Abstract
Sleep disturbance may increase risk of Alzheimer's disease and related dementias (ADRD). Acute sleep deprivation increases cerebrospinal fluid (CSF) levels of amyloid-β, but the impact of chronic insomnia remains unclear. We compared 13 adults aged 30–60 with Insomnia Disorder to matched good sleepers. After overnight polysomnography a lumbar puncture was performed to collect CSF for assays of ADRD biomarkers. Contrary to our hypotheses, we found no significant differences in objective sleep measures or any CSF biomarkers, except for lower levels of neurofilament light. These findings suggest that mild-to-moderate Insomnia Disorder may not increase ADRD biomarkers, contrasting with effects of acute sleep deprivation.
Introduction
Individuals with Alzheimer's disease and related dementias (ADRD) frequently experience insomnia that gradually worsens with increasing severity of the disorder. 1 A longstanding view has been that insomnia emerges as a consequence of dementia and is the result of progressive neuronal damage. However, there is growing evidence that the direction of causation may go both ways, with sleep disturbance potentially increasing vulnerability to dementia. Sleep disturbance often precedes and increases the risk for dementia by several years (e.g., 2 ). In a cross-sectional study of older adults, self-reported insomnia was found to be associated with greater beta amyloid burden as assessed with PET imaging. 3 One mechanism that could explain this relationship is the glymphatic system, through which potentially toxic byproducts of neural metabolism, such as amyloid-β (Aβ), are cleared from the nervous system. 4 Metabolic solutes are excreted into the interstitial fluid, which then flow through interstitial space and pass into cerebrospinal fluid (CSF) for clearance during sleep. 5 Acute sleep deprivation produces 30–50% increases CSF concentrations of Aβ.6,7 These studies support a growing consensus that disturbed sleep produces elevations in markers of ADRD and may increase vulnerability to the disorder. 8
While the mechanisms underlying glymphatic coupling to AD are still under investigation, there is increasing evidence implicating the orexin peptide. Orexin, also known as hypocretin, is produced in the hypothalamus and plays a crucial role in sleep regulation, energy balance, and other vital physiological functions. 9 There is emerging evidence that orexin is also involved in glymphatic clearance. In humans, high CSF concentration of orexin-A was associated with increased phosphorylated tau. 10 In an animal model of Alzheimer's disease, genetic deletion of orexin led to only moderate increases in sleep duration but strongly suppressed the formation of amyloid plaques. 11 In a second study of this mouse model, delivery of an orexin antagonist significantly inhibited amyloid plaque formation. 12 There is a need to better understand the role that orexin plays in the link between insomnia and ADRD. 13
Much of the literature in this area utilizes models of acute sleep disturbance, usually total sleep deprivation. Less is known about the impact of chronic sleep disturbance as it occurs in the general population, most notably in the form of chronic insomnia. Given that Aβ levels accumulate for up to 15–20 years prior to the onset of dementia, 14 improved understanding of the mechanisms through which deposition occurs could dramatically shift the landscape from treatment of dementia to one of prevention.
The goal of this pilot study was to examine CSF markers of dementia and orexin in individuals with chronic Insomnia Disorder and Good Sleeper controls. It was hypothesized that insomnia would be associated with higher levels of both dementia biomarkers and orexin.
Methods
Participants
All study participants were recruited from the Philadelphia metro area using flyers and online advertisements. Inclusion criteria for the insomnia group included a total score on the Insomnia Severity Index (ISI) > 14 15 and meeting DSM5 Diagnostic Criteria for Insomnia Disorder 16 including: dissatisfaction with sleep quantity or quality; adequate opportunity for sleep; clinically significant distress or impairment in functioning due to insomnia; frequency of occurrence at least 3 times per week for at least 3 months; and indication that the sleep disturbance was not better explained by a medical or psychiatric condition or based on the effects of a substance. Inclusion criteria for the Good Sleeper group included no lifetime history of Insomnia Disorder. Good sleepers were matched to the Insomnia Disorder group by age (within 5 years) and sex. Participants in both groups had to be between the ages of 30 and 60 years. Exclusion criteria included a diagnosis or evidence of sleep disorders other than insomnia, women who have been pregnant or lactating within the past six months, non-fluency in spoken or written English, current or past month shiftwork defined as working during the evening or night shift, and current use of medications or over-the-counter (OTC) products that impact sleep. Participants also could not show evidence of neurological abnormalities that could increase risks associated with lumbar puncture (e.g., papilledema, mass lesion, Chiari malformation) on neurologic exam. All participants provided written informed consent, and the protocol was approved by the Institutional Review Board.
Procedures
The study compared individuals with Insomnia Disorder to matched good sleepers. An initial screening visit was conducted either in person or virtually in which potential participants were provided with a description of the study and were provided with the informed consent form to read and sign. They then completed standard questionnaires consisting of the Insomnia Severity Index, Generalized Anxiety Disorder screener (GAD-7), 17 Pre-sleep Arousal Scale, 18 and the Morningness-Eveningness Questionnaire 19 to assess eligibility and for sample characterization. Eligible participants were then sent a home sleep apnea testing device (Zmachine, General Sleep Corporation) to screen for sleep disordered breathing.20,21 Subjects with significant sleep disordered breathing, defined as an apnea hypopnea index (AHI) > 15, were excluded from the remainder of the study and provided with information to review with their healthcare provider.
Participants who continue to be eligible following home sleep monitoring were scheduled for standard overnight polysomnography in the Penn Sleep Center. Electrode placements of FpZ, CZ, and OZ were be placed according to the International 10/20 system. 22 Bedtime and wakeup time were set to the individual's habitual patterns. Starting at 8:00 PM participants began fasting. In the morning study staff escorted participants to the Center for Human Phenomic Science (CHPS) at the Hospital of the University of Pennsylvania. Approximately 8:30 AM a lumbar puncture was performed by a neurologist. After the lumbar puncture participants remained in the CHPS unit and were monitored by nursing staff for at least 30 min to assess for any adverse effects of the procedure.
Sample collection, processing, and analyses
Lumbar punctures were conducted by a licensed neurologist, who first conducted a neurological assessment including fundoscopic exam in order to rule out any contraindications to the procedure such as a coagulopathy or central mass lesion. If no contraindications were found, lidocaine was injected using 25 g 1/2” needle for local anesthesia. A sprotte or standard needle was used to collect 25 mL of CSF. CSF was spun down at 2000×g for 10 min in a refrigerated centrifuge and transferred into polypropylene aliquot tubes. Within 60 min of CSF collection, aliquots were flash frozen in a combination of dry ice and ethanol, and then stored at -80°C. Participants were instructed to minimize activity and increase oral hydration for 24 h following the lumbar puncture to help reduce risk of headache.
CSF samples were assayed for levels of orexin A (OX-A), Aβ40, Aβ42, neurofilament light chain (NFL), and glial fibrillary acidic protein (GFAP). OX-A was measured using a fluorescent enzyme-linked immunosorbent assay (ELISA) (Phoenix Pharmaceuticals, Inc. kit #FEK-003-30). Samples and standards were pipetted into precoated wells in a 96 well plate format, 50 µL/well, mixed with 25 µL primary OXA antibody and incubated overnight at 4°C with shaking. 25 µL/well of biotinylated OXA was added to each well and incubated at room temperature (RT) for 1.5 h with shaking. The liquid was then discarded and the plate washed 4× with 1X assay buffer. A streptavidin-horseradish peroxidase complex (100 µl/well) was added and incubated at room temperature with shaking for 1 h, followed by repeat discarding of the liquid and washing the plate 4 times with 1X assay buffer. 100 µL/well substrate solution was added and incubated at room temperature, with shaking for 20 min. Finally, a stop solution was added, and the plate fluorescence was read with 325 nm excitation and 420 nm emission. The signal was inversely proportional to OXA. Data was analyzed using Graphpad Prism v10.2.2.
Measurements of Aβ40, Aβ42, GFAP and NFL were conducted with a Quanterix assay (Quanterix Simoa Neuro 4-Plex E kit for HD-X #103670). Calibrators were equilibrated and the samples were left at room temp for 1 h. RGP substrate was secured in a heated shaker at 30°C for 30 min at 800 rpm. Samples were diluted and pipetted 150 µL/well. Samples were diluted 1:200 or 1:400 and pipetted 150 µL/well. The shallow well polypropylene plate was sealed containing standards and samples with Quanterix X-Pierce XP-100 plate seal. The Neat protocol for Neuro 4-Plex E Advantage kit was run in Quanterix HD-X. A 4- parameter logistic curve fit, 1/y2 weighted for GFAP and NF-light and a 5-parameter logistic curve fit, 1/y2 weighted for Aβ1−40 and Aβ1–42 was generated after the assay was completed.
Statistical analyses
A series of general linear models was conducted to compare each of the sleep and CSF measures between the Insomnia Disorder and Good Sleeper groups, with age and sex as covariates. Given the pilot nature of this study no Type I error correction was performed.
Results
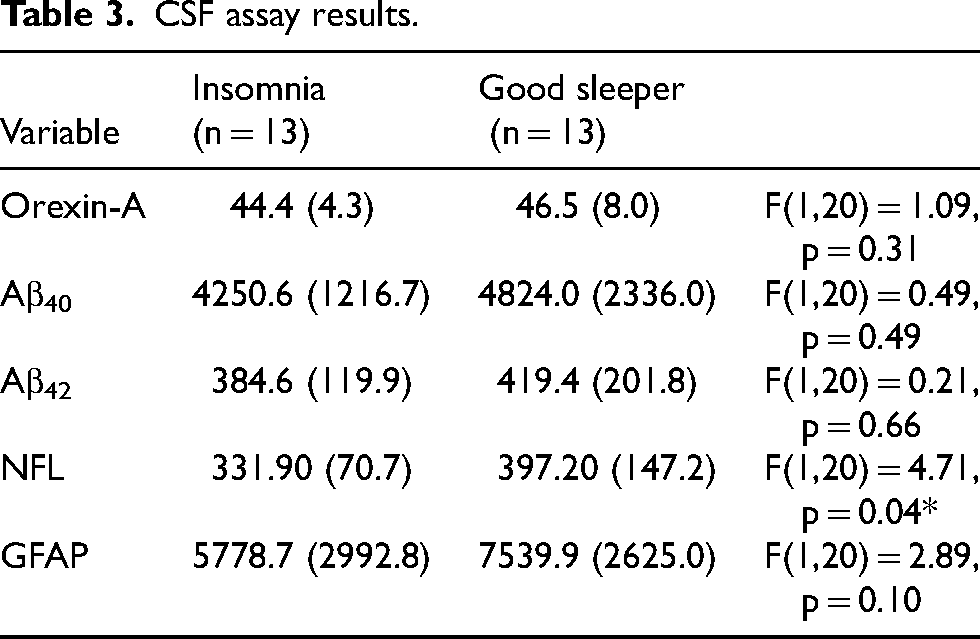
Thirteen individuals with chronic insomnia and thirteen matched good sleepers participated in the study. Sample characteristics are reported in Table 1. The insomnia group had significantly higher levels of insomnia severity, anxiety, and pre-sleep arousal, as expected. Contrary to expectations, there were no significant differences in any objective polysomnography (PSG) measures (Table 2). There were also no significant differences between groups on any CSF measures with the exception of NFL, which was significantly lower in individuals with insomnia compared to good sleepers (Table 3).
Demographic and baseline characteristics.
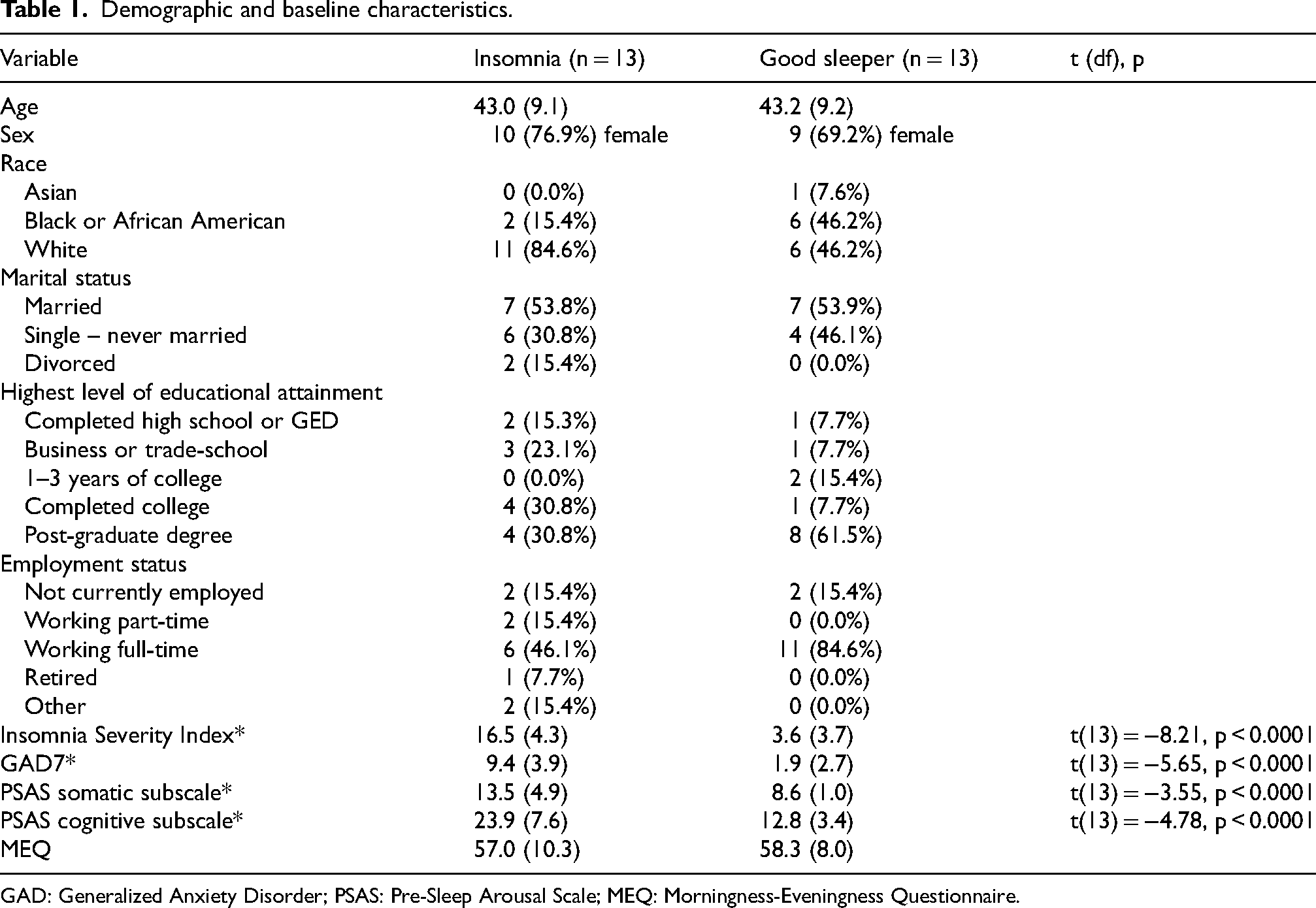
GAD: Generalized Anxiety Disorder; PSAS: Pre-Sleep Arousal Scale; MEQ: Morningness-Eveningness Questionnaire.
Objective sleep characteristics.

CSF assay results.
Discussion
This study examined whether Insomnia Disorder is associated with elevations in levels of orexin and ADRD biomarkers. Contrary to hypotheses there were no significant differences in any sleep variables compared to good sleepers, and only a single CSF biomarker was significantly different between groups. This study adds to the very mixed evidence from prior studies. In the Healthy Brain Cohort Project, higher CSF Aβ42 was associated with greater severity of insomnia on the ISI, higher actigraphic wakefulness after sleep onset and more awakenings, but not sleep onset latency. 23 There were no associations between sleep measures and tau or NFL. 23 In this study we did find an association with NFL, but it was in the opposite directions as would be expected to related to risk for ADRD. 24 The number of studies on insomnia and CSF ADRD markers is small, and there is not yet any clear pattern of findings.
There are several factors that likely contribute to the lack of associations in the present study as well as the mixture of prior results. First, the severity of insomnia in our sample was on the milder side, and it may be that greater severity and/or chronicity would be needed to find significant associations. This is also reflected in the absence of objectively poorer sleep on PSG. Second, insomnia is frequently comorbid with a range of other conditions, such as depression, anxiety, and chronic pain disorders. It is possible that associations reported in prior studies are attributable to these non-sleep factors. Third, there is significant heterogeneity across studies in age range. This study was intentional in focusing on middle-aged adults in order to avoid confounding by normal age-related increases in these biomarkers, even in individuals without ADRD symptoms, but the younger age group may be less likely to show elevations in these biomarkers and suggest that older samples might show stronger effects. Finally, this pilot study had a small sample size and was not powered to find small effects that should be investigated in larger studies.
It is also important to note that insomnia is not the same as acute, experimental sleep deprivation. The effects of acute sleep deprivation on CSF concentrations of Aβ are quite strong,6,7 contrary to the effects of insomnia. While much can be learned from sleep deprivation protocols, the extent to which they model real-world, chronic effects of insufficient sleep and sleep disorders is unknown.
These results suggest that Insomnia Disorder may not be associated with elevated risk for ADRD based on CSF biomarker levels, which is important information for the significant portion of the population who suffer from this common sleep disorder. However, these results add to a series of mixed findings across studies that makes it difficult to draw firm conclusions. These results highlight the need for larger, well-characterized samples across a diverse age range.
Footnotes
Ethical considerations
This study was approved by the Institutional Review Board of the University of Pennsylvania.
Consent to participate
All participants provided written informed consent to participate.
Author contribution(s)
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded with an investigator initiated grant from Merck & Co.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Authors TM and MH are employees of Merck & Co. There are no other competing interests to declare.
Data availability statement
Data are available in de-identified format upon request.
