Abstract
Phosphodiesterase-5 (PDE-5) inhibitors may be beneficial in Alzheimer's disease (AD). We assessed the PDE-5 inhibitor tadalafil effect on plasma biomarkers of neurodegeneration in 15 individuals with type 2 diabetes post-hoc in a randomized placebo-controlled trial (ClinicalTrials.gov: NCT02601989) at Sahlgrenska University Hospital. Tadalafil reduced plasma amyloid-β 40 and 42 but not the 42/40 ratio over a 6-week treatment period. Glial fibrillary acidic protein was reduced, but not phosphorylated tau217, neurofilament light protein or growth/differentiation factor 15. Tadalafil reduced plasma levels of biomarkers for amyloid metabolism and astroglial activation in patients with diabetes. Designated clinical trials are warranted to validate these results.
Keywords
Introduction
Patients with type 2 diabetes have almost twice the incidence of dementia relative to other patient groups and prioritization of neurodegenerative outcomes in diabetes trials has been proposed. 1 According to the National Institute on Aging-Alzheimer's Association, knowledge on how comorbidities and medications affect circulating brain biomarkers is a top research priority. 2 This is of relevance when assessing the effects of pharmaceutical treatment in patients with diabetes given the bidirectional relationship between cognitive dysfunction and diabetes management. 3
Well-established blood-based biomarkers allow for a sensitive evaluation of several aspects of the neurodegenerative spectra. Decreased concentrations of the 42-amino acid isoform of amyloid-β (Aβ42) is the defining marker in Alzheimer's disease (AD), 2 and is often adjusted for by the most abundant Aβ isoform, Aβ40, through the Aβ ratio. Tauopathy in AD can be assessed by measuring phosphorylated tau217 (pTau217) in plasma. 4 Aβ42 and pTau217 may also be used together for screening of AD through the pTau217/Aβ42 ratio. 5 Neurofilament light protein (NfL) is a sensitive yet nonspecific marker of neurodegeneration and axonal degeneration 6 and glial fibrillary acid protein (GFAP) is a prototypical marker of astrocytic activation, which increases over the AD continuum. 7 Furthermore, growth/differentiation factor 15 (GDF-15) is a cellular stress marker related to metabolic disease and inflammation. 8
A longitudinal cohort study recently reported that treatment with a phosphodiesterase-5 (PDE-5) inhibitor is associated with a lower incidence of AD. 9 A phase III randomized controlled clinical trial (RCT) of a PDE-5 inhibitor is ongoing (ClinicalTrials.gov, NCT05531526). In a recent placebo-controlled RCT in patients with diabetes, we showed that chronic treatment with the PDE-5 inhibitor tadalafil lowered mean blood glucose as measured by hemoglobin A1c (HbA1c). 10 In this post-hoc analysis of the RCT, we hypothesized that tadalafil would modulate blood-based biomarkers of neurodegeneration and cellular stress in apparently cognitive healthy patients with diabetes.
Methods
Participants
In this post-hoc analysis, we measured biomarkers of neurodegeneration and cellular stress in plasma samples from fifteen individuals with diabetes who completed a double-blind, randomized, placebo-controlled, cross-over phase 2 trial using tadalafil 20 mg once daily over 6 weeks per-protocol (ClinicalTrials.gov, NCT02601989) (Figure 1a). 10 Of the 23 individuals originally enrolled, 15 completed the trial with attendance at all visits, daily use of the study medication and valid measurements for all pre-registered endpoints. Participants were examined at Sahlgrenska University Hospital and the Wallenberg Laboratory, Sahlgrenska Academy, University of Gothenburg, between 22 January 2016 and 31 January 2019. Cognitive impairment symptoms were exclusion criteria as they could negatively affect compliance with the study protocol.
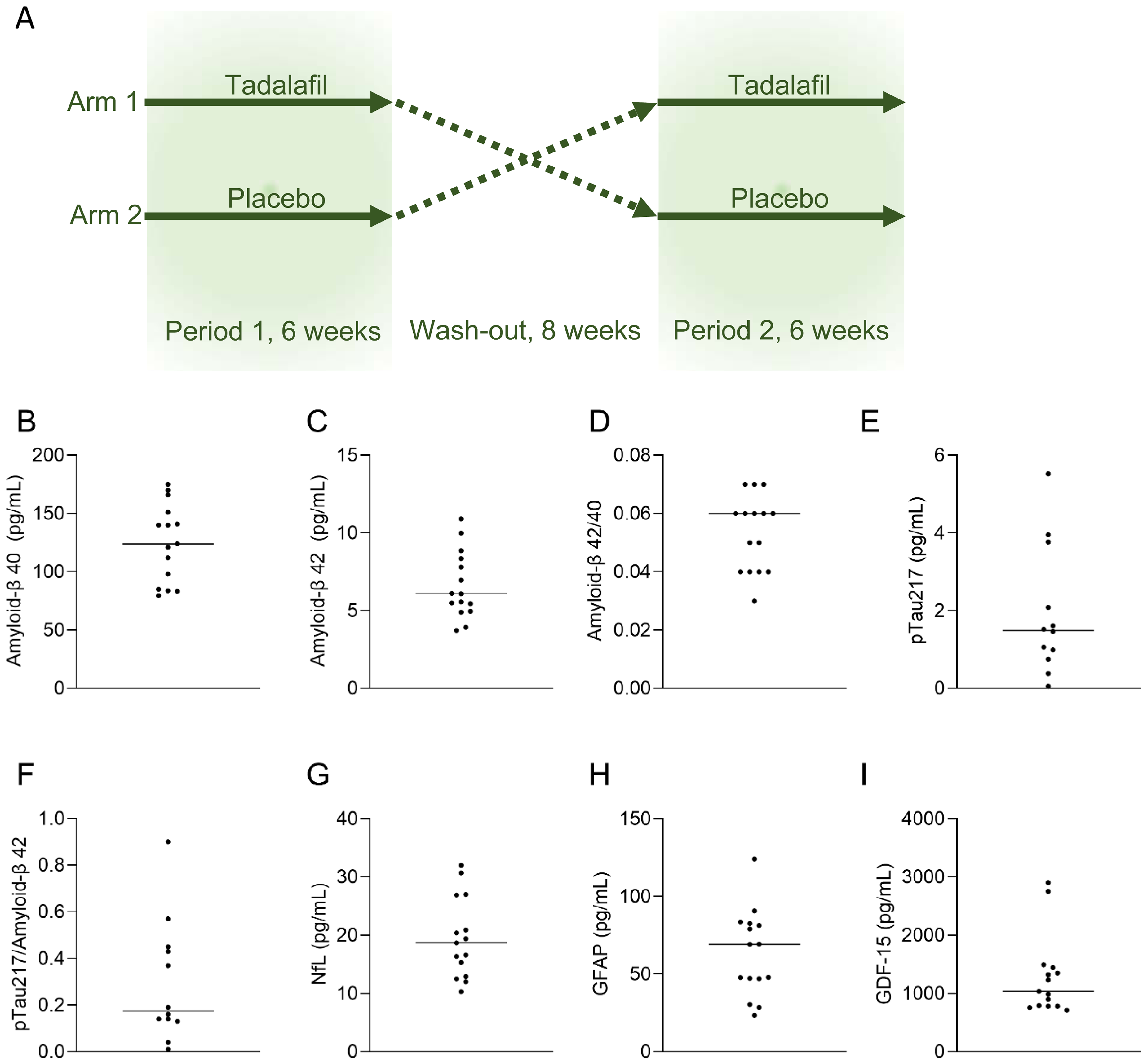
Schematic illustration of the MAKROTAD study. 10 Patients with type 2 diabetes were randomized to start with either 6-week daily administration of 20 mg tadalafil, or placebo. After 8 weeks of washout, participants shifted treatment for the second treatment period of 6-weeks. Fasting venous blood samples were collected before and after each treatment period (A). Biomarkers of neurodegeneration at randomization for participants in the per protocol analysis (before treatment, n = 15, except pTau217, n = 12), line indicates median value (B-H). pTau217: phosphorylated Tau217; NfL: neurofilament light protein; GFAP: glial fibrillary acid protein; GDF-15: growth/differentiation factor 15.
Measurements
At an initial screening visit, participants were interviewed about their health and lifestyle habits. At the start (baseline) and end of each 6-week treatment period, participants attended the clinic after an overnight fast for measurements including weight, height, waist circumference, blood pressure, and venous blood sampling. Plasma was stored at −80 °C awaiting analysis. Blood HbA1c, plasma glucose, cholesterol and lipoprotein cholesterols, triglycerides, creatinine, alanine aminotransferase (ALT) and aspartate aminotransferase (AST) in serum and creatinine/albumin ratio in urine were analyzed with accredited methods at the Clinical Chemistry Laboratory at Sahlgrenska University Hospital, Gothenburg. Plasma Aβ40, Aβ42, NfL, and GFAP concentrations were measured using a commercially available Single molecule array (Simoa) assay on the HD-X platform (Quanterix, Billerica, MA), pTau217 with the Lumipulse immunoassay (Fujirebio) and GDF-15 using an Elecsys electrochemiluminescence immunoassay on the Cobas platform (Roche Diagnostics, Rotkreuz, Switzerland). A single batch of reagents was used, and all measurements were performed in a single round of experiments by board-certified laboratory technicians blinded to clinical data and treatments. Intra-assay coefficients of variation were below 10% for all analytes. For pTau217, plasma was available for 12 and 9 individuals at baseline and follow-up, respectively, and levels were below the limit of quantification (1.2 pg/mL) in 43% of the samples. Measurements below level of quantification were also included in the analysis and were set to 0.05. Biomarkers of neurodegeneration were analyzed at the Neurochemistry Laboratory, Sahlgrenska University Hospital, Mölndal, Sweden.
Statistics
Data in Table 1 is presented as n (%) for categorical variables and mean (SD) of continuous variables for participants in the per protocol analysis. For treatment outcomes in Table 2, period-adjusted p-values were calculated by standard cross-over analysis method using Fisher's non-parametric permutation test. The limited sample size did not allow for adjustment of confounding variables. SAS 9.4 by SAS Institute Inc., Cary, NC, USA was used for the analyses.
Characteristics of study participants.

Data are presented as n (%) for categorical variables and mean (SD) of continuous variables for participants in the per protocol analysis, n = 15 (10).
ALT: alanine aminotransferase; AST: aspartate aminotransferase; B: blood; fP: fasting plasma; HbA1c: hemoglobin A1c; HDL: high-density lipoprotein; LDL: low-density lipoprotein; S: serum; U: urine.
Normal reference for HbA1c ≤ 48 mmol/mol.
Change in mean (95% CI) biomarker concentrations following treatment with tadalafil or placebo.

Period-adjusted p-value was calculated by standard cross-over analysis method using Fisher's non-parametric permutation test. B: blood; fP: fasting plasma; GDF-15: growth/differentiation factor 15; GFAP: glial fibrillary acidic protein; NfL: neurofilament light protein; pTau217: phosphorylated Tau217. Statistical significance was defined as p < 0.05, n = 15.
*n = 9 in longitudinal analysis.
Statistical significance in bold was defined as p < 0.05.
Results
Characteristics of study participants
At baseline, participants had the following mean values: HbA1c 45 mmol/mol, BMI 31.5 kg/m2, blood pressure 148/88 mmHg, and LDL cholesterol 3.4 mmol/L (Table 1). Baseline levels of Aβ42/40, pTau217, pTau217/Aβ42, NfL, GFAP, and GDF-15 are presented in Figure 1.
Effects of tadalafil treatment
Tadalafil treatment, compared with placebo, did not alter BMI, blood pressure or fasting plasma glucose, but decreased HbA1c, as previously shown in the full analysis set. 10 Tadalafil also decreased plasma levels of Aβ40 and Aβ42 but did not affect the Aβ42/40 ratio, pTau217 or the pTau217/Aβ42 ratio in plasma (Table 2). Further, tadalafil decreased the plasma level of GFAP, while plasma levels of NfL and GDF-15 did not change compared to placebo. Changes in Aβ40, Aβ42, Aβ42/40, pTau217, pTau217/Aβ42, NfL, and GFAP did not correlate with changes in HbA1c when comparing tadalafil and placebo. However, a moderate inverse correlation was observed between GDF-15 and HbA1c (Supplemental Table 1).
Discussion
We performed a post-hoc analysis of a placebo-controlled RCT to assess the effect of 6-week daily treatment with tadalafil 20 mg on blood-based biomarkers of neurodegeneration in patients with diabetes. Tadalafil, compared with placebo, decreased plasma levels of Aβ40 and Aβ42 and the neuroinflammatory marker GFAP, but did not affect the AD-specific biomarker Aβ42/40 ratio or plasma levels of pTau217. The net change of the stress marker GDF-15 in plasma after tadalafil versus placebo was inversely correlated with the net change of HbA1c.
Our observation that plasma levels of Aβ40 and Aβ42, but not the Aβ42/40 ratio, were decreased by tadalafil suggests that the observed effects of tadalafil are not specific for AD plaque pathology. However, the results may indicate an effect on amyloidogenic processing of AβPP, the precursor protein of Aβ. 11 It is also possible that improvements in microcirculation induced by tadalafil could improve clearance of Aβ40 and Aβ42, or that tadalafil modulates inflammatory effects with secondary effects on both Aβ40 and Aβ42. 12 Previous studies on comorbidities in AD have reported that diabetes is associated with increased levels of Aβ40 and Aβ42, but not the Aβ42/40 ratio in plasma. 13 It is possible that the decrease in Aβ induced by tadalafil normalizes this increase. 13 Studies in animal models have previously suggested anti-neuroinflammatory effects of tadalafil related to amyloid pathology, and tadalafil has been shown to modulate both mTOR and NF-κB expression in the rodent brain. 14 Similarly, a dual PDE-4/PDE-5 inhibitor has been shown to reduce activation of astrocytes and increase neuron numbers and mitigate neuroinflammation through the NF-κB and JNK pathways in cell studies and mice. 15 Here, tadalafil also decreased levels of the astrocyte marker GFAP, indicating an improvement in neuroinflammation. This is of interest as it has previously been reported that an increased circulating GFAP level among patients with diabetes and obesity correlates with a decrease in incident mild cognitive impairment. 16 Additional experiments should be conducted to further enhance the understanding of our observations in this clinical trial.
Baseline NfL levels were slightly higher than levels seen in healthy individuals but comparable with levels previously reported in patients with chronic kidney disease. 17 Notably, the three individuals with the highest NfL levels also showed the highest pTau217 concentrations, indicating ongoing neuronal injury.
Furthermore, we did not observe any association between NfL and HbA1c. This may be explained by a low prevalence of complications, such as peripheral neuropathy, in our study cohort. 10 Several studies have recognized the influence of somatic comorbidities on the interpretation of results using blood-based biomarkers of neurodegeneration.13,17,18 Furthermore, we have previously shown that the neprilysin inhibitor sacubitril, which is commonly used for the treatment of heart failure, skews levels of AD blood biomarkers based on Aβ, either in a ratio or by itself. 19 This type of interference will be important as AD biomarkers enter clinical diagnostic use because it could lead to misdiagnosis. In our study, neither treatment with tadalafil nor modification of glycemia in patients with diabetes altered the Aβ42/40 ratio, pTau217, or NfL biomarker levels; thus, these conditions do not confound clinical interpretation of these AD biomarkers. While not seen in this study it is possible that other ratios that include Aβ42, e.g., the plasma pTau217/Aβ42 ratio, could be affected in some circumstances. 5 A clinical consequence of our finding of decreased plasma Aβ42 levels is therefore that tadalafil may be another drug affecting AD blood biomarkers that include plasma Aβ. 20
Suppression of HbA1c during tadalafil treatment was associated with increased levels of the cellular stress marker GDF-15 levels in plasma. This observation is in agreement with proposed mechanisms behind the beneficial metabolic effects of GDF-15. 8
We report several novel observations in this post-hoc analysis from a small RCT. A limitation is that the study was not specifically powered for the outcomes of this sub-study and the small sample size did not allow for adjustment of confounding variables. However, we did not observe any significant changes in BMI and blood pressure over the treatment period. The clinical significance of our findings requires future validation, but they may be helpful to guide future research. Furthermore, to improve current treatment strategies in diabetes and neurodegenerative conditions, it is important that data from clinical trials are shared regardless of the study outcome. 21 Unfortunately, we did not assess cognitive performance pre- and postintervention, and therefore it was not possible to evaluate whether differences in cognition were associated with changes in biomarkers. Our participants were cognitively healthy, and it is possible that tadalafil could have other or more pronounced effects in individuals with cognitive disease. Here, our pTau217 measurements were very low, often below limit of quantification. There was no indication that tadalafil would increase pTau217, but any lowering effect would be missed due to the low measurements.
In conclusion, daily tadalafil treatment in cognitively healthy individuals with diabetes lowered specific blood-based biomarkers of neurodegeneration. Designated randomized clinical trials are warranted to validate the effect of PDE-5 inhibitors on circulating biomarkers of neurodegenerative disease in diabetes patients.
Supplemental Material
sj-docx-1-alz-10.1177_13872877261421227 - Supplemental material for Effect of 6-week tadalafil treatment on blood-based biomarkers of neurodegeneration: A post-hoc analysis of a randomized controlled trial
Supplemental material, sj-docx-1-alz-10.1177_13872877261421227 for Effect of 6-week tadalafil treatment on blood-based biomarkers of neurodegeneration: A post-hoc analysis of a randomized controlled trial by Emanuel Fryk, Vagner Ramon Rodrigues Silva, Lena Strindberg, Magnus Gisslén, Henrik Zetterberg, Kaj Blennow and Per-Anders Jansson in Journal of Alzheimer's Disease
Footnotes
Acknowledgements
We thank the study personnel and participants of the MAKROTAD-study for their important contributions. The study was designed and reported by the authors of this manuscript without influence of funding organizations or other external actors.
ORCID iDs
Ethical considerations
This study was conducted according to the Helsinki Declaration and was approved by the regional ethics review board in Gothenburg (291–15) and the Swedish Ethical Review Authority (2024-01843-02).
Consent to participate
All participants gave written consent before study initiation.
Consent for publication
All participants gave consent for additional analyses and publication as specified in the approval (2024-01843-02).
Author contribution(s)
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by grants from:
EF: Grants from the Swedish state under the agreement between the Swedish government and the county councils, the ALF-agreement (ALFGBG-935061), Gothenburg Medical Society (GLS-973024, GLS-985243 through Fred G och Emma E Kanolds stiftelse, John o Asta Falkmans fond, Therese Sandwalls fond and Vera och Hans Albrechtssons stiftelse), R&D Centre Gothenburg and Södra Bohuslän.
MG: Grants from the Swedish state under the agreement between the Swedish government and the county councils, the ALF-agreement (ALFGBG-1005848); the Swedish Research Council (2021-05045 & 2021-06545); and King Gustaf V:s and Queen Victoriás Foundation.
HZ: HZ is a Wallenberg Scholar and a Distinguished Professor at the Swedish Research Council supported by grants from the Swedish Research Council (#2023-00356, #2022-01018 and #2019-02397), the European Union's Horizon Europe research and innovation programme under grant agreement No 101053962, grants from the Swedish state under the agreement between the Swedish government and the county councils, the ALF-agreement (#ALFGBG-71320), the Alzheimer Drug Discovery Foundation (ADDF), USA (#201809-2016862), the AD Strategic Fund and the Alzheimer's Association (#ADSF-21-831376-C, #ADSF-21-831381-C, #ADSF-21-831377-C, and #ADSF-24-1284328-C), the European Partnership on Metrology, co-financed from the European Union's Horizon Europe Research and Innovation Programme and by the Participating States (NEuroBioStand, #22HLT07), the Bluefield Project, Cure Alzheimer's Fund, the Olav Thon Foundation, the Erling-Persson Family Foundation, Familjen Rönströms Stiftelse, Stiftelsen för Gamla Tjänarinnor, Hjärnfonden, Sweden (#FO2022-0270), the European Union's Horizon 2020 research and innovation programme under the Marie Skłodowska-Curie grant agreement No 860197 (MIRIADE), the European Union Joint Programme – Neurodegenerative Disease Research (JPND2021-00694), the National Institute for Health and Care Research University College London Hospitals Biomedical Research Centre, the UK Dementia Research Institute at UCL (UKDRI-1003), and an anonymous donor.
KB is supported by the Swedish Research Council (#2017-00915 and #2022-00732), the Swedish Alzheimer Foundation (#AF-930351, #AF-939721, #AF-968270, and #AF-994551), Hjärnfonden, Sweden (#ALZ2022-0006, #FO2024-0048-TK-130 and FO2024-0048-HK-24), the Swedish state under the agreement between the Swedish government and the County Councils, the ALF-agreement (#ALFGBG-965240 and #ALFGBG-1006418), the European Union Joint Program for Neurodegenerative Disorders (JPND2019-466-236), the Alzheimer's Association 2021 Zenith Award (ZEN-21-848495), the Alzheimer's Association 2022-2025 Grant (SG-23-1038904 QC), La Fondation Recherche Alzheimer (FRA), Paris, France, the Kirsten and Freddy Johansen Foundation, Copenhagen, Denmark, Familjen Rönströms Stiftelse, Stockholm, Sweden, and an anonymous philanthropist and donor.
PAJ: Grants from the Swedish state under the agreement between the Swedish government and the county councils, the ALF-agreement (2018-74560 and 2018-75190); the Swedish Research Council (2022-01011); and the Swedish Diabetes Foundation (DIA2021-631). Eli Lilly & Company, USA, supplied medication in the form of placebo and tadalafil 20 mg for the study. Eli Lilly & Company AB, Sweden, gave financial support to this investigator-initiated trial.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: EF, VRRS, LS, PAJ declare no conflicts of interest. MG has received research grants from Gilead Sciences and honoraria as speaker, DSMB committee member and/or scientific advisor for Amgen, AstraZeneca, Biogen, Bristol-Myers Squibb, Gilead Sciences, GlaxoSmithKline/ViiV, Janssen-Cilag, MSD, Novocure, Novo Nordic, Pfizer and Sanofi. All mentioned engagements have concluded and are not ongoing. HZ has served on scientific advisory boards and/or as a consultant for Abbvie, Acumen, Alector, Alzinova, ALZpath, Amylyx, Annexon, Apellis, Artery Therapeutics, AZTherapies, Cognito Therapeutics, CogRx, Denali, Eisai, Enigma, LabCorp, Merry Life, Nervgen, Novo Nordisk, Optoceutics, Passage Bio, Pinteon Therapeutics, Prothena, Quanterix, Red Abbey Labs, reMYND, Roche, Samumed, Siemens Healthineers, Triplet Therapeutics, and Wave. HZ has given lectures sponsored by Alzecure, BioArctic, Biogen, Cellectricon, Fujirebio, Lilly, Novo Nordisk, Roche, and WebMD, and is a co-founder of Brain Biomarker Solutions in Gothenburg AB (BBS), which is a part of the GU Ventures Incubator Program (outside this submitted work). KB has served as a consultant and on advisory boards for Abbvie, AC Immune, ALZPath, AriBio, Beckman-Coulter, BioArctic, Biogen, Eisai, Lilly, Moleac Pte. Ltd, Neurimmune, Novartis, Ono Pharma, Prothena, Quanterix, Roche Diagnostics, Sunbird Bio, Sanofi and Siemens Healthineers. KB served on data monitoring committees for Julius Clinical and Novartis; has given lectures, produced educational materials and participated in educational programs for AC Immune, Biogen, Celdara Medical, Eisai and Roche Diagnostics; and is a co-founder of Brain Biomarker Solutions in Gothenburg AB (BBS), which is a part of the GU Ventures Incubator Program, outside of the work presented in this paper.
Data availability statement
Restrictions apply to the availability of sensitive personal data to protect individual privacy of participants in the study. The corresponding author will on request consider conditions under which access to original data may be provided. Data are located in controlled access data storage at Gothenburg University.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
