Abstract
Background
In individuals with cognitive deficit, neuropsychiatric symptoms (NPS) are typically assessed through informant-based reports. In contrast, affective symptoms in cognitively normal individuals are typically evaluated using self-report measures, leveraging their sufficient insight. Comparison of these assessments in previous studies indicated differences in their perspectives.
Objective
We aimed to analyze differences between self- and informant-reported NPS using the Mild Behavioral Impairment-Checklist (MBI-C) and their relationship with cognitive deficit.
Methods
The study included 127 participant-informant dyads from a memory clinic cohort, classified as subjective cognitive decline (SCD; N = 38) or mild cognitive impairment (MCI; N = 89) based on comprehensive neuropsychological assessment; MCI group was diagnosed according to core clinical criteria for MCI due to Alzheimer's disease. A control group consisted of 26 cognitively healthy individuals. Participants and their informants completed the MBI-C, evaluating five symptom domains.
Results
Both self- and informant-rated MBI-C scores were higher in the SCD and MCI groups compared to controls. Informant ratings correlated more strongly with objective cognitive deficit, particularly in memory and executive function domains, whereas self-ratings showed only one association (higher apathy with worse attention). In MCI, informants reported more severe impulse dyscontrol symptoms. Correlations between self and informant ratings were weak to moderate.
Conclusions
This study highlights discrepancies between self- and informant-rated NPS assessments, with informant ratings more strongly linked to cognitive deficits in older adults without dementia. Therefore, we suggest using both assessments to capture informants’ and patients’ subjective experiences and facilitate early identification of neurodegenerative diseases.
Keywords
Introduction
Neuropsychiatric symptoms (NPS), changes in mood, behavior, or personality in older individuals, can be part of normal aging but may also indicate early neurodegeneration including Alzheimer's disease (AD). 1 These symptoms often emerge before dementia, i.e., in mild cognitive impairment (MCI) and subjective cognitive decline (SCD). 2 New-onset NPS in these populations are associated with a higher risk of developing dementia in AD. 3 Informant reports further suggest that NPS severity increases with advancing cognitive impairment. 4
To raise awareness about NPS as early symptoms of pathological aging, a syndrome termed mild behavioral impairment (MBI) was introduced. MBI refers to persistent new-onset NPS in adults aged 50 + without dementia. These symptoms cannot be attributable to another current psychiatric disorder (e.g., major depression) and should cause at least mild functional impairment. 5 MBI can be diagnosed concurrently with MCI but can also precede any objective cognitive deficit. 6 Studies have indicated that MBI reflects neurodegenerative changes in the brain and that it can be present in any neurodegeneration including AD.7–9 To assess MBI, the Mild Behavioral Impairment Checklist (MBI-C) was developed. 10
In dementia, NPS are typically evaluated by informants (a relative or other close person) due to patients’ reduced insight. 11 In cognitively normal or mildly impaired individuals, NPS are often assessed via self–rated questionnaires.12,13 Given the subjective nature of symptom reporting through questionnaires, informants may differ from patients, potentially having significant implications. Self-informant rating discrepancies in cognitive complaints showed potential to predict incident cognitive decline in participants at risk of AD, which led to the formation of a specialized working group. 14 Similar discrepancies appear in affective symptoms, with greater differences observed in more pronounced cognitive deficit.15–17 Such differences may stem from both patient- and informant-related factors: reduced patient insight due to memory or executive deficits and caregiver burden leading to overestimation of symptoms.18–20 However, most previous studies used different instruments for patients and informants, limiting the direct comparability of their ratings. One exception is a study using the Apathy Evaluation Scale, showing higher self-rated apathy in cognitively normal individuals, but lower in MCI patients at risk of AD, compared to external assessments. 16
The MBI-C, suitable for both self and informant rating, enables direct comparison. Recently, patient administration has begun to be used more often.21–23 However, only a few studies have directly compared both perspectives.24,25 They concluded that both informants and participants with SCD and MCI most frequently reported affective dysregulation, impulse dyscontrol and decreased motivation. It is important to note that their ratings differed in severity and correlated only weakly.24,25 Notably, these studies involved community-dwelling older adults without comprehensive neuropsychological assessment. No study to date has examined MBI-C self-informant discrepancies in a clinical population.
Our study addresses this gap by examining self-informant differences in the MBI-C in a memory clinic sample of older adults without dementia. Across the cognitive continuum, we aim to describe and compare the severity of NPS reported by participants and informants and to investigate the self-informant agreement in NPS ratings and its relation to cognitive impairment. We hypothesize an increase in NPS severity across the cognitive continuum (lowest in controls, higher in SCD, highest in MCI, for both self and informant ratings). Furthermore, we expect only modest self-informant associations in NPS ratings, resulting in a discrepancy in their MBI-C scores. These self-informant differences will increase with cognitive impairment, particularly in memory and executive function.
Methods
Participants
The study included 127 participant-informant dyads referred to a memory clinic for cognitive complaints and subsequently enrolled in the Czech Brain Aging Study.
26
Based on comprehensive neurological and neuropsychological examination, participants were classified as SCD or MCI. SCD (
Additionally, a control group of participant-informant dyads was included (
Exclusion criteria included a diagnosis of dementia and other neurological or psychiatric condition significantly affecting cognition or NPS (e.g., Parkinson's disease, frontotemporal lobar degeneration, traumatic brain injury, stroke, alcohol or substance abuse, or current/past major psychiatric disorder). Demographic characteristics of the participants are presented in Table 1.
Demographic and neuropsychological characteristics of the participants.
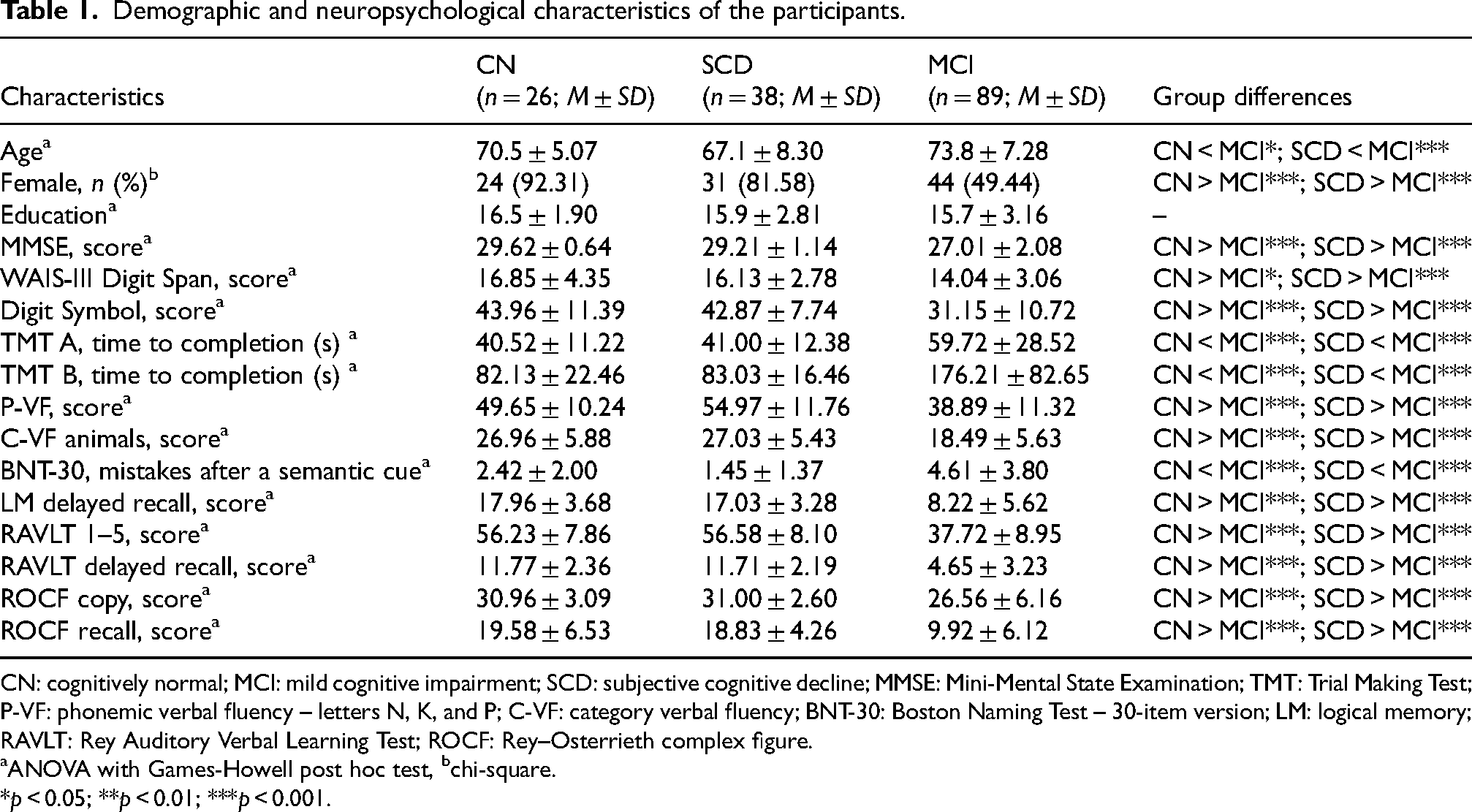
CN: cognitively normal; MCI: mild cognitive impairment; SCD: subjective cognitive decline; MMSE: Mini-Mental State Examination; TMT: Trial Making Test; P-VF: phonemic verbal fluency – letters N, K, and P; C-VF: category verbal fluency; BNT-30: Boston Naming Test – 30-item version; LM: logical memory; RAVLT: Rey Auditory Verbal Learning Test; ROCF: Rey–Osterrieth complex figure.
ANOVA with Games-Howell post hoc test, bchi-square.
*
All participants provided written informed consent according to the Declaration of Helsinki and the study was approved by the Ethics Committee of Motol University Hospital.
Neuropsychological assessment
The neuropsychological battery included the Mini-Mental State Examination (MMSE) as a screening of global cognition and the following tests covering five cognitive domains28,29: (1) memory: Rey Auditory Verbal Learning Test (RAVLT), 30 Logical Memory from the Wechsler Memory Scale-III (LM), 29 and 3-min recall of the Rey-Osterrieth Complex Figure Test (ROCFT recall) 31 ; (2) executive function: Trail Making Test B (TMT B) 29 and phonemic verbal fluency – letters N, K, P (P-VF) 32 ; (3) language: 30-item Boston Naming Test (BNT) and category verbal fluency-animals (C-VF) 29 ; (4) attention and working memory: Trail Making Test A (TMT A) and Digit Span Forward and Backward (DS) from the Wechsler Adult Intelligence Scale-III 29 ; and (5) visuospatial function: Rey-Osterrieth Complex Figure Test (ROCFT copy). 31
Neuropsychiatric assessment
The Czech version of the MBI-C was completed by both participants and their informants (partner, descendant, or another relative). 33 The MBI-C is a 34-item questionnaire designed to assess NPS before the onset of dementia. 10 It covers five domains aligned with MBI diagnostic criteria: decreased motivation (6 items), affective dysregulation (6), impulse dyscontrol (12), social inappropriateness (5), and abnormal perception and thought content (5). 5 Each item is rated for presence (yes/no) and severity (1-mild, 2-moderate, 3-severe). The symptoms should represent a change from the person's usual behavior and persist over the last 6 months. Total and domain scores are calculated by summing severity ratings (e.g., total score is 0–102). Participants with ≥4 missing MBI-C items were excluded; if ≤3 items were missing, both total and domain scores were calculated without these items. In total, 41 items (0.4%) of the 10 404 patient and informant items were omitted. No single MBI-C item was commonly omitted; missing responses were distributed across items.
Statistical analyses
Group differences in demographic and neuropsychological variables were assessed using ANOVA with Games–Howell post hoc tests, or chi-square tests for categorical data. Because MBI-C data distribution was skewed, nonparametric tests were used: Kruskal–Wallis tests (with Dwass-Steel-Critchlow-Fligner post hoc comparisons) to compare MBI-C scores among diagnostic groups, and Wilcoxon signed-rank test for paired data to compare informant- and self-ratings within each diagnostic group, visualized with ggplot2. 34 Associations between informant- and self-rated MBI-C scores were analyzed using Spearman correlations and these correlations were compared between diagnostic groups using bootstrapping.
Associations between MBI-C scores and cognitive domains (memory, attention and working memory, executive functions, language, visuospatial functioning and global cognition) were examined using linear regression analysis. Cognitive domains were composite
Only SCD and MCI participants data were included in the regression analyses, because most MBI-C scores in CN were zero. Data from SCD and MCI participants were combined to increase statistical power. MBI-C variables were log-transformed after adding 1 to MBI-C scores (see Supplemental Figures 1 and 2 for distributions before and after transformation).
The assumptions of linear regression were checked with residual plots, Q-Q plots and checking the variance inflation factors. Most models met the assumptions of linear regression, but some models, particularly those with social inappropriateness or abnormal thought and perception content as predictors had deviations from these assumptions. On the whole, these deviations were not considered sufficient to alter the analytic strategy that aimed to examine associations of all MBI domains with multiple cognitive domains.
Analyses were run in and R 4.3.2, with
Results
Clinical and demographic characteristics of the study sample
The clinical and demographic characteristics of the study sample are shown in Table 1. MCI participants were older than CN (
Neuropsychiatric characteristics of the participants – MBI-C informant and self-rating.
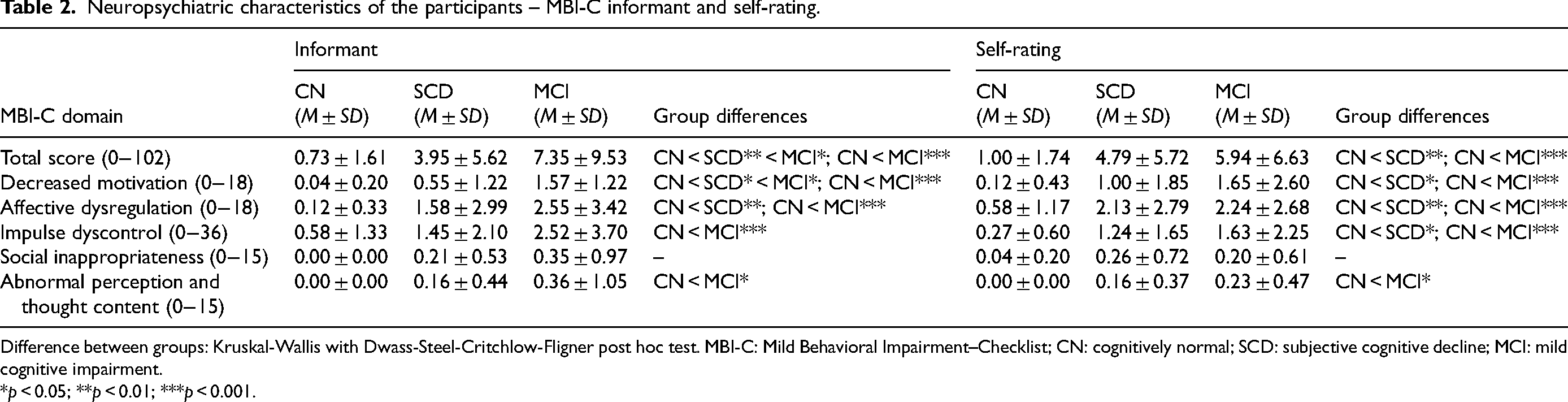
Difference between groups: Kruskal-Wallis with Dwass-Steel-Critchlow-Fligner post hoc test. MBI-C: Mild Behavioral Impairment–Checklist; CN: cognitively normal; SCD: subjective cognitive decline; MCI: mild cognitive impairment.
*
The MCI group scored higher than the CN group on the MBI-C total and domain scores in both informant and self-ratings, except for social inappropriateness (abnormal perception and thought content domain according to informant
As for the informants, most of them were spouses (94 informants; 61.4%), fewer were descendants (50 informants, 32.7%) and the remainder (6 informants; 3.9%) were others (e.g., siblings, grandchildren).
Self-informant rating discrepancies
Overall, self- and informant-rated MBI-C scores correlated weakly to moderately (
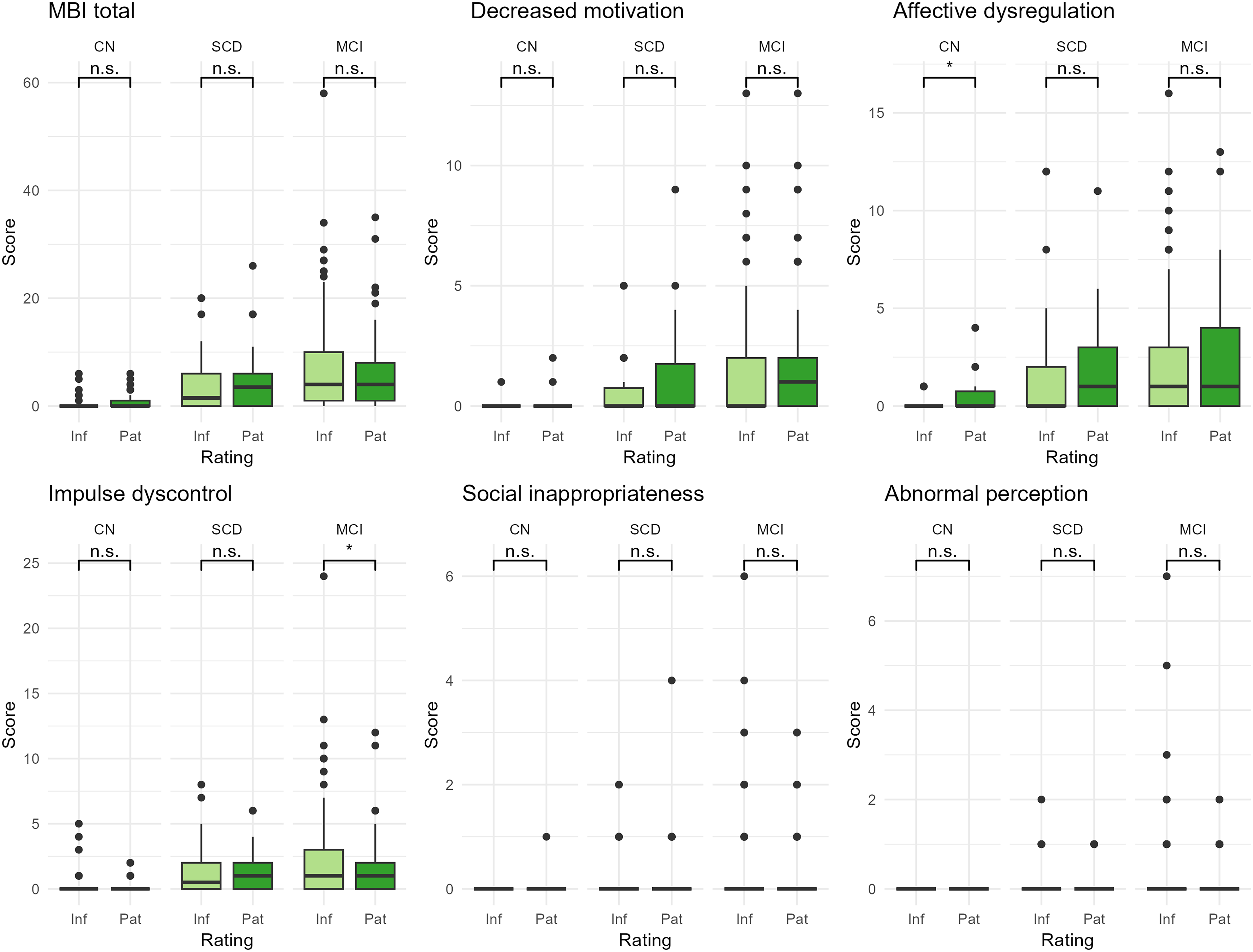
Comparison of MBI-C informant and participant scores within diagnostic groups.
Correlations between informant- and self-rated MBI-C scores.
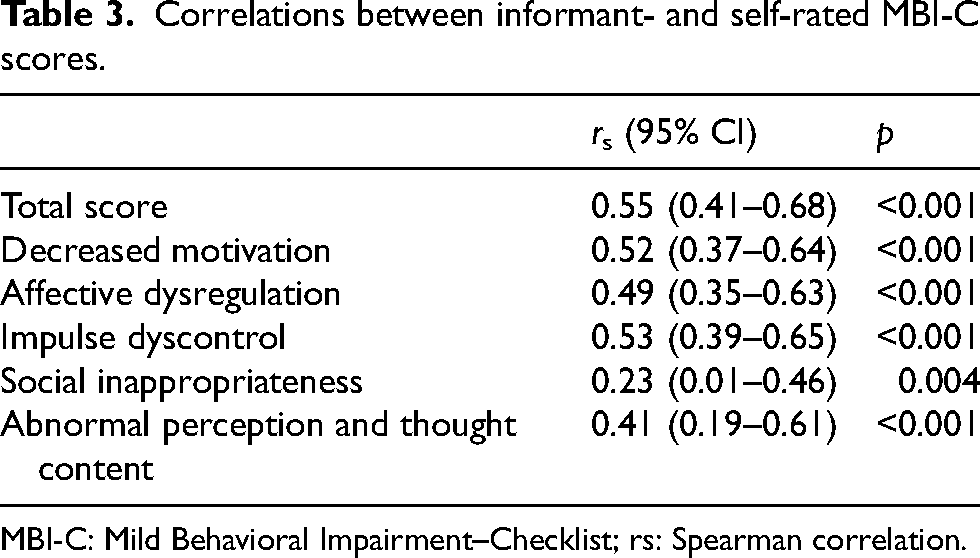
MBI-C: Mild Behavioral Impairment–Checklist; rs: Spearman correlation.
Associations with cognitive domains
The results comparing linear regression analyses with informant- and self-rated scores as separate terms versus informant-self difference scores are presented in Supplemental Table 2. In summary, the models with separate scores (mean
Having shown that using separate MBI-C informant and participant scores increase
Associations between MBI-C informant and patient scores after adjusting for covariates.
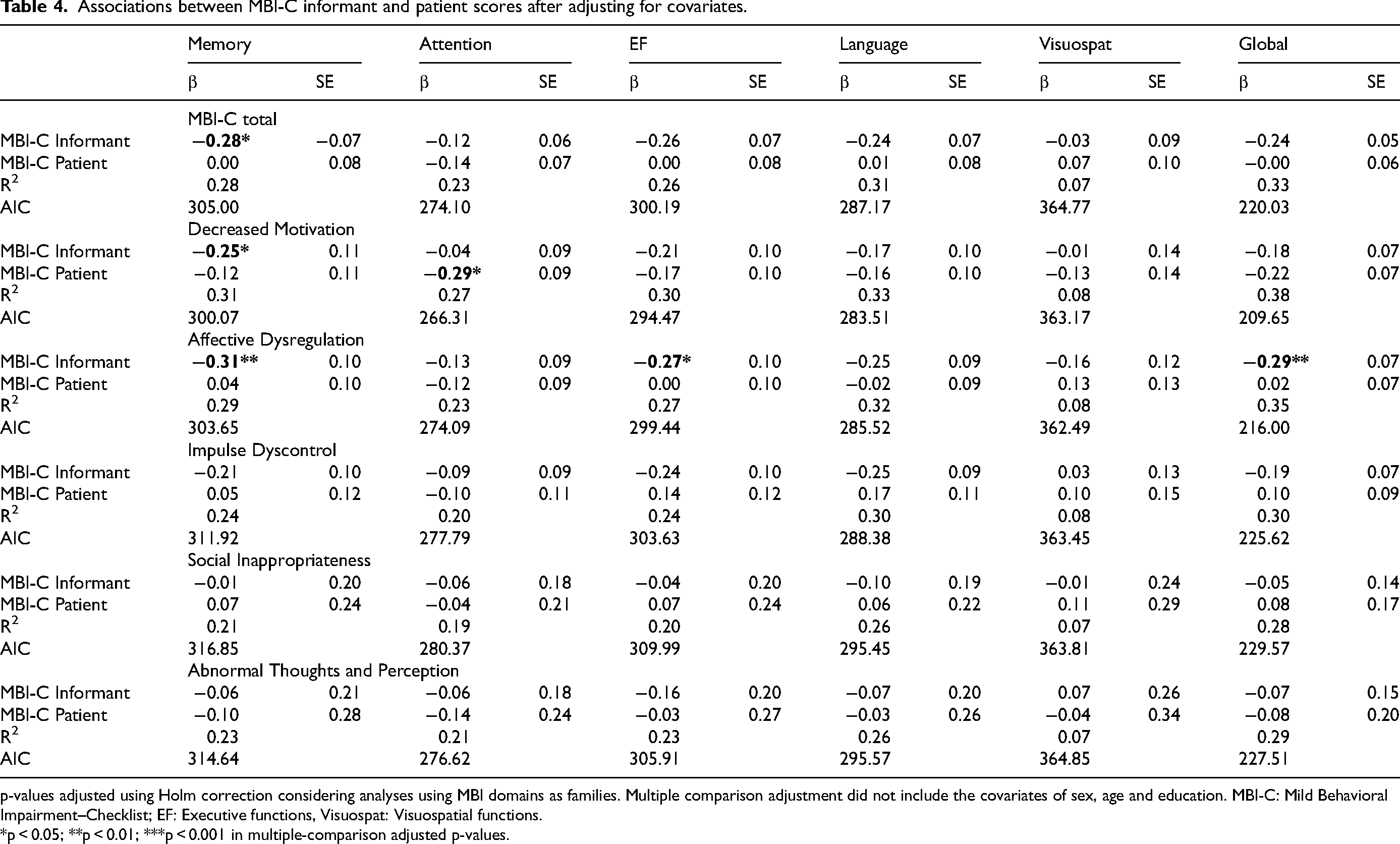
p-values adjusted using Holm correction considering analyses using MBI domains as families. Multiple comparison adjustment did not include the covariates of sex, age and education. MBI-C: Mild Behavioral Impairment–Checklist; EF: Executive functions, Visuospat: Visuospatial functions.
*p < 0.05; **p < 0.01; ***p < 0.001 in multiple-comparison adjusted p-values.
Discussion
This is the first clinical study using both informant- and self-rated versions of the MBI-C in a neuropsychologically characterized memory clinic sample of non-demented older adults. We compared self and informant ratings of 127 individuals across cognitive continuum preceding dementia and examined their associations with the patients’ cognition. Both self and informant ratings were higher in SCD and MCI compared to the control group. Importantly, informant-rated NPS severity in the clinical (SCD + MCI) group was associated with impairment across multiple cognitive domains, contrasting with the lack of substantial correlation with the self-rated NPS. On average, informants reported more severe impulse dyscontrol than participants themselves. Overall, patient-informant agreement on NPS was weak-to-moderate.
NPS across the cognitive continuum
Our study builds upon previous population-based studies by examining NPS across the cognitive continuum preceding dementia.24,25 Consistent with these studies, we found that both participants and informants most frequently reported decreased motivation, affective dysregulation and impulse dyscontrol, while social inappropriateness and abnormal perception and thought content were rarely reported. As expected, NPS were higher in both clinical groups than in controls.
We observed continuing gradient of worsening informant-rated NPS through the cognitive continuum (CN-SCD-MCI) in the total score and in the decreased motivation domain. Informants also reported higher affective dysregulation in both SCD and MCI compared to CN, with no difference between clinical groups. Our results are consistent with previous clinical studies showing increasing NPS severity across the cognitive continuum as measured by the MBI-C or the NPI-Q.2,4,38 Higher impulse dyscontrol and abnormal perception and thought content emerged only in MCI compared to CN, in line with studies showing time course of various NPS.2,39
Although patient administration of the MBI-C has begun to be used more often in studies, our study is the first investigation using MBI-C self-report in clinical population.21–25 A recent community-based study using the self-rated MBI-C found that the MCI group reported more symptoms than CN in total score, decreased motivation, affective dysregulation and abnormal perception and thought content domains. 23 Our study aligns with these results; in addition, we found higher scores in the impulse dyscontrol domain in MCI group compared to CN. On the other hand, Leow et al. did not find any difference between SCD and CN in any MBI-C domain contrasting with our study, where the SCD group had higher scores in the majority of MBI-C domains. 23 This discrepancy likely reflects sample differences: clinical SCD cohorts generally have a higher prevalence of subjective cognitive complaints, NPS, and amyloid positivity than community samples, increasing the likelihood of detecting NPS differences. 10 40–43 Indeed, other clinical studies using domain-specific self-report questionnaires have also found higher NPS not only in MCI but also in SCD group compared to CN.44–46 In contrast, we observed no significant differences between SCD and MCI in any self-rated MBI-C domain, which aligns with another clinical study reporting comparable levels of self-rated depressive symptoms in these groups. 45
Altogether, our results confirm the presence of mild NPS in both clinical groups of nondemented older adults. We observed that CN and SCD groups, although similar in cognitive performance, differed in several NPS as reported by themselves or their informants. Cognition-related anxiety and worry are among the main reasons for which SCD individuals seek medical help and may be overlapping with MBI, but our study shows that there are also other NPS, such as apathy and impulse dyscontrol, that the individuals experience and informants observe in this stage. 6 Meanwhile, SCD and MCI groups, although cognitively different by definition, only differed in informant-rated total MBI-C and decreased motivation scores, suggesting that increasing cognitive impairment is associated with greater NPS severity only in certain domains. This pattern partially aligns with a finding that Aβ-positive SCD individuals did not differ from Aβ-positive MCI in any NPI-Q score. 47 The slightly higher NPS levels of apathy observed in our study (compared to studies using NPI-Q) may indicate a higher sensitivity of the MBI-C for capturing these changes in nondemented older adults.
Self-informant rating discrepancies
Analogously to previous studies, informant and self MBI-C ratings showed only weak-to-moderate correlations.24,25 We also found that informants reported systematically more impulse dyscontrol symptoms than patients with MCI, consistent with previous research highlighting the greatest self-informant discrepancy in this domain. 25 Patients may underreport behaviors like irritability, impulsivity, or aggression—either due to lack of insight or an attempt to maintain a positive self-image—whereas informants may overestimate the severity of these symptoms based on their observations, because it occurs often in social situations and is associated with perceived burden.48–50 In a previous study, items with the greatest self–informant discrepancy were primarily related to negative personality traits, supporting this interpretation. 25
We observed no significant self-informant discrepancy in the affective dysregulation domain. Prior studies on depressive and anxiety symptoms have yielded mixed results, with some finding higher self-ratings and others higher informant ratings.16,24,51,52 Only two population studies examined self-informant differences in NPS with MBI-C: one found no self-informant difference in affective symptoms, whereas the other reported that participants endorsed more affective symptoms than their informants, which we did not confirm in our clinical groups.24,25 Notably, the raw MBI-C scores (including affective dysregulation) were higher in our clinical groups than in those community samples. 24 In a clinical population, affective symptoms are typically more pronounced and thus more apparent to informants, whereas in community samples, the symptoms tend to be milder and more difficult for informants to detect.10,42 As a result, informants may not observe the milder symptoms that participants still experience. Another explanation of these differences between self- and informant-rated depressive symptoms could be their different nature (externally observable versus internally experienced). 53
Associations with cognitive domains
Our findings highlight that informant and patient NPS ratings have distinct relationships with objective cognition. While the overall variance in cognition explained by NPS ratings was modest (maximum 15% of a cognitive domain), informant-rated NPS showed consistently stronger links to cognitive performance than self-rated NPS. We found that higher informant total and domain scores were associated with poorer memory, executive, and global cognitive function, whereas self-ratings showed only one association (higher self-rated apathy with worse attention). This pattern aligns with previous reports: informant-rated MBI-C scores correlate with worse global cognition in clinical sample and with memory and executive deficits in community sample, whereas self-rated MBI-C shows minimal cognitive associations.4,54,55 However, the association of the self-rating with attention in our study was not found in informant ratings, highlighting a unique contribution of the patient self-reports in this instance. Of note, this association was as strong as those found between informant ratings and other cognitive domains, suggesting that patient self-ratings can still provide valuable insight.
Discrepancies between patient and informant NPS ratings have been observed to widen with more severe impairment in memory and executive function, likely reflecting emerging anosognosia (unawareness of deficits) in the patient.15,18 Anosognosia is linked to memory impairment and can manifest even in prodromal stages of neurodegenerative diseases. 19 56–58 Notably, in our analyses the informant rating alone was a better predictor of cognitive outcomes than the informant-patient difference score, suggesting that the informant perspective largely drives the association between NPS and cognitive impairment.
These discrepancies between patient and informant reports also raise the question of whether clinician-administered assessments could provide added value. The MBI-C administration instructions also allow for a clinical interview. Although we did not examine this mode of administration, studies with related instruments suggest potential benefits: for instance, the clinician-rated Apathy Evaluation Scale (AES-C) predicted progression from MCI to dementia better than self- or informant-reports. 16 Also, the Neuropsychiatric Inventory clinician version (NPI-C) was developed to enhance objectivity through clinical judgment. 59 Similar advantages may apply to the MBI-C, but further validation of clinician interview administration is needed.
Limitations
This study has several limitations. First, the sample came from a memory clinic, which limits the generalizability to the general population—clinical populations tend to exhibit a higher prevalence of NPS than community samples.10,42 Second, we examined an etiologically heterogeneous sample without biomarkers. However, in this way our sample reflects clinical practice where biomarkers are often missing. Third, our informant-based measures did not account for informant characteristics. Recent research indicates informant factors (such as age, gender, or relationship to the patient) can influence reported NPS.
60
Future studies should consider adjusting for informant characteristics. Fourth, domains of social inappropriateness and abnormal perception and thought content were very infrequent in our sample, as expected in early-stage AD, which may cause small differences and lack of association with cognition in those domains.
1
Analyses using these domains did not unequivocally meet the assumptions of linear regression and might benefit from different statistical approaches than the ones employed here. However, for most models, the assumptions of linear regression were met. Finally, the correlations between self- and informant reports would ideally be interpreted with the reliability of these measures in mind. However, to estimate reliability in cross-sectional data informatively would require establishing the dimensionality of the MBI-C with factor analysis, which our sample size is not equipped to do. Therefore, it is a limitation of our study that we have used the MBI-C scales with the
Conclusion
This clinical study highlights important self-informant discrepancies in reporting NPS in older adults without dementia. Informant ratings consistently showed stronger associations with cognitive deficits particularly in memory and executive function suggesting they may offer a more accurate reflection of the patient's current cognitive status. Furthermore, impulse dyscontrol was consistently underestimated by patients. Yet, self-ratings provided relevant data, with self-perceived apathy correlating with attention deficit, highlighting a patient perspective that informants may miss. Our findings have important clinical implications supporting the need for complementing self-reports with information from their informants even in early stages of neurodegenerative diseases. Future studies should examine these discrepancies on biomarker-defined cohorts and from longitudinal perspectives.
Supplemental Material
sj-docx-1-alz-10.1177_13872877251401620 - Supplemental material for Comparing self and informant perspectives on neuropsychiatric symptoms in older adults in a memory clinic
Supplemental material, sj-docx-1-alz-10.1177_13872877251401620 for Comparing self and informant perspectives on neuropsychiatric symptoms in older adults in a memory clinic by Monika Krejčí, Veronika Matušková, Toni T Saari, Hana Horáková, Šárka Borovská, Jan Laczó, Jakub Hort and Martin Vyhnálek in Journal of Alzheimer's Disease
Supplemental Material
sj-xlsx-2-alz-10.1177_13872877251401620 - Supplemental material for Comparing self and informant perspectives on neuropsychiatric symptoms in older adults in a memory clinic
Supplemental material, sj-xlsx-2-alz-10.1177_13872877251401620 for Comparing self and informant perspectives on neuropsychiatric symptoms in older adults in a memory clinic by Monika Krejčí, Veronika Matušková, Toni T Saari, Hana Horáková, Šárka Borovská, Jan Laczó, Jakub Hort and Martin Vyhnálek in Journal of Alzheimer's Disease
Supplemental Material
sj-xlsx-3-alz-10.1177_13872877251401620 - Supplemental material for Comparing self and informant perspectives on neuropsychiatric symptoms in older adults in a memory clinic
Supplemental material, sj-xlsx-3-alz-10.1177_13872877251401620 for Comparing self and informant perspectives on neuropsychiatric symptoms in older adults in a memory clinic by Monika Krejčí, Veronika Matušková, Toni T Saari, Hana Horáková, Šárka Borovská, Jan Laczó, Jakub Hort and Martin Vyhnálek in Journal of Alzheimer's Disease
Footnotes
Acknowledgements
We would like to express our thanks to our neuropsychological team: doc. Mgr. et Mgr. Tomáš Nikolai, PhD, Mgr. Adéla Fendrych Mazancová, PhD and Mgr. Terezie Zuntychová, and our neurologists: MUDr. Ondřej Lerch, MUDr. Jiří Cerman, PhD, MUDr. Martina Laczó, PhD and MUDr. Zuzana Nedelská, M.Sc., PhD, who also participated on the data acquisition.
During the preparation of this work the authors used DeepL and ChatGPT to improve the readability and language of the manuscript. After using these tools, the authors reviewed and edited the content as needed and take full responsibility for the content of the published article.
ORCID iDs
Ethical considerations
The authors assert that all procedures contributing to this work comply with the ethical standards of the relevant national and institutional committees on human experimentation and with the Helsinki Declaration of 1975, as revised in 2008. The study was reviewed and approved by Ethics Committee of Second Faculty of Medicine, Charles University and Motol University Hospital.
Consent to participate
The participants provided their written informed consent to participate in this study.
Consent for publication
Not applicable
Author contribution(s)
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Charles University Grant Agency (grant number 62924); the National Institute for Neurological Research (Program EXCELES): Funded by EU – Next Generation EU (grant number LX22NPO5107); and Ministry of Health, Czech Republic – conceptual development of research organization, University Hospital Motol, Prague, Czech Republic (grant number 00064203).
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Martin Vyhnalek is an Associate Editor of this journal but was not involved in the peer-review process of this article nor had access to any information regarding its peer-review. Other authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
The datasets presented in this article are not readily available because of the policy of the Czech Brain Aging Study (CBAS), which allows sharing of the data only after previous approval. Requests to access the datasets should be directed to MV, martin.vyhnalek@fnmotol.cz. The study was not preregistered.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
