Abstract
Background
The Digital Assessment of Cognition (DAC) is a brief, 7-minute iPad administered-scored neuropsychological protocol.
Objective
The current research sought to investigate relationships between DAC test results and family ratings for neurocognitive decline; instrumental activities of daily living (IADL) impairment; psychiatric symptoms, and physician-determined cardiovascular risks.
Methods
179 memory clinic patients were assessed. Family members rated the severity of neurocognitive impairment, IADL decline, and psychiatric symptoms using the Everyday Cognition Scales (ECog); the Functional Assessment Questionnaire (FAQ); the Instrumental Activities of Daily Living–Compensation Scale (IADL-C); and the Neuropsychiatric Inventory (NPI), respectively. An index measuring cardiovascular risk was extracted from medical records.
Results
Partial correlations controlled for age, education, and sex found that greater functional disability and elevated cardiovascular risks were associated with lower DAC memory and executive index scores. Lower DAC memory scores were seen in relation to family ratings suggesting impaired Ecog Episodic Memory difficulty; relatively intact ECog Executive/Attention ability; and impaired IADL-C Memory/Self-Management difficulty. By contrast, lower DAC executive performance was seen in relation to family ratings suggesting impaired Ecog Executive/Planning difficulty and IADL-C Social Skills difficulty. Lower DAC-executive index scores were also associated with greater informant rated apathy.
Conclusions
The relations between DAC index scores and total informant FAQ and IADL-scores; Ecog and IADL-C subscales; and selected NPI-defined psychiatric problems suggest that the DAC is both sensitive to gross IADL decline, and specific to differing ECog and IADL-C and psychiatric problems. Combining digital assessment with family IADL ratings could help with clinical decision-making.
Keywords
Introduction
Dementia is a worldwide public health problem. In the United States alone it has been estimated that over 7 million people have been diagnosed and are living with Alzheimer’s disease (AD). 1 Importantly, medication that potentially modifies the neuropathology associated with mild cognitive impairment (MCI) and mild dementia associated with AD pathology has recently become available.2,3 Also, lifestyle modifications can have a positive impact in preventing or delaying the onset of dementia.4,5 However, to maximize the benefit of these or any other treatment strategies, clinical indications for nascent MCI and dementia must be uncovered as early as possible. For this reason, there is a renewed impetus to develop effective and parsimonious assessment strategies that can flag nascent, emergent MCI and dementia syndromes.
In addition to routine serum analyses for potentially reversable causes of cognitive decline; a CT-MRI study of the brain; and comprehensive neuropsychological assessment; the characterization and diagnosis of MCI and dementia requires querying a knowledgeable family member to ascertain the presence of decline in everyday functional or instrumental activities of daily living6,7 (IADL). Problems with episodic memory and executive abilities tend to typify dementia associated with AD pathology as well as dementia associated with MRI-defined vascular alterations. 8 Further, recent research suggests that dysexecutive difficulty appears to be disproportionately related to declining IADL abilities.9–12 For example, Libon and colleagues 13 studied a group of memory clinic patients with the Instrumental Activities of Daily Living–Compensation Scale12,14 (IADL-C). In this prior research, IADL problems tended to be highly associated with impaired performance on executive tests.
Recent research underscores links between vascular illness such as diabetes mellitus, hypertension, and hyperlipidemia with an increased risk for AD.15,16 Indeed, De Anda-Duran and colleagues17,18 found measurable dysexecutive and memory difficulty among community dwelling participants where cardiovascular risks were present but thought to be well managed. Other comorbid problems associated with MCI and dementia include psychiatric symptoms. In a recent report, Pan and colleagues 19 assessed cognitively normal, MCI, and dementia patients with the Neuropsychiatric Inventory 20 (NPI) and noted that depression, anxiety, and apathy were quite common in their sample. Other researchers have also found a high frequency of depression, anxiety, and apathy among their samples of patients characterized with AD and MCI.21,22
There is now considerable interest in digital assessment technology.23,24 However, there is comparatively little research examining how outcome variables using digital neuropsychological tests comport with clinical indications for cardiovascular risk, IADL decline, and comorbid psychiatric problems among memory clinic patients with suspected MCI and dementia. In recent research Libon and colleagues 25 studied a group of community dwelling participants. The Functional Assessment Questionnaire 26 (FAQ) was used to create groups suggesting no, subtle, versus mild FAQ-defined functional impairment. These groups were also assessed with the digital Trail Making Test-Part B where a host of new, motor and latency-based parameters were found to be associated with subtle to mild functional decline. Meaningful IADL decline was found to be associated with a host of these new, digital outcome variables measuring both motor and non-motor behavior. Similar results were described by Jokinen and colleagues. 27
In the current research, the Digital Assessment of Cognition 28 (DAC), a brief digitally administered-scored neuropsychological protocol was administered to a sample of memory clinic patients. This protocol was designed to assess verbal episodic memory, verbal working memory, and language-related abilities. The purpose of the current research was (1) to assess how knowledgeable informant ratings regarding neurocognitive, functional, and psychiatric difficulty are related to digital neuropsychological outcome measures; and (2) assess relations between digital neuropsychological test performance and clinical indications for cardiovascular risk.
In the current research, family members of memory clinic patients were queried with the Everyday Cognition Scales, 29 a questionnaire that provides informant ratings for neuropsychological abilities. Informant ratings regarding IADL abilities were obtained using the Functional Assessment Questionnaire (FAQ) 26 and the Instrumental Activities of Daily Living–Compensation Scale.12,14 Informant ratings regarding psychiatric problems were assessed with the NPI. 20 Finally, medical records were examined for indications of cardiovascular risk. Using regression and partial correlation analyses, the goals of the current research were to assess how digitally derived neuropsychological summary scores measuring episodic memory and executive control were related to these informant-based measures and cardiovascular risk.
Methods
Participants
Participants in the current research (n = 179; 56.40% female; 89.00% Caucasian) came from three sources including (1) the Rowan University, New Jersey Institute for Successful Aging, Memory Assessment Program (MAP; n = 125); outpatient referrals for neuropsychological assessment for suspected dementia (n = 24); and the Rowan University Department of Family Medicine outpatient ambulatory care service (n = 30). The NJISA MAP program provides a comprehensive, outpatient evaluation and work-up for suspected alterations involving neurocognition and personality-behavior. In addition to neuropsychological assessment, MAP patients were evaluated by a board-certified psychiatrist. A CT or MRI study of the brain and serum tests including a CBC, CMP, thyroid/B12, and an analysis of lipids was obtained. Participants referred by their primary care provider for outpatient neuropsychological assessment because of suspected dementia (n = 24) underwent the same neuropsychological evaluation as MAP patients.
Participants from the Rowan Department of Family Medicine were not referred for or assessed clinically and, therefore, did not undergo outpatient neuropsychological assessment. These participants were recruited for the current research as part of ongoing research for the development of digital neuropsychological assessment technology. Participants were excluded from this study if English was not their first language; or if there was any history of brain injury, substance abuse, a major psychiatric disorder such as major depression, other neurologic illness such as epilepsy, or metabolic disorders such as B12, folate, or a thyroid deficiency. For MAP and outpatient participants referred for neuropsychological evaluation, a knowledgeable family member was available to provide information regarding functional status. The current sample of 179 patients included the group of 77 patients described in our prior research. 28 This study was approved by the Rowan University Institutional Review Board with consent obtained consistent with the Declaration of Helsinki.
The Digital Assessment of Cognition
On average, the Digital Assessment of Cognition (DAC) requires approximately 7 minutes to administer. 28 The order of test administration was as follows: Two 6-word Philadelphia (repeatable) Verbal Learning Test 30 (P(r)VLT)-immediate free recall test trials; the semantic/‘animal’ fluency test (60 s); three trials of 5-digits backwards from the Backward Digit Span Test31,33 (BDST); a 6-item depression-anxiety screening inventory; and concluding with the P(r)VLT-delay free recall and delay recognition test conditions, respectively. Throughout the test administration, participants spoke their responses aloud, and the iPad recorded all speech for later processing and analysis. The protocol was administered using an 11-inch Apple iPad Pro. All assessments were proctored; however, all test instructions were delivered verbally by the iPad. During the assessment procedure, the iPad was kept in the portrait orientation while lying on a flat surface.
DAC episodic memory and executive control index scores
Standardized summary scores (z-scores) designed to express severity of episodic memory and executive impairment were compiled from the grand mean and standard deviation from the entire sample. The DAC episodic memory index score averaged P(r)VLT total delay free recall, and the P(r)VLT recognition ratio (i.e., [recognition hits-recognition hits + false positive foils]), a measure consistent with recognition discriminability scores as described in prior research.30,34,35 The DAC executive index score averaged total output on the ‘animal’ fluency test and BDST percent SERIAL order recall.31–33 A full description of all core digital outcome variables can be found in prior research 28 and Supplemental Table 1.
Informant neurocognitive, IADL, and psychiatric outcome measures
A spouse (64.3% of participants), adult child (17.9% of participants), other family members (14.3% of participants) or a family friend (3.5% of participants) was asked to respond to the questionnaires listed below. On average, informants have known their family member or friend for 44.60 years.
Everyday Cognition Scales 29 (ECog). Seven outcome measures are available from the ECog including the total score and six subscale scores rating neuropsychological abilities associated with episodic memory, language, visuospatial operations, executive abilities-planning, executive abilities-organization, and executive abilities-divided attention. A greater or higher raw score suggests greater informant related neurocognitive impairment.
Functional Assessment Questionnaire 26 (FAQ). The total score (range 0-30) was used in the current research where a higher score suggests greater functional difficulty.
Instrumental Activities of Daily Living – Compensation Scale12,14 (IADL-C). Five outcome variables were obtained from this measure including a total score and four subscale scores assessing money and self-management, home and daily living, travel and event memory, and social skills. A greater score suggests increasing functional difficulty.
The Neuropsychiatric Inventory 20 (NPI). The NPI provides a comprehensive assessment of the frequency and severity of 12 psychiatric symptoms (i.e., hallucinations, delusions, depression, apathy, anxiety, irritability, aggression, disinhibition, euphoria, aberrant motor behavior, disturbance of eating, and disturbance of sleep. As prior research has suggested that depression, anxiety, and apathy are quite common among patients with suspected dementia19, 21,22 only informant ratings for these three problems were analyzed in the current research.
Cardiovascular risk
Patients medical records were queried for six well-known cardiovascular risks including (1) a history of atherosclerotic cardiovascular disease (e.g., heart disease), (2) a history of cerebrovascular disease (e.g., stroke), (3) treatment with statins, (4) treatment with aspirin, (5) treatment for hypertension; and (6) treatment for diabetes. All six problems were coded as 1 = present or 0 = absent and summed to create a cardiovascular index (range 0-6).
Statistical analyses
Relations between DAC memory and executive index scores and total E-Cog, IADL-C, FAQ, and cardiovascular risk scores were assessed with partial correlations controlled for age, education, sex. Relations between the memory and executive index scores, E-Cog subscales, IADL-C subscales, and neuropsychiatric symptoms were assessed with a series of stepwise regression analyses. For all regression analyses age, education, and sex were entered into block 1 and ECog, IADL-C, or NPI variables were entered into block 2. Standardized regression coefficients (beta) were used to determine relations between dependent and independent variables.
Results
Demographic and clinical information
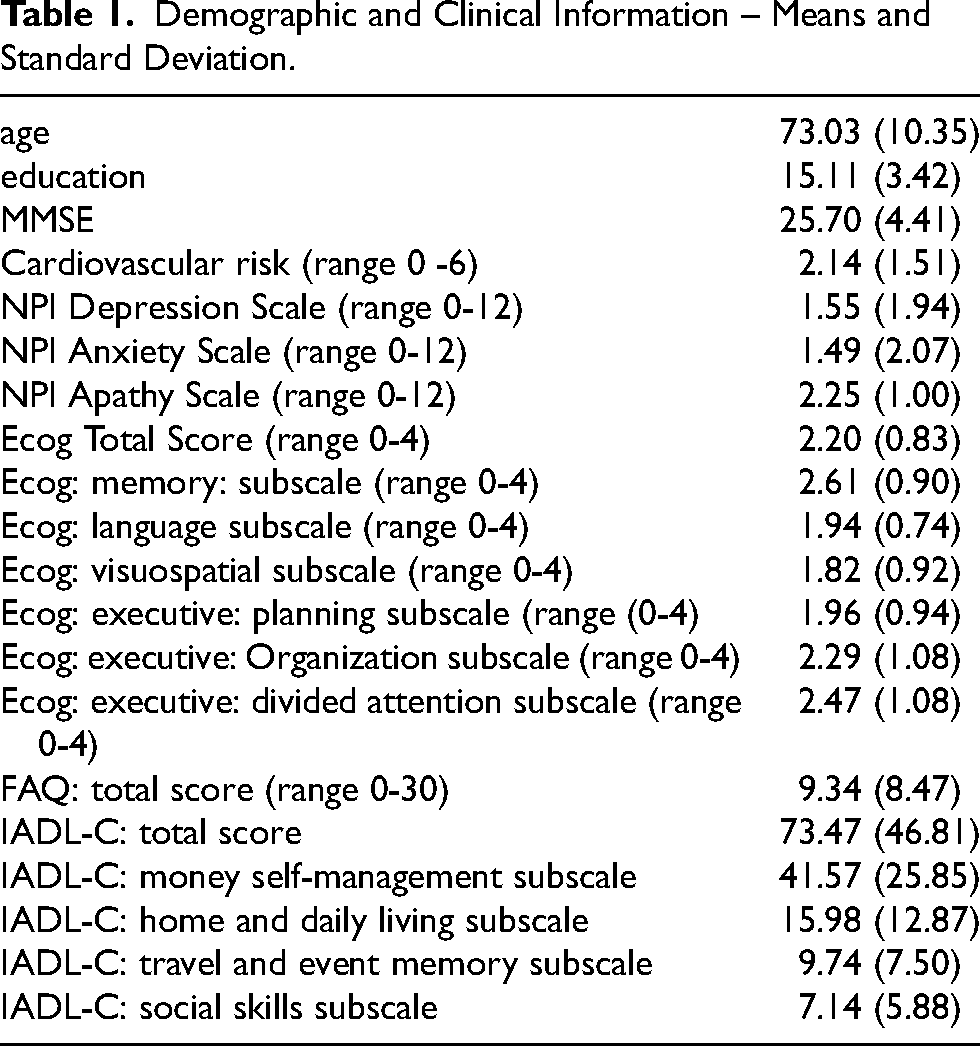
Table 1 lists demographic, MMSE and all ECog, FAQ, IADL-C scores, NPI symptoms and the cardiovascular risk score.
Demographic and Clinical Information – Means and Standard Deviation.
Digital Memory-Executive Index scores: partial correlations with total ECog, FAQ, and IADL-C scores
Significant partial correlations were found suggesting an association between worse DAC memory test performance and family ratings using the total ECog score (r = -0.378, df = 98, p < 0.001) and FAQ (r = -0.349, df = 98, p < 0.001) scores. The correlation with the total IADL-C score was only marginally significant (r = -0.186, df = 98, p < 0.065). Significant partial correlations were also found suggesting an association between worse DAC executive test performance family ratings also using the total ECog score (r = -0.338, df = 98, p < 0.001), total FAQ (r = -0.523, df = 98, p < 0.001), and total IADL-C (r = -0.256, df = 98, p < 0.010) scores. Zero-order correlations obtain separately from men and women can be found in Supplemental Table 2.
DAC Memory and Executive Index scores and ECog subscales
The stepwise regression for the DAC memory index score found worse test performance in relation to greater informant-rated difficulty for the ECog-Episodic Memory subscale followed by better informant-rated ability on the Ecog Executive-Attention scale (Ecog Memory Scale: R2 = 0.478, F = 30.82, p < 0.001; beta=-0.594, p < 0.001; Ecog Executive Attention Scale: R2 = 0.506, F = 5.50, p < 0.021; beta=0.217, p < 0.021). The stepwise regression for the DAC executive Index score found worse performance in relation to greater informant-rated difficulty on the ECog Executive-Planning subscale (R2 = 0.371, F = 6.83, p < 0.010; beta=-0.214, p < 0.010; (Figures 1 and 2; see Supplemental Tables 3 and 4 for full statistics).
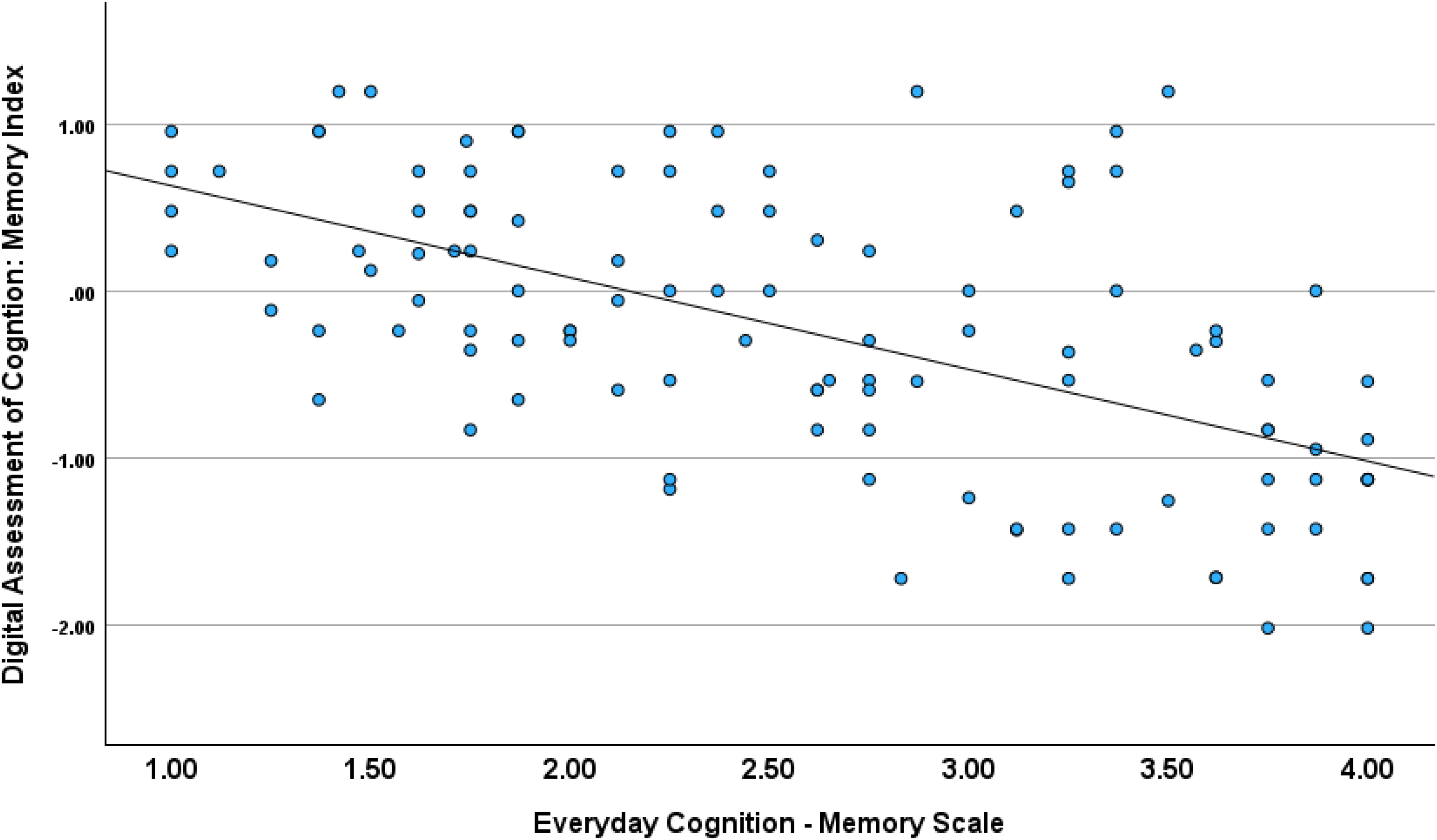
Digital assessment of cognition memory index and everyday cognition episodic memory scale.
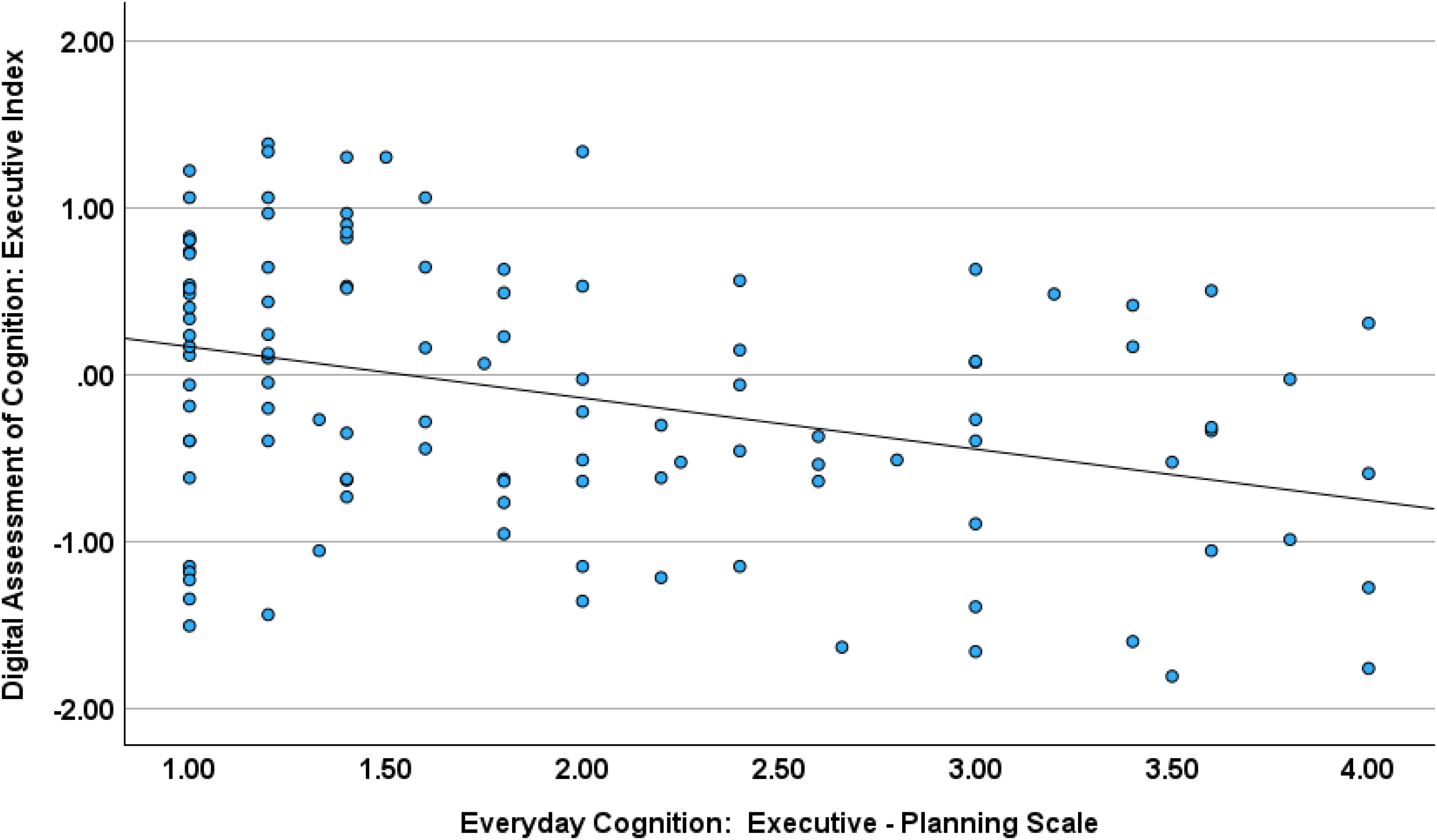
Digital assessment of cognition executive index and everyday cognition executive planning scale.
DAC Memory and Executive Index scores and IADL-C subscales
The stepwise regression for the DAC memory index score found lower test scores in relation to greater informant rated problems for the IADL-C Money and Self-Management subscale (R2 = 0.322, F = 4.15, p < 0.044; beta = -0.183; p < 0.044); and worse DAC executive test performance in relation to informant ratings suggesting greater problems on the Social Skills subscale (R2 = 0.347, F = 8.04, p < 0.006; beta = -0.243, p < 0.006; (Figures 3 and 4; see Supplemental Tables 5 and 6 for full statistics).
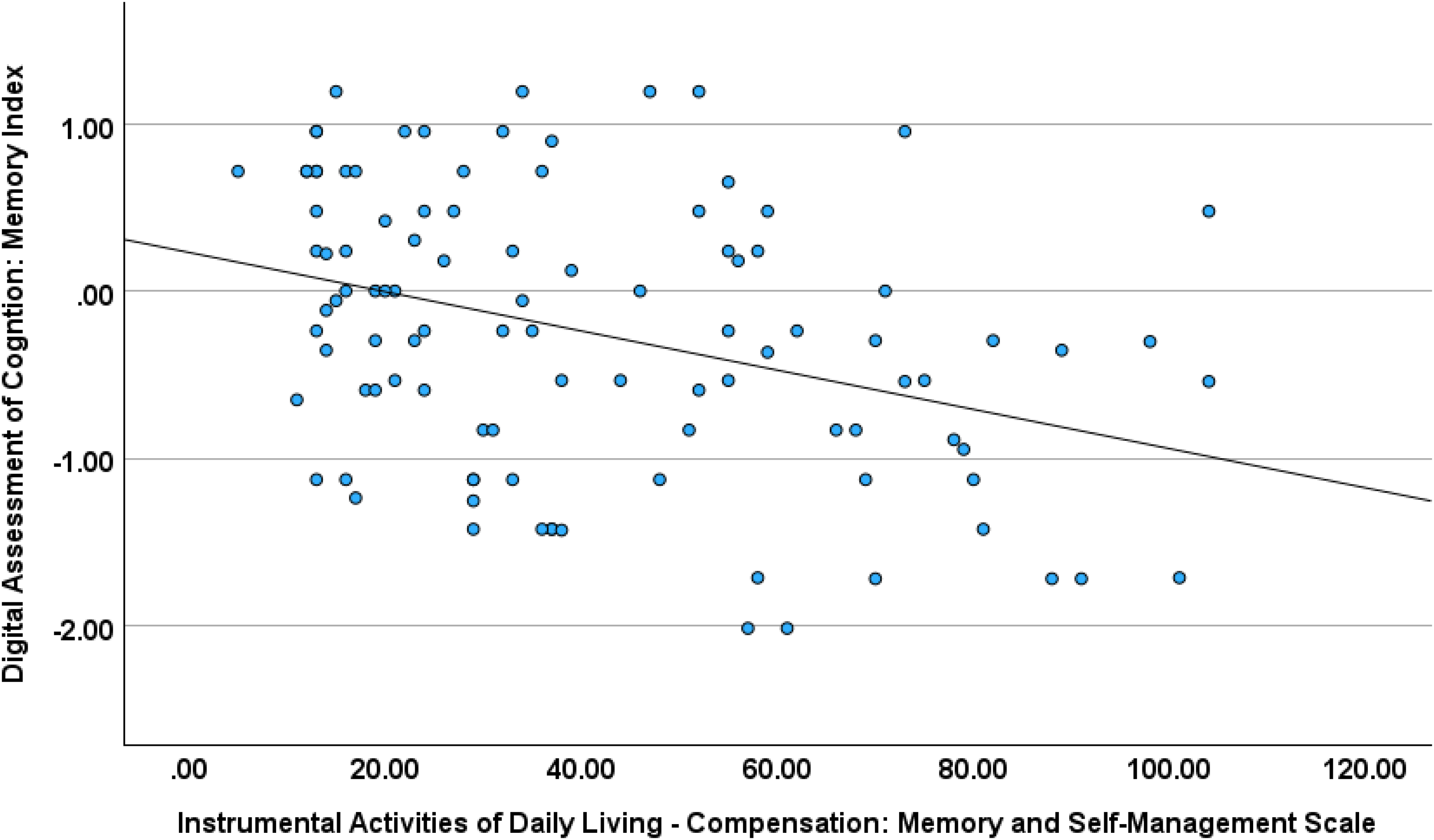
Digital assessment of cognition memory index and instrumental activities of daily living–compensation memory and self-management scale.
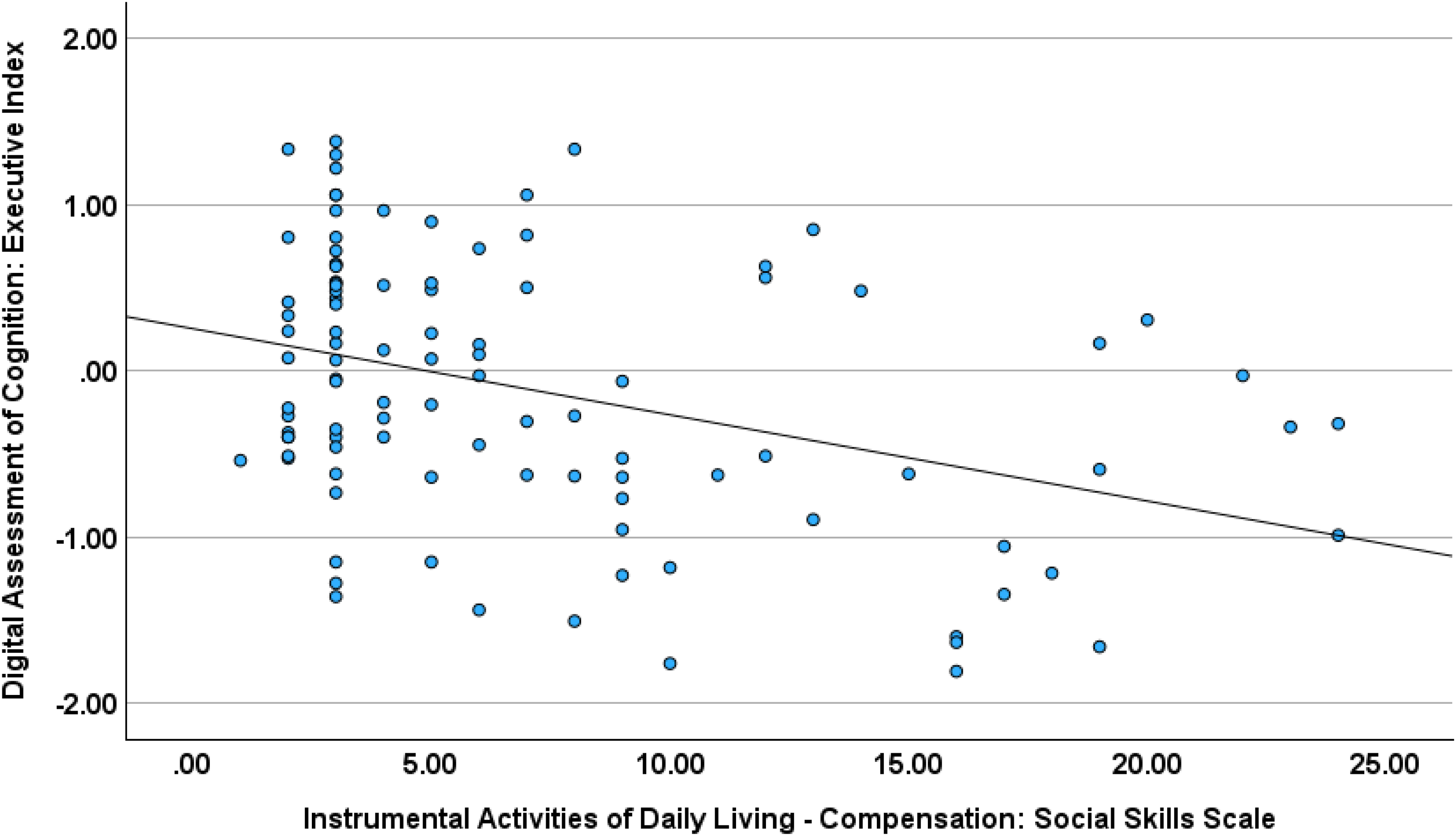
Digital assessment of cognition executive index and everyday cognition social skills scale.
Digital Memory-Executive Index scores and NPI symptoms
The stepwise regression for the DAC memory index was not significant as none of the three NPI symptoms entered the final model. The stepwise regression for the DAC executive Index score found worse test performance along with greater informant ratings only for apathy (R2 = 0.298, F = 4.90, p < 0.029; beta=-0.183, p < 0.029; (Figure 5; see Supplemental Table 7 for full details). Zero-order correlations obtain separately from men and woman can be found in Supplemental Table 2.
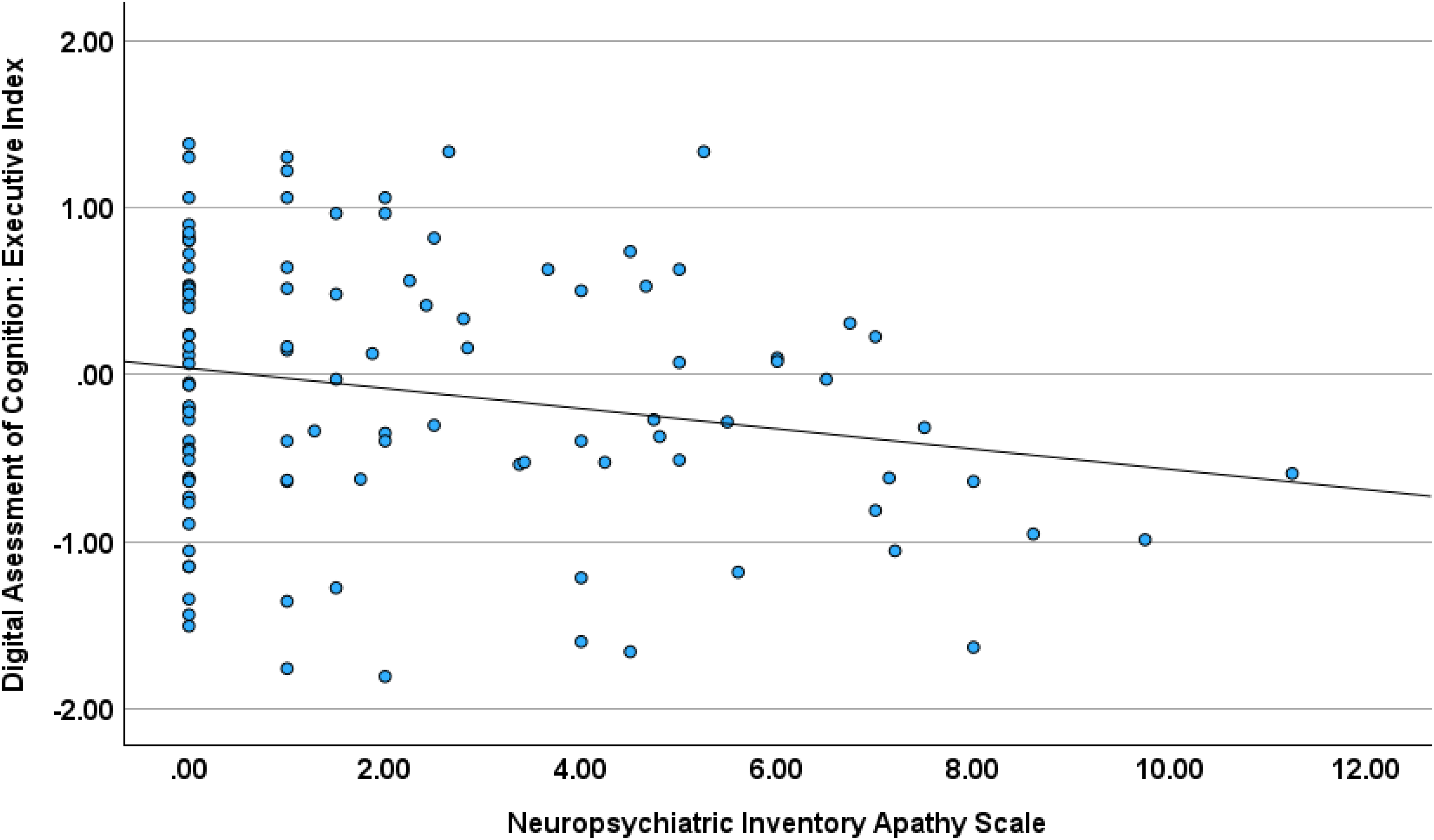
Digital assessment of cognition executive scale and the neuropsychiatric inventory apathy scale.
Digital Memory-Executive Indices and cardiovascular risk
Reduced scores on both the DAC memory and executive Indices were noted in relation to greater cardiovascular risk (memory Index-cardiovascular risk; r = -0.232, df = 137; p < 0.006; executive index-cardiovascular risk; r = -0.221; df = 137; p < 0.009).
Discussion
The DAC is a brief, iPad administered-scored neuropsychological protocol designed to assess verbal episodic memory, verbal working memory, and selected language-related operations. MCI has traditionally been viewed as a transitional state between relatively normal neurocognitive abilities and dementia such as AD. 36 Some of the early thinking regarding MCI suggested there should be little to no functional or IADL decline. 37 However, the results of subsequent research now sustain the observation that when MCI is initially diagnosed, IADL problems can be present.14,37 Indeed, data has shown that better prediction regarding the eventual emergence of dementia and rates of cognitive decline leading to dementia, are improved when IADL or functional abilities are considered.38–41
In the current research, we found that reduced performance on the DAC executive index score was associated with informant ratings for neurocognitive decline as measured with the ECog total score, and greater IADL decline as measured with total FAQ and IADL-C scores. Similarly, reduced performance on the DAC memory index score was also associated with lower total ECog and FAQ scores, but not the total IADL-C score. The observation demonstrating reduced performance on the DAC executive and memory index scores, and the total ECog score provides additional evidence supporting the criterion validity of this protocol as able to measure the two neurocognitive operations that are quite ubiquitous in MCI and dementia syndromes. Equally important, these data suggest that the DAC is generally quite sensitive to informant ratings suggesting declining neurocognitive and IADL abilities.
Functional activities have been traditionally divided into basic activities of daily living (ADLs) related to eating, dressing, hygiene, etc., and IADLs including taking medication correctly and financial management, etc. However, Frias and colleagues 29 point out that a potentially better way to understand functional loss is to assess the neurocognitive operation(s) that underlie relevant IADL activities. Frias and colleagues 29 noted that some IADL abilities may rely upon episodic memory, while IADL abilities could call upon much needed language or visuospatial operations. In support of these ideas, these researchers found that when amnestic versus mixed MCI memory clinic patients were compared on the ECog, the mixed MCI group displayed greater disability on all but the Ecog memory scale.
In the current research, and perhaps not surprisingly, reduced performance on the DAC memory index score was associated with worse Ecog informant ratings for memory. By contrast, the current research found that reduced performance on the DAC executive index score was associated with worse Ecog informant rating on the Executive-Planning scale. Implicitly, these Ecog items appear to be assessing the capacity for prospective planning and attention, i.e., “thinking things through before acting.” These results suggest that in addition to sensitivity, the DAC derived index scores may be able to dissociate between differing underlying neurocognitive operations that contribute to IADL decline.
The data described above comports well with ideas set forth by McAlister and colleagues.42,43 These researchers point out that declining IADL abilities are highly associated with dysexecutive difficulty, followed by short and long-term memory impairment. In the current research, declining DAC memory index scores were associated with difficulty on the IADL-C Money and Self-Management subscale. These IADL-C items ask informants to rate problems related to maintaining records, budgets, and related activities; operations that implicitly rely on intact episodic memory. By contrast, declining DAC executive index scores were associated with increasing informant rated difficulties on the IADL-C Social Skills subscale. This IADL-C subscale asks family members to rate such behaviors as meaningfully initiating and maintaining appropriate interpersonal discourse in conversation.
This observation is extremely interesting given the association between worse DAC executive performance and increasing apathy when informants were queried with the NPI. Massimo and colleagues 44 characterized apathy as a reduction in goal-directed behavior often resulting in difficulty in initiating activity, planning prospectively, and generating the necessary self-motivation to achieve specific goals. Massimo and colleagues 45 found that among patients with behavioral variant-frontotemporal lobe dementia apathy is a major source of caregiver distress. Also, as reported above, apathy is quite common among patients with MCI.19,21,22 Recently, Ginsberg and colleagues 46 found a robust association between increasing IADL impairment and greater NPI-defined apathy and more malignant performance on paper and pencil neuropsychological test assessing executive tests. The data described above linking reduced DAC executive index scores and greater informant rated NPI-defined apathy is consistent with prior research. However, as seen in Figure 5, for many patients in our sample apathy was absent. Thus, the relationship suggesting worse DAC executive scores and increasing informant rated apathy should be viewed with caution.
There is, without question, data linking cardiovascular health with both clinical indications and the neuropathology that typifies AD.47–51 For example, Jiang and colleagues 50 studied a group of community dwelling research participants. Cardiovascular risk was determined using the Framingham Risk Scores 52 (FHS). Elevated FRS were seen in relation to increased neurofilament light chain. VandeBunte and colleagues 53 also examined a group of community dwelling research participants and found that elevated systolic blood pressure was related to plasma phosphorylated tau181 (pTau181) and glial fibrillary acidic protein. These observations are consistent with analyses described above documenting worse DAC memory and executive test performance and elevated cardiovascular risks.
The current research is not without limitations. First, the data described above must be viewed as preliminary as only correlational relations were assessed. Future research is needed with patients classified into clinical groups, who are followed longitudinally, employing additional analyses documenting statistically determined levels for sensitivity and specificity. Second, horologic time-index measures 54 linking temporal progression using digital neuropsychological and functional outcome measures should be undertaken. Third, test-retest reliability needs to be determined. Fourth, assessing a wider array of neuropsychiatric problems would increase the external validity of the data described above. Fifth, our sample was overwhelmingly Caucasian. Data from a more diverse sociodemographic sample could increase the external validity of our findings.
Despite these limitations, the data described above suggest that digitally derived memory and executive index scores are sensitive to gross informant IADL decline and specific to differing types of IADL and neuropsychiatric symptoms. In sum, combining digital neuropsychological assessment along with informant ratings for functional and neuropsychiatric difficulty could result in better clinical decision-making regarding emergent treatment options for MCI and mild dementia related to AD.
Supplemental Material
sj-docx-1-alz-10.1177_13872877261418287 - Supplemental material for Digital neuropsychological assessment–Part 2: Relations with cardiovascular risk and informant ratings of neurocognitive decline, functional disabilities, and psychiatric symptoms
Supplemental material, sj-docx-1-alz-10.1177_13872877261418287 for Digital neuropsychological assessment–Part 2: Relations with cardiovascular risk and informant ratings of neurocognitive decline, functional disabilities, and psychiatric symptoms by David J. Libon, Rod Swenson, Sean Tobyne, Sheina Emrani, Laura Salciunas, Aeysha J. Brown, Terri Ginsberg, Mitchel Kling, Kevin Overbeck, Leonard Powell, Christian White, Adaora Okoli-Umeweni, Christopher Janson and Stephen Scheinthal in Journal of Alzheimer's Disease
Footnotes
Acknowledgements
The authors have no acknowledgments to report.
Ethical considerations
The study was conducted in accordance with the Declaration of Helsinki and was approved by the IRB Committee of Rowan University (no. pro 2016001115) on June 11, 2025, with the need for written informed consent waived.
Consent to participate
Not applicable
Consent for publication
Not applicable
Author contribution(s)
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Drs. Libon and Swenson consult to Linus Health, Inc; Dr. Libon receives royalties from Linus Health, Inc.; Drs. Libon and Swenson receive royalties from Oxford University Press. Dr. Tobyne is an employee of Linus Health. Drs. Libon and Swenson are Editorial Board members of this journal but were not involved in the peer-review process of this article nor had access to any information regarding its peer-review.
Data availability statement
The data that support the findings of this study are available from the corresponding author with Rowan University IRB approval. Some restrictions may apply.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
