Abstract
Background
Treatment resistant hypertension (TRH) is associated with increased risk of cognitive decline, which may be reduced by healthy lifestyle changes.
Objective
To examine the effects of a comprehensive, rehabilitation-based lifestyle hprogram on cognitive function during a one-year follow-up of participants from the TRIUMPH clinical trial.
Methods
Among the 140 TRIUMPH participants originally randomized, 91 (65%) were available for one-year assessments prior to the COVID-19 lockdown. Participants were originally randomized to a
Results
Participants included 91 individuals (mean age = 63.6 [SD = 8.6]), evenly distributed in biological sex and race/ethnicity, and tended to be college-educated. The C-LIFE group had more preserved cognitive functioning compared to SEPA (C-LIFE: z = −0.26 [−0.40, −0.12] versus SEPA: −0.60 [−0.81, −0.39]; d = 0.44, p = 0.008), with reduced PSQI sleep symptoms associating with more preserved cognitive function (B = −0.18, p = 0.050 per 3-points). Treatment did not improve fNIRS markers, although changes in weight and physical activity associated with fNIRS outcomes.
Conclusions
Lifestyle modification may help preserve cognitive functioning among individuals with resistant hypertension.
Keywords
Introduction
Hypertension is common and increasingly prevalent among United States adults.1–3 Recent data suggest that as many as 75% of older adults are treated with an antihypertensive medication and this is projected to increase in parallel with rising obesity levels. 3 Among hypertensive adults, those with resistant hypertension, characterized by taking at least three antihypertensives without an optimal blood pressure reduction, have been suggested to have the highest risk of stroke, cognitive decline, and Alzheimer's disease and related dementias (ADRD). 4 Strategies to improve both blood pressure and cognition among individuals with treatment-resistant hypertension (TRH) are therefore critical to mitigate risk of ADRD in TRH.
Over the past decade, a growing number of clinical trials have demonstrated that reductions in blood pressure associates with decreased risk of cognitive impairment and ADRD.1,2,5 Targeted blood pressure reductions using pharmacotherapy have been shown to reduce the risk of mild cognitive impairment (MCI) 6 and a large body of evidence from multi-component randomized trials suggests that adoption of healthier lifestyle habits helps preserve cognition in adults at risk for ADRD.7,8 Lifestyle modification may help lower ADRD risk, in part, through reducing hypertension and improving vascular function, tau hyperphosphorylation, and beta amyloid accumulation, 9 even among individuals without measurable cognitive decline. 10 The durability of these effects, however, remains in question, as many individuals are unable to sustain healthy lifestyle habits after the structure and support from the intervention team ends.11,12 Follow-up analyses from randomized trials are therefore needed to better understand the longer-term effects of lifestyle modification on cognitive function. In addition, understanding how different aspects of lifestyle change differentially influence sustained cognitive function could have important implications for prioritizing treatment targets, such as choosing to target physical activity instead of weight loss. Finally, studies incorporating additional, subclinical markers of cerebral functions are needed to enhance the understanding of inconsistent cognitive changes, as measures of cerebral efficiency may provide insight into subtle brain changes that may not necessarily be reflected on clinical neuropsychological measures.13,14
The specific lifestyle modification components that correlate with improved cognitive outcomes remain to be adequately characterized from clinical trial follow-up assessments. Although improvements in aerobic fitness, 15 weight loss,11,16–18 and reduced blood pressure 19 have all been suggested as key correlates, the relationships between changes in these factors and cognitive outcomes has been inconsistent. 20 For example, although most aerobic trial data suggest improvements in cognitive outcomes, 8 most data attempting to link changes in fitness to cognitive improvements have been equivocal. 20 In contrast, intentional weight loss appears to associate with improved cognition, 16 regardless of whether it was achieved using physical activity, weight loss surgery, or through intensive caloric restriction. 18 Recent data have extended the examination biobehavioral mechanisms to include improvements in sleep quality, which commonly parallel improvements in physical activity, diet, and weight loss.21–23 Poor sleep quality is common among individuals with hypertension and robustly associated with worse cognitive functioning, even among individuals who do not have obstructive sleep apnea. 24 In order to examine the longer-term benefit of lifestyle modification on cognitive functioning and associated biobehavioral correlates, we examined longitudinal changes in cognition and associations with changes in physical activity, dietary quality, weight maintenance, sleep quality, and cerebrovascular functioning during a one-year follow-up from the TRIUMPH trial. The present analyses focus on cognitive and cerebrovascular outcomes, whereas comprehensive findings from the overall lifestyle intervention will be reported separately (Hinderliter et al., unpublished results). For cognitive and cerebrovascular outcomes, we hypothesized that the C-LIFE treatment group would show better cognitive function and greater cerebrovascular reactivity compared to SEPA at one-year and that these effects would be associated with higher levels of physical activity and DASH diet scores, better weigh maintenance, and better sleep quality.
Methods
Sample
The protocol was approved by the Duke University School of Medicine institutional review board following the Helsinki protocol and written informed consent was obtained from all participants. Adults with overweight / obesity and resistant hypertension were eligible to participate.4,25 Treatment resistant hypertension (TRH) was defined as inadequate blood pressure response despite being optimally treated with at least three antihypertensives (including a diuretic). Specifically, as we previously reported,4,25,26 TRH was defined as treatment for at least 6 weeks with 3 or more antihypertensive medications of different classes, including a diuretic, with clinic SBP ≥130 mm Hg or DBP ≥80 mm Hg, or the need for 4 or more drugs to achieve SBP ≤130 mm Hg and DBP ≤80 mm Hg, with SBP ≥120 mm Hg, were eligible. Additional inclusion criteria included body mass index ≥25 kg/m2, lack of regular moderate or vigorous physical activity, and age 35 to 80 years. Exclusion criteria included known secondary hypertension, estimated glomerular filtration rate <40 mL/min/1.73 m2, moderate-severe ischemic heart disease, diabetes requiring insulin, and major psychiatric disorder or substance dependence, including alcohol consumption >14 drinks/week. Participants were enrolled beginning in June 2015 and ending in October 2019. Of note, because data for the present study were obtained from two separate grants with different hypotheses, the present analyses will focus on cognitive and cerebrovascular outcomes (HL130237), whereas complete data on blood pressure, physical activity, nutrition, and weight outcomes (HL122836) will be reported separately (Hinderliter et al., unpublished results).
Randomization
Participants were randomized 2:1 to a 4-month, cardiac rehabilitation-based rehabilitation program (C-LIFE) or to a standardized education and physician advice condition (SEPA). This randomization scheme was selected to leverage treatment heterogeneity in the C-LIFE condition, providing a more powerful test of efficacy. 4 Assessments of cognitive function, vascular and cerebrovascular functioning, physical activity, blood pressure, and sleep quality were collected at baseline, 4 months, and one-year after randomization.
Follow-up assessments
Following the 4-month trial, we assessed lifestyle habits, blood pressure, and cognitive function during a one-year naturalistic follow-up. Following their 4-month assessment, participants received no further intervention but were periodically contacted to maintain study contact (Figure 1). During the active intervention period, four individuals (all in SEPA) dropped out, possibly due to their dissatisfaction with treatment group assignment. The remainder of individuals with missing data were impacted by the initial COVID-19 lockdown and associated contact precautions. Individuals lost to follow-up had higher BMI and worse insulin sensitivity levels (as indicated by higher HOMA-IR [Homeostatic Model Assessment for Insulin Resistance] levels), as well as lower levels of intima medial thickness and kidney disease, but otherwise did not differ from completers. Specifically, participants lost to follow-up had higher BMI levels (37.3 [SD = 5.7] versus 35.3 [SD = 5.6]), higher HOMA-IR levels (5.5 [SD = 4.0] versus 4.4 [SD = 2.2]), lower IMT levels (0.73 [SD = 0.14] versus 0.78 [SD = 0.16]), and lower rates of kidney disease (14% versus 30%) compared to participants who were available for follow-up assessments.
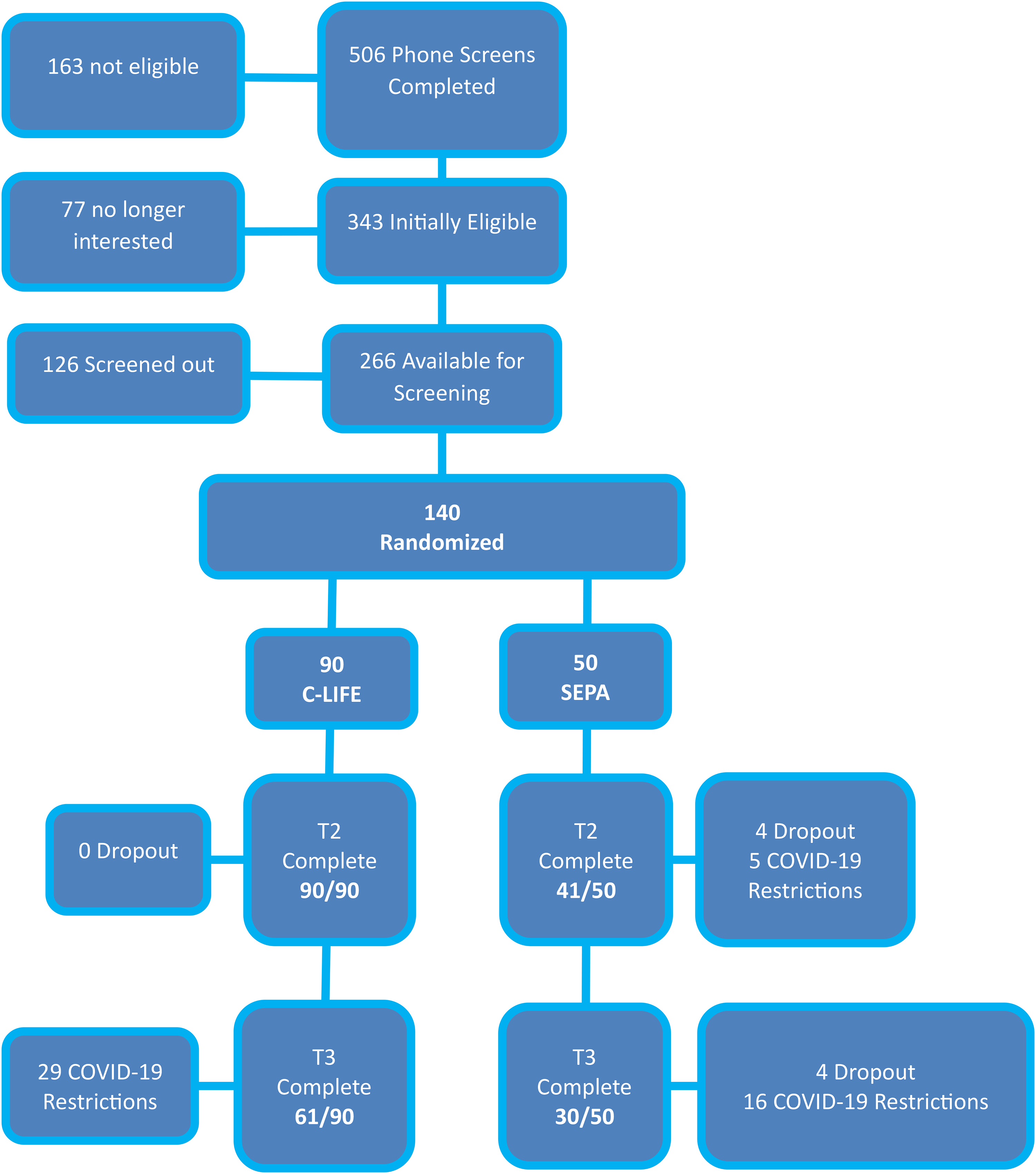
Consort chart showing the flow of participants through screening, randomization, post-treatment assessments and one-year follow-up. Participants were randomized to either a
Cognitive performance
We adopted a battery similar to that proposed by the Neuropsychological Working Group for vascular cognitive disorders. 27 As detailed below, we used reliable change indices to quantify cognitive changes using regression-based techniques, the sources of which are cited for individual subtests. Assessments included tests of Executive Function/Learning (Trail Making Test Part B [lower scores = better performance], 28 Stroop Color-Word Section, 29 Animal Naming, 28 COWA, 30 CVLT-II Discrimination Index, 31 BVMT-R Learning), 30 Processing Speed (Trail Making Test Part A [lower scores = better performance], 28 Stroop Word Section, Stroop Color Section, Digit Symbol Substitution Test, Ruff 2&7 Test), 32 and Memory (CVLT-II Total Learning [sum of trials 1–5], CVLT-II Immediate Recall, 31 and CVLT Delayed Recall).23,26,33 On the CVLT-II, we incorporated several indices that measure efficiency of encoding information, including semantic clustering and learning slope efficiency. Semantic clustering represents the degree to which participants recognized and grouped words into categories to enhance their ability to learn and recall words on the CVLT-II. 34 Total learning slope efficiency represents how quickly and efficiently individuals were able to learn CVLT-II word items, with higher score suggesting more effective learning over trials. 35
As previously reported, cognitive testing followed a fixed order of administration and utilized alternative forms for all tests where available (e.g., CVLT-II, BVMT-R, COWA, Trail Making Test, Digit Symbol Substitution Test), utilizing an alternating schedule of administration between assessment time periods (e.g., A-B-A and B-A-B). Assessments were performed by a data technician who was not involved in the participants’ treatment regimen and participants were asked not to disclose their treatment assignment during assessments.
Cerebrovascular functioning
We utilized functional near infrared spectroscopy (fNIRS) to non-invasively cerebrovascular reserve and vasomotor function in parallel with cognitive function measures. When assessed by fNIRS, the increase in cerebral blood flow in the surface layers of the cortex during localized neural activity is seen as an increase in the total concentration of hemoglobin (total-Hb) and comparative decrease in deoxy-Hb, with both variables corresponding strongly with the functional MRI BOLD signal. 36
Cerebrovascular reserve (CVR)
CVR was measured as the change in tissue oxygen saturation from baseline during a standardized breath holding index (BHI).37,38 The BHI has been used extensively as a marker of CVR, has been shown to decrease with age, 39 is impaired among individuals with cerebrovascular disease40,41 and cognitive impairment, 42 and is predictive of subsequent stroke. 43 Consistent with standard methodology, changes in TOS during BH was measured using two, near-infrared optodes of the cerebral oximetry, each consisting of two light-source fibers and one light-collecting fiber. Sensors was placed in two symmetrical points on the forehead in symmetric areas of the frontal lobe of the two hemispheres (2 cm beside the midline and about 3 cm above the supraorbital ridge) and fixed with a headband. CVR was quantified as changes in TOS from baseline to BHI, divided by baseline TOS and total BHI time (BHI = [TOSBH − TOSBAS] × 100/TOSBAS/DBH). 44 Voluntary breath holding was used to induce hypercapnia, with a duration ranging from 20 to 30 s. Assessments lasting less than 20 s were repeated.
Cerebral hemodynamic changes
We also assessed changes in oxygenated hemoglobin (HbO2) while completing cognitive challenges for selected subset of three cognitive tasks amenable to efficient assessment in an ambulatory setting. We utilized cognitive assessments known to elicit cortical activation and that have been shown to have prognostic benefit in older adults at risk for ADRD. 45 The cognitive challenge tasks therefore included semantic fluency (articles of clothing, items in a grocery store, and fruits), mental arithmetic (subtraction using serial sevens with varying start numbers; max score = 12), and phonemic fluency (letters B, F, and W; selected in order not to conflict with the COWA administration in our main test battery). As recently reviewed, 46 contemporary analyses of fNIRS data during cognitive assessments has primary utilized measures of changes in HBO2 while performing cognitive assessments, which is thought to reflect hemodynamic efficiency. 47 Cognitive tasks were administered in a fixed order (1: semantic fluency, 2: mental arithmetic, 3: phonemic fluency), counterbalanced across participants in order to minimize practice effects. Each task was conducted for one-minute, with changes in oxygenated hemoglobin (maximum – minimum HbO2 levels during one-minute assessment) used as the hemodynamic measure of interest.
Physical activity levels
Physical activity levels were assessed using 24-h actigraphy with an accelerometer (ActiGraph GT9X Link Monitor, Pensacola, FL). Wrist-worn actigraphy monitors were worn for seven consecutive days both before and after the intervention in order to assess measures of sleep quality and markers of weekly physical activity. We focused on total step count as the metric of interest, which was found to have been improved in the C-LIFE group relative to SEPA controls, as previously published.
Sleep quality
Sleep quality was assessed using Pittsburgh Sleep Quality Inventory (PSQI). The PSQI provides a subjective measure of sleep quality and is composed of nineteen individual items that generate seven “component” scores (i.e., subjective sleep quality, sleep latency, sleep duration, habitual sleep efficiency, sleep disturbances, use of sleeping medication, and daytime dysfunction).23,48
Blood pressure
Both clinic and ambulatory blood pressures (BPs) were obtained. Clinic-measured BP was determined according to JNC-7 guidelines. Ambulatory blood pressure monitoring (ABPM) was also assessed during a typical day, in which participants wore an Oscar 2 (Suntech Medical Inc, Raleigh, North Carolina) ABPM. 49 The Oscar 2 was programmed to record BP measurements 3 times per hour during waking hours and 2 times per hour during sleep, with included assessments requiring at least 70% of planned readings. 50 Total number of blood pressure medications and BP medication burden were also obtained using total count and mean Daily Defined Dose (DDD) levels. 51 The DDD was developed by the World Health Organization to systematize the quantification of medication dosing across potentially heterogeneous treatment regimens.
Nutritional and weight assessment
Dietary and nutritional content was obtained both using a retrospective food frequency questionnaire (FFQ) requiring participants to recall typical consumption during a 4-week period 52 and a 2-day (weekend/weekday) food diary. 53 The FFQ was analyzed by NutritionQuest (Berkeley, CA), and the diary data were analyzed using the Automated Self-Administered 24-h (ASA 24®) Dietary Assessment Tool (https://epi.grants.cancer.gov/asa24/).
Data analysis
Analyses were conducted in SAS 9.4 (Cary, NC) and R 4.5.0 (https://cran.r-project.org/). In order to provide a parsimonious analytical approach across cognitive outcomes, we elected to use a global score approach, which combined reliable change indices for all subtests into a unified z-score outcome. This approach parallels the “gatekeeper” methodology we utilized in the primary trial analyses for cognitive and biomarker outcomes,25,26 and is an approach we have used extensively in other clinical trials.18,54–56 Our analyses followed an intention-to-treat approach, including all 140 participants regardless of their follow-up status. Following our primary analysis, we conducted explanatory, secondary analyses of individual changes in each subtest with accompanying effect sizes to provide a characterization of where group differences were arising, if observed.57–60 This type of explanatory analytical step has been advocated as appropriate within the “gatekeeper” framework to account for multiplicity in clinical trials settings because of its control for type-I error. However, this approach also has the limitation that small-to-moderate patterns of change, if consistently observed, will yield a significant composite outcome despite relatively small changes on individual variables when examined in isolation.
To examine longitudinal associations between putative mechanistic markers and cognitive outcomes, we conducted a parallel regression in which changes in each physical activity, weight, DASH diet scores, and sleep quality were modeled together. Our primary analyses were therefore conducted using PROC REG and PROC GLM. Within our modeling approach, the one-year global RCI z-score served as the outcome variable with age, education (years), sex, baseline creatinine, baseline stroke risk (using the Framingham Stroke Risk Profile), and baseline cognitive function (mean rank across all cognitive subtests) as adjustment variables, with group assignment as the variable of interest. Similarly, to examine changes in task-based hemodynamic efficiency, we created a mean-rank, global score across all three cognitive challenge tasks, residualized on cognitive performance levels.
Changes in physical activity, weight, DASH diet quality, and sleep quality were conducted using a parallel approach, with baseline levels of the respective outcome as an additional control variable. Missing data was accounted for using multiple imputation (PROC MI within SAS 9.4), in which 200 imputations were utilized. Model diagnostics were assessed through visual inspection of residuals in comparison with observed values, as well as assessing imputations for potential influential outliers. Within our imputation models, we incorporated 1) all variables that were subsequently included in our full models (e.g., cognitive functioning, age, education, sex, creatinine, Framingham stroke risk profile, and treatment group), 2) variables predictive of missingness within our dataset (e.g., intima medial thickness, body mass index, additional markers of kidney function [e.g., eGFR], HOMA-IR), and 3) variables assessing putative mechanistic factors, such as sleep quality and blood pressure.
Follow-up, explanatory analyses of individual cognitive changes were conducted using repeated measures, linear mixed effect models through PROC MIXED. Within these models, cognitive function for individual subtests at 4-months and one-year served as the repeated measures outcome, controlling for the same adjustment variables as in our primary analyses above. Our a priori hypotheses were that C-LIFE would improve cognitive functioning across domains in comparison with controls at one-year. We further hypothesized that higher levels of physical activity, DASH diet scores, ambulatory systolic blood pressure, weight loss, and sleep quality would associate with better cognitive and cerebrovascular outcomes. Consistent with contemporary modeling approaches, we examined associations between putative mechanistic markers and outcomes regardless of whether a main effect was observed.61,62 Model assumptions regarding independence, additivity, and linearity of residuals were evaluated and found to be acceptable prior to analysis.
Results
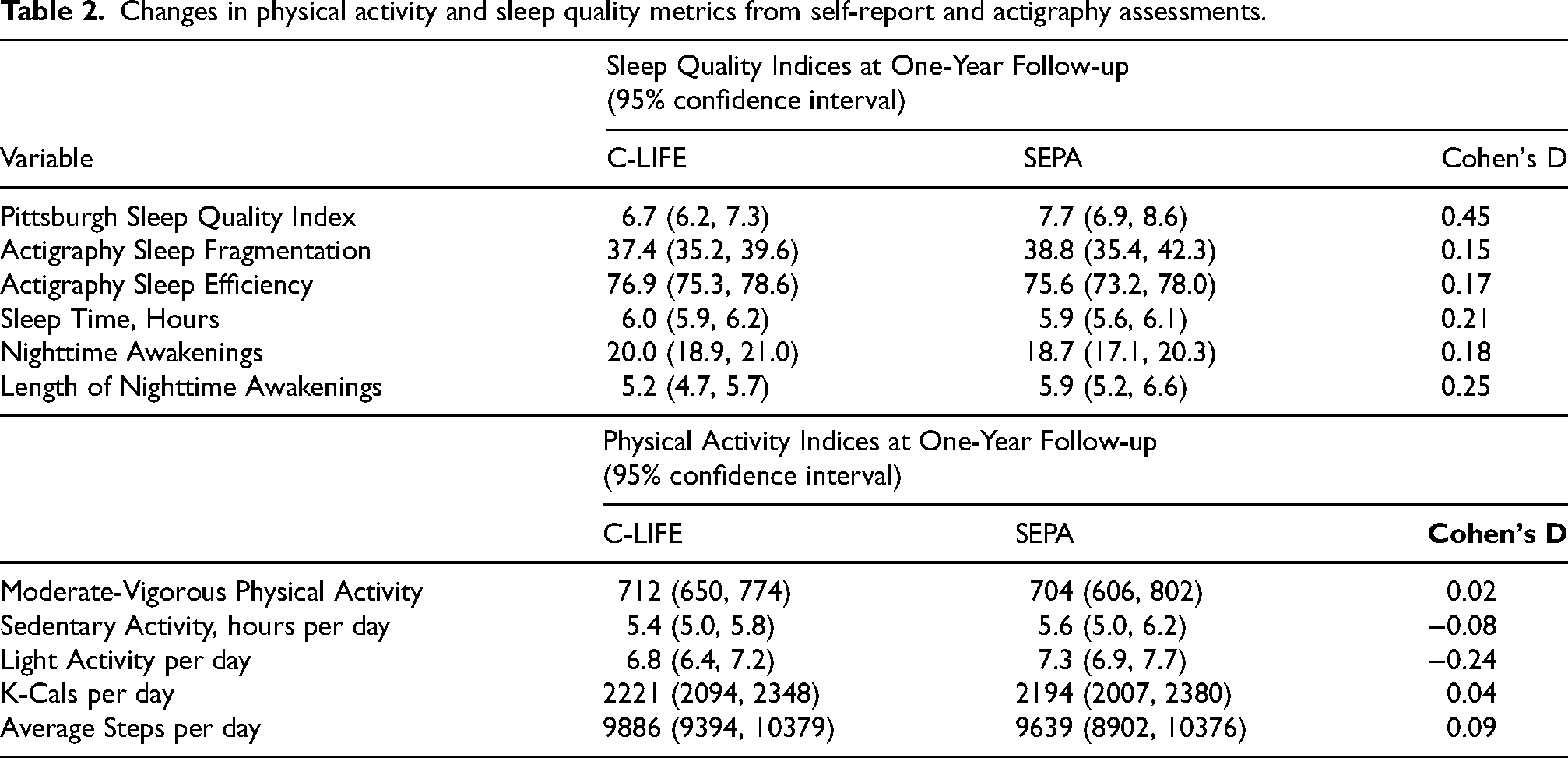
Background and clinical characteristics from baseline assessments are presented in Table 1. From the original 140 participants, 91 were available for inclusion during the one-year follow-up (Figure 1). Analyses of one-year lifestyle habits will be presented in detail elsewhere (Hinderliter et al., unpublished results) and are therefore described only briefly here. Results indicated that the C-LIFE group exhibited lower ambulatory SBP (133 mm Hg [129, 137] versus 140 mm Hg [135, 144], p = 0.044) and ambulatory DBP (71 mm Hg [69, 73] versus 74 mm Hg [72, 77], p = 0.046) compared with SEPA). In terms of other lifestyle habits, we found no group differences in dietary quality (DASH diet scores: C-LIFE = 3.8 [3.5, 4.0] versus SEPA = 3.5 [3.0, 3.9]. p = 0.171), objectively assessed physical activity (Daily Actigraphy Steps: C-LIFE = 9844 [9360, 1–320] versus SEPA = 9667 [8949, 10385], p = 0.653), or weight (C-LIFE: 219.6 [217, 222] lbs versus SEPA: 220.7 [217, 225] lbs, p = 0.282). In exploratory analyses of sleep quality changes on the PSQI, we found that the C-LIFE group continued to demonstrate improved sleep quality compared with SEPA (6.7 [6.2, 7.3] versus 7.7 [6.9, 8.6], d = 0.45, p = 0.049) (Table 2), corresponding to % changes from baseline of +5.6 [−3.7, 14.9] for SEPA versus −14.1 [−27.9, −0.4] in C-LIFE) (Supplemental Figure 1).
Background and demographic characteristics of the sample.
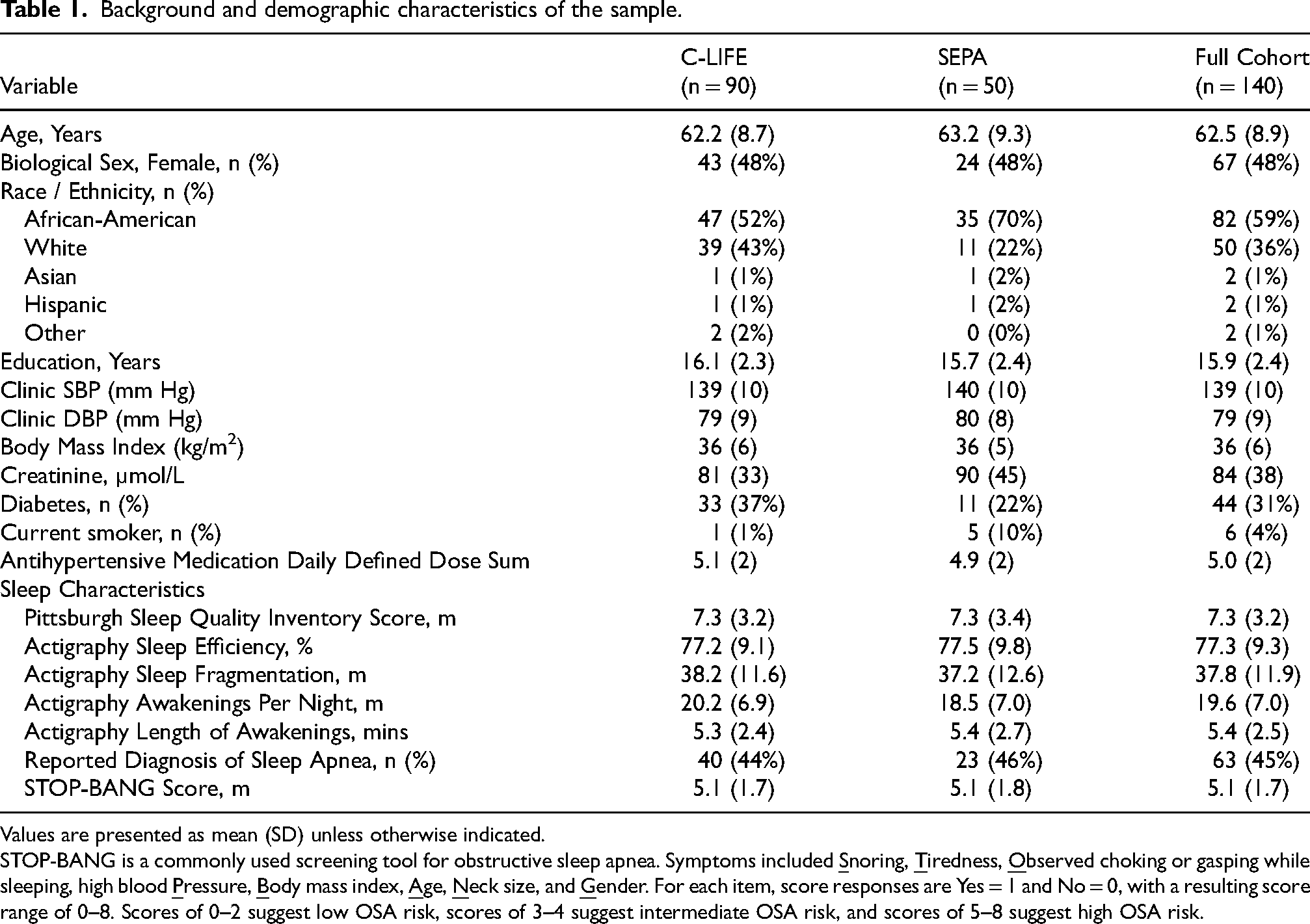
Values are presented as mean (SD) unless otherwise indicated.
STOP-BANG is a commonly used screening tool for obstructive sleep apnea. Symptoms included
Changes in physical activity and sleep quality metrics from self-report and actigraphy assessments.
One-year cognitive findings
Examination of cognitive changes from baseline to one-year follow-up were accomplished using reliable change indices. Across the cohort, most individuals showed small-to-moderate cognitive changes (mean global RCI = −0.40 [−0.52, −0.29], p < 0.001), with the largest changes in Psychomotor Speed (mean RCI = −0.63 [−0.89, −0.37], p < 0.001) and Executive Function/Learning (mean RCI = −0.50 [−0.61, −0.39], p < 0.001), without substantial Memory changes (mean RCI = 0.03 [−0.14, 0.21], p = 0.702). The majority of participants available for one-year assessments (73%) showed clinically meaningful declines on at least one cognitive subtest (i.e., < −1.96 RCI for subtest change from baseline to one-year), with 24% exhibiting one impairment, 18% exhibiting two impairments, and 31% impairments on ≥ three subtests. Examination of individual cognitive subtests revealed that the highest frequency of worsening of performance was observed on the Ruff 2&7 Test (17% and 18% for Speed and Accuracy, respectively), followed by the Stroop Color-Word Test (12%).
Examination of treatment group differences revealed main effect of treatment on our global RCI z-score, such that the C-LIFE group showed a lesser decline compared to SEPA (C-LIFE: z = −0.26 [−0.40, −0.12] versus SEPA: −0.60 [−0.81, −0.39]; d = 0.44, p = 0.008) (Figure 2). Examination of individual subtests (Table 3) demonstrated that the largest group differences were in Memory subtests, as observed on the CVLT-II Learning (d = 0.62) and CVLT-II Recall subtests (d = 0.36). Explanatory analyses of individual subtest changes in their raw form similarly revealed the largest group differences for CVLT-II Learning (C-LIFE = 49.1 [47.4, 50.9] versus SEPA = 43.8 [41.3, 46.3], p = 0.001), Immediate Recall (C-LIFE = 9.9 [9.3, 10.5] versus 8.8 [7.9, 9.7], p = 0.048), and Delayed Recall (C-LIFE = 10.4 [9.8, 11.0] versus 9.5 [8.7, 10.4], p = 0.112) (Supplemental Figure 2). Moreover, these improvements appeared to be paralleled by greater Learning Slope Efficiency, as evidenced by improvements in Semantic Clustering (C-LIFE = 1.2 [0.8, 1.5] versus SEPA = 0.5 [0, 0.9], p = 0.016) and Learning Slope (C-LIFE = 1.4 [1.3, 1.6] versus 1.2 [1.0, 1.4], p = 0.080). In contrast, we found no consistent improvements in measures of Visual Memory performance, such as the BVMT-R (BVMT-R Total Learning: d = 0.10, p = 0.272; BVMT-R Total Recall: d = 0.06, p = 0.650). Explanatory analyses of other cognitive outcomes revealed smaller and less consistent group differences in Executive Function / Learning and Psychomotor Speed subtests. For example, we observed modest changes on the Stroop Word subtest (d = 0.43, p = 0.002), Ruff 2&7 Test (d = 0.29, p = 0.039), and trends towards group differences on the Trail Making Test Part B (d = 0.35, p = 0.094) and Animal Naming (d = 0.39, p = 0.066) (Supplemental Figure 3).
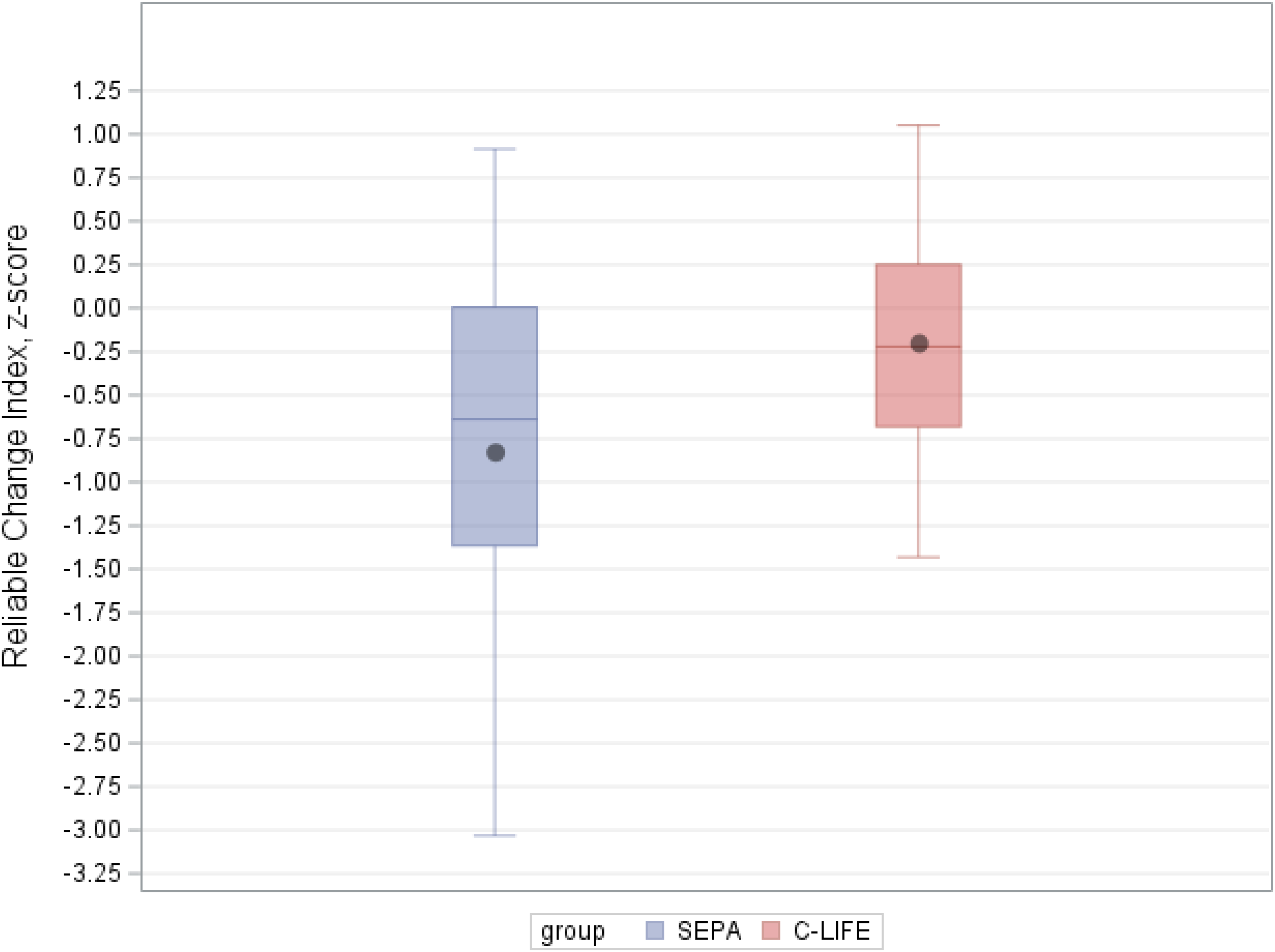
Treatment-related changes in our global, composite measure of cognitive change from reliable change indices. Participants in the
Changes in individual cognitive subtests using reliable change indices.
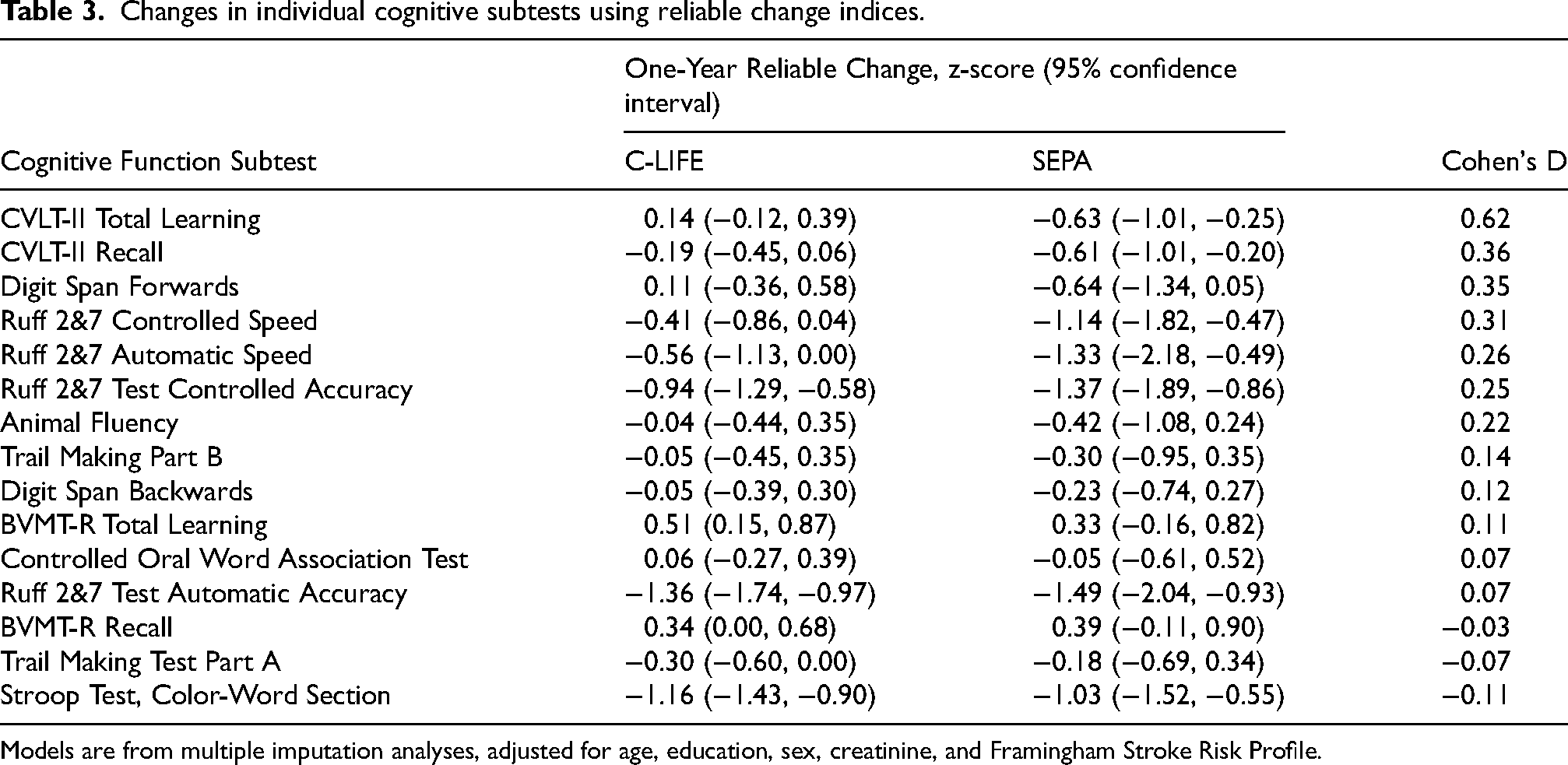
Models are from multiple imputation analyses, adjusted for age, education, sex, creatinine, and Framingham Stroke Risk Profile.
One-year cerebrovascular findings
Examination of cerebrovascular reactivity and task-related hemodynamic outcomes are shown in Table 4. As shown, we found no consistent group differences at one-year across any of our fNIRS outcomes, including changes in TOI (p = 0.489) and oxygenated hemoglobin (p = 0.964) during a breath-holding challenge. Similarly, analyses of changes in our global score of task-based oxygenated hemoglobin efficiency showed no group differences at one-year (B = −0.01 [−0.02, 0.01], p = 0.126).
Changes in cognitive and hemodynamic responses during cognitive challenge tasks.
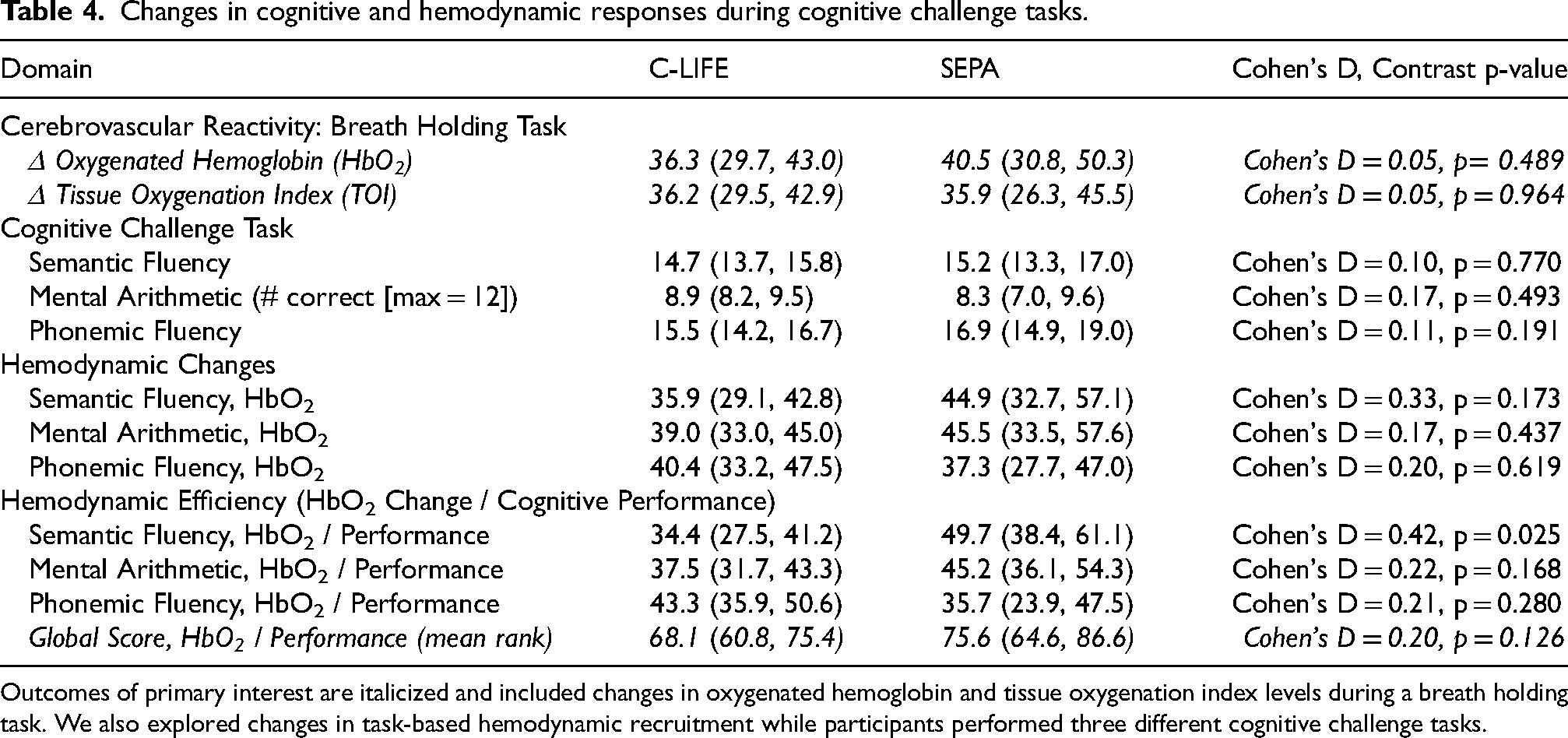
Outcomes of primary interest are italicized and included changes in oxygenated hemoglobin and tissue oxygenation index levels during a breath holding task. We also explored changes in task-based hemodynamic recruitment while participants performed three different cognitive challenge tasks.
Longitudinal associations with one-year cognitive function and cerebrovascular outcomes
Examination of correlates of one-year cognitive changes were also conducted. In an examination of changes in putative mechanistic markers and cognitive outcomes, we found that improvements in (reductions in) PSQI scores associated with global cognitive changes (B = −0.17 every 3-point PSQI reduction, p = 0.038) (Figure 3), whereas increased steps (B = −0.01 for every 2500 steps, p = 0.822), DASH diet scores (B = −0.04 for every 2-point change, p = 0.502), weight loss (B = 0.08 for every 15-lbs of weight loss, p = 0.270), and ambulatory SBP reductions (B = 0.01 for every 15-mmHg reduction, p = 0.939) were not associated with cognitive changes. Exploratory analysis of persistent treatment group effects demonstrated that treatment group differences remained significant in a final model incorporating the group contrast and putative mechanistic markers (B = 0.12, p = 0.005).
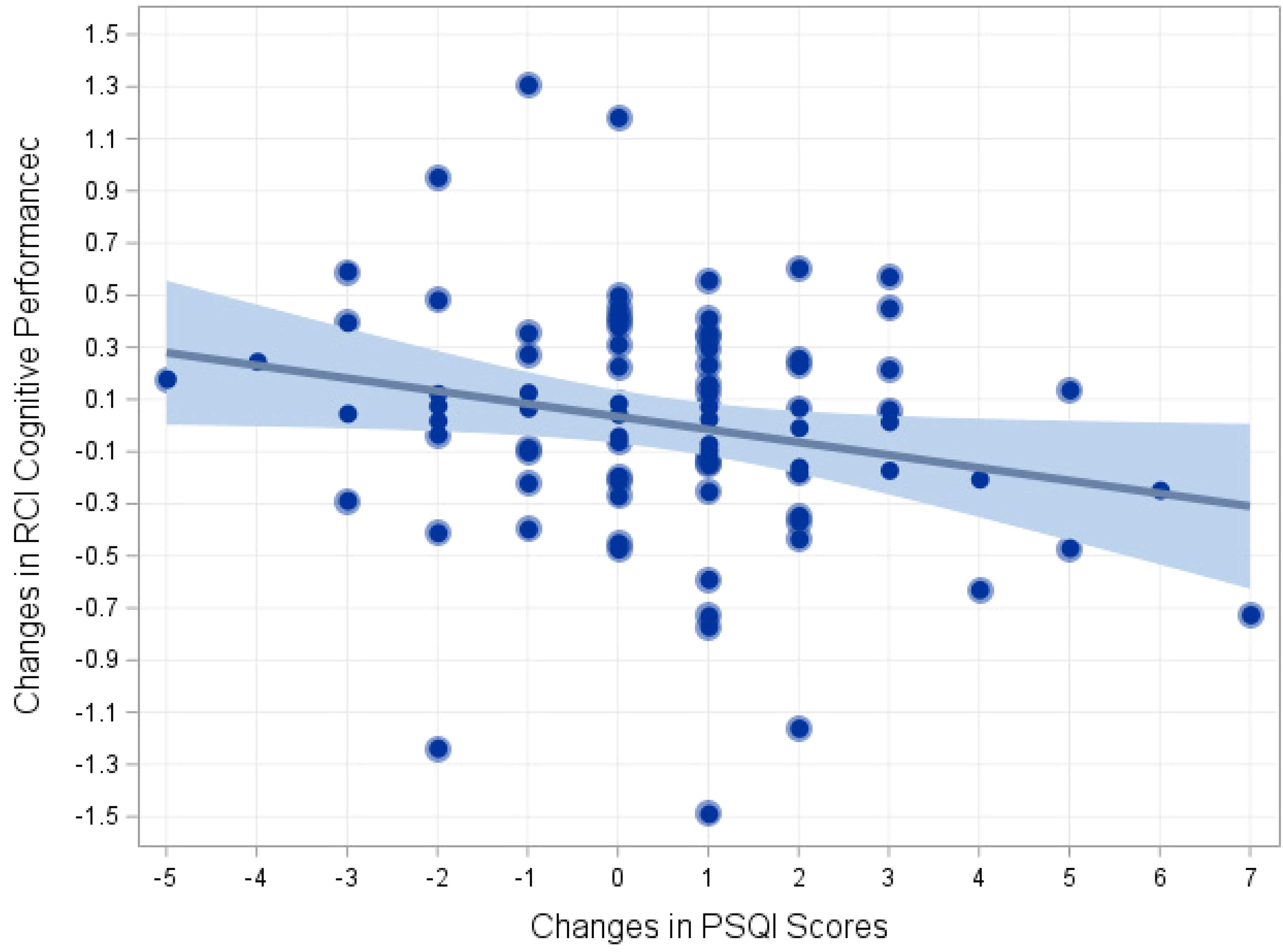
Changes in the Pittsburgh Sleep Quality Index and global cognitive scores from reliable change indices after adjusting age, education (years), sex, baseline creatinine, baseline stroke risk (using the Framingham Stroke Risk Profile), and baseline cognitive function. RCI: reliable change indices.
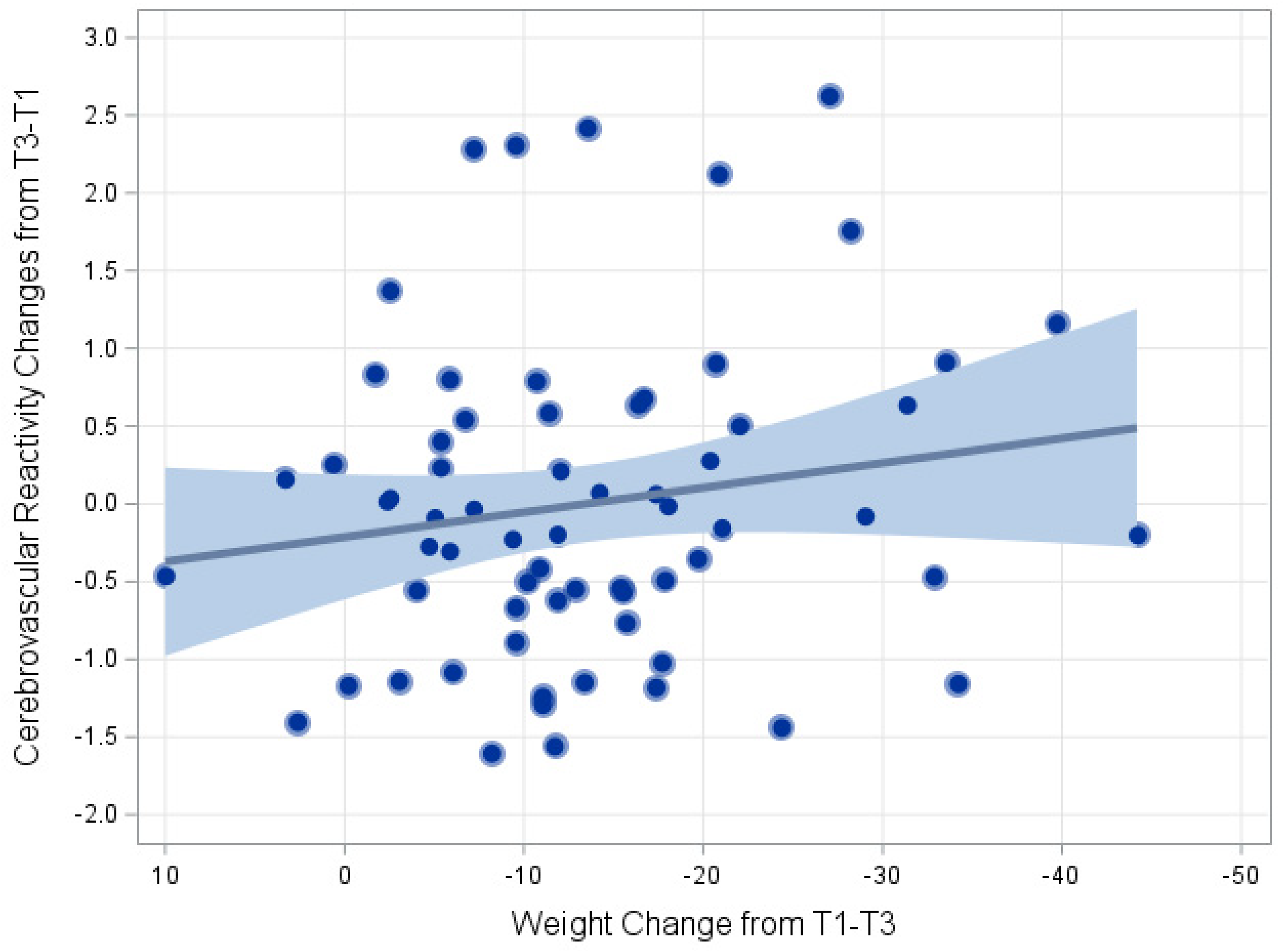
Changes in the weight from baseline to one-year follow-up and cerebrovascular reactivity (CVR) changes from functional near infrared spectroscopy (fNIRS) assessments. CVR was assessed using tissue oxygenation index changes during a breath holding provocative task for up to 30 s. CVR was residualized for age, education (years), sex, baseline creatinine, baseline stroke risk (using the Framingham Stroke Risk Profile), and baseline CVR levels.
In analyses examining correlates of cerebrovascular outcomes we found that greater amounts of weight loss (ΔTOI Saturation: 0.38 [0.06, 0.71] per 15 lbs of weight loss from baseline; p = 0.023) (Figure 4), whereas we found no such relationships with ambulatory SBP (0.06 [−0.29, 0.41], p = 0.757), DASH diet scores (−0.02 [−0.4, 0.3], p = 0.903), step count (0.01 [−0.3, 0.3], p = 0.960), or PSQI scores (0.01 [−0.4, 0.4], p = 0.980). Similarly, we found no associations between putative correlates of cerebrovascular outcomes and oxygenated hemoglobin changes: ambulatory SBP (p = 0.881), DASH diet scores (p = 0.993), step count (p = 0.371), or PSQI scores (p = 0.763).
In parallel analyses of task-based oxygenated hemoglobin changes, we found that greater levels of actigraphy-assessed physical activity associated with greater cognitive efficiency (as indicated by lower oxygenation per cognitive performance unit: B = −9.3 [−18.4, −0.3], p = 0.044). In contrast, changes in sleep (p = 0.177), ambulatory SBP (p = 0.981), DASH diet (p = 0.782), and weight (p = 0.659) were not associated with changes in cognitive efficiency levels.
Discussion
Our finding demonstrated that a cardiac rehabilitation-based lifestyle program among individuals with TRH, targeting physical activity and behavioral weight loss, associated with modestly preserved cognitive function over a one-year follow-up compared to individuals receiving an enhanced standard of care. In contrast, we did not observe parallel associations with cerebrovascular function or in several biobehavioral outcomes. These findings extend prior evidence linking lifestyle modification to improved cognitive outcomes among individuals with TRH by suggesting that sustained lifestyle adherence may help to preserve cognitive and cerebrovascular function among some individuals with TRH. Surprisingly, our findings suggested that sleep quality improvements following lifestyle modification may associate with greater preservation of cognitive function, although this association was not hypothesized and requires replication. In addition, it is notable that treatment group differences were strongest for some aspects of memory performance, whereas we observed less consistent group differences in other cognitive domains. In parallel, we found that greater weight loss and physical activity levels associated with different cerebrovascular outcomes. Finally, our findings were somewhat surprising in failing to show an association between ambulatory blood pressure, physical activity, dietary quality, and weight loss with preserved cognitive function.
Numerous prior studies have demonstrated that lifestyle modification could help sustain cognitive functioning among older adults and those with chronic medical conditions. Multimodal interventions targeting vascular risk factors using exercise and diet have provided the strongest evidence of cognitive benefits, such as from the Finnish Geriatric Intervention Study to Prevent Cognitive Impairment and Disability (FINGER) trial,63,64 the PreDIVA trial, 65 MAPT 66 and most recently the U.S. Pointer trial, 67 among others.68–70 Moreover, recent data from the SPRINT MIND trial 6 suggest that intensive blood pressure lowering results in reduced risk of MCI and dementia, as well as reducing white matter lesion burden on magnetic resonance brain imaging. 71 Finally, prior meta-analyses have demonstrated that programs increasing physical activity or implementing behavioral weight loss appear to improve cognition across multiple domains of function.72,73
The lack of improvement in cerebrovascular function, as well as the lack of association between individual biobehavioral markers (e.g., physical activity and weight loss) with cognitive function in the present study is notable and warrants additional exploration. There are several potential explanations for this finding, in addition to the possibility that our analyses were underpowered to detect associations due to missing data associated with the COVID-19 lockdown. First, as we previously reported from the primary treatment findings in the TRIUMPH trial, 25 individuals in our enhanced standard of care control condition (SEPA) showed noteworthy, within-group changes in weight (–8.5 lbs), urinary sodium [–187 mg/day) and potassium levels (292 mg/day), in addition to reducing their blood pressure levels. Although the C-LIFE treatment group nevertheless showed greater improvements in most of these outcomes, the lifestyle changes made in the SEPA group were potentially impactful and may have made it more difficult to detect associations between lifestyle factors and cognitive changes. It is further possible that the intensive lifestyle intervention we provided was harder for individuals in the C-LIFE group to maintain even before the unprecedented complexities introduced by the COVID-19 lockdown. For example, preliminary estimates from one-year follow-up outcomes suggested comparable weight loss maintenance between groups (C-LIFE: −10.6 [−13.4, −7.9] lbs versus SEPA: −9.6 [−13.7, −5.6] lbs from baseline to one-year). These findings suggest that, although the C-LIFE group showed greater weight loss after 4-months of treatment, these improvements declined over follow-up. It is possible that individuals in the SEPA group, although showing lesser lifestyle changes and blood pressure benefits immediately following trial completion, may have been better able to sustain their health habits over time. Similarly, our failure to find an association between blood pressure reductions and cognitive functions could also have been influenced by the substantive reductions in the SEPA group, making it harder to discern group differences. Finally, differential dropout in our sample could plausibly have influenced the pattern of observed findings, as BMI, metabolic function, and baseline atherosclerosis all predicted drop-out status.
One notable nuance in the present study was the differential changes in cognition observed across domains when examined using reliable change indices. The majority of TRIUMPH participants appeared to show some level of cognitive decline, albeit subtle in many cases. Cognitive changes were most commonly observed in Executive Function/Learning and Psychomotor Speed domains, whereas Memory function was more stable. Observing changes in these domains among hypertensive patients is consistent with prior literature demonstrating preferential impairments on these tasks due to their greater vulnerability to microvascular ischemic changes.1,74–78 Executive Functions, in particular, are conventionally thought of as heavily reliant on integrity within frontal-subcortical brain regions, which are sensitive to the effects of chronic hypertension.8,79,80 Our finding that Memory performance was most improved relative to other cognitive domains may partly reflect this secular decline in other domains across both groups. It is also possible that our selection criteria, which would have eliminated individuals with observable cognitive impairments on the Montreal Cognitive Assessment Battery,4,26 could have biased our sample to over-enroll individuals with cognitive impairments due to vascular risk factors and exclude individuals showing the memory changes that typify ADRD. In the present study, although treatment improvements were most notable on tasks of memory, explanatory analyses indicated improvements were likely driven by improved learning efficiency and complex information sequencing, which are conventionally associated with the dorsal attention network within the prefrontal cortex. 81 Memory is closely associated with attention and concentration abilities for many individuals and our findings seem consistent with the notation that improvements in some aspects of selective attention, a component of executive function that has an important influence on memory abilities, may have played an important role. 82
Recent studies have reported similar findings, demonstrating that physical activity and sleep quality associate independently with cognitive functioning. 83 Interestingly, an emerging body of data have extended these findings84,85 to suggest that physical activity and sleep may have moderating effects on cognitive and brain health outcomes, with greater physical activity levels protecting against the untoward effects of poor sleep.86,87 These findings underscore the need for future trials to explore the potentially moderating effects of different lifestyle components on cognitive outcomes. This is particularly important as existing evidence not only suggests that aerobic exercise training improves sleep outcomes, 88 but that treating sleep dysfunction alone does not appear sufficient to improve cognitive function. 89 Moreover, improvements in cognitive function following aerobic exercise training among individuals with MCI do not appear to be paralleled by improved sleep, 90 suggesting that both lifestyle factors interact in their impact on brain outcomes.
Although we failed to find treatment group differences on any fNIRS indices, observed associations between biobehavioral factors and fNIRS outcomes are potentially interesting and warrant further study. Our finding that changes in cerebrovascular reactivity, assessed by fNIRS, associated with changes in actigraphy-assessed is consistent with prior trial data suggesting that physical activity improves cerebrovascular functions. 91 Similarly, prior studies examining task-based changes in hemodynamic recruitment have suggested that physical activity improves hemodynamic efficiency during cognitive task performance. For example, in a one-year study examining fNIRS correlates of physical activity among individuals with MCI, MCI individuals engaging in physical activity showed decreased task-related hemodynamic responses despite stable cognitive performance. 92 Similarly, our finding that greater weight loss associated with greater CVR changes during a breath holding task parallels prior work suggesting that improved cardiorespiratory fitness (which overlaps substantially with weight) associates with increased cerebrovascular functions. 10
The present study should be viewed with several important limitations in mind. First, these findings were observed in a small and select cohort of individuals with resistant hypertension and require replication in a larger cohort. We were further limited by loss of data due to the COVID-19 pandemic lockdown, which coincided with the post-treatment assessments for final participants in the active treatment portion of the TRIUMPH trial. Second, we are limited in our ability to follow up with all participants due to distancing requirements that occurred due to the COVID-19 lockdown. Our findings are therefore encouraging in that we found improvements in memory despite our small sample size and underpowered statistical analyses. Third, our study sample was highly selected, such that more highly educated individuals with access to cardiac rehabilitation were preferentially included due to fidelity considerations. Fourth, our use of cognitive testing could have been enhanced by the inclusion of ancillary neuroimaging outcomes or ADRD biomarkers, which would have provided a more comprehensive assessment of the role lifestyle modification has on systemic markers of brain health.
In conclusion, our findings suggest that a comprehensive lifestyle modification program can improve cognitive function among individuals with resistant hypertension. These findings further emphasize the importance of comprehensive lifestyle changes as a means of protecting against the deleterious effects of hypertension among aging adults. Our findings also suggest that, even in a sample of individuals who have a high burden of cardiovascular risk factors and are refractory to pharmacological therapy for hypertension, changing lifestyle habits may nevertheless result in improved cognitive functioning and help to reduce the risk of ADRD.
Supplemental Material
sj-docx-1-alz-10.1177_13872877251409716 - Supplemental material for Lifestyle, sleep quality, and cognitive function in resistant hypertension: One-year follow-up from the TRIUMPH trial
Supplemental material, sj-docx-1-alz-10.1177_13872877251409716 for Lifestyle, sleep quality, and cognitive function in resistant hypertension: One-year follow-up from the TRIUMPH trial by Patrick J. Smith, James A. Blumenthal, Krista Ingle, Lana L. Watkins, Forgive Avorgbedor, Stephanie K. Mabe, William Kraus, Crystal Tyson, Alan Hinderliter and Andrew Sherwood in Journal of Alzheimer's Disease
Footnotes
Acknowledgements
Competing interests and sources of financial support: this work was supported by Grants HL122836 and HL130237 from the National Heart, Lung, and Blood Institute, National Institutes of Health, Bethesda, MD. All authors declare no relevant financial interests.
Ethical considerations
Participants in the current study were monitored by the Duke University Institutional Review Board to ensure ethical treatment and clinical research practices.
Consent to participate
All participants provided informed consent to participate.
Consent for publication
Not applicable
Author contribution(s)
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Grants HL122836 and HL130237 from the National Heart, Lung, and Blood Institute, National Institutes of Health, Bethesda, MD. 1R61AG080615-01 from the National Institute of Aging.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
Select data from the present analyses may be available upon request from the corresponding author.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
