Abstract
Background
Neutrophil to lymphocyte ratio (NLR) is an inflammatory biomarker for chronic disease that also may provide evidence for peripheral innate immune activation in Alzheimer's disease (AD) pathogenesis.
Objective
The objective of this study is to assess the association between NLR and AD as a biomarker or potential causal step for AD pathogenesis.
Methods
Cohorts used for analysis include the UK Biobank (n = 207100; n = 2198 AD cases) and the All of Us research program (AoU, n = 45202; n = 263 AD cases) which allowed for a larger sample size than most previous studies. Cox proportional hazard models were used to assess the association between NLR and AD, including in models adjusting for factors in the UK Biobank Dementia Risk Score. A Mendelian randomization analysis was performed to assess the potential causality between NLR and AD.
Results
NLR was associated with AD in the UK Biobank (HR: 1.03 per 1 SD, 95% CI: 1.01–1.05) but not in AoU (HR: 1.02, 95% CI: 0.92–1.14). A fixed effects model resulted in a pooled HR: 1.03 (95% CI: 1.02–1.05). These effects were robust to adjustment for C-reactive protein, APOE ε4 genotype status, APOE ε2 genotype status, and other AD risk factors. The MR analysis was performed with 214 NLR-associated variants but was not significant (IVW estimate: −0.001, p: 0.99).
Conclusions
Findings provide evidence for NLR as a biomarker for AD but not as a causal risk factor.
Introduction
Alzheimer's disease (AD) is a neurodegenerative disease resulting in impairments in cognition and functional abilities and is the leading cause of dementia. 1 In the United States, 6.5 million people 65 years or older are estimated to have AD and the prevalence is projected to increase to 13.8 million by 2060. 2 While AD is characterized by amyloid plaques and neurofibrillary tangles, 1 the complete pathogenesis of AD is not understood. Inflammation, both systemic and brain-specific, is thought to play a role in AD. 3
The peripheral immune system is the portion of the immune system that resides outside of the central nervous system and is responsible for mounting an immune response including inflammation to pathogens and injury. However, even though this system is outside of the central nervous system, changes in the peripheral immune system such as peripheral inflammation have been associated with neurodegenerative diseases including AD. 4 It has been hypothesized that systemic peripheral inflammation may lead to neuroinflammation through an increase in cytokine transport across the blood brain barrier. 5 Dysregulated cross-talk or movement of peripheral immune cells or signals crossing the blood brain barrier could lead to excessive neuroinflammation and accelerate neuron death.5–7 Systemic inflammation would result in an increase in the concentration of pro-inflammatory cytokines and therefore could increase the amount of pro-inflammatory cytokines that can cross the blood brain barrier. 5 Systemic inflammation could also disrupt the blood brain barrier and allow for the increase the movement of lymphocytes, natural killer cells, and neutrophils from the peripheral immune system across the blood brain barrier into the central nervous system. 7 Systemic inflammation markers could also correlate with much more difficult to measure neuroinflammation metrics, even if they are not causally related to AD development. Such inflammation markers have been associated with AD. For example, higher levels of the inflammatory biomarker interleukin 6 (IL-6), a cytokine with effects on multiple processes including inflammation, 8 have been associated with AD risk in a large meta-analysis. 9 Midlife C-reactive protein (CRP), a protein produced in response to inflammation, 10 also has been associated with higher AD risk.9,11–13 Prolonged elevation in CRP levels may indicate chronic inflammation possibly due to certain chronic conditions. 14 For example, a CRP level less than 0.3 mg/dL is considered normal for most healthy adults. 14 However, a CRP level between 1.0 and 10.0 mg/dL can be considered moderate elevation that is associated with systematic inflammation, for example due to rheumatoid arthritis, autoimmune diseases, or other conditions. 14 While CRP is used as a marker for inflammation, CRP, depending on the isoform, has both anti-inflammatory and pro-inflammatory effects. 15
Neutrophil to lymphocyte ratio (NLR) is a marker for systemic inflammation, reflecting the balance between innate (neutrophil) and adaptive (lymphocyte) immune system cells. 16 High NLR (reflecting greater innate immune activation in the periphery) has been associated with higher overall mortality and increased cardiovascular disease prevalence.17,18 Higher levels of NLR have also been associated with increased AD risk and age-related cognitive impairment; however, these associations have been mostly in relatively small cross-sectional case control studies which are unable to or have difficulty establishing temporality.19–24 One study with a longitudinal prospective design found higher NLR to be associated with incident dementia, of which 80% of cases were classified as AD. 25 Recent work in UK Biobank has found NLR to be associated with dementia, among other brain disorders, but it is unclear how robust this association is to adjustment for other well-known AD risk factors and important covariates such as hypertension and hypercholesterolemia. 26 Another study in the UK Biobank with 161,968 participants found higher NLR was associated with all-cause dementia (HR: 1.03, 95% CI: 1.01–1.05). 27 While this study adjusted for blood pressure, other potential confounders used in existing dementia prediction models such as parental dementia history and type 2 diabetes (T2D) were not controlled. 27 It should also be noted that since NLR involves neutrophils, the NLR will be affected by the Duffy null variant (rs2814778-CC). Duffy null status has been associated with lower neutrophil counts and lower NLR when compared to Duffy non-null status.28,29 As a result, Duffy null status may be an important component to consider when assessing the association between NLR and AD. Examining the association between NLR and AD in large biobank cohorts could provide further evidence for the role of inflammation in AD pathogenesis.
Mendelian randomization (MR) may provide further insight by assessing the potential causality between NLR and incident AD. Through using genetic factors as a proxy for the exposure of interest, MR can mimic a randomized control trial and combat common concerns for observational studies including reverse causation and confounding.30,31 However, it should be noted that certain assumptions (genetic variant is associated with the exposure, the genetic variant only affects the outcome through the exposure, and there are no confounders between the genetic variant and outcome) are required for MR to be valid. 32 We include multiple MR methods to try to address such limitations (for example variant pleiotropy).
Here, we aim to investigate the association between AD and NLR in two large cohorts (UK Biobank and All of Us (AoU)) with the hypothesis that a higher NLR would be associated with a higher risk of incident AD and that this association would be robust to adjusting for variables in the UK Biobank Dementia Risk Score (UKBDRS) 33 including age, sex, education, diabetes history, depression history, stroke history, parental history of dementia, material deprivation, hypertension, high cholesterol, and household occupancy as well as the Duffy null status (which impacts NLR) 34 and the APOE ε4 and APOE ε2 variants.
Methods
The UK Biobank is a prospective cohort study that began in 2006 and has continued to the present day. This cohort has phenotypic and genetic data on 502,649 participants, aged 40 to 69 years old, throughout 22 assessment centers in Wales, Scotland, and England.35–37 AD was defined using inpatient ICD10/9 codes and death certificates. In other cohorts, ICD-10 codes F00 and G30 have been found to have a positive predictive value (PPV) of 74.8% and a sensitivity of 71.4% in primary care. 38 ICD-9 code 331.0 has been found to have a 36.5% sensitivity, 99.0% specificity, and 97.9% PPV and death certificates using this ICD-9 code have been found to have a 28% sensitivity and 98% specificity.39,40 Follow-up time was from the date of baseline blood draw to either date of AD diagnosis, date of death, or last date of follow-up. The blood draw measurements and covariate data were from the time of baseline in-person assessment and the last date of follow-up was based on source of hospital admission (Hospital Episode Statistics for England: September-30–2021, Scottish Morbidity Record: July-31-2021, Patient Episode Database for Wales: February-28-2018). The lymphocyte count and neutrophil count came from the blood draw at the initial assessment visit. 41 Controls were restricted to individuals 65 years or older at the end of follow up.
AoU is a cohort, starting in 2018, that began recruiting participants in May 2018 at least 18 years old or older from 340 sites with the goal of creating a diverse US cohort of 1 million participants. 42 Data for this cohort comes from questionnaires, electronic health records (EHR), physical measurements, digital technology, and biospecimen collection. 42 AD was also defined using the same ICD10/9 codes as in UK Biobank. The time of blood draw was assigned as the time of the blood draw for the median value of lymphocyte count. Participants without neutrophil or lymphocyte information or with certain blood related disorders before or a year after the time of blood draw were excluded from the analysis (Supplemental Table 3). Time of blood draw and time of AD diagnosis (based on medical billing codes) were obtained by EHR. The last date of follow up for AoU (version 8) was October 1, 2023. 43 In AoU, participants could have multiple neutrophil and lymphocyte measurements over time. As a result, the lymphocyte count used for NLR was defined as the median lymphocyte count and the neutrophil count was defined as the neutrophil count taken at the time of the median lymphocyte count. Participants with a median lymphocyte count or neutrophil count equal to 0 or greater than 50 × 103/µL were excluded due to lack of plausibility of the value.44,45 These thresholds were also applied to monocyte count, while platelet count was restricted to values greater than 0 but less than 2000 × 103/µL. Controls were restricted to individuals 65 years or older at the end of follow up and AD cases were restricted to individuals 65 years or older at first ICD code. We also performed a sensitivity analysis using the first instead of median lymphocyte count.
Statistical analysis
In the UK Biobank, Cox proportional hazards models were used to estimate hazard ratios (HR) adjusting for variables previously described in the UKBDRS (Supplemental Table 1) 33 as well as self-reported ethnicity (Field 21000) and assessment center (Field 54). The variables in the UKBRDS were chosen from 28 variables using a Cox LASSO regression in the UK Biobank and the Whitehall II cohorts as the strongest predictors for incident dementia. 33 Self-reported ethnicity (and self-reported race in AoU) is adjusted for due to differential diagnosis rates of AD based on social constructs. 46 Duffy null status (defined by rs2814778-CC genotype, imputation quality = 1.0), which causes lifelong lower neutrophil counts,34,47 was also included as a covariate. This variant, rare in European ancestry populations but more common in populations with West African ancestry, 47 has been previously demonstrated to influence the association of NLR with mortality and CVD. 34 In AoU, Cox proportional hazards models were adjusted for age, sex, race and ethnicity, and Duffy null status (rs2814778-CC). AD-related risk factors used in the UK Biobank model were not adjusted for in AoU due to extensive missing data. A fixed effects model using the metafor R package was used to pool the estimates from UK Biobank and AoU with similar covariates (age, sex, race, and ethnicity).
As a sensitivity analysis, the model was further adjusted for CRP due to CRP being a more commonly measured inflammation marker, with conflicting prior evidence of association with AD.11–13 Sensitivity analyses were also adjusted for APOE ε4 and APOE ε2 status (rs429358, rs7412). We also examined models with neutrophil and lymphocyte count separately to determine if ratio associations were driven by individual cell counts. Monocyte to lymphocyte ratio (MLR) and platelet to lymphocyte ratio (PLR) were also investigated as exposures in supplementary analyses due to MLR and PLR being other complete blood count derived inflammation markers. 48 Interaction between NLR and sex was also tested for in UK Biobank due to sex differences in AD and in immune function. This interaction was not tested for in AoU due to the small number of AD cases. An expanded definition of dementia was also used as a sensitivity analysis (Supplemental Table 2). 49
In addition, a two-sample Mendelian randomization (MR) analysis was performed to further assess the association of NLR with AD. While a disadvantage of MR is risk of being underpowered, MR provides insight when reverse causality is a concern. Since AD is hypothesized to begin decades before symptoms first appear, it is possible the NLR measurement may be after AD started to develop. MR can help address this concern. First, a genome wide association study (GWAS) was performed in the UK Biobank to uncover variants for the MR analysis for NLR as NLR was not included in prior large-scale UK Biobank blood cell trait GWAS. 50 Using REGENIE (v4.1), the GWAS included 9,741,125 variants (imputation quality threshold: 0.50, minor allele frequency: 1%) and 400,131 participants who had complete NLR and covariate information (age, sex, genotyping array, first 10 principal components) and were in the European ancestry cluster based on similarity to 1000G reference panel, as described previously. 51 The p-value threshold for significance was 5 × 10−8. The top variant at each locus, excluding the highly pleiotropic APOE, ABO, and HLA regions, was used for the MR analysis. Each locus was defined as a 1 Mb region, 500 kb on each side of the lead SNP. The corresponding effect sizes for these SNPs with AD was obtained from the Bellenguez et al. GWAS summary statistics. 52 We focused on inverse-variance weighted (IVW) estimator due to the lack of nominal significance (p < 0.2) of MR-Egger intercept to test for directional pleiotropy, though we also added MR-Egger estimator (R package: MendelianRandomization version 0.10.0) to supplemental tables for sensitivity analysis. 53
Results
Of the 502,539 available UK Biobank participants, 431,812 had blood cell information and did not have blood related disorders (Supplemental Table 3).54,55 Of these participants, 207,100 had last-day of follow-up, controls 65 years or older at the end of follow up, and covariate information. Among the 207,100 UK Biobank participants eligible for our study, mean age at baseline was 61.3 years, 55.3% were female, 0.6% were Duffy null, and most were of White self-reported ethnicity (93.2%). The mean NLR was 2.33. Over a median follow-up time of 12.5 years, n = 2198 (1.1%) incident AD cases (128 cases under 65 years old at diagnosis) were identified (Table 1). For the excluded participants (n = 295,429) the mean age was 53.2, 53.8% were female, and 88.4% self-reported White ethnicity.
Population characteristics in UK Biobank and All of Us (AoU).
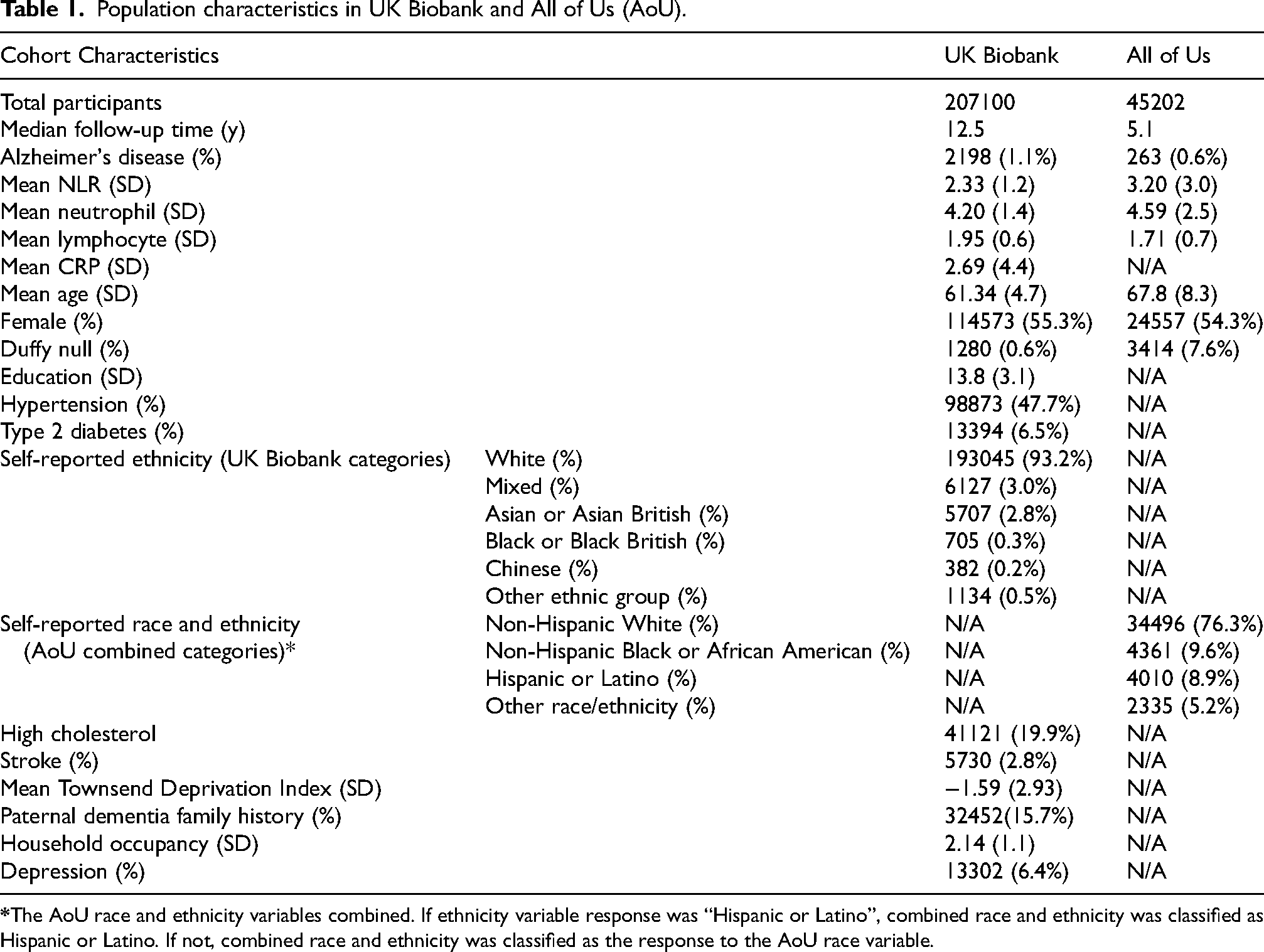
In AoU, 45,202 participants were included (Table 1). Only 0.6% (n = 263) developed incident AD and 7.6% were Duffy null. The mean NLR was 3.20 and mean age was 67.8 years. Most participants were female (54.3%) and self-reported White race (76.3%). Participants had a median follow-up time of 5.1 years.
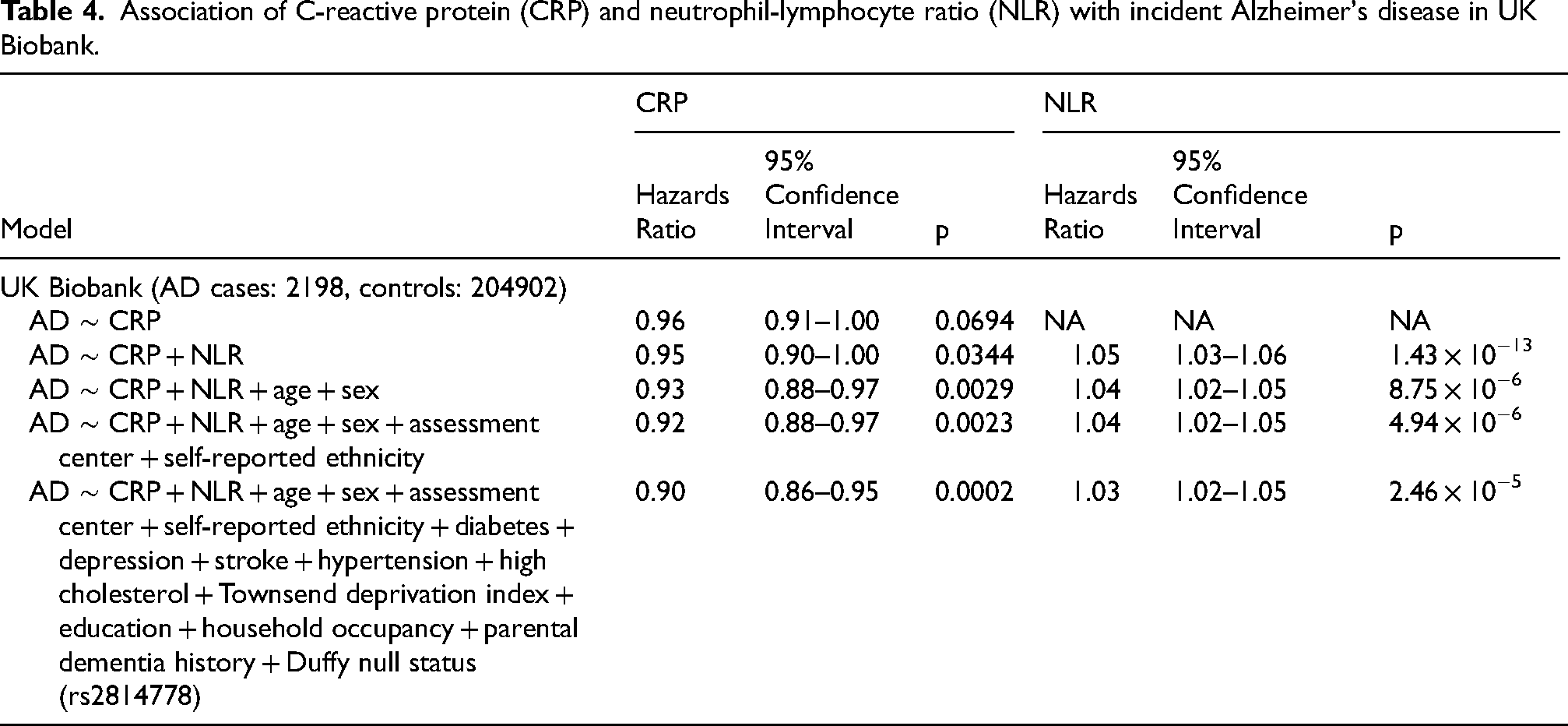
In UK Biobank, after adjusting for age and sex, a 1 standard deviation (SD) higher NLR level was associated with a 3% higher risk of AD (HR = 1.03, 95% CI = 1.02–1.05). This association was robust to adjustment for AD-related risk factors from the UKBDRS in UK Biobank 33 and the Duffy variant (Table 2) as well as APOE status (HR = 1.03, 95% CI: 1.01–1.05) to prediction models (Table 3, Figure 1). The NLR association with AD was also robust to the addition of CRP (NLR HR = 1.03 for NLR, NLR 95% CI: 1.02–1.05) after adjusting for the UKBDRS variables, age, sex, assessment center, self-reported ethnicity, and Duffy (Table 4). CRP was associated with AD after adjustment for the UKBDRS variables, age, sex, assessment center, self-reported ethnicity, Duffy, and NLR (HR: 0.90, 95% CI: 0.86–0.95) (Table 4, Figure 1). There was not a significant interaction between sex and NLR in the model adjusting for sex and age (sex NLR interaction term p = 0.70). We did not observe a statistically significant association between NLR (per 1 SD higher) and AD in All of Us (HR = 1.02, 95% CI: 0.92–1.14) (Table 2). Using the first lymphocyte measurement, increasing the number of incident AD cases to 374, the association between NLR and AD was not statistically significant (HR = 1.03, 95% CI: 0.94–1.13) (Supplemental Table 4). The pooled HR from UK Biobank adjusting for age, sex, assessment center, and self-reported ethnicity and AoU adjusting for age, sex, and race and ethnicity was 1.03 with a 95% CI of 1.02–1.05.
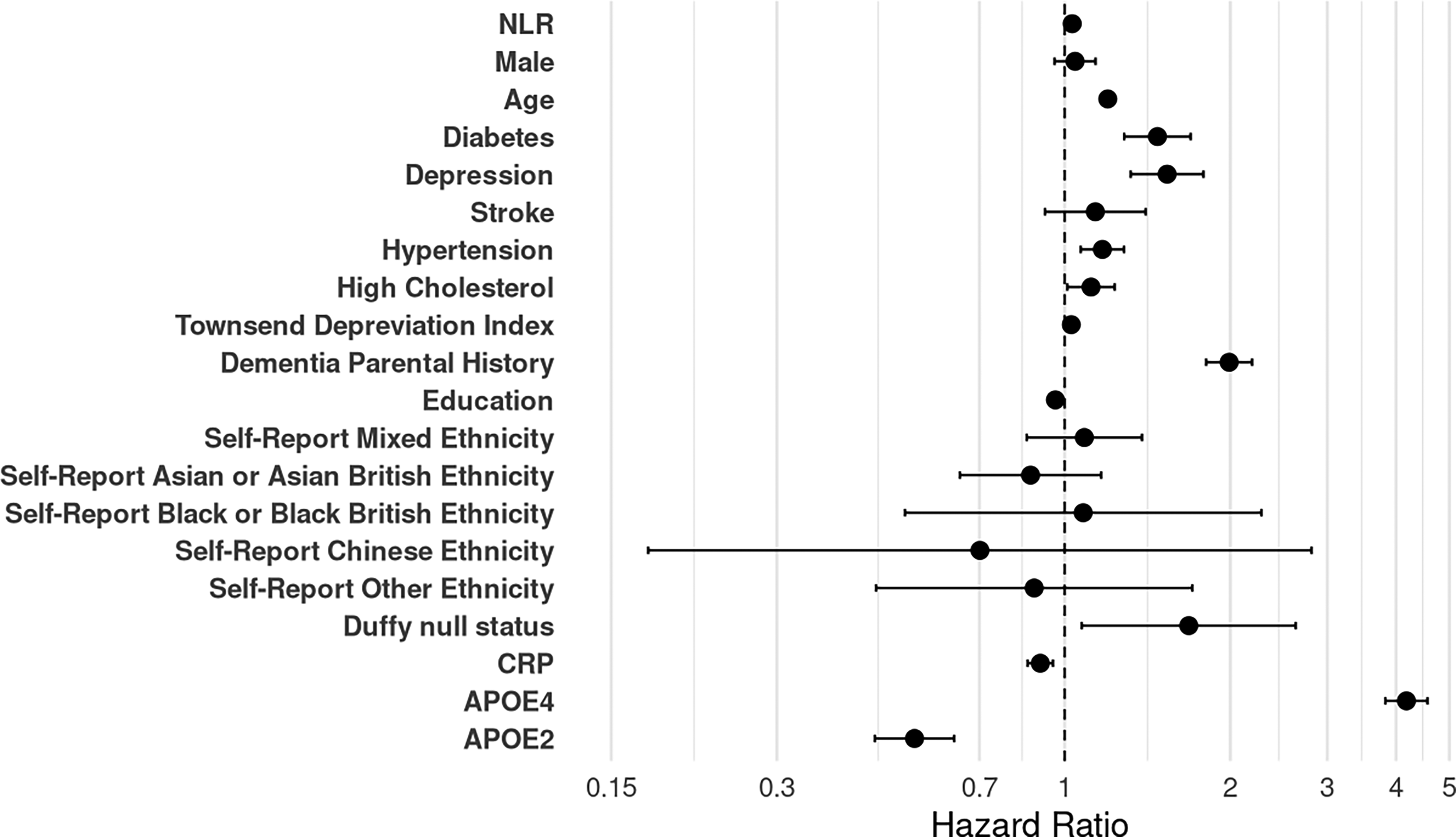
UK Biobank hazards ratios for NLR, UKBDRS variables, self-reported ethnicity, Duffy null status, CRP, APOE4, and APOE2. White self-reported ethnicity is reference group for Mixed, Asian or Asian British, Black or Black British, Chinese, or other ethnic group self-reported ethnicity. Hazards ratios for NLR, UKBDRS variables, self-reported ethnicity, and Duffy null status obtained from model adjusting for UKBDRS variables, self-reported ethnicity, Duffy null status, and assessment center. CRP and APOE variants hazards ratios obtained by adding the respective variable to the previous model.
Association of neutrophil-lymphocyte ratio (NLR) with incident Alzheimer's disease in UK Biobank and All of Us.
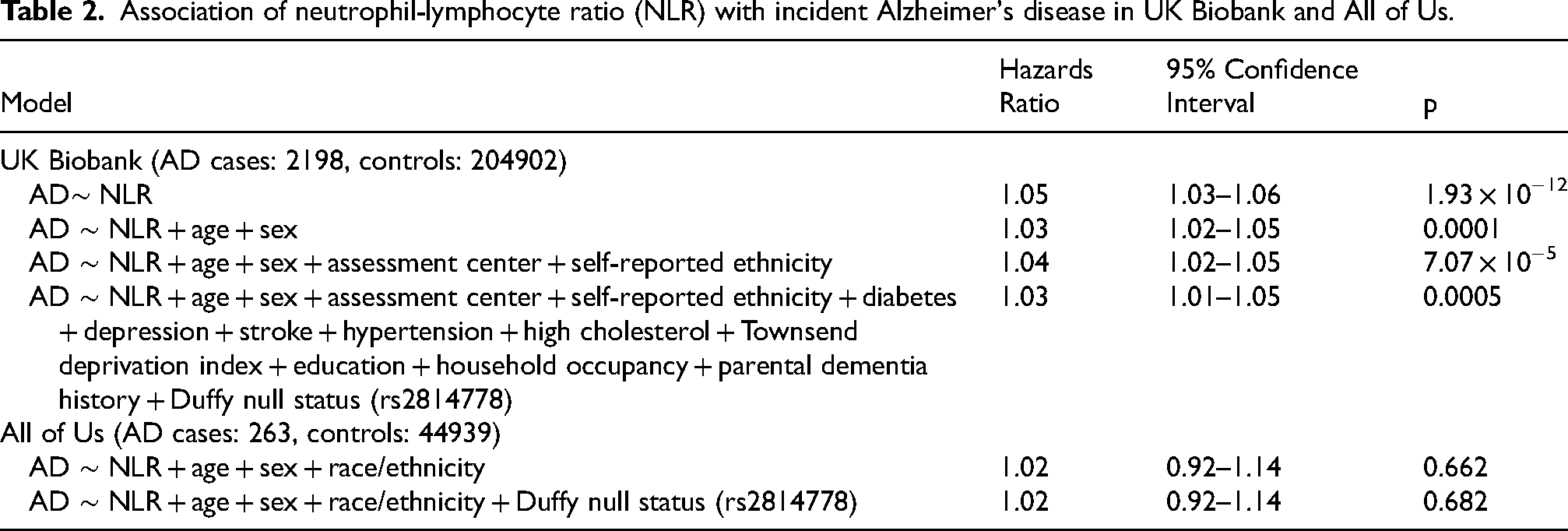
Results for addition of APOE4 variant to models in UK Biobank, with hazards ratio reported per standard deviation (sd) of NLR.
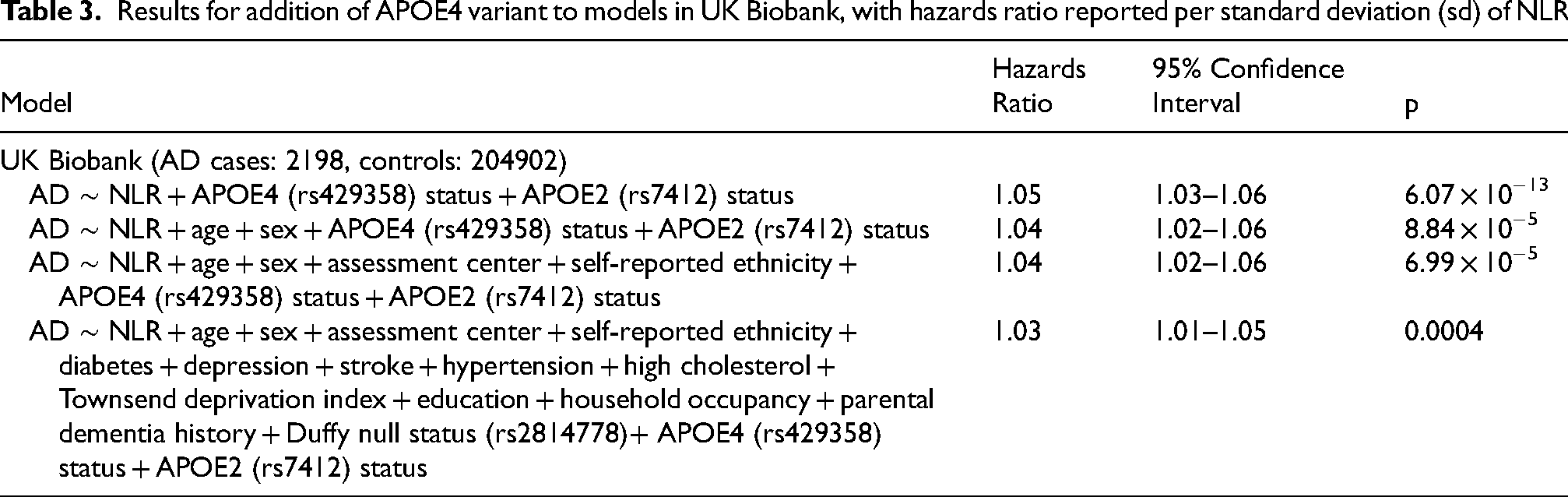
Association of C-reactive protein (CRP) and neutrophil-lymphocyte ratio (NLR) with incident Alzheimer's disease in UK Biobank.
In the UK Biobank, neutrophil count was associated with AD, robust to adjustment for potential confounders, while the lymphocyte count was not statistically significant in any model (Supplemental Tables 5 and 6). In the AoU, neither neutrophil count nor lymphocyte count was statistically significant for AD (Supplemental Tables 5 and 6).
When a more expansive definition of all-cause dementia (including AD, vascular dementia frontotemporal dementia, unspecified dementia) (Supplemental Table 2) was used, NLR was associated with dementia in the UK Biobank (HR 1.05, 95% CI 1.05–1.06) but still not in AoU (Supplemental Table 7). Monocyte to lymphocyte ratio (MLR) and platelet to lymphocyte ratio (PLR) were both not significantly associated with AD in the UK Biobank or AoU (Supplemental Tables 8 and 9).
GWAS of NLR in European ancestry UK Biobank participants resulted in 214 lead variants (all from distinct 1 Mb regions, clumping based on distance) also present in the AD summary statistics for the MR analysis (Supplemental Table 10). The MR analysis resulted in an F-statistic of 67.7. In two-sample MR, the results of the IVW and, as a sensitivity analysis, MR-Egger were both not significant (IVW beta: −0.001, 95% CI: −0.077–0.076, p: 0.99, Egger intercept p = 0.15, Egger estimate: −0.129, 95% CI: −0.318–0.056, p: 0.18) (Supplemental Table 11, Supplemental Figures 1 and 2).
Discussion
NLR was associated with AD in the UK Biobank after accounting for multiple lifestyle, environmental, and clinical risk factors as well as Duffy null status. However, this association was not significant in AoU, though effect direction was concordant, likely due to small number of detected cases and short total available follow-up time. MR results do not support a causal role for NLR in AD; this suggests NLR may instead be correlated with other true causal risk factors for AD unmeasured in this study.
Previous studies have investigated the association between NLR and AD. Multiple cross-sectional case-control studies, including a meta-analysis, found AD patients to have a higher mean NLR compared to patients with normal cognition.19,21–23 A systematic review has also been performed for NLR and AD with 12 studies, where they found a higher NLR to be associated with AD compared to healthy controls and compared to participants with mild cognitive impairment, again based on mostly cross-sectional data. 56 A previously mentioned study in the Framingham Heart Study found NLR to be associated with incident all-cause dementia. 25 This study had the strength of being a prospective study while many previous studies have had a cross-sectional design, but number of cases was small (n = 51). There have also been previous studies looking at incident AD and dementia with NLR in the UK Biobank.26,27 These studies found a higher NLR to be associated with AD and all-cause dementia similar to our study. However, a weakness of these studies is not adjusting for important covariates such as Duffy null status, CRP, and variables included in UKBDRS,26,27 and MR was not considered. Our study also had a longer median follow up time (12.5 versus 8.99 years) and more AD cases (2198 versus 1896 cases) than one study 26 and a greater overall sample size (207,100 versus 161,968 participants), longer median follow up time (12.5 versus 9.66 years), and more dementia cases in our supplementary analysis (5048 all-cause dementia versus 2958 dementia cases) than another study. 27 The use of the UKBDRS is an important addition to our study compared to previous studies since this allows for adjusting for variables that have been shown to be strong predictors in the UK Biobank for dementia. 33 However, contrary results for NLR with dementia have also been reported. 57 Compared to these previous studies, our study utilizes incident cases with longer follow up, accounts for time of follow-up from time of white blood cell measures, helps clarify the likely noncausal role of NLR in AD development using MR analysis, examines the constituent components of NLR, and also has the advantage of having an increased sample size for AD cases.
CRP is another commonly used marker for chronic inflammation 10 and has been examined for associations with AD in multiple prior efforts.10,58,59 While CRP was not associated with AD incidence in the UK Biobank when categorized into CRP < 8 mg/L and CRP ≥ 8 mg/L, 58 higher CRP (modeled as continuous) adjusted for NLR, age, sex, assessment center, self-reported ethnicity UKBDRS factors, Duffy null status (rs2814778) was associated with lower AD incidence (HR: 0.87, 95% CI: 0.82–0.91) in our study. Differences in reported effect sizes may be due to higher CRP having been associated with higher AD risk in midlife and lower AD risk at older ages.11–13 In this study, the association between NLR and AD incidence remained after adjusting for CRP. The association between CRP and AD also remained after adjustment for NLR. This suggests NLR may be an important inflammation marker to investigate for predictive value for AD, apart from the more commonly studied CRP biomarker.
While these previous studies found similar associations between NLR and AD or dementia in the UK Biobank as our study,26,27 the use of MR in our study provides further insight into whether NLR is a biomarker of AD development or a step along the causal pathway, and our more complete modelling strategy provides additional information on independence of this association from known or related risk factors. The association between NLR and AD provides evidence for systemic inflammation as either having a role in or being a biomarker for AD pathogenesis. While measured NLR had an association with AD, the MR analysis did not have a significant result. This suggests that NLR may be a biomarker for AD development but is not a causal step in AD pathogenesis. A recent MR publication suggested that lymphocyte counts may be a causal risk factor for vascular, as opposed to Alzheimer's, dementia, based on summary statistics from FinnGen, suggesting that the putative causal role of both leukocyte subpopulations and their ratios (not examined in this prior publication) should be further investigated for additional dementia subtypes in future work. 59 As previously mentioned, strengths of this study include the number of AD cases available in the UK Biobank, adjustment for known dementia risk factors, inclusion of the novel AoU dataset, as well as the prospective design of the study. However, a limitation of this analysis is missing AD cases due to low sensitivity to detect AD 39 from health records and death certificates. Another limitation, specifically for AoU, is a lack of sufficient power to detect a significant effect size due to a smaller number of incident AD cases. A power analysis in R (R package: survivalpwr) for Cox proportional hazards model assuming an HR of 1.03, standard deviation of 3.04, AD probability of 0.6%, and R2 of about 0.02 results in a power of 0.30. Also, different social and environmental exposures could play a role in population heterogeneity resulting in a lack of generalizability. Finally, use of a standardized baseline visit NLR, with standardized methods across all participants, in UK Biobank, versus electronic health record derived measures for NLR which may have a higher rate of noise or error in AoU, may also impact results. Our cohort in the AoU is currently too small to assess. Based on prior analyses of administrative record-based AD case adjudication, 60 we are possibly underestimating the true number of AD cases in both AoU and UK Biobank, which biases our results towards the null. The use of ICD-9/ICD-10 codes for AD may also include dementia cases that are not truly AD cases such as vascular dementia cases.
NLR was associated with AD in UK Biobank but not in AoU, though MR analyses suggest NLR may not be a causal biomarker for AD. These findings provide further evidence for the association between NLR and AD as well as the possible role of systemic inflammation in AD development.
Supplemental Material
sj-docx-1-alz-10.1177_13872877251386813 - Supplemental material for Association between neutrophil to lymphocyte ratio and Alzheimer's disease in large biobank cohorts
Supplemental material, sj-docx-1-alz-10.1177_13872877251386813 for Association between neutrophil to lymphocyte ratio and Alzheimer's disease in large biobank cohorts by Emily Drzymalla, Christy Avery, Priya Palta, Alexander P Reiner, Quan Sun and Laura M Raffield in Journal of Alzheimer's Disease
Supplemental Material
sj-xlsx-2-alz-10.1177_13872877251386813 - Supplemental material for Association between neutrophil to lymphocyte ratio and Alzheimer's disease in large biobank cohorts
Supplemental material, sj-xlsx-2-alz-10.1177_13872877251386813 for Association between neutrophil to lymphocyte ratio and Alzheimer's disease in large biobank cohorts by Emily Drzymalla, Christy Avery, Priya Palta, Alexander P Reiner, Quan Sun and Laura M Raffield in Journal of Alzheimer's Disease
Footnotes
Acknowledgements
This research has been conducted using the UK Biobank Resource under Application Number 25953. The All of Us Research Program was made possible by the partnership of its participants. The All of Us Research Program is supported by the National Institutes of Health, Office of the Director: Regional Medical Centers: 1 OT2 OD026549; 1 OT2 OD026554; 1 OT2 OD026557; 1 OT2 OD026556; 1 OT2 OD026550; 1 OT2 OD 026552; 1 OT2 OD026553; 1 OT2 OD026548; 1 OT2 OD026551; 1 OT2 OD026555; IAA #: AOD 16037; Federally Qualified Health Centers: HHSN 263201600085U; Data and Research Center: 5 U2C OD023196; Biobank: 1 U24 OD023121; The Participant Center: U24 OD023176; Participant Technology Systems Center: 1 U24 OD023163; Communications and Engagement: 3 OT2 OD023205; 3 OT2 OD023206; and Community Partners: 1 OT2 OD025277; 3 OT2 OD025315; 1 OT2 OD025337; 1 OT2 OD025276.
Ethical considerations
Data for analysis was obtained from the UK Biobank and the All of Us database.
Consent to participate
Informed consent was obtained from these participants at study recruitment for either the UK Biobank or the All of Us database.
Author contribution(s)
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by R01 AG075884.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
The data supporting the findings of this study are available in the UK Biobank and the All of Us databases.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
