Abstract
Background
Epigenetic dysregulation is increasingly recognized as a key mechanism in the development and progression of Alzheimer's disease (AD). Herpes simplex virus type 1 (HSV-1) infection has been proposed as a potential biological trigger that may accelerate neurodegeneration through epigenetic modifications. Among HSV-1 structural proteins, glycoprotein B (HSV-gB) may influence host–virus interactions affecting neuronal gene regulation.
Objective
This study aimed to investigate the contribution of HSV-gB to AD-related epigenetic alterations and to determine whether HSV-gB exposure exacerbates epigenetic dysregulation in two in vitro neuronal AD models.
Methods
Human SH-SY5Y neuroblastoma cells were used to establish two AD models: a differentiation-based aging model induced by retinoic acid and brain-derived neurotrophic factor (RA + BDNF), and an amyloid aggregation model induced by amyloid-β 1–42 (Aβ1–42). Cells were treated with HSV-gB (190.5 pg/ml) alone or in combination with each model. Global DNA methylation, histone H3 and H4 acetylation, histone multiplex modifications, and HDAC3 and HDAC8 levels were analyzed using ELISA-based assays.
Results
HSV-gB exposure and RA + BDNF treatment induced global DNA hypomethylation and histone hypoacetylation, accompanied by significant increases in HDAC3 and HDAC8 levels. In contrast, the Aβ1–42 model showed DNA hypermethylation and histone hyperacetylation, indicating distinct epigenetic profiles between differentiation-associated aging and amyloid-driven pathology.
Conclusions
HSV-gB contributes to AD-related epigenetic dysregulation and may amplify neurodegenerative mechanisms through HDAC-mediated chromatin remodeling. The findings highlight divergent epigenetic signatures in different AD models and support a potential role for viral factors in modulating AD-associated epigenetic pathways.
Schematic illustration summarizing the effects of herpes simplex virus type-1 glycoprotein B (HSV-gB) on epigenetic regulation in two in vitro Alzheimer's disease models. HSV-gB induces global DNA hypomethylation, histone H3/H4 hypoacetylation, and increased HDAC3/HDAC8 levels in the RA + BDNF-induced aging-like model, whereas distinct epigenetic patterns are observed in the Aβ₁–₄₂-induced amyloid model. The graphical abstract was created by the authors based on the findings of the present study.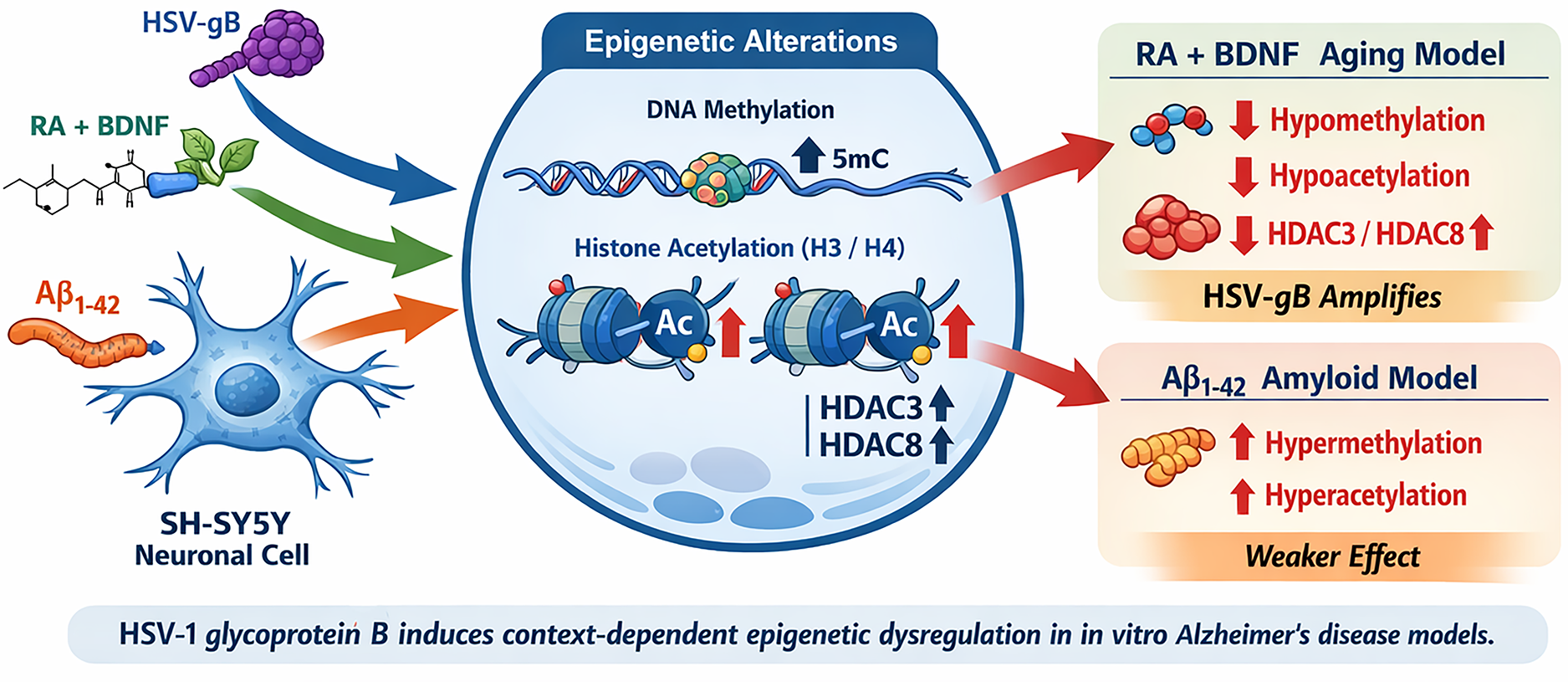
Keywords
Get full access to this article
View all access options for this article.
