Abstract
Background
CaV2.3 (R-type) channels mediate calcium signals involved in neuronal excitability, synaptic plasticity, and neurotransmitter release in the hippocampus. However, there is currently no information about their potential changes in Alzheimer's disease (AD).
Objective
This study aims to analyze the protein levels, expression, and subcellular localization of CaV2.3 channels in the hippocampus of 5xFAD mice.
Methods
We employed histoblot, western blot, qRT-PCR, and immunoelectron microscopy techniques.
Results
Using the histoblot technique, we observed that the protein content of CaV2.3 in female 5xFAD mice was downregulated in the hippocampus and caudate putamen at 5 months, with more pronounced downregulation at 10 months that also affected the septum. The hippocampus was the most affected region, where CaV2.3 immunodetection was significantly decreased in a few dendritic layers at 5 months, but this reduction extended to all layers and subregions by 10 months, demonstrating age- and laminar-dependent changes. The pattern in females differed from that seen in males and remained unchanged in APP/PS1 and P301S mice. Immunoelectron microscopy of the hippocampus showed CaV2.3 in apical and oblique dendrites of CA1 pyramidal cells, with a significantly higher presence in dendritic spines. Quantification revealed a significant decrease in the density of CaV2.3 in oblique dendrites and dendritic spines of CA1 pyramidal cells, whereas apical dendrites and presynaptic compartments were unaffected in 5xFAD mice.
Conclusions
Overall, this downregulation in postsynaptic CaV2.3 channels could impact neural circuit activity and contribute to the cognitive deficits observed in 5xFAD mice.
Introduction
Calcium (Ca2+) plays fundamental roles in different aspects of neuronal physiology, such as gene expression, neuronal growth, neuronal differentiation, synaptogenesis, membrane excitability, neurotransmitter release, synaptic plasticity, and learning and memory. 1 Accordingly, disturbances in Ca2+ homeostasis contribute to pathophysiological conditions, including Alzheimer's disease (AD).2,3 This neurodegenerative disease is characterized by three neuropathological hallmarks: extracellular deposition of amyloid-β (Aβ), intracellular neurofibrillary tangles of tau, and loss of synapses. 4 Changes in various Ca2+ channels, receptors, exchangers, and pumps have been shown to contribute to changes in the processing of the amyloid-β protein precursor (AβPP) that result in increases in the Aβ production,5,6 which can further increase Ca2+ dyshomeostasis that ultimately results in cell death.7,8
Among the different regulatory mechanisms of intracellular Ca2+ concentration, voltage-gated Ca2+ channels (CaV) mediate neuronal function due to their ability to gate high levels of this ion.1,9 CaV channels can be subdivided into CaV1 and CaV2 (high voltage-activated) and CaV3 (low voltage-activated) channels. CaV2 channels are of critical importance to synaptic Ca2+ signaling because of their spatial relationship and proximity to synaptic sites10–13 and regulation by G protein–coupled receptors (GPCRs). 14 Out of the three types of CaV2 channels, CaV2.1 (P/Q-type) and CaV2.2 (N-type) are mostly located at presynaptic sites along active zones,12,13,15–18 whereas CaV2.3 (R-type) are located at both postsynaptic and presynaptic sites11,19–22 in neurons of cortical and subcortical areas. Unique among their properties, CaV2.3 channels have membrane voltage and kinetics that are different from those that characterize P/Q, N, L, or T channels.23,24
Four subunits compose CaV2.3 channels: the pore-forming α1 subunit, designated as α1E, and two structural/regulatory α2-δ and β subunits. 25 The α1E subunit forms R-type channels, which are involved in regulating neurotransmitter release,26,27 dampening synaptic potentials through the activation of Ca2+-activated potassium (SK) channels,21,28 synaptic plasticity in the hippocampus,29,30 shaping excitability and information processing, 31 and motor learning in the cerebellum. 32 CaV2.3 knockout mice display increased resistance to seizure induction, altered pain response, protection against the neurodegeneration produced in Parkinson's disease and deficits in hippocampal theta oscillation architecture.33–36 Therefore, CaV2.3 channels have attracted special interest due to their potential as therapeutic targets.22,25,37
In the hippocampus, in situ hybridization and immunohistochemical studies showed high mRNA and protein expression for CaV2.3 in both principal cells and interneurons.11,23,24 At the subcellular level, ultrastructural reports demonstrated that CaV2.3 is primarily located at postsynaptic sites, showing a larger density of gold particles on the surface of dendritic spines and shafts compared with the somata of CA1 pyramidal cells. 11 While discrete components of Ca2+ signaling have been studied in AD, such as L-type channels, 38 it remains unknown how CaV2.3 channels are altered at the surface of hippocampal neurons in Aβ pathology, or the hippocampal pathways affected. Thus, to address these questions, we aimed to determine the protein levels and distribution of CaV2.3 channels in the hippocampus of 5xFAD mice. We provide convincing evidence for a significant downregulation of CaV2.3 in an age-, sex-, brain region- and transgenic mouse-dependent manner. We also show a reduction in the density of postsynaptic CaV2.3 channels in specific dendritic compartments in CA1 pyramidal cells in 5xFAD mice. These findings are consistent with an involvement of CaV2.3 in the synaptic dysfunctions and memory deficits in this AD model.
Methods
Animals
Transgenic 5xFAD mice (RRID: MMRRC_034840-JAX) were obtained from the Jackson Laboratory (Bar Harbor, ME, USA) and expressed human APP and PSEN1 transgenes with five mutations. 5xFAD lines from a mixed C57BL/6 and SJL genetic background were maintained by crossing hemizygous transgenic mice with C57BL/6J breeders. For analysis, three ages were selected: (i) 1 month: no sign of pathology, (ii) 5 months: when deterioration of synaptic transmission, long-term potentiation (LTP), and spatial working memory begins, and (iii) 10 months: memory deficits with significant loss of synapses and extensive deposition of Aβ.39–41 Mice were genotyped for the APP and PS1 transgenes by polymerase chain reaction.
Transgenic APP/PS1 mice (RRID: IMSR_MMRRC:034832) were acquired from the Jackson Laboratory. These mice express the Mo/Hu APP695swe construct along with the exon-9-deleted variant of human presenilin 1 [Tg(APPswe, PSEN1dE9)85Dbo/Mmjax].42,43 For analysis, 12-month-old mice, characterized by the beginning of memory deficits, significant synapse loss, and extensive Aβ deposition.44,45
Transgenic P301S mice (RRID: IMSR_JAX:024841) were obtained from the Jackson Laboratory. These mice carry a mutant (P301S) human MAPT gene that encodes the T34-tau isoform (1N4R). We selected 10-month-old animals for analysis, as they are characterized by extensive accumulation of neurofibrillary tangles, difficulties in spatial learning, deficits of LTP, impaired memory, synaptic dysfunction, and neuronal loss. 46
Animals were kept at the Animal House Facility of the University of Castilla-La Mancha (Albacete, Spain). The care and handling of the animals followed Spanish (RD 1201/2015) and European Union regulations (86/609/EC), with protocols approved by the local Animal Care and Use Committee.
For western blots, qRT-PCR and histoblots, mice were subjected to deep anesthesia through an intraperitoneal injection of a ketamine/xylazine mixture (1:1; ketamine at 100 mg/kg and xylazine at 10 mg/kg). The brain was then extracted, and the hippocampus was dissected for western blots and qRT-PCR. The tissue was frozen in liquid nitrogen, followed by its storage at −80°C. To perform the histoblot technique, brains were cut on a cryostat (Leica CM1860UV, Barcelona, Spain) to obtain horizontal 10 μm-thick sections. To carry out the pre-embedding immunogold technique, the mice were anaesthetized with sodium pentobarbital (50 mg/kg, i.p.). Then, transcardial perfusion with 4% paraformaldehyde and 0.05% glutaraldehyde in 0.1 M phosphate buffer (PB) was performed.
Antibodies and chemicals
We used a guinea pig anti-R α-1E (CaV2.3) polyclonal antibody against an epitope corresponding to the amino acid residues 2069–2087 of the mouse Cav2.3 protein (Q61290). The characteristics and specificity of this antibody have been described previously. 11
Quantitative reverse transcription PCR (qRT-PCR)
Total RNA was extracted from the hippocampus of wild-type animals (n = 6) and 5xFAD mice at 10 months (n = 6). We used a RNeasy Minikit (Qiagen #74104) and RNase-free DNase I, following the manufacturer's instructions. Using TaqMan Reverse Transcription Reagents (Applied Biosystems #N8080234), purified RNA was used for the synthesis of cDNA. Gene expression analysis by quantitative RT-PCR (qRT-PCR) was performed in triplicate according to the Fast SYBR® Green Protocol with the Fast 7500 real-time PCR detection system (Applied Biosystems). The mRNA levels of mouse riboprotein P0 were used as an internal control. Forward and reverse primers used were 5′-GTCAACAGATCCCTGTTCATCT-3′ and 5′-GGCCAGGATCATGTACTCAAA-3′ for CACNA1E (CaV2.3), and 5′- AGAAACTGCTGCCTCACATC-3′ and 5′-GGCACAGTGACCTCACAC-3′ for RPLP0. Target mRNA in samples was calculated by the 2−ΔΔCT relative quantification methodology. 47
Immunoblots
CaV2.3 protein levels in the hippocampus from wild-type (n = 4) and 5xFAD mice (n = 4) were analyzed by immunoblotting, performed as described previously. 48 Very briefly, samples were homogenized in ice-cold HES buffer, pH 7.4 (10 mM HEPES, 50 mM sucrose and a protease inhibitor cocktail) (Thermo Scientific, Pierce, Rockford, USA). We loaded in twenty-five micrograms of membrane protein onto 7.5% sodium dodecyl sulphate polyacrylamide gels (SDS/PAGE). After the transfer of proteins to PVDF membranes, the immunolabelling step was carried out using affinity-purified anti-CaV2.3 antibodies (1:1000) and antibodies against anti-Na+/K+-ATPase (1:1000). Following visualization of protein bands with an enhanced chemiluminescence (ECL) blotting detection kit, blots were quantified by densitometry using a LAS4000 MINI (Fujifilm, Japan). The optical density of the protein bands was quantified using Image Lab™ Software (Version 6.0.1, Bio-Rad). To correct for any variations in protein loading, the density of each CaV2.3 band was normalized to the density of its corresponding Na+/K+-ATPase loading control band from the same blot. Data were then expressed as a percentage of the wild-type control group. Normalization of protein loading was performed using an antibody against the α1 subunit of Na+/K+-ATPase, an integral membrane protein. The suitability of this protein as a loading control under our experimental conditions was previously validated. The linearity of the antibody signal was confirmed across a range of 10–25 µg of total protein from membrane fractions. Furthermore, we verified by Western blot that the protein density levels of Na+/K+-ATPase showed no significant differences between wild-type and 5xFAD animals.
Histoblotting
The regional distribution of CaV2.3 channels in 5xFAD, APP/PS1 and P301S mouse brains was assessed using the histoblot technique, as described previously. 49 Briefly, horizontal cryostat sections (10 µm) were arranged on nitrocellulose membranes. After several washes, the blots were reacted with affinity-purified anti-CaV2.3 antibodies (0.5 mg ⁄mL), followed by alkaline phosphatase-conjugated anti-guinea pig IgG secondary antibodies. Membranes were processed in parallel using the same incubation time for each reagent, which allowed us to compare CaV2.3 protein levels in each animal model at the experimental ages. Using a desktop scanner (HP Scanjet 8300), we acquired digital images by scanning the nitrocellulose membranes. Digital images were acquired by scanning the nitrocellulose membranes. To ensure accurate quantification, all images were processed using the same equipment and methodology. This consistency allowed for valid comparisons of grayscale image intensity across different ages and brain regions on various days. The pixel density (measured in arbitrary units) of immunoreactivity was assessed using open circular cursors with a diameter of 0.10 mm. These cursors were placed in distinct brain regions identified in adjacent cresyl violet-stained sections. We applied background correction to eliminate potential discrepancies in optical densities that could arise across different sections. The average of eight background measures taken near the protein-containing areas of the immunostained nitrocellulose membranes was subtracted from the average pixel densities measured within the brain regions. After these background corrections, the average pixel density for an entire region was counted as one data point (‘n’) for each animal. Under these conditions, the labelling performed on different days yielded very consistent results. Image analysis and processing were performed using the Adobe Photoshop software (Adobe Systems, San Jose, CA, USA). 50
Immunoelectron microscopic approaches
Quantitative immunoelectron microscopic approaches were carried out using the pre-embedding immunogold method, as we described previously. 50 In short, sections were incubated in affinity-purified anti-CaV2.3 antibodies (3–5 μg⁄ mL), followed by incubation in goat anti-guinea pig IgG coupled to 1.4 nm gold (Nanoprobes Inc., Stony Brook, NY, USA). The hippocampal stratum radiatum of the CA1 field was cut at 70–90 nm using an ultramicrotome, and the resulting ultrathin sections were picked up on copper grids and counterstained. A JEOL-1400Flash electron microscope and sCMOS camera (Jeol Ltd, Tokyo, Japan) were used for ultrastructural analyses.
Quantitative approach and analysis of immunoelectron microscopy
The amount of CaV2.3 in neuronal compartments of CA1 pyramidal cells was calculated in the stratum radiatum of 10-month-old animals (n = 6 per genotype), as described previously. 50 Quantitative analysis was assessed in three different ways:
Density of CaV2.3 channels on the surface
Analysis of CaV2.3 immunogold labelling was assessed on main dendrites, oblique dendrites and dendritic spines, as well as on the extrasynaptic plasma membrane and active zone of axon terminals. By counting the immunoparticles identified in the neuronal compartments and accurately measuring their perimeter (ImageJ, NIH, Bethesda, MD, USA), we calculated the linear density (number of immunoparticles/µm) of CaV2.3 for wild-type and 5xFAD mice.
CaV2.3 channels close to excitatory synapses
After measuring the length of the plasma membrane of dendritic spines from the edge of the PSD, we then measured the position of gold particles as a function of distance from the PSD. The number of gold particles was expressed as relative frequency in bins of 60-nm membrane segments.
Percentage of CaV2.3 gold particles
To examine the occurrence of CaV2.3 channels, we conducted a meticulous count of immunoparticles observed in each reference area and their distribution within presynaptic compartments, namely extrasynaptic plasma membrane and active zone. Data was presented as a percentage of membrane-bound gold particles within each compartment.
Controls
To evaluate the specificity of the methods, we either omitted the primary antibodies or replaced them with 5% (v/v) normal serum. No specific labelling was detected when using these conditions.
Data analysis
Before performing data analysis, experiments were performed blinded to eliminate bias. Statistical analyses were carried out using GraphPad Prism (San Diego, CA, USA). Data are presented as mean ± SEM, and significance is defined as p < 0.05. Statistical evaluation of the histoblots utilized either parametric or nonparametric t-tests, based on the nature of the data. Holm-Sidak correction was applied for multiple comparisons, while the Mann-Whitney test was used as needed.
Results
Age-specific alteration in immunodetection of CaV2.3 in female 5xFAD mice
We first determined the brain distribution pattern of α1E (CaV2.3) in female 5xFAD mice and wild-type mice at 1, 5, and 10 months of age, employing a CaV2.3 antibody and the histoblot technique. In wild-type mice, CaV2.3 staining was widely detected throughout brain regions, showing strong protein levels in the caudate putamen, hippocampus, septum and cortex (Figure 1A, C, D, F). Moderate to weak staining was detected in the thalamus and cerebellum (Figure 1A, D). In 5xFAD mice, CaV2.3 levels were quantitatively very similar at 1 month (Figure 1C). However, a significant CaV2.3 downregulation was detected in the hippocampus and caudate putamen at 5 months (Figure 1F) and in the hippocampus, septum, caudate putamen, and cortex at 10 months (Figure 1I).
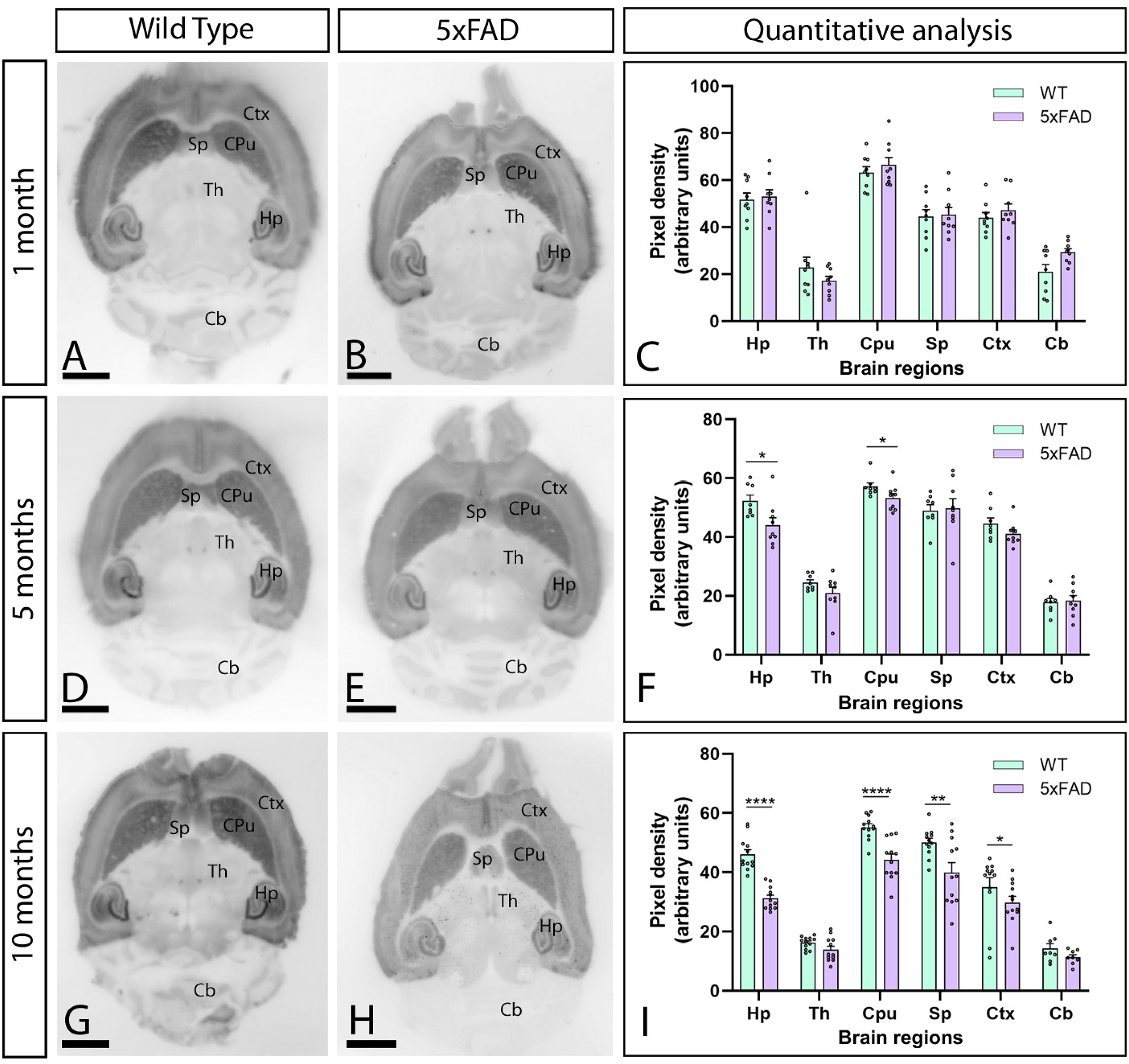
Age-dependent reduction of CaV2.3 protein level in the brain of female 5xFAD mice. (A-I) CaV2.3 protein level was visualized using histoblots of horizontal brain sections at 1, 5 and 10 months of age in female wild-type and 5xFAD mice. Densitometric analysis showed strong CaV2.3 detection in the caudate putamen (CPu), hippocampus (Hp), septum (Sp) and neocortex (Ctx), and weaker in the thalamus (Th), midbrain nuclei and cerebellum (Cb). Quantitative analysis showed no differences in CaV2.3 protein level in 5xFAD mice compared to age-matched wild-type controls at 1 month. However, a significant decrease was observed in the hippocampus and caudate putamen at 5 months and in the hippocampus, caudate putamen, septum and cortex at 10 months. Error bars indicate SEM. Scale bars: 0.25 cm.
We next analyzed the hippocampus and explored its laminar pattern of CaV2.3 labelling (Figure 2A–I). CaV2.3 was detected throughout all hippocampal subfields and layers of wild-type and 5xFAD mice but was particularly strong in CA1 and molecular layer (ml) of the dentate gyrus (DG) at 1, 5 and 10 months of age, while faint immunolabelling could be detected in dendritic layers of CA3 (Figure 2A–I). Both in wild-type and 5xFAD mice, the CaV2.3 protein level in the CA1 field was strong in the strata oriens (SO) and radiatum (SR) but moderate in the stratum lacunosum-moleculare (SLM) (Figure 2A–I). CaV2.3 level was moderate in the SO, stratum lucidum (SL), SR and SLM of the CA3 field (Figure 2A, D, G). In the DG, the CaV2.3 level was strongest in the ml but only moderate in the hilus (Figure 2A–I). Quantification of CaV2.3 staining showed unchanged hippocampal laminar staining pattern at 1 month (Figure 2C). However, the CaV2.3 protein content was significantly downregulated in a laminar-dependent manner in 5xFAD mice at 5 and 10 months (Figure 2F, I). CaV2.3 levels were downregulated in the SR and SLM of CA1 and ml of the DG at 5 months (Figure 2F) and in all subfields and layers of CA1, CA3, and DG (except the hilus) at 10 months of age (Figure 2I).
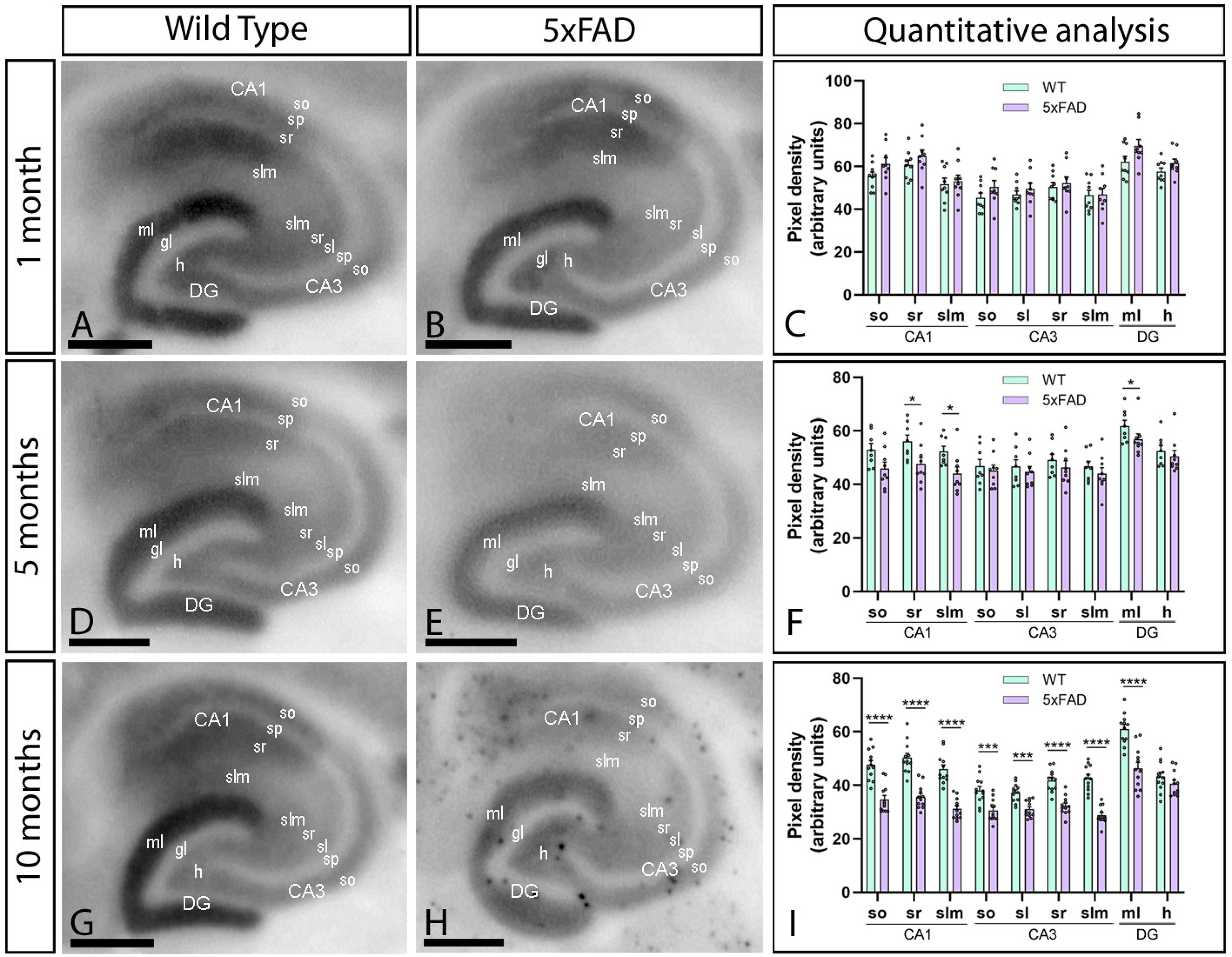
Age-dependent reduction in the laminar immunodetection of CaV2.3 in the hippocampus of female 5xFAD mice. (A-I) The laminar immunodetection of the CaV2.3 protein in the hippocampus was visualized using histoblots of horizontal brain sections at 1, 5 and 10 months of age in female wild-type and 5xFAD mice. Densitometric analysis showed strong CaV2.3 labelling in dendritic layers of the CA1 field and the molecular layer (ml) of the dentate gyrus (DG), while weaker labelling could be observed in dendritic layers of the CA3 field. Quantitative analysis showed no differences in CaV2.3 protein level in the hippocampal layers of 5xFAD mice compared to age-matched wild-type controls at 1 month. However, significant reductions were detected in the strata radiatum (sr) and lanucosum-moleculare (slm) of the CA1 field and the molecular layer (ml) of the DG at 5 months, and larger reduction in all dendritic layers of the hippocampus except the hilus (h) at 10 months. Error bars indicate SEM. so: stratum oriens; sp: stratum pyramidale; sl: stratum lucidum; gl: granule cell layer. Scale bars: 0.05 cm.
5xFAD mice exhibit decreased hippocampal levels of CaV2.3 protein but not mRNA
Next, we investigated protein levels of CaV2.3 in the hippocampus using western blots of membrane fractions from 10-month-old female wild-type and 5xFAD mice. The employment of the anti-CaV2.3 antibody showed a band with an estimated molecular mass of ∼250 kDa (Figure 3A). CaV2.3 protein levels were downregulated in 5xFAD compared to wild-type mice (Figure 3B), in agreement with histoblot data. Then, to test whether the downregulation of CaV2.3 protein involves changes in gene expression, we analyzed the mRNA expression profile from 10-month-old mice using qRT-PCR. We detected a tendency to reduced CaV2.3 mRNA expression levels, but this was not statistically significant (Figure 3C), suggesting that the decreased CaV2.3 protein level is not based on specific effects of the mutations acting at the level of Ca2+ channel gene expression. Together, these results indicate that changes in Ca2+ channel expression may occur at the cellular and posttranscriptional level.
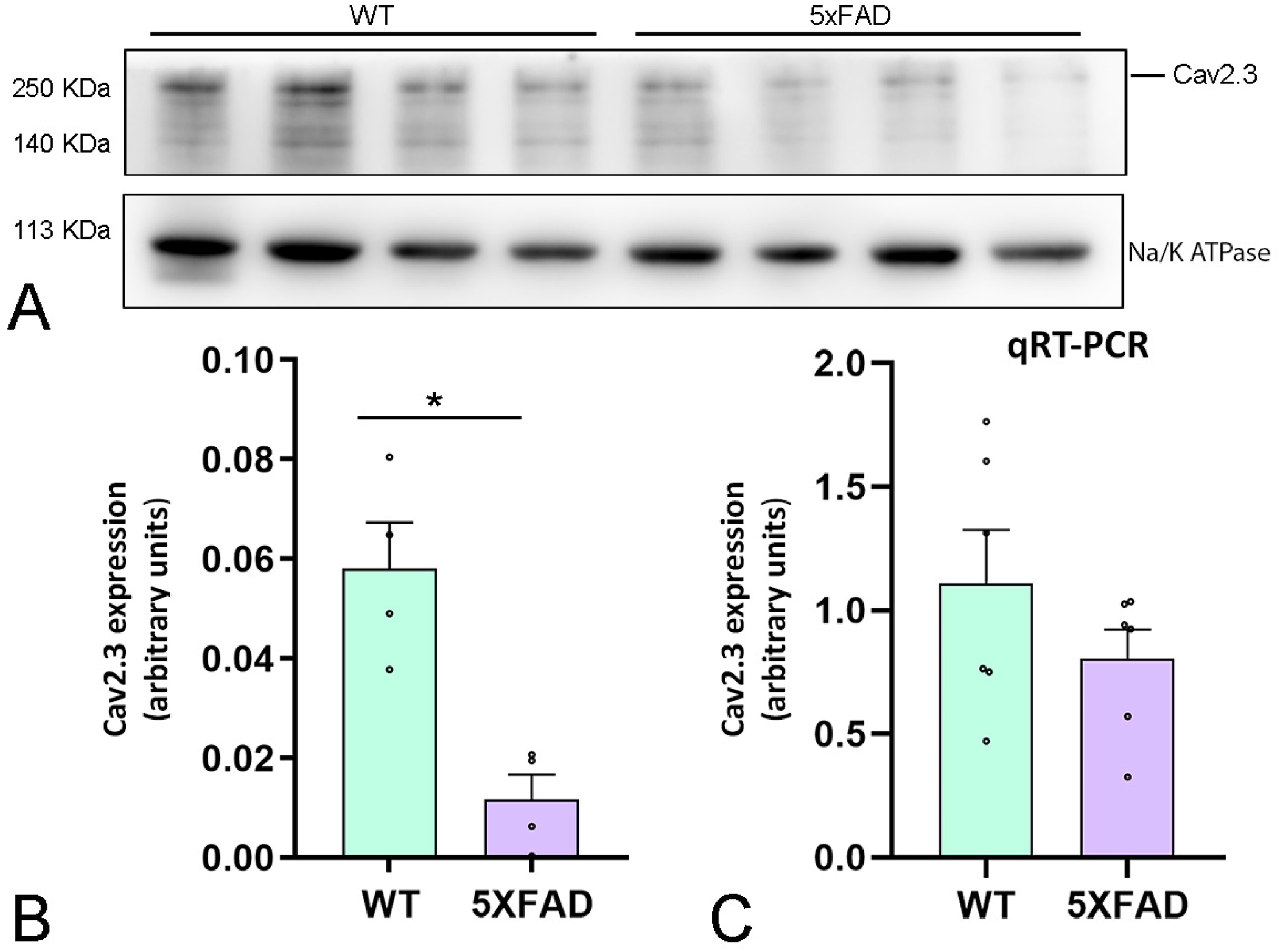
Immunoblot analysis of CaV2.3 protein level and profile of CaV2.3 mRNA in the hippocampus of female 5xFAD mice. (A) Representative western blot images showing CaV2.3 protein levels and the corresponding loading control, Na+/K+ ATPase, in total hippocampal extracts from wild-type (WT) and 5xFAD mice. All samples were run on the same gel to ensure comparability. The main immunoreactive band for CaV2.3 is detected at ∼250 kDa. (B) The quantitative data shown in the bar graph represent the densitometric values of CaV2.3 bands normalized to their respective Na+/K + -ATPase loading control. Data are presented as mean ± SEM, with individual points representing the average value from a single animal (n = 4 animals per group). Statistical analysis was performed using the Mann-Whitney test. *p = 0.0286. (C) The qRT-PCR measurements from six animals were averaged to compare the mRNA level for wild-type and 5xFAD mice at 10 months of age. Although we detected a trend toward a reduction in transcript expression, we detected no significant differences. Error bars indicate SEM.
Sex-specific alteration in the immunodetection of CaV2.3 channels in 5xFAD mice
Several studies performed in 5xFAD mice have shown that the brains of females display stronger memory deficits and Aβ accumulation compared to males.51,52 To investigate whether the expression patterns of CaV2.3 channels described for females differ in the brain of male 5xFAD mice, we performed conventional histoblotting experiments at 1, 5 and 10 months of age in males. The immunodetection patterns of CaV2.3 channels observed in the brain (Figure 4A, B, D, E, G, H) and hippocampus (Figure 5A, B, D, E, G, H) in males were virtually the same as those of females (Figures 1 and 2). However, protein densities comparison showed that CaV2.3 labelling was unaltered at 1 and 5 months (Figure 4C, F) but significantly downregulated in the hippocampus at 10 months in male 5xFAD mice (Figure 4I). In the hippocampus, the protein content of CaV2.3 was unaltered at 1 and 5 months (Figure 5C, F) and only downregulated in the SO and SR of the CA1 field, and in the SR of the CA3 field at 10 months (Figure 5I). In summary, the brain of female 5xFAD mice exhibit more profound differences in CaV2.3 levels than their male counterparts.
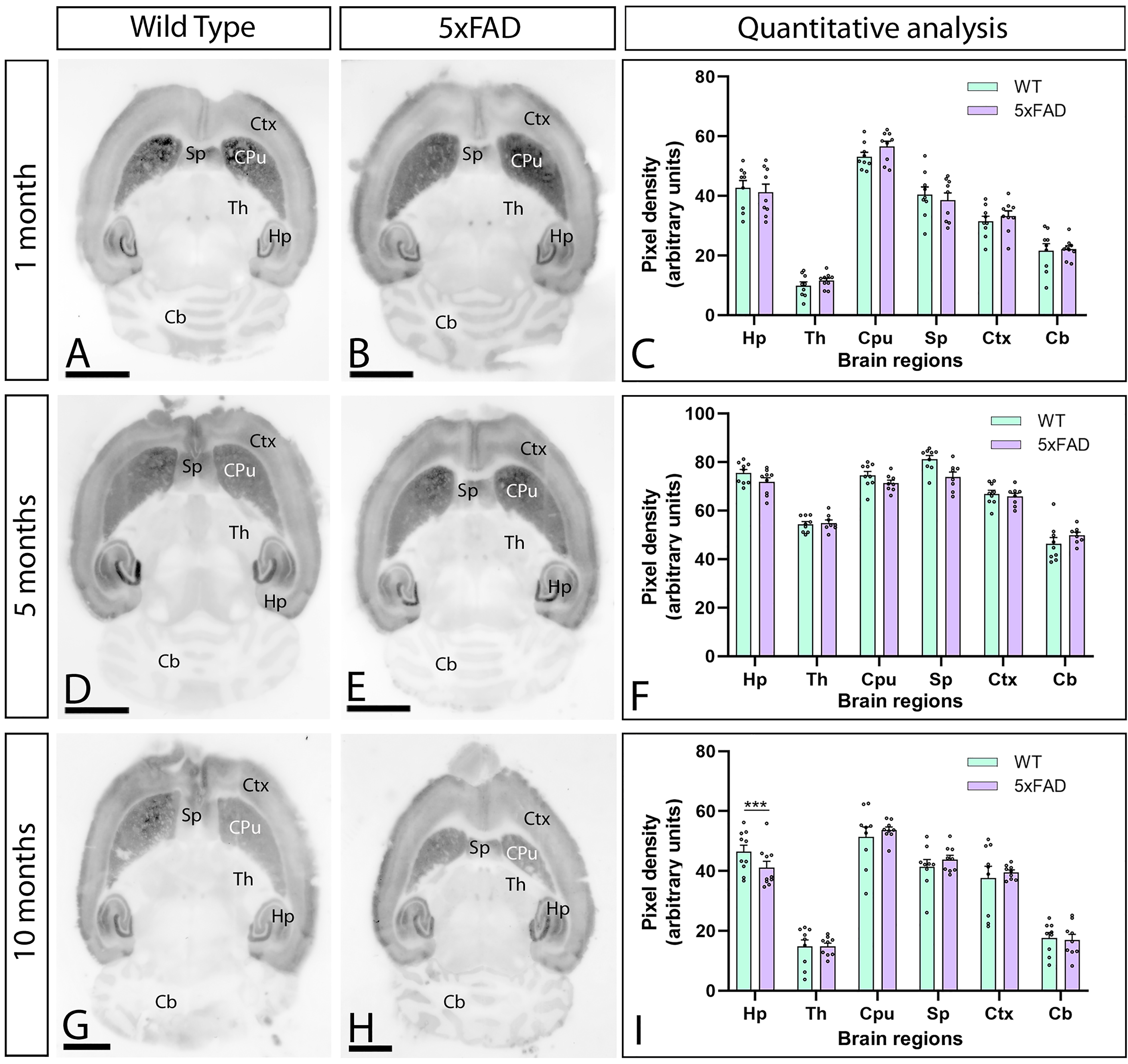
Brain immunodetection of CaV2.3 in male 5xFAD mice. (A-I) CaV2.3 protein level was visualized using histoblots of horizontal brain sections at 1, 5 and 10 months of age in male wild-type and 5xFAD mice. Densitometric analysis showed strong CaV2.3 protein level in the caudate putamen (CPu), hippocampus (Hp), septum (Sp) and neocortex (Ctx), and weaker in the thalamus (Th), midbrain nuclei and cerebellum (Cb). Quantitative analysis showed no differences in CaV2.3 protein level in 5xFAD mice compared to age-matched wild-type controls at 1 and 5 months. However, a significant decrease was observed in the hippocampus at 10 months. Error bars indicate SEM. Scale bars: 0.25 cm.
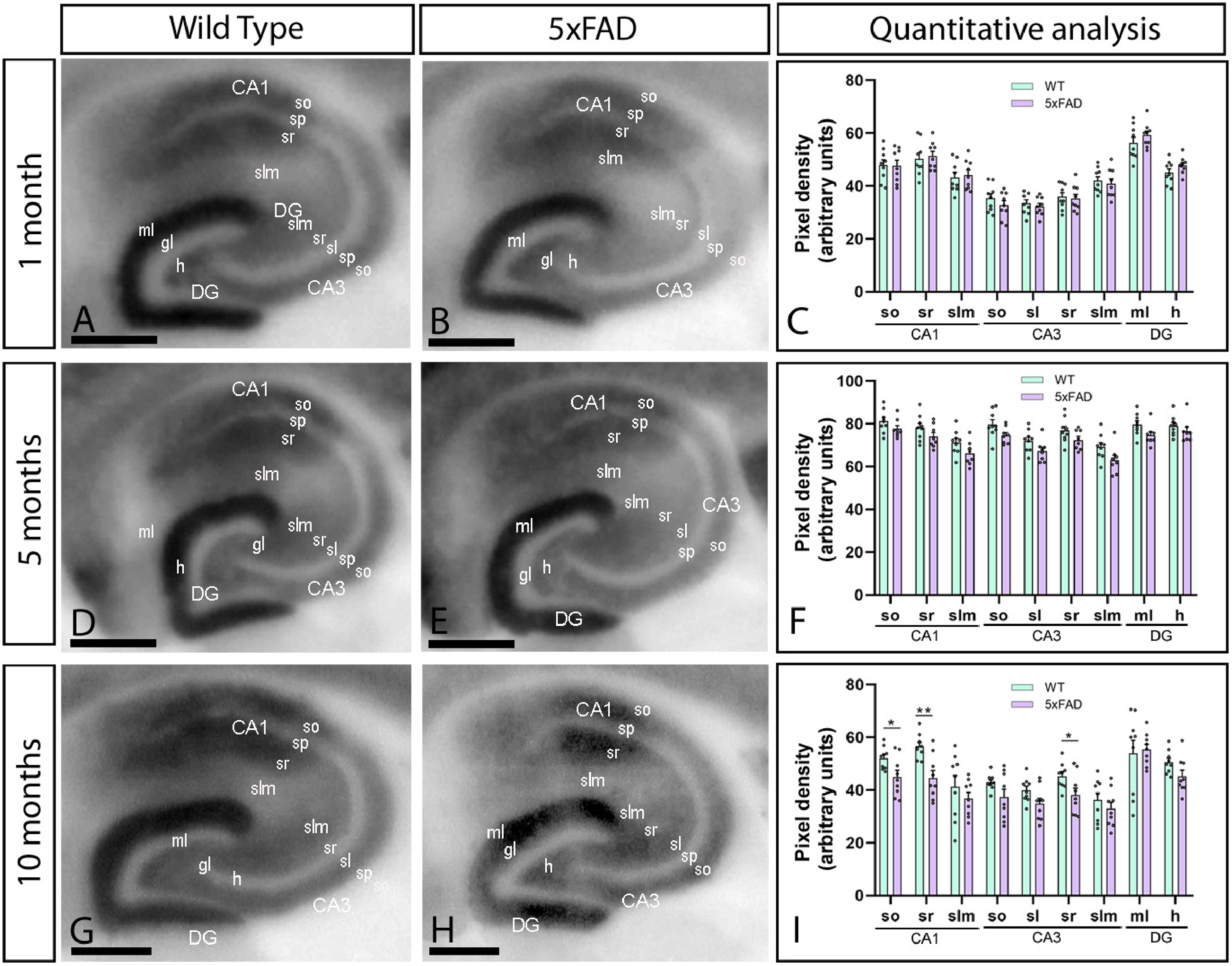
Hippocampal immunodetection of CaV2.3 in male 5xFAD mice. (A-F) The CaV2.3 protein level in the hippocampus was visualized using histoblots of horizontal brain sections at 1, 5 and 10 months of age in male wild-type and 5xFAD mice. Densitometric analysis showed strong CaV2.3 labelling in dendritic layers of the CA1 field and the molecular layer (ml) of the dentate gyrus (DG), while weaker labelling could be observed in dendritic layers of the CA3 field. No differences in CaV2.3 protein level in the hippocampal layers of 5xFAD mice compared to age-matched wild-type controls at 1 and 5 months. However, significant reductions were detected in the strata oriens (so) and radiatum (sr) of the CA1 field and the stratum radiatum of the CA3 field at 10 months. Error bars indicate SEM. DG: dentate gyrus; slm: stratum lacunosum-moleculare; sp: stratum pyramidale; sl: stratum lucidum; gl: granule cell layer; h: hilus. Scale bars: 0.05 cm.
The brain immunodetection of CaV2.3 channels is unchanged in other transgenic mice
To unravel if the downregulation of CaV2.3 protein levels found in 5xFAD mice could be observed in a different Aβ mouse model and a tau model, we investigated the brain of APP/PS1 mice at 12 months (Figure 6A–F) and P301S at 10 months (Figure 6G–L) applying the histoblot technique. As described above, CaV2.3 staining was strong in the caudate putamen, hippocampus, neocortex and septum, and moderate to weak in thalamus and cerebellum both in APP/PS1 mice and P301S mice (Figure 6A, B, G, H), showing no changes in this regional immunodetection pattern (Figure 6G, I). CaV2.3 labelling in the hippocampus of wild-type, APP/PS1 or P301S mice showed the same patterns as 5xFAD mice (Figure 6D, E, J, K). We then performed a quantitative analysis and confirmed that CaV2.3 protein levels remained unchanged between the different genotypes (Figure 6C, F, I, L).
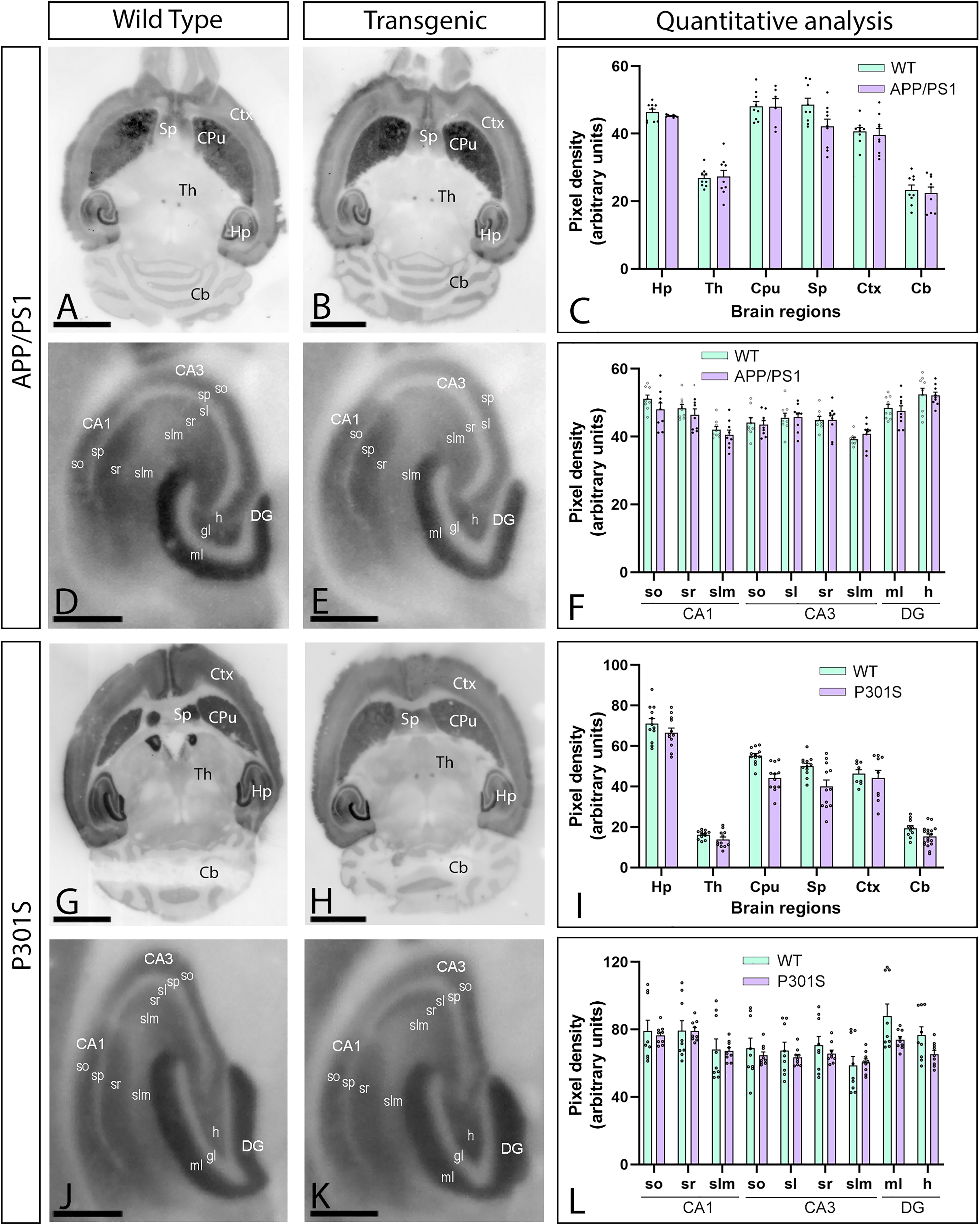
Brain immunodetection of CaV2.3 in APP/PS1 and P301S mice. (A-L) CaV2.3 protein level was visualized using histoblots of horizontal brain sections in 12-month-old wild type and APP/PS1 mice (A-F) and in 10-month-old wild type and P301S mice (G-L). In both transgenic mice, densitometric analysis showed strong CaV2.3 protein level in the caudate putamen (CPu), hippocampus (Hp), septum (Sp) and neocortex (Ctx), and weaker in the thalamus (Th) and cerebellum (Cb). In the hippocampus, labelling for CaV2.3 was strong in dendritic layers of the CA1 field and the molecular layer (ml) of the dentate gyrus (DG), and weaker in dendritic layers of the CA3 field. Quantitative analysis showed no differences in CaV2.3 expression in APP/PS1 and P301S mice compared to age-matched wild-type controls. Error bars indicate SEM. DG: dentate gyrus; so: stratum oriens; sr: stratum radiatum; slm: stratum lacunosum-moleculare; sp: stratum pyramidale; sl: stratum lucidum; gl: granule cell layer; h: hilus. Scale bars: A, B, G, H, 0.25 cm; D, E, J, K, 0.05 cm.
Hippocampal postsynaptic distribution of CaV2.3 is altered in 5xFAD mice
Previous studies demonstrated that CaV2.3 shows a compartmentalized distribution in hippocampal pyramidal cells. 11 To investigate how CaV2.3 channels are organized in primary apical dendrites, oblique dendrites, and spine heads following Aβ pathology, we explored the subcellular localization in CA1 from female wild-type and 5xFAD mice of 10 months (Figure 7). We performed this analysis in the stratum radiatum, a dendritic layer exhibiting strong labelling for CaV2.3, using a quantitative pre-embedding immunogold technique. CaV2.3 immunoparticles were localized intracellularly along the surface of postsynaptic and presynaptic compartments, according to the intracellular location of the epitope recognized by our specific antibody (Figure 7A–F). Both in wild-type and 5xFAD mice, CaV2.3 immunoparticles were preferentially localized postsynaptically, membrane-bound in dendritic shafts and spines of CA1 pyramidal cells in wild-type and 5xFAD mice (Figure 7A–F). Most immunoparticles for CaV2.3 present in spines in wild-type mice were found on the perisynaptic and extrasynaptic plasma membrane (Figure 7C). In 5xFAD mice, however, CaV2.3 immunoparticles were mainly observed extrasynaptically away from synaptic sites of spines (Figure 7F).
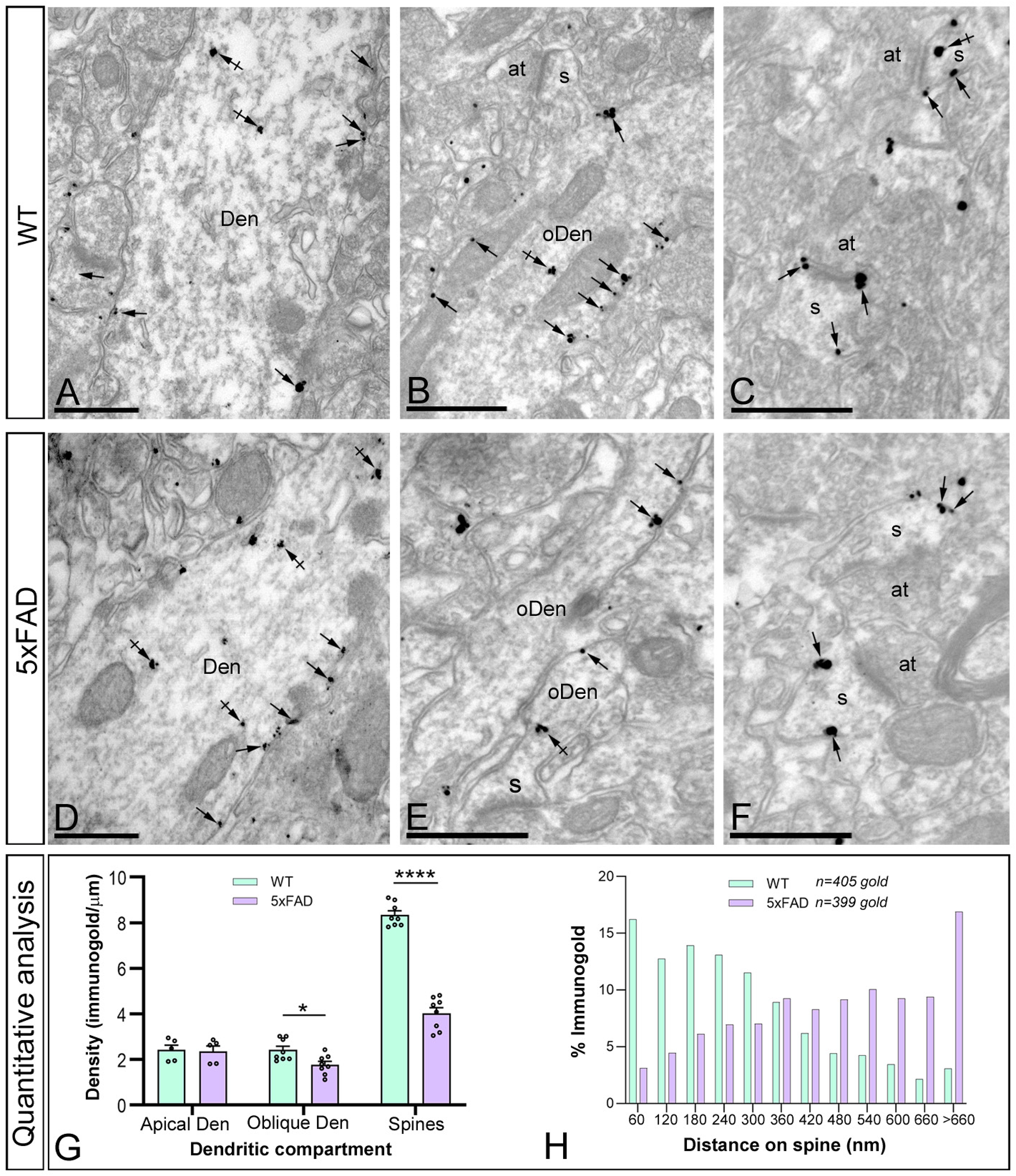
Changes in the postsynaptic localization of CaV2.3 in the hippocampus of 5xFAD mice. Electron micrographs showing immunoparticles for CaV2.3 in the stratum radiatum of the CA1 region at 10 months of age in female wild-type and 5xFAD mice, as detected using a pre-embedding immunogold technique. (A-F) In wild-type mice, immunoparticles for CaV2.3 were mostly located at the extrasynaptic membrane (arrows) of apical dendrites (Den), oblique dendrites (oDen) and dendritic spines (s) of CA1 pyramidal cells, and to a lesser extent at intracellular sites (crossed arrows). In 5xFAD mice, CaV2.3 immunoparticles in spines (s) were detected more frequently at some distances from the edge of synaptic sites. (G) Compartmentalization of CaV2.3 in CA1 pyramidal cells. Dendritic gradient of CaV2.3 along the surface of pyramidal cells showing that the density of immunoparticles is significantly decreased in oblique dendrites (oDen) and spines, but not in apical dendrites (Den). Error bars indicate SEM; **p < 0.002; ****p < 0.0001. (H) Histogram showing the lateral distances of immunoparticles for CaV2.3 measured from the nearest edge of the PSD in CA1 excitatory spines in wild-type (n = 405 gold particles) and 5xFAD mice (n = 399 gold particles). Immunoparticles were recorded in 60-nm-wide bins along the extrasynaptic plasma membrane and represented as the proportion of immunoparticles at a given distance from the edge of the synaptic specialization. The data show an enrichment of CaV2.3 in the proximity of asymmetrical synapses on dendritic spines in wild-type mice, and a redistribution towards further distances in 5xFAD mice. at: axon terminal. Scale bars: A-F, 500 nm.
To gain insight into the potential alteration of CaV2.3 compartmentalization in Aβ pathology, the average immunoparticle densities in dendritic compartments of CA1 pyramidal cells were calculated (Figure 7G). The analysis of immunogold distribution showed no differences in the CaV2.3 density in apical dendrites between wild type (2.43 ± 0.21 immunoparticles/µm) and 5xFAD mice (2.37 ± 0.23 immunoparticles/µm) (Figure 7G). However, we found significant differences in the CaV2.3 density in oblique dendrites (wild type: 2.42 ± 0.16 immunoparticles/µm; and 5xFAD: 1.77 ± 0.15 immunoparticles/µm; unpaired t-test, **p < 0.01) and spines (wild type: 8.35 ± 0.18 immunoparticles/µm; and 5xFAD: 4.03 ± 0.25 immunoparticles/µm; unpaired t-test, **p < 0.01). Our data shows that CaV2.3 channels are altered in some dendritic compartments in Aβ pathology.
Next, we investigated the distribution of CaV2.3 in spines in relation to the edge of excitatory synapses of CA1 pyramidal cell (Figure 7H). The position of CaV2.3 immunoparticles in wild type (n = 405 immunoparticles on 90 spines) and 5xFAD mice (n = 399 immunoparticles on 90 spines) was analyzed (Figure 7H). Around 76% of CaV2.3 immunoparticles were located between 0 and 300 nm from PSDs in dendritic spines in wild-type tissue. However, in 5xFAD mice, only 37% of CaV2.3 immunoparticles were located within 300 nm of PSDs and 73% away from synaptic sites (Figure 7H). In summary, the preferential localization of CaV2.3 around asymmetrical synapses observed in wild-type mice is altered in 5xFAD mice.
Unaltered presynaptic localization of CaV2.3 in 5xFAD mice
In contrast to the strong postsynaptic labelling, CaV2.3 immunoparticles were also detected presynaptically in axon terminals, where CaV2.3 was mainly found in the extrasynaptic plasma membrane and to a lesser extent over the active zone (Figure 8A–D). To investigate whether CaV2.3 changes at presynaptic sites in Aβ pathology, quantitative analysis was performed (Figure 8E, F). In the presynaptic compartments of wild-type mice, of the 247 particles counted, 29 (11.7%) particles were found along the active zone, and 218 (88.3%) particles were detected in the extrasynaptic plasma membrane (Figure 8E). These percentages were very similar in 5xFAD mice; of the 232 particles counted, 25 (10.8%) particles were found along the active zone, and 207 (89.2%) particles were observed extrasynaptically (Figure 8E). In addition, we found no differences in the density of CaV2.3 along the extrasynaptic plasma membrane of axon terminals (wild type: 1.27 ± 0.18 immunoparticles/µm; and 5xFAD mice: 1.23 ± 0.09 immunoparticles/µm) or the active zone (wild type: 0.96 ± 0.28 immunoparticles/µm; and 5xFAD mice: 0.86 ± 0.27 immunoparticles/µm) (Figure 8F). This approach demonstrated that CaV2.3 is not altered in presynaptic compartments in 5xFAD mice.
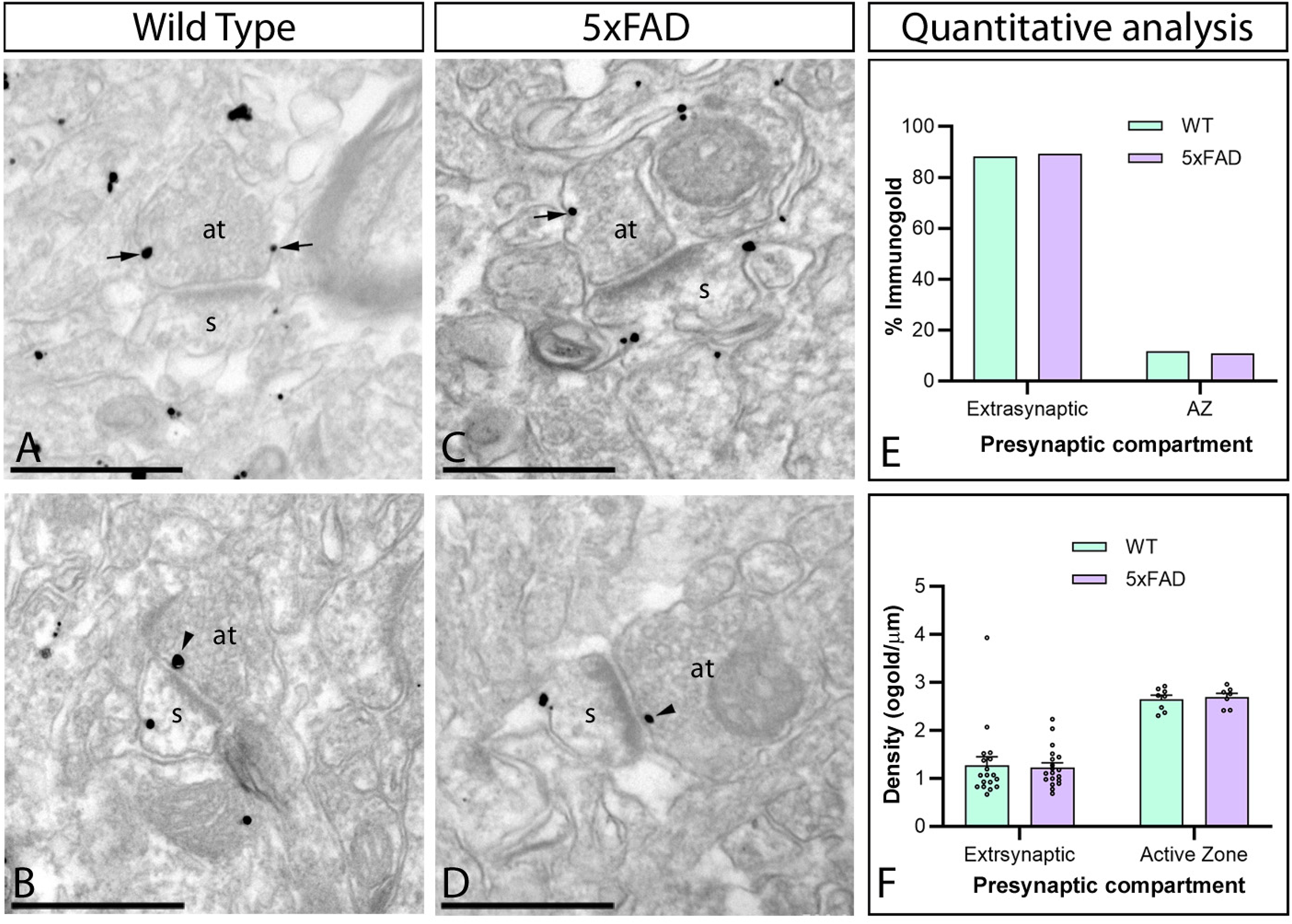
Unaltered presynaptic localization of CaV2.3 in the hippocampus of 5xFAD mice. Electron micrographs showing immunoparticles for CaV2.3 at presynaptic sites in the stratum radiatum of the CA1 region at 10 months of age in female wild-type and 5xFAD mice, as detected using a pre-embedding immunogold technique. (A-D) Both in wild-type and 5xFAD mice, immunoparticles for CaV2.3 present at presynaptic sites were primarily localized in the extrasynaptic plasma membrane (arrows) of axon terminals (at), establishing asymmetrical synapses with spines (s), and occasionally over the presynaptic active zone (arrowheads). (E) Histogram showing the percentage of immunoparticles CaV2.3 in presynaptic compartments. Most CaV2.3 immunoparticles were detected at extrasynaptic sites (88.3% in wild type and 89.2% in 5xFAD) compared to active zones (11.7% in wild type and 10.8% in 5xFAD). (F) Compartmentalization of CaV2.3 in axon terminals. No differences were detected in the density of immunoparticles at extrasynaptic sites and the active zone in wild-type compared to 5xFAD mice. Error bars indicate SEM. Scale bars: A-D: 200 nm.
Discussion
The entry of Ca2+ through voltage-gated CaV channels influences neuronal function. The high voltage-activated Ca2+ channel CaV2.3 is involved in epilepsy, metabolic stress and degeneration in Parkinson's disease,36,53 but has not yet been linked with AD. The present study is the first to provide a comprehensive investigation of the protein levels and molecular organization of the R-type pore-forming subunit CaV2.3 in female 5xFAD and how this is altered in Aβ pathology. Our findings indicate age- and region-specific downregulation of CaV2.3 channels in 5xFAD mice compared to controls, with the hippocampus as the most severely affected region. Western blot, histoblots, and immunogold analyses demonstrated a downregulation in the CaV2.3 levels in the hippocampus of female 5xFAD mice compared to wild-type. Furthermore, when analyzing the density and subcellular organization in the CA1 field, we showed that Cav2.3 channels are mainly located in dendritic compartments, particularly in spines close to synaptic sites, but are also present presynaptically. Using quantitative immunoelectron microscopy, we present convincing data showing that Aβ pathology impacts postsynaptic CaV2.3 channels in both oblique dendrites and dendritic spines of pyramidal cells, but not in apical dendrites or presynaptic axonal terminals. The observed reduction of postsynaptic CaV2.3 channels in specific dendritic regions in the hippocampus of 5xFAD mice might be involved in the synaptic dysfunction and memory deficits characteristic of this AD model.
Altered expression of CaV2.3 in the brain of 5xFAD mice
In situ hybridisation23,24,54 and immunohistochemical studies 11 reported that CaV2.3 is widely present throughout the brain, where the hippocampus shows strong expression. Consistent with these reports, we show here that the CaV2.3 protein was found in many regions of the brain where R-type currents are present, as demonstrated using electrophysiological techniques 53 and in all of them, R-type channels play a key role in regulating neuronal firing modes.21,55 Here, we report that in 5xFAD mice of 5 months, when deterioration of synaptic transmission begins, 41 there is a downregulation in regions like the hippocampus and caudate putamen. At 10 months, a stage characterized by widespread Aβ deposition and severe synapse loss, 41 this downregulation is more pronounced and extends to the septum. Therefore, there is an age- and region-dependent downregulation of CaV2.3 in the brain of female 5xFAD mice.
Focusing on the hippocampus, we found a wide distribution of CaV2.3 in female wild-type and 5xFAD mice at the three ages studied. Notably, immunoreactivity was pronounced in the dendritic layers of CA1 and the molecular layer of the DG, in comparison to the CA3 field. Studies from AD patients and animal models of AD have suggested that vulnerable neurons show synaptic dysfunction in AD. 56 In the hippocampus, the most vulnerable neurons to AD neurodegeneration are those located in the CA1 field, while neurons of the CA3 field and dentate gyrus are more resistant. Consistent with this selective vulnerability, one important finding of this study is that most of the alteration in CaV2.3 protein level found in 5xFAD at 5 months was detected in the SR and SLM of the CA1 field. However, CaV2.3 downregulation extended to all layers and subregions at 10 months, thus indicating an age- and laminar-dependent alteration of CaV2.3. Since the hippocampal CA1 field of 5xFAD mice does not exhibit neuronal loss at advanced ages, 57 the most plausible explanation for the observed downregulation is a specific molecular dysregulation of the CaV2.3 channel.
During AD development and progression, the concentration of Ca2+ is increased in the brains of patients with AD, as well as in the brains of transgenic mice. Accumulating evidence indicates that Aβ increases Ca2+ influx in synaptosomes and cultures of cortical neurons, 58 induces Ca2+ influx via ryanodine receptors 59 and promotes the entry of Ca2+ following activation of L- and T-type channels. 60 However, in our model of amyloidosis, the downregulation of CaV2.3 channels observed at 5 and 10 months suggests that Ca2+ entering through this channel is not primarily involved in the excitotoxicity driving neuronal death, but rather in different processes such as synaptic plasticity. 30 Some studies have reported in hippocampal neurons that neuronal activity modulates the recruitment of CaV2.3 channels through synaptic NMDA receptor signalling. 61 In addition, the absence of CaV2.3 function increases NMDA receptor-mediated spine Ca2+ influx and enhances AMPA receptor-mediated currents. 62
Although Aβ deposition is a pathological hallmark in both women and men suffering from AD, clinical and preclinical evidence show that women carry a higher risk of developing AD.63,64 There are also sex differences in animal models of AD, such as Tg2576, 65 3xTG-AD, 66 APP/PS1, 67 APP23, 68 AβPP/Tau, 69 APOE4 70 and 5xFAD 71 mice. Using the same technique, antibody and ages, we showed that the alteration of CaV2.3 occurs at different time points in female and male 5xFAD mice, and it is less severe in the latter. These differential expression profiles could dictate the relative contribution of calcium channels to the pathology of AD between the sexes. In other AD models, like in APP/PS1 mice, females demonstrate not only a higher prevalence of amyloid plaques and more significant spatial learning impairments compared to males, 72 but they also have elevated levels of Trem2, Clsd, and Ccl6, along with an increase in the activation of microglial morphology. 73 Furthermore, females exhibit a decrease in the expression of genes associated with neuronal plasticity. 74 In contrast, diminished activity of GIRK channels in hippocampal CA1 pyramidal neurons contributes to cognitive deficits in male mice that have an increased burden of Aβ. 75
We also found that the CaV2.3 protein level is transgenic mouse-dependent, as we did not detect any alteration in APP/PS1 and P301S mice, two models of AD that recapitulate distinct features of the disorder. Several studies have documented differential protein expression in transgenic mice. For example, the GIRK2 protein level is reduced in P301S mice, while it remains unchanged in APP/PS1 mice. 76 Additionally, AMPA receptors increase at specific excitatory synapses of 5xFAD mice, 41 but they decrease in both APP/PS1 and P301S mic.77,78 The reason why APP/PS1 and P301S mice do not exhibit downregulation of CaV2.3 in the hippocampus is unclear. This may be attributed to the distinct combinations of mutations in these models, with the 5xFAD model effectively recapitulating key pathological and behavioral characteristics of AD. Other contributing factors could include differences in gene expression levels, the ages of the transgenic mice, hormonal disparities and the promoters employed in each line. For instance, the Thy1 promoter was used in the 5xFAD model, 39 while the mouse prion protein (Prnp) promoter was utilized in the APP/PS1 and P301S mice.42,46 However, it has been reported that both Thy1 and Prnp promoters are sensitive to estrogen in the brain of transgenic mice.79,80 Thus, the downregulation of CaV2.3 in female 5xFAD mice may not be caused by estrogen but rather by other influencing factors that need further investigation.
Altered subcellular distribution of CaV2.3 in pyramidal cells of 5xFAD mice
The spatial organization of Ca2+ into distinct microdomains indicates that CaV channels are not distributed randomly along the neuronal surface. Today, we know that neuronal Ca2+ signaling is highly compartmentalized in the brain, indicating that specific subcellular localization of CaV channels is a key factor in regulating different neuronal processes. 81 Here, we provide direct evidence that CaV2.3 is mostly located postsynaptically in different dendritic compartments of CA1 pyramidal cells in the ageing hippocampus. Our ultrastructural findings agree with functional data showing that CaV2.3 channels play a synaptic role via shaping excitatory postsynaptic potentials in CA1 pyramidal cells. 31 Although CaV2.3 channels were found in both apical and oblique dendrites of those neurons, the amount of plasma membrane-bound immunoparticles was significantly higher in spines, consistent with a previous report using the same ultrastructural technique and antibody. 11
In CA1 pyramidal cells, apical dendrites give rise to oblique dendrites from which dendritic spines originate. 82 These secondary dendrites extend throughout the stratum radiatum, where they act as the primary targets of excitatory synapses, most of them originating from Schaffer collaterals. 82 In spines, the opening of CaV channels induces the accumulation of Ca2+ that is associated with back-propagating action potentials.83,84 The prevalence of CaV2.3 in spines implicates R-type channels in these processes and agrees with their involvement in modulating synaptic plasticity at the synapse between Schaffer collateral-CA1 pyramidal cells.29,30 Consistent with these findings, our ultrastructural analysis shows that the majority of the CaV2.3 immunoparticles can be found close to the PSD on dendritic spines, strongly suggesting that these channels are components of the protein networks forming the nano-environments of synaptic receptors and ion channels. NMDA receptors and CaV2.3 channels are the main origin of the Ca2+ entering into dendritic spines. Although these two proteins differ in their subcellular localization in hippocampal neurons, with NMDA receptors being present in excitatory synaptic specialization, 85 some studies have reported their functional association, showing that neuronal activity modulates the fast recruitment of CaV2.3 channels through synaptic NMDA receptor signalling. 61 Such synergism in the generation of calcium signals in dendritic spines is involved in long-term synaptic plasticity and memory formation. In addition to NMDA receptors, CaV2.3 channels act as the origin of calcium for small conductance calcium-activated K+ channels SK 21 and Kv4.2 86 in spines, and shape action potentials at somata through physical interaction with the large-conductance voltage- and calcium-activated K+ channel (BK). 31 Interestingly, we have previously demonstrated that SK and Kv4.2 channels show a similar distribution pattern in spines,87,88 suggesting that they are spatially close to fulfil functional association.
One significant finding of the quantitative ultrastructural analysis carried out in the present study is that Aβ pathology impacts R-type channels. We observed a significant reduction in the density of CaV2.3 immunoparticles along the surface of oblique dendrites and spines of pyramidal cells in the CA1 field, but not in their apical dendrites, in 5xFAD mice. This reduction was larger in spines than in oblique dendrites. In dendritic spines, alteration of CaV2.3 takes place in two different ways: (i) by a reduction in the density along the extrasynaptic plasma membrane; and (ii) by a redistribution of CaV2.3 from being concentrated at the edge of synapses to being located at more distant positions. This reduction close to synapses would influence the generation of calcium signals in association with NMDA receptors and in the amount of Ca2+ needed for SK, BK and Kv4.2 channels. In addition, given the role of CaV2.3 to attenuate the size of evoked excitatory postsynaptic potentials in hippocampal CA1 pyramidal neurons, 89 the reduction of these calcium channels in spines and dendrites might diminish their ability to perform this function, potentially leading to increased neuronal activity and excitability in 5xFAD mice. 90 Furthermore, the loss of CaV2.3 from spines and their mislocalization away from excitatory synaptic sites may further impair the capacity to modulate synaptic plasticity processes, which are known to be disrupted in 5xFAD mice. 91 While the functional relevance of our finding needs experimental testing, it is anticipated that such an impact on CaV2.3 channels may affect synaptic function in the hippocampus of 5xFAD mice.
Unaltered distribution of presynaptic CaV2.3 in 5xFAD mice
In contrast to the strong postsynaptic localization of CaV2.3 in the hippocampus, a weaker presynaptic distribution was also detected. However, in other brain regions, there is a much higher abundance of presynaptic CaV2.3, like in the interpeduncular nucleus, where axon terminals from the medial habenula show strong presynaptic immunoreactivity.11,19 Regardless of the brain region, presynaptic CaV2.3 was mainly observed at the extrasynaptic surface of axon terminals and occasionally over the presynaptic active zone. This distribution pattern differs from that of P/Q type (CaV2.1) channels, which are mainly located along active zones15,92 where they are involved in the regulation of neurotransmitter release. This suggests that CaV2.3 channels may play different functional roles, other than their known contribution to neurotransmission,20,26 in the presynaptic compartments. A significant finding from our immunoelectron microscopic study is that presynaptic CaV2.3 in axon terminals, likely from Schaffer collaterals, remains unchanged in 5xFAD mice, suggesting that Aβ pathology does not impact R-type channels presynaptically in the CA1 field.
It is important to acknowledge the limitations of our study to frame our conclusions properly. While our morphological and biochemical analyses robustly demonstrate a decrease in CaV2.3 protein levels and an alteration of the internalization machinery in 5xFAD females, it is not possible to establish a direct causal relationship with functional alterations in synaptic activity. To determine the functional impact of this protein dysregulation, electrophysiological studies, such as patch-clamp recordings to measure CaV2.3-mediated calcium currents, or calcium imaging techniques to assess its contribution to intracellular calcium homeostasis, would be necessary. Furthermore, we are aware that the function of an ion channel like CaV2.3 depends not only on its expression level but is also strongly regulated by post-translational modifications (e.g., phosphorylation, glycosylation, etc.) and by its interaction with other modulatory proteins. Therefore, a reduction in total protein expression does not necessarily imply a proportional decrease in channel activity at the plasma membrane. These considerations, far from diminishing our findings, open new and exciting avenues for future research aimed at elucidating the functional consequences of the morphological and molecular alterations we report here.
In summary, this study is the first comprehensive analysis of the subcellular distribution of CaV2.3 channels in vivo, specifically examining hippocampal CA1 pyramidal cells and how this localization is altered in Aβ pathology in 5xFAD mice using immunoelectron microscopy techniques. We have shown that CaV2.3 channels are mostly located postsynaptically in different dendritic compartments. They are especially abundant in spines, where they are concentrated close to the edge of excitatory synapses, consistent with their functional association with NMDA receptor signaling. Aβ pathology impacts CaV2.3 channels by reducing their density along the neuronal surface of oblique dendrites and spines and by moving away from synaptic sites. The dysfunction of calcium entry through CaV2.3 channels may impair postsynaptic mechanisms, affecting the activity of neural circuits in the hippocampus and possibly contributing to the pathogenesis seen in the 5xFAD mouse model of AD.
Footnotes
Acknowledgements
We thank Ms Diane Latawiec for the English revision of the manuscript. Funding sources were Spanish Ministerio de Economía y Competitividad and Junta de Comunidades de Castilla-La Mancha (Spain) and Deutsche Forschungsgemeinschaft (DFG).
ORCID iDs
Ethical considerations
All animal experimental procedures were performed in accordance with Spanish (RD 53/2013) and European Union regulations (2010/63/UE), and the protocols were approved by the local Animal Care and Use Committee.
Author contribution(s)
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Grants PID2021-125875OB-I00 and PID2024-155887OB-I00 funded by MCIN/AEI/ 10.13039/501100011033 and by “ERDF A way of making Europe” to RL. This work was also supported by Castilla-La Mancha Regional Government (JJCC) and the European Regional Development Find -SBPLY/24/180225/000007- and Universidad de Castilla-La Mancha (2025-GRIN-38362) to RL and Deutsche Forschungsgemeinschaft (DFG) CRC-TRR384 (Project-ID: 1059002414) to AK.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
All data used and/or analyzed during the current study are available from the corresponding author on reasonable request.
