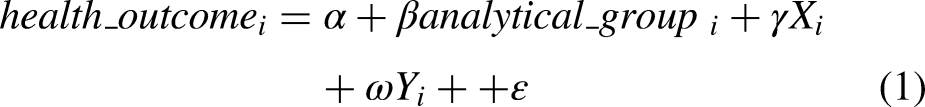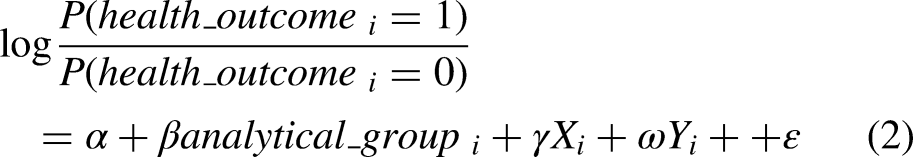Abstract
Background
Diagnosis of either diabetes or Alzheimer's disease presents substantial health challenges for older adults; however, the presence of both conditions concurrently significantly exacerbates these difficulties, complicating medical management and elevating the risk of adverse health outcomes.
Objective
Guided by syndemic theory and quantitative analyses, we examined the health profiles of older adults with dual diagnoses using national-level longitudinal data.
Methods
We utilized the Uniform Data Set (UDS) from the Alzheimer's Disease Research Centers (ADRCs) which contains demographics, neurological examination findings, and diagnoses for older adults. We analyzed 192,240 participant-visit records from 52,537 UDS participants in a pooled cross-sectional design. We conducted statistical analyses to examine disparities in health between those with dual diagnoses and those with a single diagnosis.
Results
We found significant associations between dual diagnoses and worsened outcomes across multiple domains, including systolic blood pressure, and utilization of medications, along with higher odds of anxiety and schizophrenia, compared to those with either condition alone or neither condition. For those with Alzheimer's disease only, we find higher levels diastolic blood pressure and higher odds of speech apraxia and depression compared to the other groups. The diabetes only group has higher levels of body mass index and higher odds of hypertension and sleep disorders. These findings underscore the heightened burden of comorbidity in this population.
Conclusions
Findings emphasize compounded challenges faced by older adults with dual diagnoses of diabetes and Alzheimer's disease—challenges that are not merely additive but synergistic in nature. The co-occurrence of epidemics among adults in the U.S. highlights the urgent need for policies that address their specific needs.
Introduction
The aging of the U.S. population has transformed the landscape of public health and healthcare delivery. The number of Americans aged 65 and older is projected to exceed 80 million by 2050, a demographic shift that will dramatically increase the prevalence of age-associated chronic conditions. 1 Two of the most prevalent conditions are Alzheimer's disease (AD) and diabetes. AD affected more than 6.9 million Americans aged 65 and older in 2024, while approximately one in three older adults lives with diabetes or prediabetes. 2 These conditions pose challenges to patients, families, and healthcare systems, especially as they lead to increased disability, healthcare utilization, and caregiving demands. When co-occurring, they may exert a compounded effect, amplifying challenges for patients, families, and healthcare systems. Yet, despite the growing population of older adults with both diabetes and AD, few studies have explored how these conditions jointly influence health outcomes. Addressing this gap is critical to inform integrated care approaches and health policy responses for an aging U.S. population.
A growing body of research has explored the interconnection between diabetes and cognitive decline. Type 2 diabetes has been associated with a 50–100% increased risk of developing dementia, through biological mechanisms including insulin resistance, hyperglycemia-induced oxidative stress, vascular damage, and chronic neuroinflammation.3,4 These findings have led some researchers to describe AD as “type 3 diabetes”, reflecting its connection to insulin dysfunction in the brain.5,6 While some studies provide evidence of elevated AD risk among people with diabetes,7,8 others point to a complex clinical overlap between the two conditions such as shared risk factors and challenges in disease management.9,10
Importantly, these disease patterns and their consequences are not experienced equally across demographic groups. Hispanic older adults, in particular, face disproportionately higher rates of both diabetes and AD. National estimates indicate that Hispanic individuals are nearly twice as likely as non-Hispanic Whites to develop diabetes and are projected to experience the sharpest increases in AD prevalence in coming decades.11,12 At the same time, Hispanic adults often live longer—a phenomenon known as the “Hispanic health paradox”—but spend more years managing chronic diseases. 13 This extended longevity raises concerns about a greater cumulative burden of multimorbidity. Structural barriers, including limited access to healthcare, underinsurance, linguistic isolation, and residential segregation, further exacerbate these risks among historically underserved groups. For example, Hispanic families are more likely to rely on unpaid caregivers and less likely to receive timely diagnoses or appropriate treatment. These disparities reflect not only potential biological vulnerability but also entrenched systemic inequities in care access, public health infrastructure, and social support in ethnically marginalized communities.
Despite increasing interest in the diabetes–AD nexus, relatively few studies have systematically examined health profiles of individuals with single versus dual diagnoses. Existing research often relies on small samples, self-reported diagnoses, or limited outcome measures. Our study addresses these limitations by leveraging a large, national dataset featuring clinician-assessed diagnoses and detailed measures of cognitive, physical, and behavioral health. Although we do not assess causal pathways in this work, our goal is to document differences in health outcomes and disparities across diagnostic groups, thereby advancing understanding of how dual diagnoses manifest in aging populations. To explore the intersecting challenges experienced by individuals with co-occurring diabetes and AD, we adopt a syndemic framework. Syndemic theory emphasizes that diseases do not occur or operate in isolation; rather, they cluster and interact in ways that intensify their impact. 14 In this context, the co-occurrence of diabetes and AD represents more than a dual burden; it is a syndemic condition shaped by interwoven biopsychosocial factors. A syndemic lens also carries important implications for clinical practice and public policy, underscoring the need for integrated, person-centered care models tailored to older adults managing multiple chronic conditions.
Methods
Data source
We use the Uniform Data Set (UDS), conducted at the Alzheimer's Disease Research Centers (ADRCs) across the United States. This data set has been collected on a rolling basis since September 2005 and includes participants with cognitive status ranging from cognitively normal to demented until the September 2024 data freeze. The sample for the UDS (N = 195,195) is not random but a form of convenient sampling, where each ADRC enrolls its participants using their own protocol, through clinician referral, self-referral by participants or family members, and active recruitment in community organizations. For this reason, the UDS is not a representative sample of the U.S. population and extrapolations should not be used to estimate national prevalence or incidence rates. However, its large sample size and breadth of clinical and demographic information does provide reasonable estimations of the disparities across different (diagnostic) groups.
Given the lower prevalence of comorbidities in younger populations, and to focus on older adults, we restricted our sample to those aged 50 and older, yielding 192,240 person-visit records drawn from 52,537 unique participants. Because our research question focuses on contemporaneous health status rather than within-person change, we employed a pooled cross-sectional framework in which each visit constitutes one observation, an approach consistent with previous literature focused on individual-visit outcomes rather than longitudinal progression.15,16 Throughout the paper we treat a visit as the unit of analysis but use ‘observation’ rather than ‘individual’ to avoid confusion.
Dependent variables
We explore two categories of health outcomes: clinical assessments and comorbid conditions. For test results, we use continuous measures of systolic and diastolic blood pressure, body mass index, the total number of reported medications at the visit. Regarding comorbidities and mental health outcomes, we use binary indicators for conditions reported in the UDS data set such as hypertension, sleep disorders, speech apraxia, depression, anxiety, obsessive compulsive disorder (OCD), and schizophrenia.
Independent variables
Our key independent variable is the categorical indicator for diagnoses of Alzheimer's disease or related dementias (ADRD), diabetes, both, or neither condition, based on classification available in the UDS. Demographics covariates include continuous measures for age and years of education, along with binary indicators for sex, marital status, Hispanic ethnicity, and a categorical measure for race. For other health-related factors, we include binary indicators for living alone, cigarette smoking (within the past 30 days), over consumption of alcohol, and a continuous measure for independence.
Covariates were selected a priori informed by syndemic theory and the Andersen behavioral model of health care.17,18 We controlled for predisposing (age, sex, race/ethnicity, education), enabling (marital status, living alone), behavioral (current smoking, over consumption of alcohol) and need factors (independence score, functional activities index) that are known to confound or modify the relationship between diabetes, AD and the studied outcomes. 19
More detail on the measures used is provided in Supplemental Table 1.
Analytical approach
For descriptive statistics, we conduct ANOVA tests for both the dependent and independent variables, comparing the means of those with double diagnoses or a single diagnosis. For the race category we also calculated a chi-squared test statistic. To examine the association between diagnosis status and health outcomes, we estimate a series of multivariate regression analysis following the specifications below:
Robustness checks
Because repeated visits from the same participant violate the assumption of independence, we conducted three person-level sensitivity analyses (full details in the Supplemental Methods): (i) Averaging method—all visits per participant collapsed to a single mean value; (ii) Most-recent method—only the latest visit retained; (iii) Bootstrapping method—one visit drawn at random per participant and the model re-estimated 10,000 times.
All modeling was performed in Stata/MP 18.0 (StataCorp); figures were produced in R 4.3.1 with ggplot2 and patchwork.
Results
Summary statistics
The analytical sample for this study consists of adults aged 50 and older (52,537 unique participants) in the UDS, totaling 192,240 observations between September of 2005 and the data freeze of September 2024, which includes data through August of 2024 (Table 1). Figure 1 illustrates the distribution of observation periods for individuals in the UDS. Many adults have multiple entries in the registry, where 77.3% of adults have data for 5 years or less, and that number goes up to 90.9% for 10 years of data. About 2% of individuals in the data set have interviews for 15 years or longer.
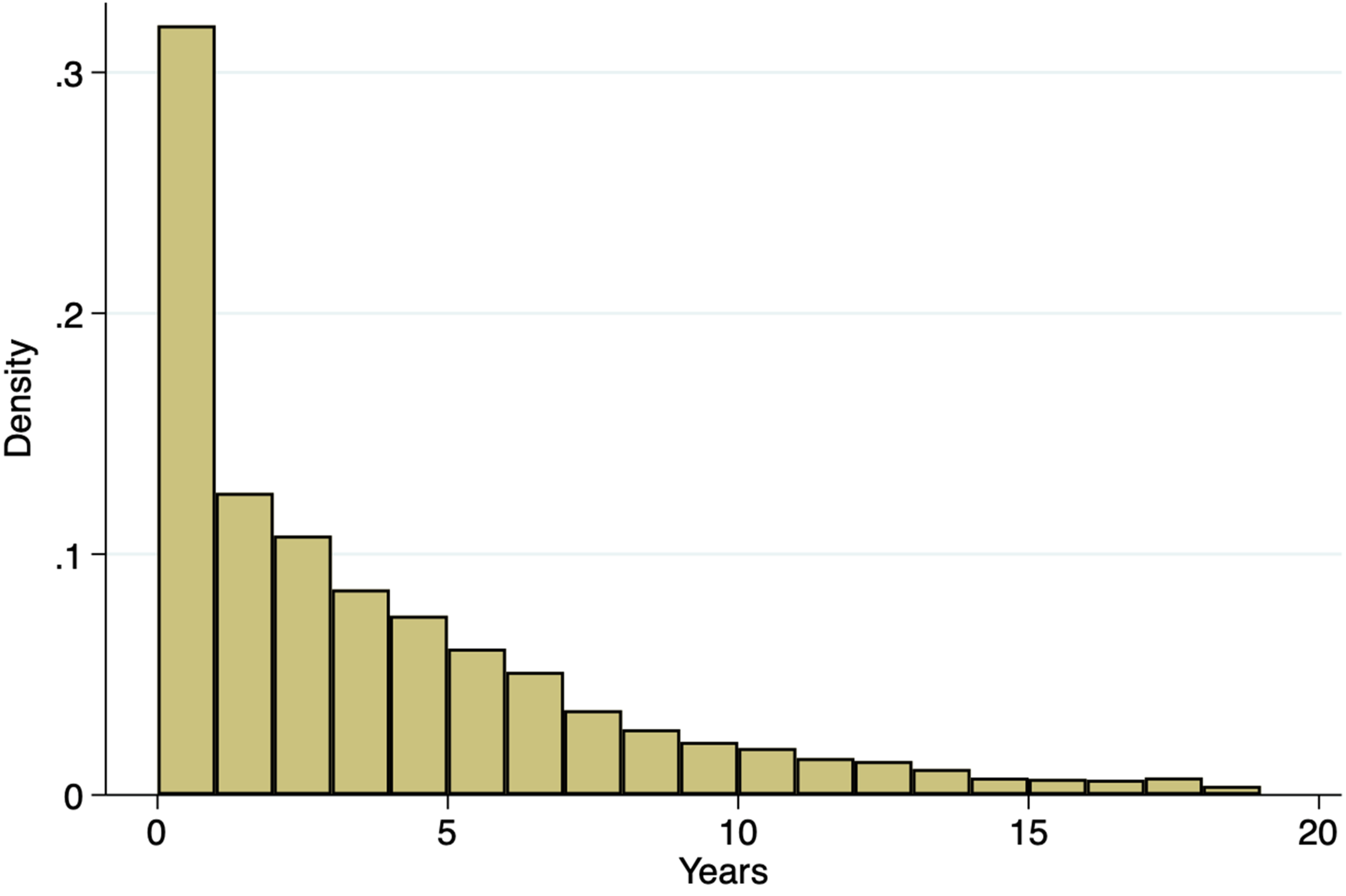
Years between first and last interview in the UDS, NACC sample (N = 192,240)
Summary statistics for NACC sample, 50 years and older (n = 192,240).
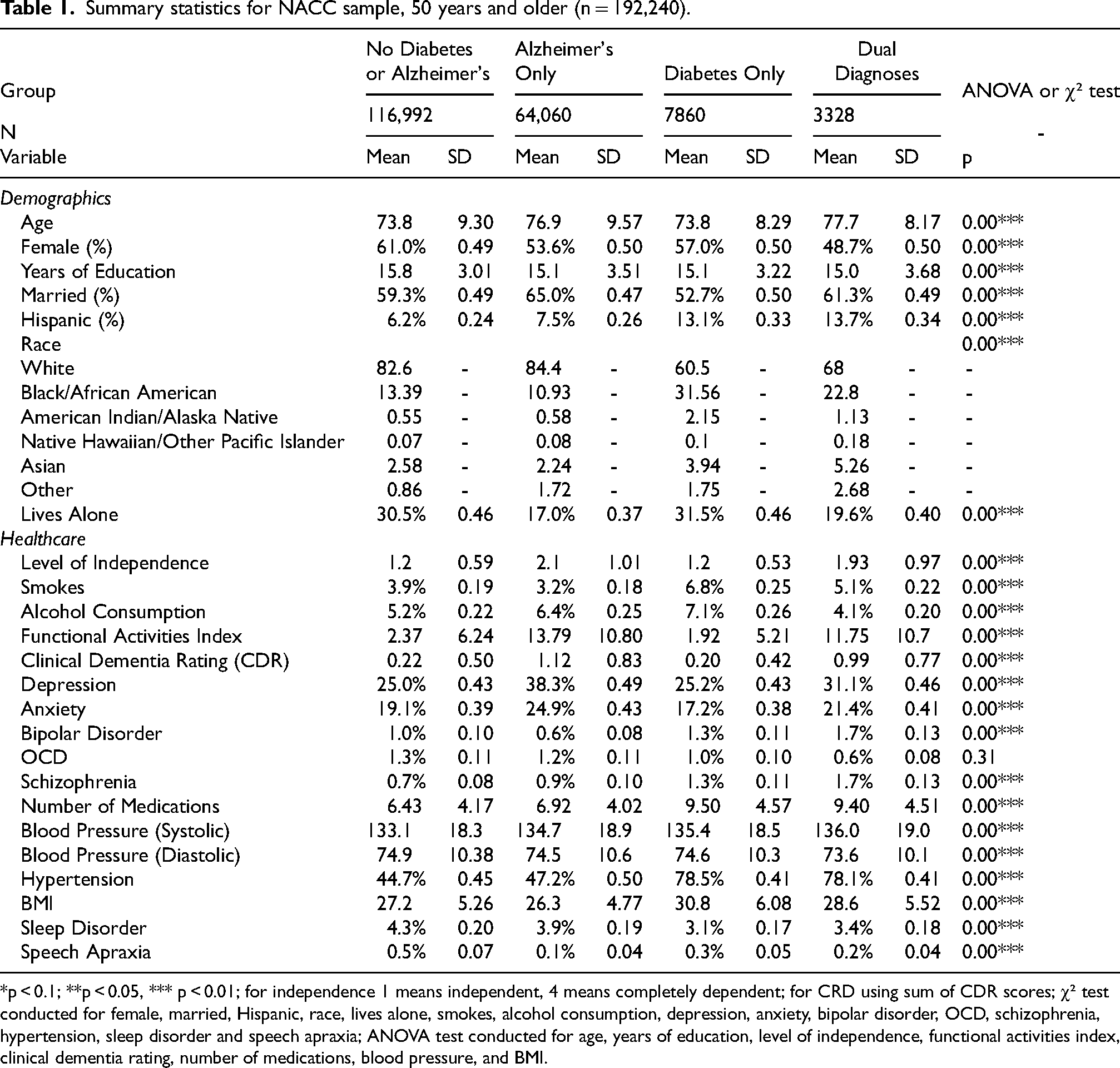
*p < 0.1; **p < 0.05, *** p < 0.01; for independence 1 means independent, 4 means completely dependent; for CRD using sum of CDR scores; χ² test conducted for female, married, Hispanic, race, lives alone, smokes, alcohol consumption, depression, anxiety, bipolar disorder, OCD, schizophrenia, hypertension, sleep disorder and speech apraxia; ANOVA test conducted for age, years of education, level of independence, functional activities index, clinical dementia rating, number of medications, blood pressure, and BMI.
Table 1 presents the descriptive statistics for demographic characteristics, health behaviors, and clinical outcomes across diagnostic groups. Overall distributions differed significantly across the four diagnostic groups for every variable shown (all omnibus ANOVA or χ² tests, p < 0.001), except for OCD, for which the difference was not significant (p = 0.31). Pairwise results of the post-hoc ANOVA tests for the continuous measures are presented in Supplemental Table 3. Among the 192,240 observations included in the analytical sample, 116,992 observations (60.9%) do not have a diagnosis of either AD or diabetes, 64,060 (33.3%) report a diagnosis of AD only, 7860 (4.1%) report a diagnosis of diabetes only, and 3328 (1.73%) report dual diagnoses of AD and diabetes.
On the demographic distribution of the sample, the age of respondents varies significantly across groups, where those with only AD and dual diagnoses (76.9 and 77.7, respectively) report higher ages than those without a diagnosis and those with only diabetes. The percentage of female respondents and average years of education are lower among those with AD only (53.6%; 15.1 years) and those with dual diagnoses (48.7%; 15 years, respectively) relative to those without diagnoses or diabetes only. Conversely, the percentage of married respondents is higher among those with AD only (65%) and with dual diagnoses (61.3%) than those without a diagnosis or those with diabetes only.
On ethnicity and race, the percentage of Hispanic respondents is notably higher among those with only diabetes and dual diagnoses (13.1% and 13.7%) compared to the other groups. The AD only group has the highest percentage of White respondents, while the diabetes only group includes the greatest proportion of Black/African American respondents. The dual diagnoses group has a more diverse racial composition than those without diagnoses or those with AD only, with 68% White and 22.8% Black/African American respondents. Finally, the percentage of respondents living alone is higher among those with only diabetes (31.5%) or no diagnosis (30.5%), than those with only AD (17%), and those with dual diagnoses (19.6%).
For health behaviors, smoking is least common in the AD only group (3.2%) and most prevalent in the diabetes only group (6.8%), with the dual diagnoses group falling in between (5.1%). Similarly, the rate of over consumption of alcohol is lower among those with dual diagnoses (4.1%) compared to those with only diabetes, no diagnosis, or only AD. For measures of cognitive impairment, the Functional Activities Index scores (11.75) and CDR® scores 20 (0.99) are lower among those with dual diagnoses compared to those with AD only (13.79 and 1.12 respectively) and higher to those with diabetes only and no diagnoses.
Mental health conditions also differ across groups. Rate of depression (31.1%) and anxiety (21.4%) are higher among those with dual diagnoses than among those without a diagnosis or with only diabetes, although lower than those with only AD. Bipolar disorder (1.7%) and schizophrenia (1.7%) are most prevalent among those with dual diagnoses. Conversely, OCD is least common in this group (0.6%) compared to all others. The number of medications taken is also higher among those with dual diagnoses (9.4), but lower than for those with diabetes only.
Overall, observations with dual diagnoses have the highest average systolic blood pressure (136 mmHg), while diastolic blood pressure remains comparable across groups. Hypertension is higher among those with dual diagnoses (78.1%) compared to those without a diagnosis and those with only AD, but lower than those with only diabetes (78.5%). Body mass index (BMI) follows a similar pattern, with the dual diagnosis group averaging 28.6 kg/m²—higher than the AD only and no-diagnosis groups, but lower than the diabetes only group (30.8 kg/m²). The percentage of respondents with sleep disorders and speech apraxia is similar across all diagnostic categories.
To provide additional therapeutic context for our findings, class-specific medication use patterns are presented in Supplemental Table 2.
Health measurements analysis
For the regression analysis of the continuous health measurements, we conducted multivariate ordinary least squares analysis (Table 2). For measures of blood pressure, we find that having dual diagnoses is associated with an increased average of systolic blood pressure (Model 1) by 5.33 points, compared to those without either condition, a much greater association than that observed in groups with only a single diagnosis. In contrast, Model 2 reveals no significant association between dual diagnoses and diastolic blood pressure. However, observations with AD only show a small but statistically significant increase in diastolic pressure (0.64 points) relative to the reference group. In both blood pressure models, differences in demographic variables such as age, sex, years of education are significantly different, although sex is not significant for systolic blood pressure, and years of education is not significant for diastolic blood pressure. Notably, observations reporting Hispanic ethnicity have, on average, 0.77 higher systolic blood pressure and 0.36 points lower diastolic pressure compared to non-Hispanics. Finally, low levels of dependence are significantly associated with lower blood pressure on both measures.
OLS regressions for NACC sample by analytical group.
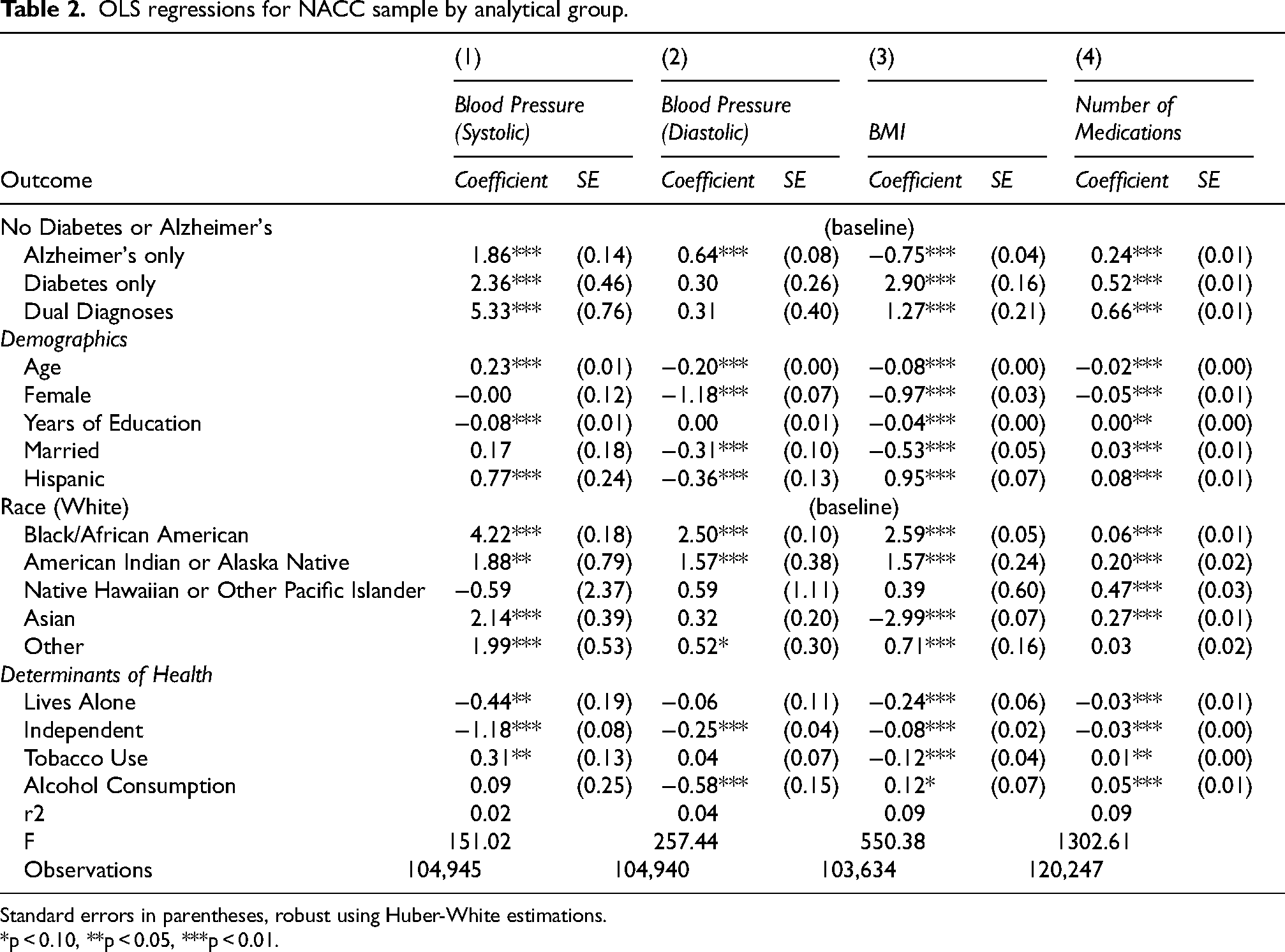
Standard errors in parentheses, robust using Huber-White estimations.
*p < 0.10, **p < 0.05, ***p < 0.01.
For the BMI outcome, Model 3 shows that individuals with AD only have significantly lower scores compared to those with no diagnosis. In contrast, having diabetes or dual diagnoses is associated with BMI scores that are 2.90 and 1.27 points higher, respectively. All demographic variables are significantly associated with differences in BMI, with Hispanic ethnicity being the important factor positively associated with higher BMI scores. Other covariates, including age, female sex, education, and marital status, show negative associations with BMI. Medication use is explored in Model 4, which reveals that all diagnostic groups have a higher number of reported medications compared to those without a diagnosis, but the diabetes only and dual diagnoses groups show higher coefficients, which corresponds with the medical needs of patients with diabetes. Demographic characteristics are significantly associated with differences in the count of medications, where age and female sex predict lower number of medications, and both married and Hispanic predict higher average number of medications.
Comorbidities analysis
For the comorbidities models, we present the results of logistic regression analyses in Table 3. Regarding hypertension in Model 1, those with diabetes only and dual diagnoses have substantially higher odds of hypertension (264% and 222%, respectively) compared to those without either condition. All demographic variables are significantly associated with hypertension. Hispanic respondents exhibit 42% higher odds of hypertension compared to non-Hispanic respondents. For sleep disorders and speech apraxia (Models 2 and 3), the groups with AD only and dual diagnoses exhibit lower odds of sleep disorders compared to the reference group, while those with diabetes only do not differ significantly. In Model 4, we find that AD diagnosis and dual diagnoses are significantly associated with higher levels of depression, 61% and 32% respectively, while diabetes diagnosis is not significantly associated with depression. Demographic variables, as well as determinants of health do associate significantly with varying levels of depression. For anxiety, in Model 5, AD only and dual diagnoses are significantly associated with higher odds for anxiety, 38% and 56% respectively. It is also worth noting that higher levels of dependence and over consumption of alcohol are significantly associated with increased odds of anxiety. For OCD, in Model 6, none of the three analytical groups are associated with changes in odds of OCD diagnosis compared to the reference group. Older age, being female, being married, and identifying as Black or African American are associated with lower odds of OCD, whereas greater independence and higher levels of alcohol consumption are associated with increased odds of OCD. Finally, Model 7 outlines the relationships with schizophrenia. In this model, the dual diagnoses group has significantly higher odds (220%) than those without a diagnosis. AD only and diabetes only respondents also have higher odds of schizophrenia, 45% and 82% respectively, compared to those with no diagnosis, but in a smaller effect size than dual diagnosis. Tobacco use and excessive alcohol consumption are strong predictors, with smokers and those with elevated alcohol intake exhibiting 22% and 76% higher odds, respectively, of a schizophrenia diagnosis.
Logistic regressions for NACC sample by analytical group (odds ratios).
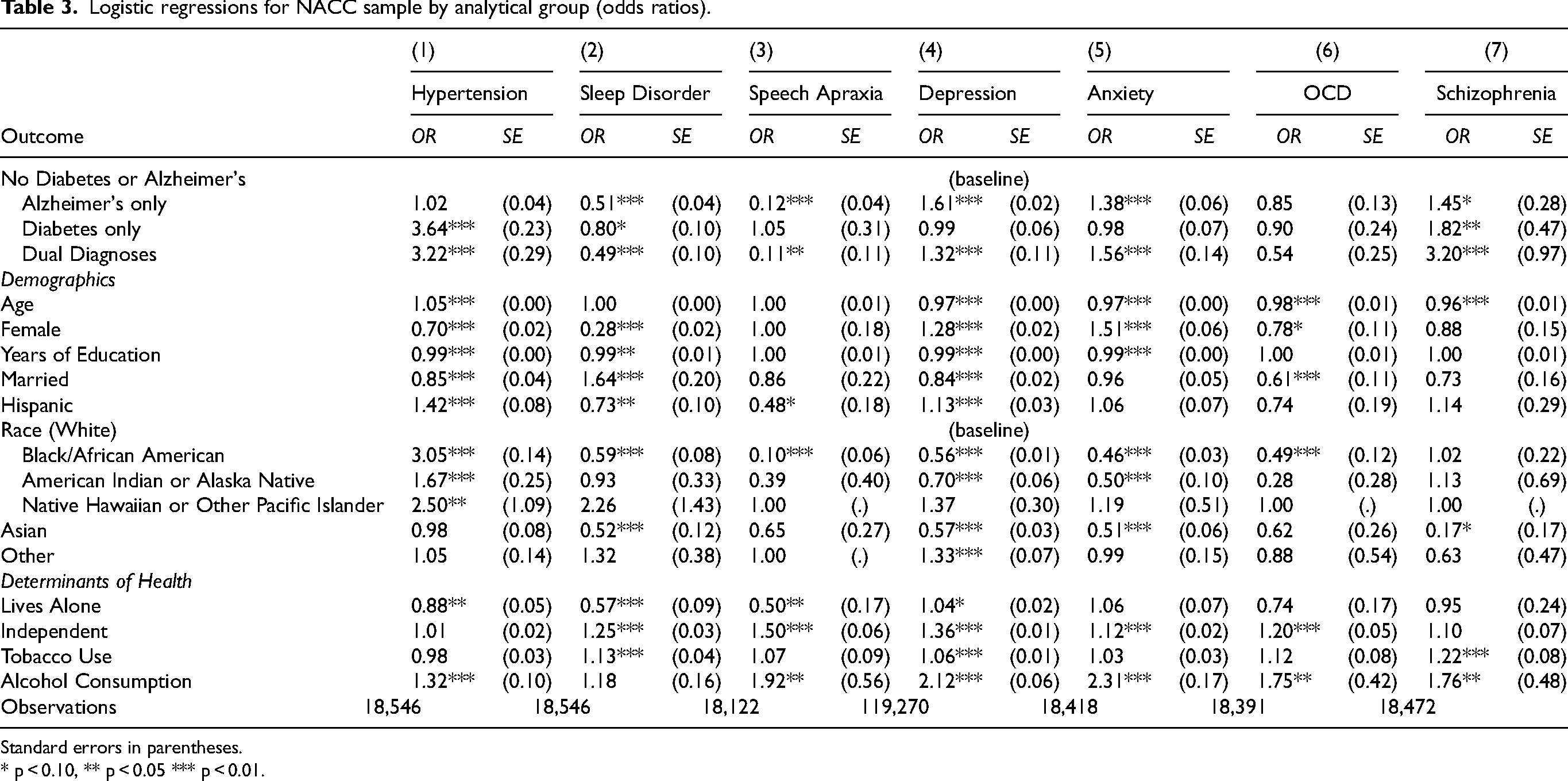
Standard errors in parentheses.
* p < 0.10, ** p < 0.05 *** p < 0.01.
Results were highly consistent when analyses were repeated at the person level rather than the visit level (Supplemental Tables 4 and 5). Direction, magnitude and statistical significance of key coefficients remained stable across the Averaging, Most-recent and Bootstrapping specifications, confirming that our pooled cross-sectional findings are not an artefact of multiple observations per participant.
Discussion
Main findings
An analysis of data from over 190,000 adults aged 50 years and older in the UDS demonstrates that the coexistence of diabetes and AD is associated with a distinctly more adverse clinical and mental-health profile than either condition alone. Participants with both conditions had the highest mean systolic blood pressure, the heaviest medication load, markedly greater odds of hypertension and schizophrenia, and a higher prevalence of depression and anxiety when compared with respondents who had neither diagnosis. In contrast, diastolic pressure and the probability of sleep disorders did not differ meaningfully from the reference group, underscoring that risk amplification is outcome-specific. These associations remained statistically significant after adjusting for demographic composition and key health-behavior covariates, lending empirical support to the syndemic hypothesis that diabetes and AD interact in a synergistic rather than merely additive fashion. 21
Biological mechanisms
Several interrelated pathophysiological pathways help to elucidate the synergistic interaction between diabetes and AD. One such pathway involves metabolic–vascular convergence, wherein hyperglycemia, insulin resistance, and microvascular injury lead to both accelerated neurodegeneration and increased arterial stiffness. 22 This mechanism likely underlies the observed elevation in systolic, but not diastolic, blood pressure in the dual-diagnosis group. Sustained mid-life hypertension, well documented as a risk factor for dementia, remodels cerebral vessels, compromises the blood–brain barrier, and fosters white-matter injury that advances both vascular and Alzheimer-type pathology. 23 Hypertension and type 2 diabetes reinforce each other through endothelial dysfunction, inflammation, dyslipidemia, obesity, and renin–angiotensin–aldosterone activation, causing disproportionate cardiovascular morbidity and mortality when both are present. 24 Low-grade systemic inflammation, characteristic of both diseases, may heighten affective symptoms and aligns with the observed excess depression and anxiety. 25 Finally, the 47% higher medication count in the dual-diagnosis group reflects the need to manage glycaemia, blood pressure, and neuropsychiatric symptoms concurrently; such polypharmacy is increasingly common in type 2 diabetes 26 and, in the context of AD, is linked to more pronounced symptom burden, functional loss, and poorer quality of life. 27
Association between AD and schizophrenia
The study also identifies a significant association between AD and elevated odds of schizophrenia. A meta-analysis covering more than five million adults has demonstrated that schizophrenia nearly doubles the subsequent risk of dementia, with an especially large excess burden in women. 28 Accelerated cognitive decline and neurodegenerative changes intrinsic to schizophrenia, together with extrinsic contributors such as vascular and metabolic comorbidities, adverse health behaviors, and long-term antipsychotic use, may converge to hasten neuronal injury. These findings highlight the importance of dementia-focused surveillance and tailored risk-reduction strategies in people living with schizophrenia.
Health-equity implications
Hispanic and Black or African-American adults were disproportionately represented in the diabetes-only and dual-diagnosis groups, corroborating evidence that structural inequities in education, income, and healthcare access concentrate metabolic risk in minoritized communities. 29 Multivariable models revealed significantly higher odds of hypertension among Black or African-American, American Indian or Alaska Native, and Native Hawaiian or Other Pacific Islander participants, and among those of Hispanic ethnicity, relative to non-Hispanic White peers, mirroring national patterns in prevalence, awareness, and control of high blood pressure. 30 Interestingly, once AD was present, Hispanic respondents exhibited lower diastolic pressure and fewer sleep disorders, suggesting cultural or familial buffering mechanisms such as multigenerational caregiving that merit further study. 31 Culturally adapted diabetes-management programs, routine cognitive screening in high-risk primary-care settings, and caregiver-centered support should therefore form the cornerstone of interventions aiming to mitigate the dual burden. 32
Clinical and policy relevance
These findings highlight the value of co-located chronic-disease clinics that integrate geriatric, endocrinological, and neurological expertise. Evidence from Hong Kong's Multidisciplinary Risk Assessment and Management Program for Diabetes indicates that such integration can reduce incident dementia by nearly a third among patients with type 2 diabetes. 33 Targeting systolic blood pressure remains critical, as shown in the SPRINT-MIND trial, where intensive systolic control below 120 mm Hg lowered the risk of cognitive impairment among older adults. 34 Routine mental-health screening is warranted given the higher prevalence of depression, anxiety, and schizophrenia in the dual-diagnosis group. 35 Effective care also relies on a trained workforce capable of managing overlapping symptoms of hypoglycemia, delirium, and dementia, while caregivers need practical guidance for synchronizing insulin administration and other treatments with fluctuating cognitive abilities.36,37
Strengths and limitations
Strengths include clinician-verified AD diagnoses, a geographically diverse sample, and simultaneous modelling of biomedical, psychosocial, and behavioral factors that together offer a granular view of multimorbidity in later life. Nevertheless, the convenience sampling strategy limits generalizability; diabetes status, though clinician-recorded, may rely partly on self-report; cross-sectional averaging obscures longitudinal trajectories; and residual confounding—for example diabetes duration, glycemic control, or APOE ε4 status—cannot be ruled out. Although we modelled participant-visits, sensitivity analyses using person-level data produced nearly identical estimates, mitigating concerns about within-person correlation.
Future directions
Longitudinal work should examine whether deteriorating glycemic control precipitates faster cognitive decline or whether emerging cognitive deficits erode diabetes self-management. Incorporating biomarkers such as HbA1c and neurofilament light chain, continuous glucose-monitoring data, and neighborhood-level deprivation indices will strengthen causal inference and illuminate contextual amplifiers of risk. Pragmatic trials of culturally concordant, team-based care are needed to determine whether integrated management can reduce the excess morbidity documented here.
Conclusion
Diabetes and AD constitute a syndemic in older U.S. adults that amplifies cardiovascular strain, polypharmacy, and neuropsychiatric morbidity, especially in racially and ethnically minoritized populations. Addressing this dual burden will require policy and clinical initiatives that transcend disease silos, prioritize equity, and support patients and caregivers across the continuum of multimorbidity.
Supplemental Material
sj-docx-1-alz-10.1177_13872877251380260 - Supplemental material for Profile and disparities in health outcomes for older adults with dual diagnoses of diabetes and Alzheimer's disease
Supplemental material, sj-docx-1-alz-10.1177_13872877251380260 for Profile and disparities in health outcomes for older adults with dual diagnoses of diabetes and Alzheimer's disease by Alfonso Rojas-Alvarez, Hyeran Chung, Jose Eduardo Cabrero Castro and Emre Umucu in Journal of Alzheimer's Disease
Footnotes
Acknowledgements
The authors have no acknowledgments to report.
Ethical considerations
Not applicable.
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Author contribution(s)
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Dr Rojas-Álvarez and Dr Chung were supported by UTEP FIRST Grant, 1U54CA280922, awarded to the University of Texas at El Paso by the National Institutes of Health. Dr Cabrero-Castro is supported by the following federal grants: NIA P30AG043703 USC AD/ADRD RCMAR, NIA 1RF1AG068988-01A1. Dr Umucu is supported by the following federal grants: AA-FAiM R01AG054059 (NIH-NIA), 1U1QHP53051 (HHS-HRSA), and 1P50MD019494-01 (NIH-NIMHD). The NACC database is funded by NIA/NIH Grant U24 AG072122. NACC data are contributed by the NIA-funded ADRCs: P30 AG062429 (PI James Brewer, MD, PhD), P30 AG066468 (PI Oscar Lopez, MD), P30 AG062421 (PI Bradley Hyman, MD, PhD), P30 AG066509 (PI Thomas Grabowski, MD), P30 AG066514 (PI Mary Sano, PhD), P30 AG066530 (PI Helena Chui, MD), P30 AG066507 (PI Marilyn Albert, PhD), P30 AG066444 (PI David Holtzman, MD), P30 AG066518 (PI Lisa Silbert, MD, MCR), P30 AG066512 (PI Thomas Wisniewski, MD), P30 AG066462 (PI Scott Small, MD), P30 AG072979 (PI David Wolk, MD), P30 AG072972 (PI Charles DeCarli, MD), P30 AG072976 (PI Andrew Saykin, PsyD), P30 AG072975 (PI Julie A. Schneider, MD, MS), P30 AG072978 (PI Ann McKee, MD), P30 AG072977 (PI Robert Vassar, PhD), P30 AG066519 (PI Frank LaFerla, PhD), P30 AG062677 (PI Ronald Petersen, MD, PhD), P30 AG079280 (PI Jessica Langbaum, PhD), P30 AG062422 (PI Gil Rabinovici, MD), P30 AG066511 (PI Allan Levey, MD, PhD), P30 AG072946 (PI Linda Van Eldik, PhD), P30 AG062715 (PI Sanjay Asthana, MD, FRCP), P30 AG072973 (PI Russell Swerdlow, MD), P30 AG066506 (PI Glenn Smith, PhD, ABPP), P30 AG066508 (PI Stephen Strittmatter, MD, PhD), P30 AG066515 (PI Victor Henderson, MD, MS), P30 AG072947 (PI Suzanne Craft, PhD), P30 AG072931 (PI Henry Paulson, MD, PhD), P30 AG066546 (PI Sudha Seshadri, MD), P30 AG086401 (PI Erik Roberson, MD, PhD), P30 AG086404 (PI Gary Rosenberg, MD), P20 AG068082 (PI Angela Jefferson, PhD), P30 AG072958 (PI Heather Whitson, MD), P30 AG072959 (PI James Leverenz, MD).
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.