Abstract
Background
Plasma phosphorylated tau217 (p-tau217) is a robust biomarker of Alzheimer's disease (AD) pathology. However, its full potential as a dynamic marker has still not been verified in very old persons, i.e., those with the highest incidence of AD.
Objective
To examine the cross-sectional and longitudinal associations between plasma p-tau217 concentration and cerebrospinal fluid (CSF) AD biomarkers. Further, to investigate the performance of p-tau217 as a predictor of amyloid status in a cohort of very old men.
Methods
CSF AD biomarkers were analyzed in thirty-five 89-year-old men. Amyloid-β (Aβ) positivity was defined according to CSF Aβ42 level. Plasma p-tau217 concentration was measured at the mean age of 82, 87, and 91. Incident dementia diagnoses in survivors were identified through medical records up to the age of 102.
Results
Plasma p-tau217 strongly correlated with CSF Aβ42 concentration in Aβ-positive (n = 16, Spearman ρ: rho = −0.63, p = 0.009) but not in Aβ-negative (n = 19, rho = 0.111, p = 0.652) men and predicted Aβ status (area under the curve, AUC 0.91). Plasma p-tau217 increased over ten years in the Aβ-positive group, while it remained unchanged in the negative group (p = 0.018).
Conclusions
Our findings indicate that plasma p-tau217 is a predictor of brain Aβ deposition also in very old individuals.
Introduction
Dementia is one of the major causes of disability and dependency among older people worldwide. 1 Alzheimer's disease (AD) is the most common dementia disorder, affecting millions of people and expecting to triple by 2050. 2 The strongest risk factor for AD is high age. Cognitive deterioration progresses slowly and the stage of mild cognitive impairment (MCI) often lasts for several years, preceding loss of functions (i.e., dementia). 3 The hallmarks of AD; brain accumulation of amyloid-β (Aβ) plaques and hyperphosphorylated tau (p-tau) neurofibrillary tangles, may be monitored by measuring the concentrations in cerebrospinal fluid (CSF) or by positron emission tomography (PET).4,5 Parallel to intracerebral aggregation, CSF Aβ42 declines over decades with the fastest rate in preclinical disease, 6 remaining stationary already by the time cognitive symptoms appear.4,7–9 Later on, from the stage of MCI, CSF p-tau181 increases before reaching its plateau. 10 Low concentrations of Aβ42 and high levels of p-tau181 in CSF strongly support a diagnosis of AD in patients with verified episodic memory decline. This holds true at the manifest dementia stage as well as in MCI.
PET is an expensive procedure and lumbar puncture may cause discomfort for the patient and it is not always possible to obtain CSF, due to anatomical reasons. Furthermore, these procedures are not available in a primary care setting where most elderly patients will present with cognitive symptoms. In primary care, the standard evaluation for diagnosing AD includes clinical examination, cognitive testing, and computed tomography (CT) scanning of the brain. Collection of blood is less invasive and less time-consuming than lumbar puncture (LP) or neuroimaging. Hence, establishing methods for analyzing AD plasma biomarkers is highly prioritized. Since it was developed, plasma p-tau has proven to be a robust biomarker of a multitude of features along the AD continuum. 11
Of various p-tau isoforms (p-tau181, p-tau217, p-tau231), tau phosphorylated at threonine 217 (p-tau217) is the most sensitive marker of multiple changes associated with AD. 12 Plasma p-tau217 has a high accuracy in discriminating Aβ-positive from Aβ-negative individuals; in identifying neuropathologically confirmed AD 13 ; and in differentiating AD from other neurodegenerative disorders.13,14 It predicts decline in cognitive test performance, accelerated cerebral atrophy and cerebral glucose hypometabolism, as well as conversion from preclinical and prodromal stages to manifest AD dementia.15–17 Further, it shows the greatest magnitude of change over time compared to the other isoforms.12,18 In brief, plasma p-tau217 increases from the stage of PET amyloid-positivity and tau-negativity before cognitive impairment, 19 and further on to AD MCI and mild AD dementia. 12 These dynamics have mainly been described in cross-sectional analyses of memory research cohorts, including participants within the AD spectrum and with a majority of them being below 75 years of age. Only a few papers have reported serial measurements of plasma p-tau21715,20–22or p-tau181 in the same individuals, and with follow-up periods up to approximately five years.23–27 Before it can become a clinical routine in primary care diagnostics, the potential of plasma p-tau217 as a dynamic biomarker needs to be verified also in community-based populations. 28 Not at least there is a need to achieve more knowledge about the dynamics in the oldest-old since several common conditions in high age may affect soluble p-tau. Cerebrovascular lesions, hypertension, diabetes and renal failure including other comorbidities 28 can influence the permeability of the blood-brain-barrier and may also affect the metabolism of p-tau and the relationship with amyloid fibrils. 29 Despite the numerous publications on plasma p-tau, to our knowledge, no previous study investigated its accuracy to predict amyloid status or the longitudinal changes in a very old cognitively unimpaired population.
In this study, we analyzed p-tau217 concentrations in plasma samples from an age- homogeneous cohort of non-demented elderly men who had also undergone LP. We included plasma sampled at three different time points over a ten-year period from the mean age of 82 to study the trajectory of plasma p-tau217, its correlations with standard CSF AD biomarkers and performance in predicting CSF amyloid status. Our secondary aim was to investigate if plasma p-tau217 correlated with performance in the Mini-Mental State Examination (MMSE) and with the degrees of global cortical atrophy (GCA) and medial temporal lobe atrophy (MTA) according to CT. Finally, we explored if plasma p-tau217 was associated with incident dementia.
Methods
The study individuals were participants in the Uppsala Longitudinal Study of Adult Men (ULSAM), a prospective cohort that started in 1970, described at ULSAM - Uppsala University (uu.se). All 50-year-old men born in 1920–1924 living in Uppsala, Sweden were invited to a health survey, initially focusing at identifying risk factors for cardiovascular disease. Eighty-two percent (n = 2322) participated in the first investigation at the age of 50. The participants were thereafter invited to examinations at ages approximately 60, 70, 77, 82 (U-5), 87 (U-6), and 91 (U-7) years. All participants were of European ancestry. Previous research on this cohort has demonstrated a strong concordance between educational level and socioeconomic status. 30 An experienced research nurse administered the MMSE 31 and collected plasma samples at U-5, U-6, and U-7.
Baseline for this study was U-6, which took place in 2008–2009 when the participants were aged 84–88 years, n = 352, as previously described.32,33 For this study we only included men without diagnosed dementia at baseline. One hundred and sixty participants were not on warfarin treatment and fit enough to be considered eligible for LP. They were invited to a sub study with CT of the brain and LP approximately two years later. Fifty-seven individuals agreed to participate and underwent CT and 52 of the 57 subjects accepted to undergo LP. Due to anatomical reasons, LP could only be completed in 35 subjects and these constitute our primary study group (Figure 1 and Table 1).
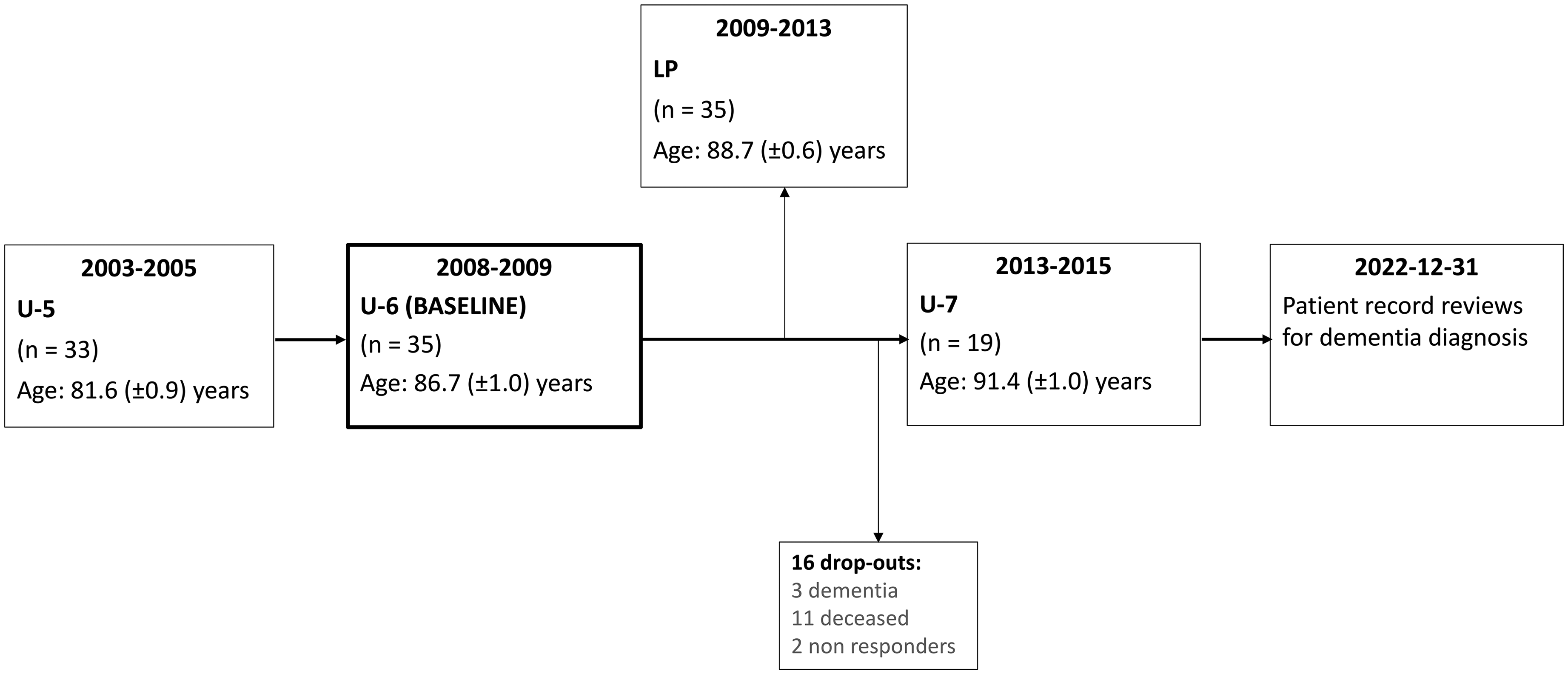
Flowchart of the participants in the CSF group. LP: lumbar puncture.
Characteristics of the participants.
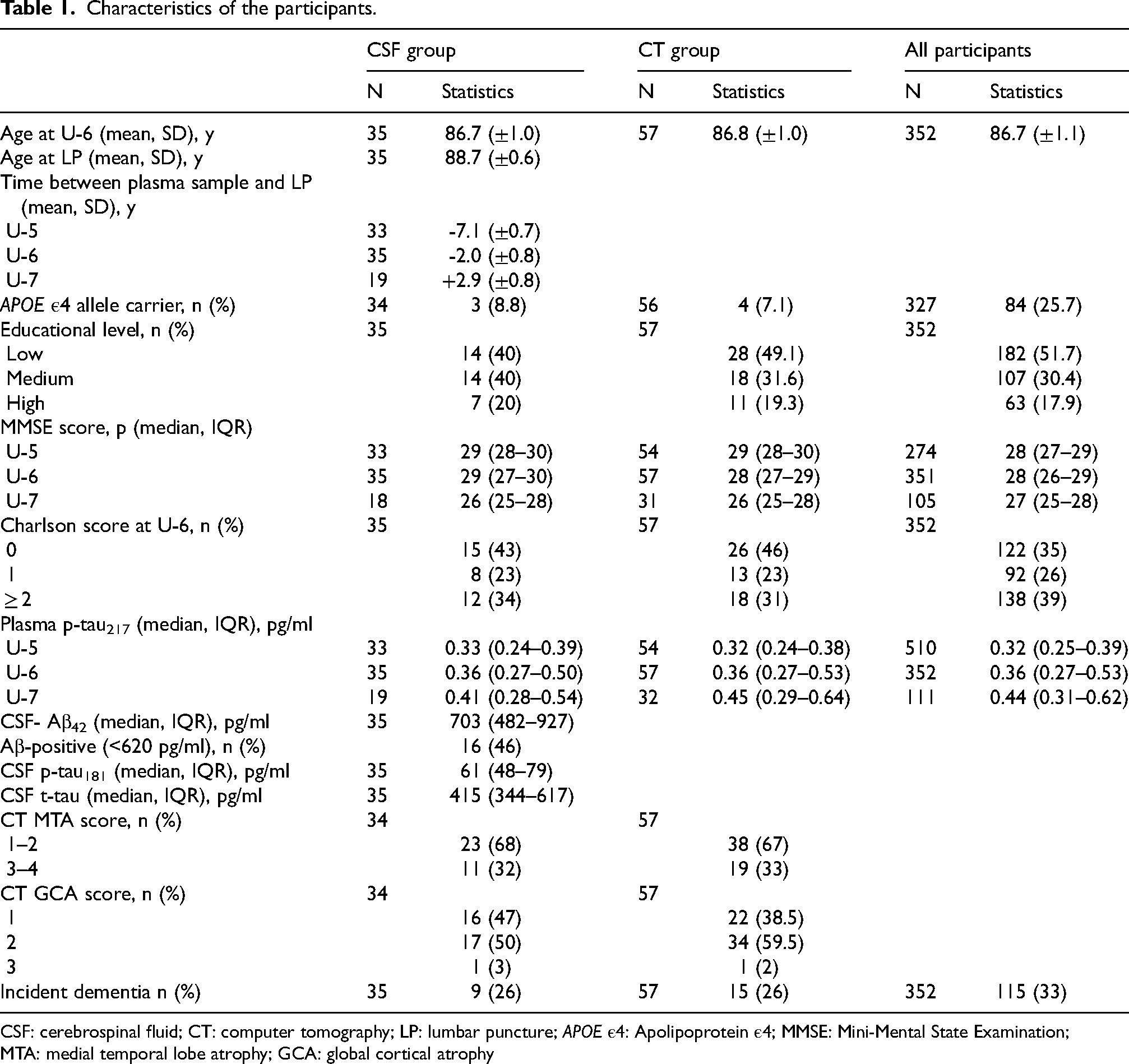
CSF: cerebrospinal fluid; CT: computer tomography; LP: lumbar puncture; APOE ε4: Apolipoprotein ε4; MMSE: Mini-Mental State Examination; MTA: medial temporal lobe atrophy; GCA: global cortical atrophy
Plasma p-tau217 concentrations were measured in archived samples from all participants in U-5 (n = 510), U-6 (n = 352), and U-7 (n = 125). In the CSF group, in samples from U-5 (n = 33), U-6 (n = 35), and U-7 (n = 19); and in the CT group in samples from U-6 (n = 57). The plasma samples were stored at −70°C until the analyses. The concentrations were measured at the Memory Research Unit, Lund University, using an immunoassay on a Meso Scale Discovery platform developed by Lilly Research Laboratories. Briefly, biotinylated-IBA493 was used as a capture antibody and SULFO-TAG-4G10-E2 (anti-Tau) as the detector and samples were diluted 1:2. The assay was calibrated with a synthetic p-tau217 peptide. All samples were analyzed in duplicates and by staff blinded to the clinical and imaging data. 0.6% (3/510) of p-tau217 values were below the lower detection limit of the assay (0.15 pg/ml).
The CT scans were performed at the Uppsala University Hospital, as previously described. 34 An 8-slice scanner (General Electric, Boston, MA) or a 64-slice scanner (Siemens Healthengineers, Erlangen, Germany) was used to acquire the images. The CT images were reformatted to axial, sagittal and coronal planes, with a slice thickness of 4 mm. All images were independently reviewed by two neuroradiologists, both blinded to cognitive status and CSF findings. The degree of the frontal atrophy was graded according to the Pasquier scale for GCA35,36 and the degree of MTA was graded using the Scheltens scale. 37 These scales are graded from 0 (no atrophy), to either 3 (Pasquier scale) or 4 (Scheltens scale), corresponding to the most severe degree of atrophy. In cases of disagreement between the neuroradiologists a consensus evaluation was made to reach the final scoring results. The CT scans were performed 1.7 (±0.9) years after U-6.
Lumbar puncture was performed by one investigator with the patient in a lying position. Twelve ml of CSF were collected in polypropylene tubes and samples with clear visual blood contamination were excluded. The samples were centrifuged and aliquoted in 1.5 ml polypropylene tubes, followed by storage at −70°C until the analyses. The lumbar punctures were performed 0.3 (±0.4) years after the CT scans (Figure 1).
The concentrations of Aβ42, t-tau, and p-tau181 in CSF were analyzed at the Clinical Neurochemistry Laboratory, University of Gothenburg, Mölndal, Sweden. The analyses were performed by board-certified laboratory technicians, who were blinded to clinical data, using sandwich ELISA (INNOTEST, Fujirebio, Ghent, Belgium) and standardized procedures accredited by the Swedish Board of Accreditation and Conformity Assessment. Aβ positivity was defined as a CSF Aβ42 level ≤620 pg/ml as defined by the laboratory's established clinical reference ranges.
Dementia diagnoses were assigned by two independent, experienced geriatricians, without information of the CSF biomarkers. 38 Follow-up diagnostics were made using all data available in medical records from Uppsala University Hospital, primary care, and nursing homes in Uppsala County until December 31, 2022, i.e., up to age of 102 in survivors. In case of disagreement, a third experienced geriatrician reviewed the case, and the diagnosis was determined by a majority decision.
Dementia was defined according to the criteria from DSM-IV, 39 in brief a persisting cognitive deterioration severe enough to interfere with activities of daily life, with other somatic or psychiatric disorders ruled out as explanations. AD was diagnosed according to the National Institute of Neurological and Communicative Diseases and Stroke and the Alzheimer's disease and Related Disorders Association (NINCDS-ADRDA) criteria, 40 including findings from the CT scans. Cases of dementia without neuroimaging and without sufficient clinical details in the medical records to set a specific dementia subtype diagnosis were classified as unspecified dementia.
Genotyping for apolipoprotein E (APOE) by minisequencing 41 was performed in 327 of the 352 participants at U-6. The National Patient Registry provided information on in-patient care before baseline at U-6, and this information was used to calculate the Charlson Comorbidity Index.42,43 The study was approved by the local ethical committee (Reference number: Dnr 02-605, Dnr 2007/338 and Dnr 2013/350), and all participants gave their written informed consent.
Statistics
Distribution or normality of variables were examined by visual assessment of histogram and standard error of skewness.
Spearman's correlation was used to calculate the correlations between plasma p-tau217 and CSF biomarkers, CT findings and MMSE, respectively. Two-tailed values of p < 0.05 were considered statistically significant. A mixed model was used to calculate the differences in plasma p-tau217 trajectory over time with respect to Aβ-status. Due to the small group sizes, it was not possible to include random intercepts in the mixed model. To examine the discriminative performance of plasma p-tau217 for predicting Aβ status we used the area under the receiver operating characteristic curve (ROC).
The Mann-Whitney U test was used to compare levels of plasma p-tau217 and CSF Aβ42 between participants with and without incident dementia, as the variables were not normally distributed.
To compare the age between non-participants (the ones with plasma samples who were not included in the CT or CSF group (n = 295)) and the participants T-test was used. Non-parametric analyses, including Mann-Whitney U and Chi-Square test, were used to compare education level, Charlson score and APOE ε4-carriership.
All statistical analysis were performed using IBM SPSS Statistics (ver.28.0.1.0 for Windows; IBM Corporation, Armonk, NY, USA)
Results
Characteristics of the participants in the study are shown in Table 1. At baseline (U-6), the mean (SD) age of the 35 participants in the CSF group was 86.7 (±1.0) years and 88.7 (±0.6) years at the time for LP. We found no significant age differences at plasma samplings, educational level or baseline Charlson score between the CSF group and all U-6 participants.
Only nine percent (3/34) of the CSF group were APOE ε4 allele carriers, which was significantly lower in comparison to 26% carriers in the total U-6 sample (p < 0.05). Sixteen participants were defined as Aβ-positive and 19 as Aβ-negative. The MMSE score distribution at baseline were within the normal range (median = 29 p, IQR = 27–30 p). Nineteen men took part in U-7; of the remaining sixteen, eleven were deceased, three were diagnosed with dementia and two did not respond.
There were no significant differences in plasma p-tau217 concentration in the CSF group compared with the whole sample at all three examinations. Plasma p-tau217 concentrations were in strong agreement across the different sampling ages (U-5 versus U-6: Spearman ρ: rho = 0.81, p < 0.001; U-6 versus U-7: Spearman ρ: rho = 0.82, p < 0.001).
Figure 2a-c shows the strong inverse correlations between CSF Aβ42 and plasma p-tau217 concentrations at U-5, U-6, and U-7 (Spearman ρ: rho = −0.59, p < 0.001, rho = −0.69, p < 0.001 and rho = −0.67, p = 0.002). In the Aβ-positive group, cross-sectionally, CSF Aβ42 inversely correlated with plasma p-tau217 at U-5 (n = 16, Spearman ρ: rho −0.55 p = 0.026), U-6 (n = 16, Spearman ρ: rho −0.63 p = 0.009) but not at U-7 (n = 8, Spearman ρ: rho −0.24 p = 0.57). In the Aβ-negative group there was no significant correlation between plasma p-tau217 and CSF Aβ42 at any of the examinations U-5 (n = 17, Spearman ρ: rho −0.0,76 p = 0.772), U-6 (n = 19, Spearman ρ: rho 0.111 p = 0.652) and U-7 (n = 11, Spearman ρ: rho −0.20 p = 0.555).
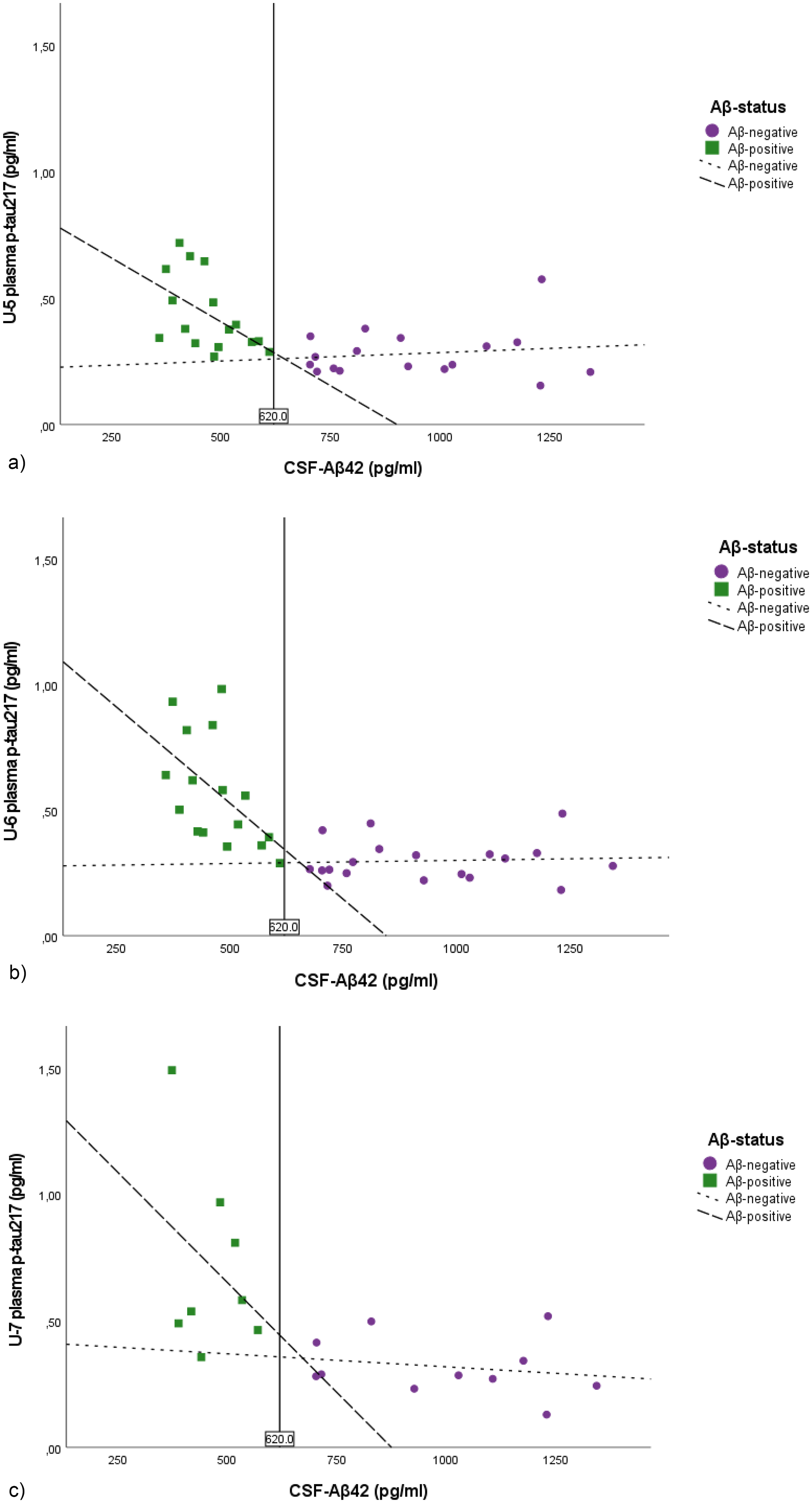
Scatter plots of CSF Aβ42 and plasma p-tau217 at the different examinations. Figure 2(a)
Figure 3 shows the correlation of plasma p-tau217 at baseline with CSF p-tau181 concentrations. The correlations of plasma p-tau217 with CSF p-tau181 concentration were of borderline significance at U-5 and U-6 (Spearman ρ: rho = 0.33, p = 0.058; rho = 0.33, p = 0.051) but did not exist at U-7 (Spearman ρ: rho = 0.20, p = 0.412), (Supplemental Figure 1a-(c)). The corresponding figures for Aβ-positive subjects were: U-5; n = 16, (Spearman ρ: rho = 0.39 p = 0.131), U-6; n = 16 (Spearman ρ: rho = 0.41, p = 0.116) and U-7; n = 8 (Spearman ρ: rho = 0.05, p = 0.911).
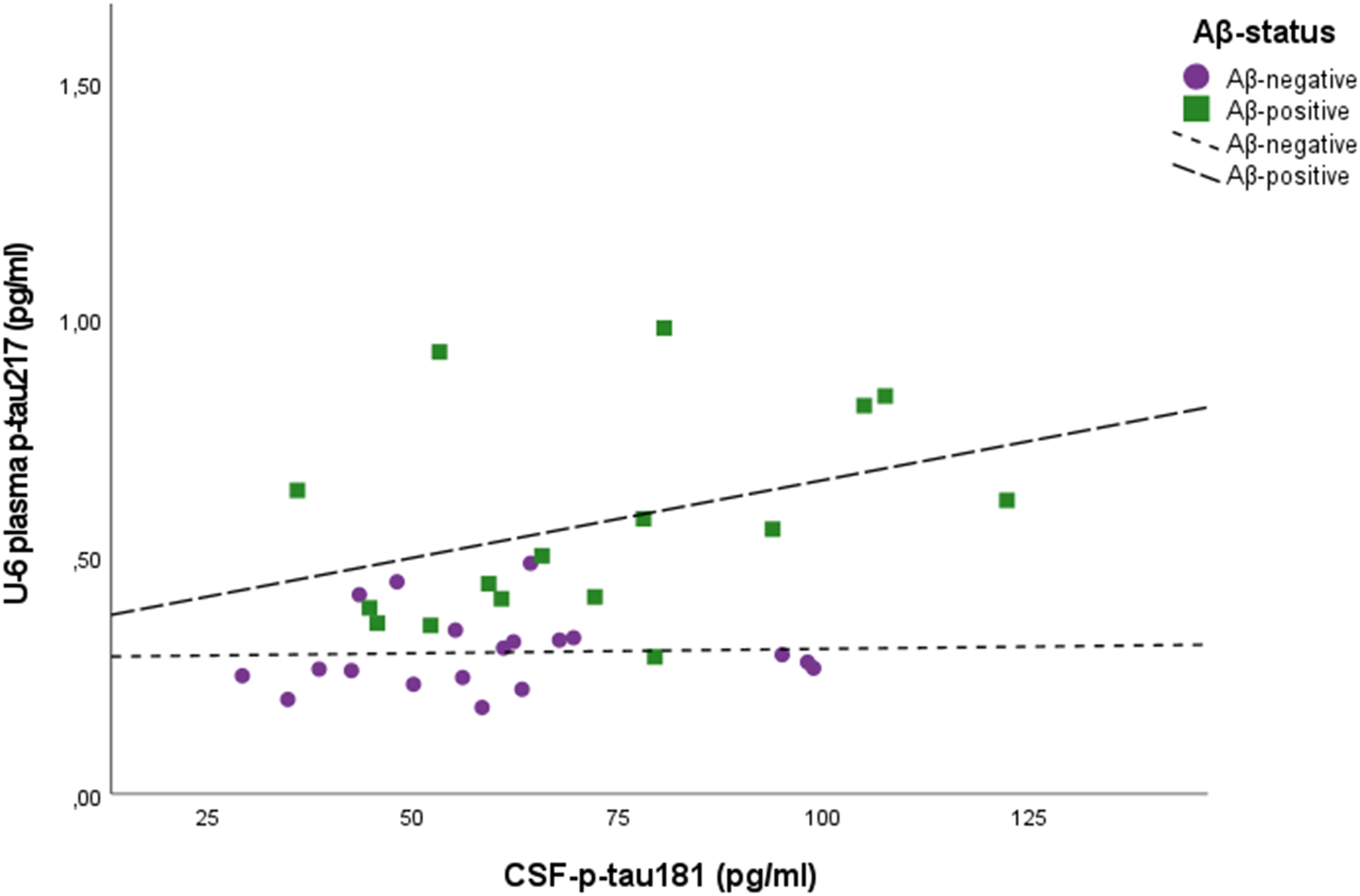
Scatter plots of CSF p-tau181 and plasma p-tau217 two years before CSF sampling (U-6, baseline).
Plasma p-tau217 concentration did not correlate with CSF total tau; at U-5, U-6, and U-7, respectively (Spearman ρ: rho = 0.24, p = 0.178; rho = 0.29, p = 0.091, and rho = 0.225, p = 0.335).
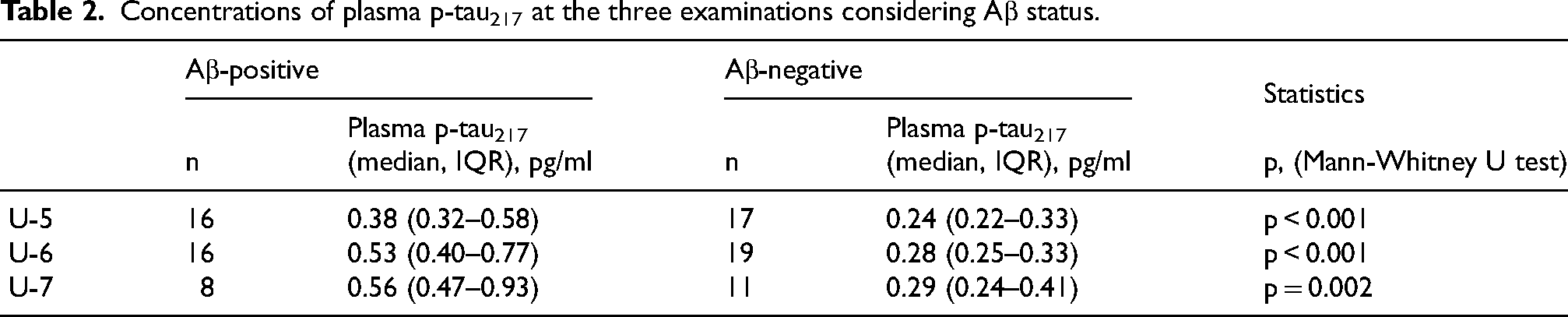
Table 2 shows that median plasma p-tau217 concentrations differed cross sectionally by CSF Aβ status at all three examinations. Figure 4 illustrates the longitudinal trajectory of plasma p-tau217, which differed significantly by CSF Aβ status (p = 0.018). There was an increasing trend in the Aβ-positive group (B = 0.137, 95% CI = 0.039–0.235, p = 0.007), whereas levels remained unchanged over time in the Aβ-negative group (p = 0.302).
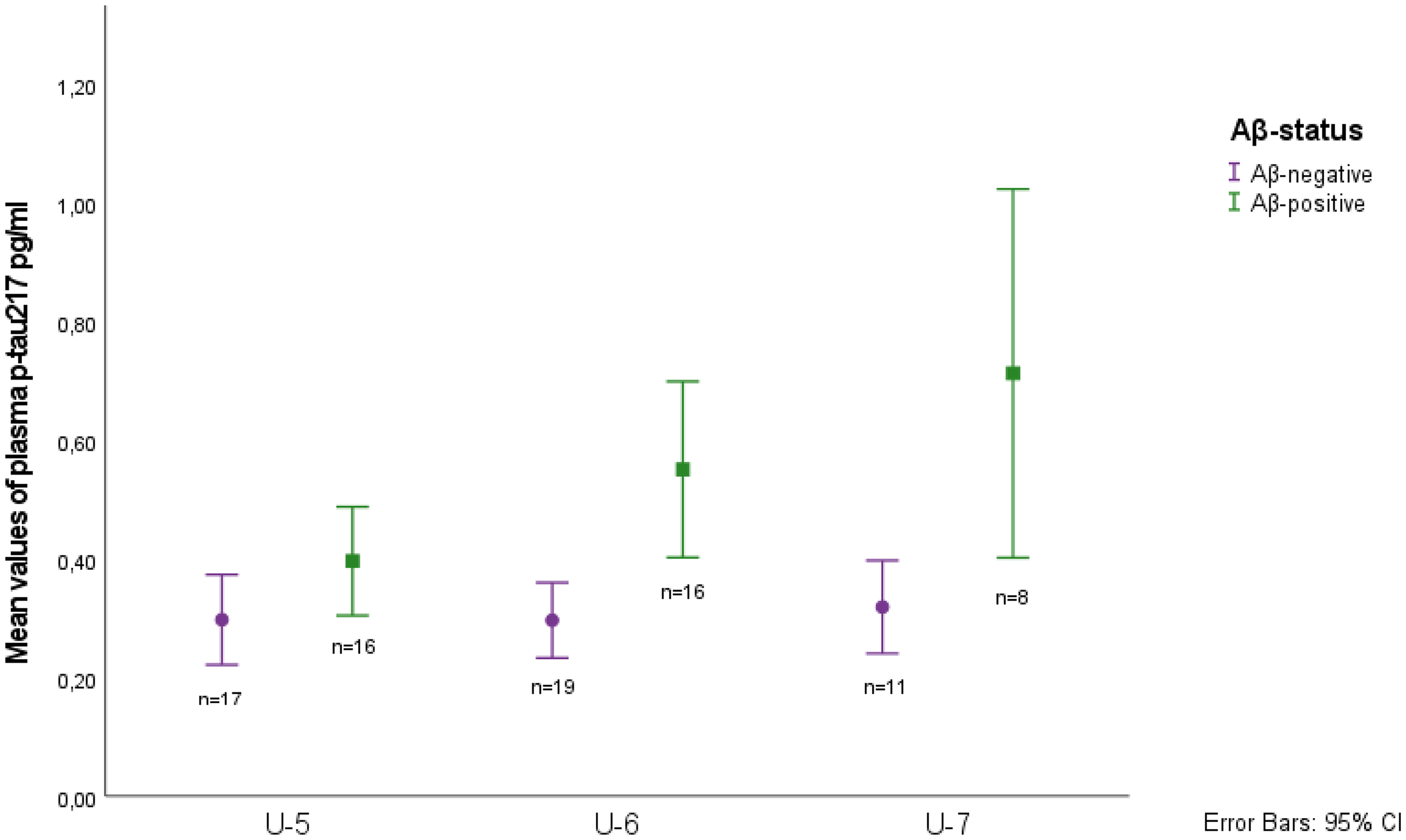
Box plots of mean values of plasma p-tau217 concentrations according to Aβ-status at the three different investigations.
Concentrations of plasma p-tau217 at the three examinations considering Aβ status.
As shown in Figure 5, plasma p-tau217 demonstrated strong discriminatory ability between Aβ-positive and Aβ-negative individuals (AUC = 0.91, 95% CI: 0.82–1.0, p < 0.001).
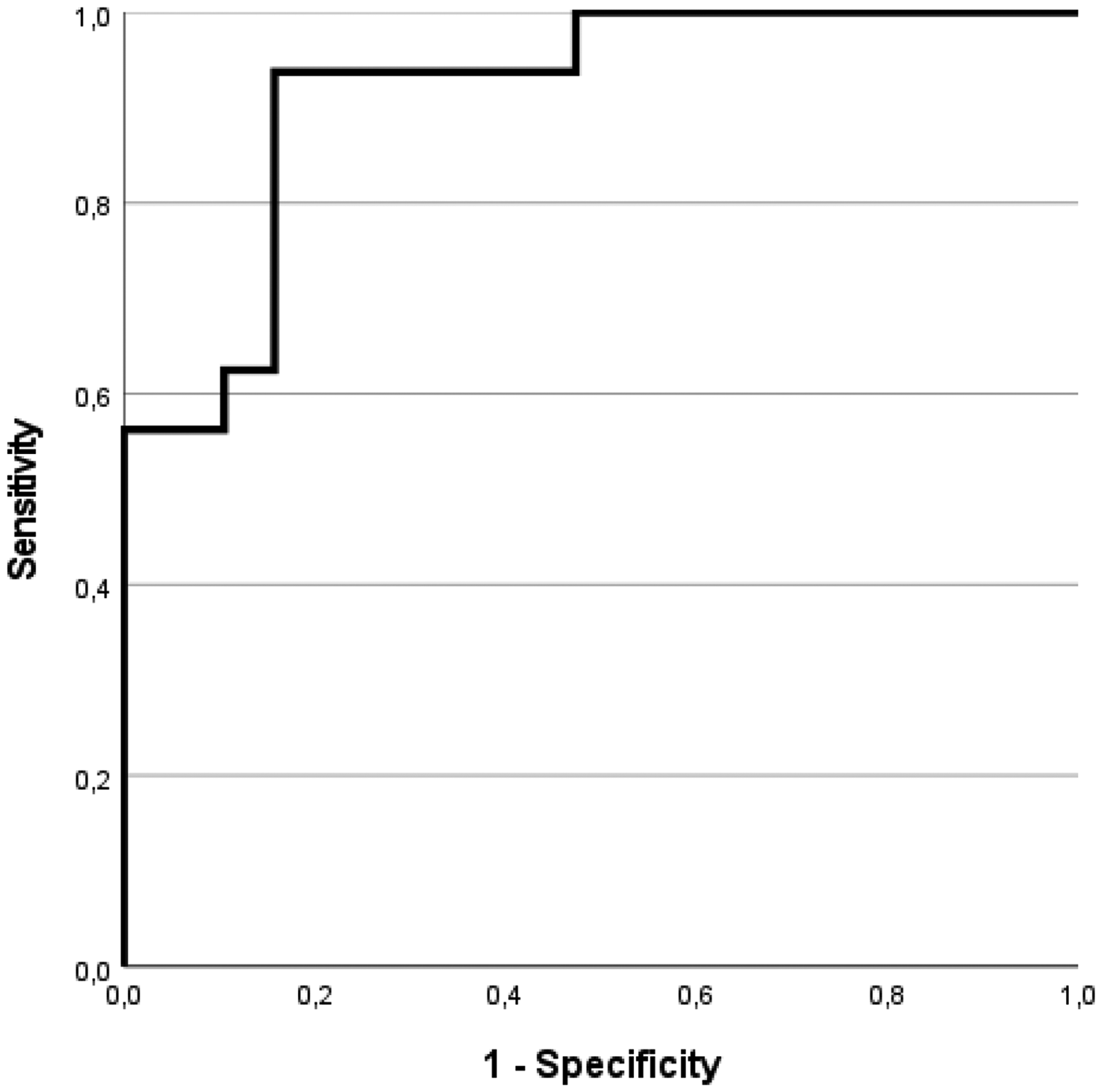
Receiver operating characteristics (ROC) curve for plasma p-tau217 in distinguishing Aβ-positive subjects from Aβ-negative subjects.
Among the 57 individuals that underwent CT scan, 33% had an MTA score of 3–4 and 62% a GCA score of 2–3. There were no significant correlations between plasma p-tau217 at baseline and any of the neuroradiological markers, for MTA (Spearman ρ: rho = −0.01, p = 0.937) and for GCA (Spearman ρ: rho = 0,109, p = 0.421).
Further, there was no significant correlation between plasma p-tau217 and MMSE scores at baseline. Spearman ρ: rho = −0.09, p = 0.619 for the 35 individuals in the CSF group.
Incident dementia in the total U-6 population (n = 352) was 33%. In the CSF group, nine men (26%; four Aβ-positive and five Aβ-negative subjects) developed dementia during follow-up to December 2022, or to the date of death. Six men had received a clinical diagnosis of AD (without knowledge of the CSF data) and in three cases no specific dementia diagnosis could be set. Although the confidence intervals were overlapping, non-significantly higher plasma p-tau217 levels were seen at baseline in men with incident dementia (median 0.42 pg/ml, IQR [0.29–0.79] versus 0.34 pg/ml, [0.26–0.46], p = 0.21), whereas there was a trend for lower CSF Aβ42 levels (median 482 pg/ml, IQR [396–807] versus 717 pg/ml, [512–1014], p = 0.12).
Discussion
In this community-based cohort of 35 very old men, free from dementia at baseline, we found a close correlation between levels of plasma p-tau217 and CSF Aβ42 in amyloid-positive subjects. Importantly, plasma p-tau217 proved to be a dynamic AD biomarker with markedly increasing concentrations in the Aβ-positive group over a ten-year period, while it was unchanged in Aβ-negative participants. These results support that elevated plasma p-tau217 primarily reflects the intracerebral aggregation of Aβ-fibrils in the preclinical AD stage in very high age, as has been shown previously among younger age groups.
In our cohort, the correlation between plasma p-tau217 and CSF Aβ42 was present already at U-5, five years prior to baseline, when plasma p-tau217 not yet differed between Aβ-positive and Aβ-negative subjects. Plasma p-tau217 shows a steep increase after Aβ biomarkers have become positive. 44 An association between plasma p-tau and amyloid accumulation according to PET, already in the asymptomatic stage, has been demonstrated in large memory research cohorts mainly of older individuals, although younger than in our cohort.10,12,15,45 Fewer studies have reported the associations between plasma p-tau and CSF amyloid markers. In BioFINDER-2, where the majority were cognitively healthy or mildly impaired and mean age approximately seventy, plasma p-tau217 had a moderate correlation with the CSF Aβ42/40 ratio. 45 Our results align with previous studies showing plasma p-tau217's performance to detect pathological CSF Aβ42/40 ratio in individuals with MCI and mean age approximately 72 years (AUC 0.858–0.947). 16 In the BioFINDER-1 cohort AUC was 0.783 for cognitively unimpaired and 0.879 for MCI subjects. 15
The underlying mechanisms behind pathological soluble tau metabolism are incompletely understood. Increasing plasma p-tau seems to represent physiological reactions to Aβ aggregation at early stages.12,29 We found no correlation between plasma p-tau and CSF Aβ in men with negative amyloid status, indicating that high age per se does not uncouple the connection between intracerebral Aβ and peripheral soluble p-tau.
In previous studies, 12 plasma p-tau217 concentrations showed weaker correlation with CSF p-tau181 than with CSF Aβ42. It was of borderline significance in our cohort probably due to lack of power. Other studies have reported a strong concordance between plasma p-tau and CSF p-tau in amyloid positive individuals across the AD spectrum, with a closer relationship for plasma p-tau217 than plasma p-tau181.16,29,45 Earlier findings suggest a close correlation between plasma p-tau217 and CSF p-tau217 already in amyloid positive cognitively unimpaired subjects as well as in MCI, and prior to insoluble tau aggregates were detected by PET. 19 In a recent narrative review of 33 heterogeneous studies, it was concluded that the strongest association between plasma and CSF p-tau exists in the MCI stage, being less pronounced in earlier stages and disappearing after plasma p-tau reaches its plateau. 29 With a few exceptions, most studies showed moderate to strong correlations (R > 0.5) between plasma and CSF values,14,16,18,45–47 i.e., higher than in our cohort. The participants in our study were cognitively unimpaired at baseline and this may have contributed to the weak correlation between p-tau in plasma and CSF. We found no correlation between plasma p-tau217 and CSF total tau, which is consistent with earlier studies, 18 and also not unexpected since total tau is a non-specific marker of neurodegeneration.
In the Aβ-positive men in our cohort, plasma p-tau217 steadily increased over a ten-year period. We have not identified any other study aiming to describe the trajectory of plasma p-tau in the same individuals over such an extended time period. A few previous studies have analyzed repeated measurements of plasma p-tau217 over 4–6 years, together with markers of AD progression. In the BioFINDER study, mean age approximately 70 years, preclinical and prodromal AD had accelerated plasma p-tau217 while it did not change over time in Aβ-negative participants, nor in MCI patients who did not convert to AD. 21 In the Wisconsin Register for Alzheimer's Prevention cohort (WRAP), mean age 63 years, plasma p-tau217 demonstrated marked amyloid-dependent changes in both preclinical and prodromal AD, and was associated with declining cognition. 15 In the same cohort, p-tau217 increased modestly with age in amyloid negative subjects. 22 In a recent study, plasma p-tau217 was analyzed with a novel commercial immunoassay repeatedly over five years in the WRAP cohort. Plasma p-tau217 increased annually only in Aβ-positive subjects, and most markedly in those who also had a positive tau PET scan. 20
We found no correlation between plasma p-tau217 and MTA, or with GCA in this small cohort. Such structural changes are commonly seen also in cognitively healthy subjects older than 80 years48,49 and CT scans (as used in the present study) are less sensitive for detecting brain atrophy than MRI imaging. Similarly, we did not find any correlation between plasma p-tau217 levels and the results on the MMSE in this small cohort, possibly due to the ceiling effects.
While the diagnostic and prognostic, i.e., conversion to AD dementia, performance of p-tau217 has been convincingly shown in large research cohorts13,15–17 our findings did not show significant associations with incident dementia, which may reflect the restricted sample size of our cohort. Dementia diagnoses were based on medical records, which may have led to underestimation of cases. However, Sweden's universal healthcare and our long follow-up to death likely reduced underreporting.
Further, five of the nine men that later were diagnosed with dementia were Aβ-negatives, reflecting that non-AD neuropathology such as cerebrovascular lesions, hippocampal sclerosis, TDP-43 accumulation and Lewy bodies are common in ninety-year-olds with dementia.50,51
Concerning representability, the number of APOE ε4 carriers was markedly lower in our sample than in the whole cohort. Although the Charlson Comorbidity Index scores did not differ between participants and non-participants, individuals eligible for LP (which were selected for the present study) were likely healthier and with less cognitive impairment compared to the others. The relatively low proportion of APOE ε4 carriers among participants may reflect this selection bias. However, the proportion of participants with positive amyloid status (46%) was very similar to other cohorts in the same age groups. One CSF study of cognitively unimpaired subjects reported that approximately 47% in 85-89-year-olds, 52 and around 40% of 80–89 year-olds without dementia were positive according to amyloid PET. 53 The present cohort included only men, which restricts the generalizability of the findings. Most studies that compared p-tau levels in serum or plasma did not find any differences between men and women, although some studies reported higher plasma p-tau181 or p-tau217 levels in men than in women, while elevated levels have been associated with greater cognitive decline in women. 54
As the population ages and more people reach a very advanced age, the incidence of dementia increases. 55 With current diagnostic tools in primary care where most elderly individuals with cognitive complaints initially present, the diagnostic accuracy for AD is around 60%. 56 This results in suboptimal treatment and care. A recent study demonstrated that a diagnostic algorithm utilizing plasma p-tau217 accurately diagnosed AD in approximately 80% of patients with MCI. 57 A blood biomarker to detect AD can be used in clinical settings for biological staging of the disease, and monitoring disease progression. This improved diagnostic accuracy can facilitate the initiation of widely accessible treatments, such as cholinesterase inhibitors. However, a pathological plasma p-tau value in older individuals with cognitive impairment should be interpreted with caution: it likely indicates Aβ plaque pathology but may not necessarily be causal, as previously mentioned. Not all individuals with amyloid pathology develop cognitive impairment. By the age of 95, individuals who die with and without dementia exhibit a similar burden of neuropathological changes. 58 Hence, a positive plasma p-tau should not be used as a standalone diagnostic test for AD but must be interpreted within a clinical context. 59 A diagnosis must benefit the patient and biomarker analysis should only be conducted when there is a reasonable suspicion of AD. Based on our findings, it is conceivable that repeated measurements in individuals with perceived cognitive complaints could guide clinical decisions. Correspondingly, normal values, especially in serial measurements, may inform the patient of a low risk of AD.
Strengths and limitations
This population-based cohort was homogeneous with respect to age and cognitive level, and with amyloid status representative for this age group. To our knowledge this cohort is unique with both CSF samples and serial plasma p-tau217 concentration measurements beyond the age of ninety years and over a decade. There was a low rate of non-response among men who were still alive at the last sampling. Compared to PET, CSF biomarkers are continuous and change earlier in the AD continuum. Despite the small sample size, we observed robust correlations between plasma p-tau217 and CSF Aβ42. There are several limitations to this study. The small and selective sample, and the difficulties recruiting very old individuals for lumbar puncture, introduce potential selection bias and limit statistical power and generalizability. The limited number of participants may have precluded us from detecting an association to incident dementia. As previously discussed, dementia cases may have been underestimated, potentially reducing statistical power and precision.
CSF was collected approximately two years after baseline plasma samplings, hence, the number of Aβ-positives may be slightly overestimated. The CSF Aβ42, t-tau and p-tau181 ELISA assays used (INNOTEST) are subject to analytical variability and manual processing, which may affect sensitivity. However, all CSF analyses followed accredited, standardized procedures.
Conclusion
In this study of very old men without diagnosed dementia, plasma p-tau217 was a reliable predictor of amyloid status, similar to younger age groups. Moreover, our data show that plasma p-tau217 is stable over a time period of at least ten years in Aβ-negative subjects even after the age of ninety. These findings indicate usefulness of this biomarker in clinical practice by aiding in disease progression monitoring and may offer the same information as CSF. This is highly relevant when deciding on pharmacological treatment, both currently and in light of future changes in the therapeutic landscape. The full potential of plasma p-tau217 as a prognostic biomarker of cognitive deterioration in very old persons needs to be further explored in larger cohorts including both genders.
Supplemental Material
sj-docx-1-alz-10.1177_13872877251390387 - Supplemental material for Plasma p-tau217 correlates strongly with cerebrospinal fluid Aβ42 and increases over a ten-year period in amyloid-positive, non-demented very old men
Supplemental material, sj-docx-1-alz-10.1177_13872877251390387 for Plasma p-tau217 correlates strongly with cerebrospinal fluid Aβ42 and increases over a ten-year period in amyloid-positive, non-demented very old men by Elisabeth Hellquist, Shorena Janelidze, Bodil Weidung, Kristin Franzon, Vilmantas Giedraitis, Martin Ingelsson, Divya Bali, Vilma Velickaite, Henrik Zetterberg, Oskar Hansson and Lena Kilander in Journal of Alzheimer's Disease
Footnotes
Acknowledgements
The authors thank all the participants in ULSAM, Käthe Ström, research nurse, for excellent care of the study participants and Professor Elna-Marie Larsson for neuroradiological expertise.
Ethical considerations
The study was approved by the Regional Ethical Review Board at Uppsala University (Approval number: Dnr 02-605 on 18-02-2003, Dnr 2007/338 on 23-01-2008 and Dnr 2013/350 on 23-10-2013).
Consent to participate
All participants gave their written informed consent before participating.
Consent for publication
Not applicable.
Author contribution(s)
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by The Freemason Foundation of King Gustaf V and Queen Victoria, The Geriatric Foundation and Thuréus Foundation. The foundations had no role in the design, conduct of research or decision of publication. HZ is a Wallenberg Scholar and a Distinguished Professor at the Swedish Research Council supported by grants from the Swedish Research Council (#2023-00356, #2022-01018 and #2019-02397), the European Union's Horizon Europe research and innovation programme under grant agreement No 101053962, and Swedish State Support for Clinical Research (#ALFGBG-71320).
Declaration of Conflicting Interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: MI is a paid consultant to BioArctic and EISAI. HZ has served at scientific advisory boards and/or as a consultant for Abbvie, Acumen, Alector, Alzinova, ALZpath, Amylyx, Annexon, Apellis, Artery Therapeutics, AZTherapies, Cognito Therapeutics, CogRx, Denali, Eisai, Enigma, LabCorp, Merry Life, Nervgen, Novo Nordisk, Optoceutics, Passage Bio, Pinteon Therapeutics, Prothena, Quanterix, Red Abbey Labs, reMYND, Roche, Samumed, Siemens Healthineers, Triplet Therapeutics, and Wave, has given lectures sponsored by Alzecure, BioArctic, Biogen, Cellectricon, Fujirebio, Lilly, Novo Nordisk, Roche, and WebMD, and is a co-founder of Brain Biomarker Solutions in Gothenburg AB (BBS), which is a part of the GU Ventures Incubator Program (outside submitted work). OH is an employee of Lund University and Eli Lilly. All other authors have no conflict of interest to report.
Data availability statement
The data supporting the findings of this study are available on request from the corresponding author. The data are not publicly available due to privacy or ethical restrictions.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
