Abstract
Background
Alzheimer's disease (AD) accounts for the majority of dementia-related disorders among aging populations. Given the irreversible nature of AD, early detection of cognitive impairment is critical for improving prognosis and facilitating timely interventions before individuals meet the clinical criteria for AD.
Objective
The objective of this review is to provide a comprehensive summary of recent advances in task-based electroencephalography (EEG), such as using attention and inhibitory control tasks, which has recently emerged as a promising non-invasive biomarker for assessing neurophysiological alterations associated with AD and mild cognitive impairment (MCI).
Methods
This systematic review evaluates the efficacy of task-based EEG biomarkers in distinguishing cognitively impaired individuals from those without impairment. A comprehensive literature search was conducted across PubMed, Semantic Scholar, and SpringerLink databases for studies published between 2017 and 2024.
Results
Findings indicate consistent neurophysiological alterations in MCI and AD, particularly reductions in event-related potential amplitudes and prolonged latencies, with P3 abnormalities observed in about half of the studies assessing selective attention and inhibitory control. Similarly, a comparable number of studies using working memory tasks report disrupted functional connectivity patterns, increased low-frequency oscillations (delta and theta activity), and reduced fast oscillations (alpha and beta activity).
Conclusions
These EEG-based indices demonstrate potential as objective biomarkers for detecting neural alterations associated with cognitive decline in both AD and preclinical dementia stages. Further research is needed to standardize EEG protocols and validate their clinical utility for early diagnosis and disease progression monitoring.
Keywords
Introduction
Dementia is a progressive neurodegenerative disease and a major cause of disability and dependency in aging societies. It is currently recognized as the seventh leading cause of death worldwide. 1 Dementia is marked by a progressive decline in cognitive functions, affecting memory, attention, reasoning, visual-spatial abilities, learning, and judgment. While age is a major risk factor, dementia is not an inevitable part of aging. As the global population grows older, dementia has become an urgent healthcare challenge, further intensified by the lack of a cure. Therefore, there is a pressing need to develop early diagnostic tools that can enable timely interventions before significant brain deterioration occurs. Cognition associated with memory, attention, executive function, language, learning, perceptual-motor function, and social cognition typically declines with advancing age. 2 Dementia represents a significant decline in one or more cognitive domains, with memory being among the first affected in many common types of disease. As such, memory impairment is often considered a potential early marker of mild cognitive impairment (MCI), a condition that carries a heightened risk of progressing to Alzheimer's disease (AD). 3 In addition to memory loss, attention—the ability to filter relevant information, ignore distractions, and guide decision-making in response to environmental changes—is also among the earliest cognitive functions disrupted in the initial stages of AD.4,5 Moreover, even in the very early phase of the disease, patients with AD exhibit impaired executive function, which is a comprehensive term encompassing the cognitive processes, including reasoning, problem-solving, and regulation of behavior.6,7 Within the non-memory domain, attention and executive function impairment precedes observable deficits in language and visuospatial abilities.8,9 Cognitive decline often precedes the clinical diagnosis of dementia by many years, highlighting the need for tools capable of distinguishing the early transitions along the AD continuum, from healthy aging to MCI and from MCI to dementia. In clinical practice, this need has traditionally been addressed through standardized screening instruments. The Mini-Mental State Examination (MMSE) and Montreal Cognitive Assessment (MoCA) are commonly used screening tools, with MoCA being superior for the identification of MCI.10–14 Alternative screening tests include the Addenbrooke's Cognitive Examination Revised (ACE-R) 15 and the Mini-Cog test. 16 Validated neuropsychological assessments such as the Trail Making test, 17 the Clock Drawing test, 18 and the Rey-Osterrieth Complex Figure test 19 are also used to evaluate specific cognitive domains. Nevertheless, these conventional pen-and-paper tests demand extensive training to ensure they are administered uniformly, and their findings are interpreted accurately.
MCI represents a transitional stage between the subtle cognitive changes of normal aging and the more pronounced decline characteristic of dementia, most often AD, which accounts for 60–70% of dementia cases worldwide.1,20 With advances in neurotechnology for age-related cognitive intervention, 21 the demand for sensitive and accessible tools to detect the earliest stage of AD is becoming increasingly urgent. Identifying biomarkers that can reliably predict the progression from preclinical MCI to overt cognitive decline and ultimately to AD is therefore critical. 22 A deeper understanding of the how AD's pathophysiological processes give rise to clinical symptoms will be essential for the development of effective disease-modifying therapies and for improving early detection of both MCI and AD.
Detecting the progression of AD before the onset of brain damage is essential. Biomarkers offer non-invasive methods for diagnosing and evaluating disease progression. While several types of biomarkers, including biochemical, genetic, and neuroimaging biomarkers, are widely used in clinical research, neuroimaging techniques can be costly, invasive, and typically detect disease-related damage only in advanced stages. Additionally, biomarkers derived from laboratory tests, including blood tests, can be unstable due to variability in levels influenced by external factors such as age, health status, and differences in laboratory methods, leading to inconsistent results. Biochemical biomarkers like amyloid-β (Aβ) and tau protein found in the cerebrospinal fluid (CSF) are hallmarks of AD pathophysiology. 23 In contrast, genetic biomarkers like the ε4 allele of Apolipoprotein E (APOE) indicate an additional risk of developing AD. 24 AD is associated with increased Aβ and tau levels in the CSF, which makes both of them potential biomarkers for AD diagnosis. 25 Neuroimaging biomarkers such as positron emission tomography (PET) and magnetic resonance imaging (MRI) reveal specific changes that characterize AD.26,27 Although amyloid PET has good sensitivity, it lacks specificity for diagnosing different dementia subtypes, and involves exposure to radiation. 28 Alternatively, MRI provides anatomical information, including the detection of structural changes associated with neurodegenerative disease such as brain atrophy, but it is not specific for AD. 29 While biochemical and genetic biomarkers reveal levels of specific compounds in the brain or body fluids, indicating biological processes or responses to interventions, neuroimaging biomarkers are non-invasive methods used to determine specific changes that characterize AD. In contrast to biochemical biomarkers, electroencephalography (EEG) offers neural activity with high temporal resolution, allowing for the identification of functional brain alterations and network dynamics in AD and MCI. Therefore, measures of neural oscillatory activity, event-related potentials (ERPs), as well as connectivity and complexity measures as revealed in task-based EEG are both promising candidate biomarkers for AD. Consequently, task-based EEG holds, for example, potential utility as a pre-screening tool prior to MRI implementation.
This systematic review aimed to identify studies on neural oscillatory activity, ERPs, and connectivity and complexity measures reflected in task-based EEG, investigating their potential as biomarkers for identifying MCI and AD. Through a systematic exploration of empirical studies, the review seeks to clarify the effectiveness and reliability of task-based EEG biomarkers in identifying distinct patterns that can aid in the early differentiation and potential prognostication of cognitive decline along the continuum from MCI to AD-related dementia. This review synthesizes recent findings on task-based EEG biomarkers and highlights their emerging role in the early detection and monitoring of MCI and AD, offering timely updates in a field where such insights are urgently needed Our systematic review will be critical for shaping future therapeutic strategies, as early detection could enable timely access to treatments and empower individuals to adopt practices that may slow disease progression. 21 In line with this goal, this work presents recent advances in task-based EEG to guide biomarker development and deepen understanding of MCI and AD neurophysiology.
Methods
Search strategies
We adhered to the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) 2020 guidelines.
30
The PubMed, Semantic Scholar, and SpringerLink online databases were searched to identify articles published in English between 2017 and 2024. This review focused on recent publications to capture current EEG methodologies and developments. Articles reporting task-based EEG biomarkers for cognitive impairment among patients with MCI or AD and in cognitively normal individuals, were included. The search strategy involved the following elements:
▪ Keywords: Four main keywords were established to retrieve articles from the databases. The first two keywords were “EEG” and “cognitive” which had to co-occur with a third keyword related to patient groups, such as “Alzheimer's disease,” “mild cognitive impairment,” or its abbreviation “MCI”. A fourth keyword defined specific cognitive tasks used to assess cognitive functions, including “oddball,” “attention inhibitory,” “working memory,” or “decision-making”. ▪ Used Combinations: Boolean operators were employed, using AND between all four main keywords and OR within the third and fourth keywords. For example: “EEG” AND “cognitive” AND (“Alzheimer's” OR “mild cognitive impairment” OR “MCI”) AND (“oddball” OR “attention inhibitory” OR “working memory” OR “decision-making”). All identified keywords and Boolean operators were adapted to the specific search features of each database. ▪ Filters: Filters were applied for publication date (2017–2024) and language (English). Additionally, filters for human subjects were applied in certain applicable databases.
Eligibility criteria
The inclusion criteria were limited to original research articles, excluding case reports, systematic reviews, and meta-analyses. The search was restricted to articles published in peer-reviewed journals, while other publication types, such as conference proceedings papers, preprints, letters, editorials, and books, were not considered. Studies met the following criteria were included in the review: 1) involved older adult populations, including individuals meeting standardized diagnostic criteria for AD or MCI, as well as those identified as at-risk MCI based on cognitive scores assessed with validated neuropsychological assessments; and 2) analyzed EEG under cognitive tasks. Meanwhile, the following studies were excluded: 1) populations with degenerative disorders in which dementia was not the primary manifestation of the disorder, such as dementia in Huntington's disease, Parkinson's disease with dementia, post-stroke dementia, and secondary dementias; 2) studies limited to healthy participants without reported memory or cognitive impairment; 3) studies on other forms of dementia or presymptomatic stage; 4) studies focusing on resting-state EEG or the effect of training or intervention on cognitive ability; and 5) studies on the development of mathematical foundations of machine learning, as they did not primarily underscore the aspects related to biomarkers for disorder detection.
Article selection
Our search terms initially identified one thousand nine hundred thirty-eight articles (n = 1938) from the PubMed, Semantic Scholar, and SpringerLink databases. Among the identified records, one thousand five hundred forty-three (n = 1543) were removed due to duplication. Furthermore, one hundred and eight studies (n = 108) were excluded for varying reasons, including fifty-four conference proceedings and/or abstract book titles, nine editorial and perspective articles, twenty-seven preprint records, and eighteen in-scope systematic reviews. Next, the titles and abstracts of the two hundred eighty-seven (n = 287) were screened according to the established inclusion and exclusion criteria in the first stage of the screening process. Out of these, fifty-six studies explored various methods for dementia screening without using EEG. Additionally, seventy studies were found to be irrelevant to dementia or focused on different clinical conditions, such as post-stroke dementia, Parkinson's disease, attention deficit hyperactivity disorder, schizophrenia, epilepsy, depression, and autism. Also, twenty-three studies were conducted on the young adult population, and three focused on tracing the developmental trajectories evident across the lifespan. Furthermore, thirty-one studies addressed resting-state EEG, while fourteen articles predominantly focused on developing machine learning models. Moreover, twenty-three records examined the influences of medication, interventions, and/or commercial product outcomes. As a result, a total of two hundred twenty articles (n = 220) were excluded from the analysis due to their failure to meet the predetermined inclusion criteria, and sixty-seven articles (n = 67) were selected for full-text review, independently screened by two reviewers (KT and AW). Out of these full-text articles, twenty-three studies (n = 23) were subsequently excluded due to the following reasons: thirteen studies on individuals with other forms of dementia or presymptomatic stage; one study involving animals; six studies on multimodal measures, which involved a combination of different approaches and could lead to deviations into non-related EEG research; and three studies with a small sample size (less than ten participants in the cognitively impaired group). Notably, to ensure a comprehensive identification of relevant studies, the reference lists of full-text articles were independently screened by KT and AW. This practice, as recommended in systematic review manuals and recent publications,22,31 proved effective yielding 30 additional articles (n = 30) that were incorporated into the review. These studies were selected based on their relevance and contributions to understanding EEG biomarkers in MCI and AD, following manual searches of the references in the identified articles. All included studies met our predefined inclusion and exclusion criteria. Hence a final number of seventy-four studies (n = 74) was included in the review. The PRISMA flow diagram of search results has been depicted in Figure 1.
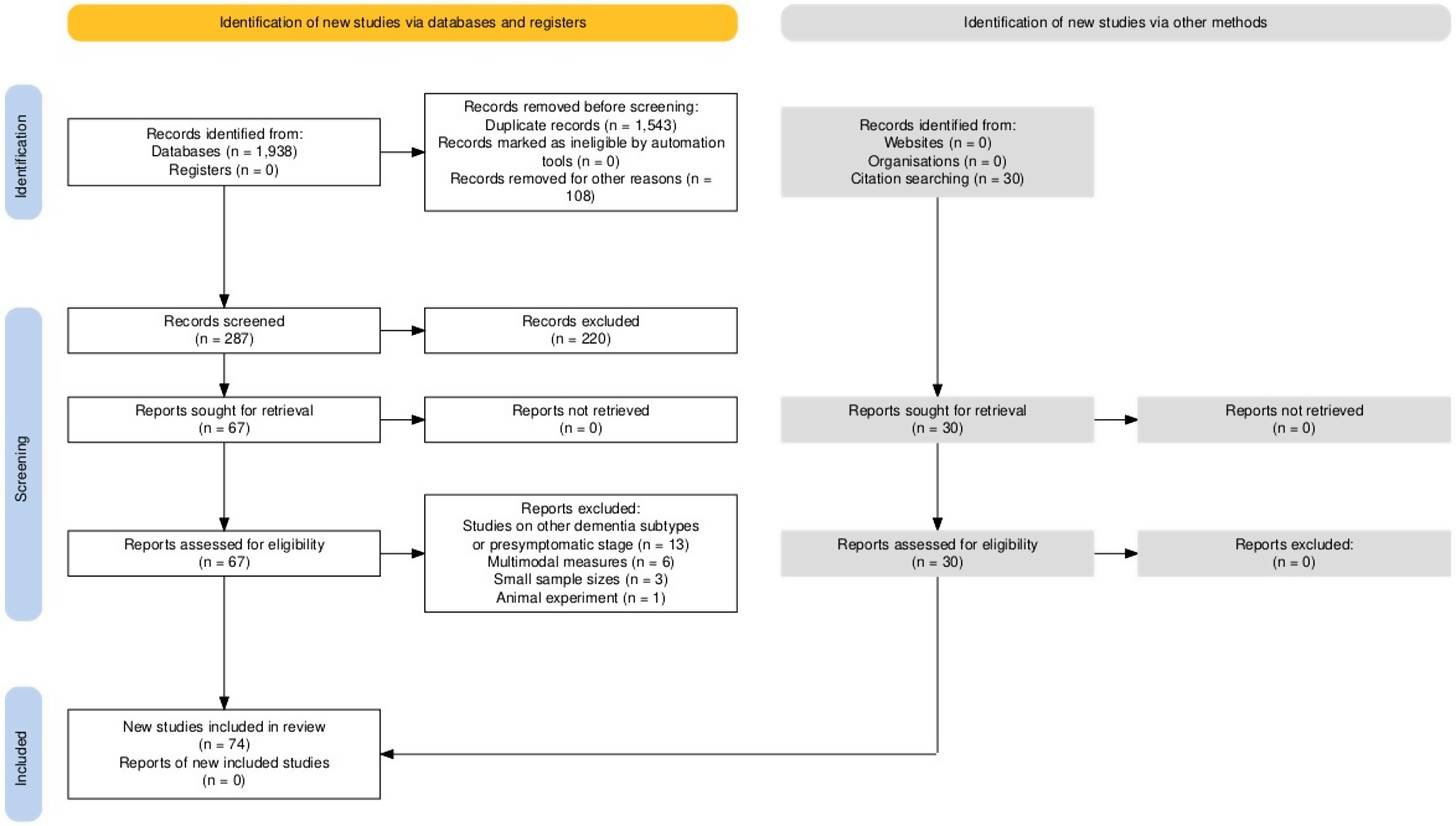
PRISMA flow diagram describing the selection process of the included studies. This output plot was generated via the https://estech.shinyapps.io/prisma_flowdiagram/. 32
Results
Task-based EEG associated with cognitive function
A detailed summary of EEG biomarkers across cognitive tasks and patient populations is provided in Table 1, while overview of the changes in ERPs and oscillatory synchrony in AD and MCI presented in this review is shown in Tables 2 and 3.
Summary of EEG biomarkers across cognitive tasks and patient groups.
‡ within-group, between-conditions; + Conflicting results have been reported, with some studies indicating an increase while others demonstrate a decrease. * This biomarker found in both AD and MCI groups.
AD: Alzheimer's disease; CPT: continuous performance task; DMS: delayed match-to-sample; ERD/ERS: event-related desynchronization/synchronization; ERO: event-related oscillation; FRN: feedback-related negativity; MCI: mild cognitive impairment; LPP: late positive potential; PSD: power spectral density; SCF: spectral crest factor; SE: spectral entropy; SK: spectral kurtosis; SNR: signal-to-noise ratio; SS: spectral skewness.
Overview of the changes in amplitude and latency of ERP components in ad and MCI presented based on the articles included in this review.

↑: increase; ↓: decrease; *: no significant difference; –: data not available/reported; AD: Alzheimer's disease; CDA: contralateral delay activity; ERP: event-related potential; FRN: feedback-related negativity; MCI: mild cognitive impairment; LPC: late positive component; LPP: late positive potential; PNwm: positive-negative working memory.
Overview of the oscillatory synchrony in AD and MCI.
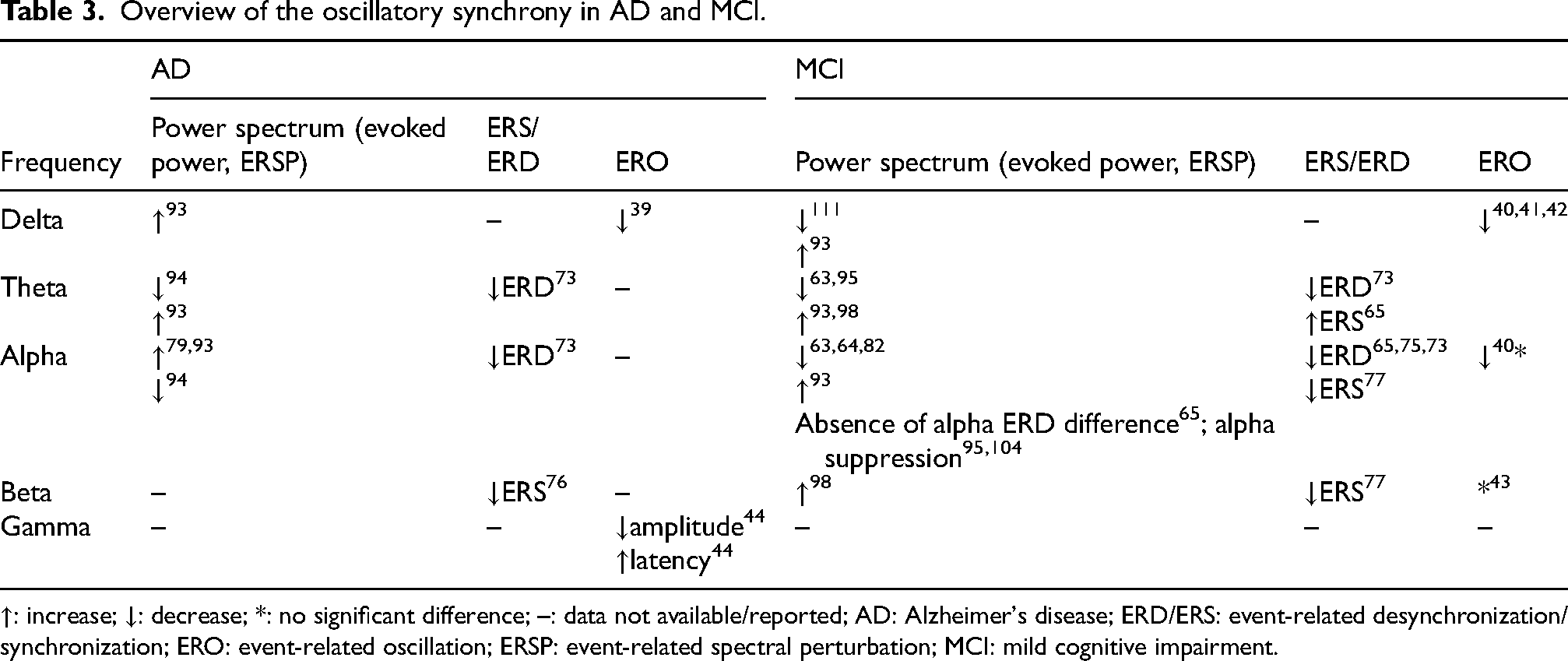
↑: increase; ↓: decrease; *: no significant difference; –: data not available/reported; AD: Alzheimer's disease; ERD/ERS: event-related desynchronization/synchronization; ERO: event-related oscillation; ERSP: event-related spectral perturbation; MCI: mild cognitive impairment.
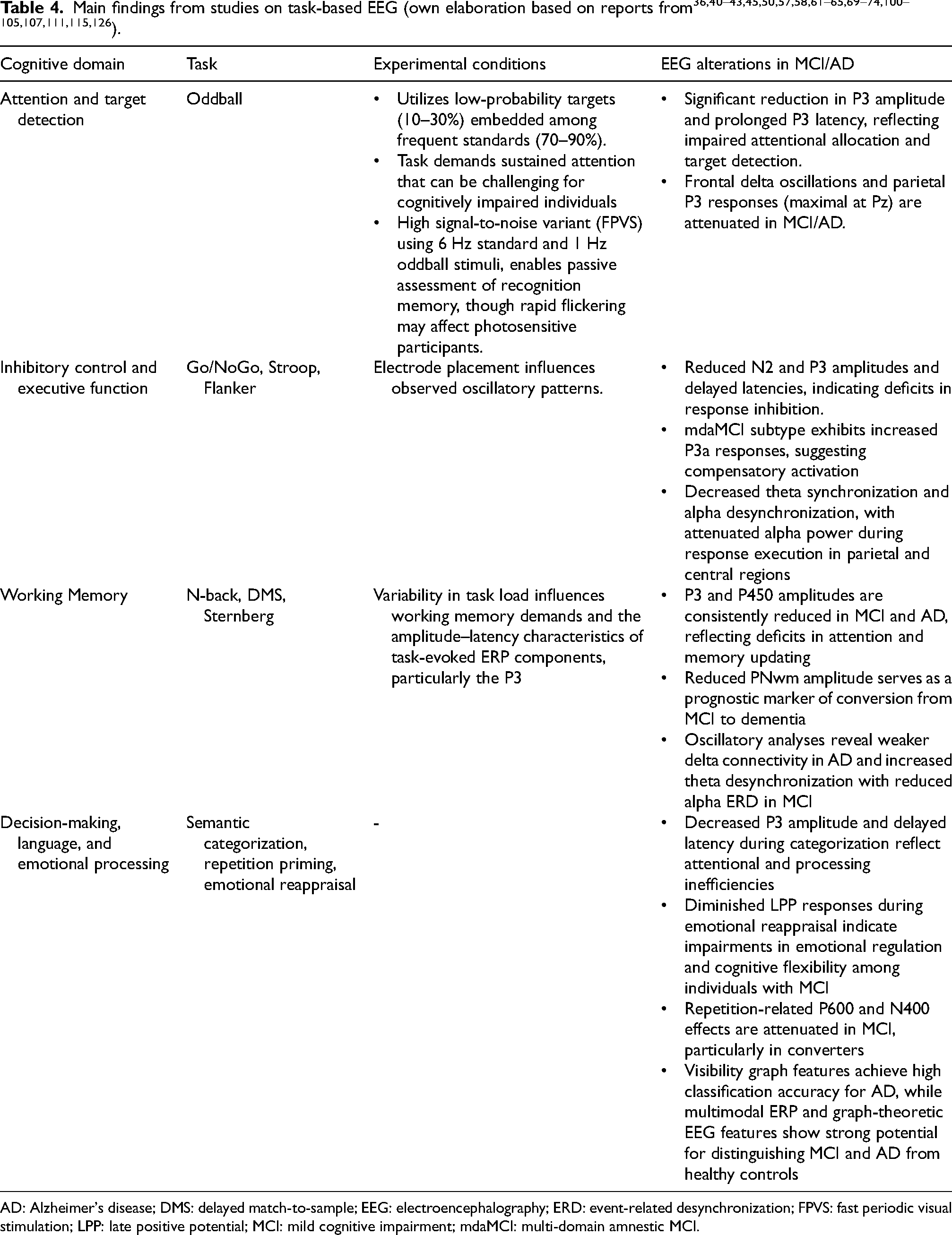
In attention and target detection tasks, such as the oddball paradigm, the task design consists of a train of frequent irrelevant stimuli (standard/non-target) interspersed with infrequent task-relevant stimuli (deviant/target or “oddball”). See Supplemental Table 1 for a summary of oddball-paradigm studies. Generally, frequent non-target auditory stimuli elicit early sensory ERP components such as P50, N1, and P2, whereas infrequent target stimuli elicit later components, most prominently N2 and P3. 33 Within this paradigm, MCI and AD groups consistently demonstrated reduced P3 amplitude and prolonged latency compared to HCs,34–36 with P3 latency showing the strongest correlation with AD severity (r = –0.512). 36 A preclinical MCI group, who had not yet been clinically diagnosed but whose neuropsychological screening indicated risk for developing MCI, also exhibited reduced P3 amplitude. 37 Notably, selective attention ERP features related to the P3 response demonstrated significant clinical utility, effectively classifying AD cases from healthy controls with effect sizes ranging from small to large (0.349–0.521 and 2.130–2.364), and achieving high predictive accuracy (AUC = 0.891) that outperformed traditional clinical measures such as the MMSE (AUC = 0.803). 38 Event-related oscillations (EROs), phase- or time-locked to a sensory or cognitive event obtained by filtering ERP signals, have also been proposed as biomarkers for detecting subtle cognitive process abnormalities in AD. Studies have reported that delta-range EROs derived from the P3 response were reduced in both AD 39 and MCI patients,40–42 whereas alpha and beta EROs showed no group differences.40,43 AD patients also showed reduced low-gamma (25–30 Hz) sensory-evoked oscillation amplitude over the left hemisphere and delayed gamma ERO latency compared to HCs. 44 Other ERP components have also been investigated. The N1 component, an indicator of cortical sensory processing, showed smaller amplitude in individuals with MCI compared to HCs. 45 In contrast, the P50 amplitude was elevated in MCI patients, and this increase was strongly associated with metabolic abnormalities in the left frontal cortex, suggesting that altered sensory gating responses may reflect underlying neuronal dysfunction. 46 Similarly, the late positive component (LPC) amplitude was enhanced in MCI, indicating alterations in memory encoding and retrieval processes. 45 Beyond ERP findings, connectivity analyses revealed decreased low-frequency coherence in AD, 47 increased theta coherence in MCI, 48 and disrupted alpha, beta, and gamma synchronization in both AD and amnestic mild cognitive impairment (aMCI). 49 Additionally, network-level synchronization parameters, including phase-locking value and amplitude coherence, demonstrated strong discriminative capacity, achieving classification accuracies exceeding 90% in distinguishing healthy controls from MCI and in differentiating AD from MCI. 49 Moreover, steady-state responses evoked during Fastball task effectively distinguished recognition memory performance between AD and HC groups, with a high AUC of 0.86. 50
In response inhibition tasks—such as Go/NoGo, Stroop, flanker, stop-signal, and continuous performance task (CPT)—executive dysfunction can be observed through impairments in conflict monitoring and conflict resolution,
51
two core processes of cognitive control. Findings from these tasks are summarized in Supplemental Table 2. Among these paradigms, the Go/NoGo task is one of the most widely used methods for assessing response inhibition, requiring participants to quickly respond to certain stimuli (Go stimuli) and withhold responses to others (NoGo stimuli). N2 and P3 are two key ERP components linked to response inhibition: N2 is typically elicited during the NoGo condition and is associated with inhibitory processes,52,53 whereas P3 is maximally generated in centroparietal areas during the Go condition and in frontocentral areas during the NoGo condition.54,55 These neural alterations are also observed in older adults, as response inhibition becomes less efficient with advancing age.
56
Additionally, paradigms that engage similar inhibitory control mechanisms—requiring selective attention and the ability to suppress responses to irrelevant stimuli—include tasks such as value-directed list learning and attentionally cued orientation discrimination. EEG studies have demonstrated that in such tasks, executive dysfunction in AD and aMCI, irrespective of subtype distinctions such as multi-domain (mdaMCI) and single-domain (sdaMCI), when compared with HCs, is indexed by reduced N2 amplitude,57–59 and prolonged N2 latency in aMCI.59–61 Aging appeared to magnify this effect, showing a stronger correlation between age and N2 latency in individuals with MCI than in healthy controls (r = 0.709 and 0.234, respectively).
60
Significant group differences in N2 latency (Cohen's d = 1.14) and amplitude (Cohen's d = 0.76) were reported between MCI and controls, whereas differences between MCI subtypes were comparatively smaller (Cohen's d = 0.33).
59
This pattern was also evident in N2 amplitude, showing a large effect size for differences between MCI and control groups (Cohen's d = 0.76) and a smaller effect between MCI subtypes (Cohen's d = 0.39).
59
Moreover, P3 measures demonstrated strong discriminative value, effectively distinguishing cognitively impaired individuals from healthy controls (Cohen's d ranging from 0.98 to 1.08;
Working memory tasks evaluate the ability to temporarily store and update information. To evaluate working memory, researchers commonly employ memory span tasks, including N-back, delayed match-to-sample (DMS), recognition memory, and Sternberg tasks (see Supplemental Table 3 for a summary of working-memory studies). In these paradigms, patients with MCI and AD consistently exhibited reduced P3 amplitude and prolonged latency,69–71 as well as smaller P450 amplitudes.72,73 Longitudinal data further showed that reduced the positive-negative working memory component (PNwm) amplitudes predicted progression to dementia. 74 Meanwhile, early visuo-cortical P1 and N1 components were comparable between individuals with and without cognitive impairment, indicating the preservation of visual processing. 75 Additional ERP and oscillatory features demonstrated predictive value, with P2 and N2 latencies and beta ERS reaching AUC values of 0.93, 0.78, and 0.72, respectively; combined, these features boosted classification accuracy to 90%. 76 Oscillatory analyses revealed reduced alpha ERD 73 and reduced alpha/beta ERS in MCI and AD cases, with individuals with MCI exhibiting more theta desynchronization than those with AD. 73,77,78 Functional connectivity was broadly reduced, particularly in delta and alpha bands.71,72,79 EEG complexity measures, characterized by higher Hurst exponent and lower entropy measures, diminished fractal dimension, and reduced mutual information, have been shown to distinguish MCI and AD from controls.80–82 Importantly, these altered connectivity and oscillatory markers not only discriminated between groups but also indicated risk of progression, with smaller alpha- and beta-band ERD in stable controls (sCON) compared to deteriorating controls (dCON) and reduced beta-band inter-trial phase synchrony in dCON relative to sCON. 75
Memory paradigms such as the DMS task, which comprises encoding, maintenance, and retrieval phases, are frequently used to assess recognition memory processes. In MCI patients, DMS tasks revealed reduced P3 amplitudes, particularly at left frontal sites,83–85 while repetition effects at P600 remained preserved 86 and even enhanced at posterior sites, 87 suggesting partial compensation in memory retrieval processes. For visuospatial memory tasks, which rely on the integrity of attention and working memory circuits, showed reduced alpha-band connectivity in MCI under high memory load conditions, with increased network centralization in both alpha and beta bands. 88 In contrast, AD patients exhibited left-hemisphere network dropout, characterized by reduced temporo-frontal connectivity during recognition and delayed signal transmission during priming, indicating lateralized memory circuit dysfunction. 89 Reduced connectivity in the upper alpha and beta bands, as measured by synchronization likelihood, was also observed in AD patients. 90
Other ERP markers of working memory and visual short-term memory showed variable results across studies. For instance, N2 and contralateral delay activity (CDA), reflecting encoding and maintenance of multiple objects, were altered in patient groups.37,91,92 Power spectral analyses provided further evidence of abnormalities: greater delta, theta, and alpha power were observed in both AD and MCI, 93 although reductions in theta power were specifically noted in AD patients 94 and in MCI individuals who later progressed to AD. 95 A consistent trend of diminishing alpha activity has been reported, with reduced alpha synchronization in MCI and diminished alpha power, particularly in the right hemisphere, in AD.82,94 Interestingly, no alpha suppression was observed in either MCI converters or non-converters, 95 suggesting that alpha modulation may differentiate between stable and progressive impairment. Beyond memory tasks, non-memory paradigms such as backward counting revealed increased total power in frontal and central regions for AD and MCI, respectively, achieving a classification accuracy of 85.4% when spectral and complexity features were combined. 96 Furthermore, substantial beta-band coherence differences, particularly in frontal and frontoparietal regions, were observed in MCI patients, with large effect sizes (Cohen's d > 0.5), suggesting that disrupted prefrontal connectivity underlies spatial navigation difficulties. 97 In addition, the MCI group exhibited increased beta- and theta-band power compared to the SCD group, 98 further implicating frequency-domain alterations in early disease stages.
In decision-making tasks, a component associated with processing negative feedback (feedback-related negativity: FRN) was reduced in aMCI during gambling paradigms, 99 while categorization tasks elicited reduced and delayed P3 responses. 100 and ERP as well as time–frequency features derived from these tasks discriminated MCI from controls with an accuracy of 87.9%, even when recorded from a single EEG channel. 101
In word repetition tasks, repeated congruent words elicited reduced P600, and incongruous words produced attenuated N400. Reduced P600 was observed in MCI and individuals progressing to MCI.102–104 N400 attenuation occurred in non-converters across right-hemisphere sites, while converters showed no effect 103 ; only one study reported prolonged N400 latency in MCI. 102 Visibility graph features from EEG differentiated AD and MCI converters from controls with 100% and 95.83% accuracy, respectively. 105 Meanwhile, EEG features derived from multimodal paradigms combining EEG, speech, and virtual reality achieved accuracy of 87% for MCI detection. 106 In emotional processing tasks, reduced late positive potential (LPP) in MCI, particularly in those at risk of dementia, reflected impaired reappraisal ability, with smaller amplitude differences between neutral and negative stimuli indicating difficulty attenuating negative responses. 107 Studies involving decision-making, language processing, and other cognitive domains are listed in Supplemental Table 4.
Discussion
This systematic review summarizes the use of EEG as a predictive biomarker for identifying different stages of the dementia spectrum, i.e., MCI and AD, and highlights the importance of cognitive paradigms over resting-state EEG. Cognitive paradigms offer more detailed insights into attention, memory, and cognitive processing. Since EEG can reveal early functional changes in the brain caused by AD, even before tissue damage or symptoms appear, this makes EEG a useful biomarker for predicting neuronal degeneration and a promising alternative or complement to current tools like MRI or PET scans. Together, these findings indicate that EEG captures disease stage–specific alterations, with MCI often characterized by early compensatory changes and AD by more extensive slowing, connectivity loss, and network breakdown. Such stage-specific differentiation between healthy aging, MCI, and early AD is especially valuable in the context of emerging disease-modifying treatments, such as monoclonal antibody therapies targeting beta-amyloid, which are most effective at the prodromal or early AD stage. By capturing functional changes before overt clinical decline, task-based EEG biomarkers provide a cost-effective and non-invasive means to support early detection, refine patient stratification, and monitor disease trajectory in clinical practice.
Prediction of MCI and AD via task-based EEG biomarkers
Several task-based EEG measures have demonstrated predictive utility in identifying individuals at risk of cognitive decline, offering insights into early neural changes that precede measurable behavioral impairments. In oddball tasks, reductions in P3 amplitude were repeatedly observed not only in clinically diagnosed MCI and AD patients but also in preclinical individuals identified as at risk on neuropsychological screening.34–37 This reduction suggests impaired allocation of attentional resources and diminished cognitive updating, processes critical to detecting and responding to salient stimuli. 112 Importantly, the strong correlation between P3 latency and disease severity underscores its value as an early marker and a progression indicator. 36 Moreover, P3-related responses have demonstrated superior discriminative accuracy compared to conventional cognitive screening tools such as the MMSE in distinguishing AD from healthy controls, 38 further reinforcing the role of P3 as a sensitive early marker of cognitive decline. This suggests that attentional ERPs, particularly those related to the P3 response, can provide both predictive and diagnostic value, reflecting heightened dementia risk more sensitively than conventional cognitive screening tools. Moreover, task manipulations such as varying stimulus presentation rates further reveal how neural responses are modulated; for instance, elevated auditory N1 amplitudes in MCI patients 109 suggest that rapid stimuli predominantly engage the auditory cortex, whereas slower rates recruit additional frontal regions,113,114 highlighting the sensitivity of ERP markers to both cognitive state and task context.
In language-based paradigms, particularly word repetition tasks, reductions in P600 and attenuated N400 responses have been associated with later conversion to MCI or AD, whereas individuals who remained stable retained N400 repetition effects.102–104 Similarly, decreases in alpha and beta synchrony differentiated deteriorating from stable control groups, 75 and reductions in theta power were specifically observed in MCI patients who subsequently progressed to AD. 95 Together, these findings suggest a consistent electrophysiological signature of disrupted semantic integration and memory retrieval processes across studies. Consequently, these measures may serve as sensitive neurophysiological indicators for identifying individuals at elevated risk of cognitive decline before the onset of overt clinical symptoms.
A consistent finding across working-memory paradigms is the emergence of compensatory frontal activity. Studies have reported enhanced left frontal ERP responses during non-target stimuli in MCI, alongside diminished responses to target stimuli, reflecting a compensatory mechanism to counter their impaired ability in inhibitory control.84,85 Importantly, these frontal ERP alterations have been detected up to five years before clinical diagnosis, 85 supporting their value of frontal memory-related neuromarkers for individualized monitoring of disease progression. Furthermore, as individuals aged, particularly with the onset of MCI, the ability to suppress distractors became more challenging. Correspondingly, reductions in P3 (memory recall), N400 and P600 (semantic memory) and alpha/beta activity (attention and inhibitory control) are frequently observed among those at increased likelihood of cognitive impairment. Additionally, variations in theta oscillations observed across studies may reflect differences in cognitive control strategies rather than uniform patterns of neural dysfunction. Therefore, despite differences in task design and analytical methods, the consistent decreases observed in these task-based EEG measures reinforce their reliability as biomarkers for detecting early cognitive impairment.
Classification of MCI and AD via task-based EEG biomarkers
Several task-based EEG features have demonstrated strong potential for classifying between groups. Network-level measures of neural synchronization and cross-frequency coupling metrics show improved classification accuracies by capturing disrupted hierarchical communication across frequency bands, even for more complex three-class classifications.49,52,115,116 This evidence reinforces the clinical value of network-based synchronization measures as robust discriminators along the AD continuum. As individuals age, EEG patterns typically show a decrease in alpha power and an increase in slower brain wave frequencies like delta and theta. These slow waves differ significantly from those typically associated with AD. Unlike AD-related slow activity, these nonpathological slow waves are not linked to a substantial asymmetry of the alpha rhythm, tend to diminish with mental activity or eye opening, and occur only sporadically as single waves or pairs rather than in continuous rhythmic trains. 117 Reductions in slow-wave activity may indicate cognitive improvement in patients responding to cholinesterase inhibitor therapy further supports their clinical relevance. 118 Desynchronization in the alpha band remarkably increased (less negative) in centroparietal regions and has been associated with attention, indicating reduced attentiveness and correlating with poor performance.119,120 Moreover, variations in theta power have also demonstrated discriminative potential in distinguishing cognitively healthy individuals from those with impairment, 63 while abnormalities in theta ERS further suggest disruptions in attentional control networks that contribute to cognitive dysfunction. 121 However, results contrary to the findings in MCI patients have emerged, demonstrating an absence of beta ERD in individuals with AD during the retrieval phase of short-term memory processing. 78 This may suggest that the AD pathology disrupts the normal desynchronization pattern of neural activity in this frequency band. These effects, known as a general “slowing” of EEG activity. 122 Given that dementia is typified by a “disconnection syndrome,” marked by diminished brain networks,123,124 individuals consistently manifest impaired functional connectivity, reduced complexity and perturbation of synchrony as AD progresses. Similarly, selective-attention ERP and spectral features have demonstrated high classification performances between cognitively impaired and healthy individuals.67,101 These findings underscore the diagnostic utility of EEG markers that capture deficits in attention, memory, and inhibitory control.
Across studies, prolonged N2 latency and reduced N2 amplitude indicate slowed cognitive processing and deficits in early attentional control among individuals with MCI,59,60 while reduction in P3 amplitude and delayed latency further reflect impairments in attentional allocation and working memory updating.34,37,62 Beyond amplitude and latency, trial-to-trial variability in P3 responses has been reported as an additional discriminative feature, reflecting the instability of attentional resource allocation in MCI and AD.57,91 The robustness of these findings supports the relevance of P3 as a potential clinical biomarker. Meanwhile, elevated P50 amplitude in MCI, which correlated negatively with left frontal metabolic abnormalities but not with MMSE performance, suggests that abnormal sensory gating may capture localized neuronal dysfunction rather than global cognitive status, limiting its reliability as a general clinical marker.36,46 In contrast, the increased LPC amplitude observed in MCI likely reflects compensatory perceptual processing in response to early sensory deficits, 45 underscoring the potential of ERP components to differentiate localized dysfunction from adaptive mechanisms.
Finally, differentiation between stable and progressive MCI remains underexplored, though it is critical for clinical decision-making. Evidence suggests that altered oscillatory activity (e.g., reduced theta power, 95 smaller alpha/beta ERD 75 ) and P3 amplitude differences62,85 may serve as indicators of progression risk, yet few studies stratify MCI patients by conversion status. Clarifying these criteria is necessary for EEG biomarkers to provide meaningful clinical guidance, particularly in identifying which patients are most likely to benefit from early interventions or monitoring. Nevertheless, classification outcomes vary widely across studies due to differences in task design, including stimulus modality, stimulus duration, and number of trials. The number of extracted features also varies widely, from large sets to only a few selected features, making comparisons between studies difficult. Preprocessing strategies also vary considerably, encompassing artifact rejection criteria, filtering approaches, and baseline correction procedures, each of which can affect the resulting EEG signals. These methodological differences highlight the adaptability of EEG but also underscore the pressing need for standardized protocols and consensus on core feature sets to ensure reproducibility and facilitate translation into clinical practice.
Monitoring disease progression via task-based EEG biomarkers
In oddball paradigms, P3 latency correlated strongly with AD severity and increased longitudinally over 18 months in probable AD patients, whereas N2 latency exhibited a weaker but still significant correlation.36,100 Findings from working memory paradigms further support this progression pattern, as reductions in PNwm amplitude have been linked to the transition from MCI to dementia. 74 Connectivity-based studies complement these results, demonstrating greater network centralization in late compared to early aMCI. 65 Moreover, oscillatory analyses revealed progressive reductions in theta and alpha power in both MCI and AD,94,95 with diminished alpha synchronization particularly evident in the right hemisphere of AD patients.82,94 Additionally, differences in theta ERS have shown promise in distinguishing between early and late aMCI groups. 65 These findings indicate that differences in theta and alpha oscillations between early and late MCI may serve as neural progression markers capable of tracking disease trajectory even when behavioral performance remains similar.
The N400 and P600 (or LPC) components reflect semantic processing and episodic memory, and their modulation by repeated (old) versus novel words, known as the repetition effect, provides insight into neural memory function . 103 In individuals with MCI, both N400 and P600 repetition effects were reduced, with the most pronounced deficits observed in those who later progressed to AD.102–104 Notably, the P600 effect was absent in MCI converters within three years, and by the second year, the N400 repetition effect had disappeared entirely in converters, while non-converters maintained a pattern similar to that observed at baseline . 103 These findings indicate that diminished or absent N400 and P600 repetition effects may serve as sensitive early markers of disease progression, offering a potential tool for identifying individuals at high risk for dementia.
Connectivity disruptions provided additional markers of progression. Left-hemisphere network dropout during recognition and delayed signal transmission during priming tasks were reported in AD, reflecting lateralized memory circuit dysfunction, 89 while reduced synchronization in upper alpha and beta bands characterized advanced disease stages. 90 Complexity measures, including higher Hurst exponent, lower entropy, and diminished fractal dimension, further distinguished controls from cognitively impaired groups and tracked worsening impairment.80–82 The observed deterioration in AD may be attributed to the presence of plaques and cell death, leading to a reduction in connectivity between cortical neurons. This reduction could result in increased regularity of brain signals, ultimately impacting communication efficiency throughout the brain. These findings support the concept of a disconnection syndrome emerging in MCI, potentially progressing to AD or other forms of dementia. Key findings from studies on task-based EEG are summarized in Table 4.
AD: Alzheimer's disease; DMS: delayed match-to-sample; EEG: electroencephalography; ERD: event-related desynchronization; FPVS: fast periodic visual stimulation; LPP: late positive potential; MCI: mild cognitive impairment; mdaMCI: multi-domain amnestic MCI.
The scope of this study pertains to manuscripts derived from the Open Access Policy, ensuring accessibility and transparency in disseminating findings. The insights summarized here, drawn from scientifically acknowledged and accessible protocols, support the future development of innovative approaches to MCI screening, consistent with other publications. 125 Methodological and practical limitations should be acknowledged. Potential omissions of relevant studies due to database or search-term constraints, variability in diagnostic criteria, and limited reporting of effect sizes reduce cross-study comparability and the robustness of clinical interpretations. Moreover, translation of EEG findings into clinical practice remains constrained by heterogeneity in task design, stimulus parameters, preprocessing pipelines, and analytical methods. The absence of standardized protocols and normative datasets makes it difficult to compare results across laboratories and to validate cut-off values that could guide clinical decision-making. Moreover, there is a notable gap between the controlled conditions of laboratory-based cognitive paradigms and the realities of clinical or hospital environments, where factors such as time constraints, patient fatigue, and comorbidities may influence feasibility and data quality. To advance clinical translation, future studies should prioritize protocol harmonization, replication in large multi-center cohorts, and integration with existing diagnostic frameworks. Such steps are critical for establishing reliable, reproducible EEG-based predictors that can complement current neuropsychological and imaging assessments in routine clinical settings.
Conclusions
This review highlights the evidence supporting task-based EEG as biomarkers for early detection and monitoring of cognitive decline across the MCI-AD continuum. Characteristic EEG alterations, most notably decreased P3 and N2 amplitudes, prolonged latencies, reduced alpha and beta activity, and disrupted connectivity, reflect impairments in attention, memory, and cortical inhibition that arise prior to overt clinical symptoms. These converging findings indicate that task-evoked EEG measures capture both early neurophysiological dysfunction and compensatory processes, offering potential utility in disease staging and prognosis. Clinically, task-based EEG provides a low-cost, noninvasive complement to traditional cognitive and neuroimaging assessments, supporting its integration into multimodal diagnostic frameworks.
Supplemental Material
sj-docx-1-alz-10.1177_13872877251413700 - Supplemental material for Biomarkers for Alzheimer's disease and mild cognitive impairment: Recent advances in task-based EEG
Supplemental material, sj-docx-1-alz-10.1177_13872877251413700 for Biomarkers for Alzheimer's disease and mild cognitive impairment: Recent advances in task-based EEG by Kornkanok Tripanpitak, Alexandra Wolf, Maryna Kapitonova, Satoshi Umeda, Tonio Ball and Mihoko Otake-Matsuura in Journal of Alzheimer's Disease
Footnotes
Acknowledgements
The authors express gratitude to the RIKEN Center for Advanced Intelligence Project (AIP) for supporting this research.
Ethical considerations
Not applicable
Consent to participate
Not applicable
Consent for publication
Not applicable
Author contribution(s)
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was partly supported by the Japan Science and Technology Agency AIP Trilateral AI Research (Grant Numbers: JPMJCR20G1) to MO-M and KT. MO-M was supported by JSPS KAKENHI (Grant Numbers: JP22H04872 and JP22H00544) and the Japan Science and Technology Agency (Grant Numbers: JPMJPF2101). AW was supported by the Special Postdoctoral Research (SPDR) grant under the RIKEN SPDR Fellowship. MO-M and SU was supported by the Japan Science and Technology Agency (Grant Numbers: JPMJMS2237). TB and MK were supported by the AI-Cog (DFG, Germany project BA 4695/4-1).
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
The data supporting the findings of this study are available within the article and/or its supplemental material.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.