Abstract
Background
Currently there is no effective treatment to reverse Alzheimer's disease, thus prevention is crucial. Patients with mild cognitive impairment (MCI) have higher rate of progression to dementia. Two commonly used cognitive tests, Mini-Mental State Examination (MMSE) and Clock Drawing Test (CDT), have only modest accuracy at predicting conversion from MCI to dementia, whereas other potentially predicting factors are difficult to implement in clinical practice.
Objective
The aim of this study was to assess the performance of a combination of simple cognitive tests and routine clinical data to predict onset clinical dementia over 3-year follow-up in a cohort of outpatients with MCI.
Methods
Medical history was collected, and an advanced neuropsychological assessment was performed at baseline and at follow-up.
Results
98 participants were included (mean age 76.6 ± 4.5 years), and 49 developed dementia. Participants who developed dementia had significantly lower MMSE and CDT scores, as well as higher presence of hypercholesterolemia at the baseline evaluation. In the multivariate binary logistic regression analysis, the OR were 0.71 (CI95% 0.58–0.87) for MMSE, 0.77 (CI95% 0.65–0.93) for CDT, and 3.9 (CI95% 1.4–10.9) for hypercholesterolemia. The ROC curve combining these three factors obtained a value of AUC of 0.825 (CI95% 0.74–0.91). A nomogram was then developed for the risk of evolution from MCI to dementia at 3 years.
Conclusions
Combining history of hypercholesterolemia with CDT and MMSE tests, may result in a simple prediction model to predict the onset of dementia over 3 years.
Introduction
Dementia presents as an important challenge in aging society. Importantly, there are no existing effective treatments to stop or reverse this medical condition and thus prevention is crucial. 1
The term mild cognitive impairment (MCI) is an intermediate stage between normal cognitive function to dementia, although it is important to note that those with MCI do not always develop dementia. In MCI, cognitive decline is greater than can be expected for an age and degree of education; but the level of cognitive decline does not significantly impact on daily activities.2,3
Prevalence estimates of MCI among populations of community-dwelling older adults are as high as 22% among those aged 71 years and older. 4 Those with MCI present a higher rate of progression to dementia than people without this condition.
Within five years, approximately 27–42% of individuals with MCI progress to dementia, while 40–50% remain stable and 9–28% may revert to normal cognition. However, even those who revert often remain at elevated risk of future decline, underscoring the need for accurate prognostic stratification at an early stage.5,6
As MCI can lead to dementia, it is mandatory to identify individuals at higher risk of progression, to select the best tailored interventions in order to slow down or stop a potential evolution.
While cholinesterase inhibitors (ChEIs), such as donepezil, have shown a lower rate of progression to Alzheimer's disease (AD) during the first 12 months of treatment, the rate of progression to AD after three years was not lower among patients treated with donepezil than among those given placebo. 7 Moreover, their use is associated with a high rate of adverse effects and no consistent improvement in cognitive scores.8,9 As a result, clinical guidelines (e.g., American Academy of Neurology) currently do not recommend ChEIs in MCI patients.
Meta-analyses indicate that physical activity, particularly resistance training, produces moderate-to-large improvements in global cognitive function in older adults, including those with MCI. 10 Multidomain, home-based lifestyle interventions incorporating diet, exercise, cognitive stimulation, and stress management have shown cognitive benefits in the majority of randomized controlled trials. 11 Socio-cognitive therapies such as music therapy, reminiscence, and validation therapy are associated with improved mood, enhanced quality of life, and reduced use of psychotropic medications in individuals with cognitive impairment. 12 Finally, digital cognitive training, including computerized cognitive training (CCT) and virtual reality (VR)-based platforms, has demonstrated efficacy in enhancing attention, memory, and executive function, and offers a scalable, patient-friendly option for cognitive rehabilitation.13,14
Even if the usefulness of neuropsychological measures for detecting early cognitive change is well established, 15 they appear to have poor accuracy in detecting people at higher risk of conversion from MCI to dementia.
Traditionally, commonly used cognitive tests for dementia screening include the Mini-Mental State Examination (MMSE) and the clock drawing test (CDT). 16 While MMSE has been found to predict conversion from MCI to AD,17–19 it is not sufficient as a stand-alone single-administration test in the identification of MCI patients who could develop dementia. 20 Similarly, even though the CDT seems to detect the degrees of cognitive impairment earlier than other cognitive screening tests, 21 its role as predictor of conversion from MCI to dementia has only limited accuracy. 22
Likewise, while several risk factors have been identified for MCI, such as age, sex, genetic factors, level of education, vascular risk factors, cardiovascular outcomes and imaging biomarkers, 23 their role on progression to dementia are unclear. Importantly, potential markers of possible evolution to dementia seem difficult to apply in everyday clinical practice, as they include advanced neuroimaging data and cerebrospinal fluid (CSF) biomarkers.24,25
As a result, even if there are several clinical prediction models in literature from the conversion of MCI to dementia, they are usually complex and frequently include machine-learning elaborations of several variables not routinely collected in clinical practice.26–30 Also adding elicited clinician knowledge to these variables seem not to improve dementia risk prediction models. 31
Given this background, the aim of the present study was to assess the performance of a combination of simple-to-administer cognitive tests and routinely collected clinical data, to predict clinical dementia over 3-year follow-up in a cohort of outpatients with MCI at enrollment.
Methods
The study followed the TRIPOD (Transparent Reporting of a multivariable prediction model for Individual Prognosis or Diagnosis) indications. 32 The checklist is reported in the Supplemental Material.
Subjects
Participants were evaluated on an outpatient basis at the Center for Cognitive Disorders and Dementia (CDCD) of Geriatrics at Dolo Hospital, during the period between 2014 and 2023. After initial assessment leading to the diagnosis of MCI, participants underwent an annual follow-up. Those who did not have complete data regarding baseline neuropsychological assessment were later excluded from the initial sample. Those who progressed to conditions other than Alzheimer's dementia or to Alzheimer's with cerebral vasculopathy (i.e., pure vascular dementia, Lewy body dementia, and primary progressive aphasia) were also excluded. All data analyzed were collected as part of routine diagnosis and treatment. The protocol of this retrospective study was approved by our ethics committee (approval number 6107/U3/24) and then included in the context of the Blood biomarkErs for the identificATion of Alzheimer's disease (BEAT Alzheimer) study on 28 August 2024. We collected the informed consent in the context of the normal clinical activities.
MCI and diagnosis of dementia
MCI was defined as the presence of a documented deficit in memory or another cognitive domain, without (single domain) or with (multiple domain) impairment in other cognitive domains, in an individual who did not meet the criteria for the diagnosis of dementia. 33 The diagnosis of MCI was made by qualified physicians, with a specific training in dementia for more than 10 years. The diagnosis of dementia was accomplished according to the criteria of DSM IV. 34
Factors
Personal data (e.g., educational degree) and remote medical history were collected by qualified personnel. The presence or absence of conditions associated with the onset of dementia, such as high blood pressure, chronic coronary artery disease, atrial fibrillation, type 2 diabetes mellitus, and the presence of hypercholesterolemia or hypertriglyceridemia, was also reported. All the medical conditions were ascertained by a trained physician using data from medical records, medications’ history, physical exam and laboratory's findings collected during the standard visit.
An advanced neuropsychological assessment was performed at baseline and at follow-up evaluations; all assessments were performed by one neuropsychologist with experience working at dementia centers. The neuropsychological symptom assessment included the following cognitive tests: MMSE, 35 CDT, 36 prose memory, 37 cognitive estimates, 38 Frontal Assessment Battery (FAB), 39 Verbal Fluency Test (FAS), 40 attentional matrices, 41 constructive apraxia test, 42 and direct and reverse digit span. 43 Residual abilities in basic activities of daily living (BADL) 44 and instrumental activities of daily living (IADL) 45 were also assessed. For most of the participants (76%), brain imaging data (CT or MRI) was also available at baseline.
Statistical analysis
The population was divided into 2 groups: patients with baseline diagnosis of MCI who remained stable at 3 years and patients who, within 3 years, had progressed to dementia. Since the cohort was limited in sample size, we decided not to perform an internal validation. Data are reported as means and standard deviations for continuous variables after analyzing normality and as frequencies and percentages for categorical variables. A chi-square test was used to compare prevalences according to progression or not and Student's t-test for independent samples for continuous variables.
Therefore, a model was prepared with the statistically significant variables between the two groups (p value < 0.05). To evaluate the relationship between the significantly different variables between the two groups at baseline and the probability of developing dementia after 3 years, a binary logistic regression, adjusted for age and education level, was performed. Data from these analyses were reported as odds ratios (ORs) with 95% confidence intervals (95%CI). All variables were tested for multicollinearity: a value of VIF (variance inflation factor) >2 was taken as a reason for exclusion, but no factor was excluded for this reason. The accuracy of the model with the statistically significant factors at the binary logistic was assessed through the area under the receiver operating characteristics curve (AUC). Traditional measures of model performance were calculated, including c-index for discrimination and the Hosmer–Lemeshow goodness-of-fit statistic for calibration. Moreover, we reported information about Mathew's Correlation Coefficient 46 and Brier Score. 47 Finally, the Youden index was calculated for the individually entered continuous variables and, therefore, a nomogram for the risk of evolution from MCI to dementia at 3 years was constructed with a score from 0 to 14, based on the weight the factors had at the multivariate analysis. For the best point, we reported data about sensitivity (Se) and specificity (Sp). 48 The prediction score was created based on the parameter estimates (β coefficients) from the final regression model.
The analyses were performed with the statistical package SPSS for Mac, version 20.
In writing this paper, we followed the guidelines for compiling predictive models in biochemical research suggested by Luo. 49
Results
Among 156 potentially eligible participants, 122 participants were included in the study who were diagnosed at the baseline visit with MCI and who had subsequently been followed up with periodic visits for at least 3 years. Twenty-four patients were later excluded from the initial sample, i.e., 11 subjects who had evolved to a form of dementia other than Alzheimer's disease or mixed degenerative and vascular dementia (7 with vascular dementia, 3 with Lewy body dementia, and 1 with primary progressive aphasia) and 13 subjects because they did not have a complete neuropsychological assessment at the initial visit. The mean age of the remaining 98 participants was 76.6 ± 4.5 years (58.2% as females).
Table 1 shows the data of the 98 initial patients grouped according to those who remained stable over the 3 years of follow-up (N = 49) and those who evolved to dementia (N = 49). Fifty-eight percent of the participants were female. The sample was homogeneous in terms of age and education level.
Characteristics at baseline between stable (n = 49) participants and those with evolution to dementia (n = 49).
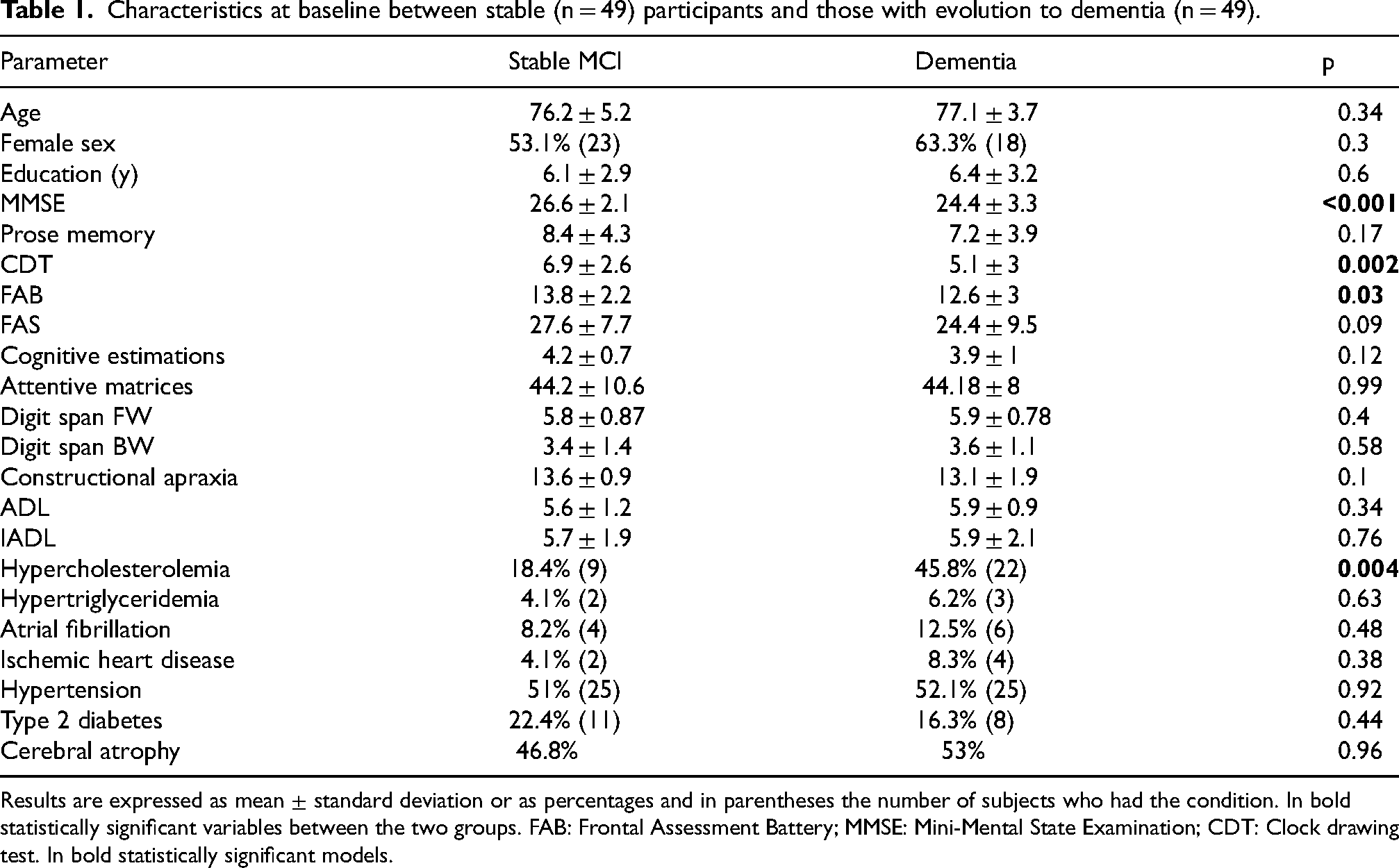
Results are expressed as mean ± standard deviation or as percentages and in parentheses the number of subjects who had the condition. In bold statistically significant variables between the two groups. FAB: Frontal Assessment Battery; MMSE: Mini-Mental State Examination; CDT: Clock drawing test. In bold statistically significant models.
Compared with participants who remained stable, patients who developed dementia had significantly lower MMSE (24.4 ± 3.3 versus 26.6 ± 2.1, p < 0.001), CDT (5.1 ± 3 versus 6.9 ± 2.6, p 0.002) and FAB (12.6 ± 3 versus 13.8 ± 2.2, p 0.003) scores. The other psychometric tests were not different between the two groups examined.
In those with dementia at follow-up, the presence of hypercholesterolemia as a comorbidity was also significantly higher (45.8% versus 18.4%, p = 0.004), while there was no difference in the prevalence of other cardiovascular diseases, the presence of type 2 diabetes mellitus (p > 0.05) or the presence of brain atrophy.
At multivariate binary logistic regression analysis (Table 2), considering the relationship between the statistically significant characteristics at baseline of the whole sample and the incidence of dementia in the follow-up period, after correction for age and sex, the FAB values (OR 1.5, CI95% 0.83–1.33, p = 0.67) did not show significant association with the development of dementia, while MMSE and CDT scores as well as the presence of hypercholesterolemia remained statistically significant. The Odds Ratios obtained by analyzing the model with only the 3 variables under consideration were OR 0.71 (CI95% 0.58–0.87, p = 0.001) for MMSE, and OR 0.77 (CI95% 0.65–0.93, p = 0.05) for CDT, while the OR for the presence of hypercholesterolemia was 3.9 (CI95% 1.4–10.9, p = 0.01). The negative OR data for MMSE and CDT should be interpreted as for each additional point on the MMSE or CDT, participants had a reduced risk of developing dementia by 29% and 23%, respectively.
Binary logistic analysis.
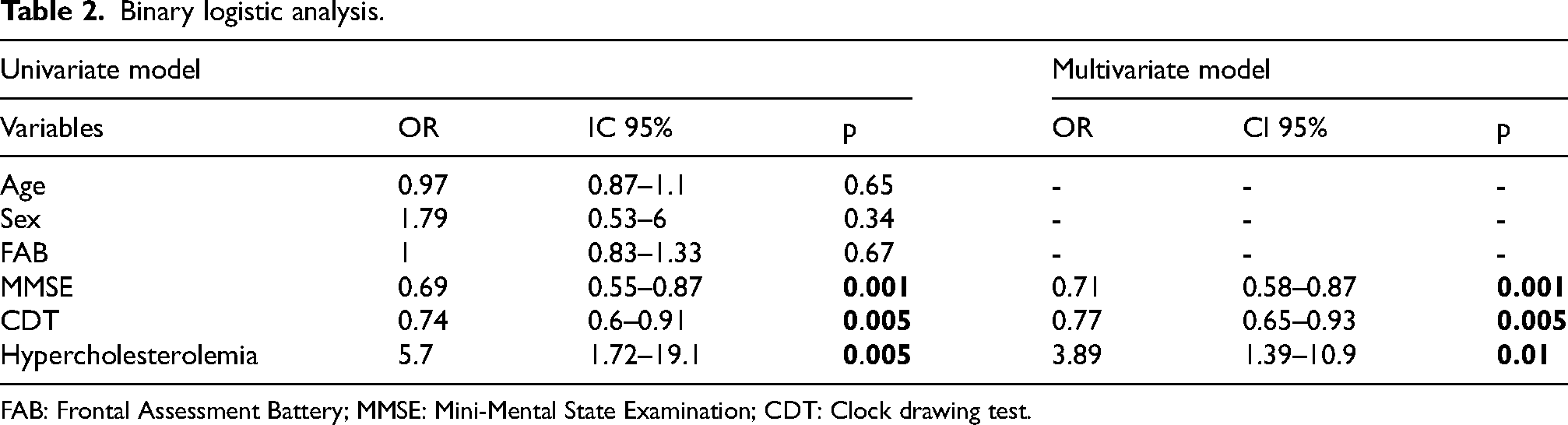
FAB: Frontal Assessment Battery; MMSE: Mini-Mental State Examination; CDT: Clock drawing test.
The ROC curve (Figure 1) was then constructed to evaluate the accuracy of the model containing the 3 statistically significant variables at the binary logistic, with an obtained value of area under the curve of 0.825 (CI95% 0.74–0.91, p < 0.001). Removing hypercholesterolemia that can still be limited to community-based settings, including primary care, outpatient clinics, and home visiting programs significantly worsens the AUC (AUC = 0.63; 0.53–0.73, p < 0.001). Adding age, sex, and FAB score to this 3-variable model did not lead to substantial changes in AUC (0.827, CI95% 0.74–0.91, p = 0.002). Using an optimal classification threshold derived from Youden's index, the model achieved a sensitivity of 82.4% (95% CI: 65.5–93.2) and a specificity of 78.6% (95% CI: 64.3–89.3), meaning that in 82.4% of the cases the algorithm correctly identified true dementia cases and in 78.6% of the cases correctly excludes non-cases. The positive predictive value (PPV) was 77.8% (95% CI: 60.8–89.9), while the negative predictive value (NPV) was 83.0% (95% CI: 68.1–92.5). The Matthews Correlation Coefficient (MCC) was 0.61 (95% CI: 0.43–0.77), indicating a strong overall correlation between predicted and actual classifications. The Brier Score, measuring the mean squared error of probabilistic predictions, was 0.128 (95% CI: 0.08–0.18), suggesting good calibration. The concordance statistic (C-statistic) was 0.75 (95% CI: 0.70–0.80), further supporting the model's discriminatory capacity.
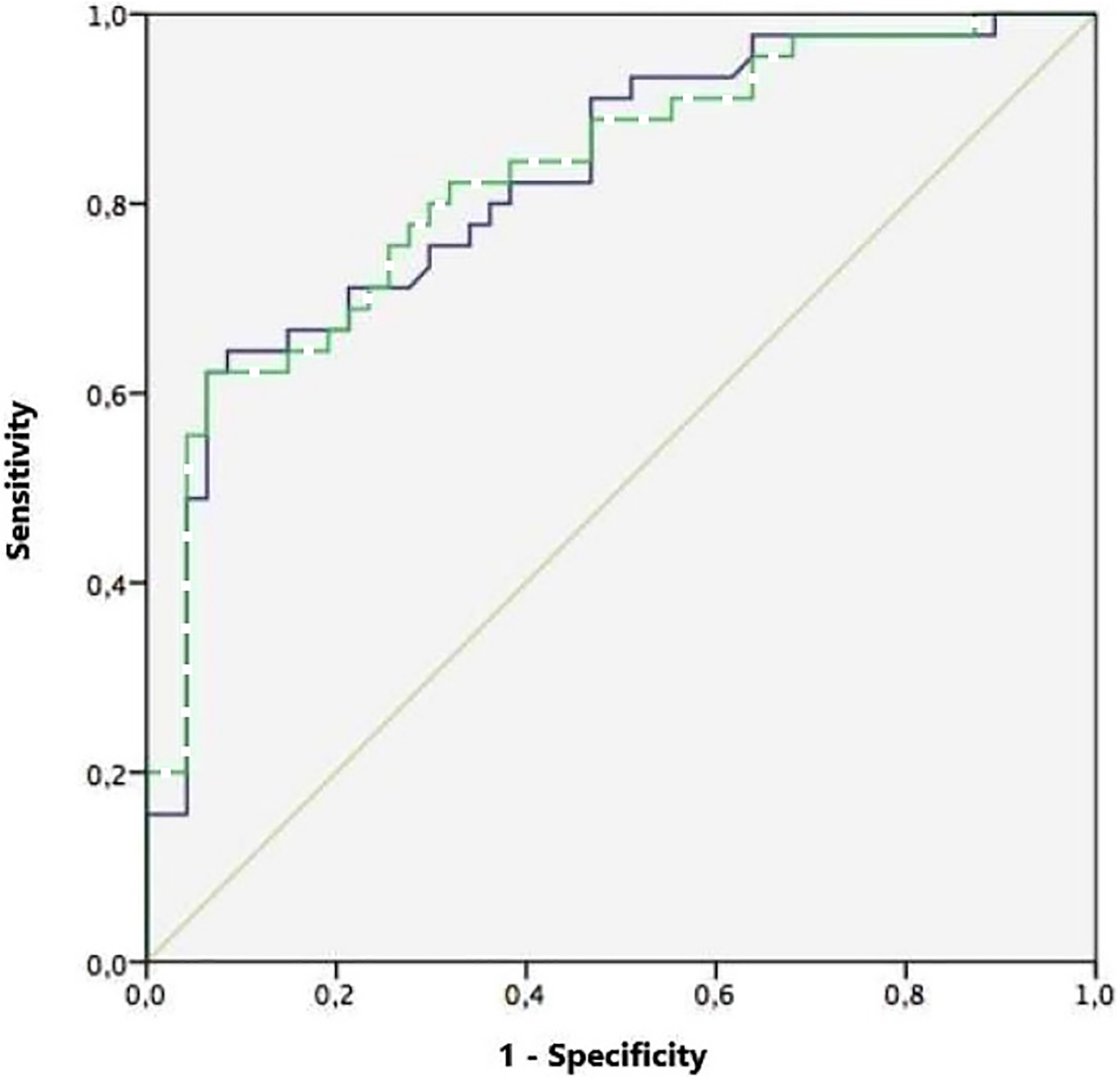
ROC curve model. In solid line the model with MMSE + Clock + hypercholesterolemia, in dotted line MMSE + Clock draw test + hypercholesterolemia + age + sex.
From the ROC curve analysis of MMSE and CDT analyzed individually, the Youden index was calculated in order to obtain an optimal cut-off value of predictivity of each test. For the MMSE, the optimal cut-off score for discriminating patients evolving to dementia was 24 (Se 0.57; Sp 0.92), and for the CDT 8 (Se 0.21; Sp 0.5). Dividing the participants among those with scores below the two cut-offs the Odds Ratios for the development of dementia was 6.3 (CI95% 2–19.43 p = 0.01) for MMSE and 4.1 (CI95% 1.4–11.9, p = 0.009) for CDT.
Finally, a nomogram (Figure 2) was then developed for the risk of evolution from MCI to dementia at 3 years with a score from 0 to 14, based on the weight of the 3 factors (MMSE, CDT, and presence of hypercholesterolemia) at multivariate analysis.
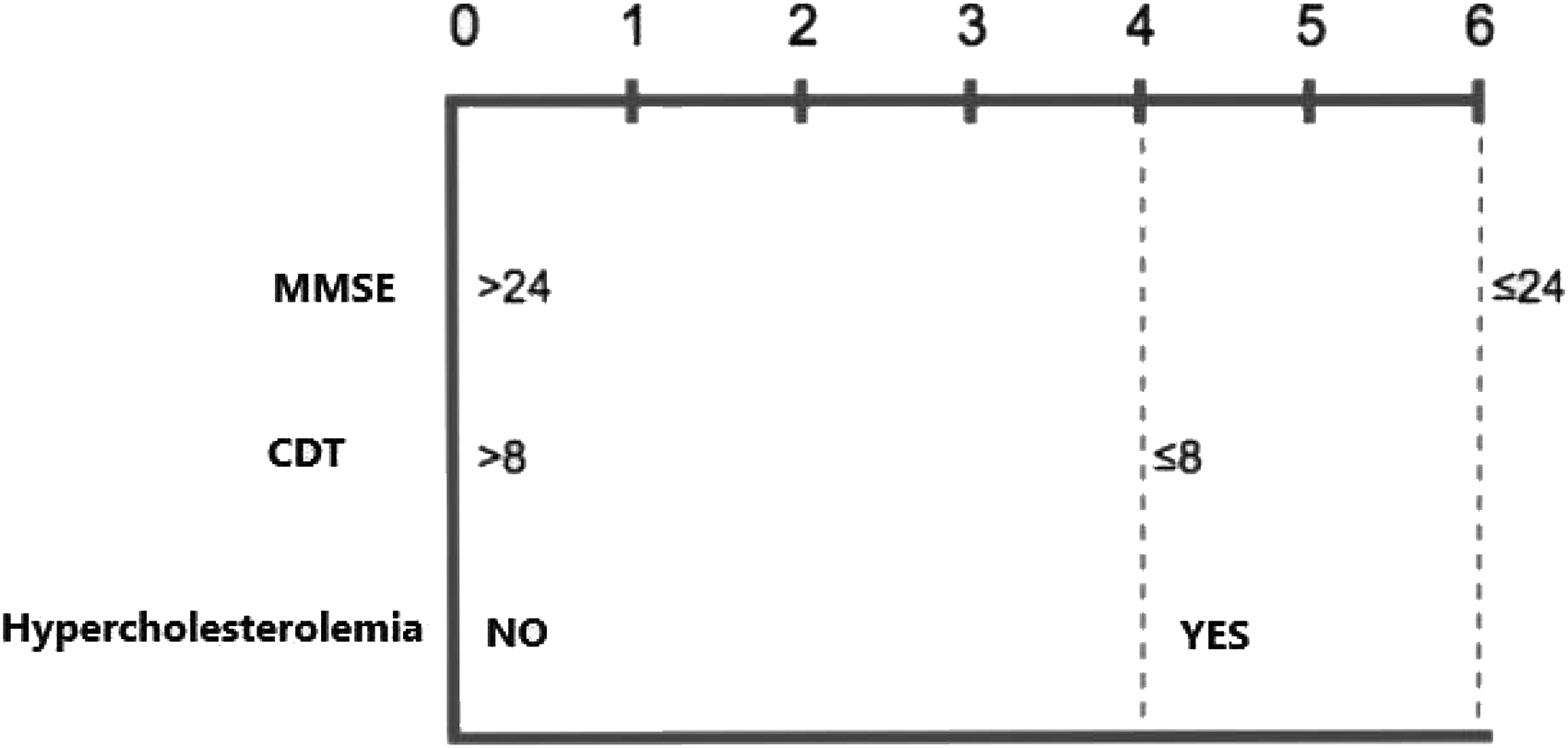
Nomogram.
Discussion
In this study, we found significant differences between MMSE score, CDT score and hypercholesterolemia prevalence in patients with MCI who developed dementia after a follow-up of 3 years, compared to patients who remained stable. Then, we evaluated a simple prognostic model to predict the onset of dementia using these three variables. We found that the combination of these cognitive tests and medical history of hypercholesterolemia was significantly better than both cognitive tests and medical history considered separately, thus providing an added diagnostic value in predicting AD in patients initially affected by MCI. The prognostic model has a significant accuracy also reflected by values of sensitivity and specificity around 80%, indicating that it should be used in daily clinical practice.
While simple and easy-to-administer cognitive tests are largely available in clinical practice, they often suffer from poor accuracy. Therefore, the development of simple tools that would allow to predict conversion from MCI to dementia would be of great clinical value to enable possible targeted interventions. Clinical prediction models are becoming increasingly important in clinical practice, with their use spreading rapidly. 50 But many of them they are usually very complex and often include machine-learning elaborations of variables such as clinical data, radiologic imaging, CSF markers, and genetic analysis, which make these models of limited applicability in routine clinical practice. 51
In a recent Cochrane review the authors did not find evidence supporting a substantial role of MMSE as a stand-alone single-administration test in the identification of MCI patients who could develop dementia. 20 Focusing only on studies including AD in the USA, in a paper published in 2009, it was found that MMSE scores are predictive of conversion from MCI to AD in a follow-up period of 3 years with an AUC = 0.78. 17 Similarly, in a Swedish study in older patients, the MMSE score and CDT score were predictive for conversion to AD during a follow-up of almost 5 years (AUC 0.79 CI95% 0.71–0.86 for MMSE and AUC 0.67, CI95% 0.58–0.75 for CDT). 16 Moreover, Mondrego et al. found low MMSE score to be predictive of dementia conversion at 3 years in a cohort of MCI patients (AUC 0.76, 95% CI = 0.6–0.88). In this latter paper, and also according to our findings, there were no differences for age and other memory performance tests. 18 The same authors in a later study confirmed MMSE to be predictive of conversion from MCI to AD (AUC 0.72; 95% CI, 0.62–0.8). 52 Pozueta in 2011 found a positive association between lower MMSE score at baseline evaluation (best cut off 26.5/30) and AD conversion over 2 years (AUC 0.77, 95% CI = 0.67–0.87); according to our results, even in this paper there were no relation with age and sex for AD conversion. 19 Therefore, MMSE seems to have a significant predictive value in detecting MCI patients at higher risk of conversion to AD. One reason MMSE can identify specifically MCI-AD, among MCI-other dementias and stable MCI, is that patients with subcortical dementias tend to produce relatively high scores on orientation and recall parts of the MMSE, while AD deteriorate early in orientation and memory. 53
Similarly to our findings, in the very recent Italian Interceptor project the authors found that MCI patients who converted to dementia, during a follow up of 3 years, were more frequently female and had lower MMSE score.
The CDT seems to differentiate the degrees of cognitive impairment earlier than is possible using other traditional screening tests. 21 Similar to the present findings, Nesset et al. found that higher CDT scores were significantly associated with a lower risk of conversion to dementia (HR 0.85; 95% CI 0.74–0.98). 54
Moreover, in a German study on 384 participants it was found that individuals with incident dementia had significantly lower CDT scores at baseline than those without dementia. CDT was a significant predictor of incident dementia, however with an AUC = 0.70. 14 Furthermore, in a recent study on almost 1600 participants a composite score, based on data collected with a digital version of the CDT, was significantly associated with multiple atrophic brain MRI measures. 55
As for MMSE, the CDT seems to identify patients at higher risk of evolution from MCI to dementia, although with a low predictive power.
Interestingly, regarding the FAB test, even if commonly used in clinical practice, studies on its predictive value in the evolution from MCI to dementia are substantially missing in literature.
Although age and female sex are well-known risk factors for dementia and MCI,56–58 we did not find a relationship with conversion from MCI to AD in our study. However, looking only at studies focusing on conversion from MCI to AD in the literature, these relationships are unclear, and the risk factors for progression were not found to be the same as in incident MCI. 23
The identification of hypercholesterolemia as an independent risk factor for progression from MCI to dementia aligns with previous evidence implicating dyslipidemia in neurodegeneration. Several mechanisms may underlie this association. First, elevated cholesterol levels contribute to cerebrovascular pathology, including atherosclerosis and microvascular damage, which may exacerbate vascular cognitive impairment.59,60 Second, cholesterol plays a crucial role in brain structure and function, and disruptions in brain cholesterol homeostasis have been linked to amyloid-beta deposition and tau pathology. 61 Third, hypercholesterolemia may promote a pro-inflammatory state that contributes to neuroinflammation and neuronal dysfunction. 62
While observational data and meta-analyses suggest that statin use is associated with a lower risk of dementia and MCI in cognitively normal individuals, 63 the evidence among those with established MCI remains inconclusive. The GEMS cohort found no protective effect in MCI subjects,64,65 and the ADNI study reported slower memory decline in early MCI but no reduction in diagnostic conversion.66,67 Large population-based studies in older adults also failed to detect a significant reduction in progression from MCI to dementia. 68
As previously stated, in literature there are many accurate clinical prediction models to predict conversion from MCI to dementia. Algorithms usually involve the combined machine-learning use of clinical, biohumoral, radiological, and genetic parameters, thus making their application unfeasible in usual clinical practice.26–29 Focusing only on studies including easily accessible and inexpensive clinical and demographic parameters, in a recent Italian study the authors found that age, MMSE score and type of MCI were significantly associated with the progression to dementia. In the resulting prediction model for the progression to dementia in the MCI patients grouped by the number (0–3) of significant predictors present in each individual (age > 78 years, MMSE < 25/30, presence of aMCI type), compared to patients with 0 predictors, a graded and significant increase in the risk of progression was found in individuals who had one factor (HR 12; 95% CI 1.7–91), two factors (HR 28; 95% CI 4.0–210) or three factors (HR 45; 95% CI 6.5–328). The ROC curve analysis for the score showed an area under the curve of 0.72 (95% CI 0.66–0.75, p < 0.001), 69 therefore lower than those reported by our group. Similarly, in a study focusing on the Framingham cardiovascular risk profile (age, gender, total cholesterol, HDL cholesterol, smoking habits and systolic blood pressure) as a predictor to conversion from aMCI to dementia, the rate of conversion increased from 12.5% in the low-risk group to 43.2% in the high-risk group, even if in this model the discriminatory power was quite low (AUC 0.592; 95%CI 0.534–0.649, p = 0.003). 70
Finally, in another paper on 105 patients with MCI, a combination of MMSE and California Verbal Learning Test Long Delayed Total Recall constituted the best predictive model for those at risk of develop dementia over 2 years, with the optimal MMSE cut off of <26/30, with high specificity (91.83%), but low sensitivity (43.58%). 19
The findings of our study must be interpreted within its limitations. First, this is a retrospective study, which inherently could lead to missing data, such as the absence of cerebral imaging with atrophy score for the entire sample, and selection and recall biases. Second, the population included is not large and included only white patients.
In our opinion, our algorithm based on simple clinical and laboratory measures, adds the possibility to better predict the onset of dementia in people affected by MCI.
In conclusion, combining clinical information, such as medical history of hypercholesterolemia with the CDT and MMSE tests, may result in a simple prediction model useful to predict the onset of dementia in a cohort of older patients over periods up to 3 years.
Future longitudinal studies are warranted to validate the predictive value of the proposed model in larger cohorts, ideally through multi-center collaborations. Longer follow-up durations (e.g., 5–10 years) are needed to assess the long-term risk of progression to dementia and determine the temporal stability of the model. In addition, future research should explore the integration of other clinical and biological variables, such as APOE genotype, cerebrospinal fluid biomarkers (e.g., Aβ42, tau), or neuroimaging data, to enhance risk stratification. These directions may contribute to more accurate, individualized prognoses and inform tailored intervention strategies.
Supplemental Material
sj-docx-1-alz-10.1177_13872877251379898 - Supplemental material for A simple nomogram to predict the onset of dementia in mild cognitive impairment: A retrospective study
Supplemental material, sj-docx-1-alz-10.1177_13872877251379898 for A simple nomogram to predict the onset of dementia in mild cognitive impairment: A retrospective study by Francesco Bolzetta, Giorgia Durante, Annalisa Tessari, Martina Lazzarin, Flavio Busonera, Anna Pontarin, Maria Villanova, Laura Vernuccio, Carlo Saccaro, Carlo Custodero, Piero Portincasa, Mario Barbagallo and Nicola Veronese in Journal of Alzheimer's Disease
Footnotes
Acknowledgements
The authors have no acknowledgments to report.
Ethical considerations
The protocol of this retrospective study was approved by our ethics committee (approval number 6107/U3/24).
Consent to participate
Where possible we obtained a written consent to participate. In accordance with our local ethics committee we proceeded to collect data without first obtaining a written consent if the outcome of the attempts made to contact subjects (in particular the verification of life status, the consultation of the data reported in the clinical documentation, the use of the telephone numbers held by the center, as well as the acquisition of contact data from the registry of patients or the resident population) resulted impossible.
Consent for publication
Not applicable.
Author contribution(s)
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was funded within the framework of the National Recovery and Resilience Plan (PNRR), Mission 6 “Health”, Component 2 “Innovation, research and digitization of the National Health Service”, Investment 2.1 “Strengthening and enhancement of biomedical research of the National Health Service”, financed by the European Union – NextGenerationEU, under project code PNRR-MCNT1-2023-12378321 CUP: I73C23000440006.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
Data are available upon reasonable request to the Corresponding Author.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
