiological measures into a single, integrated signature of cognitive health.
Footnotes
Acknowledgements
ORCID iDs
Author contribution(s)
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by an Alzheimer's Association Grant (AARG-NTF-22-928616).
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
