Abstract
Background
Compounds derived from the plant Cannabis demonstrate many therapeutic properties suggesting that they could delay the onset and progression of Alzheimer's disease (AD).
Objective
The goal of the present experiment was to observe the effects of chronic cannabidiol (CBD) administration on the behavior and brain pathology of an AD tauopathy mouse model, Tau P301S-Line PS19 mice.
Methods
Mice were orally given CBD (20 mg/kg) or vehicle, daily, beginning around 3 months of age. At 6 months old, mice were tested on a battery of tasks to assess object recognition, motor function, and spatial learning and memory. The mice were retested at 9 months old on the behavioral tasks and the fear conditioning paradigm was added. Following completion of behavioral testing, the mice were perfused for histological analysis.
Results
Chronic CBD treatments did not appear to affect the behavior nor restore the reduced hippocampal volume of Tau P301S mice. However, a deeper assessment of the changes in inflammatory markers showed a treatment effect on a measure of microglia reactivity. Robust sex differences were revealed with Tau P301S males showing more severe pathology relative to females. Finally, daily treatments of CBD did not negatively impact the behavior or brain of any of the experimental groups suggesting that its chronic administration was relatively safe.
Conclusions
Taken together, the results suggested that CBD can have beneficial effects on some of the pathology associated with AD, even in an aggressive model of this neurodegenerative disease, but the impacts on impaired behavior were minimal.
Keywords
Introduction
Alzheimer's disease (AD) is classified by, among other things, the presence of neurofibrillary tangles (NFTs), which are intraneuronal lesions related to the hyperphosphorylation of tau protein and paired helical filaments (PHFs). While NFTs are associated with a variety of neurodegenerative diseases, known as tauopathies,1,2 they progress in a distinct manner during the pathological progression of AD which aids in distinguishing AD from the alternate tauopathies. Braak and Braak 3 distinguished six distinct stages of the pathological progression of abnormal tau and NFTs. Specifically, the pathologies have been reported to begin developing in the transentorhinal region, progressing throughout the entorhinal cortex, the hippocampus (HPC), and eventually the isocortex. 3 Once a stage becomes apparent the progression of tau will not reverse. 4 This is particularly concerning because NFTs are directly linked to inflammation, synapse loss, neuronal loss, and ultimately cognitive decline among AD cases.5,6 Therefore, treatments that prolong the formation of NFTs would likely benefit AD patients by delaying the progression and accumulation of the lesions.
Specifically, treatments that prevent the hyperphosphorylation of tau are particularly intriguing. Hyperphosphorylated tau precedes PHFs and NFTs, suggestive of its early therapeutic potential as a target.7–9 While the direct cause of hyperphosphorylated tau is unknown there are several factors believed to be involved in the process. Research suggests that an imbalance between tau kinases and phosphatases might contribute to its hyperphosphorylation.9,10 In support of this hypothesis, the activation of glycogen synthase kinase 3-β (GSK3-β), a tau kinase, is increased in the brains of AD patients, 11 while protein phosphatase 2A (PP2A), a tau phosphatase has been shown to be decreased. 10 Furthermore, the phosphorylation of tau in microglial cell cultures has been correlated to increase GSK3-β activity and decrease PP2A activity. 12 Thus, treatments that decrease activated GSK3-β and/or increase PP2A activity might aid in preventing the hyperphosphorylation of tau by regulating the phosphorylation and dephosphorylation of the protein. In addition to an imbalance between tau kinases and phosphatases, alternate AD pathologies have been correlated with the phosphorylation of tau. Indeed, increased amyloid-β (Aβ),13–16 oxidative stress, 17 and inflammation 18 have been shown to promote tau phosphorylation.
Interestingly, neuroinflammation is normally an adaptive response in the brain. In the absence of disease, inflammation promotes the uptake and degradation of toxic species and helps maintain homeostasis in the central nervous system (CNS) through the release of pro-inflammatory cytokines and anti-inflammatory cytokines.19,20 More specifically, microglia, which are the primary immune response in the CNS utilize their long dendritic processes to communicate with their environment.19,20 In the presence of pathology, microglia will become active. Reactive microglia change in shape and can elicit phagocytosis. 21 However, when chronically activated or in the presence of disease, the normal function of microglia can be disrupted. The toxic effects of chronic microglia have been correlated to AD pathologies and neuronal damage in pre-clinical AD cases, further supporting their potential role in the descent into dementia.22,23 Microglia induced by Aβ are believed to contribute to oxidative stress, synapse loss, and neuronal damage.19,22,24–26 In support of this, it is widely accepted that in neurodegenerative diseases, chronic active microglia release excess amounts of pro-inflammatory cytokines thereby continuously pruning synapses and degrading neurons.19,27 Inflammatory inhibitors have been shown to reverse these negative effects, thereby supporting microglia as therapeutic targets. 28
Furthermore, microglia are thought to signal the activation of astrocytes, another inflammatory response in the brain.29,30 Once activated, astrocytes are usually capable of phagocytosis, regulating microglial synaptic pruning, and maintaining homeostasis in the CNS.29,31 However, it is believed that the function of astrocytes is disrupted in the presence of AD mutations.32,33 Indeed, reactive astrocytes are closely linked to plaques and the soluble forms of Aβ appear to alter the ability of astrocytes to use phagocytosis on toxic species.32,33 Additionally, like microglia, astrocytes have been shown to release pro-inflammatory cytokines, which have been shown to upregulate the APP gene and promote the cleavage of Aβ. 34 As such, the malfunction of astrocytes among AD brains in conjunction with the release of pro-inflammatory cytokines might contribute to progression of AD in the brain.
Within the intricate landscape of AD pathology, the relationship between tau hyperphosphorylation and inflammation emerges as a pivotal axis contributing to the pathogenesis of the disease. Tau pathology is intertwined with inflammatory processes, as activated microglia and astrocytes respond to the abnormal tau aggregates, releasing pro-inflammatory mediators. 35 The crosstalk between tau hyperphosphorylation and inflammation creates a vicious cycle that exacerbates neuronal dysfunction and accelerates the progression of AD. Treatments that target chronic neuroinflammation might improve the progression of the disease. However, multiple neuropathological changes occur throughout the course of AD. Targeting one pathology might not be as effective as treatment options that target multiple pathologies. Despite that multiple factors contribute to disease onset and prognosis; current treatment options often only target one pathology.
Cannabidiol (CBD) is a safe, readily available, and relatively inexpensive treatment option that has been shown to improve pathologies associated with AD. Certainly, in cell cultures, CBD has been shown to directly reduce the hyperphosphorylation of tau via inactivation of GSK3-β, 15 and the aggregation of tau. 36 Furthermore, CBD treated cell cultures have been shown to reduce Aβ, oxidative stress, and neuroinflammation.35,37–40 In addition to directly and indirectly preventing the hyperphosphorylation of tau, CBD has been shown to prevent neurodegeneration, while promoting neurogenesis. 41 Neurodegeneration was correlated to hyperphosphorylated tau and NFTs.5,42 Finally, CBD was able to promote spatial learning and memory in a tauopathy mouse model, further supporting the therapeutic potential of the cannabinoid compound. 43
The present study sought to assess the effects of chronic administration of CBD on the behavior and brain pathology of Tau P301S-Line PS19 mice. The dosage was chosen based on pilot work in our lab and observations of previous experiments.44–46 The Tau P301S mice and their noncarrier littermate controls were orally administered 20 mg/kg of CBD or vehicle, daily, beginning around 3 months of age. Tau P301S mice do not demonstrate cognitive deficits at this age, 47 allowing for cognitively pre-symptomatic assessment of CBD administration. Around 6 months of age, when the mice have been reported to demonstrate cognitive deficits, 48 they were tested on a battery of tasks to assess object recognition, motor function, and spatial learning and memory. Due to a lack of observed impairments at 6 months of age, the mice were retested at 9 months old on the behavioral tasks and the fear conditioning paradigm was added. Following all behavioral testing, half the mice were perfused for histological analysis, and the other half were sacrificed for fresh tissue collection. The perfused brains were used to quantify the HPC volumes of the mice and to assess the quantity of ionized calcium binding molecule adaptor 1 (Iba1), a microglia marker, and glial fibrillary acidic protein (GFAP), an astrocytic marker, in the HPC and prefrontal cortex (PFC).
It was hypothesized that the cognitively pre-symptomatic administration of CBD would improve any observed learning and memory impairments among the Tau P301S mice. CBD was not expected to impact the learning and memory of the controls. It was predicted that the Tau P301S mice would demonstrate reduced HPC volumes in conjunction with increased levels of Iba1, GFAP, and tau in the HPC and PFC. CBD was expected to mitigate the observed pathologies. No histological abnormalities were expected to be observed in the controls and CBD was not expected to impact the brains of this group.
Materials
All methods and procedures described in this experiment were in accordance with Canadian Council of Animal Care Guidelines and were approved by the University of Lethbridge Institutional Animal Welfare Committee (Protocol #1918).
Subjects
The Tau P301S-Line PS19 mice demonstrate synaptic dysfunction, mitochondrial dysfunction, oxidative stress, neuroinflammation, hyperphosphorylated tau, NFTs, and neurodegeneration in an age dependent manner.49–54 Research suggests that the progression of hyperphosphorylated tau in the Tau P301S mice progresses similarly to the characterized stages of NFTs, 3 with the exception that the brains of the Tau P301S mice progress at a quicker rate relative to the human stages. 50 Furthermore, the hyperphosphorylation of tau in Tau P301S mice has been correlated with increased levels of GSK3-β.49,51 Finally, in addition to the development of AD related pathologies, including neuroinflammation, the mice demonstrate cognitive impairments.47–49,51,54
Twenty-five heterozygous Tau P301S-Line PS19 (PS19) (male (n = 16); female (n = 9)) and twenty-two noncarrier littermates (WT) (male (n = 8); female (n = 14)) were bred on a c57BL/6N background at the University of Lethbridge rodent facility. The mice were housed with littermates in individually ventilated caging (IVC) cages with passive airflow, which contained corncob bedding, Crink-l’Nest, a cotton square, and a plastic tunnel for enrichment. The housing room was kept on a 05h00–17h00 light/dark cycle and held at a constant temperature of 21 °C, with 35% humidity. Prior to commencing the experiment, WT and PS19 mice were assigned to receive vehicle or CBD using simple randomization. More specifically, within each cage, we used simple randomization to assign each mouse to a treatment condition considering relatively equal distribution of litter, genotype and sex. The mice were divided into four groups based on genotype and treatment: noncarrier littermates-CBD (WTC) (n = 11), noncarrier littermates-vehicle (WTV) (n = 11), Tau P301S-CBD (PS19C) (n = 13), and Tau P301S-vehicle (PS19V) (n = 12). The mice had ad libitum access to food and water. Supplemental Table 1 indicates each subject's inclusion throughout the study.
Drugs preparation and administration
The CBD was diluted in PC Grapeseed oil (No Frills). The oil was filtered using a syringe and a 0.22 sterile filter unit (FroggaBio). A stock solution of 20 mg/mL CBD oil was mixed, and high-performance liquid chromatography (HPLC) was performed to confirm the concentration of the CBD oil. The solution was aliquoted in 1.5 mL tubes and stored at −20 °C. CBD oil was stored in a 4 °C fridge for up to 5 days while being used.
The drug (CBD oil or vehicle) was mixed in 0.25 g of Nutella and orally administered daily starting at 3 months old. During treatments, the mice were transferred from their housing cages into individual holding cages, which contained a weigh boat with the Nutella/drug mix. The mice were weighed every second day to determine the amount of drug (µL) they would receive (20 mg/kg, body weight). The mice were placed in the holding cages for a maximum of one hour or until every mouse had finished eating their Nutella/drug mix, any remaining Nutella was weighed and noted. Holding cages were cleaned daily with Virkon.
Behavioral assessments
Around 6 and 9 months of age, novel object recognition (NOR), followed by balance beam, tail suspension, and Morris water task (MWT) were performed. Fear conditioning (FC) was performed at 9, but not 6 months of age to avoid any training effects from the task. Videopad editor was used for behavioral tasks that were manually scored. All videos were scored by an experimenter blind to the genotype and treatment groups.
Novel object recognition. NOR was performed as previously described.55,56 A square white plastic context (46 cm×48 cm) with corncob bedding placed at the base of the arena was used. Each mouse was individually placed in the context for their given trial. The bedding was shuffled between trials to eliminate olfactory cues. All males were tested prior to females. Before the females were placed in the context, the arena was cleaned with 70% ethanol, and the corncob bedding was replaced to eliminate any odors from the males. After each trial in which objects were present, they were cleaned with 70% ethanol to eliminate olfactory cues. Habituation: The mice were placed in the context and given 5 min to freely explore, daily for 6 days. Training: Twenty-four hours after the last habituation day the mice were placed in the context with two identical objects (A, A or B, B) that were positioned at an equal distance from the walls and given 10 min to explore the context and objects. Testing: Twenty-four hours after training the mice were given a 5 min test in which one familiar object was replaced with a novel object (A, B or B, A). The behavior of the mice was recorded in the contexts for further analysis. The videos were manually scored, and exploratory behavior was defined as time (s) spent within 1 cm of the objects and facing the objects. Looking past the objects, sitting on the objects, and digging near the objects was not considered object-directed exploratory behavior. The investigation ratio (IR) for the novel object was calculated by dividing the time spent exploring the novel object by the total exploration time for both objects, combined. 55 Mice are naturally exploratory, therefore increased exploration for the novel object is suggestive of recall for the familiar object. 56 Recall for the familiar object indicates that the mice retained object recognition thought to be dependent on the function of networks related to the perirhinal cortex.57,58
Balance beam. Balance beam assessed the motor coordination of the mice. 59 During balance beam there was one training day followed by one testing day. Training and testing were performed using a beam that was elevated 50 cm above the table, 100 cm in length, and 2 mm wide. There was a black tunnel at the end of the bean for the mice to traverse to. Training: Mice were placed on the beam from different distances from the end of the beam (10 cm, 50 cm, and 100 cm). The mice did not move onto the farther start points until they were able to successfully traverse the beam. If the mice began to slip during their given trial, an experimenter assisted them back onto the beam. A trial was considered successful if the mice did not fall off prior to reaching the tunnel. Training was complete when the mice successfully traversed the beam from each start point and successfully completed three trials from 100 cm. Testing: Twenty-four hours later, mice were placed 100 cm from the end of the beam and given three consecutive trials. When the head of the mouse was inside the tunnel the trial was considered successfully complete. If the mice fell off the beam, the trial was not considered successful. The behavior of the mice was recorded during testing for analysis. The average latency of the mice to traverse the beam during successful trials and the distance travelled across all three trials were manually scored. A decreased latency and an increased distance travelled was suggestive of intact normal motor coordination.
Tail suspension. Tail suspension assessed the hind limb clasping of the mice while they were suspended by the middle of their tail for 10 s. The behavior of the mice while suspended was recorded for clasping analysis and was scored on a four-point scale as previously described.60,61 Two experimenters scored tail suspension. Any scores that varied between experimenters were observed by a third person. A score of 0 indicated a normal splay of limbs, a score of 1 indicated one limb slightly retracted forward, a score of 2 indicated two limbs slightly retracted forward with toes splayed, a score of 3 indicated two limbs slightly retracted forward with occasional clasping and toes clasped, a score of 4 indicated limbs completely retracted forwarded and clasped along with the clasping of toes. The scores were given based off the clasping behavior of the mice for 50% of the video. A higher clasping score was indicative of hind limb weakness.
Morris water task. MWT consisted of eight spatial training days, followed by one probe day, and one visible platform day. 62 A retention probe test was performed to assess the long-term memory of the mice at 6 but not 9 months of age. The test was not replicated at 9 months of age due to the spatial training impairments already observed among the PS19 mice at the latter age. Furthermore, at 9 months old the mice were trained to locate a hidden platform in a different quadrant relative to that of 6 months of age to avoid training effects. Novel distal cues were also used at this time point. Apparatus: A large white circular fiberglass pool (50 cm height and 154 cm in diameter) was elevated onto a platform and filled with water to 40 cm. Across all days, the temperature of the water was 22 °C (± 1 °C) and non-toxic white paint was placed in the pool to render the water opaque white. Each day the mice were individually placed in holding cages and allowed to acclimate for 30 min in the MWT room. Spatial training: During training, a platform was placed in a fixed location, approximately 0.5–1 cm below the water. Distal cues (posters of different geometry and size) were positioned around the pool to aid the animals in learning to locate the hidden platform. Other room cues were the computer, holding rack, sink, door, and experimenter. Each training day consisted of four trials with each mouse being placed in the pool facing the wall from a different start point (N, S, E, or W). The start points were counterbalanced across days. During each trial, the mice were given 60 s to locate the hidden platform, if they failed to locate the platform an experimenter guided the mice with their hand to the location. Once the mice were on the platform, they were left to sit for 10 s prior to being removed. Once removed, the mice were dried with a towel and placed back in their holding cage until their next trial. Probe: To assess the spatial recall of the mice, a single probe trial with the platform removed was performed 24 h after the final training day. During the probe trial the mice were placed in the pool from one of the two start locations farthest from the trained platform location. The mice were left to swim in the pool for 60 s prior to being removed. Immediately following the probe trial, the platform was placed back in the pool in the same previous location and mice were retrained for three trials. Retention test: A week following the probe trial and retraining, the retention test occurred. The platform was removed, and the mice were placed in the pool for 60 s from one of the two start locations farthest from the prior platform location. Visible platform: This test was performed 24 h after the retention test at 6 months of age and 24 h following the probe trial at 9 months old. The visible platform test was used to rule out any sensory, motor, or motivational deficits in the mice. The platform was 1 cm above the water in a novel location in the pool and a black cylinder was placed on top of the platform and visible cues were moved to novel locations. The same training procedure was used as during spatial training. HVS-Image software package tracked the behavior of the mice for analysis. Retained spatial memory suggests that networks centered around the HPC remain functional.63–65
Fear conditioning. At 9 months of age the mice were given a few days to rest after MWT before beginning FC. FC consisted of one day of conditioning, followed by context or tone recall performed over two days. 55 The order in which the mice experienced context or tone recall was counterbalanced across genotype and treatment group. During conditioning and tone/context recall, the mice were individually placed in holding cages containing corncob bedding. The mice were not placed back in their housing cages until all cage mates were tested to eliminate any additional stressors. Conditioning: The mice were brought individually into the conditioning room and left to sit for 2 min prior to being placed in the context. The context was an acrylic square box (33 cm×33 cm×25 cm) that consisted of two black walls and two white walls, with grid flooring, which was attached to a Lafayette Instruments shock generator. Once placed in the context, a plexiglass cover was laid over the top to limit access to cues in the testing room and to prevent the mice from jumping out. After a 2 min baseline period in the context a 20 s, 2000 Hz tone was presented. The tone was co-terminated with a 2 s, 0.5 mA shock. The mice received five tone/shock pairings that were spaced 2 min apart. Following the last tone/shock pairing, the mice were left in the context for 1 min prior to being removed. Context or tone recall occurred 24 to 48 h following conditioning. The mice were individually brought into a novel experiment room and left to sit for 2 min. Context recall: The mice were placed in the same context used during conditioning; however, no tone or shock was administered. The mice were given 5 min in the context to freely explore. Tone recall: The mice were placed in a novel white triangular chamber (33 cm×33 cm×29 cm) and were given 2 min to explore the context. After this period a 20 s, 2000 Hz tone was presented 3 times at 2 min intervals, however no shock was administered. Following the last tone, the mice were left for 1 min prior to being removed. During context and tone recall the behavior of the mice was recorded and their time spent freezing was manually scored. Freezing time was defined as the mice not moving. Rearing on the wall, remaining in a stretched-out positioning, and sniffing was not considered time spent freezing. The percent time spent freezing was calculated by dividing the total time the mice spent freezing by the total time being scored, times by 100. The first two minutes during tone recall was used as a baseline measure of freezing behavior for the mice. Increased freezing in the presence of the context and tone indicates recall for the context/shock and tone/shock pairings and is suggestive of intact function of networks related to the amygdala.66,67
Brain assessments
Perfusions. Around 10 months of age, 27 mice (WTV: n = 6; WTC: n = 6; PS19V: n = 7; PS19C: n = 8) were given an intraperitoneal injection of sodium pentobarbital (300 mg/kg, body weight) and transcardially perfused with phosphate buffered saline (PBS) and 4% paraformaldehyde (PFA) in PBS. The brains were extracted and kept in 4% PFA for 24 h prior to being placed in 30% sucrose with PBS and 0.02% sodium azide. The brains were stored at 4 °C and were serially sectioned coronally at 40 µm with a freezing microtome. The remaining mice were sacrificed for fresh tissue collection under deep anesthesia with Isoflurane.
Nissl stain. Nissl was performed on all perfused brains for volumetric analysis of the HPC. The cells in the dentate gyrus (DG), cornu ammonis 1 (CA1), and cornu ammonis 3 (CA3) were counted using the Cavalieri method on a stereology microscope at 10 X magnification. The Allen mouse brain atlas was used to ensure the proper regions were being counted. 68
Immunohistochemistry (IHC). IHC procedures were performed on all perfused brains for neuroinflammation, cholinergic status, and tau protein accumulation. Microglia were stained with Iba1, astrocytes were stained with GFAP, cholinergic neurons were stained with choline acetyltransferase (ChAT), and phosphorylated tau were stained with AT8. The brain slices were free float mounted in PBS onto positively subbed slides. Washes were done in tris buffered saline (TBS). To prevent nonspecific staining, sections were blocked in TBS with 0.3% Triton-X and 3% goat serum for 2 h. Sections were then washed and incubated in primary antibodies anti-GFAP (rabbit, Abcam, Ab7260, 1:2000); anti-Iba1 (rabbit, Wako, 019–19741, 1:1000); anti-ChAT (monoclonal, rabbit, Ab178850, Abcam, 1:500)); anti-AT8 (monoclonal, mouse, Thermo, MN1020, 1:500) in a dark humid chamber for 24 h. Following the primaries, the sections were washed and placed in the secondaries (anti-rabbit-alexa-594 (IgG [H + L], goat, Invitrogen, A11037, 1:1000; anti-mouse-alexa-488 (IgG (H + L) goat, Abcam, Ab150113, 1:100) for 24 h. Finally, sections were counterstained with Dapi. A stock solution of 20 µg/mL of Dapi was diluted at a 1:2000 concentration in TBS. The diluted solution was pipetted onto the slides and were coverslipped for 1 h in a dark humid chamber. Following the incubation period in Dapi, slides were washed and coverslipped with Vectashield H-1000. Full slides were imaged using Nanozoomer microscope (Nanozoomer 2.0-RS, HAMAMATSU, JAPAN). The CA1 and CA3 of one dorsal HPC slice and one ventral HPC slice were imaged for each animal using an Olympus VS-120 digital slide scanner using a 100 X oil objective (UPlanSApo, 100x/1.40 oil, ∞/0.17/FN26.5) and Olympus VS-ASW FL software. Z stacks were comprised of 17 images spaced 0.59 µm apart with a total working distance of 10 µm.
Quantification. ImageJ was used to quantify the number of cholinergic neurons in the medial septum and diagonal band (MSDB) and percentage of phosphorylated tau in HPC. ImageJ and Ilastik were used to quantify the percentage of Iba1 and GFAP in the whole PFC and HPC. Neurolucida® 360 was used to quantify the precise process volume, convex hull, and cell body volume in the CA1 and CA3 regions of the Iba1 stained brain slices. Obtaining the precise process volume, convex hull, and cell body volume allowed us to infer reactivity of the microglia. ImageJ and Ilastik: IHC stains were quantified separately. Cholinergic neurons were quantified using every 4th slice in the MSDB region with 4–6 slices being counted per brain. The cholinergic counts were divided by the total number of slices to obtain a count/slice. Tau quantification involved manually cropping HPC in ImageJ using polygon selections and then splitting the cropped images into RGB channels. The blue channel was subtracted from the green channel using the Image Calculator. The resulting image was automatically thresholded, and the area covered by AT8-positive tau staining was quantified. Area measurements from all slices of each brain were summed, converted from pixels to mm2, and divided by the total HPC area to calculate the average percentage of tau coverage per brain slice. For GFAP and Iba1, the HPC and PFC of the exported images were cropped using ImageJ and the area of the HPC and PFC for each slice was obtained. The cropped images were batch processed through Ilastik. When training Ilastik to quantify inflammation (GFAP and Iba1), markers that appeared like active microglia or active astrocytes were counted. Active microglia were characterized by enlarged cell bodies with short processes. Active astrocytes were characterized by enlarged cell bodies with long dendrites. Ilastik provided an excel file with the size of each marker counted for every brain slice which were combined to obtain data for the whole brain for each animal. The sum size of every marker counted for the whole brain was obtained and divided by the number of slices analyzed to receive the average size per slice. The average size of the markers per slice was then divided by the average volume of the brain region for each animal and times by 100 to receive the percent area covered (mm2). Neurolucida® 360: Using Neurolucida® 360 the cell body and processes of two microglia were traced per image, making a total of 4 tracings completed for each animal. A total of 108 microglia were traced. Microglia were chosen at random, with the guideline that the whole microglia must be visible within the z-stack. The cell body and processes were traced at every micron imaged and each process that was in direct contact with the cell body was individually traced. The tracing began where the process touched the cell body and continued until the branch had no further connections. If the process split into multiple branches, bifurcating node or trifurcating node were appropriately chosen and the tracing continued. The tracings appropriately matched the width of the process and branches. Once the whole process along with the branches were completed the tracing was terminated for that process. Once all the processes in direct contact with the cell body were traced, the tracing for the microglia was terminated. The following data was collected from the traced microglia: (1) process volume (2) convex hull surface area (3) cell body volume.
Results
Data analysis
The statistical analysis was performed using SPSS statistical software package, version 27.0. Results are presented as mean ± SEM. ANOVAs were performed to elucidate significant differences in genotype, treatment and their interactions for the behavioral data. Two-way ANOVAs were performed to determine significant differences in genotype, treatment, and their interactions for brain pathology measures. A p-value < 0.05 was considered as statistically significant. Bonferroni corrections were used for post hoc comparisons. For behavioral testing, larger group sizes were required for statistical power, thus males and females of the same genotype and treatment were grouped together for statistical analysis. For brain pathology measures, more robust data can be obtained from each subject and therefore males and females were analyzed separately for each genotype and treatment. Supplemental Table 1 indicates the behavioral tasks and histological assessment each subject was included in for the study.
Behavioral assessments
Novel object recognition. The training data was analyzed and any mouse that did not spend a total of 20 s exploring the objects during training was excluded from the testing data. No mice were excluded from the testing data at 6 months of age. However, 3 controls treated with CBD were excluded from the testing data at 9 months old. Therefore, the end group sizes at 9 months of age were: WTV (n = 11), WTC (n = 8), PS19V (n = 12), PS19C (n = 13).
An IR above 0.5 indicates that the mice explored the novel object more than the familiar object. As seen in Figure 1 (A) all the mice, regardless of genotype, had an IR value well above 0.5 at 6 months of age. Whether the mice received CBD, or the vehicle did not impact object recognition of the mice. A two-tailed one-samples t-test was conducted on the IR to determine if the exploration for the novel object was significantly above chance. The results suggest that the WTV (t (10) = 3.467, p < 0.006), WTC (t (10) = 3.859, p < 0.003), PS19V (t (11) = 4.694, p < 0.001), and PS19C (t (12) = 4.622, p < 0.001) explored the novel object significantly above chance. A two-way ANOVA assessed the object recognition of the mice and indicated no significant effect of genotype (F (1, 43) = 0.13, p = 0.72), treatment (F (1, 43) = 0.003, p = 0.96) nor genotype x treatment interaction (F (1, 43) = 0.51, p = 0.48). Thus, at 6 months of age the PS19 and WT mice retained object recognition and CBD did not impact their object recognition.
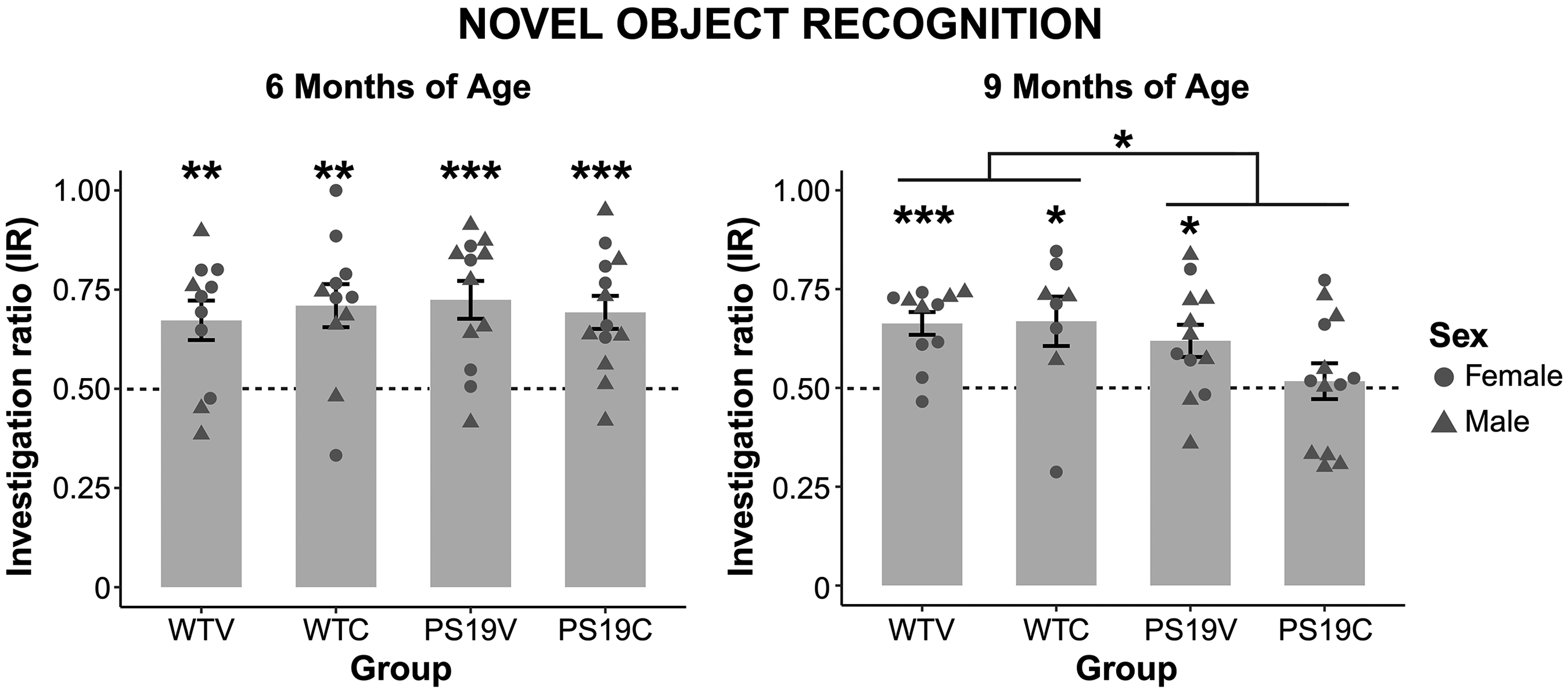
The average investigation ratio (IR) for the novel object during novel object recognition (NOR) for noncarrier littermates (WT) and Tau P301S (PS19) mice treated with a vehicle (V) or 20 mg/kg of CBD (C). (left) The NOR data at 6 months of age. Each group had an IR value significantly above 0.5, indicating that they explored the novel object above chance relative to the familiar object (WTV (**p < 0.006), WTC (**p < 0.003), PS19V (**p < 0.001), and PS19C (***p < 0.001). (right) The NOR data at 9 months of age. The object recognition of the PS19 mice was impaired relative to the WT mice (*p < 0.034). The WTV (***p < 0.001), WTC (*p < 0.031), and PS19V (*p < 0.014) explored the novel object significantly above chance, but the PS19C group did not. There was no effect of treatment at either age.
The NOR data collected from the groups of mice around 9 months of age can be seen in Figure 1 (B). The IR value of the PS19C mice was the lowest followed by the PS19V mice. The performance of the WT groups was similar. CBD might be impairing the object recall of the PS19 mice, but CBD does not appear to impact the memory of the WT mice. A two-tailed one-samples t-test revealed that the IR of the WTV (t (10) = 5.665, p < 0.001), WTC (t (7) = 2.669, p < 0.031), and PS19V (t (11) = 2.925, p < 0.014) was significantly above chance, but the novel object exploration for the PS19C mice (t (12) = 0.37, p = 0.72) was not. A two-way ANOVA suggested that there was a significant effect of genotype (F (1, 40) = 4.794, p < 0.034), but no effects of treatment (F (1, 40) = 1.17, p = 0.29), nor genotype x treatment interaction (F (1, 40) = 1.44, p = 0.24), Therefore, at 9 months of age the PS19 mice were impaired relative to the WT mice. CBD might be further impairing the object recall of the PS19 mice. However, CBD did not impact the object recognition of the WT mice.
Balance beam. The average latency to traverse the beam during successful trials was assessed. Figure 2 (A) demonstrates the latency of the mice depending on genotype and treatment. The CBD treated mice had a shorter latency relative to the vehicle mice. There were no observable differences between genotypes. A two-way ANOVA suggested that there was a significant effect of treatment (F (1, 43) = 4.168, p < 0.047) but no effects of genotype (F (1, 43) = 0.003, p = 0.96), nor interaction (F (1, 43) = 0.09, p = 0.77) on the latency of the mice. The average distance travelled across the three trials is depicted in Figure 2 (B). There were no observable differences depending on treatment. The PS19 mice appear to have a slight shorter distance travelled relative to the WT mice, suggesting that they successfully completed fewer trials than the WT mice. However, a two-way ANOVA revealed no effects of genotype (F (1, 43) = 0.62, p = 0.43), treatment (F (1, 43) = 0.16, p = 0.69), nor interaction (F (1, 43) = 0.02, p = 0.88) on the average distance travelled. The average number of paw slips while traversing the beam (Figure 2 (C)) was also assessed with a two-way ANOVA, indicating no significant effects of genotype (F (1, 43) = 2.56, p = 0.12), treatment (F (1, 43) = 0.36, p = 0.55) nor interaction (F (1, 43) = 2.61, p = 0.11). In summary, at 6 months of age the motor coordination of the PS19 mice was unimpaired compared to WT mice. CBD, in part, appeared to be promoting the motor coordination of the mice, independent of genotype at this age.
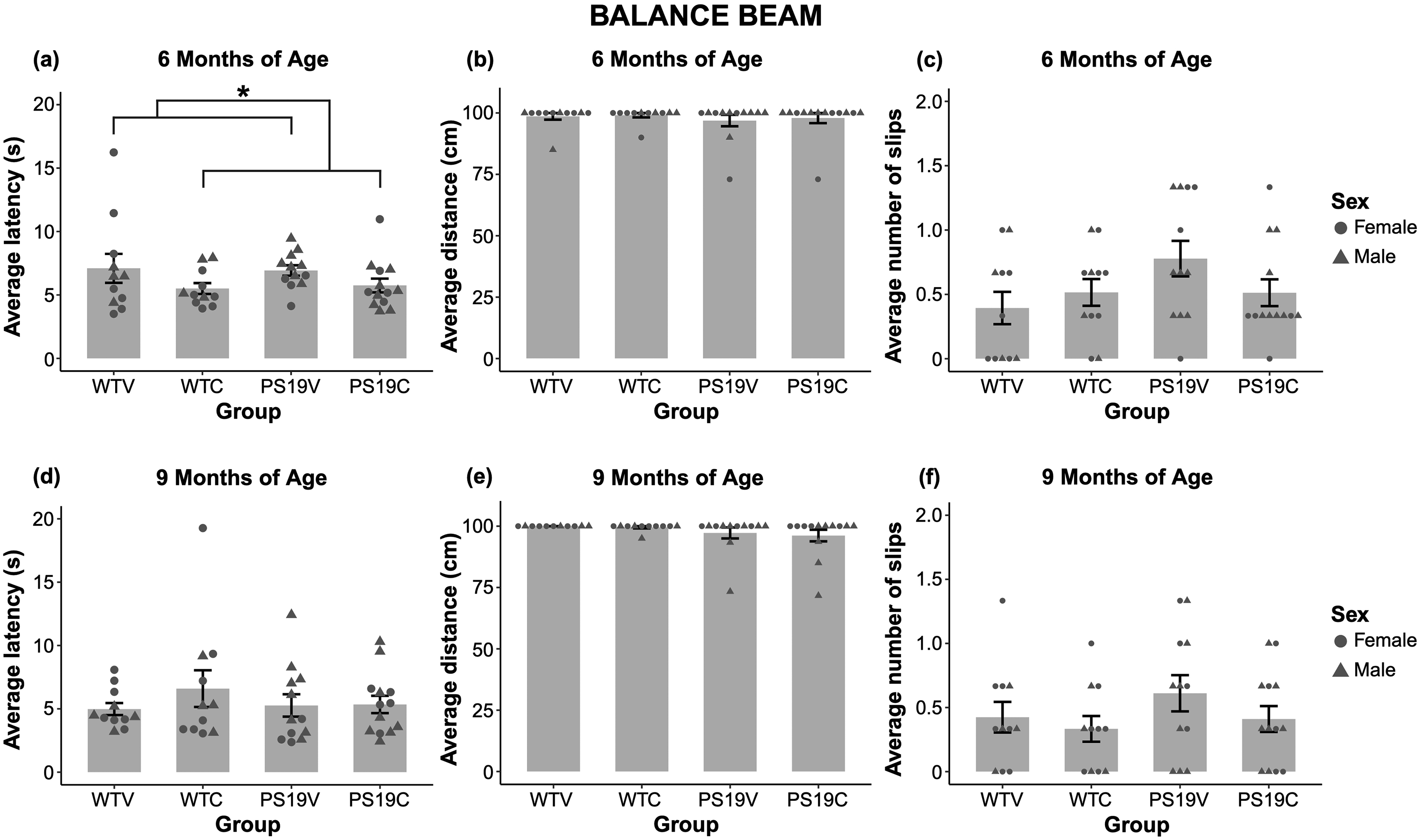
The balance beam data collected from the WT mice and PS19 mice treated with vehicle (V) or CBD (C) at 6 months old (top row) and 9 months old (bottom row). (A) The average latency of the mice to traverse the beam during successful trials. The CBD treated mice appear to have a shorter latency relative to the vehicle treated mice (*p < 0.047). There was no significant effect of genotype. (B) The average distance travelled across all three trials was similar, regardless of genotype and treatment. (C) There were no significant effects of genotype nor treatment on the average number of paw slips during successful trials. (D) The average latency of the mice to traverse the beam during successful trials was similar, regardless of genotype and treatment. (E) There was no significant effect of genotype nor treatment on the average distance travelled across all three trials. (F) The average number of paw slips during successful trials were similar in all groups.
At 9 months of age the average latency to traverse the beam during successful trials did not appear to differ depending on the treatment and genotype of the mice (Figure 2 (D)). A two-way ANOVA confirmed no significant effects of genotype (F (1, 43) = 0.08, p = 0.78), treatment (F (1, 43) = 0.86, p = 0.36), nor genotype x treatment interaction (F (1, 43) = 1.16, p = 0.29). As seen in Figure 2 (E) the PS19 mice have a shorter distance travelled across all three trials relative to the WT mice, suggesting that their motor coordination may have been beginning to decline at this age. CBD does not appear to impact the average distance travelled. A two-way ANOVA analyzed the data and despite the observed results there was no significant effects of genotype (F (1, 43) = 3.03, p = 0.09), treatment (F (1, 45) = 0.18, p = 0.67) nor interaction (F (1, 43) = 0.28, p = 0.87). A two-way ANOVA performed on the average number of paw slips also indicated no significant effects of genotype (F (1, 43) = 1.93, p = 0.17), treatment (F (1, 43) = 1.56, p = 0.22) nor interaction (F (1, 43) = 0.53, p = 0.47) (Figure 2 (F)). Overall, the PS19 mice maintained intact motor coordination at 9 months of age and CBD treatments did not impact the motor coordination of the mice at this age.
An age-analysis was performed to compare the mice at 6 to 9 months of age on ability to traverse the beam. A three-way ANOVA revealed that there were no significant effects of age for genotype, treatment or distance travelled on the beam (all p's > 0.05). Similarly, an assessment of paw slips indicated no difference in genotype or treatment at the different ages (all p's > 0.05), An analysis on latency to traverse the beam indicated no significant effects of genotype or treatment, but a marginally significant age x treatment effect was observed (F (1, 43) = 3.922, p = 0.054). To further elucidate this interaction, post hoc comparisons with Bonferroni corrections indicated that CBD reduced latency compared to vehicle at 6 months (p < 0.047) but not 9 months of age (p = 0.36).
Tail suspension. The hind limb clasping of the mice was scored on a four-point scale (0–4). At 6 months of age there were no observable differences in the hind limb clasping of the mice depending on genotype or treatment (Figure 3 (A)). A two-way between subjects ANOVA confirmed no significant effect of genotype (F (1, 43) = 0.004, p = 0.95), treatment (F (1, 43) = 2.31, p = 0.14), nor genotype x treatment interaction (F (1, 43) = 0.97, p = 0.33). However, as seen in Figure 3 (B), at 9 months of age, the PS19 mice did demonstrate increased clasping relative to the WT mice. There were no observable differences in the clasping of the animals receiving CBD or a vehicle. A two-way between subjects ANOVA confirmed a significant effect of genotype (F (1, 43) = 10.644, p < 0.002), but no significant effects of treatment (F (1, 43) = 0.93, p = 0.34) nor interaction (F (1, 43) = 2.43, p = 0.13). The results suggest that the PS19 mice demonstrated hind limb weakness at 9, but not 6 months of age.
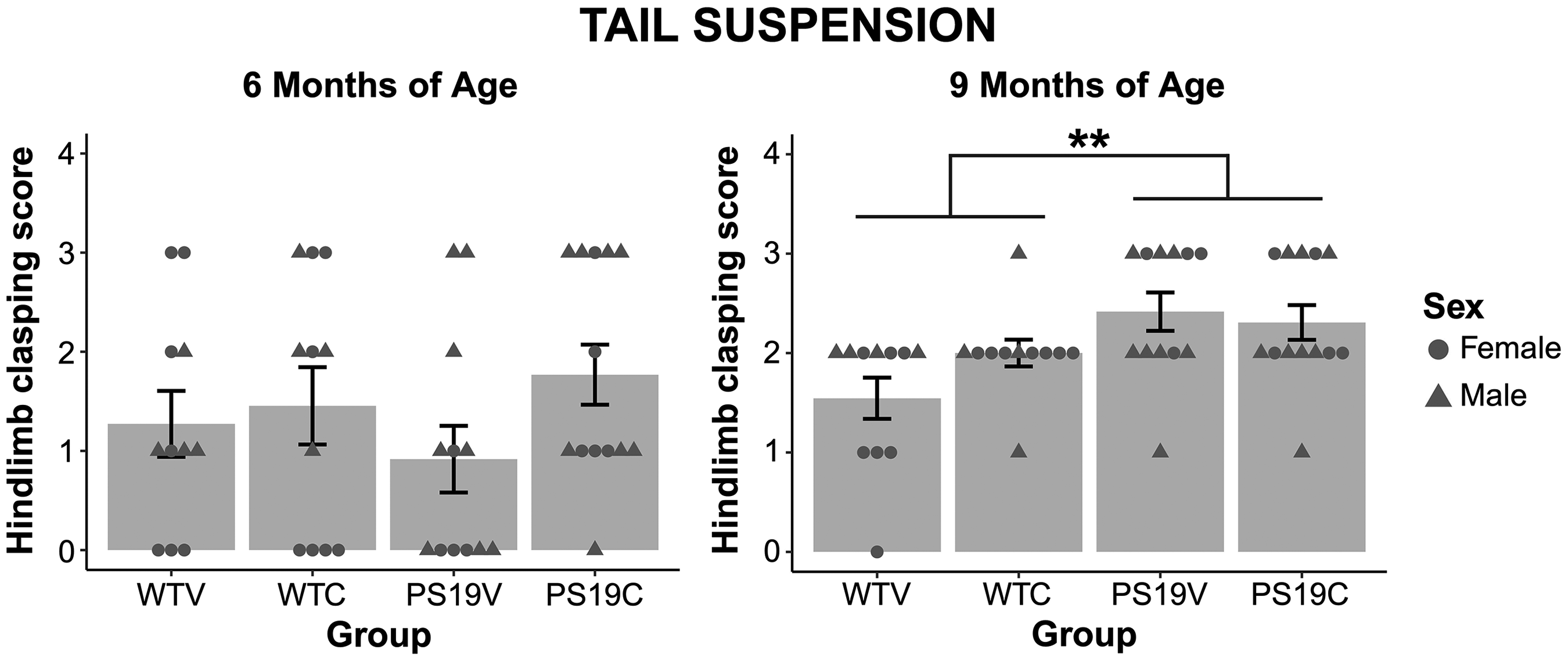
The hindlimb clasping score of the WT mice and the PS19 mice treated with vehicle (V) or CBD (C). (left) At 6 months of age the hindlimb clasping of the mice was similar regardless of genotype and treatment. (right) The hindlimb clasping score of the PS19 mice was higher relative to the WT mice, suggestive of hindlimb weakness at 9 months old. There was a significant effect of genotype (**p < 0.002). CBD did not impact the clasping of the mice. An age analysis revealed an age x genotype x treatment interaction with the WTV and PS19V mice significantly different at 9 months old (p = 0.002) but not at 6 months old (p = 0.47) while the WTC and PS19C mice were not different at 6 or 9 months old. This suggests that as PS19 mice age, CBD treatment reduced the progression of hindlimb weakness compared to vehicle treatment.
An age analysis performed on the hind limb weakness confirmed that there was a significant difference between 6 and 9 months of age (F (1, 43) = 20.06, p = 0.001), and a trend towards an age x genotype interaction (F (1, 43) = 3.66, p = 0.062) and an age x genotype x treatment interaction (F (1, 43) = 3.745, p = 0.06). There were no effects of genotype (F (1, 43) = 1.66, p = 0.20), treatment (F (1, 43) = 2.44, p = 0.13) nor genotype x treatment interaction (F (1, 43) = 0.15, p = 0.90). Pairwise comparisons with Bonferroni correction performed to explore the age x genotype interaction revealed that the WT and PS19 mice performed similarly at 6 months old (p = 0.95), but at 9 months old the PS19 mice exhibited greater hindlimb weakness compared to the WT mice (p < 0.002). Further, the results showed that the WT mice were not significantly different in their hindlimb clasping at 6 and 9 months of age (p = 0.086), but the PS19 mice were (p < 0.001). Post hoc comparisons assessing the age x genotype x treatment interaction revealed that the WTV and PS19V mice were significantly different at 9 months old (p = 0.002) but not at 6 months old (p = 0.47) while the WTC and PS19C mice were not different at 6 (p = 0.51) nor 9 months old (p = 0.23). This suggested that as PS19 mice age, CBD treatment reduced the progression of hindlimb weakness compared to vehicle treatment.
Morris water task. At 9 months of age one PS19V mouse and one PS19C mouse were excluded from the MWT data due to an inability to swim. Thus, the group sizes at this time point were: WTV (n = 11), WTC (n = 11), PS19V (n = 11), PS19C (n = 12).
Spatial training: At 6 months of age all the mice were able to learn the location of the hidden platform, evidenced by a decreased path length across training days, although the performance of the WT mice was better than the PS19 mice. There did not appear to be a consistent effect of CBD on the spatial learning of the mice. The path length of the mice to reach the hidden platform during spatial training was plotted from day 1 to day 8 as seen in Figure 4 (A). A three-way repeated measures ANOVA analyzed the path length of the mice and indicated a significant effect of genotype (F (1, 43) = 13.804, p < 0.001), but no effects of treatment (F (1, 43) = 0.91, p = 0.35), nor genotype x treatment interaction (F (1, 43) = 0.095, p = 0.76). Within subject tests indicated a significant decrease across days (F (7, 301) = 58.07, p < 0.001), but no interaction effects of day x genotype (F (7, 301) = 0.57, p = 0.78), day x treatment (F (7, 301) = 0.79, p = 0.59), nor day x genotype x treatment (F (7, 301) = 0.72, p = 0.66).
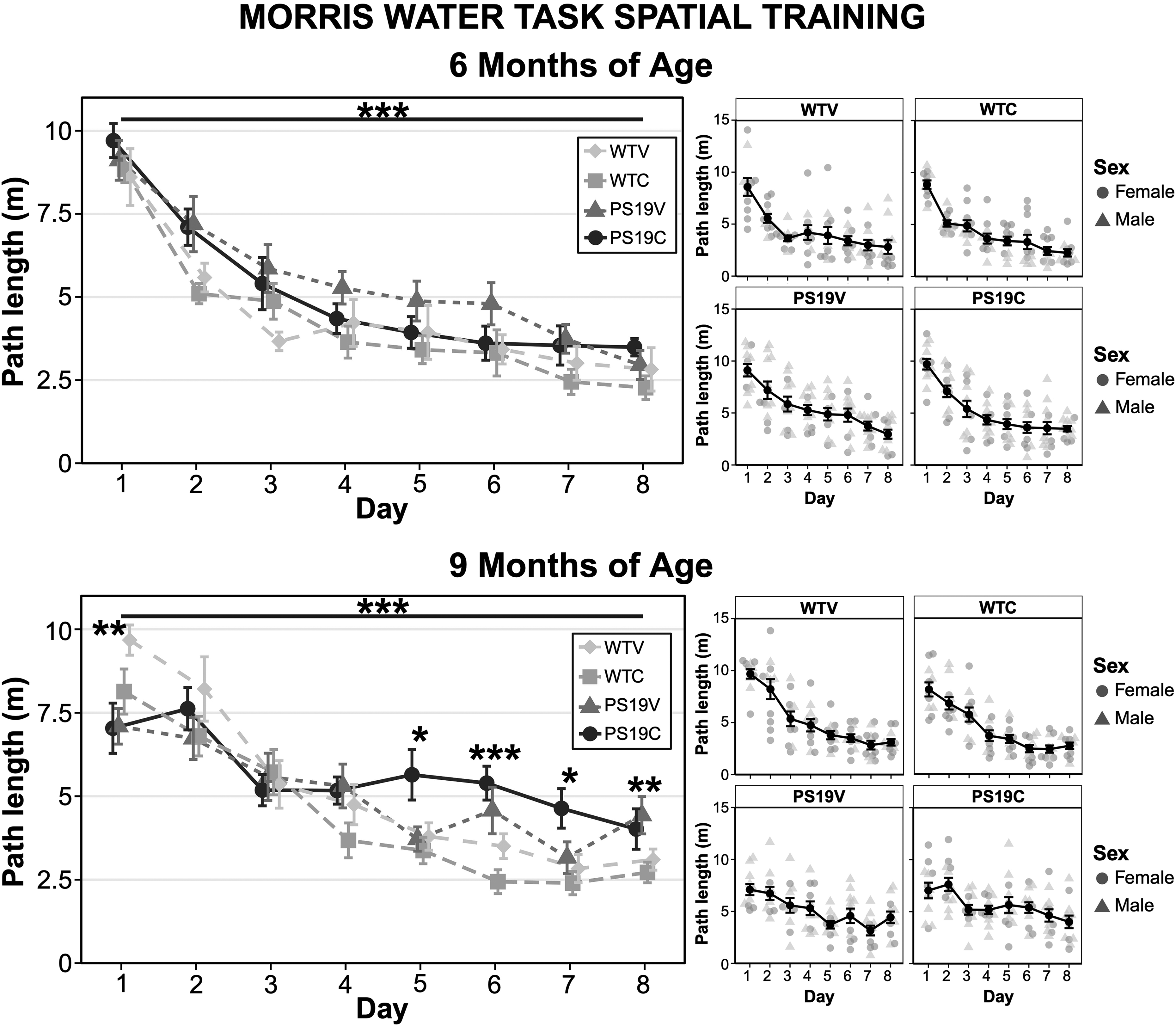
The path length of the PS19 mice and the WT mice treated with vehicle (V) or CBD (C) during the Morris water task (MWT) spatial training. (top left) The path length of the mice from training day 1 to 8 at 6 months old. (top right panels) The training days with male and female data points illustrated. All mice were able to learn the location of the platform over the training days (***p < 0.001). There was no significant effect of genotype nor treatment on the path length of the mice. (bottom left) The path length of the mice at 9 months old. (Bottom right panels) The training days with male and female data points illustrated. Although all mice were able to learn the location of the hidden platform (***p < 0.001), the PS19 mice were impaired compared to the WT mice (p < 0.028). Pairwise comparisons with Bonferroni corrections revealed that the WT and PS19 groups differed significantly on day 1 (**p < 0.004), day 5 (*p < 0.034), day 6 (***p < 0.000), day 7 (*p < 0.011), and day 8 (**p < 0.006). CBD did not restore the spatial learning of the PS19 mice but did improve performance in the WTC group as compared to the WTV group (p < 0.037).
As seen in Figure 4 (B), at 9 months of age, the path length of the PS19 mice to reach the hidden platform was increased relative to the WT mice, suggesting impaired spatial learning. CBD did not appear to impact the spatial learning of the mice. A repeated measures three-way ANOVA indicated significant within subjects effects of day (F (7, 287) = 40.473, p < 0.001), and a day x genotype interaction (F (7, 287) = 5.055, p < 0.001). No significant effects of day x treatment (F (7, 287) = 0.93, p = 0.49), nor day x genotype x treatment were observed (F (7, 287) = 1.1, p = 0.39). Between subjects, there was no treatment effect (F (1, 41) = 0.23, p = 0.63), but significant effects of genotype (F (1, 41) = 5.207, p = 0.028), and a genotype x treatment interaction (F (1, 41) = 6.789, p = 0.013) were obtained. To further assess the learning difference, pairwise comparisons with Bonferroni corrections on the genotype effect indicated that the WT outperformed the PS19 mice on day 5 (p < 0.034), day 6 (p < 0.000), day 7 (p < 0.011), and day 8 (p < 0.006). Conversely, the WT mice performed worse than the PS19 mice on day 1 (p < 0.004). Pairwise comparisons performed to elucidate the genotype x treatment interaction revealed that CBD treatment improved performance in the WT but not PS19 mice (p < 0.001). Furthermore, WT mice treated with CBD performed better than WT mice treated with vehicle (p < 0.037), while there was no difference in path length in the PS19 mice treated with CBD or Vehicle (p = 0.14). An analysis of swim speed was also conducted to rule out learning deficits being due to decreased motor ability at 9 months old and indicated that all mice increased swim speed across days (F (4.017, 164.686) = 9.528, p < 0.0013), regardless of genotype (F (1, 41) = 1.07, p = 0.31) or treatment (F (1, 41) = 0.55, p = 0.46).
Ultimately, at 9 but not 6 months of age the spatial learning of the PS19 mice was impaired relative to the WT mice. These results suggest that the function of networks centered around the HPC were beginning to decline in this group of mice. CBD did not impair nor restore the spatial learning of the PS19 mice, however it did improve spatial learning of the WT mice.
Probe: Figure 5 (A) demonstrates the time spent in the target quadrant relative to the time spent in the alternate quadrants for all groups at 6 months of age. All the mice spent more time in the target quadrant relative to the average of the alternate quadrants and there were no observable differences in the mice regardless of genotype or treatment. A three-way ANOVA reported a significant effect of quadrant (F (1, 43) = 121.304, p < 0.001), but no effects of genotype (F (1, 43) = 2.43, p = 0.13) nor treatment (F (1, 43) = 0.10, p = 0.75). Despite the quadrant preference results, the annulus crossing of the PS19 mice were reduced relative to the WT mice at 6 months of age (see Figure 5 (B)). Therefore, the spatial specificity of the PS19 mice may be beginning to decline. CBD does not appear to impact the spatial specificity of the mice. A two-way ANOVA confirmed a significant effect of genotype (F (1, 43) = 4.542, p < 0.039), but no effect of treatment (F (1, 43) = 0.60, p = 0.44), nor interaction (F (1, 43) = 1.09, p = 0.30) on annulus crossings.
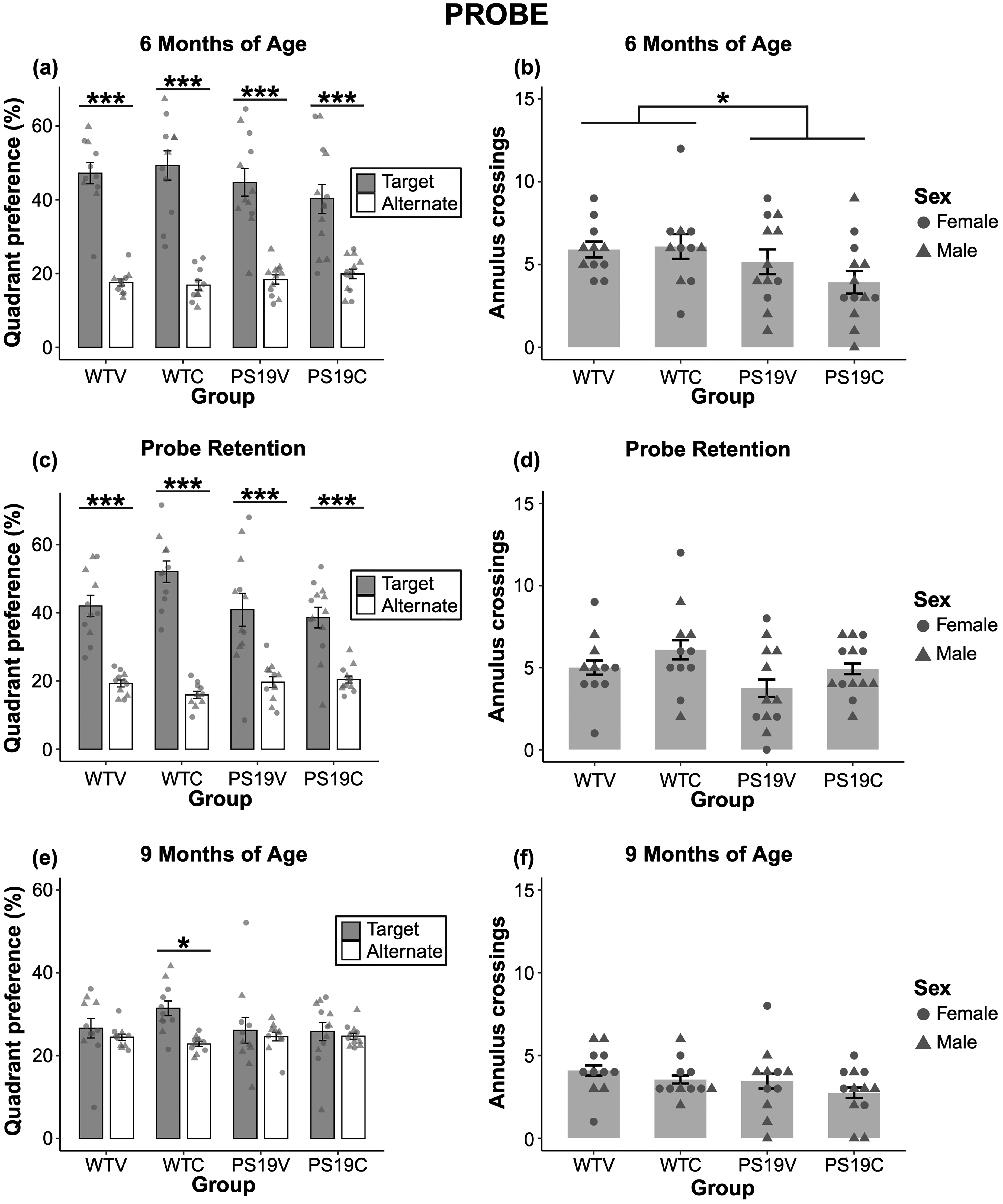
The probe data of the WT mice and PS19 mice treated with vehicle (V) or CBD (C). (A) The quadrant preference of the mice at 6 months old during the probe trial performed 24 h after training. All mice spent a greater percentage of time in the target relative to average of alternate quadrants (***p < 0.001). There were no significant effects of genotype nor treatment on quadrant preference. (B) The number of annulus crossings for the PS19 mice were lower relative to the WT mice (*p < 0.039). CBD did not restore the spatial recall of the mice. (C) The data collected during the probe retention performed 1 week after training at 6 months old. All mice recalled the previous platform location (***p < 0.001). There was no significant effect of treatment. (D) The number of annulus crossings during the probe retention. Overall, the performance of the mice was similar. (E) The quadrant preference during the probe trial when the mice were 9 months old. Only the WTC group demonstrated a significant target quadrant preference (*p = 0.012). There were no effects of genotype nor treatment on quadrant preference. (F) The number of annulus crossings during the probe trial. There were no significant effects of genotype or treatment on this measure.
Retention test: Figure 5 (C) depicts the quadrant preference of all groups during the retention probe, performed one week after training. The WTC mice demonstrate the strongest preference for the target quadrant. The performance of the other groups was similar. A three-way ANOVA within subjects effects revealed a significant effect of quadrant (F (1, 43) = 102.818, p < 0.001) and a marginally significant effect of quadrant x genotype (F (1, 43) = 4.014, p = 0.051). There were no interaction effects of quadrant x treatment (F (1, 43) = 1.13, p = 0.29) nor quadrant x genotype x treatment (F (1, 43) = 2.88, p = 0.10). To assess the quadrant x genotype effect, pairwise comparisons with Bonferroni corrections were completed that revealed marginally significant differences between the WT and PS19 groups as the WT mice spent more time in the target quadrant (p = 0.051) and less time in the alternate quadrant (p = 0.052) compared to the PS19 mice. Between subjects tests revealed a marginally significant effect of genotype (F (1, 43) = 4.02, p = 0.051), but no treatment (F (1, 43) = 1.12, p = 0.30) nor genotype x treatment interaction (F (1, 43) = 2.87, p = 0.097). Due to an a-priori assumption that the CBD treatment would improve retention based on the WTC mice having improved learning, post hoc comparisons with Bonferroni corrections were performed that confirmed the WTC mice exhibited stronger retention of the escape location than PS19C mice (p < 0.011), as well as a trend towards significance in comparison to WTV (p = 0.066). As seen in Figure 5 (D) the PS19V mice have the lowest number of annulus crossings during the retention probe. The performance of the PS19C and the WTV was similar, while the WTC have the highest number of annulus crossings. Despite these impressions, a two-way ANOVA revealed no significant effects of genotype (F (1, 43) = 3.34, p = 0.075), treatment (F (1, 43) = 2.93, p = 0.094), nor interaction (F (1, 43) = 0.004, p = 0.95) on the annulus crossings during the retention probe.
At 9 months old, the quadrant preference of the mice depending on genotype and treatment during the probe trial can be seen in Figure 5 (E). It appears that only the WTC mice were able to recall the previous platform location. The performance of the WTV, PS19C, and PS19V groups was similar. A two-way ANOVA suggests that despite the slight quadrant preference observed in the WTV, PS19C, and PS19V groups, there was a significant effect of quadrant (F (1, 41) = 4.348, p < 0.043). There were no interaction effects of quadrant x genotype (F (1, 41) = 1.16, p = 0.21), quadrant x treatment (F (1, 41) = 0.88, p = 0.35), nor quadrant x genotype x treatment (F (1, 41) = 1.10, p = 0.30). Post hoc pairwise comparisons with Bonferroni corrections were performed to further elaborate the quadrant effect and revealed that only the WTC mice spent significantly more percent time in the target versus average of alternate quadrants (p = 0.012), suggesting that CBD improved memory in this group at 9 months old. As seen in Figure 5 (F), there does not appear to be any observable differences in the number of annulus crossings between the groups. A two-way between subjects ANOVA confirmed no significant effects of genotype (F (1, 41) = 2.26, p = 0.14) nor treatment (F (1, 41) = 1.72, p = 0.20) on the annulus crossings of the mice.
Overall, the probe data suggests that the spatial recall of the PS19 mice began to decline at 6 months of age, as evidenced by a significant effect of genotype on path length swam during training and the number of annulus crossings during the probe. There was a marginally significant effect of genotype on the quadrant preference during the retention test, further supporting that the spatial recall of the PS19 mice was beginning to decline. At 9 months old, there were no significant effects of genotype nor treatment. The quadrant preference of each group of mice was weak, except for the WT mice treated with CBD which showed a significant preference for the target quadrant.
Visible platform: Figure 6 (left) demonstrates the path length of the mice during spatial training day 1, when the platform was hidden, relative to their path length during the visible platform test, when the platform was visible, at 6 months of age. The PS19 and WT groups had a shorter path length when the platform was visible relative to when it was hidden. CBD did not appear to impact the performance of the mice. A three-way ANOVA confirmed a significant decrease in path length during the visible platform test compared to spatial training day 1 (F (1, 43) = 743.317, p < 0.001). There were no effects of genotype (F (1, 43) = 1.41, p = 0.24) nor treatment (F (1, 43) = 0.66, p = 0.42) on the performance of the mice.
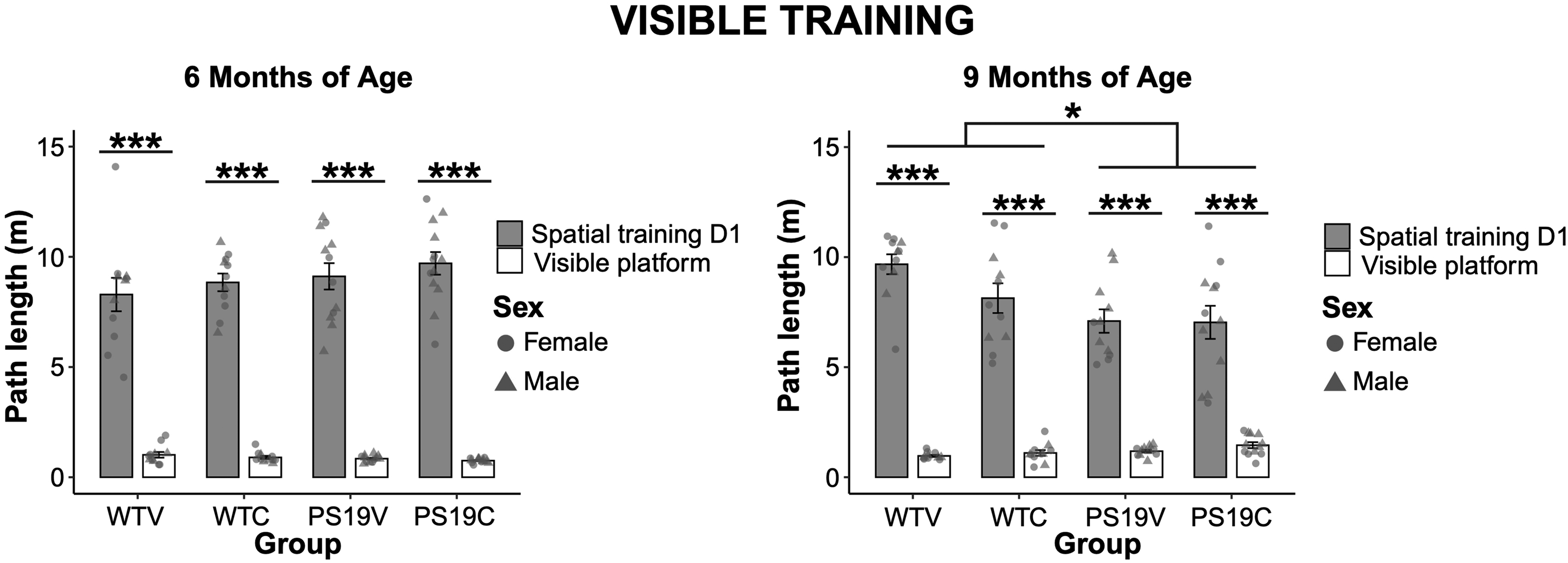
A comparison of the path length of the WT mice and PS19 mice treated with vehicle (V) or CBD (C) during spatial training day 1 relative to the visible platform test. (left) At 6 months old, all the mice demonstrated a decrease in path length when the platform was visible (***p < 0.001). (right) At 9 months old, although all the mice demonstrated a decreased path length during the visible platform test (***p < 0.001), there was a significant effect of genotype (*p < 0.012). There was no effect of treatment on the performance of the mice.
As seen in Figure 6 (right), at 9 months old, all the mice showed a drastic decrease in path length when the platform was visible relative to when it was hidden on day 1 of spatial training. It did not appear that genotype nor treatment were impacting the sensory, motor, or motivational behavior of the mice at this age. A three-way ANOVA indicated a significant decrease in path length during the visible platform test relative to training day 1 for all groups (F (1, 41) = 414.799, p < 0.001), a genotype effect (F (1, 41) = 6.903, p = 0.012), and a significant path x genotype interaction (F (1, 41) = 10.1, p < 0.003). Pairwise comparisons with Bonferroni corrections further analyzed the path x genotype interaction and revealed that the PS19 mice performed significantly better than the WT mice on spatial training day 1 (p < 0.005), but the WT performed better than the PS19 mice on the visible platform test (p < 0.01). There was no significant effect of treatment on the performance of the mice (F (1, 41) = 1.02, p = 0.32). To further elucidate the genotype difference, a repeated three-way ANOVA was performed to analyze the visible platform test across the four swims that the mice received (Figure 7). Mauchly's test of sphericity was violated (ε = 0.753) thus Greenhouse-Geisser degrees of freedom were used. The results indicated significant effects of visible swim trial (F (2.259, 92.627) = 13.716, p < 0.001) and genotype (F (1, 41) = 7.334, p = 0.01), and a trend towards treatment effect (F (1, 41) = 3.738, p = 0.06). No interaction effects were found. Post-hoc comparisons with Bonferroni corrections revealed that the WT and PS19 mice were different on the first swim trial (p = 0.002), but not for the remaining trials. A two-way univariate ANOVA on swim speed to the visible platform indicated no significant effects of genotype (F (1, 41) = 1.96, p = 0.17), treatment (F (1, 41) = 0.38, p = 0.85), nor their interactions.
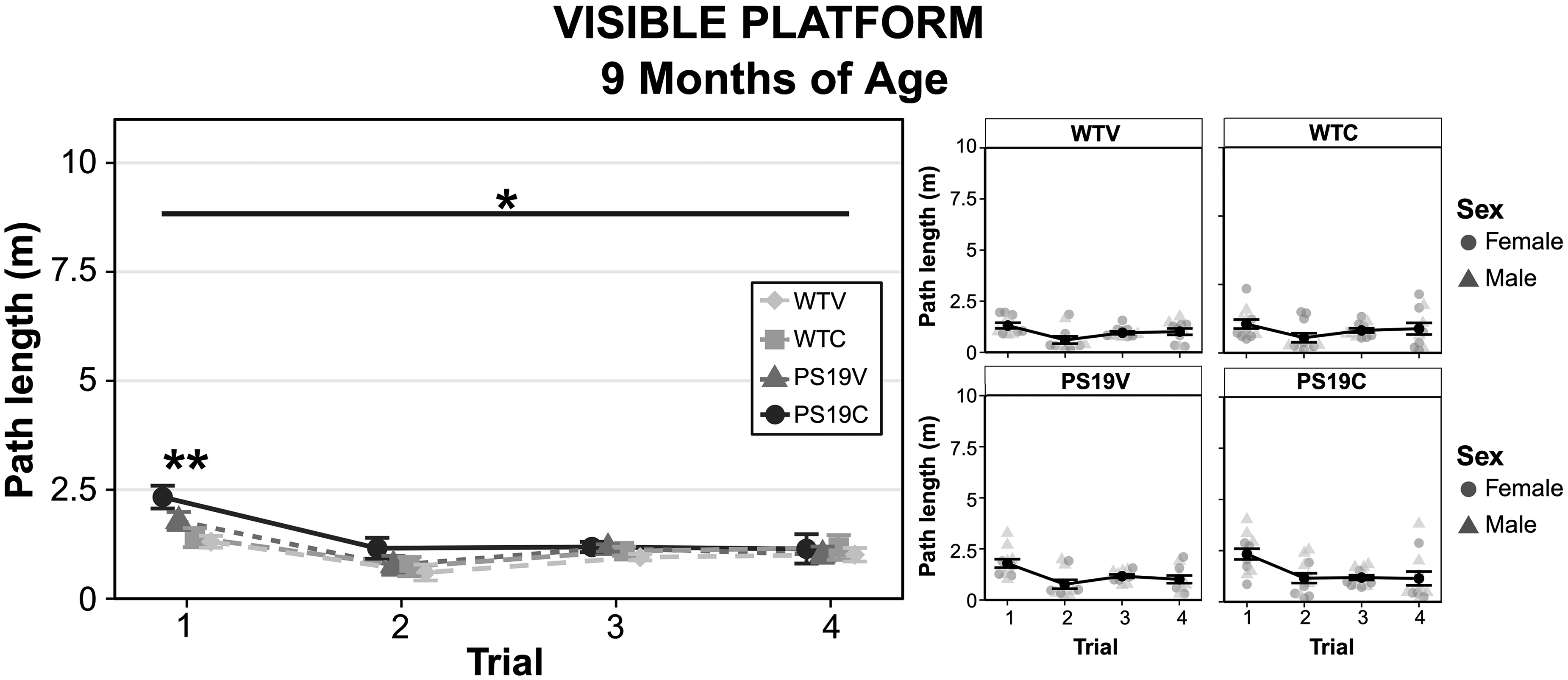
A comparison of the path length of the WT mice and PS19 mice treated with vehicle (V) or CBD (C) during the four swims of the visible platform test (left). (right) The swim trials with male and female data points illustrated. Both the WT and PS19 mice had significantly longer path length to reach the visible platform on the first swim versus remaining swims (p < 0.005). Post hoc comparisons with Bonferroni corrections revealed that the trial x genotype interaction was due to the WT mice performing better than the PS19 mice on the first trial (**p = 0.002). The groups were not different in trials 2–4.
Overall, the visible platform data suggests that the general motor, motivational, and sensory behavior was not impaired. It is important to note that while the performance of the PS19 was statistically subpar relative to controls, this difference was only evident on swim trial 1, as performance on trials 2–4 were indistinguishable from the WT mice. CBD did not impact the sensory, motor, or motivational behavior of the mice.
Fear conditioning (FC). One PS19V mouse was humanely euthanized and perfused prior to FC due to the onset of paralysis, characterized by a hunched back posture comorbid with weight loss. See Supplemental Figure 1 for a demonstration of the posture of the PS19 mice as they descend into the final stages of the disease. The final group sizes during FC were: WTV (n = 11), WTC (n = 11), PS19V (n = 11), PS19C (n = 13).
As seen in Figure 8 (left), the PS19 mice demonstrate increased freezing in the novel context, whereas the WT mice spend minimal time freezing. Furthermore, the freezing response of the PS19 mice in the presence of the conditioning context was slightly lower than the WT mice. There were no observable differences between the mice treated with CBD or vehicle. A three-way within subjects ANOVA assessed the percent time spent freezing during the novel context compared to during context recall and reported no significant effect of genotype (F (1, 42) = 0.05, p = 0.83) as both groups showed a significant increase in the freezing response during context recall compared to the novel context (F (1, 42) = 19.747, p < 0.001). However, a significant interaction effect of context x genotype was observed (F (1, 42) = 10.916, p < 0.002). Pairwise comparisons with Bonferroni corrections revealed that the significant interaction effect was applicable to a difference in freezing of the groups in the novel context (p < 0.012), but not to the recall context (p = 0.11). Furthermore, the WT mice showed increased freezing in the recall versus novel context (p < 0.001), whereas the PS19 mice did not (p = 0.42). There was no significant effect of treatment on the freezing of the mice (F (1, 42) = 0.05, p = 0.83).
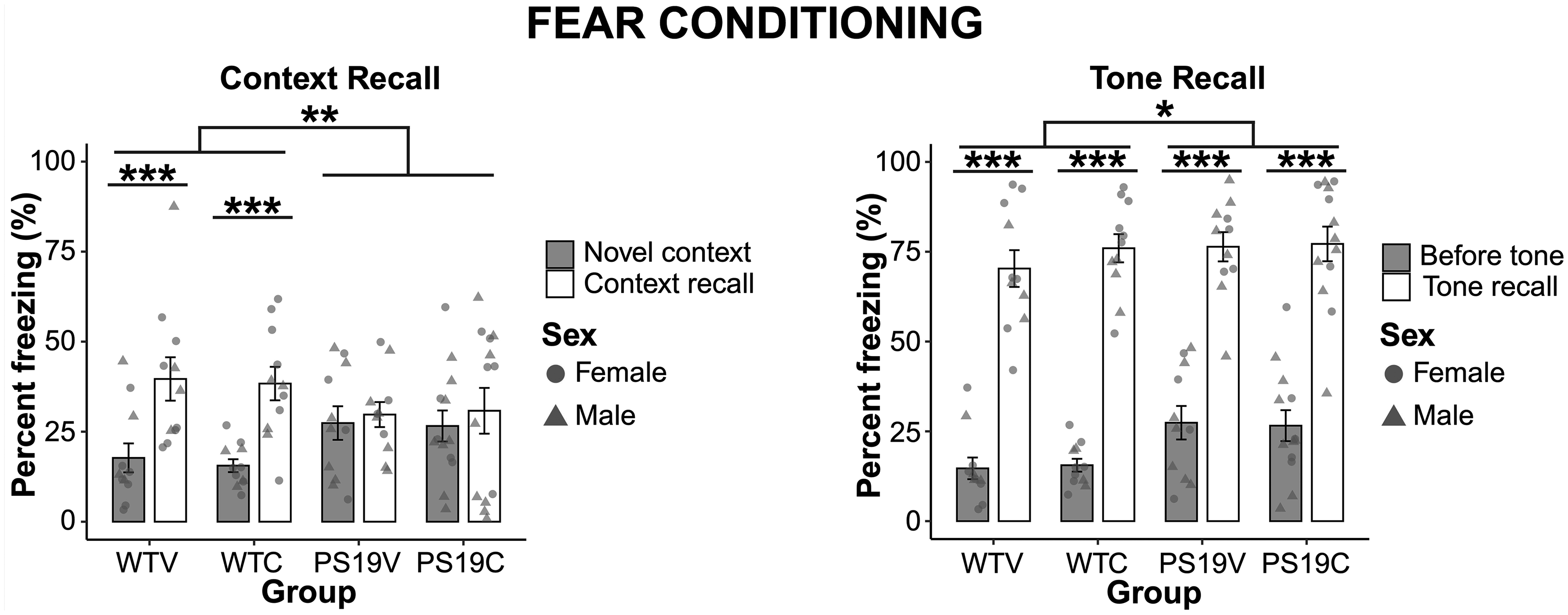
The fear conditioning data of the WT mice and the PS19 mice treated with vehicle (V) or CBD (C). (left) A comparison of the freezing response of the mice during the novel context relative to context recall. Overall, all groups displayed increased freezing behavior during context recall compared to the novel context (p < 0.001). There was a significant effect of genotype x context on the freezing response of the mice (**p < 0.002). Pairwise comparison with Bonferroni corrections showed that the freezing response of the WT and PS19 mice only differed significantly in the novel context (p < 0.012) but not during context recall. Further, the WT mice had increased freezing response during context recall compared to the novel context (***p < 0.001), while the PS19 mice did not show discriminative freezing between the contexts. CBD does not appear to impact the context recall of the mice. (right) All groups demonstrated a higher percentage of freezing during the tone compared to before the tone (***p < 0.001). There was also a significant effect of context x genotype (*p = 0.021) as the PS19 mice exhibited more freezing behavior before the tone than the WT mice. There was no effect of treatment on the freezing response.
Figure 8 (right) depicts the percentage of freezing exhibited by the mice before and during the tone presentation conditions. As can be seen in this figure, each group of mice were able to recall the tone/shock pairing as evidenced by an increased freezing response during the tone relative to before the tone. A three-way repeated measures ANOVA confirmed that although all mice displayed more freezing during the tone as compared to before the tone (F (1, 42) = 427.536, p < 0.001), a significant effect of genotype (F (1, 42) = 5.695, p = 0.022) was reported. Post hoc comparisons with Bonferroni corrections showed that the genotype difference stemmed from the PS19 mice freezing more than the WT mice before the tone (p = 0.003) but not during the tone presentation (p = 0.43). There was no effect of treatment (F (1, 42) = 0.26, p = 0.61). A time bin analysis of the 2-min period before the tone presentation was also performed to further assess the freezing behavior and revealed that while all mice displayed more freezing in the second 60 s compared to the first 60 s, the PS19 mice froze more than the WT mice. A repeated measures ANOVA confirmed these observations indicating significant effects of genotype (F(1,42) = 10.257, p = 0.003) and freezing bin (F (1,42) = 25.799, p < 0.001), but no effects of treatment (F(1,42) = 0.001, p = 0.98).
These results are suggestive of retained fear associative memory, an assay of learning and memory supported by neural networks centered on the amygdala. The PS19 mice exhibited more freezing behavior in the novel context and prior to the tone presentation relative to the WT mice. The potential cause for these results will be explored in the discussion.
Brain assessments
The group sizes for the histological analyses were: WTV male (n = 3), WTV female (n = 3), WTC male (n = 3), WTC female (n = 3), PS19V male (n = 4), PS19V female (n = 3), PS19C male (n = 5), PS19C female (n = 3).
Hippocampal volume. The brains of the perfused mice were stained with Nissl and HPC volumes were quantified. As seen in Figure 9 (left), overall, the WT mice have larger HPC volume relative to the PS19 mice. The HPC volume of the male WT mice was slightly larger than the female WT mice. In contrast to this, the HPC volume of the female PS19 mice was much larger compared to the male PS19 mice. CBD does not appear to impact the HPC volume of the animals. A three-way between subjects ANOVA was conducted to determine any effects of genotype, treatment, and sex on the HPC volume of the animals. The results showed significant effects of genotype (F (1, 19) = 40.419, p < 0.001) and a genotype x sex interaction (F (1, 19) = 8.837, p = 0.008). No effects of treatment (F (1, 19) = 1.12, p = 0.30) nor sex (F (1, 19) = 0.89, p = 0.36) were observed. A pairwise comparison with Bonferroni corrections analyzed the significant genotype x sex interaction and showed the effect was applicable to the PS19 (p = 0.009, ηp2 = 0.307), but not the WT mice (p = 0.19). The size difference of the HPC depending on genotype and sex can be clearly observed by looking directly at the stained brains in Figure 9 (right).
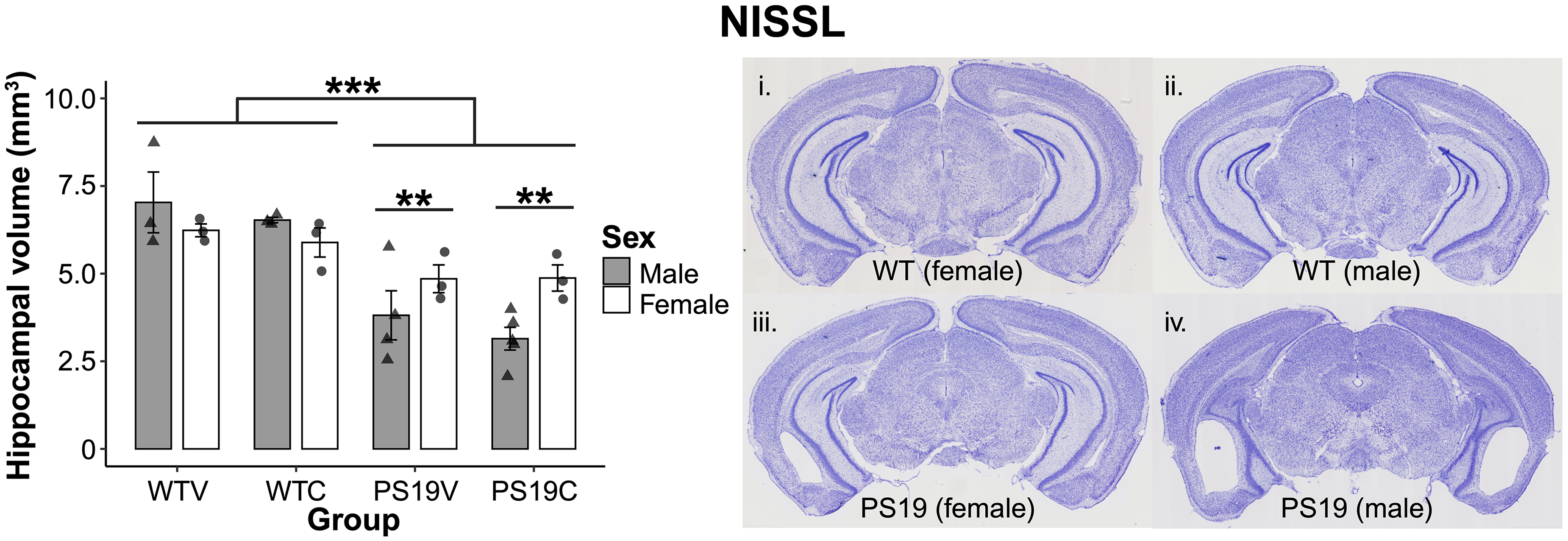
The hippocampal (HPC) volume of the PS19 mice and WT mice treated with vehicle (V) or CBD (C). (left) Overall, the HPC volume of the WT mice was larger than the PS19 mice (***p < 0.001). There was a significant interaction of genotype x sex (p < 0.01). A pairwise comparison with Bonferroni corrections revealed that the HPC volume of the female and male mice only differed among the PS19 group (**p = 0.009). CBD did not appear to have an effect on the HPC volume of the mice. (right) The Nissl-stained brains of the mice depending on genotype and sex. Image (i) demonstrates a WT female mouse, (ii) demonstrates a WT male mouse, (iii) demonstrates a PS19 female mouse, and (iv) demonstrates a PS19 male mouse. All the brains shown were mice treated with CBD.
Inflammation quantification via ImageJ and Ilastik
Microglia: A representation of microglia staining in the HPC of the mice depending on genotype and sex can be seen in Figure 10 (A). There was a visibly larger quantity of Iba1 in the HPC of the PS19 mice relative to the WT mice. The percentage of Iba1 in the HPC of male and female PS19 and WT mice treated with CBD or vehicle can be seen in Figure 10 (B). The PS19V mice had the highest percentage of Iba1 in the HPC, followed by the PS19C mice. The percentage of Iba1 was similar in the male and female WT mice treated with vehicle or CBD, with the CBD treated mice having a slightly reduced quantity. A three-way between subjects ANOVA confirmed the observed results. There was a significant effect of genotype (F (1, 19) = 76.741, p < 0.001), but no significant effects of treatment (F (1, 19) = 3.0, p = 0.1) nor sex (F (1, 19) = 1.46, p = 0.24). A priori assumptions were made that CBD would reduce the quantity of Iba1 in the HPC of the PS19 mice. A pairwise comparison with Bonferroni corrections revealed a marginal trend towards the PS19C mice having a reduced quantity of Iba1 compared to the PS19V mice (p < 0.070, ηp2 = 0.162). There was no significant effect of treatment for the WT mice (p = 0.56). Overall, the PS19 mice had more Iba1 stained cells in their HPC relative to the WT mice. There was a marginal trend towards CBD decreasing the quantity of Iba1 in the HPC of PS19 mice. Sex did not statistically impact the quantity of Iba1 in the HPC of the mice.
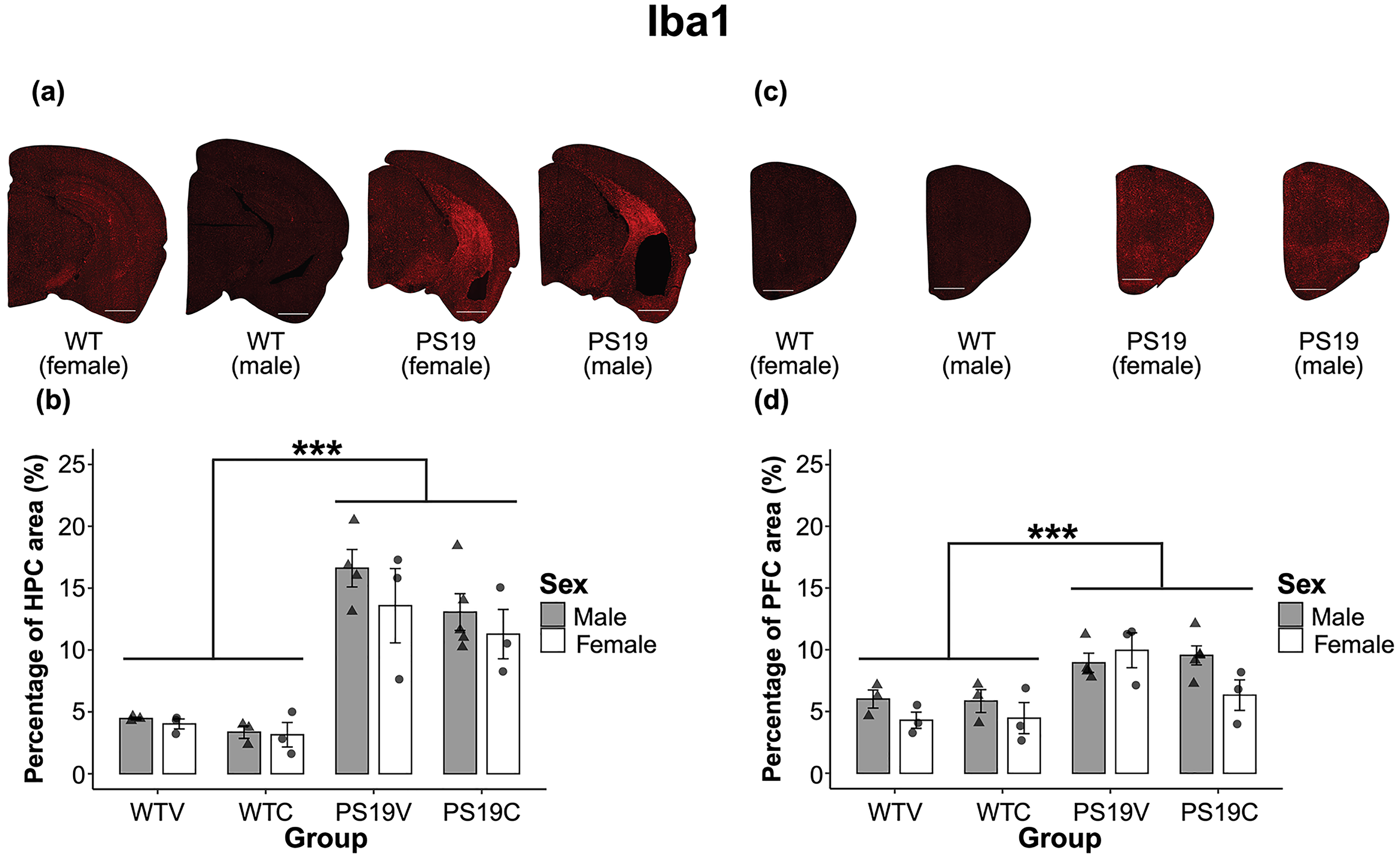
A representation of the distribution of microglia, stained with Iba1, in the brains of the mice depending on genotype and sex. Brain images were taken with a Nanozoomer slide scanner and the scale bar represents 1 mm. (A) Demonstrates the microglia staining in the hippocampus (HPC) of mice treated with CBD. (B) The percentage of Iba1 in the HPC of male and female PS19 mice and WT mice treated with vehicle (V) or CBD (C). The PS19 mice had a higher percentage of Iba1 in the HPC relative to the WT mice (***p < 0.001). The PS19C mice appeared to have a slightly reduced quantity of Iba1 in the HPC than the PS19V mice, however, there were no significant effects of treatment nor sex. (C) Represents the microglia staining in the PFC of mice treated with CBD. (D) The PS19 mice had a higher percentage of Iba1 in the PFC compared to the WT mice (***p < 0.001). There was no significant effect of treatment. There was a trend towards a significant effect of sex (p < 0.074).
The difference in the quantity of Iba1 in the PFC, depending on genotype, was less pronounced when observing the stained images (see Figure 10 (C)). Figure 10 (D) represents the percentage of Iba1 in the PFC of the male and female mice depending on their genotype and treatment group. The male PS19C mice and the female PS19V mice had the highest percentage of Iba1 in the PFC, followed by the male PS19V mice and the female PS19C mice. Among the WT mice, the males consistently have more Iba1 relative to the females. Overall, the PS19 mice have more Iba1 in the PFC relative to the WT mice. A three-way between subjects ANOVA confirmed a significant effect of genotype (F (1, 19) = 25.49, p < 0.001). There was no effect of treatment (F (1, 19) = 1.16, p = 0.30), but there was a marginal trend towards an effect of sex (F (1, 19) = 3.577, p = 0.074). A post hoc comparison performed to elucidate the genotype x treatment x sex effect revealed that CBD treatment significantly reduced Iba1 in the female PS19 mice compared to the male PS19 mice (p = 0.024, ηp2 = 0.241). CBD did not have the same effect in the WT mice. Therefore, Iba1 was increased in the PFC of the PS19 mice relative to the WT mice. CBD was only effective at reducing the quantity of Iba1 in the female PS19 mice. Sex might be in part impacting the quantity of Iba1 in the PFC.
Astrocytes: When observing the staining of GFAP in the HPC of the mice, the male PS19 mice followed by the female PS19 mice have a visibly larger quantity of the inflammatory marker relative to the WT mice (see Figure 11 (A)). The quantity of GFAP in the PFC varied among male and female PS19 mice and there was little GFAP observed in the PFC of the WT mice (see Figure 11 (B)). No effect of treatment was observed when looking at the stained images. The quantity of GFAP in the HPC of the male and female mice depending on their genotype and treatment can be seen in Figure 11 (C). The PS19V mice had a higher percentage of GFAP in the HPC, relative to the PS19C mice. Overall, the PS19 males and WT males demonstrated a higher percentage of GFAP relative to the female mice, however the sex difference between the WT mice was slight. A three-way between subjects ANOVA confirmed the observed results indicating a significant effect of genotype (F (1, 19) = 27.729, p < 0.001), and sex (F (1, 19) = 4.747, p = 0.042, ηp2 = 0.20), but no effect of treatment (F (1, 19) = 0.10, p = 0.75). Pairwise comparisons with Bonferroni corrections revealed that there was a significant effect of sex among the PS19 mice (p = 0.018, ηp2 = 0.26) but not between the WT mice (p = 0.56). The results suggest that the PS19 mice had a higher percentage of GFAP in the HPC than the WT mice. Additionally, the male PS19 mice had a higher percentage of GFAP relative to the female PS19 mice. CBD was unable to delay the accumulation of GFAP in the HPC of the mice.
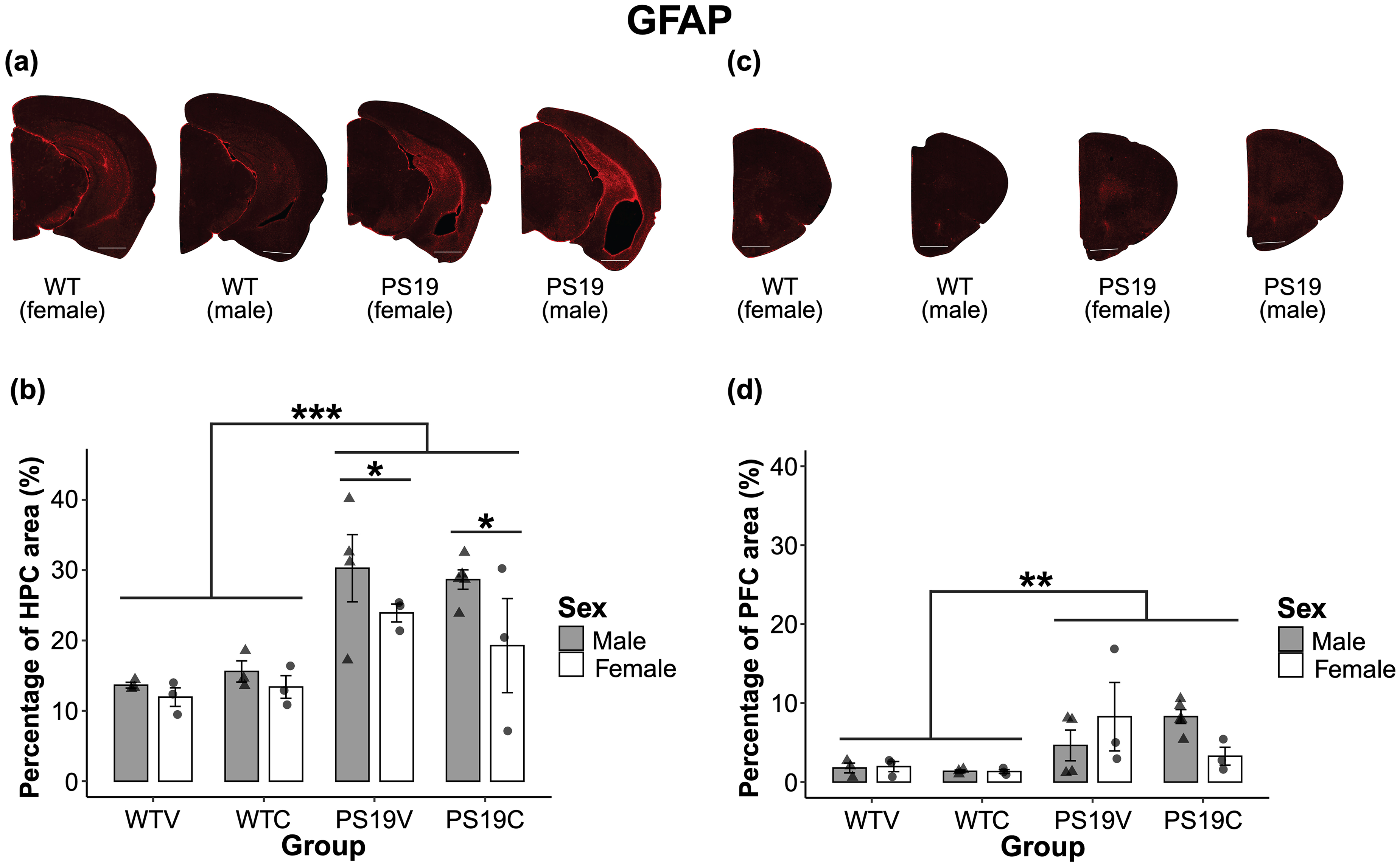
A representation of the distribution of astrocytes, stained with GFAP, in the brains of the mice depending on genotype and sex. Brain images were taken with a Nanozoomer slide scanner and the scale bar represents 1 mm. (A) Demonstrates astrocytes staining in the hippocampus (HPC) of mice treated with CBD. (B) The percentage of GFAP in the HPC of male and female PS19 mice and WT mice treated with vehicle (V) or CBD (C). The PS19 mice have a higher percentage of GFAP in the HPC relative to the WT mice (***p < 0.001). There were no clear trends towards CBD impacting the percentage of GFAP in the HPC. Overall, the male mice in each group appeared to have more GFAP in the HPC compared to the female mice (*p = 0.042). A pairwise comparison with Bonferroni corrections revealed that there was only a significant effect of sex in the PS19 mice (*p = 0.018). (C) Represents astrocytes staining in the prefrontal cortex (PFC) of mice treated with CBD. (D) The PS19 mice had a higher percentage of GFAP in the PFC compared to the WT mice (**p = 0.002). There was no significant effect of treatment nor sex on percent GFAP in the PFC.
The quantity of GFAP in the PFC of the mice depending on genotype, treatment, and sex can be seen in Figure 11 (D). Among the vehicle treated PS19 mice, the females demonstrated a higher percentage of GFAP relative to the males, however among the CBD treated mice the males demonstrated a higher percentage of GFAP relative to the females. Overall, the PS19 mice had a higher percentage of GFAP in the PFC relative to the WT mice. A three-way between subjects ANOVA assessed the observed results and revealed that there was a significant effect of genotype (F (1, 19) = 13.507, p = 0.002). There was no effect of treatment (F (1, 26) = 0.24, p = 0.63) nor sex (F (1, 19) = 0.06, p = 0.82). The results suggest that the PS19 mice have a higher quantity of GFAP positively stained cells in the PFC, suggestive of reactive astrocytes. CBD did not reduce the quantity of GFAP, and sex did not impact the quantity of GFAP in the PFC.
Inflammation assessment via neurolucida® 360
Due to the trend towards a treatment effect on the percentage of Iba1 in the HPC of the Tau P301S mice, Neurolucida® 360 was used to trace individual microglia. Previous research suggests that full photomicrographs, like those used while quantifying with Ilastik, are not sensitive enough to detect morphological alterations. 69 Rather single cell analyses are more reliable and can more accurately speak to morphological alterations, which are suggestive of reactivity.
Processes volume: Figure 12 (A) demonstrates the average volume of the processes of the traced microglia for the groups of mice. Overall, the dendritic volume of the PS19 mice appeared larger than the WT mice. Whether the mice were treated with CBD, or the vehicle did not impact the dendritic volume of the traced microglia. A mixed design repeated measures ANOVA confirmed the observed results. Mauchly's test of Sphericity was violated (ε = 0.715), thus Greenhouse-Geisser degrees of freedom were used. Within subjects there was no significant effect of dendritic volume for each microglia traced (F (2.144, 49.313) = 2.779, p < 0.068). Additionally, there was no significant effect of dendritic volume x treatment interaction (F (2.144, 49.313) = 1.72, p = 0.19), nor dendritic volume x genotype (F (2.144, 49.313) = 1.54, p = 0.22). However, while there was no significant effect of treatment (F (1, 23) = 0.82, p = 0.38), there was a significant effect of genotype (F (1, 23) = 7.963, p < 0.010). The results suggest that CBD was unable to reduce the volume of the processes for the PS19 mice. Overall, the volume of the processes was significantly larger among PS19 mice relative to that of the WT mice.
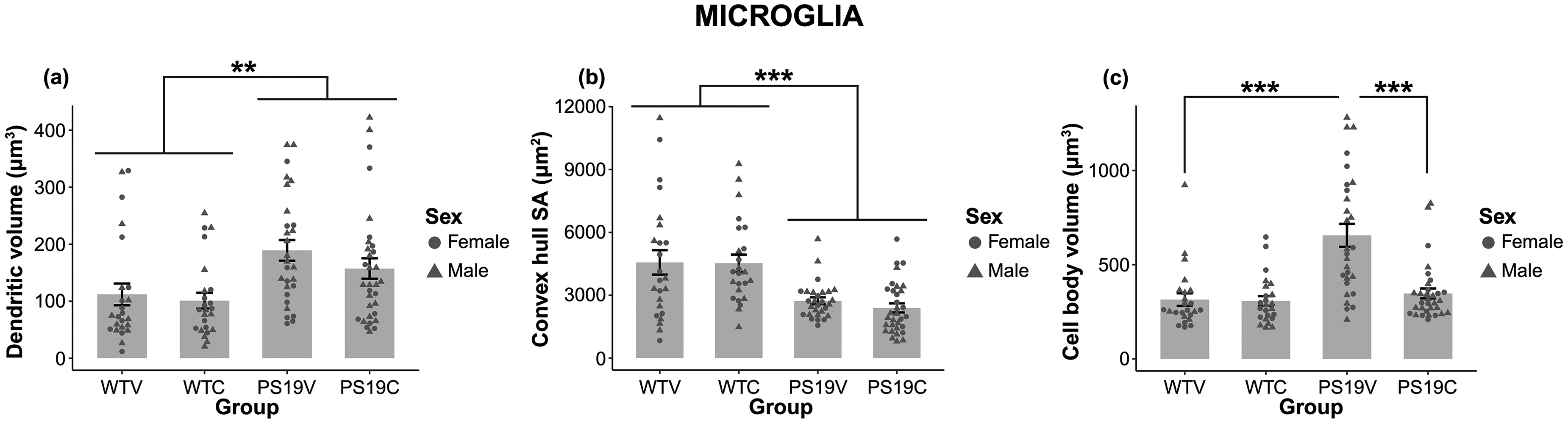
The collected data of the traced microglia in the CA1 region of the hippocampus (HPC). (A) The volume of the processes for all groups of mice. The PS19 mice appeared to have larger processes relative to the WT mice (**p < 0.01). CBC did not appear to affect the volume of the processes of the PS19 or WT mice. (B) The total convex hull surface area (SA) is suggestive of the area in space that the microglia consume. The total convex hull SA of the microglia in the PS19 mice was smaller relative to the WT mice (***p < 0.001), regardless of treatment group. (C) The total cell body volume of the traced microglia for all groups. There was a significant effect of genotype (**p < 0.004) with the PS19 mice having larger cell body than the WT mice. There was also a significant effect of treatment group (p < 0.004). The PS19V mice had the largest cell body volume, while the PS19C, WTV, and WTC groups were similar in size. Finally, there was a significant interaction of genotype x treatment group (p < 0.005). Bonferroni corrections revealed that total cell body volume was significant between the WT and PS19 mice when they received vehicle treatment (***p < 0.001), but not when they received CBD (p = 0.559). Furthermore, total cell body volume was significantly reduced in PS19C compared to PS19V mice (***p < 0.001). Overall, the results showed that CBD appeared to reduce the cell body volume of the PS19 mice to a size similar to that of the WT mice.
Convex hull: Graphed in Figure 12 (B) is the total convex hull surface area of the traced microglia of the groups of mice. The total convex hull surface area is suggestive of the area in space that the microglia consume. The total convex hull of the PS19 mice is smaller than the WT mice, regardless of treatment group. A mixed design repeated measures ANOVA was used to analyze the results. Within subjects there were no significant effects of total convex hull (F (3, 69) = 0.76, p = 0.52), convex hull x treatment group interaction (F (3, 69) = 2.02, p = 0.12) nor convex hull x genotype interaction (F (3, 69) = 1.17, p = 0.33). Between subjects, while there was no significant effect of treatment (F (1, 23) = 0.14, p = 0.71), there was a significant effect of genotype (F (1, 23) = 15.649, p < 0.001). Overall, the PS19 mice had a smaller convex hull surface area compared to the WT mice. Whether the mice received the vehicle or CBD did not impact the convex hull surface area of the traced microglia.
Cell body volume: Figure 12 (C) demonstrates the cell body volume of the microglia for all groups. The PS19V mice had the largest cell body volume. The cell body volume of the PS19C, WTV, and WTC were similar in size. A mixed design repeated measures ANOVA was used to analyze the observed results. Mauchly's test of Sphericity was violated (ε = 0.75), thus Greenhouse-Geisser degrees of freedom were reported. The results suggest that within subjects there was no significant effect of the total cell body volume for each microglia traced (F (2.250, 51.753) = 0.65, p = 0.54). There were also no significant interaction effects of cell body volume x treatment (F (2.250, 51.753) = 0.32, p < 0.76), nor cell body volume x genotype (F (2.250, 51.753) = 2.11, p = 0.13). There was a significant effect of treatment (F (1, 23) = 10.558, p < 0.004), genotype (F (1, 23) = 15.356, p < 0.001). and a genotype x treatment interaction (F (1, 23) = 9.57, p < 0.005). Due to the significant interaction, pairwise comparisons with Bonferroni corrections further analyzed the results. The results revealed that total cell body volume was only significant between the WT and PS19 groups that received vehicle (p < 0.001), but not when they received CBD (p = 0.56). Furthermore, while the total cell body volume was not significant between the WT mice depending on treatment group (p = 0.92), it was significant between the PS19 mice depending on treatment group (p < 0.001). Overall, the results show that CBD appears to reduce the cell body volume of the PS19 mice to a size similar to that of the WT mice.
Cholinergic neurons in MSDB. Research suggests that the cholinergic system plays a vital role in memory and attentional processes and that cholinergic tone is decreased in aging and AD. 70 As illustrated in Figure 13, the MSDB was stained with ChAT antibody to identify cholinergic cells. A univariate three-way ANOVA indicated no significant effects of genotype (F (1, 19) = 1.98, p = 0.18), treatment (F (1, 23) = 0.8, p = 0.39), nor sex (F (1, 23) = 0.76, p = 0.39). This result revealed that in the PS19 mice had similar numbers of cholinergic cells in the MSDB as the WT mice. CBD did not impact cholinergic tone in these regions.
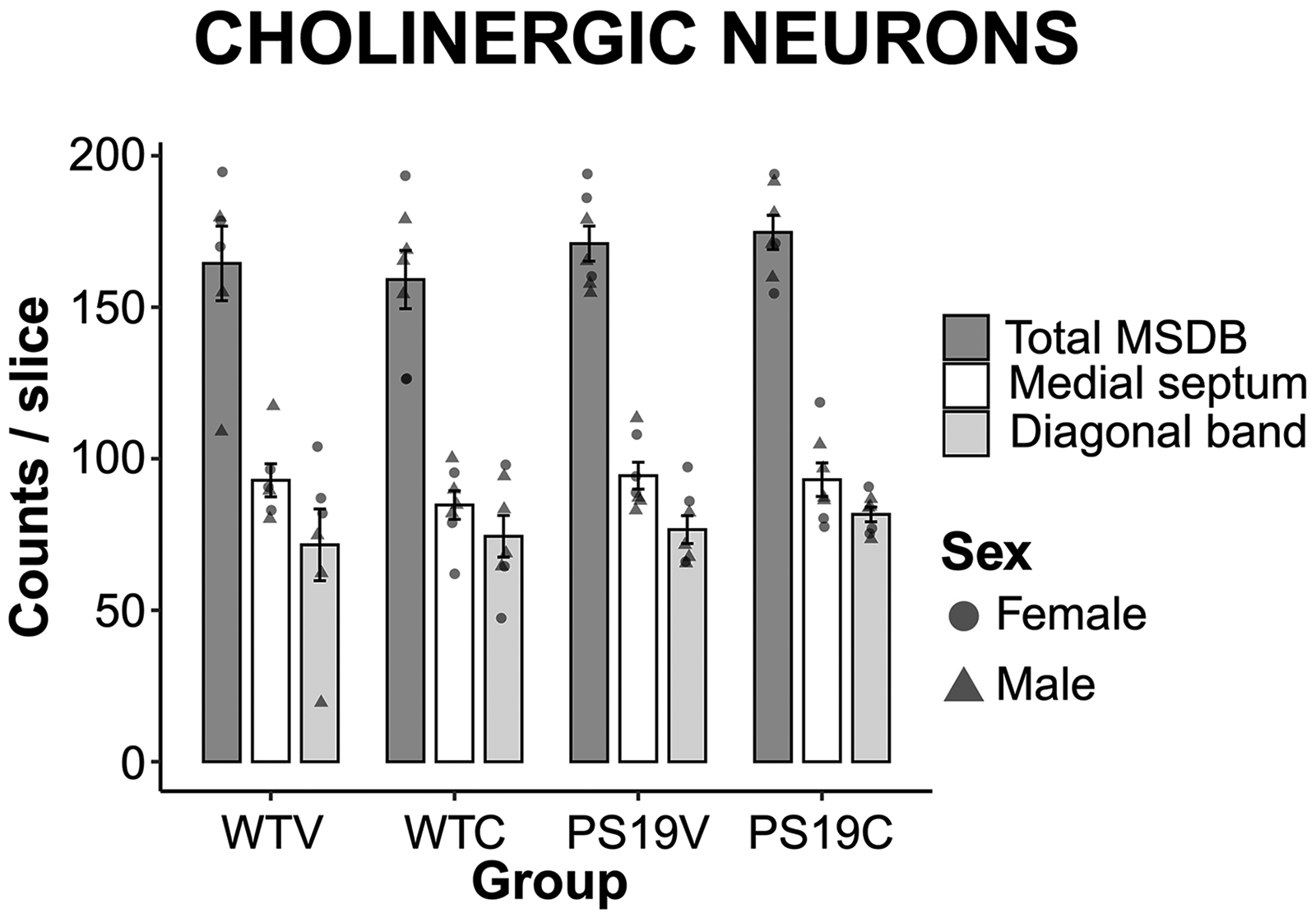
Cholinergic neurons in the medial septum and diagonal band (MSDB). The number of cholinergic neurons per slice in the medial septum, diagonal band or combined total in the WT mice and PS19 mice treated with vehicle (V) or CBD (C). There was no impact of genotype nor CBD on cholinergic neurons in these regions.
Phosphorylated tau in hippocampus. Four mice were removed from the analysis because of autofluorescence which made the AT8 quantification unreliable. The group sizes for the tau analysis were: WTV male (n = 3), WTV female (n = 3), WTC male (n = 2), WTC female (n = 2), PS19V male (n = 3), PS19V female (n = 3), PS19C male (n = 4), PS19C female (n = 3). As can be seen in Figure 14, the PS19 mice had more AT8-positive tau in HPC than the WT mice.
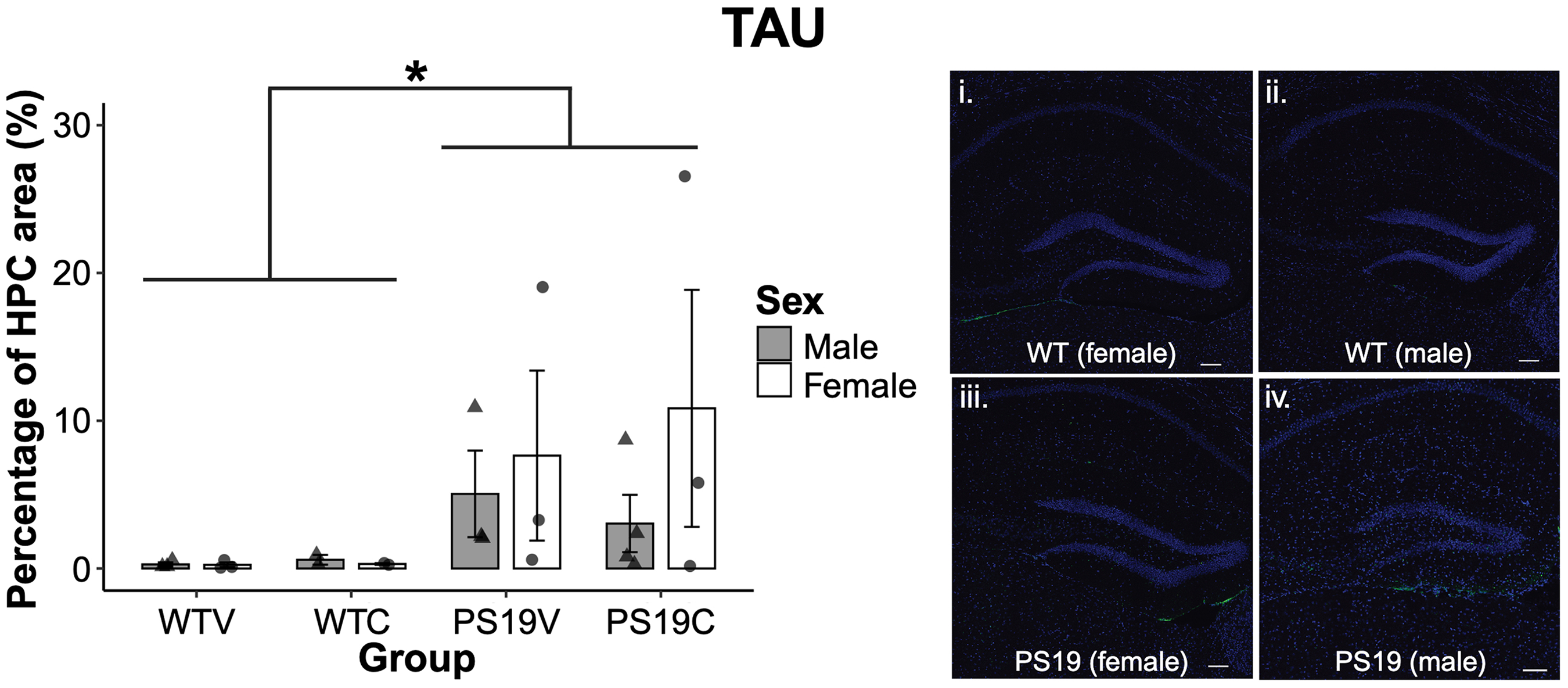
A representation of the distribution of tau, stained with AT8, in the hippocampus (HPC) of the mice depending on genotype and sex. Brain images were taken with a confocal microscope, and the scale bar represents 100 µm. (left) The percentage of AT8-positive tau in HPC of male and female WT mice and PS19 mice treated with vehicle (V) or CBD (C). The PS19 mice had significantly higher percentage of tau compared to WT mice (*p = 0.047). CBD did not impact the amount of tau in the PS19 mice. Image (i) demonstrates a WT female mouse, (ii) demonstrates a WT male mouse, (iii) demonstrates a PS19 female mouse, and (iv) demonstrates a PS19 male mouse. All the brains shown were mice treated with CBD.
A univariate three-way ANOVA indicated a significant effect of genotype (F (1, 15) = 4.734, p = 0.046) but no effects of treatment (F (1, 15) = 0.02, p = 0.90), sex (F (1, 19) = 0.75, p = 0.40), nor their interactions. This suggests that CBD did not reduce the amount of phosphorylated tau in the PS19 mice. It should be noted that the percentage of tau reported in the PS19 mice was likely an underestimation because of our conservative quantification method employed due to similar fluorophores used for Dapi (staining of neurons) and AT8 as well as autofluorescence artifacts.
Discussion
The aims of the present experiment were to characterize the Tau P301S-Line19 mice and littermate controls by assessing (1) cognitive and motor abilities at 6 and 9 months of age; (2) the effects of chronic, pre-symptomatic administration of CBD on improving any observed learning or motor impairments and reducing brain pathology; and (3) the safety and tolerability of chronic oral CBD (20 mg/kg) administration. The results suggest that the spatial recall of the Tau P301S mice began to decline around 6 months of age, while the object recognition, spatial learning, and hind limb strength began to decline around 9 months of age. The Tau P301S mice retained motor coordination and fear associative memory. No cognitive impairments were reported for the controls. CBD did not restore the cognitive functions of the Tau P301S mice but did improve performance on the MWT in the WT mice in comparison to the WT mice treated with vehicle. CBD also improved motor coordination in both groups at 6 months old, and improved hind limb strength in the PS301S mice at 9 months old. In addition to the cognitive impairments, the Tau P301S mice had pronounced HPC volume loss, as well as increased inflammation in the HPC and PFC relative to the controls around 10 months of age. Neurodegeneration and the percentage of astrocytes in the HPC of the male Tau P301S mice was more severe than the female Tau P301S mice. The pathology in the PFC varied among all Tau P301S mice. CBD was unable to restore the severe HPC volume loss observed in the Tau P301S mice. Furthermore, CBD did not affect the quantity of astrocytes in the HPC and PFC of the mice. However, there was a marginal trend towards CBD decreasing the quantity of microglial in the HPC of the Tau P301S mice, suggesting potential anti-inflammatory effects of the cannabinoid compound. Further support for this claim came from a more in-depth analysis of the morphology of the microglia, which showed that CBD appears to reduce the cell body volume of the microglia in the HPC of the Tau P301S mice to a size similar of that of the controls. CBD did not differently impact the pathology of the mice depending on sex. CBD did not impact the brains of the controls, which did not demonstrate HPC cell loss or pronounced astrogliosis and microgliosis. Finally, CBD was well tolerated regardless of genotype, suggesting its chronic oral consumption was safe at 20 mg/kg, in mice. It is important to note that although our concentration of CBD was 20 mg/mL and confirmed with HPLC, the bioavailability of the 20 mg/kg dose administered orally could not be confirmed due to the brain tissue being collected at least 12–18 h post-treatment. Thus, pharmacokinetic analysis performed on these brains would not accurately assess the amount of CBD that reached the brain after such duration as the optimal time for this analysis is 20 min post-administration and this work clearly showed that similar doses are available in the brain at this time point. 71 Nonetheless, we did obtain reduction in microglia supporting our claim that CBD did reach the brain.
CBD treatment effects on the learning and memory of Tau P301S mice and controls
The NOR results demonstrate that at 9, but not 6 months of age the Tau P301S mice had an overall decreased preference for the novel object relative to controls suggesting impaired object recognition and dysfunction of a network centered around the perirhinal cortex. Previous authors have found Tau P301S mice to be impaired during NOR at 7, 51 8, 72 10 and 12 months of age. 73 In contrast, López-González et al. 47 found that the object recognition of the mice was not impaired at 9 months of age, but it was impaired at 10 months of age. Therefore, there is variability as to when Tau P301S mice demonstrate object recognition impairments.
In addition to the overall impairment among the transgenic mice at 9 months old, the Tau P301S mice treated with CBD were the only group that did not explore the novel object above chance. Importantly, there were no observed impairments on the object recall of the controls treated with CBD. Furthermore, there were no negative effects of CBD on any group of mice at 6 months of age. Hence, this CBD induced impairment requires caution and further research because it relies on a single data point and could be a statistical anomaly (Type I error), and the declined object recognition correlated to the treatment might be specific to the tauopathy mutation. To our knowledge there are no reports of the effects of CBD on the object recognition of a tauopathy mouse model. Previous experiments have found that CBD was able to improve object recall on Aβ mouse models of AD.44,74–76 During the course of the disease, Aβ and NFTs occur in conjunction. 77 As such, future studies should assess the effects of CBD on a mouse model that develops Aβ and NFTs to further observe if the benefits of the cannabinoid compound on Aβ mouse models persists in the presence of tau abnormalities.
Similarly, the spatial learning of the Tau P301S mice was impaired at 9, but not 6 months of age. Previous experiments have reported that the Tau P301S mice learn the location of the platform at a slower rate relative to controls at 6, 48 7, 51 10, and 12 months of age. 73 In contrast to these reports, a trend towards a spatial learning impairment at 10 but not 7 months of age was found. 49 Importantly, these studies only trained their mice for 4–5 days.49,73 In the present experiment we trained our mice for 8 days, and the learning of the Tau P301S mice was only impaired from day 5 to 8. We have previously argued that it takes 8 days of training to get asymptotic levels of performance on this task and for the subjects to exhibit strong spatial specificity to the trained platform location during the probe trial.55,62
Despite the intact spatial learning, the spatial recall of the Tau P301S mice began to decline at 6 months of age. While the quadrant preference of the mice was unaffected 24 h after training, the Tau P301S mice had a reduced number of annulus crossings relative to the controls, a strong indicator of impaired spatial specificity. Furthermore, during the retention test, performed one week after training, there was a strong trend towards an effect of genotype on the quadrant preference of the mice. CBD did not restore nor impair the spatial recall of the mice at 6 months of age. At 9 months old, only the controls treated with CBD demonstrated a preference for the target quadrant relative to the alternate quadrants. As such CBD might be promoting spatial recall in the controls as they age.
Previous experiments have found Tau P301S mice to have impaired spatial recall at 6–7 months of age.48,51 Wild type mice are generally unimpaired during spatial recall tasks at 9 months of age. The impairments observed in the present experiment by the control mice might be a result of retraining. While there was a three-month delay between MWT testing, and visual cues in the training environment were changed, it is plausible that at 9 months old, the mice recalled some form of spatial representation for the platform they were trained to locate at 6 months of age. This could have resulted in potential interference between competing memories acquired in that training context.78,79 Indeed, some room cues such as the door, the animal holding rack, shelf and sink would have remained the same and the mice could have also used the shape of the pool combined with the head direction system to orient themselves. 80 This was supported by the fact that the WT mice performed significantly worse than the PS19 mice on the first day of spatial training, suggesting that the increased path length to locate the platform could have been due to prior learning in the environment.
CBD did not impact the spatial learning and memory of the Tau P301S mice at 6 or 9 months of age. Interestingly, CBD might be promoting spatial recall in the controls at 9 months of age. Previous papers have reported the benefits of 20 mg/kg of CBD on the spatial learning of an Aβ mouse model. 46 Furthermore, 100 mg/kg of CBD improved the spatial learning and recall in a tauopathy mouse model. 43 Perhaps in the presence of tau mutations higher dosages of CBD are needed to observe behavioral benefits of the drug. In support of this theory, 50 mg/kg of CBD was found to be sufficient to improve cognitive impairments in an Aβ mouse model of AD. 81 However, when the same laboratory assessed the effects of 50 mg/kg of CBD on a tauopathy mouse model they found no benefits from the cannabinoid compound. 82 As such, future studies should observe the effects of higher dosages of CBD on tauopathy mouse models.
Additionally, there was a significant effect of genotype during the visible platform performance of the mice at 9, but not 6 months of age. Importantly, the Tau P301S mice did have a significantly reduced path length to locate the platform when it was visible, their path length was simply longer than that of the controls. In fact, the difference in genotype stemmed from the first trial of visible platform training as they were similar for the remaining trials. This suggests that the Tau P301S mice were initially reluctant to approach the platform, rather than indicating a decline in motor, motivational, and/or sensory behaviors. Consistent with this, a previous experiment did not report an effect of genotype during the visible platform test at 10 months of age. 49
Furthermore, the balance beam data contradicts the claim that the Tau P301S mice had motor impairments. Indeed, there was no effect of genotype during balance beam at 6 or 9 months of age. In support of this, Dumont et al., 49 did not find motor impairments during balance beam in Tau P301S mice at 2, 7, or 10 months of age. Interestingly, the CBD treated mice had a reduced latency to traverse the beam at 6 months of age, suggesting CBD might be improving the motor coordination of the P301S mice. At 9 months of age there was no longer an influence of CBD on the performance of the mice.
In contrast to our findings, the effects of 50 mg/kg of CBD was tested on Tau58/2 mice, the tau mice used in their experiment demonstrated motor impairments on balance beam and the CBD treated mice had an increased number of paw slips compared to the vehicle mice. 82 In the present study, CBD did not impact the paw slips of the mice. However, Giuliano et al., 83 found that when CBD was administered to a mouse model of Parkinson's disease, the motor coordination of the mice was improved on a cylinder task, but not on the rotarod task. Like the present experiment, no negative effects of CBD were found on the motor coordination of the animals. 83 More studies are needed to further observe the potential benefits or detriments of CBD on the motor coordination of mice, particularly with varying dosages, treatment lengths, and on various tasks.
While the Tau P301S mice did not demonstrate motor impairments, they did demonstrate hindlimb weakness at 9 but not 6 months of age as increased clasping behavior was observed during tail suspension. Yoshiyama et al., 53 have previously reported that Tau P301S mice demonstrate increased clasping behavior beginning at 3 months of age, while Dumont et al., 49 did not find muscular weakness assessed with a grip test at 10 months of age. The differing results further suggests variability as to when behavioral abnormalities commence within this mouse model. Regardless, in our present study, Tau P301S mice treated with CBD had improved hindlimb strength than Tau P301S mice treated with vehicle.
Finally, the fear conditioning data suggests that the Tau P301S mice did not demonstrate contextual recall or tone recall impairments around 10 months of age. However, the freezing response of the Tau P301S mice before the tone, in a novel context, was increased relative to the controls. Due to the increased freezing in the novel context, the contextual recall of the Tau P301S mice was less pronounced than the controls. Interestingly Takeuchi et al., 48 also found that the freezing of Tau P301S mice during context recall was less pronounced, but not statistically different from controls. Furthermore, previous experiments have found that the Tau P301S mice were significantly impaired during context recall, but not tone recall, at 8 months of age. 72
The increased freezing response of the Tau P301S mice during the novel context does not necessarily contradict the fear conditioning data. Previous papers have found a correlation between increased freezing prior to the tone with increased freezing during the tone, 84 suggesting that the “baseline” freezing measure used throughout these kinds of experiments was imprecise. As such, future studies should record the freezing of their mice prior to conditioning. Moreover, the increased freezing prior to the tone, in the novel context, might be suggestive of contextual generalization. Increased contextual generalization would not indicate that the mice did not form the context/shock or tone/shock pairing, rather it would suggest that they did not retain a distinct representation of the conditioning context.84,85 Some have shown that ventral HPC dysfunction is responsible for this kind of context generalization effect, but others have implicated the orbital frontal cortex.86,87 While some may argue that the Tau P301S mice are less mobile than the controls, this is unlikely as the total exploration during NOR was the same for these groups. Nevertheless, CBD did not impact the fear associative memory of the mice in the present experiment. Like the present experiment, AβPP/PS1 mice treated with 20 mg/kg had increased baseline freezing response, but the freezing of the transgenic mice during context and tone recall did not differ from controls. 45 Similarly, CBD did not impact the fear conditioning of their mice.
In general, when observing the age of cognitive decline among the Tau P301S mice, there are clearly conflicting results between laboratories. The conflicting results might be due to experimental differences (i.e., training duration, apparatuses, housing). It may also be a result of the background strain of the mice. The Tau P301S mice in the current experiment were kept on a c57BL/6N background, whereas previous papers have used the Tau P301S mice on a B6C3H/F1 background.51,53 A direct comparison of the transgenic mice depending on the background has not yet been characterized, however previous research suggests that the background of Aβ mouse models of AD has implications on the severity and onset of cognitive decline and pathology.88,89 In our view, it is important to consider the background of the mice when comparing experimental results as it might have implications on the onset of symptoms and development of pathologies. In support of this, the ethnicity of AD cases impacts the risk factors associated with the onset and severity of the disease in humans. 90 Future studies should directly compare Tau P301S-Line PS19 mice on a c57BL/6N background to the mice on B6C3H/F1 background to further characterize the differences in their behavior and brain pathology at different ages. Based upon the findings in the present experiment, there is reason to believe that the Tau P301S mice decline at a slower rate when bred on a c57BL/6N background. Despite the potential delay in the onset of cognitive decline, the pathology of the mice was similar with previous experiments.
Additionally, while there were no clear effects of CBD on the behavior of the Tau P301S, it is important to note that the male and female mice were grouped together for the behavioral analysis. Grouping the behavior of the mice regardless of sex may have resulted in increased variability rendering it difficult to view statistically significant results. In support of this, Sun et al., 52 have reported that the onset and severity of learning and memory, as well as motor impairments among Tau P301S mice differs significantly between males and females. The same observation was made in the present experiment. Furthermore, the pathology of the male and female mice differed significantly in the present experiment and in previous experiments. 52 Future studies should include sex as a factor when assessing the behavior of Tau P301S mice to limit variability. Unfortunately, this was not possible in the present experiment as there would not have been enough animals to include sex as a factor when analyzing the behavioral results.
The effects of CBD on AD-related pathologies in Tau P301S mice
Overall, the 10-month-old Tau P301S mice had visible degeneration within the HPC formation, particularly the male Tau P301S mice. The observed HPC volume loss and sex difference was statistically supported. Consistent with our findings, previous experiments have reported significant HPC cell loss in Tau P301S mice at 8–9 months of age.53,72 CBD was unable to restore the HPC volumes of the Tau P301S mice around 10 months of age. In contradiction to these reports, CBD has been shown to prevent cell death, promote cell survival and promote neurogenesis in vitro,15,35,37,40,41 and in vivo.41,91 However, to our knowledge, the effects of CBD on cell loss in a tauopathy mouse model has not been reported. Neurodegeneration in Tau P301S mice is related to the toxic effects of tau, as such CBD might be ineffective in delaying this cascade of events. Future studies should assess neurogenesis to observe if CBD is able to promote neurogenesis in the presence of tau abnormalities.
In addition to the reduced HPC volumes, the Tau P301S mice demonstrated increased positively stained GFAP cells and Iba1 cells in the HPC and PFC around 10 months of age. Specifically, the morphology of the GFAP and Iba1 stained cells appeared reactive. Reactive astrocytes were classified to have enlarged cell bodies and long, thick processes, whereas reactive microglia had enlarged cell bodies and short processes. In support of this, previous papers have also found that astrogliosis and microgliosis to be increased in the brains of Tau P301S mice beginning at 3–6 months of age,51,53 increasing from 9–10 months of age.47,52 Furthermore, the HPC of the male Tau P301S mice demonstrated increased levels of GFAP relative to the female Tau P301S mice. The effect of sex was not observed on the quantity of Iba1 in the HPC or PFC of the Tau P301S mice. This result is supported by a previous experiment that also found GFAP, but not microglia to be increased in the HPC of the male Tau P301S mice relative to females. 52
While the findings that the pathology of the male Tau P301S mice progressed faster relative to the female Tau P301S mice was consistent with previous reports on this strain, 52 it contradicts AD as seen in humans. AD related pathologies are more severe in females than males, 92 and female AD patients have more severe atrophy of regions within the medial temporal lobe. 93 Furthermore, the prevalence of AD among females is higher than the prevalence of AD among males. 90 Despite the discrepancy between sex differences observed within this mouse model and AD as seen in humans, there was no consistent trends on CBD impacting the pathology of the male and female mice differently. To our knowledge there have been no previous papers that report the effects of CBD on the progression of AD depending on sex. Future studies should further assess if the cannabinoid compound impacts male and female AD mouse models differently, particularly given the differences in the prevalence and presentation of AD in humans.
There was a marginal trend towards an effect of CBD on reducing the quantity of Iba1 in the HPC, but not PFC of the Tau P301S mice. We further explored this trend by using a more sensitive measure of assessing reactive microglia to elucidate the effects of CBD. Hovens et al. 94 suggest that cell body to cell size and dendrite size to cell size are accurate measurements of reactive microglia. Accordingly, we used a 3-dimensional imaging technique to assess some of these microglia characteristics which revealed that CBD significantly altered the conformation of at least one of the inflammatory markers. This was an important result as it suggested that the treatment was getting into the brain and reduced at least one aspect of the pathology associated with AD.
Previous experiments have reported anti-inflammatory effects of CBD in vitro and in vivo in the presence of AD toxicities.39,41,45,46,74,81 To our knowledge this is the first experiment to assess the effects of pure CBD, in vivo, on the pathology of a tauopathy mouse model. As such there is a need for more experiments to further understand the impact of CBD on inflammation related to the hyperphosphorylation of tau. Researchers have reported that microglia, but not astrocytes precede the accumulation of hyperphosphorylated tau among Tau P301S mice.52,53 Therefore, the marginal trend towards a decrease in the percentage of microglia in the HPC and the clear reduction in cell body size in the HPC, but not astrocytes might suggest that CBD could also delay the hyperphosphorylation of tau within this strain of animals, which would further support its therapeutic usage for AD. In support of this, the phosphorylation of tau was increased in the presence of inflammation. 53 Specifically, within this strain, the distribution of inflammation is believed to correlate with the distribution of tau pathology. 52 Interestingly, when Tau P301S mice were treated with immunosuppressants at 2 months of age, their survival rate increased and the mice demonstrated reduced inflammation, atrophy, and ultimately a reduction in phosphorylated tau and insoluble tau. 53 Similarly, depleting microglia in Tau P301S mice was found to reduce phosphorylated tau by 47% and 70%. 95 Therefore, there is reason to believe that reducing inflammation would reduce the phosphorylation of tau. Our assessment of tau pathology in Tau P301S mice did not reveal a significant effect of CBD reducing the progression and accumulation of hyperphosphorylated tau. However, our group sizes on this measure were small, limiting the conclusions that can be drawn from these results.
Finally, the Tau P301S did not show reductions in cholinergic neurons in the medial septum/diagonal band, which are found in human AD patients and other preclinical mouse models of AD.70,96,97 These neurons project to the HPC modulating learning, memory and attentional processes, and can be impacted by treatment interventions like exercise and certain forms of cognitive training.96,97 The lack of cholinergic depletions in this tau model of AD but significant depletions in an APP model of AD suggests that the cholinergic system alterations are somehow linked to Aβ pathologies. 98
Conclusion
This study investigated the effectiveness of CBD as a treatment for AD using a tauopathy mouse model, Tau P301S-Line PS19. The results showed that chronic and daily CBD intake did not improve memory, restore HPC volume reductions, or attenuate brain inflammation increases in the Tau mice. Importantly, a deeper look at changes in brain inflammatory markers showed a treatment effect by reducing microglia reactivity. More broadly, these experiments revealed significant sex differences in the expression of the tauopathy phenotype with males showing more severe pathology on some measures relative to females. Finally, to our knowledge the present experiments represent one of the longest chronic treatment regimens ever completed, with mice receiving 7 months of daily CBD treatment. This is important to note because these chronic treatments of CBD at 20 mg/kg did not negatively impact the behavior or brain of any of the experimental groups based on our metrics, suggesting that long-term administration is relatively safe.
Supplemental Material
sj-docx-1-alz-10.1177_13872877261421654 - Supplemental material for The effects of chronic cannabidiol administration on brain pathology and behavioral deficits found in the tau P301s-line PS19 mouse model of Alzheimer's disease
Supplemental material, sj-docx-1-alz-10.1177_13872877261421654 for The effects of chronic cannabidiol administration on brain pathology and behavioral deficits found in the tau P301s-line PS19 mouse model of Alzheimer's disease by Abigail G. Nixon, Nancy S. Hong, Behroo Mirza Agha, Jackson R. Ham, Merrin Monteith, Tina Kaloa, Angela Kyriazopolus, Matt Nielsen, David R. Robertson, Andrey Golubov, Robert J. Sutherland, Igor Kovalchuk, Andrew Iwaniuk, Majid H. Mohajerani and Robert J. McDonald in Journal of Alzheimer's Disease
Footnotes
Acknowledgements
The authors would like to thank Val Lapointe for histological advice and assistance, and Kortney Dyck and Brianna Olson for providing some assistance with treatments. We also thank Di Shao for mice breeding and genotyping, and the animal welfare staff for care of the mice. This research project would not be possible without funding from Canadian Institutes of Health Research (CIHR) and the University of Lethbridge Research Fund, and trainee scholarships from CIHR, Alzheimer's Society of Canada, Alberta Innovates, and Chinook.
Ethical considerations
The animal study was approved by the University of Lethbridge Animal Welfare Committee. All methods and procedures described in this experiment were conducted in accordance with the Canadian Council of Animal Care Guidelines and the University of Lethbridge Institutional requirements.
Consent to participate
Not applicable
Consent for publication
Not applicable
Author contribution(s)
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by a Canadian Institutes of Health Research Project grant [PJT-175091] and University of Lethbridge Research Fund awarded to RJM. AGN received funding from CIHR. JRH received a postdoctoral fellowship from Alzheimer's Society of Canada. TK, MM and DR received funding from Alberta Innovates summer research studentship. MN received funding from Chinook summer research award.
Canadian Institutes of Health Research (grant number PJT-175091), University of Lethbridge Research Fund .
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
The data is available upon reasonable request to the corresponding author.
Supplemental material
Supplemental material for this article is available online.
