Abstract
Background
Empowering people living with memory problems, including Alzheimer's disease, dementia, or mild cognitive impairment, and their carers to be engaged in the shared decision-making process about their medicines could reduce their risk of experiencing medicine-related harm. More co-designed resources to support them are needed.
Objective
To co-design and test a conversation-starter tool to empower people living with memory problems and their carers (consumers) to have a conversation with their healthcare professional (HCP) about their medicines.
Methods
We employed a consumer participation method with an eleven-member Steering Group consisting of six consumers and five HCPs from Australia and Maryland, United States of America. We conducted one-on-one interviews with consumers and HCPs to test the tool. We analyzed data deductively using the integrated-Promoting Action on Research Implementation in Health Services (i-PARIHS) framework and the Communication-Health Information Processing (C-HIP) model. Inductively, we also drew emergent themes that did not fit within the i-PARIHS.
Results
We successfully co-designed the PRIME tool and recruited 35 participants (26 consumers; 9 HCPs) to improve the tool's readability and comprehensibility. We identified four major themes including: Theme 1: Variable consumer self-advocacy; Theme 2: Value of the tool; Theme 3: Changing behavior, empowerment, and motivation; Theme 4: Future use, dissemination, and implementation.
Conclusions
Our participants valued the PRIME tool as an empowerment resource. They believed it would remind them of their permission to ask questions about their medicines. This may lead to a closer alignment of medicines with a person's goals of care.
Keywords
Introduction
According to the World Health Organization, there are nearly 10 million new cases of dementia every year. 1 Specifically, Alzheimer's disease affects 55 million people worldwide. A dementia diagnosis impacts people's health and their ability to manage their medicines. 2 Mild cognitive impairment (MCI) is defined as cognitive decline greater than expected for an individual's age that does not interfere notably with activities of daily life. 3 It is estimated that the prevalence of MCI in people aged 65 years or older is 3–19%. 4 MCI is distinctly different to dementia, yet people living with MCI may also face similar health and medicine-related challenges.
Despite the growing number of people living with memory problems including dementia or MCI, limited co-designed resources to support them to make complex decisions about their medicines exist. 5 People living with dementia or MCI often experience other long-term health conditions, such as diabetes or cardiovascular disease. 6 This contributes to their overall multimorbidity. The single-disease model and disease-specific prescribing guidelines encourages the prescription of multiple medicines without holistically considering the impact of multimorbidity on medicine appropriateness. 7 This contributes to people living with dementia including people with Alzheimer's disease, taking, on average, seven or more medicines. 8 This increases their risk of experiencing negative consequences of polypharmacy, commonly defined as taking five or more medicines. 9
As memory problems progress, one or more medicine(s) may become potentially inappropriate or misaligned with the person's current goals of care.10,11 Such a change in goals of care could create an opportune time to review medicines. It could also trigger deprescribing, reducing or stopping, potentially inappropriate medicines that may be causing more harm than good.12,13 This is especially important as people living with dementia or MCI are at an increased risk of being prescribed at least one or more potentially inappropriate medicine and therefore are at an increased risk of experiencing medicine-related harm. 14
What matters most from the perspective of the person and their carer should center shared decision making. 15 However, engaging consumers (people living with memory problems including dementia or MCI, and their carers) in decision making is challenging 16 given that dementia or MCI symptoms may include memory loss or trouble communicating. Carers may feel burdened by regularly engaging in proxy decision-making for their care recipient. 17 Healthcare professionals also navigate challenges such as complex clinical environments, competing professional responsibilities and limited resources including time. 18 Due to this and various other barriers, shared decision-making, reviewing medicines and deprescribing in practice remain limited. 19
Consumer empowerment is a key strategy for supporting deprescribing in practice. 20 This may include using direct-to-consumer brochures and campaigns that aim to increase consumer awareness about the benefits and harms of medicines. 21 Additionally, limited resources have been co-designed or tested with people living with dementia or MCI. 5 Despite their perceived effectiveness, using these resources in clinical practice remains limited. 19
Co-design involves meaningful end-user engagement and can occur through the entirety of the research process. 22 Co-design can be a powerful approach in consumer-engaged research to proactively include the consumer and ensure the output takes into consideration the consumer's needs, priorities, and values. 23 Therefore, the acceptability, relevance, and uptake of research outputs in practice can be improved using this approach. 24
Our preliminary research has shown that improving the process of shared decision-making about medicines is a top priority for people living with memory problems, their carers and their families. 25 Specifically, consumers have reported limited self-efficacy to initiate conversations about reviewing and deprescribing medicines with their healthcare professional. 26 Consumers have reported they would value direct-to-consumer (i.e., patient-directed), easy-to-use, accessible tools to support them in understanding the continued need for their medicines. 27 Co-designing resources (tools, educational materials) to nudge consumers to engage in deprescribing conversations with their healthcare professional may improve consumers’ self-efficacy and advocacy. 26 This could encourage a greater alignment between current goals of care and medicines taken by consumers. It may also help alleviate carers’ decision burden. 28
Together with consumers, healthcare professionals, and our partners from The University of Maryland, United States (US), we aimed to co-design a conversation-starter tool for people living with dementia and their carers. The PRIME
Methods
We aimed to develop a tool relevant to both an Australian and American audience due to shared challenges in both countries including a high burden on healthcare systems and the prevalence of medication-related harm. 29 Therefore, we co-designed the PRIME tool with a Steering Group (SG) consisting of Australian and American members. We appreciate language differences between the two countries. Therefore, in our study, we will use the term ‘carers’ to refer to people who care for people living with memory problems unless referred to as ‘caregivers’ in quotations reported by our participants from the US.
The timeline of the co-design and the alpha-testing process is demonstrated in Figure 1. To co-design the tool, we:
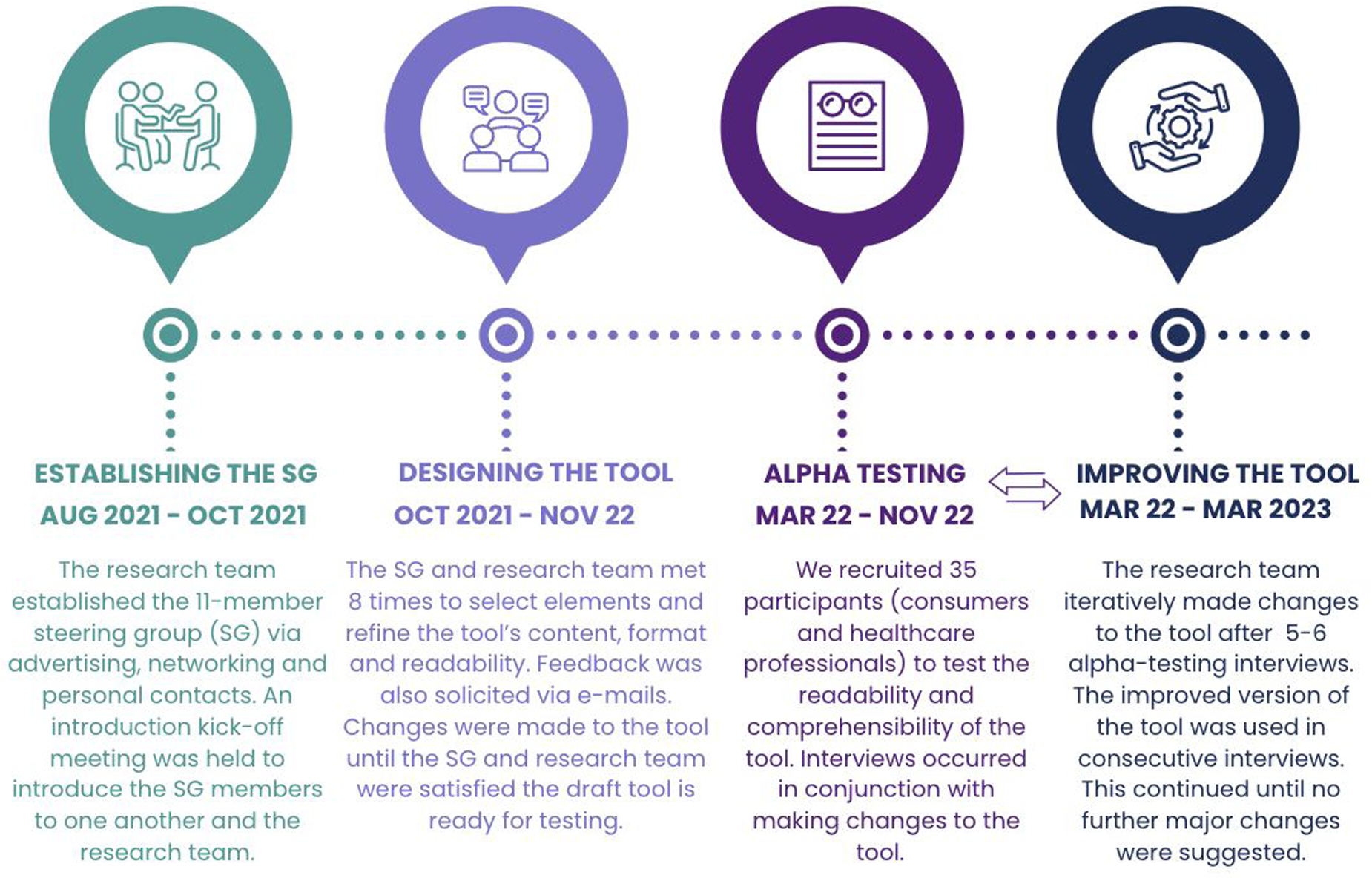
Timeline of co-designing the PRIME tool.
Established a stakeholder SG
In 2021, we established an 11-member stakeholder SG with 5 members from the US and 6 from Australia. This consisted of 2 people living with dementia, 4 carers and 5 healthcare professionals (three geriatricians, a nurse practitioner, and a social worker). Our research team consisted of three pharmacist-researchers (NJA, NB, ER) and a public health researcher (KRW). Further details regarding establishing and partnering with the SG is detailed in another article. 30
Partnered with consumers
We employed consumer participation methods to partner with consumers in our SG. 31 Between 2021–2023, we consulted consumers on the design and format of the PRIME tool (Figure 1). We also co-developed recruitment strategies, data collection instruments (e.g., interview guide) and our analysis approach with our consumers. We remunerated consumers for their contribution per hour of their involvement according to local guidance. 32
Co-designed the PRIME tool
The concept of the PRIME tool is based on the behavioral economics idea of nudging, the process of providing subtle cognitive cues to change behavior. 33 The research team proposed the initial key components to include in the PRIME tool. The research team presented these potential elements to the SG. In smaller groups of 3–4 members of consumers and 1–2 healthcare professionals led by a research team member (NJA, KRW, ER, NB), the SG selected and refined elements to include.
The research team incorporated feedback from the SG to improve the PRIME tool. This continued until April 2022, when the SG and research team agreed that no further changes were needed prior to alpha-testing. In March 2022, the penultimate version of the PRIME tool draft was ready for testing. To assess the tool's readability, we used the Sydney Health Literacy Editor. 34
Alpha-testing the PRIME tool
To alpha-test the readability and comprehensibility of the PRIME tool, we conducted one-on-one interviews with consumers and healthcare professionals in Australia and Maryland, US. We completed the COREQ-32 checklist (Supplemental Table 1) to summarize our qualitative approach.
To recruit participants, we advertised via consumer organizations (Dementia Australia) and an online research participation service (Step Up for Dementia Research). We also invited participants via personal contacts and snowballing techniques. We included (a) Consumers: people living with memory problems including MCI or mild-to-moderate dementia who could provide their own consent and/or their carers. We included people living with memory problems who are prescribed at least one regular medication from all healthcare settings and (b) Healthcare professionals (nurses, pharmacists, medical doctors, physician assistants, etc.) who have current or previous experience caring for people living with dementia. We excluded people who were aged <18 years old or could not speak or understand English.
Interviews
We developed the interview guide using the Communication-Human Information processing (C-HIP) model 35 and pilot tested it (Supplemental Table 2). We chose the C-HIP model to analyze how consumers receive health information. In Australia, pharmacist-researchers NJA and LSH conducted the interviews. In the US, interviews were conducted by pharmacist-researcher NB. Interviews were video, and audio recorded. The audio-recordings were transcribed and checked for accuracy.
Conceptualization of the analysis framework
We developed a qualitative analysis codebook using the Integrated Promoting Action on Research Implementation in Health Services (i-PARIHS)36,37 and the C-HIP model 35 (Supplemental Table 3). Using these frameworks enabled us to determine contextual factors needed for the successful facilitation and implementation of the PRIME tool by consumers in real-world settings.
The i-PARIHS framework outlines that successful implementation is a result of the facilitation of an
Analysis framework.

*C-HIP: Communication-Human Information processing model.
Analysis
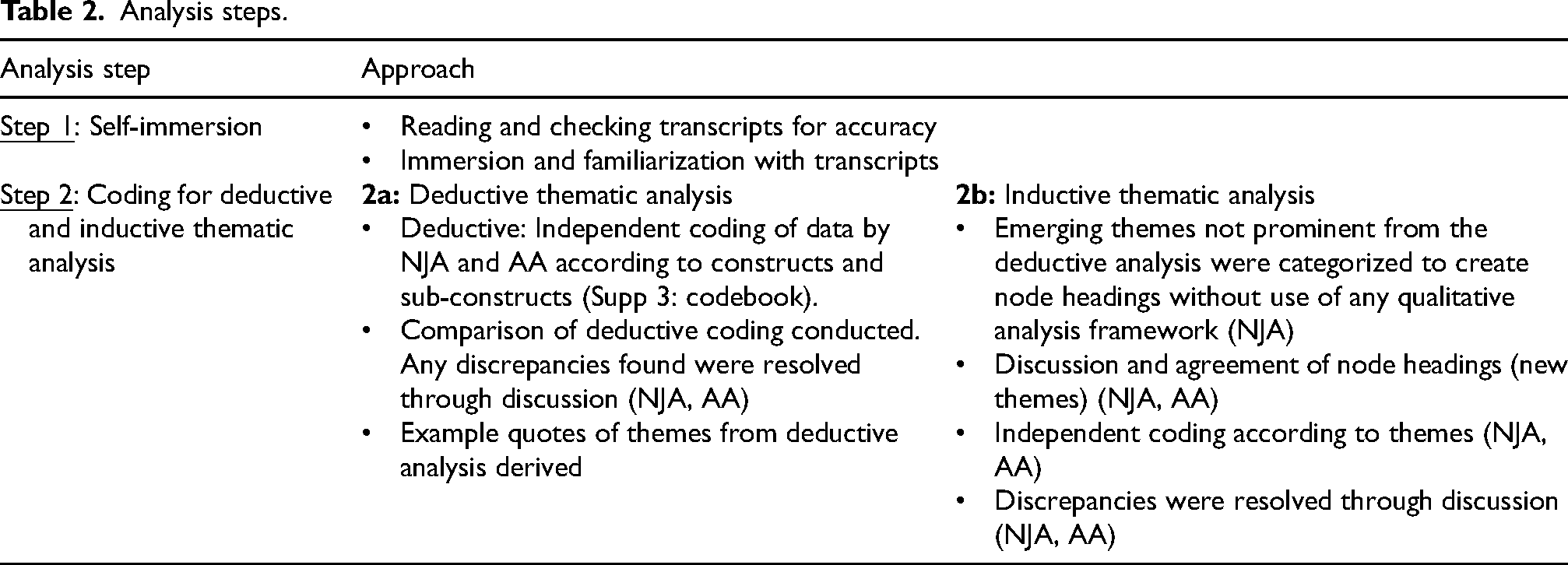
NJA and AA led a deductive and an inductive qualitative analysis based on the Braun & Clarke's Six Steps to Thematic Analysis. 38 Analysis steps are outlined in Table 2.
Analysis steps.
We employed a co-design, iterative approach to make necessary changes to improve the tool. A deliberative process between NJA and LSH to improve the readability, format, and content of the tool was taken. After conducting 5–6 interviews with participants, LSH met with NJA to discuss themes and to implement suggestions. We placed a greater weighting on changes suggested by consumers versus healthcare professionals. This is because we aimed to co-design a tool tailored and designed specifically for consumers, our main group of end-users. NJA made changes to the tool. The improved version of the PRIME tool was used in subsequent interviews. This continued until saturation of themes and no further major changes were suggested by participants. Saturation was determined via discussion amongst the research team.
Results
By conducting 35 qualitative one-on-one interviews in Australia and the US, we alpha-tested the co-designed tool to ensure it is both readable and acceptable by people living with memory problems and their carers. We refer to the individual who said the quote by using ‘HCP’ for a ‘healthcare professional’, ‘PLWD’ for ‘a person living with dementia’, ‘PLMCI’ for ‘a person living with MCI’ and ‘C’ for ‘carer’.
Our participants included 6 people living with dementia, 6 people living MCI, 13 carers, and 9 healthcare professionals in Australia and the US (Table 3). Of the participants, there were two dyads consisting of a person living with dementia and their carer. Healthcare professional participants had experience caring for people living with memory problems but did not care for the consumer participants in our study. Apart from the dyads, no other participants knew each other. Most consumer participants were from Australia and 30.8% of consumer participants were from the US. Just over half of our healthcare professional participants were from the US (55.5%). Most participants identified as Caucasian. Some participants identified as Asian, Black, or African American, Indian, or Aboriginal and Torres Strait Islander (First Nations People of Australia).
Demographics.

The PRIME tool's components and its utility
By conducting co-design meetings with the SG, we reached consensus to include three main sections in the PRIME tool draft (Supplemental Figure 1). These include (a) Background, (b) Self-reflection 39 and (c) Call-to-action. 30
Changes to the PRIME tool draft
Based on our participant's feedback, the original version of the tool (Supplemental Figure 1) was revised over 4 rounds to develop the penultimate PRIME tool (Figure 2).

The main changes included:
“My first thought when I saw it in the first version was that it looks very busy. I sort of looked at it and went like obviously it will be folded. But my first impression was there's a lot on there.” HCP10 “I think the same messaging could be done with …less questions, less conversation prompts.” HCP31
“Not all a lot of it's a good size, but some of it. Starting for me to get a bit small, I can read it. The bottom of the left-hand side. The top of the middle the just above the logos. That's a bit small.” PLMCI8
“We don't talk about doctor stuff at all until I get a little list out and say this is what we've got to talk about.” PLMCI4 & C5 “Us with dementia … with spatial awareness and things that we have with our vision, you need a space between the lines and things like that. It's just too close together and if you have a full dot, okay, start another line.” PLWD7 “How to prime a conversation about your medicines with your healthcare professional” to “How to start a conversation about your medicines” so that it is more comprehensible and relevant to our target audience. “Some people may not think of prime as a verb in the way that I'm thinking of it, but I know that it's important to put the name of the tool right on the front page.” C16 “My first thought that I think and the other thing that I thought is it's very much towards, it gives me the impression that taking medicines is a bad thing if that makes sense.” HCP10 “[You must consider] if you can’t read… and many people [may be] from a low social economic background. We have a lot of illiterate people in the community. These are the people who are taking too many medications…You know these are [the people] going to multiple doctors and getting medication from multiple doctors.” PLWD17
By the end of the alpha-testing phase, the reading level of the penultimate tool was reading grade level of 8. A grade of 8 or lower is deemed to be acceptable for most lay readers. We received positive feedback from our participants that the revised penultimate tool is readable by most consumers. “I’m perfectly happy because it [PRIME tool] just includes the target information. Not too, not too much, not too little, fantastic.” PLWD11
Thematic analysis
A list of constructs and sub-constructs is presented in Table 4 under
Deductive analysis displayed as constructs and sub-constructs of our analysis framework (i-PARIHS/C-HIP) with example quotes.

*HCP: healthcare professional; PLWD: person living with dementia; PLMCI: person living with mild cognitive impairment (MCI); C: carer or caregiver of a person living with memory problems (dementia or MCI). We define Innovation as the PRIME tool; Recipient as the person living with dementia or caregiver and Context as healthcare setting in which the tool is being implemented; Facilitation as how to implement the PRIME tool in practice.
Inductively, we extracted four emerging themes. These include:
Theme 1: variable consumer self-advocacy
Self-advocacy of consumer participants varied. Some reported that they have limited self-empowerment to broach a conversation with their healthcare professional. This underpinned their hesitancy to ask questions about their medicines. A dyad of consumers explained that people living with dementia may lose confidence which impacts their self-empowerment.
“Especially people [living] with dementia because they lose a bit of their self-empowerment. They lose their confidence.” C19 from (PLMCI18 & C19)
“I feel like it [the tool] is a way to give patients and their caregivers permission to ask about this. I think a lot of patients and caregivers think that they can’t question their doctor about things like their medicine. That the doctor knows best, and I love that this is sort of a way to say - hey, you can talk to your provider about your medicine.” HCP31
Some consumers felt that asking such questions would lead the healthcare professional to be defensive and that using the PRIME tool might help to mitigate this. Other consumers commented on how having this conversation might become more challenging as dementia progresses.
“So, if you take away the defensiveness and give them something [the tool] that they can talk to the doctor [about] a medication and start that [conversation] first. I think that will definitely help.” C15
“I'm already quite involved in at least the medication management side of things as part of my care responsibilities, but I just think…as time goes on [and dementia progresses], I think this will become more of an issue.” C16
Some reported that despite them wanting to ask questions, they didn’t know how to have this conversation, especially given time constraints of medical appointments.
“Health appointments can turn into a case of, well, all these instructions are dictated to you. You don't get much time at the end to ask about anything.” C16
Consumers’ self-advocacy to start the conversation heavily depended on the rapport between the consumer and their healthcare professional. Cultural factors also impacted this. Some consumer participants reported that they believe they have the confidence and ability to have this conversation, with the acknowledgement that this might not be the case for other people. People reported feeling vulnerability and shame related to the cognitive challenges they may be facing. This played an important factor for some participants who relatedly limited the number of conversations they have with their healthcare professional.
“Sometimes, you realise people from some countries [cultures] aren’t even aware sometimes that they have a right to say.” PLWD11
“I'm not intimidated by my doctor; I find her absolutely wonderful and she's a great listener. But I mean, [sometimes], some people …. might seek information [from the doctor], but then [the doctor] doesn’t give much back, so there's not this natural give and take…equalizing of the relationship, which closes off some people especially if you’re feeling an element of shame about … the cognitive difficulties you’re having and you feel that you’ll be written off and not taken seriously.” PLMCI13
Differing views emerged regarding whose responsibility it is to start a conversation. Some believed that the healthcare professional should initiate these conversations whereas others thought that healthcare professionals are time poor, leaving them to initiate the conversation. Some consumers felt they should be having these conversations with mostly one central person, often their General Practitioner (GP).
“I’ve learned that doctors are so busy that they can’t think without us initiating the conversation about the care of our loved ones.” C6
“[My] opinion is that the doctor's office should initiate. We are so conditioned into just doing what the doctors say, that we're not questioning the medication that we're on.” C23
“If you would like to try to decrease, come off or maybe even increase the medication that that's yeah, it's up to you and you can discuss it well, hopefully with your GP.” PLMCI8
Theme 2: value of the tool
The tool was widely accepted by our participants to have the potential to start a conversation with a healthcare professional about their medicines. In particular, the ‘call to action’ section (Figure 2) was favorited by many and was seen as a valuable resource to start the conversation with their healthcare professional. Carers especially found much value in the tool and believed it could reduce their decision burden. Carers also believed they can use the tool as a reminder of the questions and points they would like to raise with the healthcare professional. Consumers believed the tool could raise awareness about the potential harms of medicines and remind them they have permission to have a conversation about their medicines.
“What I like about it [the tool] is, it gives people permission to do that [have a conversation], you know, and I mean it depends on your doctor and a lot on the … the relationship you have with your doctor.” PLMCI13
Consumers believed that having a conversation about their medicines is important and a starting point to optimally managing their medicines. One consumer commented on the uniqueness of the tool and expressed that it “means business”.
Consumers reported a gap in terms of support, such as resources, that are available to them to manage their medicines. For example, one consumer reported they wished the PRIME tool was available to them when they were caring for their spouse who had dementia. Our consumer participants recognized that the tool could potentially fill this gap. One consumer valued the broad and global application of the tool to all medicines.
“[The tool] is providing a step. But at the same time, it's cleverly saying that it's not providing detailed information about the details of medication, etc, [the purpose of the tool] is just to start a conversation.” PLWD9
“If I had when my husband was diagnosed [with dementia], that would be helpful. But that was a long time ago. Even today [the tool] is helpful too, for myself anyway, it is too late for my husband already but for myself.” C15
“[The tool] looks as if we really mean business, and let's talk about something, yes.” P7
Healthcare professional participants commented that they would value consumers using the tool and filling it out prior to their appointment (e.g., during pre-screening). They recognized that they or the consumer could use the tool to broach and facilitate a focused conversation about medicines. They also recognized that the tool might support healthcare professionals to empower people they care for to be part of the conversation. They also reported that this tool fills a gap and focusses on having a conversation rather than only providing guidance on how to deprescribe.
“One of the hardest parts about doing this [a medicine review] … is initially approaching the conversation, and I think this [the tool] is an icebreaker. They’ve already partly considered the topic and then you can get the insights early to then have a more focused discussion about medicines.” HCP1
“We are underutilising patients or carers to have these discussions. I think people [pharmacists] would … quite like that [tool] to help to have conversations with people to think about whether they still need all their medicines.” HCP10
“A lot of deprescribing materials we have are for providers on how to do it [deprescribing] and less for patients that they can initiate that conversation as well.” HCP33
Theme 3: changing behavior, empowerment, and motivation
Consumers spoke about the need for changed behavior in the clinical environment. Specifically, they reported they would like more acceptability for them to question the continued need of their medicines. They reported they would value becoming empowered and having more ownership of their medicine list, their goals of care and health information. Some consumers reported that the tool could provide them with the knowledge and assertiveness they need to become empowered to have a conversation.
“It's giving them permission to have the ability (to have a conversation) and I suppose (consider) to stop their medication.” C5
“It [the tool] gives people permission to be a bit more assertive about what they're doing and I'm relatively assertive about medical things.” PLMCI13
“I think this tool empowers you with knowledge. And it gives you the control. Puts you in control…and then you're then able to go and start the conversation.” PLWD17
In a clinical sense, a consumer commented that the tool would make them more likely to have the confidence to go and speak with their geriatrician about their medicines.
“With the only one geriatrician [we have in town] and everybody [living] with dementia knows who he is, and a lot of people find him not particularly helpful, but…if I went in there next month with a document like this, I think he would have a bit more respect for us and my concern for my husband's medications.” C6
A need for changed behavior related to the interaction between healthcare professionals and consumers emerged. Some reported the need to balance the relationship.
“They [people living with dementia] feel that it's a bit like a head headmaster and student situation. Whereas this [tool] empowers.” PLMCI8
Some healthcare professionals also reported the need for healthcare professionals to increase their self-competency to have conversations about optimizing medicines. This was thought to require changed behavior through education efforts for example.
“How could we help them [healthcare professionals] in finding the right words or language to start those conversations? And then you get the question is, how do I, you know, still [implement] deprescribing?” HCP10
Theme 4: future use, dissemination, and implementation
Participants broadly thought that the tool can be implemented in various healthcare settings including primary, aged care and hospital. In primary care, consumer participants reported that the tool could be presented in medical clinics and/or pharmacies for them to fill out whilst they are waiting to see their doctor or whilst their medicines are being dispensed. Consumers often wanted to bring in a filled-out copy of the tool to their medical appointment for them to discuss alongside their healthcare professional.
“So, it would be part of my list to go to the GP. It's certainly every six months you know the care plan thing or whatever, please review.” PLWD14
“Could be a prompt for them [consumers] to have it there when they pick up a prescription, or they [pharmacists] could put it in the prescription bag.” PLWD7
Most healthcare professionals expressed that reading a filled-out tool prior to their appointment would help them to conduct a comprehensive medicine review. Consumers also wanted their healthcare professional to read the filled-out tool as well. A healthcare participant also made comment about incorporating it in the paperwork consumers fill out before their appointment. They also suggested it could be used by hospital pharmacists to conduct medicine reconciliations for inpatients.
“It would be a good starting point if it was available in GP's and that would also suggest that they may have looked at it as well.” PLWD9
“If it's out in the waiting room… they could fill that out…. with our workflow or medical assistants [physician assistants] will go through med rec [medicine reconciliation] forms that the patient fills out, and then the physician assistant kind of reviews that. This could be kind of integrated into the med rec [medicine reconciliation] even in the hospital so that might be an opportunity to implement and include this tool.” HCP35
Additionally, participants reported that it could be incorporated at timely points in a consumers’ health journey, including during annual wellness visits and at transfer points of healthcare such as upon admission into hospital or aged care and upon hospital discharge.
“I think [about] our work in the annual wellness visits. It [the tool] would tie in nicely…Oftentimes, if there is someone…with cognitive impairment, we reach out to their caregivers too.” HCP33
“I'd like to see this document in aged care facilities, residential care facilities. I'd love to see that available to residents and their families, their primary family.” C6
Participants expressed that they could envision the tool being adapted to suit audiences beyond those who live with memory problems, including older people in general and people living with disabilities. Overall, consumers reported they would share it with their friends in support groups including people without memory problems.
“I see this tool as a great tool for people with disabilities, so I would like to see this tool be used in the disability sector.” PLWD17
“I would recommend it to all my friends. Not because all my friends have memory problems or anything like that, but I think it's just a good tool to have.” PLMCI8
Discussion
We co-designed and tested the PRIME tool for people living with memory problems including dementia or MCI, and their carers, to start conversations with their healthcare professional about their medicines. It has been recognized that increased efforts to bridge the deprescribing to practice gap are needed. 40 A key priority area to reduce polypharmacy includes person and carer engagement. This can be encouraged by using co-designed tools and resources for medicine management in clinical practice. 15 Powered by the behavioral economic concept of nudging, the PRIME tool can be used as a low-touch, high-value implementation strategy to sustainably reduce the use of potentially inappropriate medicines. 33 It is a novel, co-designed, conversation-starter international tool for people living with memory problems and their carers. Partnering with our diverse SG from the US and Australia enabled us to genuinely co-design the tool with the values and needs of people living with memory problems and their carers in mind. 24
The PRIME tool is designed to empower consumers to start and engage in a global, holistic conversation about reviewing the continued need of their medicine(s). It was designed to be translational and usable in everyday clinical practice. To the best of our knowledge, limited communication aids and supportive resources have been specifically designed and evaluated with people living with memory problems to date. 5 Most consumer-directed deprescribing resources are for older people who do not experience memory problems. 41 A 2019 environmental scan found that most online, consumer-directed deprescribing resources are written above the average reading level, deeming them unsuitable for people with lower to average health literacy. 5 Moreso, just above a third (37%) of the resources presented balanced information about the benefits and harms of deprescribing. 5 Another 2023 review echoes these findings and reported that most available resources have not specifically been tested or evaluated with the intended end-user population. 41 The PRIME tool specifically fills this gap providing people living with memory problems and their carers with a resource to support having a conversation about their medicine needs. 5 It has been co-designed, tested and revised to ensure that it is readable and usable by consumers, carers and healthcare professionals who care for them. While the onus is not solely on the consumer to start a conversation about their medicines, evidence suggests that consumers who are empowered to initiate this conversation with their healthcare professional are more likely to take fewer inappropriate medicines.26,42
We designed the tool with the needs of people living with memory problems and carers at the forefront. For example, we included information in the “background” section that would be useful for people living with memory problems, including those living with Alzheimer's Disease, to know about medicines.10,43 We included potential common medicine-related adverse events or side effects that may significantly impact people living with dementia's health and wellbeing. This information is particularly relevant as dementia or MCI may progress and their goals of care may change. 43 The tool is unique because it is not limited to specific medicine-class(es) and it does not specifically focus on a step-by-step guide to taper or discontinue potentially inappropriate medicines. Rather, its focus is to support the person living with memory problems and/or their carer to have a conversation about their medicines. 44 Changes to medicine(s), including continuing or deprescribing the medicine(s), is a collaborative decision made with the healthcare professional and is a secondary outcome.
Whilst the tool has been co-designed alongside consumers for consumers, various healthcare professionals, individually or in multidisciplinary teams (facilitators) can use the tool to navigate a deeper conversation about reviewing medicines and more generally the person's goals of care. Various barriers to optimizing medicine use in practice exist. These include time constraints, poor communication between prescribers at transitions of care and lack of incentives. 18 We believe the PRIME tool may encourage healthcare professionals to review medicines and implement medicine-related changes to address the concerns of the person they care for. An unintended consequence could be that some consumers may feel it is their responsibility to start the conversation about their medicines with their healthcare professional after using the tool. The intent of the PRIME tool is not to place the responsibility of starting these conversations or making decisions related to appropriate medicine use on the consumer. Rather, the PRIME tool may overcome some of these barriers by using a soft nudging approach and empowering consumers to be engaged in the conversation and advocate for their health priorities and medicine needs.
Our healthcare participants reported they would recommend the inclusion of the tool in their pre-medical appointment paperwork and in medicine review processes. To create change, the tool could be embedded in real-world clinical workflows and integrated, for example, in comprehensive annual reviews. The tool may be used as a complement to other policy and practice reforms needed to optimize medicine use.
Changing behavior and especially, consumer engagement, sustainably can be challenging. 45 Our consumers expressed various challenges that may limit the occurrence of medicine-related conversations. These included consumers feeling overwhelmed and unempowered and challenges with juggling medical advice from multiple healthcare professionals. Consumers, the person living with memory problems and their carer together or individually, can use the tool independently and choose to discuss their PRIME responses with their healthcare professional when they next visit them. Most of our consumer participants considered the PRIME tool readable and expressed they would read and fill the PRIME tool prior to their medical appointment. They also reported that they would like to bring it to their appointment and use it as a reminder to ask their healthcare professional questions about their medicines.
Our findings suggests that the PRIME tool may be used in a variety of healthcare scenarios (medical appointments; annual wellness visits) and during transitions of care. For example, our participants suggested the potential integration of the tool and use upon hospital admission or discharge and for residents living in aged care.
Our consumers expressed that using the PRIME tool might increase their confidence to start a conversation. This may mitigate potential feelings of shame, isolation, stigma, and the fear of the unknown as dementia progresses and memory problems worsen. 46 From a healthcare professional perspective, limited access to guidance to optimize medicine use and time poor health environments were viewed as challenges. These echo other barriers previously reported in the literature. 18 Similarly, healthcare professionals reported that using the PRIME tool may address some of these challenges and encourage goal-oriented care. 28
Previous research suggests that people are willing to take less medicines with their healthcare professional's agreement. 47 Our findings suggest that various potential facilitators (healthcare professionals, individually or in multidisciplinary teams) can use the PRIME tool to support their conversation with consumers about reviewing medicines. We also identified key facilitation strategies to increase the consumer's uptake of the tool (Table 4). These included the tool's clarity, the consumer's ability to read it in their own time, the consumer filling it out prior to their medical appointment in and the consumer individualizing the tool with their own questions that they can bring and refer back to in their medical appointment.
Strengths of our research included partnering with a SG which included consumers and healthcare professionals. We used a consumer participation method to ensure collaboration and increase the relevance and practicality of the PRIME tool. Additionally, the use of the i-PARIHS and C-HIP frameworks enabled a robust, rigorous analysis to determine contextual determinants that may influence the PRIME tool's implementation in practice. However, some of our findings could not be categorized within the i-PARIHS constructs and sub-constructs. For example, the use and dissemination of the tool was not captured by i-PARIHS. Therefore, we theorized additional themes by conducting an independent inductive analysis.36,48 Given our small sample size, and that most of our participants were Female from a Caucasian background, our findings may have limited generalizability to all people living with dementia and carers.
Our findings align with previously reported successful deprescribing conversation strategies. 27 Engaging consumers via the provision of educational materials (e.g., brochures) about the benefits and harms of medicines, has been positively associated with having more deprescribing conversations and supporting goal-concordant care. 49 Some interventional deprescribing studies resulted in a high discontinuation rate of medicines by providing patients and doctors with deprescribing leaflets for medicines (e.g., benzodiazepines; proton pump inhibitors). 42 Similarly, the PRIME tool could be implemented in practice to encourage conversations about medicine appropriateness.
Future research
We plan on leveraging our findings to implement the PRIME tool in clinical practice to explore its impact on changing behavior and increasing the number of deprescribing conversations. An overview of systematic reviews found that deprescribing evidence related to vulnerable groups or settings is limited. 50 Relatedly, resources for vulnerable or minority groups such as people from culturally and linguistically diverse (CALD) populations is limited. 21 This was also evident in our study with some participants suggesting the need to translate the tool into different languages (e.g., Chinese; Italian) (Table 4). Future research should aim to adapt and translate existing resources, such as the PRIME tool, to suit CALD people. Our participants reported mixed preferences about the mode of the tool (i.e., hard copy versus website or mobile application). Future research is needed to develop resources into various modes to cater for a diverse community.
Conclusion
People living with dementia or MCI, carers and healthcare professionals in our study recognized the need for a resource to empower them to have conversations about medicines. They believed our co-designed PRIME tool could fulfill this gap and nudge more conversations about medicines to occur in practice. Consumers using the PRIME tool alongside their healthcare professional may prompt a higher uptake of clinical recommendations that align their medicines with their current goals of care. This may improve a person's overall satisfaction with managing their medicines. It may also reduce their risk of experiencing medicine-related harm by deprescribing medicines that may cause more harm than benefit improving their overall health and wellbeing.
Footnotes
Acknowledgements
We would like to acknowledge Dementia Australia, Step Up for Dementia Research, and our stakeholder steering group members who supported this research. We would also like to acknowledge the US Deprescribing Network and the University of Washington's Plein Centre for Geriatric Pharmacy's support towards this research. We would also like to thank Professor Gillian Harvey for their mentorship.
Ethical considerations
Ethical approval was provided by the University of Queensland Human Research Ethics Committee (2022/HE000102).
Consent to participate
We gained written informed consent from participants who were people living with dementia or carers. We gained verbal informed consent from participants who were healthcare professionals.
Consent for publication
We have obtained written informed consent from our participants to publish deidentified data collected from this study.
Author contribution(s)
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work is supported by a National Health and Medical Research Council (NHMRC) Investigator Grant (NJA GNT2025939), (KRW GNT2017295), (ER GNT1195460), and the US Deprescribing Network, National Institute of Ageing (NJA NIA:1R24AG064025).
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
Due to ethical considerations and in line with our ethical approval, we will not make our data set available.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
