Abstract
For years, the understanding of Alzheimer's disease (AD) has been shaped by the amyloid hypothesis, which suggests that pathological markers like amyloid-β (Aβ) and phosphorylated tau are the primary drivers of the disease. This hypothesis has guided the development of major treatment strategies, including monoclonal antibodies targeting Aβ. However, most of these treatments have failed to produce clinically significant results, highlighting the urgent need for a new therapeutic approach. It is now evident that AD is a complex, multifactorial disease that develops over decades, ultimately leading to Aβ and tau accumulation. Therefore, addressing the underlying causes of these depositions is crucial. One well-supported yet underrecognized theory is the infection hypothesis, which links infections to AD pathology. Despite substantial scientific evidence, this perspective has faced significant resistance. In this review, we describe how chronic infections contribute to AD by triggering neuroinflammation and Aβ accumulation. We also explore the barriers to accepting the infection hypothesis and the steps necessary for its integration into drug development and early-stage treatment strategies. Persisting with an amyloid-centric approach will only exacerbate the societal burden. Embracing the infection hypothesis could transform AD research, diagnosis, and treatment, bringing new hope to millions.
Introduction
Alzheimer's disease (AD) and related dementias are currently the most pressing public health challenge due to their high prevalence in the world-wide aging population, paucity of treatment options, profound impacts on quality of life, and population trends towards increased longevity. 1 After more than 100 years of scientific investigation, we still do not know what the causes of AD are. The initial description by Dr. Alzheimer himself on the deposition of amyloid-β (Aβ) under the form of plaques was thought to be the cause. 2 Recently, this hypothesis was extended by adding the neurofibrillary tangles accompanied by extracellular phosphorylated tau (p-Tau) deposition. Brain pathological hallmarks such as Aβ and p-Tau are also present in the blood and cerebrospinal fluid (CSF) as peripheral measurable clinical biomarkers at all stages of the AD progression from pre-clinical to clinical stages.3–7 According to the current scientific view, these biomarkers, especially Aβ, are considered causal factors in AD because they are consistently found in the brains of patients post-mortem and can also be detected in vivo using amyloid PET.8–10 Therefore, Aβ emerged as the most prominent druggable target, and monoclonal antibodies directed against various forms of Aβ are now in clinical use, where they are reported to slow the progression of AD.11–13 However, growing scientific evidence accumulated over the past decades suggests that Aβ deposition is one observable consequence of AD pathogenesis but neither a necessary nor sufficient cause of the disease.14,15 For example, several animal species that develop AD-like dementias do not exhibit Aβ accumulation. Likewise, post-mortem analyses of the brains of individuals diagnosed with AD show that Aβ is rarely the sole pathological feature and is absent in many cases. Conversely, many people without clinical symptoms of AD display substantial Aβ deposition. Moreover, reducing Aβ levels in AD patients’ brains with monoclonal antibodies has generally failed to produce meaningful clinical benefits.16,17 Although a contributory causal role for Aβ cannot be ruled out and may indeed be relevant in some cases, it is increasingly implausible that Aβ represents the sole or primary cause of AD. Nevertheless, despite these findings, much of the scientific community continues to focus predominantly on Aβ and tau as therapeutic targets. Current evidence calls into question the idea that AD is a single, unified disease with a single causal pathway. Although the infectious-origins hypothesis has gained increasing empirical support, it has been largely overlooked for more than three decades, including in the latest Lancet commission report. 18 This review aims to disentangle how infections and antimicrobial roles of Aβ may contribute to AD progression and what it will take for the richness of our accumulated knowledge on AD to benefit the millions of patients suffering from it.
The past: amyloid hypothesis of Alzheimer's disease
The disease stigmata/markers discovered by Dr. Alzheimer at the beginning of the twentieth century in the case of a 51-year-old woman are now described in the medical world as the hallmarks of dementia. 2 At the beginning of the 1980s, the medical establishment started to consider what was known for a long time as senile dementia as the new AD, having the same presentation and markers that Dr. Alzheimer described decades before. 19
The pathomorphological hallmarks (and causes) were clearly established as the Aβ plaques and the neurofibrillary tangles. It is important to note that Aβ is initially secreted as a monomeric peptide but rapidly oligomerizes, acquiring neurotoxic properties through interactions with neuronal receptors (e.g., TLR2) that initiate proinflammatory cytokine production, oxidative stress, and mitochondrial dysfunction. 20 Subsequently, these oligomers fibrillate and assemble into plaques, which remain toxic by perpetuating microglial activation and neuroinflammation, thereby promoting neurodegeneration.21,22 This created the assumption of a clear link between the pathomorphology and the clinical presentation. Consequently, nearly all researchers in the field devoted their energy and sometimes their entire scientific life to finding the holy grail, i.e., to free the brain from the deposited Aβ and, if possible, as early as possible.23–26 From the beginning, it was clear that this approach might not succeed, as Dr. Alzheimer and later Dr. Fischer had drawn attention to the possibility that Aβ is only a consequence and not the cause of the disease. 27 Surprisingly, they also advocated that an infection may be present.
It is now known that many older subjects accumulate amyloid in their brains without developing dementia and, vice versa, that many demented people do not have significant amyloid deposits in their brains. 28 Furthermore, it is clear that Aβ has many advantageous physiological roles, notably including its function as an antimicrobial peptide (AMP).29–34 Accordingly, the research focus on AD has been shifting to p-Tau.35–37 Its accumulation can be measured in the sera, CSF and brain, and it was found that this accumulation is triggered by Aβ.3,5,38–40 The new paradigm now proposed is that, if Aβ elimination was not a success, perhaps p-Tau elimination would be, based on the belief that p-Tau is more linked to the clinical progression of AD.41,42 However, after numerous trials, this has also not proven to be a valuable path. 43 Finally, a third marker was added by the proponents of the amyloid hypothesis, which is neuroinflammation elicited/triggered by the plaques and the neurofibrillary tangles.44,45 At the center of this neuroinflammation are the microglia supported by the changes in astroglia known as astrogliosis.46,47 Recognition that neuroinflammation is part of AD represents great progress in understanding the pathomechanisms of the disease. However, neuroinflammation is not a late but a very early event, in fact preceding Aβ deposition and consequently damaging when it becomes chronic. 48
It is important to note that AD occurs in two main forms: familial and sporadic. 49 The familial form is caused by specific mutations that lead to pathological Aβ deposition and the development of AD.50,51 This form accounts for approximately 5% of all AD cases and can manifest at almost any age. In contrast, the sporadic (or idiopathic) form shows no mutations in the APP-processing machinery, and its incidence increases predominantly with aging.. 52 There is also a well-defined genetic form in individuals with Down syndrome, who carry three copies of chromosome 21, which includes the gene encoding amyloid precursor protein (APP). The resulting overexpression of APP leads to excessive Aβ production, promoting plaque formation and contributing to AD in this population. 53 The familial AD and Down syndrome genetics have often been interpreted as supporting a causal role for Aβ in the pathogenesis of AD.
After decades of failure with previous drugs removing Aβ without improvement of cognition, the proponents of the amyloid hypothesis were reinvigorated by the slight clinical success of the anti-amyloid monoclonal antibody treatment with aducanumab and donanemab.54,55 This was claimed as an enormous success and a historic step forward for the entire scientific and medical community. However, clinically eligible patients are only about 8% among those corresponding to the already stringent eligibility criteria and clinical effects are marginal. 56 This led to the assumption that this treatment improves clinical endpoints based on simply removing Aβ. The limited clinical success of these interventions, despite decades of effort, strongly suggests that Aβ is likely not the primary cause of AD but rather a downstream consequence or, at most, a significant biomarker of the disease. 57
However, we are not suggesting that Aβ and p-Tau do not contribute to the AD pathogenesis. It might be that modest levels of each may play an adaptive physiological role, but that too much is part of an excessive response to other underlying conditions. 58 It also could be that targeting Aβ and p-Tau too late produces few benefits for patients, and that interventions are needed decades earlier. Lastly, it may be that cognitive reserve and buffering mean that even some patients with advanced Aβ pathophysiology show few symptoms. These observations highlight the complex nature of AD, which arises from the interplay of both significant genetic and environmental factors. Nonetheless, it has become increasingly clear that i) targeting Aβ and p-Tau is not the most efficient or productive way to treat or prevent AD; ii) many other factors play crucial roles upstream; iii) Similar symptoms arise from other pathophysiological pathways, indicating that AD defined by protein aggregation is not a distinct clinical entity but rather one component of a broader AD spectrum59,60; and iv) understanding common causes of various pathophysiological mechanisms is likely to be a more productive approach. Considering the significant clinical failure of targeting Aβ and p-Tau in AD, we should ask whether we have alternatives. Therefore, we should first ask a fundamental question: what generates the recognized hallmarks of AD in certain individuals?
Alzheimer's disease as a syndrome with complex multifactorial causes
Clinicians understand that AD is still best regarded as a syndrome 61 or spectrum of disorders, 59 not a single disease, although many in the AD research community view it as such because of its defining biological markers (Aβ and p-Tau). 62 The clinical manifestations of the disease may be highly variable and depend largely on the history of the individual who is presenting it. 63 Not only are the clinical manifestations particularly heterogeneous, but there are also many paths leading to AD. There is no simple one-to-one mapping of etiology and symptomatology. 64
One way to conceptualize this is as a series of upstream factors—pollution, infections, lipid metabolism, glucose uptake, mitochondrial dysfunction, dysbiosis, etc.—that collectively influence a range of pathophysiological mechanisms. Some causes have a stronger impact on particular mechanisms than others, and these mechanisms can interact, forming a complex pathophysiological network where certain processes reinforce each other. Nevertheless, these pathophysiological changes do not directly lead to specific symptoms, as they are moderated by cognitive buffering (also known as cognitive reserve or cognitive resilience) and the individual's unique neural architecture, ultimately determining whether a distinct set of symptoms emerges. If most individuals presenting advanced dementia symptomatology suffer from multiple pathophysiological mechanisms, it could explain why targeting one single mechanism may have minimal clinical impact, particularly if the treatment is administered late in the etiological process. The challenge is that cognitive buffering often delays the onset of symptoms, meaning that by the time symptoms are noticeable, treatment may already be too late. In this model, the most promising strategies for treatment and prevention likely involve identifying the key contributing factors and intervening early, before the pathophysiological mix fully forms. Among these contributors, infections and immune state are one of the most promising potential targets.
From the earliest history of research on neurodegenerative diseases, it was suspected that somehow infections may be involved in their pathogenesis. The best-known example is tertiary syphilis leading to dementia. 65 The next alarm was given to the medical community by the increase in AD after the influenza pandemic in 1918–1919. 66 It was later learned that HIV infection leads to what is called HIV-associated neurocognitive disorders (HAND), the incidence of which is highly decreased by antiretroviral treatment (although not completely eradicated). 67
Recently, the COVID-19 pandemic demonstrated how a peripheral viral infection may cause neurocognitive disorders. 68 It is widely recognized that although COVID-19 was initially regarded as a primarily respiratory infection, it soon became evident in clinical practice that many patients developed neurocognitive and psychiatric symptoms, including confusion, delirium, brain fog, and anxiety.69,70 It was found that SARS-CoV-2 can invade the brain either directly—since neurons express ACE2 receptors—or indirectly via infected innate immune cells such as monocytes. 71 Within the brain, SARS-CoV-2 triggers pathological changes reminiscent of those observed in AD, including Aβ deposition, p-Tau neurofibrillary tangles, marked neuroinflammation through activation of microglia and astroglia, breakdown of the blood–brain barrier (BBB), vascular alterations leading to early synaptic loss, and subsequent neurodegeneration.72,73 Although COVID-19 is an acute infection, these hallmarks strongly suggest that it may contribute to long-term risk for AD development. Indeed, COVID-19 joins a long list of historic infections associated with the onset of dementia resembling AD. Furthermore, from a pathophysiological perspective, sickness behavior has long been recognized as a prototype for understanding how cognitive function is altered in response to local or systemic insults, particularly infections. 74
Despite the predominance of the Aβ hypothesis, researchers tried to advance the understanding of other pathophysiological mechanisms. One of the most successful possibilities for an alternative trigger of AD was the vascular hypothesis.75,76 Vascular problems may lead to AD through several potential mechanisms. The first very important study was the Nun study, which showed that none of the participating nuns could be clinically diagnosed as AD if they did not present cerebral vascular alterations. 77 Hence, the vascular injuries clinically revealed the underlying AD pathology in the brain. How could this be? It is now known that ischemia, even for a short time or in a silent region, leads to Aβ production as a protective factor. 78 Hypertension may also be a powerful risk factor which can lead to Aβ production followed by p-Tau via repetitive hypoxia. 79 Furthermore, ischemia may lead to oxidative stress and inflammation, which will also generate the Aβ deposition resulting in neuronal death. Finally, it is now well-recognized among vascular scientists, cardiologists and vascular surgeons that atherosclerosis either in coronary or peripheral arteries can cause vascular lesions in the brain, especially at the blood brain barrier or the choroid plexus. The vascular contribution is further supported by a study showing that brains of patients with severe dementia rarely have pure Aβ pathology, and that it is often mixed with vascular issues. 80 This further supports the multiple etiologies-multiple symptomatology model.81,82
It is also recognized that brain trauma, either by fall or concussion, led to Aβ deposition and in the long run to AD.83,84 This was contested for a long time before becoming widely accepted. This reinforces the idea that Aβ is a by-product of various injuries to the brain, released and acting as a protective factor. 85 When the aggression is maintained through decades, it will excessively deposit and become harmful. Too much of a good thing can turn a bad thing. Moreover, brain trauma has been linked to reactivation of the herpes simplex virus (HSV) as well as to hypoxia resulting from vascular injury, further supporting the notion that Aβ deposition may be an adaptive response and that multiple etiologies and risk factors interact in AD.86–88 Recently, life-long exposure to toxic substances was also accepted as one pathway which could lead to the development of AD.89,90
Finally, other systemic insults, including socioeconomic, psychological, or metabolic stress, dysbiosis at any part of the body, or adverse early life events could lead to the development of some AD-like dementia.91–93 As the most widely studied “off track” cause of AD is infection, we will examine it as a prototype cause for the development of AD.
How infections can lead to Alzheimer's disease
As mentioned above, certain infectious diseases such as syphilis, HIV-AIDS, and SARS-CoV-2 (COVID-19) may lead to dementia resembling AD.65–68 This observation has prompted researchers to dive into the putative causal connection between infections and AD.94,95 What is the current evidence, either experimental or clinical, to state that infections lead to AD? First, we wish to be clear that we are not using the term infections in the broadest lay sense (acute infections culminating in an acute infective disease) but rather to chronic and often latent infections.96–98 Moreover, under this hypothesis and considering the complex nature of AD, not all chronically infected persons are expected to develop AD. 99 With that in mind, we will examine the experimental, clinical, and epidemiological evidence for these claims.
A key starting point is to emphasize the importance of studying AD and related dementias (ADRD) rather than just AD. AD was originally defined based on the pathomorphological presence of Aβ plaques, without proof that these plaques were causally implicated in all cases with similar symptomatology. The presence of multiple types of dementia, including Lewy body dementia and vascular dementia, is now well recognized, and advanced clinical dementia is generally characterized by mixed pathology.92,100–103 In this context, looking for evidence that infections cause AD specifically is not likely to be productive. Rather, we should ask what the evidence is that infections relate to ADRD or similar clinical presentations, and how this might work mechanistically.
Some of the first experimental evidence beside those evoked by Dr. Alzheimer and Dr. Fischer came from the groups of Dr. MacDonald and Dr. Itzhaki. Dr. Macdonald first showed that, in the brains of AD patients, spirochetes bacteria were more frequent than in those not presenting AD symptoms.104,105 The Itzhaki group later identified HSV1 DNA in the brain of AD patients.106,107 However, it was detected at similar frequencies in age-matched controls, suggesting that additional risk factors, such as the apolipoprotein E4 (APOE4) genotype, may influence whether HSV-1 contributes to AD development. Indeed, the difference was that the prevalence of APOE4 was high in the patients and low in the normal subjects, suggesting a joint HSV1-genetic risk for AD. 108 The key to explain this apparent contradiction is that only subjects having a lifelong reactivation of the virus will produce Aβ in the long run, resulting in uncontrolled chronic neuroinflammation; the related intrinsic and extrinsic risk factors are starting to be elucidated.109–113 All these steps may be contained if the immune system is capable of adaptation to challenge the infection. If the immune system is not able to adapt, as can happen with aging (inflammaging), this can lead to neuronal destruction.114–117 The state of trained innate immune memory also determines whether the pathological process leads either to protection or to immune paralysis.118–120 The exposome may exert its influence via toxic products, diet, or exercise.121–125 Pathological stresses also contribute to the constant reactivation of the virus. Furthermore, in susceptible individuals, i.e., especially APOE4 homozygotes, the reactivation of one virus may lead to the reactivation of other pathogens such as Varicella-zoster, making AD often the consequence of a chronic polymicrobial infection. 126
Additionally, it is now becoming evident that APOE4 is not uniformly detrimental. For instance, APOE4 can confer protection against HSV-1. In Amazonian forager–horticulturalist populations with a high parasite burden, APOE4 has been associated with improved cognitive performance, 127 and another study showed that APOE4 modulates the relationship between infectious burden and cognition. 128 Moreover, APOE4 has also been reported to protect against liver injury caused by hepatitis C virus (HCV). 129 These paradoxical findings may be accounted for by the ways in which APOE4 modulates cellular membrane cholesterol which influences viral entry, as well as by its capacity to modify the receptor-binding affinities of different viruses that compete with APOE isoforms.130–135 These studies show that the relationship between infections and genetic susceptibility is very complex and that it may modulate the infection—AD relationship. This also points to the presence of numerous possible risk factors for developing AD decades after the original infection.
In addition to viruses, bacteria were also proposed early on to be involved in AD pathophysiology. Pioneering work by Dr. Miklossy with Borrelia burgdorferi (the cause of Lyme disease) being able to invade the brain and leading to AD some decades later gave further credit to the (bacterial) infection hypothesis. 136 Although the mechanistic link could not be definitively established, clinical treatment against B. burgdorferi greatly improved cognition of patients. 137
Another pioneering scientist, Dr Balin, showed in experimental models and in human brains that intracellular Chlamydia pneumoniae infection may also result in AD development. 138 Since then, many other groups have also demonstrated the presence of this bacterium in the neurons of AD patients. 139 Not all AD patients have C. pneumoniae infections, and not all infected individuals will end up with AD. Since these infections last for decades, many aspects of the immunobiography and envirobiography can influence who will proceed further towards clinical AD. This also means that many infected subjects may also efficiently combat their infections and never get AD. This could explain why older subjects may have Aβ plaques and not present AD. These Aβ plaques may act as biofilms containing the invading microbes.140,141
One of the most studied bacteria in ADRD pathophysiology is Porphyromonas gingivalis, a keystone bacterium in the mouth leading to gingivitis (inflammation of the gingiva) and progressing ultimately to periodontitis, with the loss of bone and teeth. 142 Almost half of people presenting an infection by P. gingivalis in their 50 s or 60 s will develop ADRD some decades later. 143 P. gingivalis makes biofilms, making the bacteria difficult to eradicate. Therefore, once the process in the mouth starts, most individuals will develop AD if they live long enough.144,145 In this context, the work of Dr. Dominy demonstrating the gingipains in the brain of AD individuals was also seminal. 146 This has also been demonstrated many times in experimental animals, mainly in mice. 147
Fungi and parasites may also be considered among the microbial causes of AD, either via direct infection or via the gut-brain axis.98,148 This raises the possibility that a dysbiosis in the microbiome from any part of the body may be related to the brain and cause AD. This also reinforces the notion of the constant communication between the periphery and the brain either directly or by the vagus nerve for these microbes and their metabolic products.149,150
Microbiome-gut-brain axis and its potential involvement in AD
The gut microbiome, as well as other body microbiomes such as those of the nose and mouth, is increasingly being linked to AD. Growing evidence indicates that microbial dysbiosis, an imbalance in the normal microbial flora, can influence AD development through the microbiota–gut–brain (MGB) axis. 151 This dysbiosis, coupled with increased gut permeability, can contribute to key AD hallmarks, including neuroinflammation, abnormal Aβ aggregation, tau hyperphosphorylation, and other detrimental effects on brain cells, potentially promoting disease progression. Recent experimental studies have confirmed the presence of dysbiosis in AD patients, although the specific bacterial alterations reported vary across studies.152,153 What appears consistent, however, is that changes occur throughout the AD continuum. Moreover, while significant interpersonal variability exists in gut microbial composition and diversity, all studies reported alterations associated with AD. Recent studies have also emphasized that the functional properties of the various bacteria may be more important than their taxonomy (phylogenetic lineages), offering new avenues for targeted microbiome manipulation.
How can alterations in the microbiome at multiple sites in the body, including the gut, lead to pathological changes in the brain characteristic of AD? Several indirect mechanisms have been proposed, including the production of neuroactive compounds, modulation of the immune system and metabolism, and regulation of the BBB, all of which may contribute to the development of AD hallmarks in the brain. 151 Direct microbial invasion has also been suggested: pathogens such as P. gingivalis, Borrelia burgdorferi, and Chlamydia pneumoniae may reach the brain via peripheral nerves, including the vagus nerve, or directly through the olfactory bulb or trigeminal ganglion. 154
These findings represent associations and do not yet establish that dysbiosis directly causes AD. However, emerging research suggests that interventions targeting the microbiome, such as probiotics or dietary modifications, may hold therapeutic potential for preventing or mitigating AD, although larger and long-term clinical trials are needed to confirm these effects. One particularly compelling line of evidence comes from fecal microbiota transplantation (FMT). In some patients with both C. difficile infection and AD, FMT has been reported to produce significant improvements in cognitive function.155,156
Mechanisms by which infections cause AD
A question raised by the infection hypothesis is what mechanisms cause AD when the pathogen in the brain cannot be found most of the time. Despite this contention, one obvious mechanism is the invasion of the AD patient brain by microbe(s). Microbes can reach the brain through multiple routes. Some cross the BBB independently, for example via transcytosis, while others are transported inside white blood cells in a “Trojan horse” mechanism.157–160 Certain pathogens can also propagate along neuronal axons, and others may gain direct access to the central nervous system through the olfactory pathway or the trigeminal nerve.161–163 These diverse mechanisms provide plausible routes by which peripheral infections could contribute to the pathological hallmarks observed in diseases such as AD.
Many studies showed the direct presence of microbe(s) in the brain of patients suffering from AD, but some others were unable to demonstrate them. 164 The issue could be technical, such as the conservation of post-mortem brains. But microbes could still cause AD even while being undetectable in the brain. Recent studies have shown that microbial byproducts can be detected in the brains of many AD patients at higher frequencies than in control subjects. These include gingipains, bacterial lipopolysaccharides (LPS), and viral proteins such as HSV-1–linked ICP27. 165 This remains controversy regarding the causal role of microbes, as they can also be found in the brains of non-AD individuals. Nevertheless, these findings are consistent with the multifactorial and multicausal model of ADRD proposed here, in which dysbiosis may contribute to disease development under specific circumstances.166,167
Microorganisms, when they are in distant places such as mouth, nose, or intestine, may communicate with the brain by liberation of various signaling molecules. One of the best known is lipopolysaccharide (LPS). Repetitive injection of LPS in mouse models can result in the appearance of typical pathological hallmarks of AD. 168 Another means of communication is by the extracellular vesicles called outer membrane vesicles (OMVs), liberated by microbes and carrying important substances to communicate and influence target cells. 169 Injection of OMVs originating from P. gingivalis in mouse brain induced identical pathomorphological changes to human AD brains.170,171 In our recent experiments, OMVs derived from P. gingivalis were injected peripherally into zebrafish and were detected in the brain just three hours later, where they induced pronounced neuroinflammation (unpublished data). There could also be other still unknown pathways existing between latent infections and the brain.
There are many ways organisms combat infection at the cellular level. The infected cell could intentionally trigger its death in order to not propagate the infection (e.g., apoptosis) but could also elicit its antimicrobial machinery.172–175 Neurons are no exception, and they secrete many different antimicrobial peptides such as Aβ, α-synuclein, and tau.176–180 These small molecules are well known in plant biology and are used in a similar fashion to bacterial curli to kill other bacteria.181–184 The first demonstration that Aβ is antimicrobial came from the Tanzi group demonstrating that it can kill bacteria and fungi. 34 Others, including our group, have related this finding to Aβ antiviral activity.29,30,185 The mechanism by which it can “kill” viruses was also elucidated. These findings highlighted Aβ's physiological role.186,187 The same was also demonstrated for tau and α-synuclein, the latter being the typical protein characterizing Parkinson's disease.176–180 These data unify the causal theory of AD by integrating the amyloid hypothesis in the infection hypothesis, with Aβ deposition as a consequence of multiple infectious insults during life, particularly chronic latent infections. 140 The crucial implication of these findings is that, while Aβ may under some circumstances contribute to ADRD pathology, it may under others be a crucial mechanism of antimicrobial control and protection. Precisely distinguishing the two may be a nearly impossible task, and this may further explain the limited efficacy of anti-Aβ therapies, which may produce benefits in some individuals but harms in others.
The second important effect of infection is to initiate the activation of the peripheral and brain innate immune system, resulting in neuroinflammation which largely precedes Aβ deposition. This inflammation, being sustained by the various byproducts of the infective agents, may result in neuronal destruction without the presence of an active acute infection.44,188,189 All the events or processes described above lead to a vicious cycle, starting with an infection resulting in inflammation and Aβ production which in turn, will sustain inflammation during reactivations.190,191 This suggests that AD may, at least in part, result from the chronic accumulation of noxious inflammatory events in the brain. Under normal conditions, such local neuroinflammation may persist at low levels throughout life with minimal adverse effects. However, repeated exposures to infections or other insults can trigger acute inflammatory responses that become particularly severe and sustained in the elderly. 192 Over time, these insults can compromise the integrity of the BBB, allowing brain-derived inflammatory mediators to enter the periphery, stimulating systemic innate and adaptive immune responses, and enabling peripheral inflammatory signals to access the brain. 193
Neuroinflammation can also originate from the periphery, where infections in various tissues or disturbances in any body microbiome trigger systemic inflammation. Microbial byproducts, such as infectious peptides, curli, free radicals, and pro-inflammatory cytokines, can reach the brain via hematogenous or neuronal pathways, where they activate glial cells. This leads to the stimulation of microglia and astrocytes, initiating and perpetuating neuroinflammation. Microglia serve as the first line of innate defense in the brain and play a critical role in maintaining homeostasis. However, upon activation, microglia adopt a pro-inflammatory M1 phenotype characterized by cytotoxic activity and the production of inflammatory mediators such as IL-1, IL-6, IL-12, TNFα, reactive oxygen species (ROS), and nitric oxide.194,195 Chronic or repeated M1-type activation suppresses microglial phagocytic function, allowing the persistence of microbes, dead cells, protein aggregates, and other soluble antigens in the CNS. 196 The accumulation of Aβ plaques, combined with pro-inflammatory factors released by microglia, further drives and sustains the neuroinflammatory response, ultimately leading to neuronal death and contributing to AD pathology. 197 Regardless of the initial trigger (central or peripheral), the outcome is the same: sustained neuroinflammation that damages neurons and synaptic connections, resulting in neurodegeneration.
In this context, a key player is TREM2, a receptor predominantly expressed in microglia that regulates their proliferation, survival, migration, and phagocytic activity. TREM2 and its ligands have CNS protective effects in AD by modulating microglial functions and promoting the clearance of neurotoxic substances, including Aβ.198,199 In addition, TREM2 plays a critical role in regulating inflammatory responses and various cell signaling pathways that influence microglial immune and regulatory functions. 199 Consequently, certain TREM2 variants are associated with an increased risk of AD, representing an important genetic risk factor similar to ApoE.200,201
Inflammaging per se and all the immunological history from birth to aging (immunobiography) shaped mostly by various successive infections will together contribute to neuroinflammation and will lead to AD in conjunction with other contributing factors. 202 Chronic neuroinflammation is therefore the common endpoint for all infections in the brain to perpetuate the pathology and the progression of AD.
Epidemiological evidence for a role of infections in ADRD
Epidemiological studies have sometimes been viewed skeptically due to potential limitations, including their retrospective design, variability in AD diagnosis, and reliance on treatment records. However, many recent independent studies have addressed these limitations in various ways, and the growing convergence of results is increasingly difficult to ignore. Several studies have reported associations between antiviral treatment and reduced AD risk, as well as between HSV-1 infection and AD development across diverse populations and countries, helping to mitigate some of the aforementioned limitations. For example, a recent large-scale, nationwide cohort study of over 120 million U.S. patients found a significant link between HSV-1 or HSV-2 infection and increased risk of AD (odds ratio = 2.44), 166 while antiviral medications appeared to provide protective effects. These findings suggest that targeting the underlying mechanisms of HSV infection could potentially reduce dementia risk in HSV-infected individuals. Another recent study reinforced the association between HSV-1 infection and AD. 203 Moreover, vaccination appears to contribute to a decreased incidence of AD, providing quasi-experimental evidence. The protective effect seems to increase with the number of vaccines administered, including those given in early childhood. 204 Our recent meta-analysis further supports the role of vaccination in lowering dementia risk (Maggi et al., in press). Additionally, large epidemiological studies from Taiwan, Sweden, and France have shown that even short-term HSV-1 treatment with acyclovir, for as little as 10 days, is associated with a reduced incidence of AD.205–207 While individual epidemiological studies have limitations, many studies provide compelling evidence. For instance, a large Taiwanese cohort of 33,448 individuals found that HSV-1 infection was associated with a > 2.5-fold increased risk of dementia, 207 an effect too large to be explained by residual confounding. Treatment with acyclovir reduced this risk by over 90%, highlighting the quasi-experimental strength of such studies and demonstrating that epidemiological research can provide more than merely correlative insights.
Lines of evidence that infections can cause ADRD
Under the model proposed here, multiple lines of evidence support a role for infections in the pathogenesis of many cases of sporadic ADRD. First, experimental studies in model organisms demonstrate that infections can trigger ADRD-like pathology and symptoms.111,112 Second, in humans, chronic infections co-occur with ADRD more frequently than expected by chance.65,67,136,146 Third, as discussed above, a mechanistic framework now explains many counterarguments to the infection hypothesis, including why predominantly chronic latent infections contribute to ADRD, why effects vary across individuals and contexts, and how Aβ and neuroinflammation remain central to disease processes. Fourth, epidemiological studies increasingly converge, showing protective effects of vaccination, antiviral treatments, or absence of infection.204–209 Together, these lines of evidence suggest a nuanced picture: rather than a single infectious agent causing ADRD, multiple types of infections and microbes interact with the immune system, genetics, and environmental factors to contribute substantially to ADRD etiology.
Why it is so difficult for the infection hypothesis to be accepted by the Alzheimer research community?
Despite these various types of direct proof of a causal relationship between infections and AD, this conclusion has not been fully accepted and consequently translated into clinical practice. This might be the consequence of the modest success of the clinical trials using the anti-Aβ antibodies creating an overemphasized hope in a therapeutic desert. The recent acceptance by the Food and Drug Administration (FDA), after decades of research, of the two monoclonal antibodies as disease-modifying drugs diverted attention from more fundamental causes of AD. While an acute infection occurring decades before the clinical onset of AD is unlikely to be causal, relapsing, repeated, or chronic infections can plausibly contribute. Nevertheless, dozens of articles have emerged during the COVID-19 pandemic attempting to explain how acute SARS-CoV-2 infection might lead to or increase the risk of AD. Moreover, it is always pointed out that no clinical trials to prevent or treat AD have been successful, neither with antiviral nor antibiotics nor anti-inflammatory drugs.210–213 The only exception appears to be the study by Loeb et al., in which treatment with antibiotics (rifampin and doxycycline) led to clinical improvement. 214 Unfortunately, the recent failure of valacyclovir to produce clinical benefits in patients with mild-cognitive impairment (MCI) has reinforced this critique. While these results are valid, they are largely consistent with the predictions of the model proposed above, which emphasizes the complexity, timing, and multifactorial nature of infections in AD pathogenesis. The trials were done mostly too late, in patients whose AD was quite advanced—and probably not in sufficient dosage and time. Once the disease has progressed to AD, it could be very difficult to mitigate the long-term effect of persistent infections. These treatments should be done in a preventive way, when the damage has not yet led to chronic inflammation and Aβ can be eliminated. The ADRD research community is still too focused on the consequences of AD (Aβ and p-Tau) rather than the causes that led to the production of these final pathological hallmarks. Finally, the ADRD research community often remains entrenched in a reductionist paradigm, focusing on single pathways with discrete causes and effects in the hope of identifying a “silver bullet” treatment, even though it is now recognized that AD arises from complex interactions between genetic and environmental factors. 215 The body of evidence accumulated to date suggests that this approach is unlikely to be fruitful, highlighting the urgent need for a more integrative and multifactorial framework.
What would it take to accept the infectious hypothesis?
The scientific community should first accept that an old paradigm or hypothesis might be replaced or better complemented by a new theory. An open-minded, innovative community should consider the possibility they may be wrong, and that patients’ interests are furthered by testing a diversity of approaches and hypotheses. There are already ample experimental, clinical, and epidemiological data to support the infection hypothesis as one of the causes of AD.216–218 As scientists and clinicians increasingly accept the data and mechanistic theory supporting this infection hypothesis, much more intensive research and clinical trials will be initiated to tackle this new paradigm. 219 The community should also be firmly convinced that the infection hypothesis is valid and should not present it only as a possibility, but as an established cause of AD/ADRD. 219 Funding agencies and pharmaceutical companies should also support this research endeavor by new approaches and clinical trials towards the prevention of AD. Researchers working on the infection hypothesis should also integrate the pathogenesis of all other AD contributing factors, as all these mechanisms may interact. Thus, an apparent weakness in one specific pathway will not dismantle or discredit the whole hypothesis. Therefore, a constructive and timely dialogue should be established between researchers studying the traditional hallmarks of AD and those investigating alternative pathways, such as infections, to accelerate progress and ultimately benefit patients.
What would a paradigm shift mean for the prevention and treatment of Alzheimer's disease?
Once the infectious origins hypothesis is accepted, major research efforts should be devoted at various levels to arrive quickly at clinical approaches to treat and prevent AD(RD). From the basic research aspect, the use of 3D minibrain human models with various infections combined or not with other AD risk factors is critical. 220 This human model may be complemented with choroid plexus, blood-brain barrier and human immune cells to mimic the human brain. The study of the extracellular vesicles (EVs) and OMVs in this experimental setting and from patients with various stages of the disease could give insight on the physiopathology. 221 The contribution of the immune system should be also studied more deeply either in the innate or in the adaptive part.
From the clinical aspect, an important point should be to build clinical trials in the disease course to find ways to prevent the development or progression of the disease. Blood biomarker-based diagnostics of ADRD are now increasingly available and have been shown to predict symptomatology onset decades in advance, in the spirit of the amyloid hypothesis based largely on Aβ and p-Tau 217.5,38,39 Most clinical trials classify patients using traditional biomarkers in the blood (since CSF collection is more invasive) along with MRI or occasionally PET scan results. Even as of 2025, the majority of trials continue to focus on the amyloid or tau hypotheses, with only a small number targeting alternative pathways such as inflammation, metabolic changes, or neurodegeneration. 222 None to date have specifically targeted infections, although one trial has explored probiotics. Notably, the FDA has cleared the investigational new drug (IND) application for LHP588 (Lighthouse Pharmaceuticals, San Francisco, CA), a next-generation small-molecule gingipain inhibitor being investigated as a potential treatment for Porphyromonas gingivalis–positive AD. LHP588 is designed to target the infectious P. gingivalis pathogen, which has been associated with certain chronic degenerative and inflammatory disorders, including AD. IND clearance was based on phase 1 human trial data demonstrating that LHP588 met safety and tolerability endpoints and achieved plasma levels sufficient to engage the P. gingivalis target with once-daily dosing. Following clearance, the FDA issued a “Study May Proceed” letter, enabling the initiation of the planned phase 2 clinical trial called SPRING. This study will be a randomized, double-blind, placebo-controlled trial assessing the safety and efficacy of once-daily LHP588 in patients with mild to moderate P. gingivalis–positive AD.
In the future, longitudinal studies should be put in place to follow various populations at different stages of the disease over decades in order to find increasingly precise biomarkers of the disease, such as predictive, diagnostic, prognostic, or response biomarkers. We also need to characterize the holistic microbiome by state-of-the-art methods at different stages of the disease from various part of the human body as well as proof-of-concept interventional studies on a multimodal basis and very early in the disease process. 223 This paradigm change could also lead to a more widespread acceptance that vaccination is one of the best ADRD prevention measures, as demonstrated recently by several studies. All possible other causes leading to AD should be integrated, including environmental pollution and ischemia. Together, these studies and joint effort between researchers and clinicians will improve the prevention of the disease for the benefit of the patients and will help identify druggable, clinically useful targets to reduce the incidence of AD. As a result, a more holistic approach will be born, integrating all the possible causes which not exclusively but concomitantly lead to disease (Figure 1). This shift will ultimately lead to prevention and better treatment of AD.
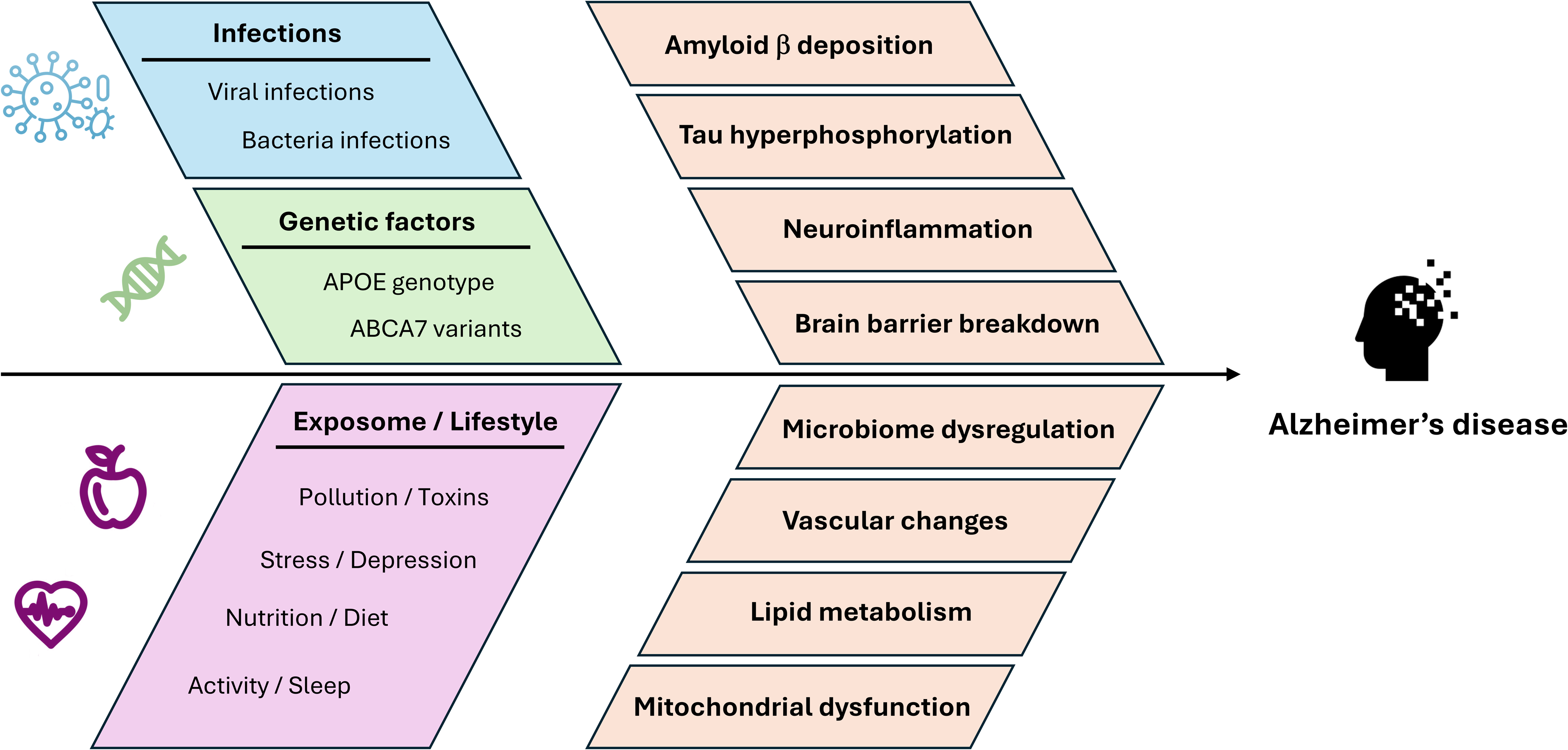
An integrated model of multimodal contributors to Alzheimer's disease.
Perspectives
There is now substantial experimental evidence supporting a shift toward an infectious origins’ paradigm for ADRD. Key lines of evidence include: (i) presence of pathogens (e.g., HSV, B. burgdorferi—see a full list in Table 1) or their pathogenic proteins (e.g., gingipain, ICP27, LPS) in the brain; (ii) indirect triggering of AD pathological hallmarks by microbes from outside the body (e.g., LPS) or from within (e.g., neurotransmitters, short-chain fatty acids, pro-inflammatory cytokines, curli); (iii) successful modulation of microbes through the MGB axis (e.g., FMT, probiotics) or direct antimicrobial treatment (e.g., syphilis, HIV); (iv) reduction of AD risk through various vaccinations, as supported by epidemiological studies; (v) prevention of infection by Aβ in AD mouse models. 32 Collectively, these findings provide a strong rationale for considering infections as a central factor in ADRD pathogenesis and for reevaluating therapeutic strategies accordingly.
Pathogens involved in AD pathogenesis.

This paradigm shift has the potential to attract greater investment from both public and private sources, enabling the rapid development of clinical trials aimed at preventing or slowing the progression of AD, guided by predictive biomarkers in a multimodal approach. Future trials should be longitudinal, reflective of real-world settings, and personalized, capturing the full clinical spectrum of AD. To achieve this, state-of-the-art multi-omics approaches combined with machine learning and artificial intelligence (AI) techniques must be employed to identify reliable predictive biomarkers. Such strategies will also foster greater engagement from civil society in the fight against AD, while prompting a transformation in clinical practice. AD societies representing patients and caregivers should be mobilized to advocate for a more inclusive conceptualization of the disease, paving the way for truly disease-modifying therapies. Clinical criteria that rely solely on Aβ and p-Tau will become insufficient. Instead, a comprehensive and multi-causal approach should be developed, integrating all potential contributing factors to AD. Scientists are likely to recognize that Aβ and p-Tau represent consequences rather than the sole causes of the disease, arising from a constellation of stresses including chronic infections. Initially protective, these factors later become detrimental byproducts. Adopting this approach could transform AD research and clinical care, ultimately benefiting millions by preventing or mitigating this devastating disease.
Conclusion
In conclusion, research on the AD spectrum has reached a turning point. Targeting only the traditional pathological markers of the disease is no longer sufficient, nor can the clinical spectrum be fully defined by these markers alone. A new era should be initiated, guided by a holistic, multimodal, and multi-hit approach to understanding AD spectrum. Encouragingly, recent funding initiatives from the European community are fostering collaboration among researchers from diverse fields to investigate the impact of chronic infectious diseases on non-communicable disorders such as AD. These efforts integrate state-of-the-art experimental techniques with advanced machine learning and AI approaches, ultimately aiming to benefit the millions of patients affected by this devastating disease.
Footnotes
Acknowledgements
The authors have no acknowledgments to report.
Author contribution(s)
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: We apologize to our colleagues whose work we were unable to cite due to space limitations. This effort was supported by a grant from Fondation Antoine-Turmel to B.L. and T.F.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
