Abstract
Background
Early detection of amnestic mild cognitive impairment (aMCI) is crucial for early intervention. The Loewenstein-Acevedo Scales for Semantic Interference and Learning (LASSI-L) identify risk for cognitive decline by measuring proactive semantic interference (PSI). PSI correlates with critical Alzheimer's disease (AD) biomarkers, predicting decline. A digital version of the LASSI™, the LASSI-D™, was highly convergent with the LASSI-L in older participants.
Objective
We developed three alternative forms of the LASSI-D™, examining convergence across forms and retest changes, to evaluate utility as a repeated-measure.
Methods
Alternative forms were developed in English and Spanish. 77 participants meeting criteria for aMCI and 92 participants with normal cognition were tested twice at a 2-week interval. Participants who were assessed first with the LASSI-D were retested with the LASSI-L and vice versa. Critical variables for the LASSI™ are indices of PSI, including reduced learning of the second word list and recall intrusions when attempting to recall the second list.
Results
There were no form or group differences on any PSI-related variable across alternative forms, all ps > 0.6, with the digital forms and LASSI-L equivalently sensitive to aMCI, (all ps < 0.01). Intrusion errors were essentially identical for LASSI forms across, (all p > 0.86), without significant practice effects. Performance did not differ across language for any variable across groups, forms, and assessments, all ps > 0.010.
Conclusions
Three digital alternative forms of the LASSI™ were highly convergent with each other and the LASSI-L. The findings provide evidence of the utility of a cloud-based, self-administered cognitive challenge test with alternative forms.
Keywords
Introduction
Efforts to develop early therapies for Alzheimer's disease (AD) and AD related disorders (ADRD) are occurring on many fronts. To develop more efficacious therapies, it is critical to facilitate earlier detection of cognitive changes associated with ADRD, prior to extensive progression of central nervous system deterioration. It is equally important to ensure that such measures are broadly accessible and easily disseminated to clinicians and care providers. Many traditional assessments aimed at mild to moderate AD are generally insensitive to mild cognitive impairment (MCI) 1 and even less effective in detecting the features of early MCI. 2 One strategy developed to address these challenges involves the use of “cognitive stress tests”, which are designed to be particularly challenging for individuals with early cognitive impairment. 3
One cognitive stress test that has been extensively validated is the Loewenstein-Acevedo Scales of Semantic Interference and Learning (LASSI-L). 4 This assessment strategy examines vulnerability to proactive semantic interference, 5 which is known to increase in older individuals. 6 In the LASSI, participants first learn a list of 15 words from three semantic categories (5 words each): fruits, musical instruments, and articles of clothing (i.e., list A). After list A is presented and recalled twice, a competing target list of words from the same three semantic categories is presented (i.e., list B). Proactive semantic interference (PSI) can then be measured, defined as interference occurring when previously learned information interferes with the ability to learn new, semantically similar, information. These effects are indexed by both reduced learning of the second list and intrusions of words from the first list into the recall of the second. A unique feature of the LASSI is a second presentation of list B that offers the opportunity to recover from PSI, designated as “Failures to Recover from Proactive Semantic Interference” (frPSI).7–8 frPSI is manifested by continued learning deficits after a second presentation of list B and continued intrusions from list A.
frPSI has been found to be related to total and regional amyloid load in older apparently neuropsychologically normal community-dwelling individuals; 9 to differentiate between aMCI patients with suspected AD from cognitively unimpaired older controls cross sectionally and longitudinally (CN);10–12 and has been associated with volumetric loss in AD-prone areas in amnestic MCI.13–14 In contrast, standard memory tests were not related to volumetric findings and progression to MCI. 2 Particularly relevant to specificity to AD/ADRD are findings that semantic intrusion errors on both first (i.e., PSI) and second (i.e., frPSI) presentation of list B are greater in persons with amnestic MCI (aMCI) who are amyloid positive on PET compared to individuals with aMCI caused by other neurological or neuropsychiatric conditions, who are amyloid negative.15–17 This provides substantial evidence of the potential to differentiate those with AD pathology from those without, even among those with similar levels of global cognitive performance. This construct is indexed by the Percentage of Intrusion Errors (PIE) for list B presentations 1 and 2, which has been consistently found as the best discriminator between people who are amyloid positive versus amyloid negative based on other AD biomarkers such as plasma p-tau 181 18 as well as plasma Aβ42/Aβ40 ratios using mass spectrometry.
The LASSI-D™ is a self-administered abbreviated, cloud-based digital version of the LASSI-L, which is administered at the direction of a healthcare professional or researcher. A previous computerized version of the LASSI, the LASSI-BC, 19 was developed to digitize the assessment. The LASSI-BC showed excellent convergence with the LASSI-L 20 but requires the presence of an examiner. The fully self-administered LASSI-D™ has demonstrated excellent convergence with the key outcomes of the LASSI-L in a mixed samples of aMCI and NC older adults. 21 Thus, it has potential for broader clinical applications as it eliminates the need for a trained psychometrist to be present to administer and score the assessment.
While the LASSI-D™ demonstrated convergence with the LASSI-L, the LASSI-L only exists in a single form. This same stimulus set was used for the development of the primary form of the LASSI-D™, making comparisons of performance on digital versus the legacy instrument more direct. However, serial assessments, whether for tracking cognitive decline or evaluating treatment effects, requires careful consideration of potential practice effects. Practice effects are a normal feature of serial assessments, but when not controlled, they have the potential to mask both subtle elements of cognitive decline and the effects of treatment. Practice effects are a concern even in the presence of memory challenges. 22 Individuals with MCI and those with more significant ADRD have been consistently reported to manifest practice effects at retesting, although reduced as compared NC. 23 Wang et al. reported that reduced or absent practice effects have been associated with faster disease progression in AD subjects based on performance at baseline.
A further consideration of any assessments for ADRD is the cross-cultural validity of the assessment. The LASSI has been extensively validated in English and Spanish speakers in south Florida, 24 as well as in Spanish speakers outside of Florida, including Spain. 25 Effects of Spanish-English bilingualism in the US have been studied. 26 Also, the performance of American Black participants was specifically examined 27 and studies in other countries and languages 28 have examined the utility of the LASSI, in terms of replicability of the PSI findings.
The use of equivalent alternate testing forms in serial assessments has been shown to reduce practice effects across cognitive and functional measures. 29 In this paper, we present the development of alternative forms for the LASSI-D™, designed to enable retesting participants with the LASSI™. The goals of the study were to evaluate the convergence of three different LASSI-D forms with each other and with the legacy LASSI-L, to examine similarity of the alternate forms at their first administration, and to provide insight into potential practice effects for each of the digital forms. Convergence is defined by equivalent performance on critical variables across forms (lack of significant form-related differences), equivalent sensitivity to aMCI (defined by equivalent between groups effect sizes across forms), equivalent re-test changes (similar effect sizes across forms at retest), and high cross temporal correlations across forms within individuals.
The original LASSI-L and three alternate versions of the LASSI-D™ (A, B, and C) were included in the retesting protocol, with form A being identical to the LASSI-L and reported on previously. All four forms were administered to all participants, selected for the presence of aMCI or NC, in a counterbalanced order with retesting at 2-week intervals. The LASSI-L and each of the three LASSI-D™ forms were each administered first to a subset of the participants. All participants tested first with a form of the LASSI-D were retested with the LASSI-L. Additional performance-based assessments were administered at each LASSI™ testing session to evaluate the similarity of the 4 participant subsamples, defined by which variant of the LASSI™ was administered first, as well as to compare retest changes in the LASSI™ forms to those seen in the sample with those seen with two brief typical neuropsychological assessments, Trail Making Test Part A and Animal Naming Fluency.
Methods
Overall study design
This report is based on the second Phase of a longitudinal observational study conducted at seven community centers/locations in South Florida and four in New York City. As reported before, participants received an in-person assessment to characterize their cognitive status and were tested with the legacy LASSI-L and the different forms of the LASSI-D. Participants who met inclusion criteria and adequately completed the initial assessment progressed to later parts of the study. The WCG Institutional Review Board approved the study (WCG# 2022-1540), and all participants provided signed informed consent. In this report, we present the data from the baseline and first reassessment of participants, which examined the similarity of the different forms (LASSI-L, the LASSI-D Form A [identical to LASSI-L], and LASSI-D forms B and C, at baseline and a two-week retest assessment.
Participants
The sample included English or Spanish-speaking adults over the age of 59 who lived in the community, had at least 20/60 vision, were able to read a computer screen, and had adequate hearing for an auditory-based assessment. Males and females were recruited, without restrictions on racial or ethnic status. MCI status was ascertained with a neuropsychological assessment using the Jak-Bondi criteria. 30
Exclusion criteria included a Montreal Cognitive Assessment (MoCA) 31 score of <19 or a reading score, in their commonly spoken language, at less than a 6th grade level. Other exclusions included the inability to undergo assessments in either English or Spanish, having been tested with the LASSI-L in the past 12 months, a diagnosis of a serious psychiatric condition apart from clinical depression, a previous medical history of brain disorder such as stroke, seizures, tumor, or significant traumatic brain injury with extended loss of consciousness.
Reading performance
The literacy level of English speakers was examined with the Wide Range Achievement Test (WRAT), 32 3rd edition. Spanish speakers were assessed with the Batería III Woodcock-Munoz Identificacion de letras y palabras subtest. 33 We used the 3rd edition of the WRAT (Published in 1993) because the participants in this study were an average age of 43 when those norms were developed and later norms might present a disadvantage to them because of intervening improvements in performance expected based on the “Flynn Effect.” 34 The Flynn effect refers to the continuous improvement in intelligence seen over time seen in Western cultures. Older participants would therefore have their cognitive performance underestimated when compared to later norms, reflecting population based normative changes that occurred after they completed their education.
Cognitive assessments
Cognitive assessments were used to collect data for the performance-based MCI criteria. The LASSI™ measures were not employed in diagnostic determination. All assessments were performed in each participant's commonly spoken language (English or Spanish).
Montreal cognitive assessment
This test examines cognitive performance with scores ranging from 0–30. Assessments were performed by certified bilingual raters, and we used the official Spanish translation for Spanish speakers.
Wechsler memory scale- revised, logical memory i and ii (Anna thompson story)
Participants are asked to read a story and then asked for immediate recall, followed by a 20-min delayed recall filled with other non-verbal assessments. 35 We chose this assessment, administered in a single form with delayed recall, because of its wide-ranging inclusion in standardized assessments and databases
Hopkins verbal learning test-revised (HVLT-R)
We administered the HVLT-R prior to assessment with the BAC, substituting this shorter 12-item, 3 trial verbal learning assessment for the much harder 15-item 5-trial BAC word list test. 36 We also examined delayed recall to contribute to the MCI classification. We obtained translated versions of the test from the publisher.
Brief assessment of cognition (BAC): app version
The BAC-app measures domains of cognition known to be related to everyday functioning. 37 The BAC-app delivers the same assessments with cloud-connected tablet delivery for ease of administration and standardization. The BAC-App has been widely used in studies of MCI. The cognitive domains assessed include: Digit Sequencing, Token Motor; Verbal Fluency; Symbol Coding; Tower of London (as well as a verbal learning test not used in this study). The assessment has 6 different forms and has validated translations into multiple languages. We used form 1 and the official Spanish translation for the baseline assessments.
“Warm-up” pretesting prior to each LASSI™ assessment
Two short tests, Trail-Making Test part A and Animal Naming Fluency were administered immediately prior to the form of the LASSI™ form administered in that session. This procedure was used to ensure that the participants were ready to be assessed with the LASSI™. These data were also used to examine practice effects arising from retesting with identical standard NP tests and to generate comparative information about the similarities in cognitive performance of the subsamples first assessed with different versions of the LASSI™.
The LASSI™
Participants were administered two variants of the LASSI™: the legacy tester-delivered in- person paper version: the LASSI-L and three different forms of the LASSI-D™. The LASSI™ employs controlled learning and cued recall to maximize encoding of a list of 15 target words representing three semantic categories: musical instruments, fruits, or articles of clothing (five words per semantic category). Participants are shown each of the words, one at a time, and asked to read the word aloud, thus allowing for monitoring of processing the stimulus. Words that are read incorrectly are immediately cued in the following way: “The word is XYZ. Please say XYZ”. Advancement to the next word does not occur until the current stimulus is correctly pronounced.
A graphical depiction of the overall assessment sequence with the LASSI™ is presented in Figure 1. After immediate free recall of the first word list, the examinee is presented with each category as a recall cue (e.g., articles of clothing) and asked to recall the words from that category, which are summed across the three semantic categories into a cued recall score. A second learning trial, identical to the first, and subsequent cued recall immediately follows, providing a second opportunity for maximum encoding of the learning information. As the LASSI™ targets PSI in the absence of major memory challenges, previous studies excluded participants unable to learn at least 11 of the 15 list A stimulus words over 2 learning trials.18–20 In this study, any such participants were also excluded from the PSI analyses.
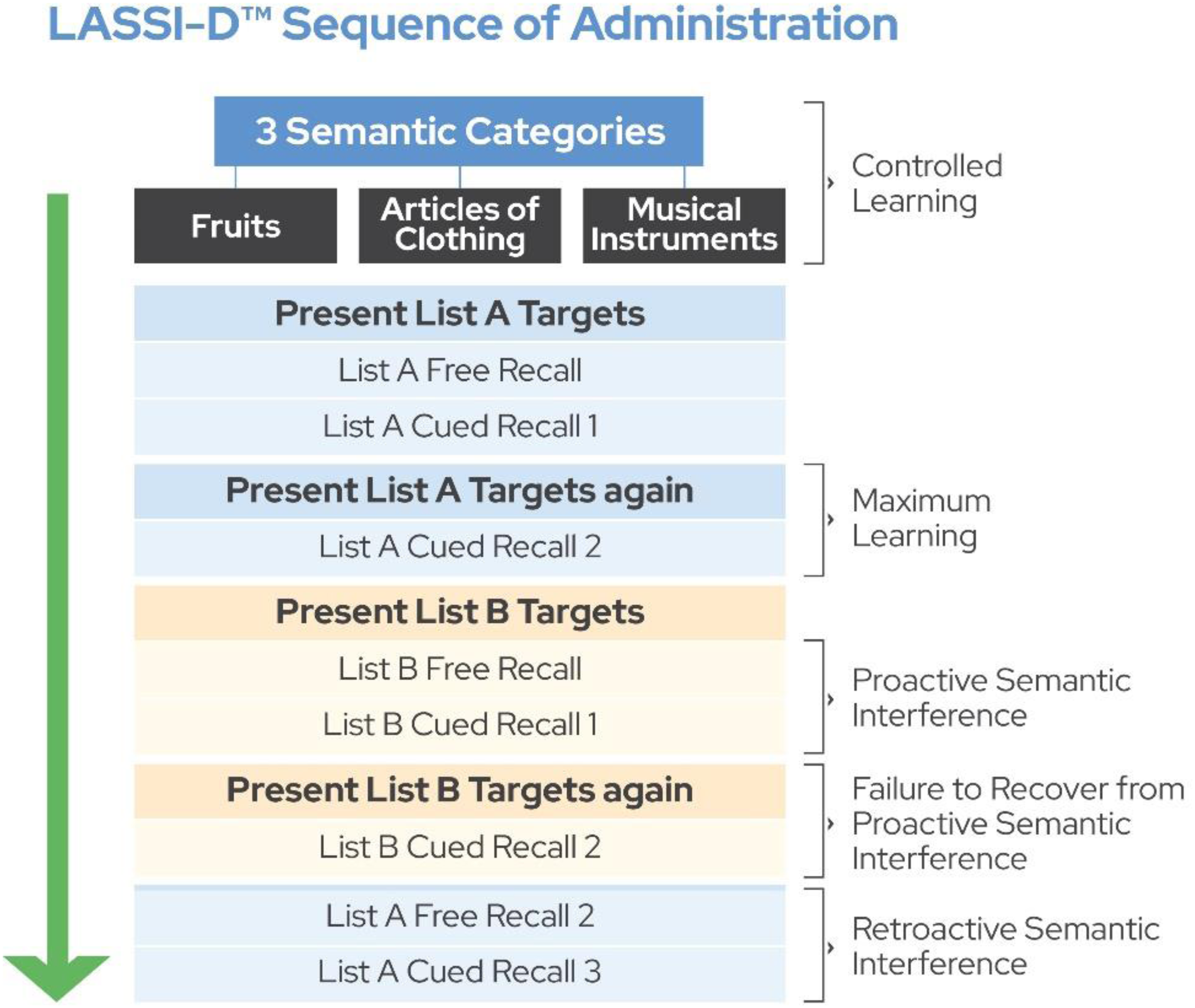
Full assessment sequence of the LASSI, including administration of List A and List B.
Following the second presentation of list A, the participant is presented with a new semantically related stimulus set (list B) which consists of 15 new words from the same three semantic categories (five from each) and is again asked to read them aloud. The participant then performs free recall of the list B words (B1 Free recall). Then, the participant is semantically cued to recall the list B words. The difference in performance on cured recall compared to proximal cued recall performance for the first list defines immediate proactive semantic interference (PSI). Next, the participant reads the list B words again, followed by a second cued recall trial (Cued Recall B2). Differences in cued recall performance on the second trial compared to the first list B cued recall quantifies the extent to which the examinee recovers from PSI, with smaller incremental benefits from the second presentation defining frPSI.
The most critical set of outcomes are PSI intrusion scores, including either targets from the original list or (far less frequently), words belonging to one of the three semantic categories but not belonging to list A or B target words. During both cued recall trials for list B, any words that are not from that list are recorded. The scores for correct recall and intrusions are calculated into an index, dividing the number of intrusion errors by the total words recalled (correct and intrusions), yielding “percentage of intrusion errors: “PIE”. PIE has been shown to be the most sensitive predictor of risk for subsequent cognitive decline.16,38
Digital migration of the LASSI™
As described above, the digital LASSI-D™ is cloud-based and self-administered without the requirement for a human tester. The LASSI-L has been translated into multiple languages, including Spanish. We migrated the English and Spanish versions in this study. The LASSI-D™ uses speech recognition technology (both Google and Microsoft) to capture and correct participants’ reading of the stimulus words on the learning trials for list A and B, as well as capturing and scoring all vocalized recall scores. The LASSI-D™ also employs a realistic AI generated human avatar. The avatar vocalizes the same instructions and feedback during assessment that are read by a human tester administering the LASSI-L. A digitally administered pre-testing orientation and preparation session was also included at each LASSI-D™ assessment to ensure comprehension on the part of the participant. This orientation tests the functionality of the internet connection, the microphone, and speakers and asks the participant to name some simple shapes that appear on the screen, continuing until correct responses are detected by the software.
Creation of alternate forms
LASSI-D™ form A is identical in all features to the paper and pencil LASSI-L. Form A is designed to be the primary digital form and thus we devoted extra effort to comparing digital Form A to the LASSI-L. In order to create forms B and C, the same 30 target words from LASSI-L and LASSI-D™ form A were reorganized into forms B and C. We attempted to keep 50% of the words in the same presentation list (first versus second) as previously and moved the other words across lists in making forms B and C.
We chose to re-use the same 30 words (10 per semantic category) because of concerns that if we retained the same three semantic categories and tried to develop novel category exemplars (requiring 20 new words per semantic category), the word frequencies would be markedly mismatched between the original stimuli (LASSI-L and LASSI-D™ form A) and the newly created forms B and C. In terms of creation of novel categories, we could not determine in advance if other categories would manifest the same learning and interference parameters. An important factor in our decision to reuse the 30 words of LASSI-L, was that the original creators of the LASSI-L tried to use several novel categories to create alternative forms but that they were unsuccessful in developing forms with similar PSI characteristics. To re-iterate, our primary goal was developing a close to identical digital migration for LASSI-L and LASSI-D form A and we were willing to regroup if we could not succeed in development of alternative forms with the same 30 stimulus words.
Procedure
Participants were screened for cognitive functioning as described above. They were designated as having normal cognition (NC) or one of three different MCI subtypes: amnestic MCI (impairments on two or more memory tests but no other cognitive domains; aMCI); Multi-domain MCI: MDMCI (two or more impairments on both memory and other cognitive domains); Non-Amnestic MCI: NAMCI (impairments on two non-memory cognitive domains, but no more than one memory domain). Normative standards for the screening tests were used to evaluate performance, with impairment defined as 1.0 SD below normative values on each test. The performance data for participants with NAMCI were collected, but their data was not examined because the LASSI™ is focused on aMCI. We did not inform the research participants whose data were being excluded, because they may have known other participants and could potentially discussed the research procedures with them. A fully bilingual testing assistant was present during all assessment sessions and administered all the paper and pencil tests as well as the LASSI-L.
Testing sequence
The testing order of the LASSI™ variants was sequential and separated by two weeks. The LASSI-L was administered first until we had at least 50 confirmed eligible participants (MoCA > 19, list A2 recognition > 10, Does not meet criteria for NAMCI). For those participants, we administered LASSI-D™ forms B or C at the two-week retest, in an alternating order. In the next sequence, we administered the LASSI-D™, Form A until we were certain we had 50 eligible participants, with all these participants retested at two weeks with the LASSI-L. Because of delays in confirmation of MCI status, more than 50 cases were included in the final assessment sequences. We did not attempt to balance the samples for potential factors that could influence performance (language, aMCI vresus NC, sex).
After completing the first assessment with either the LASSI-L and LASSI-D™ Form A as described above, we switched to administration of LASSI- D™ Forms B or C first while continuing to collect more participants. We alternated LASSI- D™ forms B or C, with the LASSI-L always administered as the reassessment. There were 53, 56, 30, and 30 participants tested first with the LASSI-L, LASSI-D™ form A, and LASSI-D™ forms B and C, respectively. Thus, 116 total participants were tested first with digital forms A, B, and C and all these participants were retested with the LASSI-L. Figure 2 presents a graphical depiction of the flow of the assignment sequence.

Sequential assignment of participants to alterative testing forms.
Data analyses
The overall goal of the analyses was to examine the similarity of the three newly created LASSI-D™ forms as well as confirming that forms B and C also performed similarly to the LASSI-L, as did LASSI-D form A. Since LASSI-D™, form A is identical in stimuli to the LASSI-L, our previously published study of the convergence of performance on these two forms was a direct test of the digital migration. Analyses were performed for participants who achieved a qualifying cued A2 score (Cued Recall 2 list A > 10), focused on 6 list B variables relevant to PSI: Cued Recall of list B trial 1 (B1), cued recall 2 of list B (B2), cued recall intrusions on list B trial 1 (B1) and 2 (B2), and percentage of intrusion errors (PIE) for list B trials 1 and 2 (PIE1 and PIE2). Analyses compared the 4 forms at Baseline. For these analyses, we used one-way analysis of variance with follow-up Tukey HSD tests. For all analyses, we did not correct for multiple comparisons, because we did not want to obscure critical differences in performance across forms with corrections. We used the same strategy to compare performance on the LASSI-L to LASSI-D™ forms B and C at retest, noting that form A was not administered at retest.
To examine retest differences, we used paired t-tests comparing the scores at baseline and retest for all six list B variables and calculated the Pearson correlations between performance at baseline and retest. We present the retest effects where the LASSI-L was administered first versus second separately because these are not same form test-retest effects.
We used t-tests to compare cross sectional performance of NC and aMCI participants on all 6 critical list B PSI variables in each of the 4 forms (LASSI-L vs. LASSI-D™ forms A, B, and C) at baseline and retest. We also used a repeated-measures Analysis of variance with aMCI status as the between-groups variable to examine group differences in retest effects. We compared English and Spanish speakers on all 6 PSI variables at each assessment for each of the 4 tests. To confirm that the samples were comparable in their overall cognitive performance, subsamples were compared for their baseline Trail Making Part A and Animal Naming performance across the 4 different subgroups. Trail Making Part A and Animal Naming were also examined for retest effects.
Results
Participant characteristics
There were 24 cases with non-amnestic MCI whose data were not examined. There were also 42 participants who did not meet the list A learning criteria.
Table 1 presents information on the eligible participants, characterized by which version of the LASSI™ they received first. There were no significant differences in age, education, and MoCA scores across the participant subsamples. There were slightly more Spanish speakers in the LASSI-D™ cohort, but there were no differences in any of the 6 critical LASSI™ PSI variables at baseline or retest, age, or MoCA scores across the Spanish and non-Spanish speakers, all ts < 1.64, all ps > 0.10.
Demographic information on participants based on their first testing form.
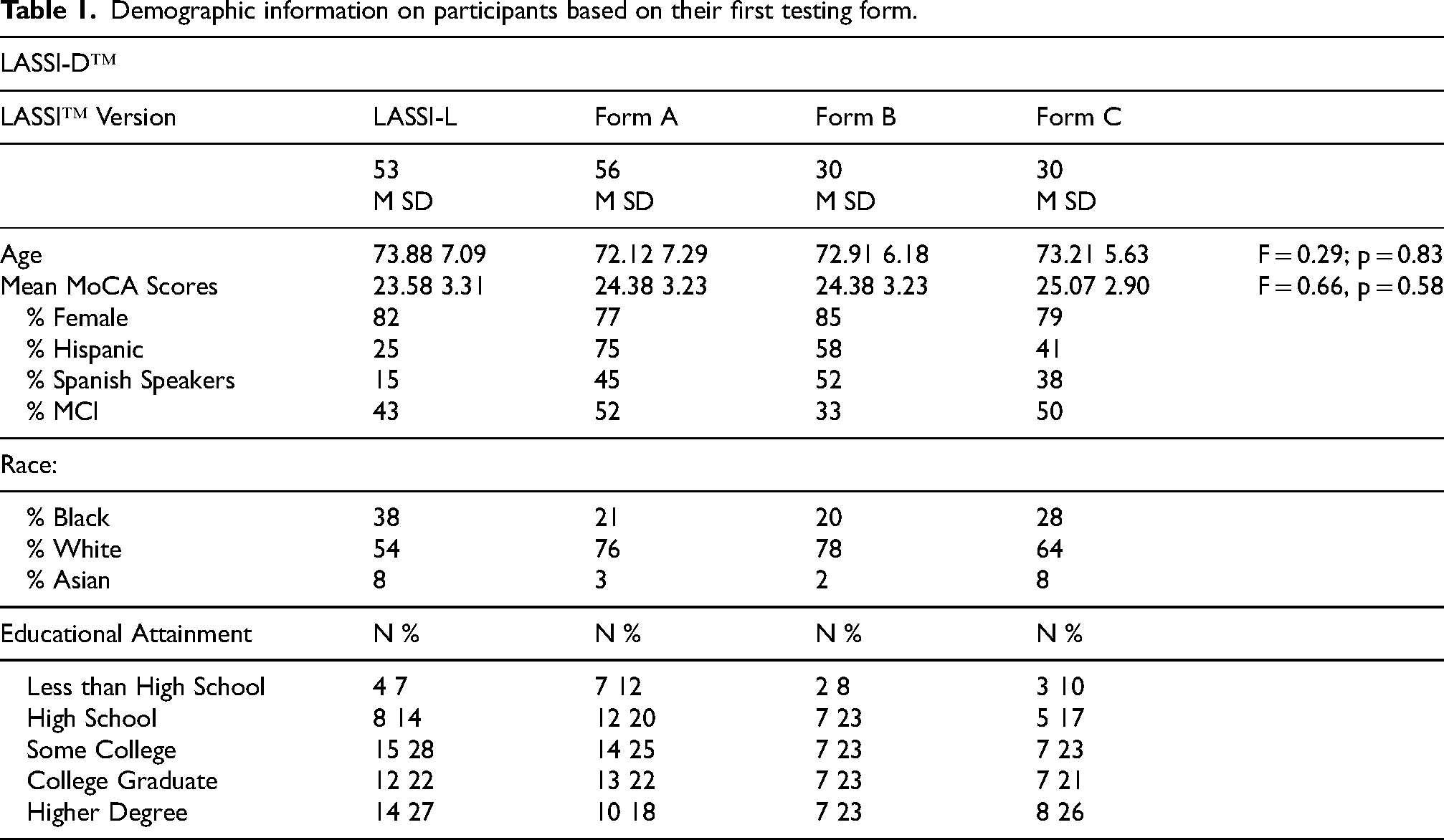
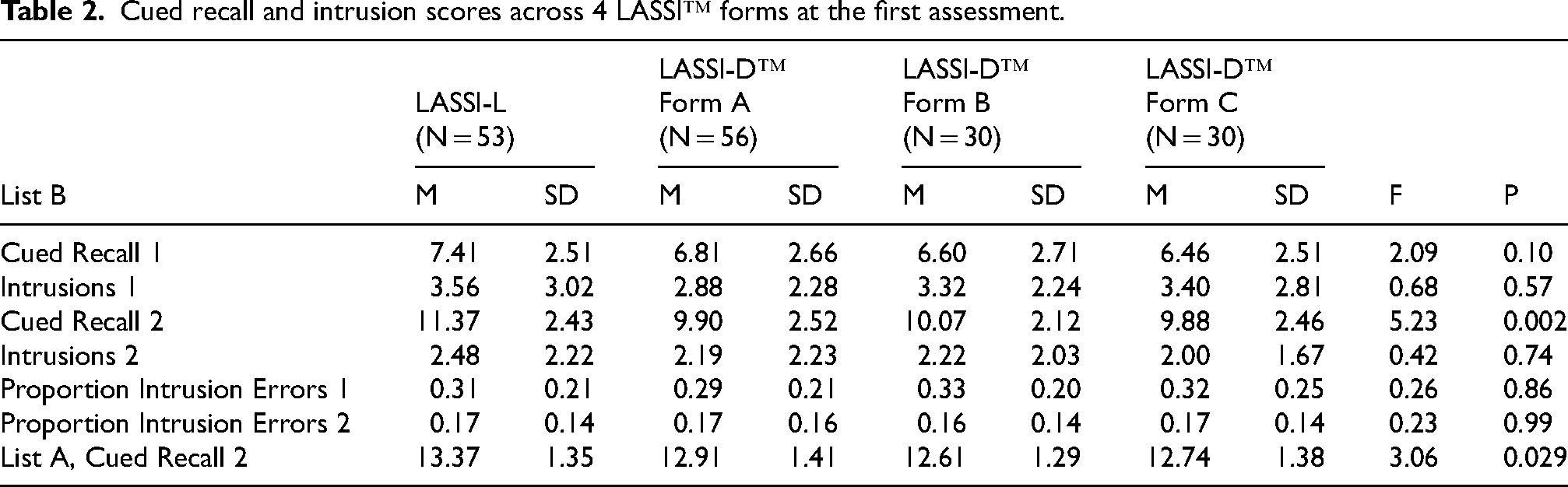
Table 2 presents the results of the comparison of baseline performance across the 4 forms of the LASSI™. As can be seen in the table, despite the restricted range of possible scores (11–15), list A cued recall 2 differed significantly across the forms, with LASSI-L performance being significantly better than digital form A, and no other differences being significant. There was only one of the PSI variables, list B cued recall 2, that differed between the 4 forms overall, with best performance on the LASSI-L. For that variable, Tukey follow-up tests indicated that the differences between the three digital forms of the LASSI™, A, B and C, achieved a p-value of p = 0.90, indicating no performance difference. In fact, the list B Cued recall 2 for LASSI-L was not significantly different from digital forms A and B, p = 0.06, although performance on Form C was significantly less than the LASSI-L on Cued Recall B2. When list B Cued Recall 2 was compared across the forms with list A Cued Recall 2 as a covariate, the effect of form on list B Cued Recall 2 became nonsignificant, F = 2.66, p = 0.11, while the covariate effect of list A Cued Recall 2 was significant, F = 33.89, p < 0.001.
Cued recall and intrusion scores across 4 LASSI™ forms at the first assessment.
When cross sectional performance differences at baseline between aMCI and NC participants were compared, for the LASSI-L and LASSI-D™ form A, all 12 tests were significant, all t > 2.45, all p < 0.001, and all d > 0.62. LASSI-D™ forms B and C had smaller sample sizes, with 10 and 15 MCI participants. For form B, PIE 2 did not differ significantly, t = 1.94, p = 0.064, but the effect size was still d = 0.65. For form C, cued recall 2 intrusions were not significant, t = 1.67, p = 0.10, but the effect size was 0.47. With all three LASSI-D™ forms aggregated, all comparisons of aMCI and NC participants were significant, all t > 2.66, all p < 0.009, and all d > 0.50. LASSI™ list A cued recall 2 was also compared across the groups. For all three digital forms, despite the restricted range of possible scores of 11–15, MCI participants performed more poorly than NC, all t > 2.70, all p < 0.001. For the LASSI-L, the two groups did not differ, t = 1.32, p = 0.19.
Table 3 presents the results of the LASSI™ forms at the retest assessment. There were no statistically significant differences between the LASSI forms on any variable for the sample as a whole. We compared aMCI and NC participants on their scores at reassessment. We performed t-tests comparing aMCI and NC participants on the 6 critical PSI variables for the LASSI-L and LASSI-D forms B and C. For the LASSI-L, all 6 tests were significant, all t > 2.30, all p < 0.024, and all d > 0.44. For forms B and C, all differences were also significant, all t > 2.10, all p < 0.045, all d > 0.82
Cued recall and intrusion scores across 3 LASSI™ forms at the reassessment.
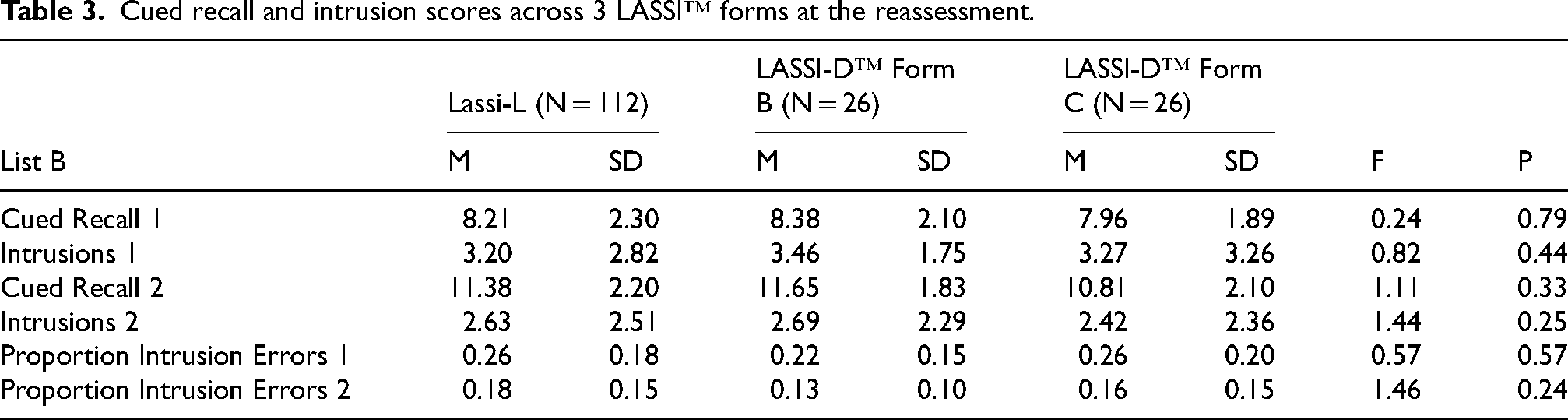
Table 4 presents the results of comparisons of baseline and follow-up assessments across the different forms. For the two cued recall variables, LASSI-L scores at retest were higher than the previously administered LASSI-D™ scores at baseline. However, for the critical intrusion variables, PIE 1 and PIE 2, the differences between baseline and retest were not significant for either testing sequence and all effect sizes for the differences between baseline and retest were all less than d = 0.20. Pearson correlations between baseline and retest performance (presented in Table 4) were all 0.50 or higher, reflecting 25% shared variance between the two assessments for all LASSI™ variables. Also, effect sizes for differences from baseline to retest for the digital forms were all less than 0.20, other than for cued recall 1, which was still a small effect size. All of the important intrusion variables had retest effect sizes less than 0.18, either with retesting with the LASSI-L or digital forms B and C.
Retest effects at the first reassessment across 4 LASSI™ forms: presented as effect sizes for changes from the first assessment.
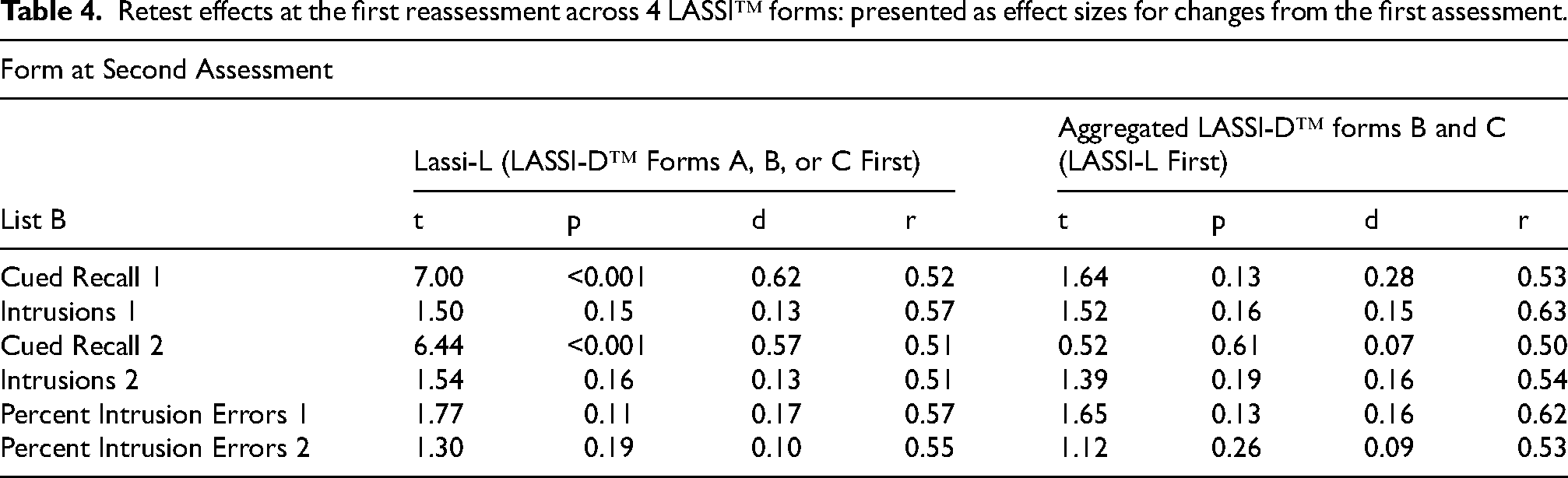
MCI group differences in practice effects were examined with a repeated-measures analysis of variance, with baseline and retest as the repeated factor and aMCI status as the between subjects variable. Analyses were done separately for each of the 4 assessment sequences and for all 6 PSI variables. Across 24 ANOVAs, there was only one interaction of assessment and aMCI status that was close to statistically significant, list B presentation 1 cued recall intrusions for LASSI-D™ form A first and LASSI-L second, F(1,55) = 3.89, p = 0.054. Thus, there is essentially no evidence for aMCI group differences across forms in practice effects on the list B learning and intrusion variables.
Finally, when baseline performance on Trail-Making Test Part A and animal naming, as well as MoCA scores were compared across the different subsamples, there were no differences, all F < 1.04, all p > 0.36, all eta2 < 0.04. Retest performance on the Trail Making test manifested a statistically significant, but modest practice effect, t(168) = 2.12, p = 0.02, d = 0.26. Animal naming performance was essentially identical across the two assessments: Baseline = 20.5; Retest = 20.7.
Discussion
The present study evaluated convergence between the LASSI-L™ and the performance of three alternative, digitally migrated forms of the LASSI-D™ in terms of the measures’ similarity on variables found to be sensitive to PSI and that have correlated with biomarker positivity in aMCI samples. While we previously found that the LASSI-D™ form A was essentially equivalent to the LASSI-L after digital migration, our goal here was to determine if alternative newly created digital forms manifested substantial similarity in performance to LASSI-D Form A and the LASSI-L. The results indicate similar overall performance between the LASSI-D™ forms at baseline and upon retest, with the three digital forms performing essentially identically. The aMCI and NC comparison showed that the LASSI-D™ alternative forms were especially sensitive, with substantial effect sizes, to aMCI. Better retest recall scores were seen with the LASSI-L, but these differences were accounted for by greater list A Learning. There were no differences among the LASSI-D™ alternative forms on any variables or any differences in PIE. Further, there were no statistically significant aMCI group interactions with practice effects on any variable on any form.
The notable similarity in the most critical LASSI™ variable, PIE, between the legacy form of the LASSI™ and its alternative digitally migrated forms reinforces the digital measure's potential for large-scale administration. As the PIE from the second list B recall trial is the critical variable that optimally classifies each group, aMCI and NC, suggests that LASSI-D™ will allow for increased accessibility to validated measures in early aMCI by virtue of widely scalable administration potential.
The modest practice effects seen in a subset of the learning variables are not unexpected. The LASSI-D™ form A and the LASSI-L are identical, so retesting with the LASSI-L after LASSI-D™ Form A administration would likely manifest some learning effects. Quite interestingly, LASSI-L administered after the identical LASSI-D™ form A did not lead to any differences in performance compared to administering forms B or C after the LASSI-L. The words used to create the alternative digital forms are also all contained in the LASSI-L, so some improvements with repeated exposure could be expected. The LASSI-L was also associated with higher scores on list A learning which explained the differences in list B cured recall. However, intrusion errors or their ratio compared to total responses were not influenced by repeated testing and all the evaluated combinations of forms found no differences in intrusions from the first to second assessment. Recent reviews of the literature have suggested that practice effects can serve as a classifier in clinical trials, so their detection remains critical. While previous analyses suggest that the addition of alternative forms and specialized tests can help reduce practice effects, 24 the more testing points there are, the more alternative forms must be available to account for a potential increase in practice effects. 22
Additionally, this study was conducted at non-clinical community centers, characterizing it as an ecologically valid test of the similarities of the different forms of the LASSI-D™. Our administration protocol for the LASSI-D™ is designed to be a suitable assessment strategy for these types of centers, where there are no professional assessors available.
There are limitations in our research design. Our primary goal in the study was to develop an identical digital form using the exact stimuli used in the LASSI-L. Thus, we prioritized collecting more participants tested first with the LASSI-D, form A, resulting in smaller samples for initial assessments with forms C and D. That sample mismatch seems to be obviated by the finding that baseline performance across digital forms A, B, and C was close to identical and that retesting with the LASSI-L led to identical scores across all three samples: the larger Form A sample and the smaller form B and C samples. LASSI-D™ form A was not administered at retest, but since the LASSI-L is identical to the LASSI-D form A and was administered to many participants at retest, there is little reason to believe that Form A would perform differently. The recommendation for use of the LASSI-D™ is to always administer Form A first to capitalize on the similarity to the legacy LASSI-L. There were higher list B cued recall trial 2 scores for the LASSI-L, which was significant when compared to one of the digital forms. This potential difference across the forms was associated with better learning of list A as well. However, at re-testing there were no differences in performance on any variables across all testing sequences, within the limitations that form A was administered second in this study. A final limitation is that the LASSI-D could not be subjected to rigorous cross-cultural validation in this development study.
Intrusion errors on the LASSI-L have been shown to correlate closely with amyloid PET, tau PET, structural MRI, CSF biomarkers, and blood-based biomarkers. The application of a widely accessible LASSI-D™ will expand the reach of screening of participants for pre-aMCI related to ADRD. The alternative forms now expand the LASSI™ and introduce the possibility of its use as a possible treatment outcomes measure for treatments targeting biomarker deposition. It could also be used as an assessment strategy in longitudinal studies related to AD/ADRD, with its use supported by the findings that critical intrusion errors are not reversed by practice. Our findings show excellent levels of convergence between the LASSI-L and the alternative LASSI-D™ forms and excellent discriminant sensitivity for aMCI across forms. This sensitivity enables further research into the mechanistic features that underlie semantic intrusions and can broaden biomarker relevant wide-ranging cognitive assessments.
Footnotes
Ethical considerations
IRB approval was obtained before any research procedures were performed. WCG IRB Approval number was: WCG# 2022-1540. No individuals who were under guardianship participated.
Consent to participate
All participants signed an informed consent form and any individuals who could not read the consent form or who not competent to consent were not eligible.
Author contribution(s)
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was funded by National Institute of Aging Grant number: 5R44AG074818, to Peter Kallestrup.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Drs. Harvey and Czaja are co-founders and Co-CSIs of i-Function.
Mr Kallestrup is co-founder and CEO of i-Function.
Dr Rivera-Molina and Ms. Mueller were full time employees of i-Function.
Drs. Curiel-Cid, Loewenstein, and Crocco were compensated as consultants during this study.
Dr Loewenstein is an Editorial Board Member of this journal but was not involved in the peer-review process of this article nor had access to any information regarding its peer-review.
Ms. Huang-Ouyang reports no biomedical conflicts of interest.
Dr Harvey receives royalties for the Brief-Assessment of Cognition, i-Pad version (BAC-App) from Clario Digital outcomes, Formerly WCG, formerly Verasci, Inc.
Dr Harvey has received consulting fees or travel reimbursements from Alkermes, BMS (Karuna Therapeutics), Boehringer Ingelheim, Kynexis, Minerva Neurosciences, and Neurocrine Biosciences.
Data availability statement
The data supporting the findings of this study are available on request from the corresponding author. The data are not publicly available due to the fact that this is a commercialization grant.
