Abstract
Background
The FMR1 premutation (FXPM) is a common genetic variant (∼1:151 females) linked to increased risk for neurodegenerative disease. Midlife cognitive phenotypes remain poorly defined.
Objective
To characterize episodic memory performance in midlife FXPM women and examine potential risk moderation via genetic (i.e., FMR1 CGG repeat expansion size) and environmental (i.e., college degree attainment) influences.
Methods
Eighty-eight FXPM women and 84 matched controls, aged 30–55, completed the Loewenstein-Acevedo Scales for Semantic Interference and Learning (LASSI-L), a measure of specific episodic memory processes sensitive to subtle Alzheimer's disease (AD)-related cognitive and neuropathological changes.
Results
FXPM women demonstrated deficits in proactive semantic interference (PSI), recovery from PSI, and intrusion errors relative to controls. College education buffered these effects: college-educated FXPM women performed comparably to controls, while those without a college degree showed deficits. Gene-environment interactions showed patterns consistent with differential susceptibility: LASSI-L performance in women with mid-range CGG repeats (∼80–100) was strongly influenced by educational attainment, whereas education effects were absent in women with lower/higher CGG lengths.
Conclusions
Midlife FXPM women showed episodic memory deficits, paralleling LASSI-L deficits seen in prodromal AD. College education offered protective benefits, particularly for women with mid-range CGG expansions. Findings highlight a critical midlife window for cognitive monitoring, identify education as a potential protective factor, and inform personalized risk assessment based on CGG length to promote earlier detection and targeted prevention for FXPM women. Findings suggest potential overlapping mechanisms with AD that merit further study.
Keywords
Introduction
The Fragile X Messenger Ribonucleoprotein-1 (FMR1) premutation (FXPM) is a prevalent genetic variant caused by an expansion of 55–200 CGG repeats on FMR1, estimated in 1:151 females and 1:468 males. 1 Because the FXPM is more than twice as common in females, delineating clinical effects in women is a priority. The FXPM is linked to increased disease burden in women, as reflected by heightened risk for conditions such as premature menopause, depression, anxiety, migraines, chronic pain, thyroid disease, and sleep disorders. 2 Additionally, ∼16% of FXPM women develop fragile X-associated tremor/ataxia syndrome (FXTAS), a late-onset neurodegenerative disorder characterized by tremor, gait ataxia, and cognitive decline. 3 Notably, FXPM women, but not FXPM men, can pass fragile X syndrome to their children, adding complexity to the female FXPM experience as caregiving for a disabled child is a cause of chronic stress that can worsen health. 4
Cognitive and memory dysfunction in FXPM women
Despite the high prevalence and associated health risks, the neuropsychiatric phenotype of the FXPM in women remains undefined. Initially considered “asymptomatic carriers”, FXPM women showed no global neuropsychiatric differences in early systematic reviews and large cohort studies.5–7 However, domain-specific cognitive vulnerabilities have since been noted, including disinhibition,8–11 attention problems,12,13 and cognitive-linguistic deficits,14–16 with neuroimaging evidence supporting alterations in associated brain regions including the hippocampus, prefrontal cortex, and cerebellum.17,18 This literature has primarily focused on young/midlife adults, suggesting FXPM women may experience cognitive deficits decades before the typical age of FXTAS onset in the early 60s. 19 Moreover, growing cross-sectional and longitudinal evidence suggests that these cognitive problems progress with age,9–11,14,16,20 calling attention to cognitive dysfunction as a potential prodrome of FXTAS with implications for early detection and intervention.
Memory impairment, particularly in the episodic memory processes of encoding and retention, is observed in FXTAS across early and late stages of disease.21–23 Early evidence, based on males, also suggests that the FXPM could increase risk for dementia up to 6-fold, irrespective of FXTAS comorbidity. 24 However, research on episodic memory in FXPM women is sparse, which limits clinical understanding of the cognitive limitations experienced by these individuals. One study reported intact initial learning with impaired long-delay and marginal short-delay recall differences in a sample of 19 FXPM women, 25 while another found no differences in short or long-delay recall in 25 FXPM women compared to controls. 26 Thus, data on episodic memory on small samples of women is inconclusive. However, an intriguing case series by Tassone et al. 27 presented neuropathological findings from eight FXPM women, with detection of a high rate of Alzheimer's disease (AD)-type cortical amyloid-β plaques and neurofibrillary tangles—present in half of the women autopsied, with and without corresponding clinical AD signs and/or FXTAS. Given that episodic memory impairment is a hallmark feature of AD and amnestic mild cognitive impairment (aMCI; the prodromal stage of AD), preliminary evidence of AD neuropathology in FXPM women underscores the need to better delineate the episodic memory profile of females.
Genetic and environmental influences on cognitive dysfunction
Most studies of FXPM men have suggested a positive linear relationship between CGG length and the severity of various cognitive problems,24,28–30 with nonlinear patterns reported only rarely. 31 In contrast, research on FXPM women has shown nonlinear CGG associations, with risk for psychiatric, health, and reproductive symptoms increasing at mid-range CGGs of ∼80–100 repeats and decreasing at lower or higher CGG repeat lengths.32–35 Early evidence suggests similar curvilinear patterns of CGG moderation in relation to certain cognitive skills, including inhibition and language fluency,9,15 but there remains a need to establish CGG risk patterns in relation to distinct neurocognitive functions such as episodic memory, which differ in their associated brain circuitry and mechanistic influences.
It has been long recognized that exposure to environmental stressors can compound FXPM genetic vulnerability, exacerbating clinical symptoms.36,37 However, the interplay between genetic and environmental risk in the FXPM may be more complex than once thought, with recent work suggesting that environmental influence may vary by CGG repeat length. This research suggests that sensitivity to the environmental context is heightened at mid-range CGG repeats, with environmental influences playing a larger role (positive as well as negative) in shaping the health of FXPM women with mid-range CGG repeats than those with lower or higher repeat lengths.38–41 This new understanding of how CGG length may moderate plasticity to environmental influence could provide a pathway to improve clinical care by identifying at-risk subgroups based on CGG length and directing environmental risk reduction strategies towards individuals genetically primed to benefit from them. One modifiable environmental factor that may be particularly salient to the cognitive outcomes of FXPM women is educational attainment. In the general population, higher education has been found to reduce risk for cognitive decline and dementia, presumably by increasing brain resilience to age/disease-related changes, or cognitive reserve.42,43 Few studies have addressed educational neuroprotection in the FXPM, but a recent report by Hong et al. 44 documented milder and slower progressing FXTAS-type motor/cognitive symptoms in FXPM women with college degrees, even after accounting for confounding socioeconomic and lifestyle factors such as income and health behaviors. This finding suggests that higher education could buffer risk for FXPM neurodegenerative phenotypes.
The present study
This study sought to characterize cognitive dysfunction in episodic memory processes in FXPM women, via novel application of the Loewenstein-Acevedo Scale for Semantic Interference and Learning (LASSI-L), an assessment of initial and continuing proactive semantic interference in memory and learning that is sensitive to subtle cognitive changes in neurodegenerative disease, particularly those observed in prodromal AD. The LASSI-L has been studied extensively in relation to brain atrophy and other biological markers of neurodegeneration, such as amyloid-β burden and plasma AD biomarkers,45–47 which makes its application particularly useful for understanding neurocognitive vulnerabilities associated with the FXPM genotype, where such biomarkers have been observed. We posed two research questions: (1) Do middle-aged FXPM women show deficits on the LASSI-L relative to controls, and does educational attainment moderate LASSI-L performance? (2) Is LASSI-L performance in FXPM women associated with CGG repeat length, and is this association moderated by educational attainment? We hypothesized that FXPM women would show poorer LASSI-L performance relative to controls, with college degree attainment mitigating the severity of cognitive deficits. We also anticipated a curvilinear CGG risk pattern that interacts with college degree attainment in a manner consistent with differential susceptibility, such that the influence of college education would be more pronounced among women with mid-range CGG repeats than at higher or lower repeat lengths.
Methods
Participants
Participants were n = 88 FXPM women and n = 84 control women, aged 30–55 years, drawn from a larger study on FXPM language phenotypes. Participants were fluent English speakers with normal/corrected-to-normal vision and hearing. Controls were mothers of biological children who had not been diagnosed or treated for developmental disorders, to reduce the possibility of undiagnosed FMR1 conditions in the control sample. The FXPM was confirmed via genetic testing. We also tested 79% of controls to rule out the FXPM (funding limitations precluded testing the full sample). FXTAS was not an exclusion criterion, but no participants reported having a FXTAS diagnosis.
FXPM women were recruited nationally in the US through advertisements on social media, parent groups, word-of-mouth, the National Fragile X Foundation, and the Fragile X Research Registry of the Carolina Institute for Developmental Disabilities of the University of North Carolina-Chapel Hill. Controls were recruited through advertisements on social media, word-of-mouth, and flyers posted within the local community. The groups did not differ in age, race, education, income, marital status, number of children, or nonverbal IQ (Table 1). FXPM women were mothers to children with fragile X syndrome (70%), FXPM (8%), children without FMR1 expansions (13%), or did not have children (9%). Because the groups were not matched on caregiver status, sensitivity analyses were conducted to rule out caregiver status as a potential confound (see Data analysis).
Group characteristics.
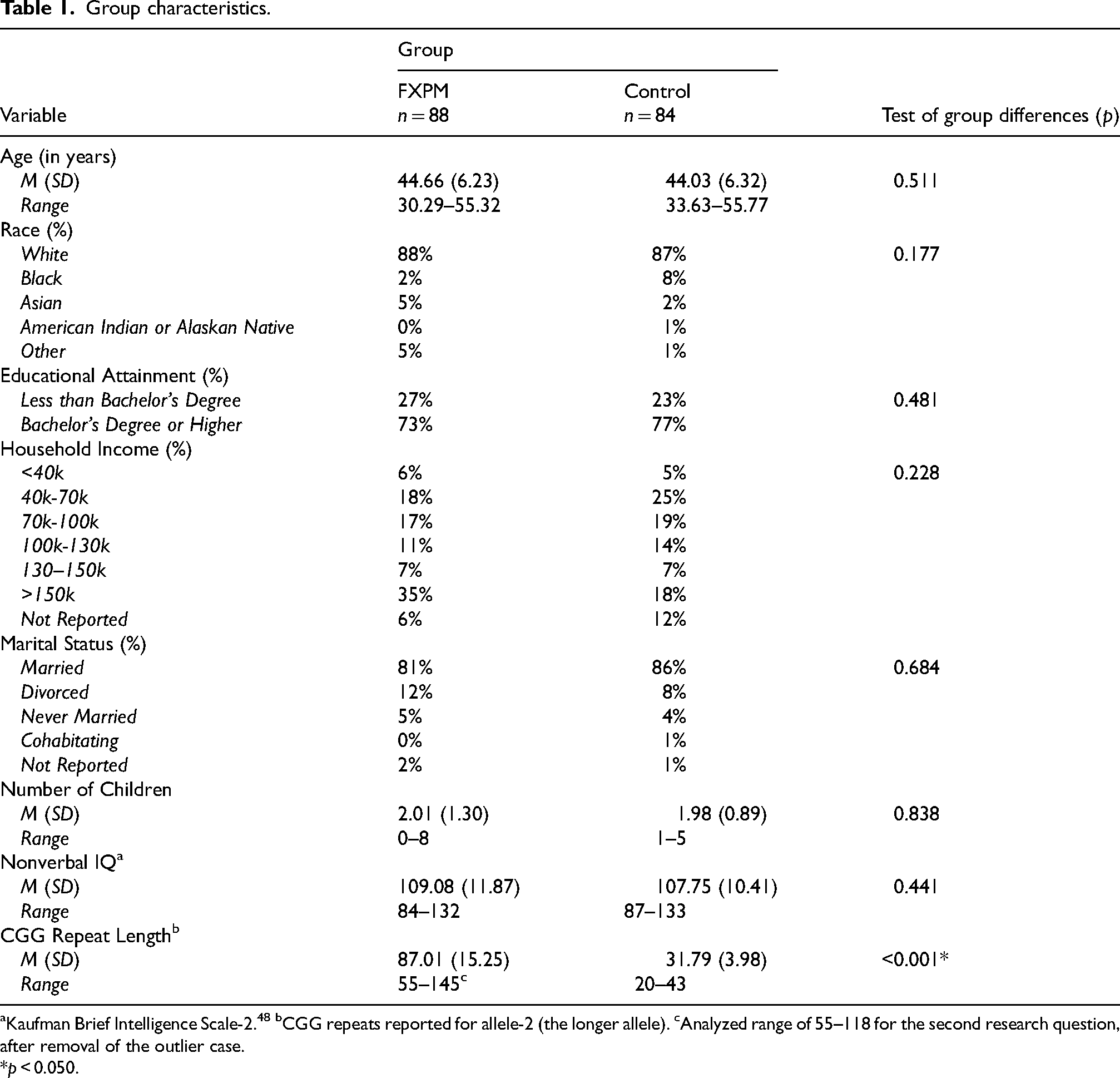
Kaufman Brief Intelligence Scale-2. 48 bCGG repeats reported for allele-2 (the longer allele). cAnalyzed range of 55–118 for the second research question, after removal of the outlier case.
*p < 0.050.
Procedures
Assessments spanned about four hours across two sessions and were conducted in-person in a university laboratory or remotely via videoconferencing (Zoom Communications Inc) during the COVID-19 pandemic. Telehealth comprised 68% of assessments, with similar proportions across groups (χ2 = 0.139, p = 0.709). Telehealth assessments were completed from a quiet room, using provided noise-reducing headphones with attached microphone. Across both assessment modes, measures were administered in a standardized order with the LASSI-L at the beginning of the second session. Compensation and travel reimbursement were provided. Procedures were approved by the Institutional Review Board of the University of South Carolina (Pro00067288, approval date 6/13/2017). Participants provided written informed consent.
Measures
Loewenstein-Acevedo Scale for Semantic Interference and Learning (LASSI-L). The LASSI-L49,50 is a verbal learning paradigm that measures interference in learning and memory. The paradigm involves encoding and recall of two competing word lists, allowing for the evaluation of the episodic memory processes of proactive semantic inference (old learning interfering with new learning), failure to recover from proactive semantic interference (persistent interference of old learning with new learning) and semantic intrusions (failure to inhibit non-target words). The LASSI-L is sensitive to subtle cognitive changes in a range of neurodegenerative conditions, but has been studied most extensively in prodromal AD. It discriminates aMCI from healthy aging with high sensitivity (0.86–0.88) and specificity (0.82–0.92),49,51–53 and is correlated with AD neuropathological changes, such as amyloid-β load and atrophy in brain regions vulnerable to AD.54–60 Psychometric properties are strong, including test-retest reliability and concurrent, discriminative, and construct validity.61,62
LASSI-L administration begins with presentation of 15 words from three categories (i.e., fruits, musical instruments, articles of clothing); List A. Each word is presented typed in black on a white background for four seconds while participants read the words aloud. Participants then freely recall all words (60 s allotted) and are cued to recall the words in each category (20 s per category). Then, presentation of List A and category-cued recall are repeated (A2 Cued Recall). Next, List B, 15 new words from the same categories, is presented and free recall and category-cued recall (B1 Cued Recall) is completed. Fewer target words recalled in B1 Cued Recall indicates increased proactive semantic interference (PSI). Finally, presentation of List B and category-cued recall (B2 Cued Recall) is repeated. Failure to recover from PSI (frPSI) is marked by fewer target words recalled in B2 Cued Recall. Semantic intrusion errors (i.e., incorrect, non-target words) are tallied during B1 and B2 Cued Recall, and the percent of intrusion errors (PIE) is computed by dividing the number of intrusions by the total number of correct and incorrect words recalled. The LASSI-L was adapted for telehealth by presenting stimuli virtually through screensharing over Zoom. While data collection for the present study was initiated prior to its development, a computerized version of the LASSI-L has since been validated for telehealth with high test-retest reliability and discriminant validity. 63
CGG repeat length. CGG repeat sizing of FMR1 5`-UTR was conducted on DNA derived from buccal cells via FMR1 triplet repeat-primed polymerase chain reaction (PCR) with capillary electrophoresis using the Asuragen AmplideX® PCR/CE FMR1 kit. Alleles in the premutation range had confirmatory testing and activation ratio determination using the Amplidex® mPCR FMR1 kit. Allele-2 CGG repeat length (the longer allele) was used in analysis. Two FXPM participants were missing CGG data due to insufficient DNA.
Data analysis
Analyses were conducted using SAS 9.4 (SAS Institute, 2013). Descriptive statistics were examined. The distributions of PIE were right skewed, so transformation of λ = 0 was applied. However, inference was the same using the original variables, so the untransformed data are presented. The first research question was tested with general linear models including group, education (college degree attainment/non-attainment) and their interaction as predictors of LASSI-L performance. Age (centered at 45), race (collapsed into levels of White/Black/Asian/Other, to ensure sufficient cases at each level), and assessment mode (telehealth/in-person) were covaried. Significant interactions were followed by interaction contrasts, with FDR correction to adjust for multiple comparisons. 64 The second research question was tested with general linear models including CGG (centered at 90), education, and their interaction, as predictors of the LASSI-L, covarying for age and assessment mode. Race was considered as a covariate but did not improve fit or account for significant variance, so was not retained. Quadratic and cubic CGG terms and their interactions with education were probed and retained if they improved fit. Diagnostics identified an outlier case with 145 CGG repeats with an unduly large influence, with Cook's Distance far exceeding Di > 4/n-k-1. 65 This case was thus omitted from the models for the second research question. All other CGGs were less than 118. This was viewed as a conservative approach to ensure that patterns at high CGGs were not driven by a single, unduly influential case. 66 Effect size was investigated via partial eta squared (η2p) estimates using the conventional rule-of-thumb interpretations of 0.01 as “small, 0.06 “medium”, and 0.14 “large”. 67
Finally, sensitivity analyses assessed the potential influences of baseline intellect and caregiver status on findings. Repeating models while covarying for nonverbal IQ, or excluding non-mother FXPM women, resulted in no changes to statistical inference.
Results
Descriptive statistics
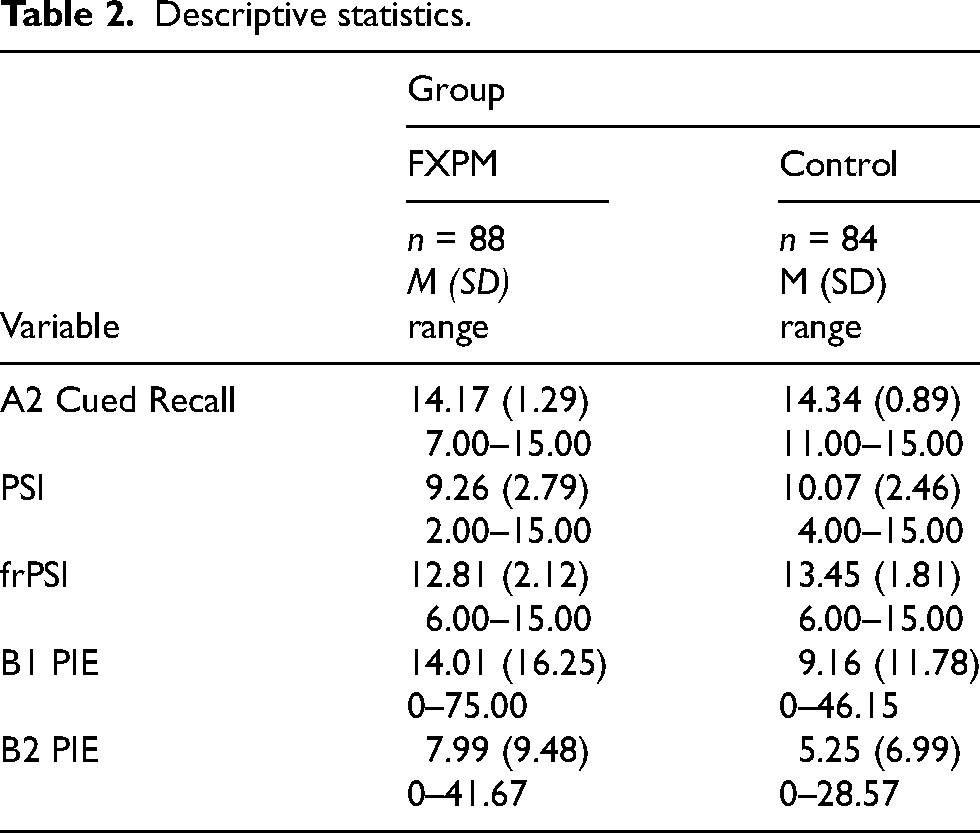
Table 2 presents descriptive statistics. The groups did not differ in A2 Cued Recall, an index of initial acquisition/encoding ability (p = 0.303, η2p = 0.01).
Descriptive statistics.
Group and education effects on LASSI-L performance
Proactive semantic interference and recovery. Significant main effects for group indicated that FXPM women performed worse, on average, than controls on PSI (p = 0.001, η2p = 0.06) and frPSI (p = 0.001, η2p = 0.06); Table 3. Group-by-education interactions for PSI (p = 0.004, η2p = 0.05) and frPSI (p = 0.010, η2p = 0.04) were also significant. FDR-corrected interaction contrasts showed that FXPM women without college degrees performed worse on PSI and frPSI than FXPM women with college degrees (p = 0.005, η2p = 0.05; p = 0.007, η2p = 0.05, respectively), controls with college degrees (p = 0.003, η2p = 0.06; p = 0.002, η2p = 0.07, respectively), and controls without college degrees (p = 0.002, η2p = 0.07; p = 0.002, η2p = 0.07, respectively). In contrast, FXPM women with college degrees did not differ on PSI or frPSI from controls with college degrees (p = 0.794, η2p < 0.01; p = 0.534, η2p < 0.01, respectively) or controls without college degrees (p = 0.239, η2p = 0.01; p = 0.301, η2p = 0.01); see Figure 1. Education effects were specific to the FXPM; control performance did not differ by college education (ps > 0.269, η2ps < 0.01).
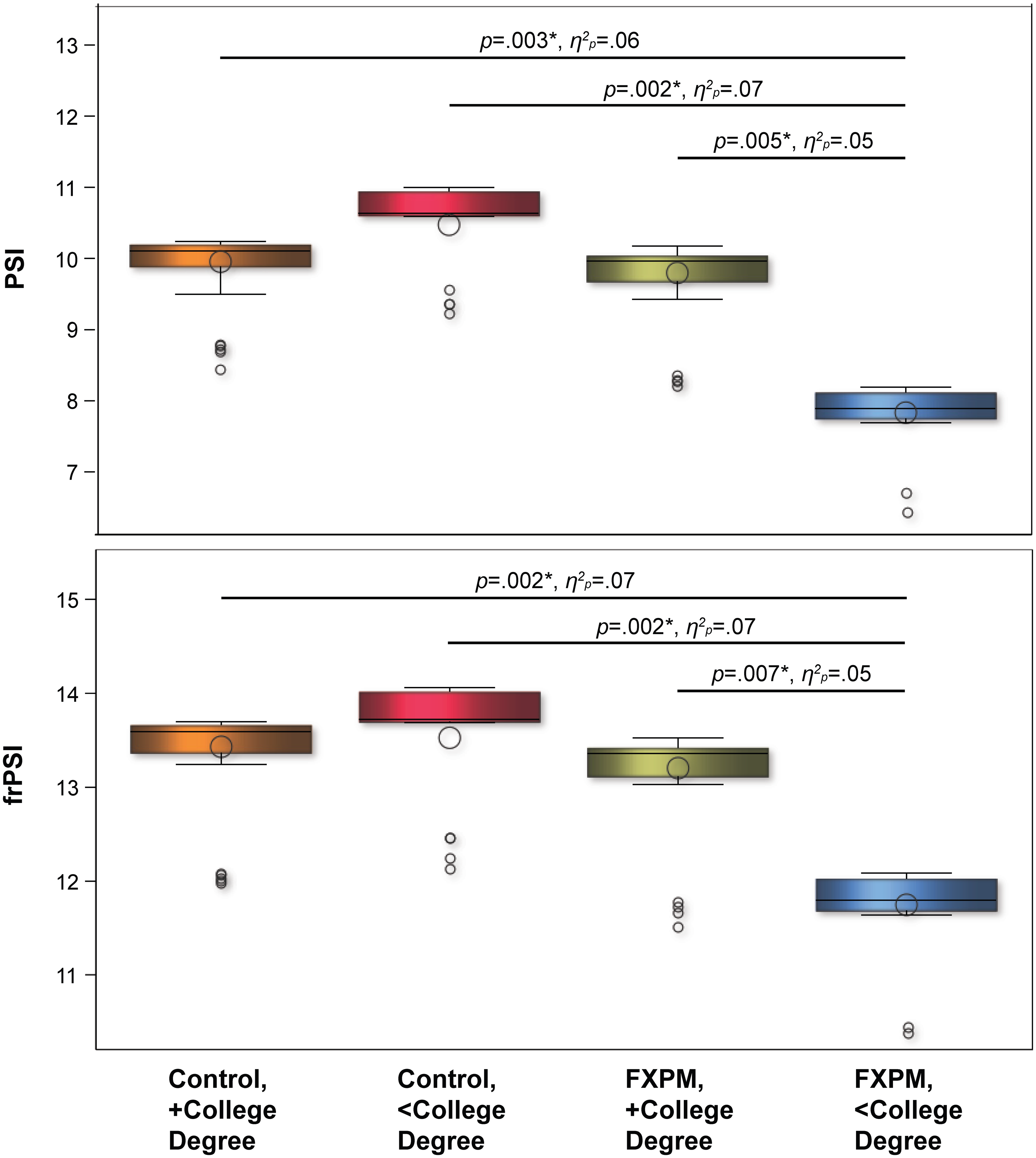
College education buffers FXPM genetic risk for proactive semantic interference and recovery deficits. Model-predicted values shown to account for covariates. *p < 0.050.
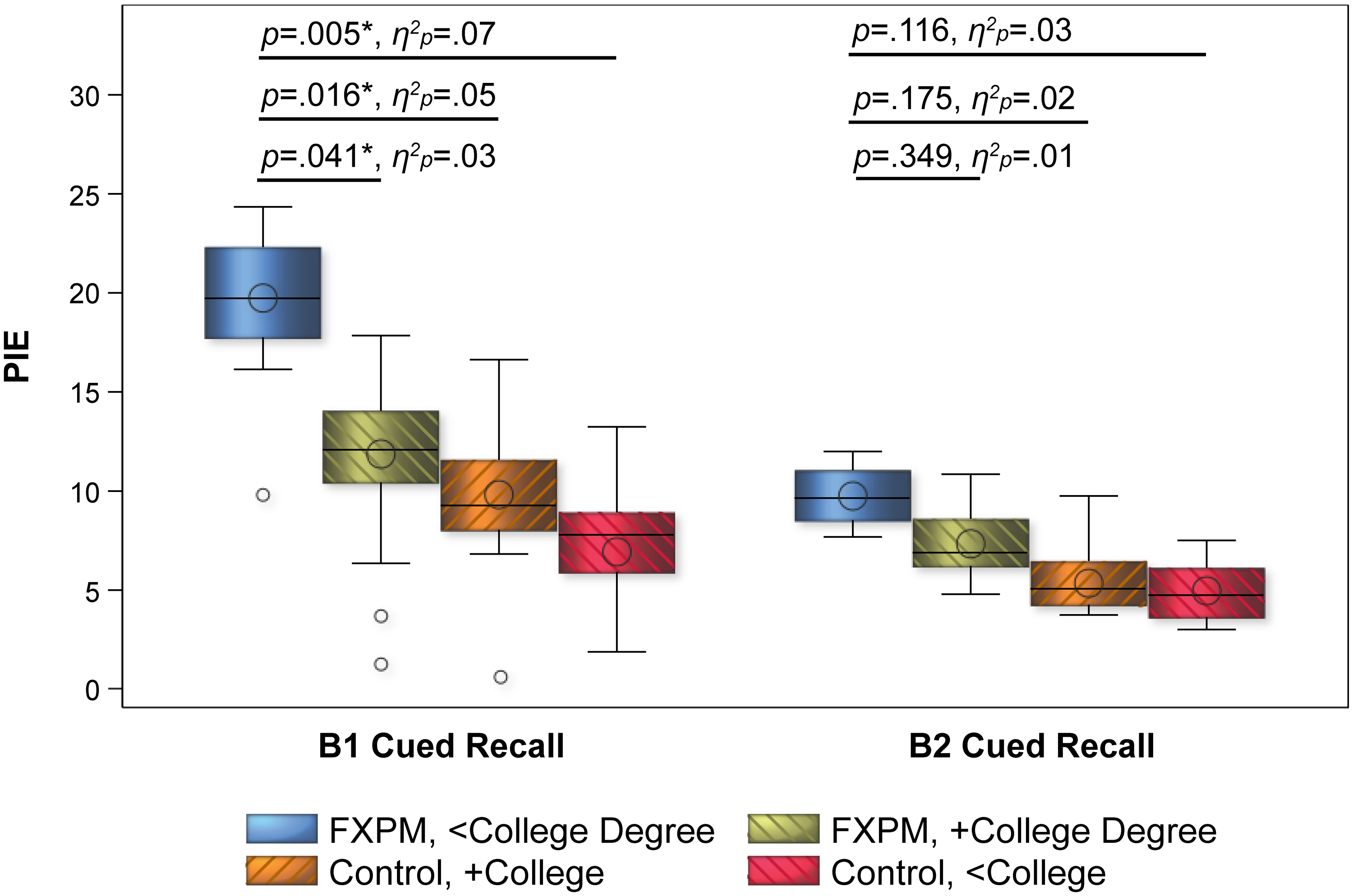
College education buffers risk for semantic intrusion errors in FXPM women. Model-predicted values shown to account for covariates. *p < 0.050.
Model results: group and education effects on PSI and frPSI.

*p < 0.050.
Intrusion errors. Group had a significant main on B1 PIE (p = 0.001, η2p = 0.06) and B2 PIE (p = 0.014, η2p = 0.04), with significantly higher (i.e., worse) PIE in FXPM women; Table 4. The group-by-education interaction for B1 PIE was also significant (p = 0.010, η2p = 0.04), with patterns mirroring the PSI and frPSI results. As shown in Figure 2, FDR-corrected interaction contrasts indicated that FXPM women without college degrees had significantly higher B1 PIE (M = 20%) than FXPM women with college degrees (M = 12%; p = 0.041, η2p = 0.03), controls with college degrees (M = 10%; p = 0.016, η2p = 0.05), and controls without college degrees (M = 4%; p = 0.005, η2p = 0.07). In contrast, college-educated FXPM women performed similarly to controls with college degrees (p = 0.524, η2p < 0.01) and controls without college degrees (p = 0.099, η2p = 0.02). Control performance did not differ by college education (p = 0.193, η2p = 0.01).
Model results: Group and education effects on PIE.
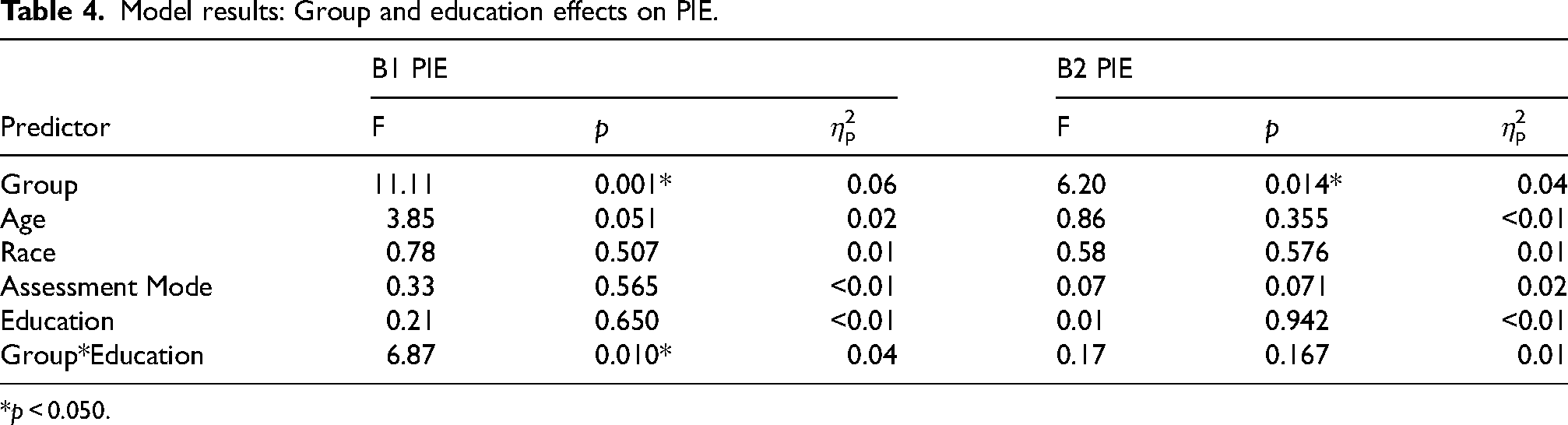
*p < 0.050.
CGG and education effects on LASSI-L performance in FXPM women
Quadratic CGG models including lower and higher-order interactions with education demonstrated the best fit for PSI (F[7,77] = 2.77, p = 0.013, R2 = 0.20) and frPSI (F[7,77] = 3.08, p = 0.006, R2 = 0.22); Table 5. Education-by-quadratic-CGG interactions for PSI (p = 0.035, η2p = 0.06) and frPSI (p = 0.001, η2p = 0.08) indicated that the influence of college degree attainment varied by CGG length following a U-shaped curve; Figure 3. FDR-corrected interaction contrasts tested education effects at CGGs representing mid-range (90), low (70), and high (110) repeat lengths. At 90 CGGs, FXPM women with college degrees outperformed those without college degrees on PSI (p = 0.002, η2p = 0.14) and frPSI (p = 0.002, η2p = 0.15). Performance did not vary by college degree attainment at 70 CGGs (ps > 0.914, η2ps < 0.01) and 110 CGGs (ps > 0.811, η2ps < 0.01). No CGG associations were observed for PIE.
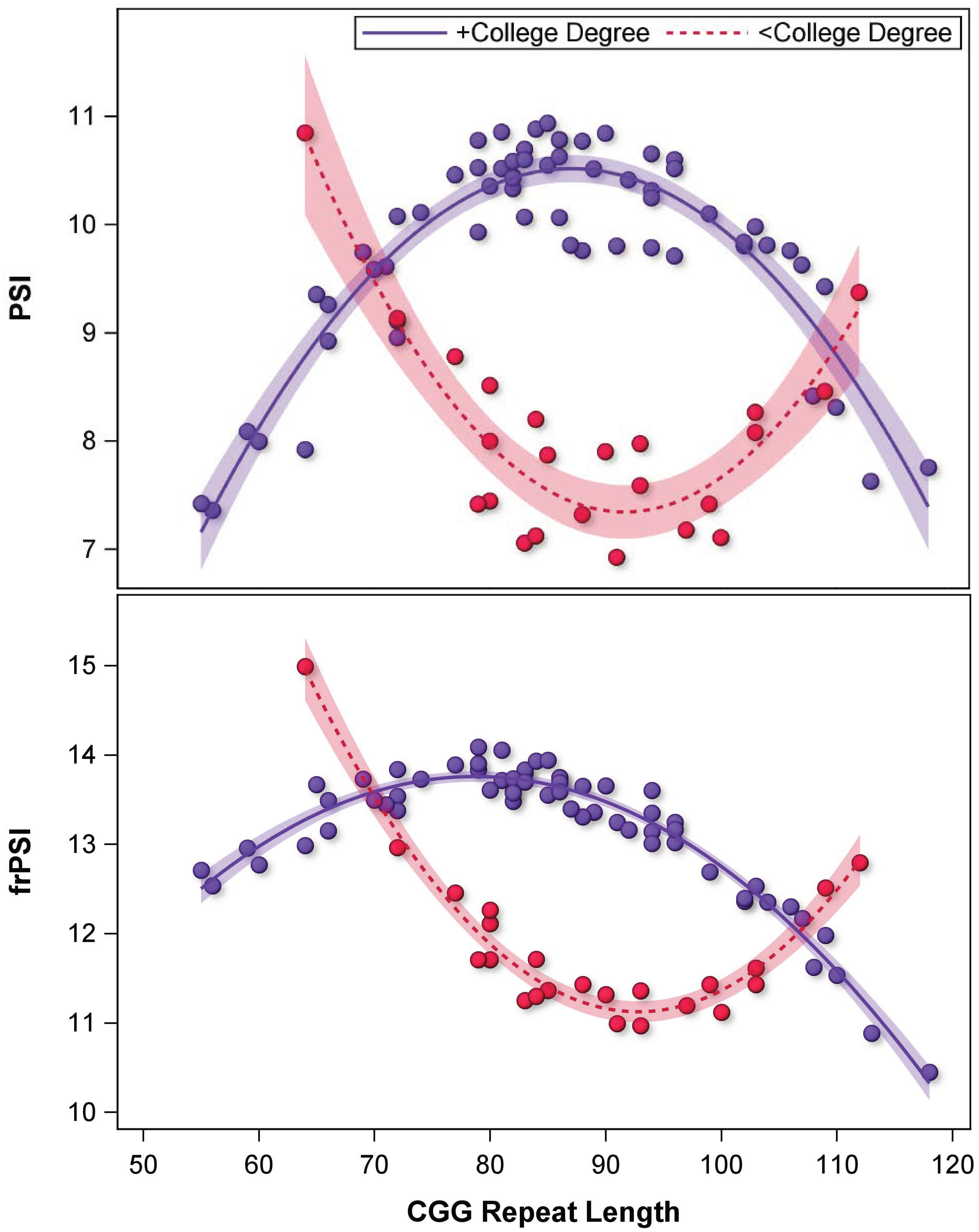
Sensitivity to the influence of college education in FXPM women is heightened at mid-range CGG length. Model-predicted values shown to account for covariates. *p < 0.050.
Model results: CGG and education effects on PSI and frPSI in FXPM women.

*p < 0.050.
Discussion
The LASSI-L revealed subtle cognitive deficits in FXPM women aged 30–55 years, suggesting midlife cognitive differences that may signal early-stage neurodegenerative processes. As a group, FXPM women performed worse than controls on indices of PSI, recovery from PSI, and semantic intrusions that tap cognitive processes involved in episodic memory. However, college education emerged as a major neuroprotective factor that rescued FXPM women from cognitive deficits. Moreover, the effect of education varied across CGG length, following a gene-environment interaction pattern consistent with differential susceptibility to the environmental context. Specifically, women with mid-range CGGs showed heightened plasticity to environmental shaping, demonstrating significant risk-reduction in association with college degree attainment (and, conversely, risk-enhancement in association with non-attainment), whereas midlife women with higher and lower CGGs performed similarly on the LASSI-L regardless of their educational background. Findings point to education as a potential modifiable factor for FXPM women, which might be applied within personalized medicine framework to target early detection and risk reduction towards CGG-defined at-risk women. Notably, the observed LASSI-L deficits have been associated with early cognitive and pathophysiological changes of AD in prior literature, which may suggest a role of interacting AD disease pathways in the cognitive expression of FXPM neurodegenerative disease.
LASSI-L indicates specific episodic memory deficits in FXPM women during midlife
The LASSI-L was highly effective at differentiating the cognitive performance of FXPM women from that of controls, particularly in those who had not attained a college degree. Notably, these cognitive differences were evident at 30–55 years, suggesting that FXPM women may begin to experience subtle cognitive decline during midlife, decades prior to the typical age of FXTAS onset. Evidence of midlife cognitive dysfunction in FXPM women has implications for clinical care. Brain aging research suggests that midlife could mark a turning point in the neural trajectory of cognitive decline, with structural and functional brain changes emerging by the fourth decade of life, well before overt cognitive symptoms become apparent.68,69 Building on this research, and the fact that LASSI-L can detect subtle deficits in midlife, future research and intervention in this population could pursue not only higher education as a preventative strategy, but also lifestyle changes.70,71 Considering that FXPM women are at known risk for neurodegenerative disease, and subtle cognitive differences have been observed as early as the 40 s, clinical monitoring—beginning in midlife—may be warranted.
FXPM women showed deficits across PSI, frPSI, and PIE, which tap cognitive processes involved in episodic memory such as encoding, storage, and retrieval. 72 This suggests episodic memory deficits are a component of the FXPM neuropsychological phenotype in females, consistent with Shelton et al. 25 Memory deficits have been studied more extensively in FXPM men, who show encoding and retrieval differences.21–23 Interestingly, however, episodic memory deficits in men are thought to be better accounted for by weak initial acquisition, rather than specific deficits in long-term storage or retrieval, per se. This study does not support initial acquisition deficits in FXPM women, as A2 Cued Recall did not differ from controls. While methodological differences may contribute to discrepant findings across males and females, sex differences in the FXPM neurocognitive profile would not be unexpected. Sex differences in cognitive function are well-documented across both healthy and pathological aging, with differences arising from multifactorial genetic, epigenetic, hormonal, physiological, and environmental influences. 73 Regarding the FXPM, it is notable that FXPM women experience disproportionate sex/gender-specific risk factors for cognitive decline, such as premature menopause, depression, and chronic parenting stress. When considering these clinical risk factors, occurring alongside sex-specific genetic influences, divergent FXPM neurocognitive profiles across the sexes are highly plausible. This underscores the need for further research focused specifically on the FXPM neurocognitive profile in females.
LASSI-L deficits suggest potential overlap with AD
The LASSI-L has been well-validated as a tool that is sensitive to cognitive, neural, and pathological changes in early AD. Specifically, the persistent semantic inference effects marked by frPSI and intrusion errors elicited during conditions of frPSI (i.e., B2 PIE) are considered a defining feature of aMCI, the prodromal stage of AD. frPSI is sensitive to the presence of underlying AD pathophysiology, with moderate-to-strong correlations with AD biomarkers such as amyloid-β load and Aβ42/40 ratio and atrophy in brain regions vulnerable to AD.54,57,58,72,74,75 Therefore, frPSI deficits in FXPM women might indicate cognitive phenotypes that are shared with prodromal AD. This is consistent with well-established molecular genetics evidence demonstrating that FMR1's protein, fragile X messenger ribonucleoprotein (FMRP), plays a role in regulating the synthesis of amyloid-β protein precursor which is central to AD pathophysiology. 76 Early clinical reports also suggest interaction between FMR1 and AD disease pathways. For example, risk for FXTAS increases 12-fold when FXPM carriers also carry APOE ε4, an AD risk allele 77 and high rates of AD-type amyloid-β plaques and neurofibrillary tangles have been detected in FXPM women in early case series data. 27 The finding of elevated frPSI and B2 PIE deficits in FXPM women is consistent with this growing evidence of potential interface between FMR1 and AD disease pathways.
We also documented elevated B1 and B2 PIE in FXPM women. Intrusion errors, marked by PIE, have also been shown in the literature to correlate with brain atrophy in AD-prone regions and other biomarkers of AD neurodegeneration.45,56,57,78 However, intrusions are thought to provide a more specific indicator of AD pathology than other LASSI-L indices, with B1 PIE offering stronger discriminatory and predictive power than B2 PIE.51,79 B1 PIE differentiates amyloid-β-positive aMCI from aMCI without amyloid-β-positivity45,56 and is not elevated in individuals with MCI due to other neurological disorders or individuals with MCI of non-neurological causes (e.g., depression), who perform similarly on B1 PIE as cognitively-normal controls. 80 Therefore, elevated B1 PIE in FXPM women suggests a level of specificity that more directly implicates AD-related pathophysiological processes in the observed cognitive deficits.
Implications of LASSI-L deficits for disease prediction
It is important to note that the intrusion errors exhibited by the middle-aged FXPM in this study are not as severe as those typically observed in older adults with aMCI. In the literature, B1 PIE cut-off scores of 39–44% have been shown to differentiate amyloid-β-positive aMCI from normal cognition with sensitivity and specificity exceeding 80%.51,53 In the present sample, 1% of controls and 6% of FXPM women exceeded 44% PIE. An alternative cut-off combining several LASSI-L indices has also shown utility in discriminating early stage MCI with amyloid-β positivity, 79 and may be more applicable to the detection of early stage deficits. This cut-off identified 2% of controls and 10% of FXPM women (17% of those without college degrees, 8% of those with college degrees) as at-risk. While further research is needed, findings suggest that clinically meaningful cognitive impairment could present in as many as 10% of FXPM women aged 30–55. Moreover, the poor B1 PIE and frPSI performance observed in middle-aged FXPM women raises concerns about progression over time. In longitudinal studies of older adults, B1 PIE and frPSI deficits at baseline are associated with steeper functional decline and faster disease progression.53,81,82
Longitudinal investigations are needed to clarify how cognitive deficits in FXPM women progress with age, and intersect with FXTAS. We speculate that the observed LASSI-L deficits reflect an early neurodegenerative process tied to FMR1 dysfunction, consistent with prior cross-sectional and longitudinal evidence of age-related cognitive decline in FXPM women beginning as early as midlife.9–1114,16,20 However, without longitudinal data it remains unclear whether these cognitive differences reflect an early manifestation of the FXTAS disease process, or a broader FXPM cognitive effect independent from FXTAS. In this middle-aged sample, the LASSI-L performance gap between FXPM women without college degrees and controls was of a medium effect size. We would expect this gap to widen with age as neurodegenerative processes unfold, though further research is needed to confirm this hypothesis. While moderate effect sizes suggest that the LASSI-L alone may lack sufficient robustness for diagnostic use in midlife, our findings highlight potential utility for prescreening in clinical trials to enrich for vulnerable individuals, or in combination with other markers to more efficiently identify high-risk individuals. Importantly, diagnostic applications should be revisited in older samples, as differentiation is often less robust during preclinical stages when pathology is less advanced. 83
Genetic risk buffered by education, particularly at mid-range CGG repeats
Although FXPM women, as a group, performed worse than controls on the LASSI-L, college education emerged as a key modifier of cognitive performance in this group. FXPM women without college degrees showed deficits relative to FXPM women with college degrees and controls, whereas FXPM women with college degrees performed similarly to controls. Therefore, educational experience shaped the cognitive expression of FXPM genetic vulnerability during midlife, to the extent that FXPM women showed normalized cognitive performance in association with college education. Findings suggest potential educational neuroprotection, consistent with prior reports suggesting moderation of FMR1 CGG-related health risks via higher education.44,84 While we interpret the observed education effects through the explanatory framework of cognitive reserve, we acknowledge that other explanations could account for educational advantage. For example, women with higher preexisting intellectual abilities could have self-selected into higher education, whereby educational advantage would reflect baseline ability rather than a cognitive reserve mechanism. However, sensitivity analyses controlling for IQ did not yield support for this hypothesis. Socioeconomic advantages tied to higher education (e.g., better medical care, cognitively-stimulating occupations) could also contribute to neuroprotection, rather than the educational experience of college itself. In this case, although the outcome is the same, the focus of intervention would shift from higher education to the lifestyle or behavioral factors that accompany it. However, some evidence suggests that controlling for IQ, socioeconomic status, and other financial, psychosocial, health, and family factors, does not alter the CGG-by-education protective effects on other outcomes (e.g., early mortality). 84 Of note, in the present sample FXPM women with and without college degrees did not differ in CGG repeat length, suggesting that educational advantage could not be accounted for by differing repeat length in college-educated women (see Supplemental Table 1). Cognitive reserve hypotheses may be tested more directly in future studies through animal models allowing for experimental environmental manipulation, or neuroimaging methods capturing neural markers of reserve.
The effect of education varied nonlinearly across CGG repeat length, with gene-environment interactions suggesting differential susceptibility to the environmental context.85,86 Specifically, educational attainment was a strong moderator of cognitive performance at mid-range CGGs, but not higher or lower CGGs, consistent with prior evidence suggesting heightened sensitivity to other aspects of the environment at mid-range CGGs.38–41 This resulted in disproportionate vulnerability in FXPM women with mid-range CGGs when they lacked the benefits of college education. A personalized medicine approach might leverage this finding to target risk modification strategies towards at-risk subgroups. For example, FXPM carriers with mid-range CGGs might be prioritized for programs and policies that increase access to high-quality education (e.g., Title 1, Community College Access initiatives), given that education may be particularly crucial for the long-term cognitive outcomes of those with mid-range CGG repeats.
High environmental sensitivity may increase risk in unsupportive contexts, but differential susceptibility models suggest that this trait may also allow greater benefit to be derived from supportive contexts (i.e., “vantage sensitivity”), supporting outcomes that surpass those of less sensitive individuals. 87 Figure 3 suggests this phenomenon, with college-educated women with mid-range CGGs outperforming those with lower and higher CGG lengths. The pattern in Figure 3 is strikingly similar to the differential susceptibility pattern reported by Seltzer et al. 39 in relation to psychiatric symptoms, raising the question whether this could be a generalized FXPM effect that spans symptom domains. Clinically, this effect might be leveraged to target environmental risk reduction and enhancement strategies to those who are most genetically primed to respond positively to these strategies.
Despite growing evidence of heightened environmental sensitivity at mid-range CGGs, the mechanism underlying this phenomenon remains unclear. One possibility is that clinical risk factors that affect women with mid-range CGGs at higher rates, such as fragile X-associated primary ovarian insufficiency (FXPOI),32,88 contribute to increased vulnerability. Given that reduced lifetime estrogen exposure has been linked to increased risk for cognitive impairment, e.g.,89,90 future research should address whether hormonal dysregulation predisposes women with mid-range CGGs to cognitive difficulties, which has implications for the use of estrogen-based hormone therapies. 91 However, hormonal dysregulation alone cannot explain for the vantage sensitivity patterns observed here and in prior reports38–41 (e.g., above-average outcomes achieved by mid-range women in supportive/enriching environments), which warrant future investigation.
Strengths, limitations, and directions
Study strengths include application of the LASSI-L that proved effective in discriminating midlife cognitive dysfunction in FXPM women. Our relatively large sample is a strength that allowed for identification of curvilinear CGG effects that might otherwise have been obscured by environmental influences. Despite the large sample, we lacked sufficient representation above ∼120 CGG repeats, which occur less frequently in the FXPM population and are often poorly represented in research. Efforts to study very high CGG repeats may be informative considering they have been linked with a second elevation of risk. 9 Our sample had limited racial diversity, limiting generalizability; ascertainment of representative samples must be an intentional goal in future research. Additionally, college degree holders were overrepresented in our sample relative to the US population (75% versus 39%). 92 However, analyses adjusted for education; future research should replicate these results with more representative rates of college education.
Clarifying the timing and intensity of education needed to achieve a protective response is key to optimizing cognitive enrichment interventions. We focused on college degree attainment as a meaningful educational gradient linked to population health and longevity,93,94 but future work should identify the “active ingredients” (e.g., years of study, credential earned, full-time versus part-time) that drive the college effect to better inform public policy and clinical implications. Unpacking the importance of educational timing is also critical, as the brain may show heightened plasticity during certain “sensitive periods” that occur prior to adulthood, and non-normative timing of college education may yield fewer health advantages.95,96 Finally, education is not the only path to increasing cognitive reserve and other activities—particularly physical exercise—may effectively promote neuroplasticity. 97 Identifying alternative pathways for FXPM women to build cognitive resilience will inform a broader array of therapeutic options.
Conclusions
The LASSI-L proved sensitive to subtle midlife cognitive deficits in FXPM women, with patterns suggesting potentially shared mechanisms of neurodegeneration across the FXPM and AD. Importantly, higher education emerged as a modifiable environmental factor that conferred neuroprotection, especially in women with mid-range CGG lengths. These findings not only underscore the importance of early cognitive monitoring and intervention in FXPM women but also point to the promise of personalized medicine to mitigate risk and promote successful cognitive aging in this population.
Supplemental Material
sj-docx-1-alz-10.1177_13872877251414950 - Supplemental material for Cognitive dysfunction in women with the FMR1 premutation during midlife: The LASSI-L reveals curvilinear CGG-dependent risk buffered by college education
Supplemental material, sj-docx-1-alz-10.1177_13872877251414950 for Cognitive dysfunction in women with the FMR1 premutation during midlife: The LASSI-L reveals curvilinear CGG-dependent risk buffered by college education by Jessica Klusek, Jillian Gierman, Amanda J. Fairchild, Andreana M. Benitez, Elizabeth Berry-Kravis and Marsha R. Mailick in Journal of Alzheimer's Disease
Footnotes
Acknowledgements
We thank the women who gave their time to support this research. During the preparation of this work the authors used ChatGPT in order to improve tone and styling of the authors’ written work. After using this tool/service, the author(s) reviewed and edited the content as needed and take full responsibility for the content of the publication.
Ethical considerations
Procedures were approved by the Institutional Review Board of the University of South Carolina (Pro00067288, approval date 6/13/2017).
Consent to participate
Participants provided written informed consent.
Consent for publication
Not applicable.
Author contribution(s)
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by National Institutes of Health [grant numbers R21DC017804, 3R21DC017804-02S1, R01AG073374, 3R01AG073374-02S1, 1R01AG073374-04S1, P50HD118708]; and the Research Participant Registry Core of the Carolina Institute for Developmental Disabilities [grant number U54HD079124].
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Dr. Berry-Kravis has received funding from Acadia, Alcobra, AMO, Asuragen, Avexis, Biogen, BioMarin, Cydan, Engrail, Erydel, Fulcrum, GeneTx, GW, Healx, Ionis, Jaguar, Kisbee, Lumos, Marinus, Mazhi, Moment Biosciences, Neuren, Neurogene, Neurotrope, Novartis, Ovid, PTC Therapeutics, Orphazyme/Kempharm/Zevra, Retrophin, Roche, Seaside Therapeutics, Taysha, Tetra, Ultragenyx, Yamo, Zynerba, and Vtesse/Sucampo/Mallinckrodt Pharmaceuticals, to consult on trial design or run clinical or lab validation trials in genetic neurodevelopmental or neurodegenerative disorders, all of which is directed to Rush University Medical Center in support of rare disease programs; Dr. Berry-Kravis receives no personal funds and Rush University Medical Center has no relevant financial interest in any of the commercial entities listed. The authors have no other conflicts of interest to disclose.
Data availability statement
The data presented in this study are available on request from the corresponding author due to privacy restrictions.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
