Abstract
Tauopathies, including Alzheimer's disease (AD), are characterized by the pathological aggregation of tau proteins, culminating in progressive neurodegeneration and cognitive decline. Conventional therapeutic strategies remain limited by inadequate blood-brain barrier (BBB) penetration, insufficient specificity for tau pathology, and lack of real-time disease monitoring. While carbon-based nanomaterials have been extensively investigated for amyloid-β related applications, their potential for tau-directed theranostics remains at an early yet rapidly advancing state. Carbon-based nanomaterials–including carbon dots, fullerenes, graphene derivatives, and carbon nanotubes–exhibit exceptional physicochemical versatility, demonstrating potential to inhibit tau aggregation, scavenge reactive oxygen species (ROS), enable precision drug delivery, and facilitate ultrasensitive detection of tau biomarkers. Their inherent capacity for BBB penetration and dual diagnostic-therapeutic functionality positions them as transformative candidates for next-generation management of tauopathies. Despite this promise, most evidence remains preclinical, and major translational challenges persist. These include long-term biocompatibility and toxicological uncertainties, lack of standardized synthesis and functionalization protocols, production scalability, and the molecular heterogeneity of tau pathology across distinct tauopathies. This review provides a comprehensive assessment of carbon nanomaterials to date in the context of tau pathology, critically examining their theranostic potential and the barriers impeding clinical translation. By delineating current limitations and strategic priorities for future research, it underscores the urgent need for coordinated interdisciplinary efforts to convert these versatile nanoplatforms from conceptual promise to clinically actionable technologies for precision tau-targeted therapy.
This is a visual representation of the abstract.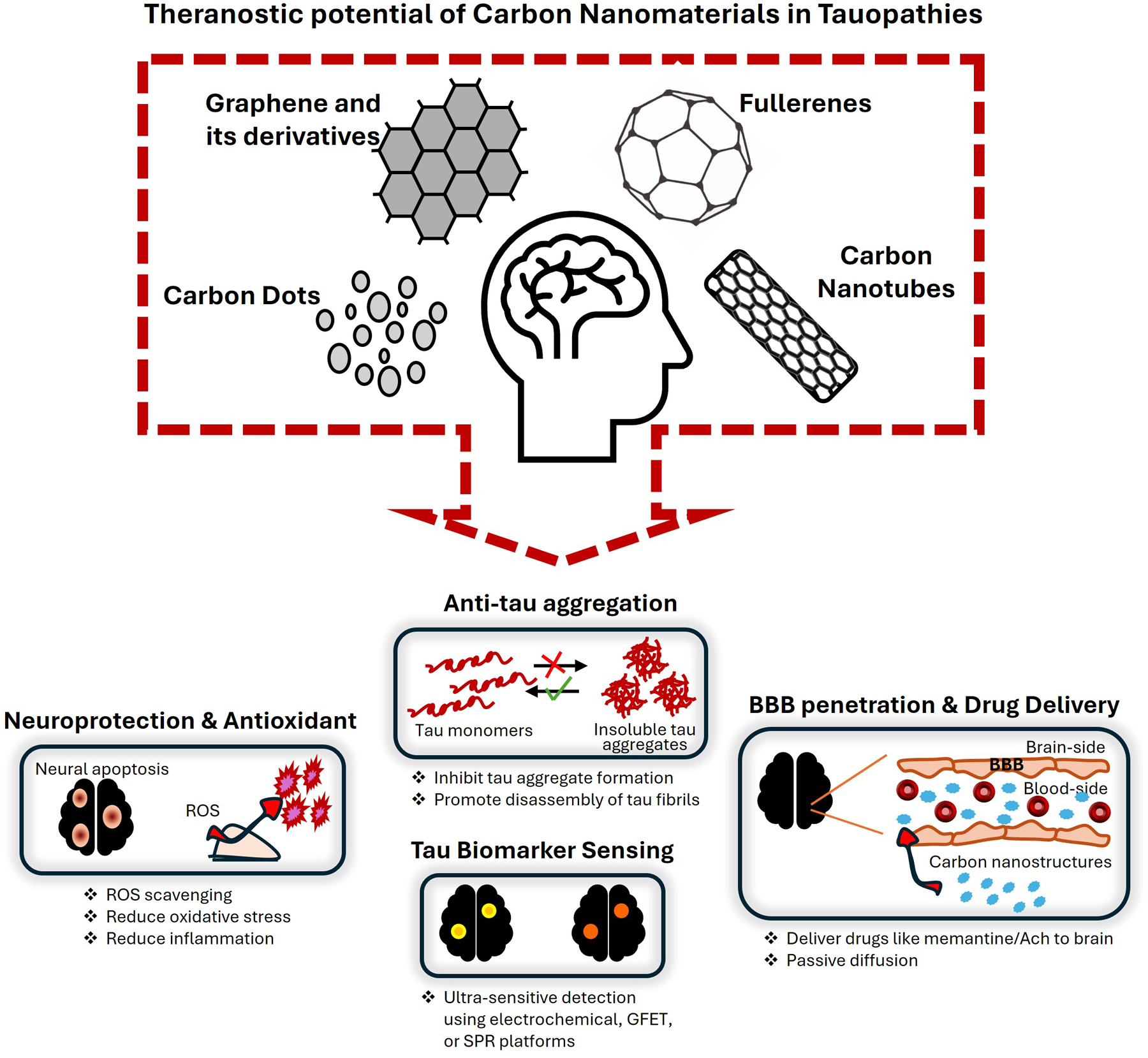
Keywords
Get full access to this article
View all access options for this article.
