Abstract
Background
Antidepressant and anxiolytic medication use in people with dementia (PwD) may contribute to potentially inappropriate prescribing and be associated with mortality.
Objective
To investigate trends in prescribing of these medications and their association with mortality risk among PwD.
Methods
A nested case-control study was conducted in Northern Ireland (NI) using linkage of five administrative population-based data sources within a cohort of dementia patients (identified if a medication indicated for dementia was prescribed). Dementia patients who died were matched to one control who lived at least as long as their matched case after dementia diagnosis (matched on age, sex and year of dementia). Exposure to antidepressant and anxiolytic medications was assessed from two years prior to study entry. Odds ratios (ORs) and 95% confidence intervals (CIs) were calculated using conditional logistic regression after adjusting for demographic factors and comorbidities.
Results
The study included 14,420 dementia cases. Antidepressants were prescribed to 59.2% of cases and 54.7% of controls while 44.8% of cases and 36.0% of controls were prescribed anxiolytics. There was evidence of a weak increased risk of mortality in PwD prescribed antidepressants (fully adjusted OR = 1.08; 95% CI 1.02–1.14) and a strong increased risk in those prescribed anxiolytics (fully adjusted OR =1.26; 95% CI 1.19–1.33) compared to nonusers.
Conclusions
In this large NI population-based cohort of PwD, elevated levels of antidepressant and anxiolytic prescribing were observed. The use of anxiolytic medications was strongly associated with mortality in PwD.
Keywords
Introduction
Dementia is a progressive neurodegenerative condition characterized by a gradual decline in cognitive functioning. 1 It is associated with age and as the proportion of older people aged 65 years and over increases, the prevalence of dementia is also projected to increase. 2 It was estimated that there were 885,000 people with dementia (PwD) in the United Kingdom (UK) in 2019, equating to a prevalence of 7.1%, which is projected to rise to 1.6 million by 2040.3,4
The cognitive symptoms associated with dementia are generally well understood; however, the non-cognitive symptoms present additional challenges for management. 1 Up to 90% of PwD can experience non-cognitive symptoms such as apathy, depression, irritability, agitation and anxiety, 1 with depression and anxiety shown to be among the most clinically significant. 5 The management of depression and anxiety is regarded as more challenging in PwD than the general older population because of greater multimorbidity, 6 more complex medication regimens, polypharmacy and potentially inappropriate prescribing (PIP) 7 with patients more susceptible to medication side effects. 8 Pharmacological interventions may be prescribed to treat non-cognitive symptoms of dementia and include the use of antidepressant and anxiolytic medications. The effectiveness of these agents for treating depression and anxiety in PwD is not well established with some research suggesting they might worsen symptoms, cause adverse effects and increase mortality, while other population-based studies have demonstrated reduced mortality rates during sustained antidepressant use.9–13 Systematic reviews have concluded that although depression is common in PwD and many patients are prescribed antidepressants, there is limited published evidence to support the efficacy of antidepressants for the treatment of depression in this patient group.14–16 Similarly, the use of anxiolytics has been shown to cause adverse effects and increase mortality. For example, Norgaard et al. reported that concomitant use of antipsychotics with benzodiazepines was associated with an increased risk of death when compared with antipsychotic treatment alone. 17
As the prevalence of dementia increases, the need for a greater understanding of the factors that influence mortality in PwD (such as exposure to antidepressant and anxiolytic medications) becomes more critical. 18 However, it is first important to fully understand the trends and patterns of prescribing of these medications. Therefore, the aim of this study was to describe prescribing trends of antidepressant and anxiolytic medications and to evaluate the risk of mortality associated with their use among a retrospective cohort of PwD in Northern Ireland (NI), from 1 January 2010 to 31 December 2020.
Methods
Setting
NI is part of the UK and has a population of approximately 1.9 million. 19 Healthcare in NI is provided under Health and Social Care Northern Ireland (HSCNI) which integrates both health and social care services under a single framework. The HSCNI operates under the Department of Health and ensures that services are free at the point of delivery. This is similar to the National Health Service (NHS) which provides healthcare to other regions within the UK. 20
Data access
The Health and Social Care (HSC) Honest Broker Service (HBS) is the Trusted Research Environment for accessing healthcare-related service user data for analysis in NI and is hosted within the HSC Regional Business Services Organisation (BSO). 20 The HBS provides access to de-identified or aggregated data for approved health and social care-related research.
Data sources
Data were obtained using linkage of five administrative population-based data sources in NI. The National Health Application and Infrastructure Services (NHAIS) contains a record of all patients registered with a GP in NI along with basic demographic information, e.g., Health and Care Number (HCN; this 10-digit number is used from birth, throughout life, and uniquely identifies a patient within HSCNI), sex, age, postcode. 21 In addition, the NHAIS receives a quarterly indicator to identify when a patient has been admitted to a care home. The Enhanced Prescribing Database (EPD) contains detailed information about all prescriptions dispensed in primary care in NI; however, it does not contain clinical information about patients nor does it include data relating to prescriptions issued privately, in hospital, or items obtained over the counter. Furthermore, the EPD does not contain any clinical or diagnostic data about patients.22,23 The Patient Administration System (PAS) holds demographic information about patients such as their sex, date of birth, age, postcode, marital status and GP number. 21 All deaths in NI are registered as a legal requirement with the General Register Office (GRO), which is a branch located within Northern Ireland Statistics and Research Agency (NISRA). The Northern Ireland Neighbourhood Information System (NINIS) is a service provided by NISRA containing datasets for statistical and administrative geographies and area profiles including the Northern Ireland Multiple Deprivation Measure 2017 (NIMDM 2017). NINIS also provides area-level geographical information including urban or rural classification.
Study design and population
A nested case-control study was conducted within a retrospective cohort of PwD in NI. Approval was granted by the HBS Governance Board (Ref: E078). The study is reported according to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) checklist (Supplemental Table 1). 24 Study participants were identified by a computerized search of the EPD, conducted by BSO data custodians. The study population comprised all individuals in the EPD who were dispensed a drug for the management of dementia (donepezil, galantamine, rivastigmine, memantine) during the study period 1 January 2010–31 December 2020. The dispensing of these medications was used as a proxy for diagnosis of dementia in the absence of clinical information about individuals. A “washout period” of one year was applied between 1 January 2011 and 31 December 2011; any individual dispensed a drug for dementia during this time was excluded from the base cohort ensuring only those who were dispensed a drug for dementia for the first time from 1 January 2012 onwards were included in the base cohort. The date of first dispensing of a drug for dementia after this time was therefore used as a proxy for the date of diagnosis. Cases were identified as individuals within the base cohort who died any time between the start of the follow-up period (1 January 2012) and the study end date (31 December 2020). The event date for cases was defined as the date of death. For all cases, the date (month and year) of death was extracted from GRO death registrations to define when death occurred. Controls were identified from the base cohort and matched on age category, sex and date (year) of dementia diagnosis to one case living at least as long after diagnosis as their matched case.
Exposures
Data on drug use were extracted using British National Formulary (BNF; a standard drug reference text used in the UK) codes. 25 The primary exposure of interest was the use of any antidepressant or anxiolytic medication(s) as described in the BNF 68. The exposure period in cases was from two years prior to dementia diagnosis date up to six months before death. The exposure period in controls was of the same duration as their matched cases starting from two years prior to dementia diagnosis. Antidepressant or anxiolytic prescriptions were assessed from two years prior to dementia diagnosis to ensure appropriate exposure and the six-month period prior to the event date was excluded as this may have reflected end-of-life treatment or increased exposure to healthcare professionals. Exposed individuals were defined as “any” users.
Confounders
The numbers and types of medications prescribed for comorbidities were assessed and any association with an increase in mortality was investigated. Prescribing of the following classes of medications, as described in the BNF 68, commonly used for the management of the comorbidities were assessed: anti-arrhythmic drugs (BNF Section 2.3); anti-hypertensive drugs (BNF Section 2.5); lipid-regulating drugs (BNF Section 2.12); hypnotics (BNF Section 4.1.1); anxiolytics (BNF Section 4.1.2); antipsychotic drugs (BNF Section 4.2.1); antidepressant drugs (BNF Sections 4.3.1, 4.3.2, and 4.3.3); antidiabetic drugs (BNF Section 6.1.2). As antidepressants and anxiolytics are frequently used for the management of common comorbidities they were both recorded as confounders as well as exposures. Other potential confounders investigated for their association with mortality among study participants included level of deprivation, urban-rural classification, marital status and place of residence.
Outcomes
The primary outcome was to evaluate the risk of all-cause mortality associated with antidepressant and anxiolytic medications.
Analysis
Descriptive statistics (such as frequencies and proportions) and cross tabulations were used to describe prescribing trends of antidepressants and anxiolytic medications. Adjusted logistic regression analyses were used to calculate odds ratios (ORs) and 95% confidence intervals (CI) to assess the association between the prescribing of antidepressant and anxiolytic medications and mortality. Sensitivity analyses were conducted, restricting the exposure period to between six to twelve months prior to death or the end of the study period. Analyses were also conducted stratified by the number of prescriptions issued for each exposure medication (i.e., 1–11; 12–23; 24–47 and 48–1000 prescriptions), comparing with non-users during the exposure period. Analyses were performed using STATA/SE 18 (StataCorp, College Station, TX).
Results
Characteristics of the study population
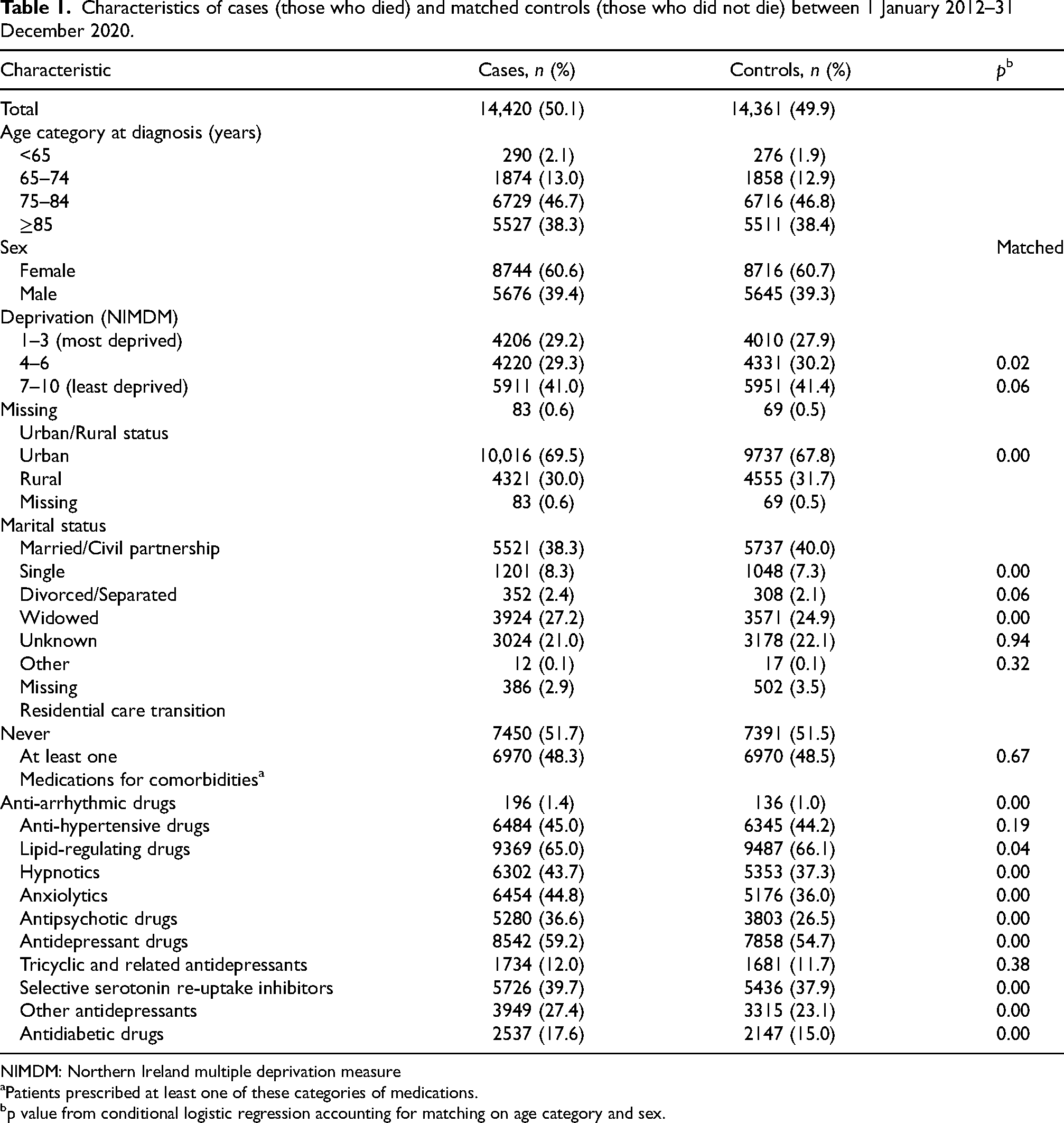
For the study period, 31,525 persons identified in the EPD were eligible for inclusion in the study. The final case-control dataset for analysis contained 16,302 deaths of PwD. The exclusions applied to the final cohort included 1,721 individuals who had received a diagnosis of dementia within two years of the end date of the study. Additionally, 126 individuals were excluded because they had died within two years of their dementia diagnosis and another 35 were excluded because they either had a missing date of dementia diagnosis or date of death. This base cohort was converted to case-control data with 14,420 cases (i.e., those who died) and 14,361 controls (i.e., those who did not die) between 1 January 2012 and 31 December 2020 (Table 1). For 59 cases, suitable matched controls could not be identified as shown in Table 1.
Characteristics of cases (those who died) and matched controls (those who did not die) between 1 January 2012–31 December 2020.
NIMDM: Northern Ireland multiple deprivation measure
Patients prescribed at least one of these categories of medications.
p value from conditional logistic regression accounting for matching on age category and sex.
Of these cases, a significant proportion was older adults, with nearly half (n = 6,729, 46.7%) aged between 75 and 84 years and over a third (n = 5,527, 38.3%) aged 85 years or older at the time of dementia diagnosis. Only a very small proportion (n = 290, 2.1%) was aged less than 65 years. A greater proportion of women (n = 8,744, 60.6%) was diagnosed with dementia during the study period than men (n = 5,676, 39.4%).
Prescribing trends of antidepressant and anxiolytic medications
Anxiolytic medications were prescribed to nearly one-half of cases (n = 6,484, 44.8%) and over one-third of controls (n = 5,176, 36.0%). The most commonly prescribed anxiolytic was diazepam which accounted for 81.2% of the overall anxiolytic prescriptions dispensed, followed by lorazepam (12.1%) and buspirone (4.3%). Antidepressant medications were also commonly prescribed to over half of cases (n = 8,542, 59.2%) and controls (n = 7,858, 54.7%). The most commonly prescribed antidepressants were the selective serotonin reuptake inhibitors (SSRIs), which were prescribed to approximately two-fifths of cases (n = 5,726, 39.7%) and controls (n = 5,436, 37.9%). The most frequently prescribed SSRIs to the study cohort were sertraline, citalopram and fluoxetine, which accounted for 37.2%, 35.1% and 17.5% respectively of SSRIs dispensed. The dispensing of other antidepressant drugs, which included agomelatine, duloxetine, flupentixol dihydrochloride, mirtazapine, reboxetine, tryptophan, venlafaxine and vortioxetine accounted for just over a quarter of cases (n = 3,949, 27.4%) and over one-fifth of controls (n = 3,315, 23.1%). The most frequently prescribed were mirtazapine, venlafaxine and duloxetine which accounted for 63.8%, 23.2% and 11.0% respectively of other antidepressants dispensed. The tricyclic antidepressants (TCAs) were the least commonly prescribed class of antidepressant drugs among both cases (n = 1,734, 12.0%) and controls (n = 1,681, 11.7%). The most frequently prescribed medications in this class were amitriptyline, dosulepin, and trazadone, which accounted for 68.8%, 15.1%, and 8.8% respectively of all TCAs dispensed.
Association of antidepressant and anxiolytic use with mortality in PwD
Table 2 displays the conditional logistic regression models applied to the matched case-control data exploring the association between exposure to antidepressant and anxiolytic medications and the risk of mortality in the study cohort.
Association between exposure to antidepressant and anxiolytic medications in the two years prior to study entry and up to six months before index date, and mortality in people with dementia.

CI: confidence interval; OR: odds ratio.
*Other antidepressants included medications such as duloxetine, mirtazapine, tryptophan, venlafaxine. **Anxiolytics included medications such as diazepam, lorazepam, buspirone.
Model contains age and sex.
Model 1 plus adjustment for marital status, urban/rural status, deprivation, residential care transition, lipid regulating medications, anti-arrhythmic medications, hypnotics, antipsychotics, anti-hypertensives, diabetic medications, anxiolytics, tricyclic and related antidepressants, selective serotonin re-uptake inhibitors and other antidepressants.
Model 1 plus adjustment for anxiolytics.
Model 1 plus adjustment for selective serotonin re-uptake inhibitors, other antidepressants and anxiolytics.
Model 1 plus adjustment for tricyclic and related antidepressants, other antidepressants and anxiolytics.
Model 1 plus adjustment for tricyclic and related antidepressants, selective serotonin re-uptake inhibitors and anxiolytics.
Model 1 plus adjustment for all antidepressants.
There was evidence of a 21% risk of mortality in PwD who were exposed to all antidepressants, which included the use of TCAs, SSRIs, and other antidepressants when compared with no use (OR =1.21, 95% CI 1.16–1.27). There was a sizeable reduction in mortality risk but with a slight association still apparent after adjustment for all confounders (i.e., marital status; urban/rural status; deprivation; residential care transition and medications for comorbidities) (OR =1.08, 95% CI 1.02–1.14). There was no association between TCA use and mortality compared with no use when fully adjusted (OR =0.95, 95% CI 0.88–1.02). The association between use of SSRIs and mortality was similar to that seen with the TCAs (fully adjusted OR =1.01, 95% CI 0.96–1.06). The use of other antidepressants displayed evidence of an increased risk of mortality compared with no use (OR =1.26; 95% CI 1.20–1.33). This decreased after adjustment for confounders, but an 11% increased mortality risk was still observed (fully adjusted OR =1.11, 95% CI 1.05–1.18) with statistical significance demonstrated across all models.
The use of anxiolytics displayed the strongest association with mortality before adjusting for confounders (OR =1.46, 95% CI 1.39–1.53) when compared with no use. After adjustment for all confounders, the increased risk of mortality was attenuated but still statistically significant (fully adjusted OR =1.26, 95% CI 1.19–1.33).
Sensitivity analysis
The findings were generally consistent across each of the exposure medications. The adjusted OR estimates shown in Table 3 demonstrate a similar pattern of association with mortality risk and all antidepressants, with an OR estimate of 1.07 (95% CI 0.99–1.15) for the individuals dispensed 1–11 prescriptions and a slight increase in effect size for those individuals dispensed 12–23 prescriptions (OR =1.13, 95% CI 1.04–1.23) compared to nonusers. A decreased effect size was shown with the use of TCAs for those dispensed 1–11 prescriptions and 12–23 prescriptions in the adjusted analyses (OR =0.90, 95% CI 0.82–1.00) and (OR =1.00, 95% CI 0.85–1.18), respectively. A slight increase in association with mortality risk was demonstrated, after adjusting, with the use of SSRIs when 12–23 prescriptions were dispensed (OR =1.07, 95% CI 0.98–1.17) compared to the risk associated with 1–11 prescriptions (OR =1.01, 95% CI 0.96–1.06). The other antidepressants demonstrated a similar association with mortality across all models of stratified prescription numbers compared to any use of other antidepressants (adjusted OR =1.11, 95% CI 1.05–1.18). Finally, the mortality risk associated with exposure to anxiolytics showed some consistency across the numbers of prescriptions with a higher risk demonstrated for those receiving 12–23 prescriptions (adjusted OR =1.32, 95% CI 1.18–1.48). In a sensitivity analysis (Supplemental Table 2) when the definition of exposure period was altered to use of exposure medication between 12 months and six months before death, this demonstrated similar associations to the main analysis (which investigated use in the period 2 years before diagnosis to 6 months before death).
Adjusted sensitivity analyses for the association between exposure to antidepressant and anxiolytic medications and mortality in people with dementia.
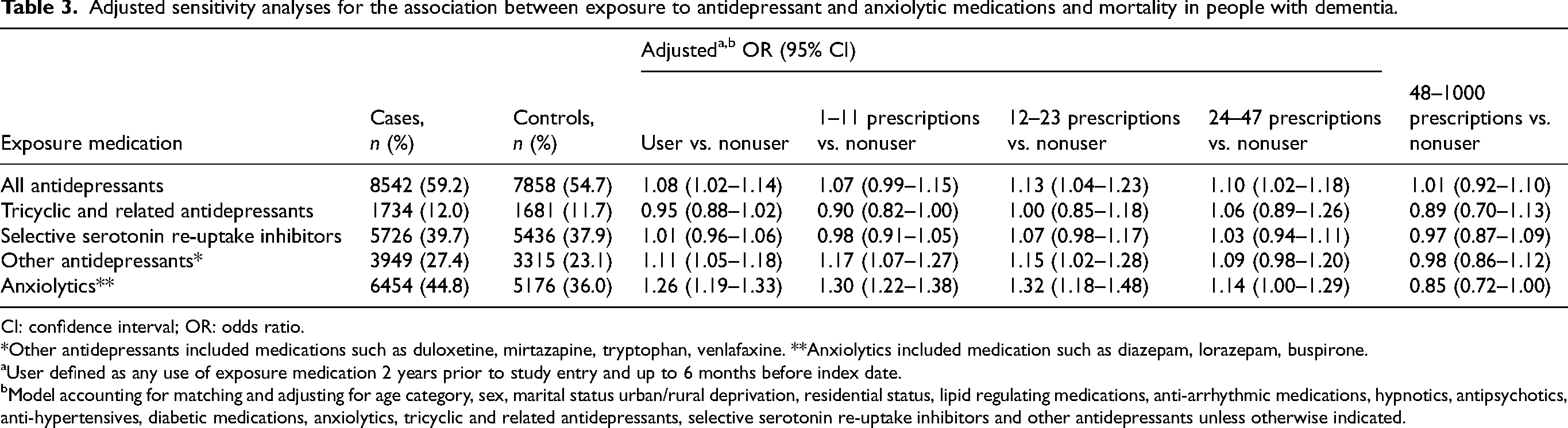
CI: confidence interval; OR: odds ratio.
*Other antidepressants included medications such as duloxetine, mirtazapine, tryptophan, venlafaxine. **Anxiolytics included medication such as diazepam, lorazepam, buspirone.
User defined as any use of exposure medication 2 years prior to study entry and up to 6 months before index date.
Model accounting for matching and adjusting for age category, sex, marital status urban/rural deprivation, residential status, lipid regulating medications, anti-arrhythmic medications, hypnotics, antipsychotics, anti-hypertensives, diabetic medications, anxiolytics, tricyclic and related antidepressants, selective serotonin re-uptake inhibitors and other antidepressants unless otherwise indicated.
Discussion
Findings
This study investigated the prescribing trends of antidepressant and anxiolytic medications and assessed their association with mortality during the study period 1 January 2010–31 December 2020. Based on the data from a large dispensing database of 31,525 dementia patients in NI, we found that prescribing of antidepressant and anxiolytic medications was prevalent among this community-dwelling patient population over the study period. Some association between exposure to antidepressant medications and mortality was observed among PwD. Even more notably, the risk of mortality in PwD increased upon exposure to anxiolytic medications.
Prescribing trends
Overall, there was a high proportion of cases within the matched cohort who were prescribed an antidepressant with almost 60% having been dispensed at least one prescription over the study period. This is unsurprising given that PwD are twice as likely to be diagnosed with depression; however, the prevalence of antidepressant medication use was higher than previously reported.26,27 Previous studies have reported antidepressant medication use between 25% to 50%, although these studies focused on PwD residing in the community and in nursing homes and therefore their findings are not entirely generalizable.28–30 Evidence suggests that certain antidepressants may be prescribed in the management of other non-cognitive symptoms such as agitation, psychosis and anxiety and that PwD may be prescribed antidepressant medication without a formal diagnosis of depression, hence adding to the observed increased level of prescribing.31,32 The high level of prescribing in the current study may also indicate that prescribers in NI are not adhering to guidance, such as that published by the National Institute for Health and Care Excellence (NICE). The 2018 NICE dementia guideline recommended that antidepressant medication may be considered for moderate to severe depression in PwD, especially where non-drug therapies are insufficient or not possible. 1 However, as the EPD does not provide clinical information about patients such as diagnoses they have received, the severity of depression in these patients or other factors impacting clinical decision making was not known; hence, further work would be necessary to gain an in-depth understanding as to why prescribers may be less inclined to adhere to clinical guidance.
This study revealed that anxiolytic medication was widely prescribed during the study period with almost 45% of cases being dispensed at least one anxiolytic. This is higher than previous studies which demonstrated up to 38.1% of PwD were prescribed anxiolytic medications.28,33 Again, these studies are difficult to compare as they involved participants from multiple settings including long-term care institutions. Anxiolytics are generally prescribed for PwD because they induce sleep when given at night and sedate when given during the day, while benzodiazepines are often prescribed when required. 33 This may have added to the increase in the overall level of prescribing in this study as the definition of use was based on the use of any prescription of the medication during the study period. Additionally, anxiolytics are often used for the management of other non-cognitive symptoms such as agitation and aggression as they are regarded as being a safer alternative to antipsychotics and can effectively reduce the severity of these symptoms. 34 Benzodiazepines are a common cause of PIP in PwD and the high level of prescribing of these medications in the current study may suggest that PIP was prevalent among the study cohort which has been reported previously. 23
Association with mortality
The association demonstrated between the prescribing of antidepressants and the risk of mortality in this study adds to the growing evidence showing increased mortality risk with antidepressant use in PwD.10,30,35,36 This association could be partly explained by previous research which reported that antidepressants are associated with adverse outcomes which can increase the risk of mortality including falls, hyponatremia, cardiovascular events and cerebrovascular conditions.35,37 Additionally, the physiological changes that occur with ageing can lead to altered pharmacokinetic and pharmacodynamic responses to medications such as antidepressants, decreasing the individual's tolerability of medications and leading to greater risk of adverse effects such as falls and drug-drug interactions, thereby contributing to an increased risk of mortality in PwD. 38 An alternative explanation for the association between the prescribing of antidepressants and the risk of mortality shown in this study may be attributable to the severity of dementia itself and the symptom burden. Non-cognitive symptoms of dementia, including depression, have been shown to be associated with accelerated cognitive decline, higher rates of institutionalization and increased mortality in PwD, independent of medications. 39 Furthermore, antidepressants are generally reserved for PwD at more advanced stages of dementia, with more severe or recurring depression and who may be more likely to be burdened by multimorbidity which, in turn, may increase the risk of mortality. 1 However, a number of studies demonstrate the potential benefits of the use of antidepressant medications in this population, for example, some research suggests that certain antidepressants exert anti-inflammatory effects within the central nervous system, reducing neuroinflammation that may contribute to cognitive decline. 40 In this study, it appeared that the association of antidepressants with mortality was largely driven by the prescribing of the medications from the group of other antidepressants, which included mirtazapine, duloxetine and venlafaxine. However, as the explanation for the observed risk of mortality could not directly determined, the weak increase in mortality may be attributable to residual confounding.
The use of anxiolytics was associated with the highest increased risk of mortality after adjusting for all confounders. This is in line with several previous studies which demonstrated a marked increase in mortality in PwD; however, these studies used different methodologies and included different confounders.10,30 Other studies which have examined large groups of patients with other conditions such as chronic insomnia and patients on dialysis, highlighted concerns regarding increased mortality rates particularly in combination with psychotropic medications including anxiolytics and highlighted the need for further evaluation of this group of medications.10,41 However, judicious and short-term use of anxiolytic medications may help in patients experiencing severe insomnia or agitation by restoring their continuity of sleep and by potentially reducing harm to patients/others in certain clinical situations. 42 Almost half of PwD in our study received an anxiolytic with diazepam being the most frequently prescribed. This is concerning because anxiolytics may cause sleep disruption, confusion, dizziness, low heart rate and blood pressure and should therefore be avoided in older people because they are at greater risk of becoming ataxic and confused, leading to falls and injury.10,41,43,44 Patients often respond positively to benzodiazepines, with a reduction in the severity of their behavioral problems; however, these medications fail to improve functioning, care needs or quality of life in PwD. 45
Implications
Patients, carers and clinicians face a dilemma when it comes to deciding how best to manage the symptoms of depression in PwD. 46 While non-pharmacological treatments may be recommended first line for managing depressive symptoms, it is not always possible or accessible for patients and their carers to avail of them and therefore many will consider seeking a pharmacological approach. The pressure “to do something” and prescribe was evident in a recent study which reported that clinicians found managing non-cognitive symptoms such as depression and anxiety as “very difficult to deal with” and were frequently faced with the need to balance the benefit of prescribing antidepressants and anxiolytics with the potential increased risk of adverse events, mortality and efficacy.46,47 The findings from our study emphasize the need for safe, targeted interventions aimed at optimizing the management of depression and anxiety in PwD and future work should be undertaken to understand the experiences and perspectives of key stakeholders (i.e., carers, patients, general practice-based pharmacists and prescribers) involved in the management of depression and anxiety in PwD.
Strengths and limitations
This is one of the largest epidemiological studies to provide a comprehensive overview of antidepressant and anxiolytic prescribing in PwD in NI. The BSO provided robust data on individuals’ settlement band, level of deprivation and residential status. Additionally, mortality records were provided to ascertain time to death from the date of diagnosis of dementia to provide the mortality risks associated with the exposure to antidepressant and anxiolytic medications in the study population. The nested case-control design of this study was a strength as it was efficient for analyzing the large numbers and accounted for immortal time bias ensuring that cases were matched on sex, age and date of dementia diagnosis to one control, who had dementia and lived at least as long after diagnosis as their matched case.
Medication data provided by the EPD dataset has been used with ease in previous studies of the older population in NI.22,23,48 The EPD holds information on all prescriptions dispensed in NI, and the high scan rate of prescriptions has generated a reliable database for researchers. Although we can be confident in the generalizability of the results to the wider dementia population in NI, there are some methodological limitations which may limit the generalizability of the findings to other settings. The EPD does not contain clinical information about patients, notably diagnosis. Therefore, individuals with dementia were identified using the first date that a dementia medication (i.e., donepezil, galantamine, rivastigmine and memantine) was prescribed after a “washout period” as a proxy for the date of diagnosis. It is therefore likely there was an underestimation of the prevalence of dementia and individuals with certain types of dementia were identified (e.g., those with frontotemporal dementia and vascular dementia were likely to be omitted from the study cohort as the drug treatments listed above are not indicated for these). In addition, patients who were previously prescribed dementia medications but were unable to tolerate them due to side-effects or contraindications would not have been included in the cohort. Another limitation to note is that antidepressants and anxiolytics can be used for indications other than depression and anxiety and as the EPD does not contain clinical information about patients, the prescribing of these medications was assumed to be for depression and anxiety only. Medication adherence among the study participants was assumed, i.e., individuals took their dispensed medication as prescribed by the doctor. However, previous research has shown that even if a patient has collected their medication from a community pharmacy, they may not take them as the clinician has prescribed them, or indeed, may not take them at all. 49 The final limitation of this study is that the marital status of participants was from data recorded at hospital discharge and so approximately 35% of the data is missing. Additionally, marital status was recorded at the time of diagnosis of dementia and was not recorded throughout the study period, therefore the findings in relation to the role of marital status should be interpreted with caution. This has been encountered in previous studies and was anticipated during study planning. 50
Despite these limitations, the prescribing of antidepressant and anxiolytic medications is elevated in PwD in NI. This study demonstrated some association between the exposure to antidepressant medications and mortality in PwD and a strong association between the exposure to anxiolytic medications and mortality in PwD. This study has added to the limited body of epidemiological work undertaken on the prescribing trends of antidepressant and anxiolytic medications and the associated risk of mortality in PwD. Further pharmacological studies should be undertaken to validate the findings from the current study in other settings such as the rest of the UK or Europe.
Supplemental Material
sj-docx-1-alz-10.1177_13872877251414888 - Supplemental material for Antidepressant and anxiolytic medications and risk of mortality in people with dementia: A nested case-control study in Northern Ireland
Supplemental material, sj-docx-1-alz-10.1177_13872877251414888 for Antidepressant and anxiolytic medications and risk of mortality in people with dementia: A nested case-control study in Northern Ireland by Catherine J. Sinnamon, Carmel M. Hughes, Chris R. Cardwell and Heather E. Barry in Journal of Alzheimer's Disease
Footnotes
Acknowledgements
The authors would like to acknowledge the help provided by the staff of the Honest Broker Service (HBS) within the Business Services Organisation Northern Ireland (BSO). The HBS is funded by the BSO and the Department of Health (DoH). The authors alone are responsible for the interpretation of the data and any views or opinions presented are solely those of the author and do not necessarily represent those of the BSO.
Ethical considerations
This study was approved by the Honest Broker Governance Board (E078). It was confirmed that ethics approval was not required for this project as it did not involve linking external datasets to HSC Regional Data Warehouse data.
Consent to participate
Not applicable
Consent for publication
Not applicable
Author contribution(s)
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
Researcher access to anonymised Health & Social Care NI data is facilitated via the HSC Honest Broker Service (HSC HBS), the Trusted Research Environment for HSC in NI. This service has been established by the HSC Business Services Organisation (BSO) and enables the provision of de-identified health and social care data to researchers within a secure setting. Individuals wishing to utilize HSC data for research purposes must apply to the HBS Governance Board for formal assessment to ensure the project's feasibility, health and social care relevance, and its implications for the delivery or improvement of public services. Data external to the HBS can also be requested subject to approval and the appropriate legal arrangement with the relevant data custodian. Research proposals can be submitted via the HBS Application Procedure and Data Access Approval must be secured for a research project through the HSC Data Access Committee. Applications are considered and provided in line with the Five Safes’ framework, a set of principles established to ensure the safe and responsible use of data: for further information contact HonestBrokerService@hscni.net.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
