Abstract
Background
Mitochondrial dysfunction is an important pathogenic factor in Alzheimer´s disease (AD) progression. Most studies analysed disturbances in the mitochondrial metabolism and oxidative stress or focussed on mitochondrial dynamics such as mitochondrial trafficking, fusion-fission and mitophagy.
Objective
Very limited data exist regarding changes in the mitochondrial numerical density at different levels of AD neuropathologic changes (ADNC) in human brains.
Methods
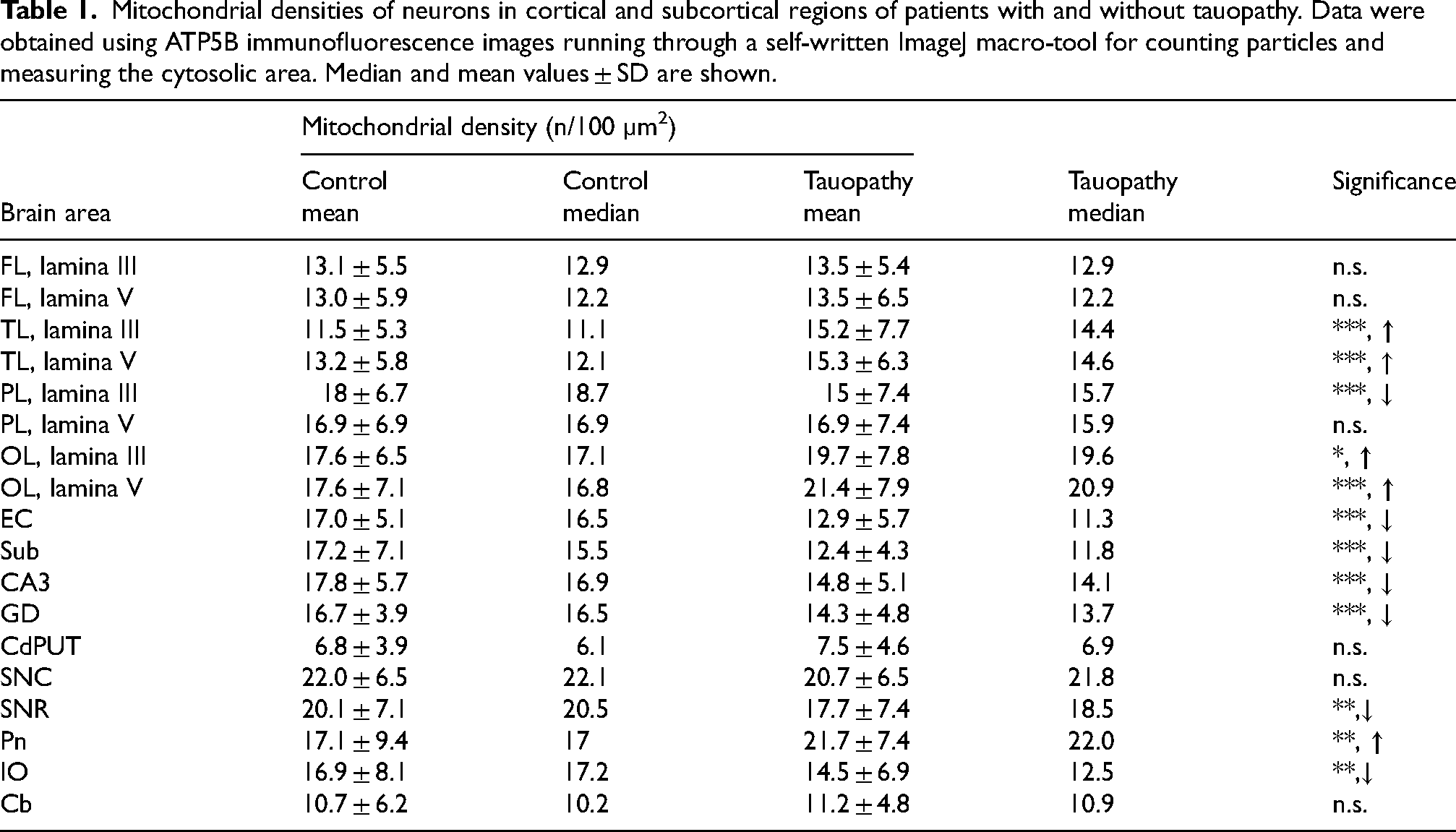
Mitochondrial numerical densities were analysed by morphometry using the marker protein ATP5B in sections of 13 brain areas of 8 patients with either low, mid or high ADNC, 6 patients with tauopathy and 10 control patients. Patient samples were classified according to the ABC score.
Results
In comparison to control patients, we detected increases in mitochondrial densities at low (not in all cases), mid and high ADNC in neurons of the frontal (25%) and temporal (11%) neocortices, pontine nuclei (30%) and Purkinje neurons of the cerebellum (30%). Contrarily, mitochondrial densities decreased by 20% in hippocampal neurons of the entorhinal cortex and CA3 region at mid and high ADNC. Only minor changes occurred in other brain regions investigated (e.g., parietal and occipital neocortices, inferior olive, substantia nigra, striatum). In tauopathy patients, changes in mitochondrial densities were comparable to those in AD patients, except for a stronger decrease in the entorhinal cortex (40%) and a greater increase in the temporal neocortex (30%).
Conclusions
In the neocortex, primarily affected by extracellular amyloid-β (Aβ) deposits, mitochondrial densities in neurons increased, whereas they decreased in the hippocampus, at first enriched in intracellular neurofibrillary tangles.
Keywords
Introduction
Mitochondrial dysfunction is involved in the pathogenesis of many diseases, including neurodegeneration as found in patients with Alzheimer's disease (AD). Hallmarks of AD are extracellular amyloid-β (Aβ) deposits and intracellular neurofibrillary tangles (NFTs) which both increase during the disease progression in a region-specific manner. While accumulation of Aβ deposits starts in the frontal and temporal neocortex, 1 spreading of NFTs is initiated in the entorhinal cortex, followed by the CA1 region, subiculum, CA3 region and dentate gyrus of the hippocampus. 2 A positive loop between Aβ and NFT aggregation is thought to accelerate AD neuropathologic changes (ADNC). Whether mitochondrial dysfunction occurs at early or late stages in AD pathogenesis is still unclear and debated in two different, so-called primary and secondary mitochondrial cascade theories. 3 Experiments (mostly using animal models) focused on specific changes of the mitochondrial function but less is known on changes of the organelle itself (e.g., its morphology or density) in AD and control human brains. Only a few studies exist, in which their morphology and intraneuronal localization, or the protein amount of mitochondrial fractions, mitochondrial DNA (mtDNA) copy number or the organelle density have been analyzed in distinct human brain regions. 4 Regarding changes in their morphology and localization, oversized mitochondria in length and size with broken cristae have been described in pyramidal neurons of hippocampal autopsy material from AD patients. 5 In addition, an accumulation of mitochondria in the perinuclear space has been detected in fibroblasts derived from AD patients,6,7 and in hippocampal pyramidal neurons from an AD patient with a presenilin-1 mutation. 8
Two electron microscopic studies further investigated the subcellular presence of mitochondria in AD human cortical biopsies.9,10 In AD compared to control human brains, the percentage of pre- and postsynaptic sides of synapses containing mitochondria 9 and the number of mitochondria in the neuronal soma have been shown to be significantly reduced by half. 10 The reduced number in the neuronal soma in AD patients was accompanied by a 2-fold increase in mitochondrial size with a 2-fold higher percentage of damaged organelles compared to controls. 9 However, in the study of Wang et al., 10 the number of mitochondria was not related to area and thus does not reflect their density. Such an increase in mitochondrial size accompanied by a reduced number pinpoints to a disturbed axonal transport and a dysfunction in mitochondrial dynamics in favor of a reduced fission. The latter hypothesis is supported by findings of decreases in dynamin-like protein-1 (DLP-1) in some,6,7,11,12 but not all studies. 13 Indeed, also fragmented mitochondria, indicating excessive fission have been described in the brain of AD patients. 14 Such controversial data in different studies might be due to the variety of the patient samples (e.g., brain regions, neuropathological stages or type of AD (sporadic or rapid developing forms of the disease). 14 Measurement of the mtDNA copy number is also a possible method to quantify mitochondria. Thubron et al. 15 analyzed the amount of mtDNA and observed a decrease in the mtDNA level in the parietal, but not in the frontal cortex and cerebellum of patients with AD compared to controls. However, in these studies, mtDNA content per tissue weight or the total number of mitochondria in the neuronal soma or synapses with no relation to area, and thus not mitochondrial density was measured. Interestingly, semi-quantitative western blot analyses of mitochondrial fractions using mitochondrial marker proteins such as the non-selective voltage-gated ion channel (VDAC), 16 the mitochondrial import receptor, subunit 20 (TOM20) 11 and cytochrome c oxidase subunit IV (COX4) 11 revealed no differences between human control and AD neocortices. In only one other study, mitochondrial densities were measured in different regions of a human brain with negative neuropathologic alterations by mitotracker staining of the frozen postmortem tissue. 17 Mitochondrial density was calculated by measuring the amount of mitotracker fluorescence in defined voxel-sized homogenates; higher values of mitochondrial densities were measured in the frontal and temporal neocortices than in the hippocampus. 17
In the present study, morphometric analysis of mitochondrial density (using ATP5B as a mitochondrial marker protein) was performed in 13 distinct brain regions of formalin-fixed paraffin-embedded (FFPE) postmortem samples from human patients with different ADNC or patients with tauopathy compared to control patients. Density values reflect the number of mitochondria (particles) per cytosolic area of the neuronal soma.
Methods
Postmortem human brain material
Twenty-four human brains with neuropathology diagnosis of AD, 6 human brains with tauopathy and 10 age- and gender matched control human brains were included in the study. We used formalin-fixed brain samples with a postmortem interval of 1 to 5 days. In the Supplemental Table 1, the data of each patient are listed including age, gender, ABC score and whether they had diabetes type II or not. Since we used subsequent serial sections of paraffin blocks of human brain regions that we have analyzed for peroxisome densities in our previous study, 18 details on the histopathological evaluation for AD classification and details on comorbidities and on the causes of death have been described in this publication. 18 The following regions were dissected out of the brains and were later embedded in paraffin in a defined stereological pattern: the entorhinal cortex (EC), subiculum (Sub), CA3 region (CA3), and dentate gyrus (GD) of the hippocampal formation, defined areas of the frontal (FL), temporal (TL), parietal (PL), and occipital (OL) lobes as well as of the striatum (caudate-putamen, CdPut), the substantia nigra, pars compacta (SNC) and pars reticularis (SNR), pontine nuclei (Pn), the inferior olive (IO), and the cerebellum (Cb). Anatomical nomenclature and abbreviations are based on the “Atlas of the Human Brain” (https://www.thehumanbrain.info/database/nomenclature.php).
All patients relatives signed a written informed consent and agreement that the brains would enter the brain donation bank of the Institute for Neuropathology to be used for diagnosis, research and teaching. The project has been approved by the ethics committee of the Justus-Liebig University (AZ 07/09).
Immunofluorescence staining to characterize the density of mitochondria in human brain tissue
Two µm FFPE sections of the human brain samples were used for immunofluorescence stainings for detection of the mitochondrial marker protein ATP5B (ATP synthase, F1 subunit beta), Aβ and paired helical filament (PHF) tau. Prior to the staining, the paraffin was removed from the sections by incubating the slides in xylene followed by rehydration through a descending graded alcohol series. Antigen retrieval was performed by microwave irradiation (3 × 5 min) at 900 Watt in 10 mM citrate buffer (pH 6.0) and subsequent cooling to room temperature for 30 min. Non-specific binding sites were blocked with 4% bovine serum albumin in phosphate-buffered saline (PBS) with 0.05% Tween 20 for 2 h. Thereafter, the sections were incubated with the primary antibodies diluted in 1% bovine serum albumin in PBS with 0.05% Tween 20 overnight in a moist chamber at room temperature. The following primary antibodies were used: rabbit anti-ATP5B IgG (1:2.000, Sigma-Aldrich Chemie GmbH, HPA001520, Steinheim, Germany), mouse anti-Aβ IgG (1:5.000, 4G8, Covance, SIG-39220, through antibodies-online GmbH, Aachen, Germany) and mouse anti-PHF tau IgG (1:2.000, AT8, Invitrogen, MN1026, Schwerte, Germany). The next day, sections were washed with PBS and donkey anti-rabbit IgG coupled with Alexa488 (1:300, Invitrogen Laboratories, A11070, Schwerte, Germany) or donkey anti-mouse IgG coupled with Alexa555 (1:300, Invitrogen Laboratories, A31570, Schwerte, Germany) were added for 2 h. Thereafter, sections were washed with PBS and nuclei were counterstained with DAPI in a 1:750 dilution in PBS from a stock of 1 mg DAPI/ml. Slides were mounted with a mixture (2:1) of Mowiol® 4–88 mounting medium and n-propylgallate as anti-fading agent.
Analysis of the mitochondrial density in the cytoplasm of the neurons
Laser scanning microscopy was performed using the Leica Confocal Laser Scanning Microscope TCS SP2 (Leica, Bensheim, Germany) equipped with a 63× oil-immersion objective and Leica Confocal Software for image acquisition. For comparability of the data between the different patient samples, all images were taken at the same acquisition settings for the detection of ATP5B. For each brain region of each patient, up to 20 images were taken from double-fluorescence images for ATP5B combined with Aβ (4G8 antibody) and double-fluorescence images for ATP5B combined with NFTs (AT8 antibody). Fluorescence images were stored in TIF format. For the analysis of the mitochondrial densities, original TIF data were imported as an unedited green format into the open-source Java-based image processing program ImageJ. In addition, original TIF data was imported into Photoshop CS3 for the preparation of representative micrographs. Mitochondrial density represents the number of counted particles within the cytosolic area of neurons. Discrimination of mitochondria from neighboring lipofuscin granules, which all exhibit a strong autofluorescence, was achieved using several intermediate steps to isolate mitochondria-sized particles and by setting of the correct particle size for mitochondria (ranging from 4–50 pixel diameter; their average size is 10–30 pixel diameter for pyramidal neurons of the frontal neocortex). Lipofuscin granules are of much bigger size (>51 pixel diameter, their average size is 120–300 pixel diameter for pyramidal neurons of the frontal neocortex). However, the distribution pattern and the sizes of lipofuscin granules are variable depending on the brain region. To avoid counting of even rare small lipofuscine granules, we took advantage of the fact that most of the lipofuscin granules are in range of the background fluorescence which was subtracted using the automatic “Yen dark” threshold macro function.19,20 For an automatic analysis of a series of images, an ImageJ macro tool including these pre-processing steps was established (Supplemental Figure 1A). Steps of this analysis starting with the opening of the original image (Supplemental Figure 1B) and ending with the counting of ATP5B-positive particles are shown in Supplemental Figure 1C, 1D, and 1F. In Supplemental Figure 1C-F, we demonstrate that lipofuscin granules are of bigger size (C, E, G) and that their autofluorescence is in the range of the background fluorescence (D, F). All quantitative results are presented as box plots overlaid with dot plots, mean values are shown as a black square in the center of the box, median values as a crossbar.
Mitochondrial densities of neurons in cortical and subcortical regions of patients with and without tauopathy. Data were obtained using ATP5B immunofluorescence images running through a self-written ImageJ macro-tool for counting particles and measuring the cytosolic area. Median and mean values ± SD are shown.
Statistics
First, the Shapiro-Wilk-test was used to test data normality. In case of a normal data distribution, homogeneity of variances was analyzed by Levene test followed by one-way analysis of variance (ANOVA-1) and post-hoc pairwise t-test (homogeneous variances) or by Kruskal-Wallis test and post-hoc Dunn test (non-homogeneous variances) to determine the significance of differences between the groups. In case of non-normality of the data, significance of differences between the groups was evaluated by Kruskal-Wallis test and post-hoc Dunn test. When we compared two patient groups only (e.g., controls versus tauopathy), significance of difference was analyzed by Wilcoxon test. For statistical analysis together with the graphics, we used an R-based self-written program. In the box plots, the level of significance is shown in all figures and tables as follows: *p < 0.05; **p < 0.01; ***p < 0.001).
Results
Brain-region specific differences in mitochondrial numerical abundances in control human brains
In control human patients, mitochondrial densities in the neuronal somata varied between different brain regions. In the neocortex, median densities between 10.8/100 µm2 and 12.1/100 µm2 were found in laminae III and V of the frontal and temporal lobes. Higher values between 15.9/100 µm2 and 17.7/100 µm2 were noted in the parietal and occipital cortices (see blue boxes in Figure 1). In all four neocortical regions of control patients, no differences were detected comparing the mitochondrial densities of pyramidal neurons of the laminae III and V. The mitochondrial densities of neurons of the occipital and parietal cortices were similar as found in the hippocampal formation (entorhinal cortex, subiculum, CA3 region, dentate gyrus) with values between 14.3/100 µm2 and 15.7/100 µm2 (see blue boxes in Figures 1 and 2). Neurons of the pontine nuclei and inferior olive of the medulla oblongata contain 17.2 mitochondria/100 µm2, and Purkinje neurons of the cerebellum 10.2 mitochondria/100 µm2. Interestingly, very low values of mitochondrial densities were found in the striatum (6.1/100 µm2) in contrast to the high values in the substantia nigra (20.6/100 µm2 in the pars reticularis and 22.1/100 µm2 in the pars compacta, respectively).
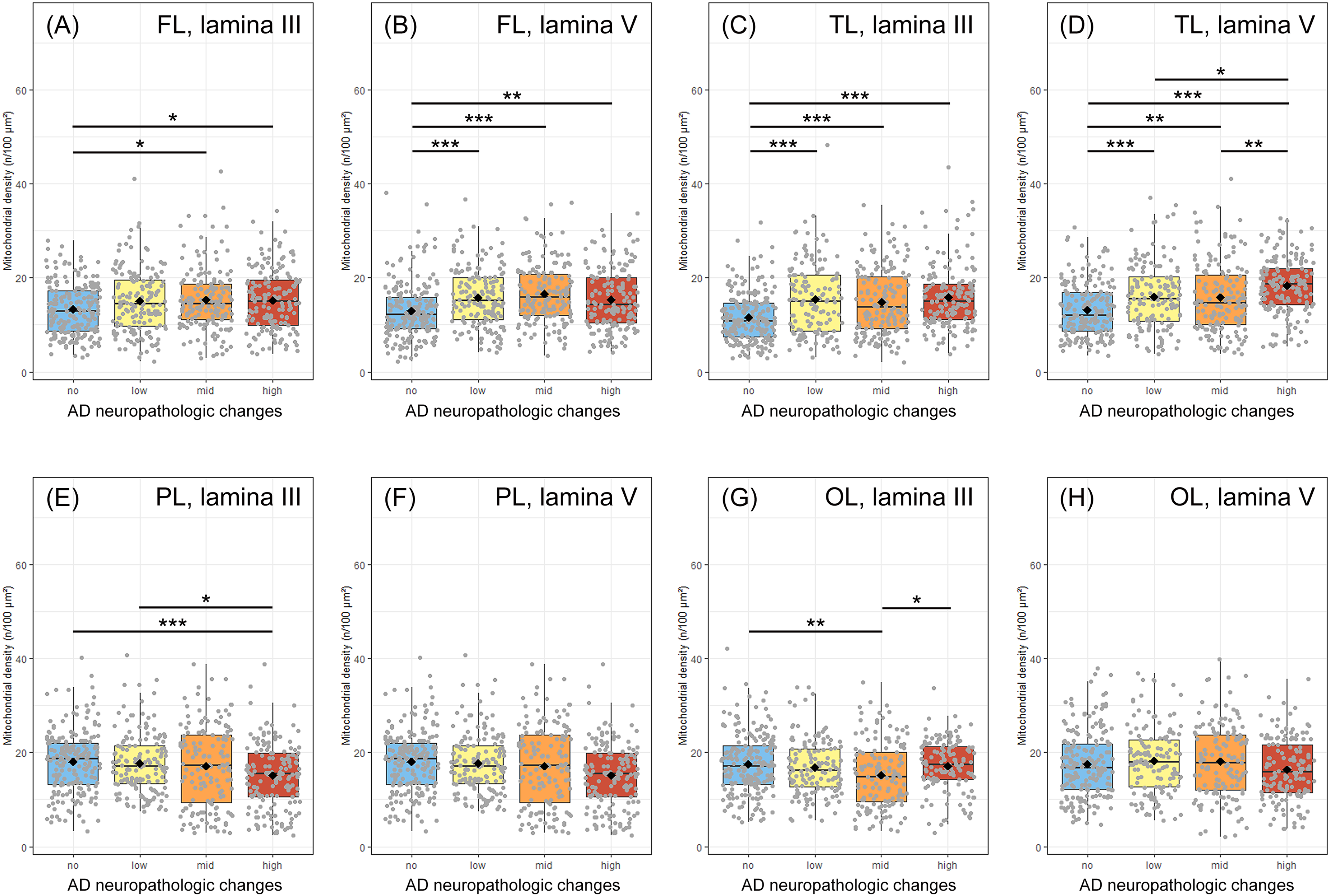
Increase in the neuronal mitochondrial densities in laminae III and V of the frontal (FL, A, B) and temporal (TL, C, D) lobes of patients with mid and high ADNC compared to the respective controls. Only minor changes in mitochondrial densities were observed in the parietal (PL, E, F) and occipital (OL, G, H) lobes. Data were obtained using ATP5B immunofluorescence images running through a self-written ImageJ macro-tool for counting particles and measuring the cytosolic area. Twenty individual neurons were analyzed from each patient and plotted as a point on the graph. Crossbar = median value, black diamond in the box center = mean value, vertical lines above and below each box = SD values. (Color figure available online).
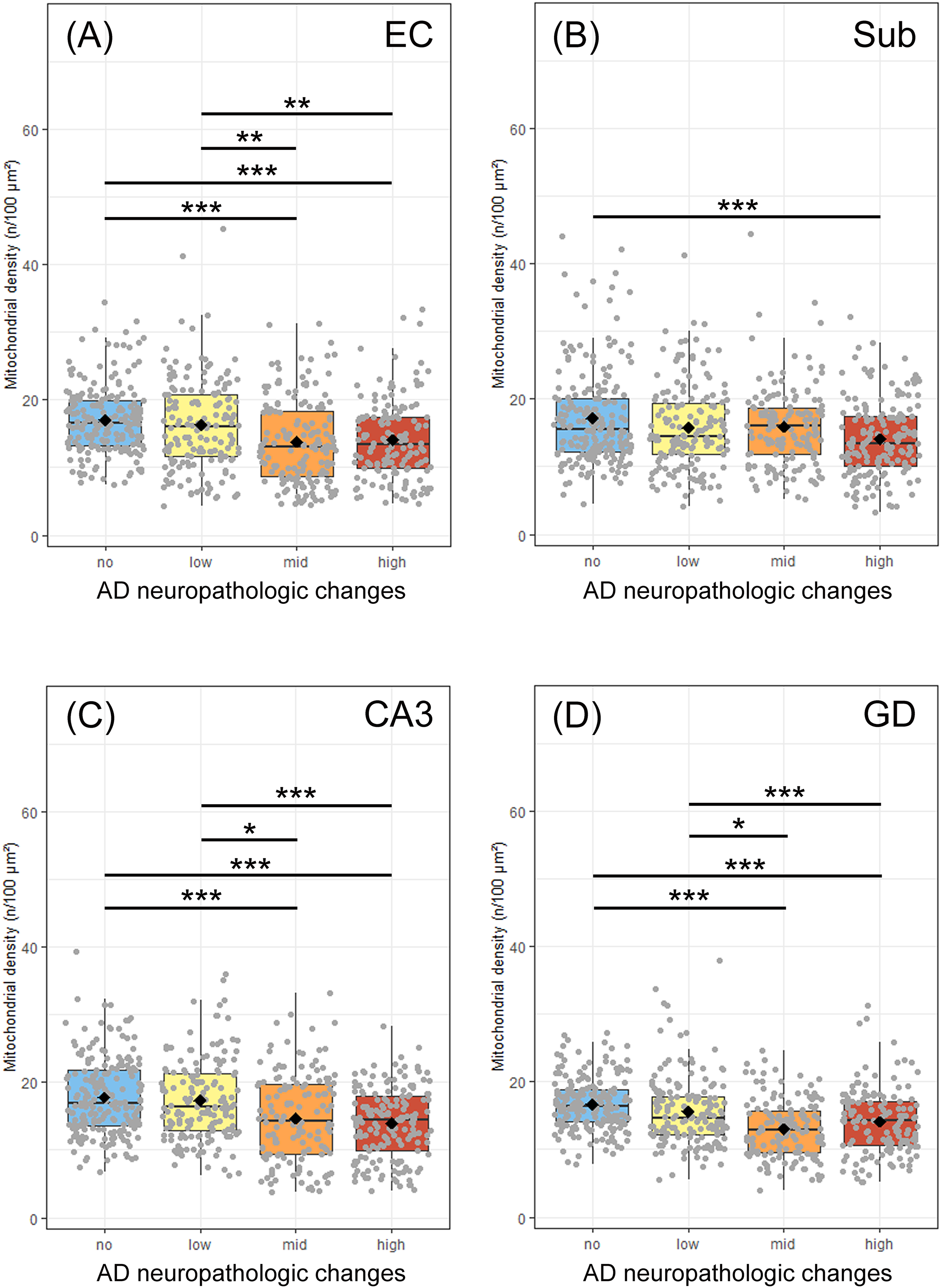
Mitochondrial densities of neurons in the entorhinal cortex (A), CA3 region (C) and gyrus dentatus (D), but not in the subiculum (B) were lower in patients at mid and high ADNC in comparison to control ones. Data were obtained using ATP5B immunofluorescence images running through a self-written ImageJ macro-tool for counting particles and measuring the cytosolic area. Twenty individual neurons were analysed from each patient and plotted as a point on the graph. Crossbar = median value, black diamond in the box center = mean value, vertical lines above and below each box = SD values. (Color figure available online).
Increases in mitochondrial densities in the frontal and temporal neocortices of patients at low to high ADNC compared to control patients
Neuropathology in AD follows a spatial and temporal hierarchy. In case of the neocortex, formation of extracellular Aβ deposits occurs at early and that of intracellular NFTs at very late time points of AD (reviewed by 21 ). As shown by PET image analyses of 85 AD patients, 1 the accumulation of Aβ appears chronologically in the basal part of temporal cortex (stage I), in the anterior cingulate cortex (stage II) and in wide parts of frontal, temporal and parietal association cortices (stages III-IV) in 98% of all patients. Even at stage IV, less Aβ was detected in parietal and occipital neocortices and the striatum; the thalamus was free of Aβ. 1
In our study, mitochondrial densities increased in pyramidal neurons of the frontal and temporal cortices of patients with low, mid and high ADNC compared to controls (Figures 1 and 3). Only in the large pyramidal neurons of the lamina V of the temporal cortex, increases in mitochondrial density differ between low/mid and high ADNC. Interestingly, the frontal and temporal cortices are the regions containing very low levels of mitochondrial densities in control patients.
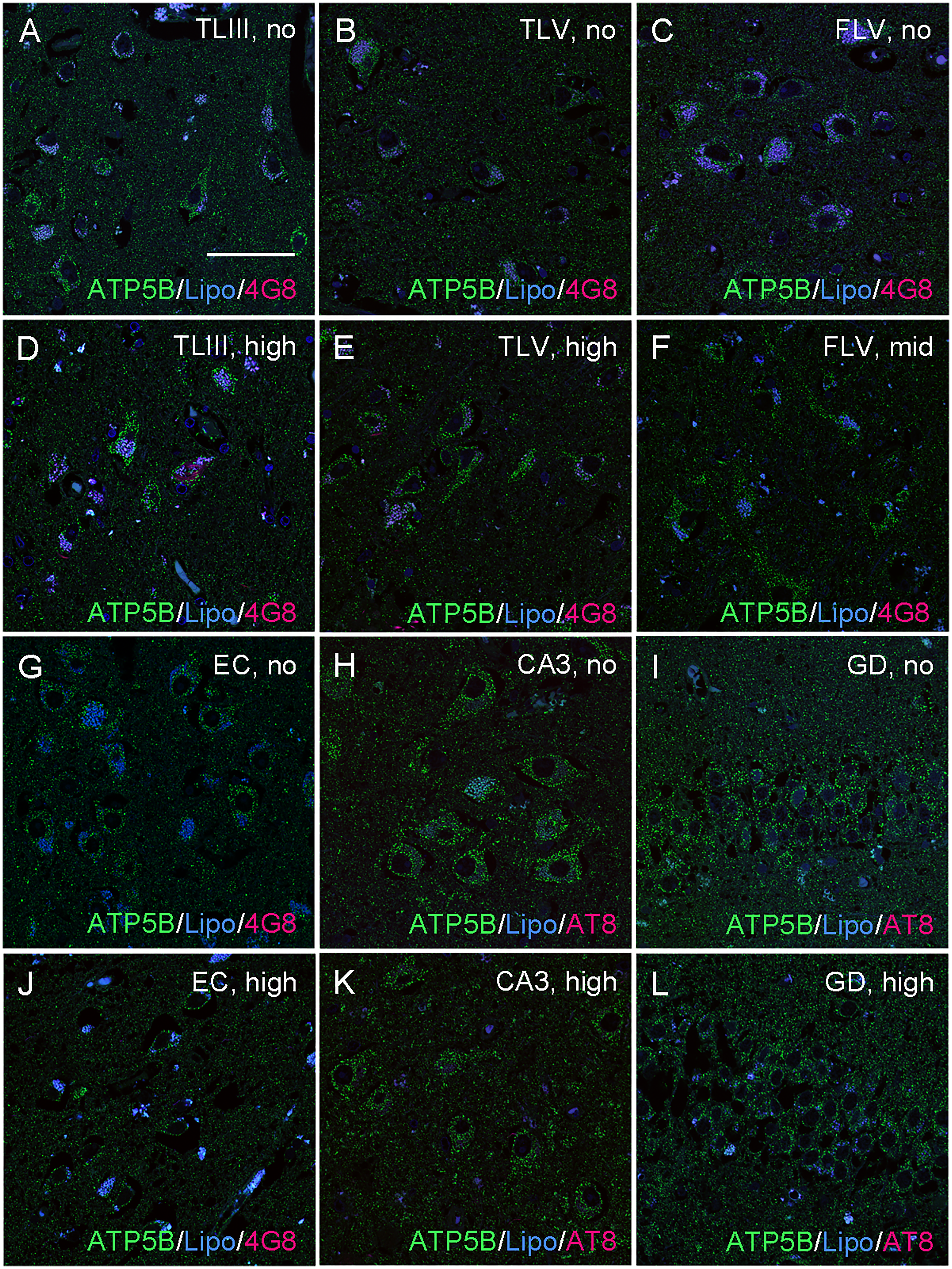
Mitochondrial densities increased in neurons of patients with mid and high ADNC in the temporal lobe, laminae III (TLIII; D) and V (TLV; E) as well as in the frontal lobe, lamina V (FLV; F) when compared to the respective controls patients (A-C). Mitochondrial densities decreased in neurons of patients with mid and high ADNC in the entorhinal cortex (EC; J), CA3 region (CA3; K) and dentate gyrus (GD; L) when compared to the respective control patients (G-I). Representative photomicrographs of immunostainings for the mitochondrial marker ATP5B (green), Aβ (4G8), NFTs (AT8) and autofluorescent lipofuscin granules (Lipo) are shown. Scale bar = 50 μm. (Color figure available online).
Mitochondrial densities decreased in the entorhinal cortex and CA3 region of patients with mid and high ADNC compared to control patients
In the hippocampal formation, aggregation of NFTs is the first major hallmark of AD. In the human brain, NFTs appear inside the neuronal perikarya and proximal processes of the entorhinal cortex (phase 1), CA1 region (phase 2), subiculum, thalamus (phase 3), CA3/4 region and putamen (phase 4) and finally in layer III (phase 5) and V (phase 6) of the neocortex.21,22 Aβ deposition in the hippocampus follows the same spatial chronology, but occurs at later time points in the presence of pre-existing intracellular NFTs.
When we analyzed mitochondrial densities in the hippocampus in this study, we observed decreases between 14% and 20% in the CA3 region, the dentate gyrus and the entorhinal cortex; but no changes were detected in the subiculum (Figures 2 and 3).
Mitochondrial densities are differentially altered in subcortical regions
In case of the subcortical regions, alterations of mitochondrial densities were especially detected at mid or even low, but not at high ADNC. The highest increases in mitochondrial densities of 28% and 23% at mid ADNC were measured in neurons of the pontine nuclei and Purkinje neurons of the cerebellum, smaller increases of 10% at low ADNC in the substantia nigra, pars compacta and pars reticularis (Figures 4 and 5). In the inferior olive, mitochondrial density decreased by 20% and no changes were measured in the striatum (Figures 4 and 5).
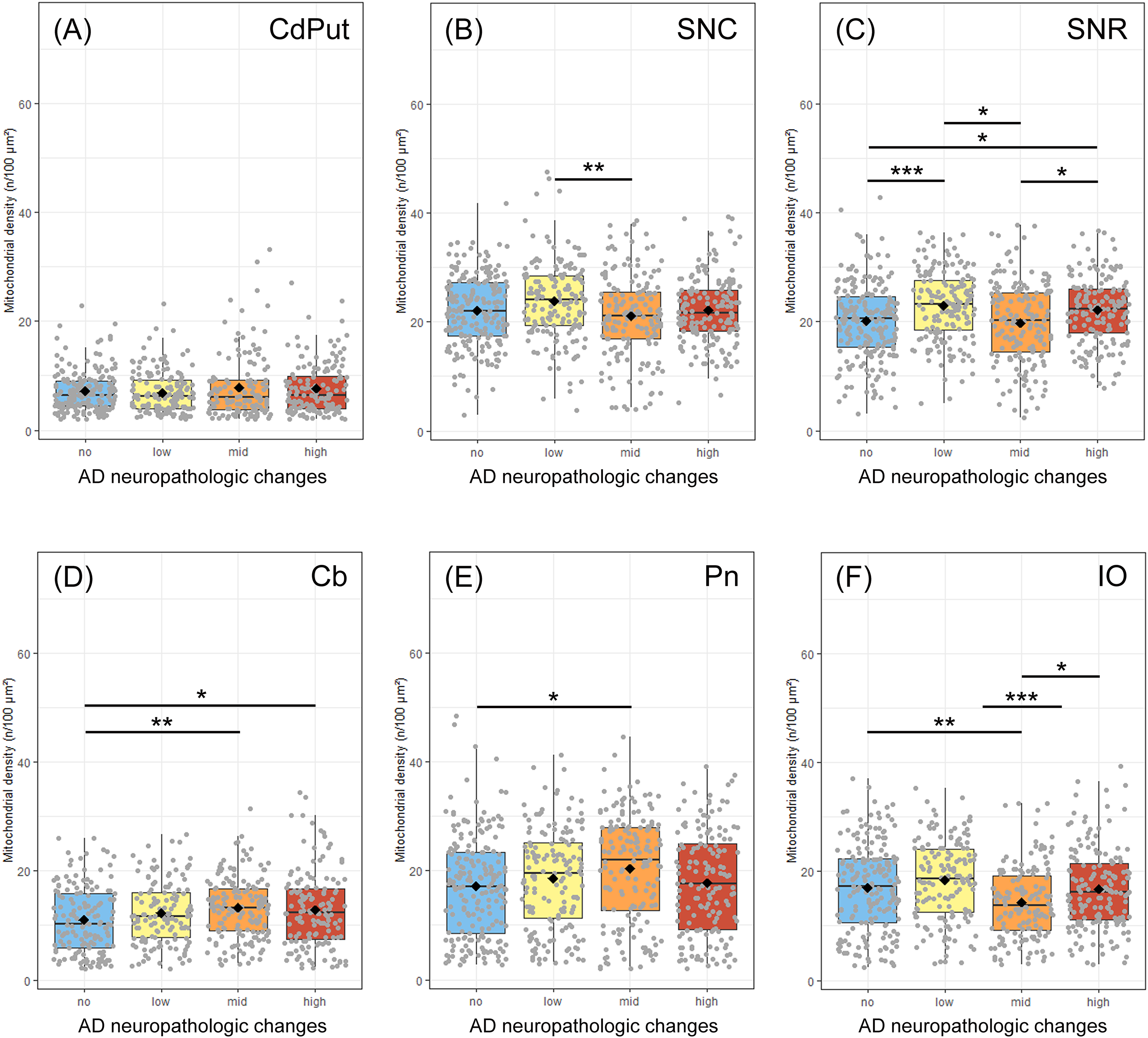
Mitochondrial densities of neurons in subcortical areas such as the striatum (CdPut; A), the substantia nigra, pars compacta (SNC; B) and reticularis (SNR; C), the Purkinje layer of the cerebellum (Cb; D), pontine nuclei (Pn; E) and inferior olive (IO; F) in patients with no, low, mid and high ADNC. Data were obtained using ATP5B immunofluorescence images running through a self-written ImageJ macro-tool for counting particles and measuring the cytosolic area. Twenty individual neurons were analysed from each patient and plotted as a point on the graph. Crossbar = median value, black diamond in the box center = mean value, vertical lines above and below each box = SD values. (Color figure available online).
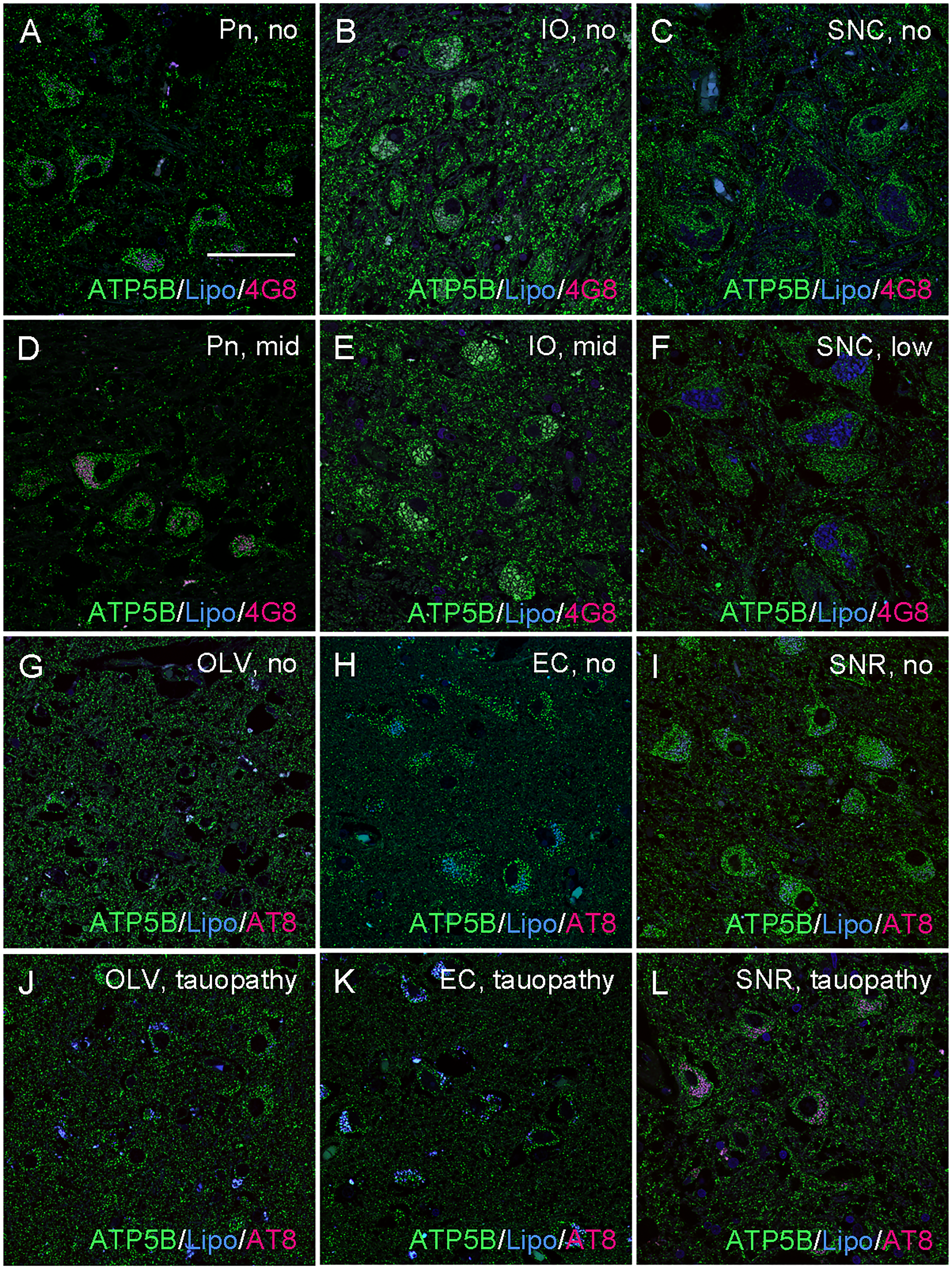
Mitochondrial densities were higher in neurons of patients with low and mid ADNC in the pontine nuclei (Pn, A), inferior olive (IO; B) and the substantia nigra, pars compacta (SNC; C) compared to the respective control patients (D-F). Mitochondrial densities were lower in neurons of patients with tauopathy in the occipital lobe, lamina V (OLV; J) entorhinal cortex (EC; K) and substantia nigra, pars reticularis (SNR; L) compared to the respective control patients (G-I). Representative photomicrographs of immunostainings for the mitochondrial marker ATP5B (green), Aβ (4G8), NFTs (AT8) and autofluorescent lipofuscin granules (Lipo) are shown. Scale bar=50 μm. (Color figure available online).
Mitochondrial densities in patients with tauopathy
Patients diagnosed for tauopathy contain high amounts of NFTs especially with first deposits in the parietal, temporal, and occipital association cortices and later in all CA fields of the hippocampal formation; all of these brains areas were free of Aβ deposits. Hence, the tauopathy patients analyzed in this study show scores of A0 and C0 (Supplemental Table 1). Mitochondrial densities were higher in the frontal, temporal, parietal and occipital cortices in tauopathy compared to control patients (Table 1). Similarly, but to a higher extent in comparison to AD, mitochondrial densities were reduced in the entorhinal cortex, subiculum, CA3 region and dentate gyrus of patients with tauopathy compared to control patients (Table 1, Figure 5). Moreover, such as in AD, neuronal mitochondrial densities were higher in pontine nuclei and lower in the inferior olive in tauopathy compared to control patients (Table 1). No differences were found for the striatum, substantia nigra and cerebellum (Table 1).
Next, we correlated the mitochondrial density with the number of NFTs independent of the ABC score. In all neocortical and hippocampal brain regions, there was no significant correlation between the number of NFTs and mitochondrial densities (Supplemental Figure 2). We also compared mitochondrial densities in otherwise healthy (no AD) control patients with and without diabetes type II. In non-diabetic control patients, densities of 14.8 ± 4.5 and 15.9. ±4.2 mitochondria/100 µm2 were quantified in the frontal neocortex and hippocampal CA3 region, respectively. In the diabetic compared to the non-diabetic control patient group, lower values of 9.8 ± 5.8 mitochondria/100 µm2 were measured in the frontal neocortex and higher ones of 19.9 ± 6.5 mitochondria/100 µm2 in the CA3 region. Within our group of 24 AD patients, a low number of only 5 patients suffer from diabetes (Supplemental Table 1). Therefore, we did not analyze the influence of diabetes as comorbidity factor for AD in order to avoid overinterpretation of our data.
Discussion
The important role of mitochondria for AD pathogenesis is increasingly recognized 23 and a dysfunction of this organelle was supposed to account for the loss of synaptic plasticity and transmission, finally ending in AD symptoms such as cognitive decline, memory deficits, and neurodegeneration. To the best of our knowledge, no elaborate morphometric study on the numerical density of mitochondria of many different selected areas of human brain exists. In this study, we compared mitochondrial numerical densities in the different cortical (neocortex, hippocampal formation, striatum) and subcortical (substantia nigra, cerebellum, pontine nuclei, inferior olive) brain regions of neuropathological well characterized patients with no, low, mid and high ADNC as well as of patients with tauopathy compared to controls using the nuclear-encoded protein ATP5B as mitochondrial marker (Figure 6).
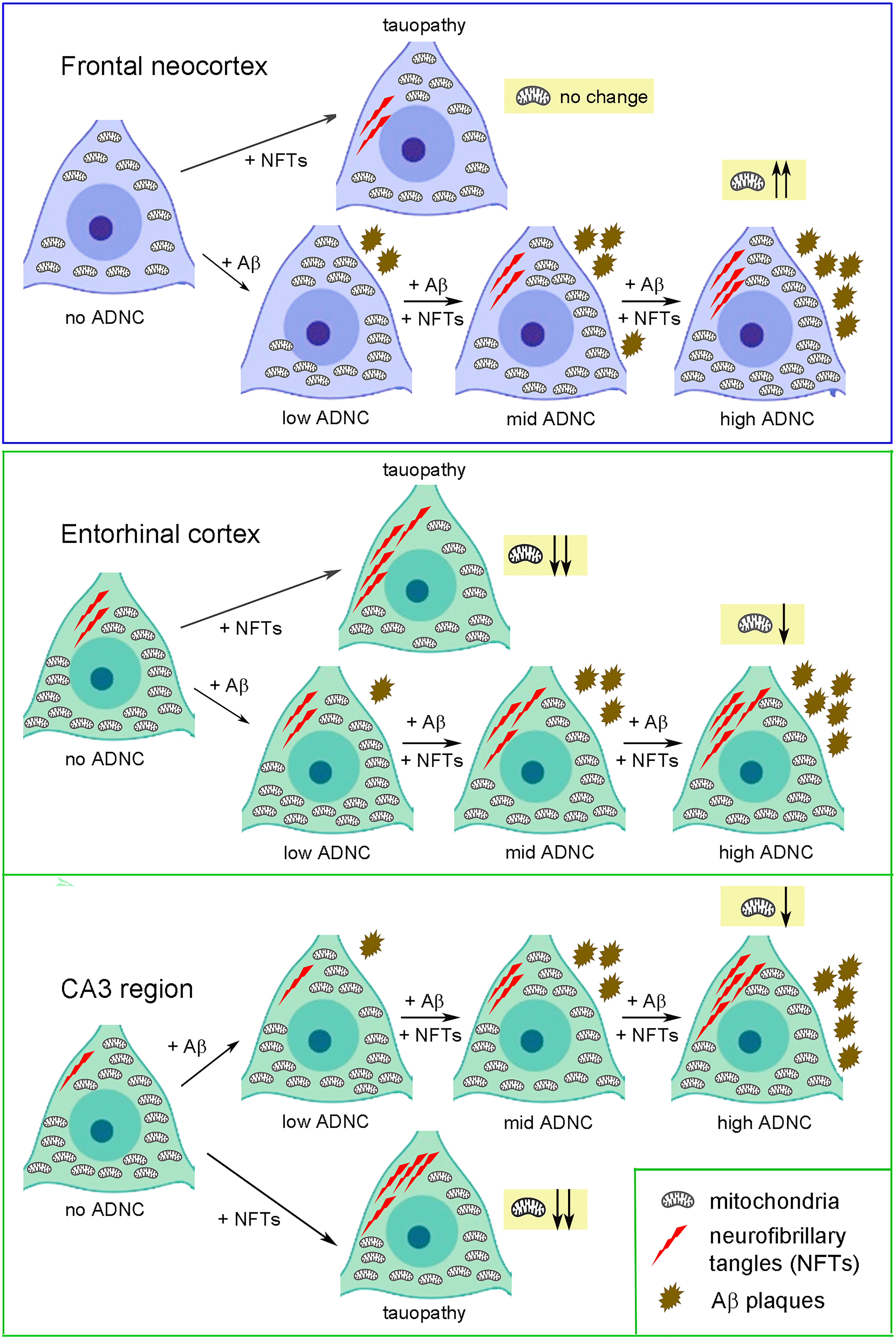
Summary of AD-induced changes in mitochondrial densities of pyramidal neurons of the neocortex and hippocampal formation. Neuronal soma of the pyramidal neurons of the frontal and temporal lobes are colored in blue; those of the entorhinal cortex and CA3 region appear in green. Low, mid and high ADNC are represented by increasing amounts of Aβ (brown stars) and/or NFTs (red arrows) or of NFTs only in case of tauopathy. In the graphic, alterations in mitochondrial (black tubes) densities are visible; Figure was created with BioRender.com. Balsliemke e. (Color figure available online).
Region-specific changes in mitochondrial densities in human brain during AD progression
As an interesting finding, we observed increases in the mitochondrial numerical densities in the frontal and temporal neocortices (Figures 1 and 3) and decreases thereof in the entorhinal cortex and CA3 region of the hippocampus (Figures 2 and 3) in patients with low (not in all cases), mid and high ADNC compared to controls. Regarding disturbances of mitochondrial dynamics in AD patients either excessive (reduced levels of dynamin-like GTPase OPA1, mitofusins 1 and 2) or reduced fission (reduced levels of DLP-1) has been detected in previous studies (reviewed by14,24). It was suspected by different groups that controversial findings were due to the variety of the sample material. 14 The sample material varied in terms of the brain region, analytical method, as well as of the disease stage and types of AD. Since in our study opposite alterations in mitochondrial densities were found in the neocortex and hippocampus of the same patients, we suggest that the variability may also depend on the brain-region analyzed. Moreover, we hypothesize that the predominant presence of either Aβ in the neocortex or PHF tau in the hippocampus during early stages of the disease might have caused these observed regional differences. Recently, Xiyang et al. 11 analyzed the temporal neocortex of control in comparison to rapidly progressive or typical AD patients. The three patient groups differed in the level of Aβ (low levels in control, high levels in rapidly progressive and typical AD patients) and PHF tau (low levels in control and rapidly progressive AD patients, high levels in typical AD patients). 11 Even though the protein levels of COX4 and TOM20 supposed to reflect mitochondrial quantity, both proteins remained unchanged in all 3 groups, whereas dynamin-like GTPase OPA1 and mitofusins 1 and 2 were reduced in rpAD patients 11 indicating an excessive fission and thus a higher number of smaller mitochondria in the presence of Aβ and absence of PHF tau. These results are consistent with our findings of increases in mitochondrial densities in the frontal and temporal neocortices with high Aβ, where PHF tau appeared only at late stages.
Many previous studies discussing theories on AD pathogenesis based on animal models that might not necessarily reflect the “real” human AD pathomechanism, especially the involvement of Aβ, PHF tau, and mitochondrial dysfunction in this complex process. In spite of this and due to the yet not satisfying drug therapy, mitochondria are more and more considered as a possible target for AD.25,26 Dietary strategies with a positive impact on mitochondrial bioenergetics and function include caloric restriction, a ketogenic diet and supplementation with carnitine, riboflavin, creatine and ubiquinone. 26 Drugs mostly aimed to inhibit mitochondrial reactive oxygen species (ROS) production by inhibiting (i) mitochondrial complex I (metformin, capsaicin, coenzym Q2, mucidine, stigmatelline, blood-brain barrier permeable antioxidants), (ii) mitochondrial peroxidation of lipids (MitoVitE) and cardiolipin (peptide SS-31), and (iii) by enhancing the activity of mitochondrial antioxidant enzymes such as NADPH oxidase 2 (MitoApocyanin) or superoxide dismutase 2 (MitoTEMPO). 25 Other drugs which are considered as therapy for AD affect mitochondrial dynamics. They either regulate mitochondrial fusion (mitochondrial division inhibitor-1), increase DLP-1 (ceramides), inhibit fission (ceramides, SH-BC-893, PAQ-22, DDQ, P110) or activate mitophagy (UMI-77, logamin).25,26 In animal models, gene therapy using adeno-associated virus vectors has been performed either (i) to restore NADPH dehydrogenase (which is reduced in AD due to mtDNA mutation), (ii) to increase mitochondrial antioxidant capacity, e.g., thioredoxin (AAV-Trx-1) or (iii) to affect mitochondrial dynamics (AAV9- PGC1a, AAV9-CEND1, AAV-PINK1). 26
ATP5B as a marker protein for mitochondria
An ideal mitochondrial marker should meet the following requirements: (i) exclusive expression in mitochondria, (ii) high abundance to ensure complete detection of all organelles, (iii) stable, metabolic-independent expression and (iv) constitutive and equal expression in different tissues or cells. 27 Common commercially available mitochondrial markers are proteins of the inner and outer mitochondrial membranes and of the electron transport chain such as TIM23, TOM20, TOM70, heat shock protein 60, COX4, different subunits of the ATP 5 synthase, VDAC, mitofusins 1 and 2, mitofilin and ubiquinol-cytochrome c reductase complex 1 (UQCRC1) (https://www.bio-techne.com/research-areas/cell-biology/cell-markers/mitochondrial-markers).27 All these proteins are nuclear-encoded protein and thus independent of the heteroplasmic mtDNA mutations often found in the brains of AD (reviewed by24,28), but also in aged patients. 29 Such mtDNA mutations occur mostly in the regulatory D loop region, leading to a reduced mtDNA copy number and a reduced level of proteins encoded by mtDNA such as COX1 and NADPH dehydrogenase. 30 In our study, we used an antibody against ATP synthase F1 subunit beta (ATP5B), because earlier studies of our group demonstrated that the ATP5B protein is highly abundant, for example in the lung, 31 and is also a useful mitochondrial marker for morphometric studies in the brain. 32 Indeed, protein expression profiles of all the above mentioned mitochondrial marker proteins revealed the highest abundance for COX4 and ATP5B with 400 normalized transcripts per million (nTPM) (https://www.proteinatlas.org/ENSG00000110955-ATP5F1B/brain). All other proteins were between 100 and 300 nTPM (ATP5A, ATP5C, TOM20, VDAC, UQCRC1) or even below 100 nTPM (mitofusins 1 and 2, mitofilin, TOM22, TIM23). Further information on ATP5B showed similarly expression levels in the range of 350–450 nTPM in all brain regions analysed in this study (neocortex, hippocampus, midbrain, pons, cerebellum, medulla oblongata).
Regarding possible AD-induced changes (e.g., strong decreases) in ATP5B expression, ATP5B does not appear in the list of proteins altered in AD after performing a cluster analysis of five different GEO AD platforms. 33 Decreases in the ATP5B mRNA levels have been found in the entorhinal cortex of human AD patients 34 and in laser-captured mouse CA1 hippocampal neurons where the decrease correlated with the NFT burden. 35 In another study, the ATP5B mRNA level was only slightly changed (less than 10%) in the temporal neocortex and hippocampal areas in AD compared to control human brains.36,37 Nonetheless, the risk of a disease-induced reduction of protein levels exists for all mitochondrial marker proteins. We counted all dots (mitochondria) with detectable signal intensities; that means independent of their ATP5B protein levels, provided that they are above the detection limit. In future studies, mitotracker dyes which are able to label mitochondria in postmortem17,38 and formalin-fixed human brain tissue could be used not only to describe general changes of mitochondria in AD,38,39 but to carry out morphometric analyses.
Interplay between mitochondria and peroxisomes especially in AD-related pathologies
Mitochondria closely interact with other membrane-bound intracellular organelles, such as the endoplasmic reticulum, lysosomes, lipid droplets 40 and especially peroxisomes 41 to coordinate lipid and ROS metabolism. In addition, mitochondria and peroxisomes share the machineries for organelle dynamics (fission-fusion), e.g., the fission proteins dynamin-related protein 1, fission 1, mitochondrial division protein 1,42,43 all three preferring peroxisomes. 44 Fission starts with the fragmentation (of damaged parts) of the organelle followed either by a repair and fusion (in case of the mitochondria) or by a removal through macroautophagy (mitophagy or pexophagy). Moreover, axonal transport of both organelles occurs via Miro-GTPases (reviewed by 45 ).
With regard to AD, increased levels of very long chain fatty acids 46 and reduced levels of plasmalogens and docosahexaenoic acid46–51 were measured in postmortem neocortices of patients with high ADNC compared to control patients. These metabolic alterations pinpoint to an additional peroxisomal dysfunction either up- or downstream to mitochondrial dysfunction.
In addition, both organelles are affected during inflammation, another pathogenic factor in AD. Generally, peroxisomes inhibit inflammation by synthesizing eicosapentaenoic and docosahexaenoic acids which both restrain the production of the pro-inflammatory prostaglandins and cytokines. 52 Mitochondria are pro-inflammatory organelles through the production of ROS and mitochondrial cardiolipin; cardiolipin mediates increase in cytokine production53–55 and promotes inflammasome assembly. 43 Furthermore, ROS-mediated signaling is involved in AD and is the result of a crosstalk between redox-sensitive organelles such as peroxisomes, mitochondria, and the endoplasmic reticulum.56,57 Thus, the dysfunction of mitochondria or peroxisomes will affect the metabolism as well as biogenesis and degradation, and thus also the number of both organelles.
In a previous study of our group, using the same paraffin-embedded human brain material (same AD patients and same specific brain regions), we analyzed the numerical densities of peroxisomes. 18 The peroxisome biogenesis protein PEX14 served as marker protein 58 for the quantitative analysis of immunofluorescence stainings prepared with the same protocols. Since we used the same material, the same method to label the organelle and the same method for quantification in the previous and in the present study, we are able to directly compare our data on numerical densities of mitochondria and peroxisomes as well as their eventual distinct region-specific alterations in different ADNC. In the frontal, temporal and parietal neocortices (primarily exposed to Aβ), decreases up to 30% of the peroxisomal densities 18 were accompanied by increases of 10 to 25% of the mitochondrial densities in patients at mid and high ADNC (Supplemental Figure 3). In the hippocampus (primarily exposed to NFTs), peroxisome density first increased by 20% until mid ADNC, but returned to control levels at high ADNC. 18 In contrast, mitochondrial densities decreased by 20% already at mid ADNC and stayed at the same level at high ADNC (Supplemental Figure 3). We found a negative correlation between the peroxisomal density and Aβ, but no correlations exist between mitochondrial density and Aβ as well as between peroxisomal and mitochondrial densities and NFTs. Thus, both organelles seemed to respond differently to AD-induced (metabolic) changes: mitochondrial numerical densities increased in areas with deposits of Aβ only (e.g., in the neocortex at low ADNC) and in contrary decreased in areas in which first NFTs were elevated (e.g., in the hippocampus); peroxisome numerical densities changed either by a decrease in case of the presence of Aβ deposits only (e.g., in the neocortex) and by an increase if NFTs appeared first (e.g., in the hippocampus; Supplemental Figure 3). 18 To the best of our knowledge, no previous study exists directly comparing the numerical abundances of mitochondria and peroxisomes in serial sections of the same brain areas of identical patients with different levels of ADNC.
As is the case in experimental research, our study has some limitations: e.g., (i) we analyzed a relatively sample size, (ii) morphometric analysis was time-consuming (just for one brain area from one patient, we acquired and analyzed a large number of fluorescent images) and (iii) we cannot completely exclude that rare highly fluorescent lipofuscin granules smaller in size than 50 pixel diameter were counted as mitochondria and that mitochondria labelled for ATP5B below the detectable level were not counted. In addition, this is a descriptive study, and no direct conclusion can be drawn from our results for AD pathomechanism. Still, we analyzed for the first time mitochondrial densities in 13 different brain-regions of the same postmortem brain samples of control patients and those with three different ADNC. Our results show that alterations in mitochondrial densities are opposite in the neocortex (increase) and hippocampus (decrease) which should be taken into account when mitochondria are considered as a therapeutic target. Future investigations on the mechanisms of the distinct AD-induced changes in mitochondrial and, in accordance with our previous study, peroxisomal densities might help to identify new therapeutic strategies for AD.
Supplemental Material
sj-docx-1-alz-10.1177_13872877261418994 - Supplemental material for Region-specific ups and downs in mitochondrial numerical densities during Alzheimer's disease progression: A pilot study in human brains
Supplemental material, sj-docx-1-alz-10.1177_13872877261418994 for Region-specific ups and downs in mitochondrial numerical densities during Alzheimer's disease progression: A pilot study in human brains by Anna-Lena Balsliemke, Barbara Ahlemeyer, Eugen Schmidl, Till Acker, Anne Schänzer and Eveline Baumgart-Vogt in Journal of Alzheimer's Disease
Footnotes
Acknowledgements
The authors thank Prof. Dr Peter Rehling, Division of Cellular Biochemistry, Georg August University, Göttingen, for recommending the ATP5B antibody (HPA001520 from Sigma-Aldrich, Germany) as mitochondrial marker in immunofluorescence stainings and for providing us with a small amount of this antibody for a test trial. The authors are also highly indebted to Prof. Dr. Jörn Pons-Kühnemann and his team, Division of Medical Statistics, Medical Faculty, Justus-Liebig University, Gießen, for continuous help in statistical analysis of our data. We further thank Susanne Pfreimer and Elke Rodenberg-Frank for excellent technical assistance.
Ethical considerations
The project was approved by the local ethics committee (AZ 07/09, Justus Liebig University Giessen).
Consent to participate
Written informed consent was obtained from the patients’ relatives.
Consent for publication
Not applicable.
Author contribution(s)
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: EBV received performance-related resource allocation-funds of the Medical Faculty of the Justus-Liebig University Gießen, so-called “Leistungsorientierte Mittel” (LOM).
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
All data generated or analyzed during the current study are mostly included in this published article and supplemental files. Further data are available from the corresponding author on reasonable request.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
