Abstract
Background
Individuals with mild cognitive impairment (MCI) are at an increased risk of developing Alzheimer's disease. Anatomical and functional brain alterations associated with this condition are still elusive.
Objective
This study explored the cognitive correlates of cortical thickness, white matter (WM) volume, and resting-state connectivity among people with MCI.
Methods
A total of 56 older participants (aged 51 to 92 years) with amnestic MCI were recruited. Cognitive abilities were measured using the Trail Making Task, the Stroop Color-Word Test, the Forward and Backward Digit Span test, and computerized n-back tasks. Morphometry was used to measure cortical thickness and WM volume from 3 T MR images, while functional connectivity was measured using resting-state fMRI and calculated using Independent Component Analysis. Voxel-wise regressions were used to test associations between cognitive scores and brain measures.
Results
Worse working memory updating (n-back) performance was associated with lower cortical thickness of the left middle temporal gyrus. Additionally, at a lower demand, working memory performance was linked to frontoparietal network (FPN) intrinsic connectivity, while WM volume within the anterior segment of the left arcuate fasciculus and default mode network (DMN) resting-state connectivity were relevant when the demand was higher. Lower DMN connectivity was also associated with worse conflict monitoring (Stroop) performance (all cluster-corrected ps < 0.05).
Conclusions
The findings highlight the relevance of the perisylvian region to working memory updating and conflict monitoring in people with MCI.
Keywords
Introduction
Mild cognitive impairment (MCI) can be seen as the transitional stage between normal aging and dementia, at which individuals experience cognitive declines of insufficient severity to warrant a dementia diagnosis and demonstrate essentially normal functional activities.1,2 A recent meta-analysis estimated a worldwide prevalence rate of 15.56% among community-dwelling older adults. 3 Although this condition can revert to normal aging, 4 people with amnestic type of MCI are at least eight times more likely to develop Alzheimer's disease (AD) and other dementias as compared to healthy controls. 5 As diagnosis of MCI is so far based on cognitive symptoms, 6 a clearer link between cognitive decline and brain alterations could advance early detection, help refine diagnostic criteria, and improve prognosis of disease progression. While both AD and MCI show clear anatomical and functional brain abnormalities,7,8 the direct relationship between these alterations and levels of cognitive functioning remains poorly understood.
Structurally, widespread cortical thinning has been observed in people with MCI, including in the temporal and parahippocampal areas.9,10 There is also evidence that cortical thickness is correlated with cognitive measures (e.g., global cognition, episodic memory, and working memory) among MCI patients, but not among healthy controls.11,12 However, Julkunen et al. 9 observed a link between global cognition and cortical thickness only in AD, not in MCI. Additionally, Li et al. 13 reported white matter (WM) atrophy among AD patients in the structures relevant to memory functioning. Among people with MCI, however, the evidence has been inconsistent with some studies reporting abnormalities and an association with memory, while others not. 14 This might be because age-related macrostructural decline (including volumetric changes) does not affect all WM tracts equally. 15 Therefore, it is warranted to investigate WM atrophy in specific WM tracts.
The superior longitudinal fasciculus (SLF), the arcuate fasciculus (AF), and the cingulum bundle are of particular interest. The SLF is a major associative WM tract connecting the ipsilateral perisylvian regions (i.e., the frontal, temporal, and parietal lobes). 16 This broad connection is important for attention and executive function. Traditionally known as a part of the SLF, 17 the AF links the frontal and temporal lobes and is associated with various cognitive functions, including short-term and working memory. 18 Similarly, the cingulum has a longitudinal projection that runs from the frontal lobe, along the cingulate cortex, and towards the temporal lobe. Due to its projection, this tract is implicated in memory, working memory, and executive functions.19,20 Microstructural alterations of these tracts have been documented in MCI patients. 21 However, evidence of macrostructural changes and their cognitive correlates in individuals with MCI is scarce.
Functional aberrations have also been observed in MCI and studies showed that as compared to healthy controls, individuals with MCI demonstrated abnormal functional connectivity in the default mode network (DMN), 22 the frontoparietal network (FPN), 23 and the salience network (SN). 24 The DMN is the largest resting-state network which typically shows decreases in activity during cognitive tasks. 25 Its activity has been shown to be associated with executive function in older adults 26 and alterations of its connectivity pattern has been reported among people with MCI. 27 Meanwhile, the FPN and the SN are both task-positive systems. Connectivity in both networks was shown to be related to executive functions, such as working memory, inhibition, and set-shifting ability of older adults. 28 Yet, there is limited evidence of the association between connectivity in these three networks and cognition in MCI populations.
This study aimed to explore the cognitive correlates of cortical thickness, WM volume, and resting-state connectivity in older adults with MCI. It used T1-weighted and resting-state T2*-weighted magnetic resonance images which have several benefits. As a standard medical imaging modality, T1-weighted image is easily available and has been shown to reliably provide markers for MCI. 29 Moreover, resting-state functional magnetic resonance imaging (fMRI) can also be easily incorporated into the standard practice as it is relatively simple, requires little cooperation from the patient, and allows the investigation of different functional networks simultaneously. 30
In this study, we tried to expand previous evidence by directly examining how the severity of cognitive decline was associated with brain measures in people with MCI. As cortical thinning in MCI has been shown to affect regions important for short-term and working memory, we hypothesized that it would correlate with these domains of cognition. Furthermore, based on previous studies in healthy individuals, we hypothesize that the WM volume in the SLF would be associated with executive function, while volume in the AF with short-term and working memory. For the cingulum, associations with short-term memory, working memory, and executive function were expected. Lastly, following the existing evidence from studies with older adults, we hypothesized that intrinsic DMN connectivity would correlate mainly with executive function, while connectivity within the FPN and the SN with both working memory and executive function.
Methods
Inclusion criteria and recruitment
The current study used the baseline data from the COGMAX project (ABR number NL64677.042.18) which investigated the effect of transcranial alternating current stimulation (tACS) on frontoparietal connectivity and executive functions. The project recruited people with amnestic MCI from the memory clinic of the University Medical Center Groningen (UMCG) and from the community through advertisements. To be included in the project, participants must score at least 1.5 standard deviations below the normative control values in a verbal memory test (i.e., the 15-Word Test; see Table 1). The diagnosis of amnestic MCI (aMCI) was then evaluated and confirmed by a neurologist prior to inclusion. Meanwhile, the exclusion criteria include: (1) history of psychiatric or neurological illnesses other than MCI; (2) any metal implants (e.g., pacemaker, heart valves) or particles; (3) tattoos containing iron peroxide; (4) pregnancy or breastfeeding; (5) claustrophobia; (6) alcohol or drug abuse; (7) severe lesions on the scalp; (8) color-blindness; and (9) refusal to be informed of structural brain abnormalities detected by MRI. Participants were also instructed to avoid alcohol use for two days prior to the MRI measurement.
Sample characteristics (n = 56).

T-scores based on the Dutch normative data;
Percentile scores based on the Dutch normative data;
n = 53.
A total of 56 participants with anatomical MRI (aMRI) data were included in the current study. The clinical diagnosis of AD-type MCI was made in four participants who were referred to the study by neurologists, while the other participants were included based on their MCI clinical profile. Thirteen participants exhibited deficits in non-memory cognitive functions (i.e., multiple domain aMCI), of whom 11 showed impairment in task-switching ability, one in conflict monitoring, and one in both domains. Participants’ age ranged from 51 to 92 years (mean = 71.67, SD = 7.65). Most participants were male (71.43%) and right-handed (92.9%). Additionally, 48.21% of the participants completed high-level secondary education, while 19.64% had university-level education. Participants’ scores on the Mini-Mental State Examination (MMSE) ranged from 21 to 30 (mean = 26.63, SD = 2.20). Scores in the 21–24 range indicate mild global cognitive impairment consistent with an MCI profile, and no participants scored in the range typically associated with moderate severe dementia. Of all included participants, only 53 had resting-state fMRI scans.
Cognitive measures
To measure various aspects of cognitive function, participants underwent a neuropsychological evaluation. This included the following tests: (1) Trail Making Task (TMT); (2) Stroop Color-Word Test (SCWT); (3) the Wechsler Adult Intelligence Scale (WAIS) Digit Span subtest; and (4) computerized n-back tasks. In TMT, participants were asked to connect randomly placed numbers sequentially (TMT-A) and to connect alternately between randomly positioned numbers and letters (TMT-B) as soon as possible. The first part is thought to measure processing speed, while the latter taps more into task-switching ability.31,32 Originally developed to assess cognitive interference, 33 SCWT comprised three parts where participants were instructed to read out words of colors (SCWT-1), to name the color in which a set of rectangles were printed (SCWT-2), and to name the color in which words of colors were printed (suppressing to mention the written words; SCWT-3). Only the corrected TMT-B score (for TMT-A score) and the corrected SCWT-3 score (for SCWT-2 score) were used in the current study as they provided a better indication of executive functions (i.e., task switching and conflict monitoring, respectively). Both scores were transformed into percentile scores in accordance with the Dutch normative data, 34 adjusting for sex, age, and education level.
The WAIS Digit Span subtest is a measure of short-term memory (Forward DS) and working memory (Backward DS). 35 In this subtest, participants were instructed to repeat sequences of numbers in the same order they were orally presented (Forward DS) and in the reverse order (Backward DS). Another common measure of working memory (updating) is the n-back task. 36 Here, participants were asked to identify if a letter was the same as the letter presented one time (1-back condition) or two times (2-back condition) before. The task was performed during fMRI measurements (not included in this study). One participant did not complete this measurement. In total, participants were given eight blocks of trials with four blocks for each n-back condition in a pseudorandomized order. For each block of trials, d’ was calculated using the “psycho” package in R. 37 Then, d’ values were averaged across blocks for each condition. d’ reflects a signal discriminatory ability and is thought to be a better index of n-back performance as it is calculated from both hit and false alarm rate. 38 The sample characteristics based on the included cognitive measures are summarized in Table 1.
Magnetic resonance imaging acquisition
All MRI data acquisition was conducted with a 3-Tesla Siemens Magnetom Prisma scanner. As anatomical data, 3D magnetization-prepared rapid gradient-echo T1-weighted sequences were acquired (repetition time (TR) = 2300 ms; echo time (TE) = 2.98 ms; field of view (FoV) = 256 × 256 × 256 mm3; voxel size = 1 × 1 × 1 mm3; flip angle (FA) = 9°; 176 slices; duration = 9 min 14 s). For resting-state functional data, 2D echo-planar T2*-weighted sequences were obtained with multiple echo times (number of volumes = 220; TR = 2170 ms, TE = 9.74 ms, 22.10 ms, and 34.46 ms; FoV = 224 × 224 × 224 mm3; voxel size = 3 × 3 × 3 mm3; FA = 60°; 39 slices; duration = 8 min 28 s) while participants kept their eyes closed.
Anatomical image preprocessing: voxel-based and surface-based morphometry
Anatomical data were preprocessed using the Computational Anatomy Toolbox (CAT12), 39 in Matlab R2023a. 40 The preprocessing pipeline involved tissue segmentation to obtain white matter volume and spatial registration using the DARTEL approach. 41 Segmented maps were then modulated to preserve the total amount of white matter and normalized to the MNI space. After that, smoothing with an 8 mm full width at half maximum (FWHM) Gaussian kernel was conducted to the modulated-normalized maps. Additionally, cortical thickness was also estimated. Thickness data were then resampled to 32k template space and smoothed using 12 mm kernels.
To show the validity of our data, we examined the correlations between hippocampal volumes and MMSE scores. To that end, we extracted the hippocampal gray matter (GM) volumes (see Supplemental Analysis: Hippocampal volume analysis). Hippocampal atrophy has been reliably documented in MCI and is associated with impairment severity.42,43
Functional image preprocessing: independent component analysis
Resting-state functional images were preprocessed using the fMRIPrep 23.1.4. 44 In a nutshell, this preprocess involved estimation of head motion parameters, a slice timing correction, resampling into the native space for motion correction, the combining of the multiple echo images using the nonlinear regression method, a co-registration to the skull-stripped T1-weighted image with six degrees of freedom, and finally resampling into the standard MNI space. No initial volumes were removed during this preprocessing as the first two volumes were already discarded during acquisition and fMRIPrep also employed automatic detection of nonsteady-state volumes. The outputs of fMRIPrep were then post-processed using the eXtensible Connectivity Pipeline -DCAN (XCP-D).45,46 The post-processing pipeline involved the framewise displacement (FD) calculation with an automatically estimated head radius for each participant. The data was despiked before a denoising followed using 24 nuisance regressors (including the six motion parameters with their temporal derivatives and the quadratic expansion of the six motion parameters with their temporal derivatives), in addition to linear trend and intercept terms. Any censored volumes were then interpolated and filtered with a fourth-order Butterworth filter to retain signals between 0.01 and 0.1 Hz. After that, the filtered and interpolated time series were re-censored to remove volumes with FD values greater than 0.5 mm. Finally, the data were smoothed with a 6 mm FWHM Gaussian kernel.
Subsequently, the post-processed functional images were subjected to a group independent component analysis (gICA) using the GIFT toolbox (http://icatb.sourceforge.net/) in Matlab R2023a. The gICA was run using the infomax algorithm and the ICASSO method with “randinit” mode and five runs. In order to further clean the data from noise, the gICA was conducted in two rounds. In the first round, 25 independent components (ICs) were extracted. After visual inspection of the component spatial maps, two noise-dominant ICs were removed. A second round was then run using the modified data from the first round as the input to extract 20 ICs. Whole-brain spatial maps and time courses of each IC were back-reconstructed for each participant. Both back-reconstructed data were transformed into standardized Z-scores. ICs belonging to the DMN, FPN, and SN were identified through visual inspection of the spatial maps and the help of GIFT's component labeler utility. Figure 1 shows these ICs. The spatial maps of each IC represent the contribution of each voxel to the functional connectivity within each particular IC.
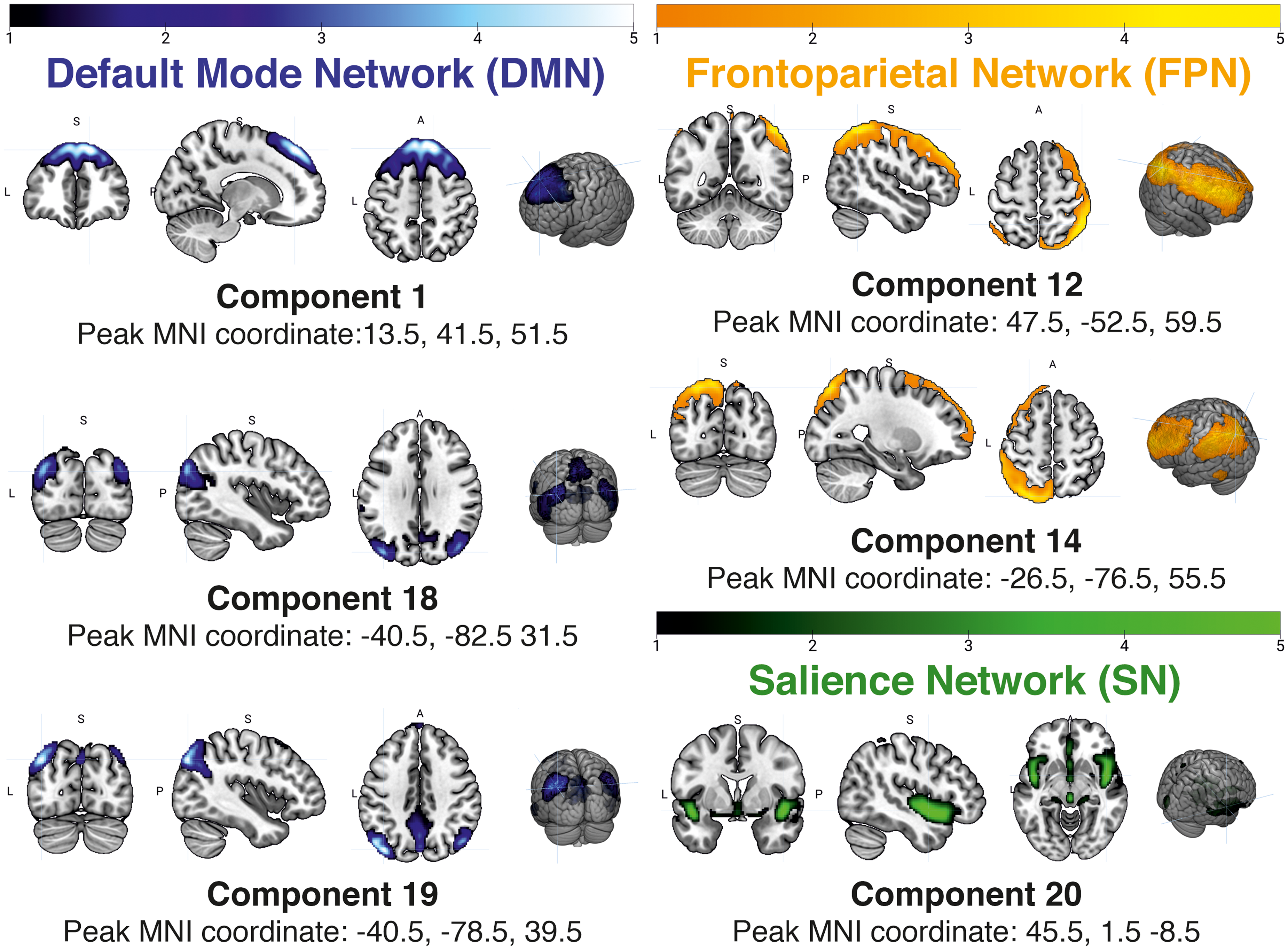
Spatial maps of the extracted independent components. Left panel: the three ICs shown belong to the DMN; Upper right panel: the two ICs belonging to the FPN; and Bottom right panel: one IC belonging to the SN. Images are shown in the neurological convention: the left hemisphere is shown in the left side of the image.
We further investigated the correlation between ICs and the cognition (see below). In addition, an exploratory analysis was performed to investigate the synchronization within and between the DMN, the FPN and the SN (see Supplemental Analysis: Time course correlations between independent components).
Statistical analysis
After the preprocessing of the anatomical and functional images, voxel-wise statistical analyses were conducted in SPM-12. In the morphometric analysis, one participant was excluded for having an overall bad anatomical image quality (i.e., IQR lower than 75%). Separate multiples regressions were run to estimate correlations between each cognitive scores and cortical thickness or WM volume using the CAT12 outputs. For WM volume analysis, correction for total intracranial volume (TIV) was done with proportional scaling. We investigated correlations of the following six regions with cognition: the SLF, the dorsal and parahippocampal cingulum, and the three segments of AF. More specifically, binary masks (voxel size = 1 × 1 × 1 mm3) of the bilateral SLF (number of non-zero voxels (nnz) = 13,212 voxels), bilateral dorsal cingulum (cingulate gyrus segment; nnz = 5093 voxels), and bilateral parahippocampal cingulum (hippocampus segment; nnz = 2391 voxels) from the JHU ICBM-DTI-81 White-Matter Labels atlas in FSL, as well as binary masks of the anterior (nnz = 57,666 voxels), posterior (nnz = 85,928 voxels), and long segment (nnz = 40,935 voxels) of the bilateral AF from Thiebaut de Schotten et al. 47 were applied.
Furthermore, the relationships between resting-state functional connectivity and cognitive measures were also analyzed with separate multiple regressions using the back-reconstructed spatial maps from the gICA. As previously mentioned, only 53 participants had functional images. The T2* sequence of one participant also deviated from the aforementioned acquisition parameters and thus was excluded from the analysis, resulting in a final sample of 52 participants for the resting-state analysis. Binarized spatial maps (voxel size = 1 × 1 × 1 mm3) of the DMN (nnz = 11,201 voxels), FPN (nnz = 2455 voxels), and SN (nnz = 1173 voxels) from Neurosynth were used as explicit masks for each IC to constrict the analysis to only include the voxels within these networks.
Results were evaluated using a cluster-corrected threshold of p < 0.05, which consisted of a minimum number of voxels (depending on the mask size) combined with an alpha level of p < 0.001. For the cortical thickness analysis, the native cluster threshold estimation in SPM12 was used. Meanwhile, for WM volume and resting-state functional connectivity, the cluster thresholds were calculated through Monte Carlo simulation using AFNI's 3dClustSim program. When significant results were observed, a family-wise error (FWE) corrected threshold of p < 0.05 was also applied to demonstrate the robustness of the findings under a conservative correction standard. Lastly, for exploratory analysis of hippocampal volumes and temporal synchronization of ICs, a stringent correction for multiple testing was used, namely the Bonferroni correction.
Results
Cortical thickness
Lower cortical thickness was associated with worse performance in both 1-back and 2-back tasks. Associations with 1-back performance were found in various brain regions (Left panel in Figure 2). On the left hemisphere, two significant clusters were found in the temporal (Z = 4.34, k = 1302, MNI coordinate = -67 −29 4, AAL label = left Middle Temporal Gyrus) and near the insular area (Z = 3.85, k = 252, MNI coordinate = -37 −19 −3, AAL label = (Near) left Insular cortex). Other two clusters were found on the right hemisphere, specifically in the parietal (Z = 4.30, k = 312, MNI coordinate = 49 −54 33, AAL label = right Angular Gyrus) and temporal lobe (Z = 3.89, k = 306, MNI coordinate = 59 −31 11, AAL label = right Superior Temporal Gurys). It is interesting to note that, even under the most stringent threshold (i.e., FWE-corrected p < 0.05), the clusters in the left temporal lobe and the right parietal lobe still survived. The left temporal cluster was split into four significant clusters, while nine voxels of the right parietal cluster remained significant (see Supplemental Table 1 which summarize the statistical results at FWE-corrected threshold of p < 0.05). In addition, cortical thickness in the left temporal area was also linked to 2-back performance (Z = 3.48, k = 149, MNI coordinate = -56 −44 7, AAL label = left Middle Temporal Gyrus; see the right panel of Figure 2). No significant correlations were found between cortical thickness and other cognitive domains.
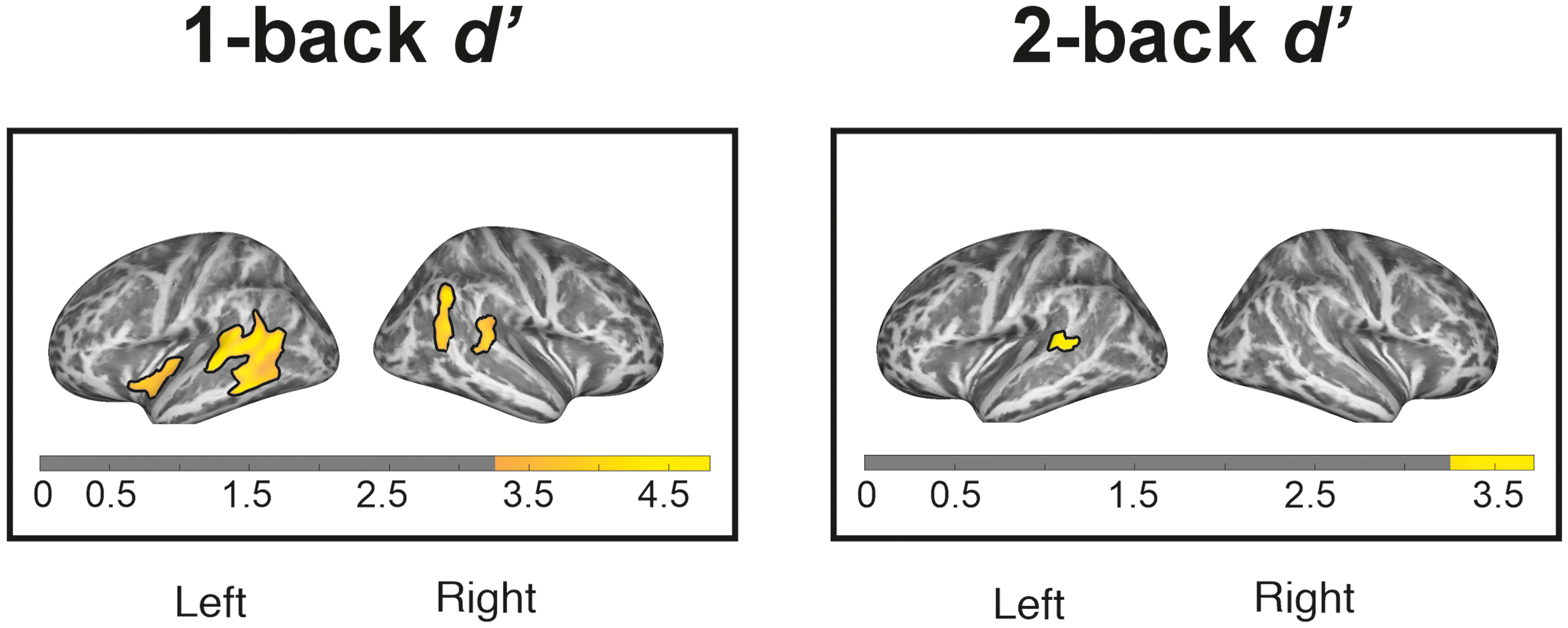
Relationship of cortical thickness with 1-back (left panel) and 2-back (right panel) performance. Significant clusters at p < 0.001 with cluster thresholding (cluster-corrected p < 0.05) are marked with solid outlines.
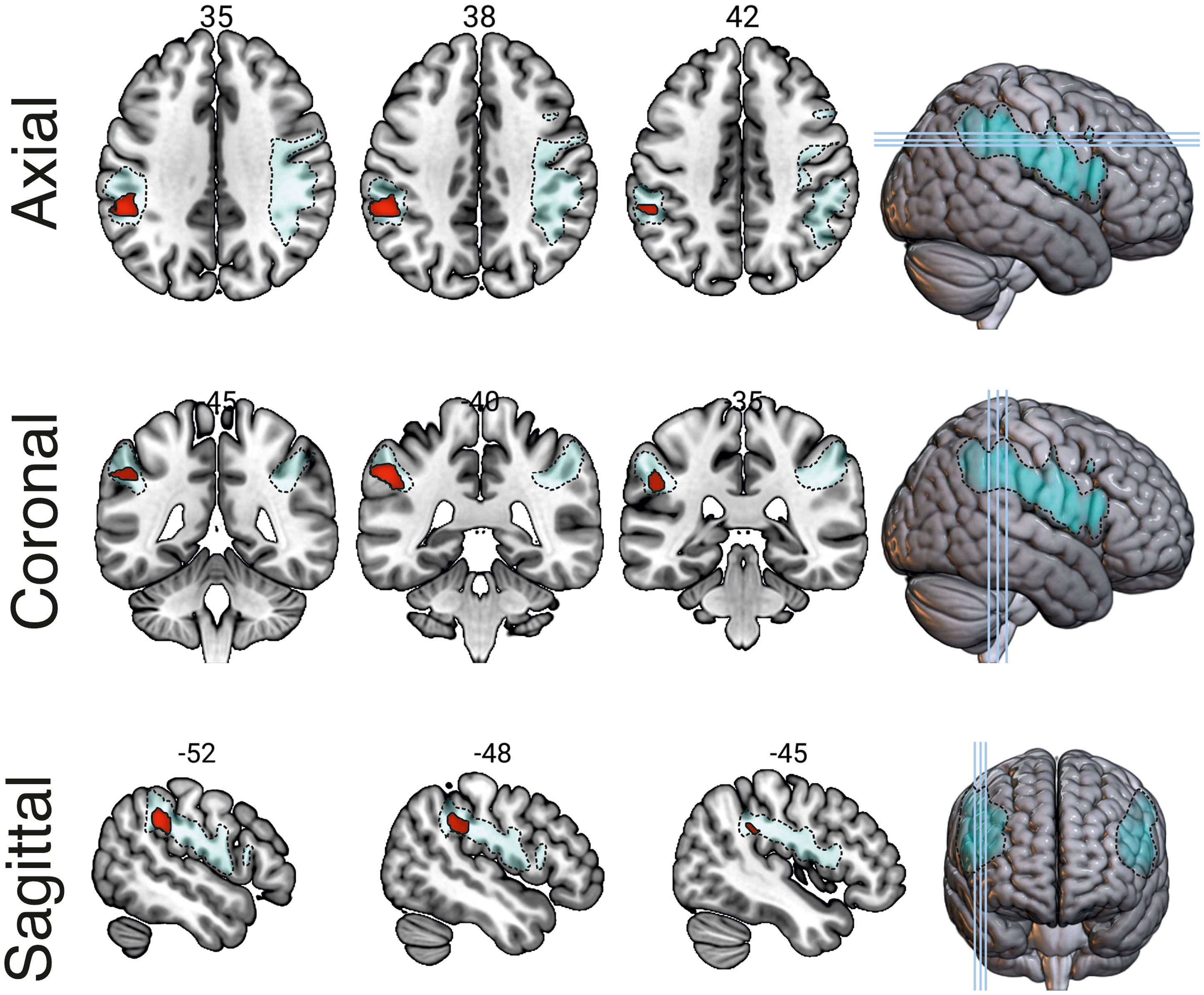
Relationship between white matter volume within the anterior arcuate fasciculus (marked with dashed outlines) and 2-back performance. The region enclosed by a solid outline indicates significant clusters at p < 0.001 with cluster thresholding (cluster-corrected p < 0.05). Images are shown in the neurological convention: the left hemisphere is shown in the left side of the image.
White matter volume
Lower WM volume in the left anterior AF was correlated with lower 2-back performance (Z = 4.97, k = 512, MNI coordinate = -56 −40 38, AAL label = left Inferior Parietal Lobe; see Figure 3). This significant cluster was still observed, albeit with a much smaller number of voxels, when implementing a stringent FWE-correction at 0.05 without cluster correction (k = 2, cluster pFWE−corrected = 0.038, MNI coordinate = -56 −40 38, AAL label = left Inferior Parietal Lobe). Meanwhile, no significant correlations were found between WM volume and other task performance.
Resting-state functional connectivity and cognition
Within the FPN, a significant positive correlation was observed between functional connectivity in IC number 12 (i.e., the left FPN) and 1-back task performance (Z = 4.02, k = 53, MNI coordinate = -12 −60 54, AAL label = left Precuneus). This effect is shown in Figure 4. Additionally, upon further exploratory analysis, it was found that the time course of IC number 12 was significantly correlated with that of IC number 14 (i.e., the right FPN, t(51) = 13.27, puncorrected < 0.001, pBonferroni < 0.001; see Supplemental Analysis that shows time course correlations between ICs).
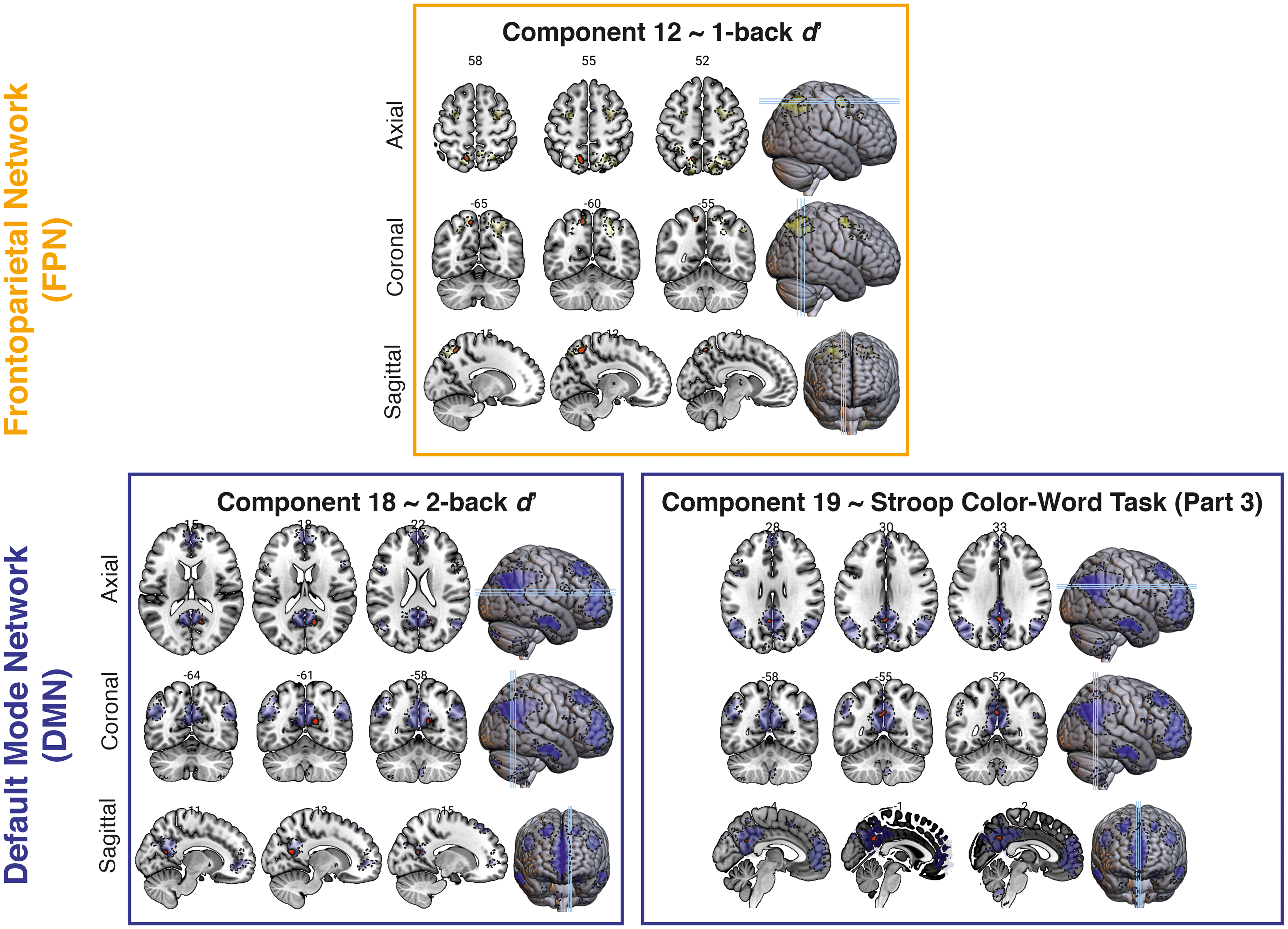
Relationship between resting-state connectivity and cognitive performance. Upper panel: Correlation between the frontoparietal network (dashed outlines) connectivity and 1-back performance. Lower panels: Correlation of the default mode network (dashed outlines) connectivity with 2-back performance (left panel) and performance in the third part of the Stroop Color-Word test (right panel). The regions enclosed by solid outlines indicate significant clusters at p < 0.001 with cluster thresholding (cluster-corrected p < 0.05). Images are shown in the neurological convention: the left hemisphere is shown in the left side of the image.
Furthermore, for the DMN, functional connectivity in IC number 18 (i.e., medial temporal DMN) was positively correlated with 2-back task performance (Z = 4.27, k = 28, MNI coordinate = 14 −60 18, AAL label = right Calcarine cortex). A positive correlation was also found between functional connectivity in IC number 19 (i.e., posterior DMN) and corrected SCWT-3 scores (Z = 3.51, k = 30, MNI coordinate = 0 −52 34, AAL label: left Posterior Cingulate Cortex). In the exploratory analysis, the correlation between the time courses of these two ICs of the DMN did not survive Bonferroni correction (t(51) = 2.27, puncorrected = 0.03, pBonferroni = 0.41), although both ICs are significantly correlated with the ICs belonging to the FPN (all puncorrected < 0.001, highest pBonferroni = 0.001; see Supplemental Analysis). Meanwhile, no significant correlations were found between any cognitive performance and functional connectivity within the SN component.
Hippocampal volume
In line with previous findings, 42 the current study found that MMSE scores were significantly associated with the total GM volume in both the left (ρ = 0.33, puncorrected = 0.01, pBonferroni = 0.03) and the right hippocampus (ρ = 0.37, puncorrected = 0.01, pBonferroni = 0.01).
Discussion
This study explored the cognitive correlates of cortical thickness, WM volume and resting-state functional connectivity in individuals with MCI. Of all the included cognitive measures, only n-back task performance was found to be associated with all three brain measures (i.e., cortical thickness, WM volume, and resting-state connectivity). SCWT performance was related to only the resting-state connectivity within the DMN. Meanwhile, performance in other cognitive tests (i.e., TMT and WAIS Digit Span tasks) was not found to be correlated with any of the brain measures.
The n-back task provides a valid measure of working memory updating 48 and poorer performance has been observed in aging adults. 49 Working memory involves many brain regions, including Broca's area, Wernicke's area, and the parietal lobe.50–52 In this study, better performance in both 1-back and 2-back task was correlated with higher cortical thickness in the left middle temporal gyrus (BA 22), which corresponds to the core Wernicke's area. 53 Known for its role in language, this area is also important for phonological analysis of verbal working memory. 54 Previous studies have rather consistently reported cortical thinning of the temporal area in people with MCI.55–57 In line with the current finding, Vaqué-Alcázar et al. 58 reported that older adults who experienced working memory decline, exhibited cortical thinning in the left middle and inferior temporal gyri. Such thinning has been shown to also predict subsequent cognitive impairment. A longitudinal study found that individuals who developed cognitive impairment within eight years had higher rates of cortical thinning. 59 As cortical thinning and cognitive decline are expected in old age,60,61 the current evidence implies that higher cortical thickness is important for performance in the n-back tasks.
Also, the aforementioned perisylvian regions are connected by the AF, from the frontal cortex to the lateral temporal cortex, through the inferior parietal lobule (IPL or the Geschwind's territory).16,62 More specifically, the anterior segment of the AF bridges the Broca's area and the Geschwind's territory.47,63 There has been evidence of elevated functional activity during a verbal working memory task but reduced activation during a non-verbal task in the Broca's area among older adults when compared to younger counterparts. 64 Older adults also show lower gray matter volumetric asymmetry of this region. 65 Moreover, lesions in the Geschwind's territory have been linked to impaired working memory, particularly in the phonological storage aspect. 66 Studies also showed gradient reductions of cortical thickness 67 and its asymmetry 68 in this area in people with MCI and AD. The current findings showed that performance in the 2-back task was associated with WM volume in the left anterior AF. This suggests that the less WM volume within this bundle, the worse the performance in verbal n-back for individuals with MCI.
Furthermore, the current study also found correlations between n-back performance and resting-state connectivity within the FPN and the DMN. This expands a previous finding of a reduced brain activation in the lateral prefrontal cortex (a region involved in the FPN) and reduced deactivation of insula, lingual gyrus, and medial temporal lobe in MCI patients during n-back tasks, despite comparable performance to that of healthy controls. 69 Similarly, Zanchi et al. 70 reported decreased FPN activation during an n-back task in people with MCI as compared to the control sample. Interestingly, they also noted that FPN activation was also lower in control participants who later developed subtle cognitive deficits when compared to those who remained cognitively healthy. A similar finding came from a study using resting-state fMRI which demonstrated reduced connectivity between the dorsolateral prefrontal cortex (DLPFC) and the IPL in MCI patients, while the connectivity between the DLPFC of both hemispheres heightened. 71 They interpreted these findings as an ipsilateral disconnection within the FPN, accompanied by a compensatory mechanism. Consistent with this, Sambataro et al. 72 reported a compensatory higher DMN connectivity in older adults, along with reduced FPN connectivity during a memory task. Together, these studies suggest that internetwork functional connectivity, both during tasks or at rest, is important to counteract diminished intranetwork activity.
In this study, worse 1-back task performance was associated with lower intrinsic connectivity within the IC number 12 which represented the right FPN, but the significant cluster was found on the left hemisphere. In addition, as suggested by the further exploratory analysis, a significant positive correlation was observed between time-courses of the left (IC number 12) and right (IC number 14) FPNs. This suggests the importance of interhemispheric connectivity for working memory performance in older adults with MCI. Meanwhile, lower 2-back performance was correlated with lower resting-state connectivity within IC number 18 (i.e., medial temporal part) of the DMN. In healthy young adults, DMN connectivity has been previously shown to be related to 3-back performance, suggesting the role of attentional resource in a highly demanding executive task. 73 Similarly, in older adults with and without cognitive impairment, the link between working memory and DMN intrinsic connectivity has also been demonstrated.74,75
Moreover, DMN connectivity was also associated with SCWT performance in the current study. This implies that DMN is also important for conflict monitoring. However, prior research has yielded mixed results, with some studies reporting a significant relationship between SCWT performance and DMN connectivity (e.g., Duchek et al. 76 ), while others did not.75,77 This could be due to the use of different methods. Additionally, in the current study, not all ICs identified as part of the DMN showed correlations with SCWT performance. This possibly suggests that conflict monitoring is only correlated with a certain subnetwork of the DMN. Andrews-Hanna et al. 78 propose that the DMN comprises a core and two distinct subsystems, with these two subsystems having been documented to dissociate functionally. 79 More research is needed to confirm whether only some parts of the DMN are crucial for executive functions.
Limitations and future directions
There are some limitations in the current study. Firstly, the research question of this study essentially pertained to how severity of cognitive impairment is associated with brain measures and thus did not call for healthy control group. However, it is also relevant to evaluate how the brain-cognition associations might differ between cognitively healthy and impaired older individuals. For instance, Setiadi et al. 80 found that, despite no between-group differences in white matter microstructures, apathy was associated with diffusion tensor imaging (DTI) parameters only in MCI patients. Similarly, Kaskikallio et al. 81 found that white matter hyperintensity in the left temporal lobe was linked to processing speed impairment in MCI/AD patients, but not in healthy participants. This kind of analysis could offer insight into brain measures that may play an important role in compensatory mechanisms.
Additionally, although some previous studies used large datasets such as those from the Alzheimer's Disease Neuroimaging Initiative (ADNI, e.g., Edmonds et al. 55 ), the sample size of the current study was moderately large and comparable to that of other single-center studies (e.g., Seo et al. 56 and Migo et al. 69 ). Nonetheless, our sample is not representative of the aMCI population at large due to the overrepresentation of male participants. Consequently, the generalizability of the findings may be limited. While a metanalysis indicated that prevalence and incidence rates of aMCI are comparable between sexes, 82 Berezuk et al. 83 demonstrated that risk factors of subsequent dementia may differ between men and women with aMCI.
It is also important to note that the current study used the 15-Word Test, the Dutch version of the Rey Auditory Verbal Learning Test to characterize and establish a criterion for aMCI. On average, our sample showed a higher delayed recall ability than immediate recall ability. Previous studies using similar tests have demonstrated that delayed recall performance is a good predictor of conversion to AD dementia.84,85 Meanwhile, Lim et al. 86 reported that AD is more closely associated with learning deficits rather than with memory impairment per se. Although distinguishing between learning and memory processes is a relevant consideration, it falls beyond the scope of the present study. Notably, the COGMAX study also collected data using the extended FNAME task, 87 an associative memory assessment designed to separate learning from retrieval processes. These data offer a valuable opportunity to explore this distinction in future research.
Furthermore, this study investigated the importance of WM volume in aMCI. While WM properties are relevant to study in relation to cognition, T1-weighted imaging such as in the current study cannot discriminate between different WM tracts. 88 This is possible using DTI which can provide investigation of microstructure properties of specific WM bundles. Hence, the current finding about WM volume within the left AF should be interpreted carefully. Nonetheless, a recent DTI study provided converging evidence pertaining to the association between AF volume and working memory performance. 89 Additionally, future studies could also include artificial intelligence-based synthesis of multimodal neuroimaging data, in order to use them for diagnostic purposes. Following replication of our findings, the integration of T1-weighted and resting-state functional imaging may support clinical applications in characterizing disease severity.
Lastly, the current study did not measure all cognitive domains and included mostly measures of executive functions. Future studies focusing on other domains such as attention and memory might be useful to provide a more comprehensive picture. Moreover, even within the investigated domains in this study, findings seem to be task-specific. The associations with working memory were only observed for the n-back task. The Backward DS was not found to be correlated with cortical thickness, WM volume, or resting-state connectivity. Indeed, n-back performance has been reported to be affected in individuals with orbitofrontal damage, but performance in DS tasks is not. 90 However, brain regions associated with both working memory tasks seem to highly overlap.91,92 Additionally, while n-back performance has been shown to distinguish people with MCI from healthy control, 93 conventional DS scores lacks the sensitivity to achieve the same. 93 Future research might want to consider using more complex DS-derived metrics, which have demonstrated greater sensitivity to MCI. 93
Conclusion
The current study explored how the severity of cognitive decline in MCI is associated with cortical thickness, WM volume and resting-state connectivity, expanding previous evidence from studies comparing MCI and healthy controls. The findings suggest that diminished cognitive abilities such as working memory and conflict monitoring in individuals with MCI are reflected in the structural and functional brain characteristics, notably within the perisylvian region. Lower cortical thickness in the Wernicke's area was linked to worse n-back task performance. Additionally, while WM volume within the anterior segment of the AF and DMN resting-state connectivity are relevant in higher working memory demand (i.e., 2-back task), the intrinsic connectivity of the FPN is implicated in the lower demand (i.e., 1-back task). Moreover, evidence of an association between DMN intrinsic connectivity and conflict monitoring was also found. Associations with the FPN and the DMN highlight the potential contribution of intrinsic brain connectivity to cognitive functioning, even during the resting state. While these results provide initial insight into possible neural correlates of cognitive performance, further research is needed to confirm and specify these associations.
Footnotes
Acknowledgements
We are much obliged to all volunteers and participants who took part in the COGMAX project. We also thank all research assistants and interns who contributed to the project during the conception of the project, participant recruitment, and data collection.
ORCID iDs
Ethical considerations
The COGMAX project (ABR number NL64677.042.18) was in accordance with the Declaration of Helsinki and the protocol was approved by the medical ethical board of the UMCG (METc number METc2018.089; 13 November 2018).
Consent to participate
Written consent to participant in the study was obtained from all participants.
Consent for publication
Not applicable
Author contribution(s)
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The COGMAX project was funded by ZonMW Memorabel grant (Project number: 733050836).
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
The data used in this report will be made available upon request after the core COGMAX analyses are finalized.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
