Abstract
Background
The association between lifestyle factors and Alzheimer's disease (AD) pathophysiology remains incompletely understood.
Objective
The aim of this study was to assess the association of alcohol consumption, smoking behavior, sleep quality and physical, cognitive, and social activity with cerebral amyloid pathology.
Methods
For this cross-sectional study, we selected participants from the Amyloid Biomarker Study data pooling initiative. We used generalized estimating equations to assess associations of dichotomized lifestyle measures with amyloid pathology.
Results
We included 9171 participants with normal cognition (NC) and 2555 participants with mild cognitive impairment (MCI) from the Amyloid Biomarker Study. Of participants with NC, 58% were women, 34% were APOE ε4 carrier, and 27% had amyloid pathology. Of participants with MCI, 48% were women, 47% were APOE ε4 carrier, and 57% had amyloid pathology. In NC, cognitively active participants were less likely to have amyloid pathology (OR = 0.77, 95%CI 0.66–0.89, p < 0.001). In MCI, participants who had ever smoked or had sleep problems were less likely to have amyloid pathology (OR = 0.85, 95%CI 0.73–0.99, p = 0.029; OR = 0.62, 95%CI 0.45–0.86, p = 0.004).
Conclusions
In NC, cognitive activity was associated with a lower frequency of amyloid pathology. In MCI, favorable lifestyle behaviors were not associated with a lower frequency of amyloid pathology. The results of the current study contribute to the broader evidence base on lifestyle and AD by further characterizing the role of lifestyle behaviors in AD pathology across different clinical stages.
Keywords
Introduction
Evidence on the role of lifestyle behaviors in the development of cognitive impairment and dementia has been steadily accumulating, with evidence from meta-analyses and systematic reviews showing clear associations between lifestyle factors and dementia risk. 1 However, the relation between lifestyle factors and Alzheimer's disease (AD) pathophysiology is not yet well defined. A better understanding of the association between lifestyle factors and AD pathology (i.e., amyloid plaques and tau tangles) in early disease stages may help to identify those at risk and contribute to the development of targeted prevention strategies.
Findings from previous studies on the association between lifestyle factors and AD biomarkers of amyloid in cerebrospinal fluid (CSF) or on positron emission tomography (PET) are mainly based on cross-sectional analyses and are heterogeneous.2–16 While some studies report clear associations between lifestyle behaviors such as physical and cognitive activity and amyloid burden,2–4,6,7,9,10,12–16 others find no associations between lifestyle behaviors and amyloid pathology,5,8,11 or identify associations specific to APOE ε4 carriers or non-carriers. 7
Differences in composition and size of study cohorts, associated differences in distributions of demographic factors, timing (midlife versus late life), and method of lifestyle assessment as well as lack of power contribute to heterogeneity and likely account for a proportion of the divergence in results. Assessment in a large, harmonized study sample could therefore provide more insight into the role of lifestyle factors in the development of AD.
The aim of the current cross-sectional study was to examine the associations of alcohol consumption, smoking behavior, sleep quality, physical activity, cognitive activity, and social activity with amyloid pathology in a large group of persons without dementia by harmonizing lifestyle and amyloid measures across 42 cohorts and 11,726 participants included in the Amyloid Biomarker Study (ABS).
Methods
Standard protocol approvals, registrations, and patient consents
Written informed consent was obtained from all participants and data were de-identified. All individual sites contributing to the ABS have obtained local ethical approval and the ABS was approved by the Medical Ethics Committee of the Maastricht University Medical Center which declared that the Medical Research Involving Human Subjects Act does not apply to the study and waived the informed consent requirement because de-identified data were used.
Participants
Participants for this cross-sectional study were recruited from the ABS, a data pooling initiative that was started in 2013. Study design and data collection have been described previously.17–19 The ABS currently includes over 24,000 participants from 95 cohorts. For the present study, we selected 11,726 participants from 42 centers based on availability of amyloid biomarkers, age, cognitive status, and information on any lifestyle factor (n = 3399 excluded due to missing information on lifestyle; cohort characteristics are provided in Supplemental Table 1). 9171 participants had normal cognition (NC) as defined by normal scores on cognitive tests and absence of cognitive complaints (cohort-specific tests and measurements), and 2555 participants had mild cognitive impairment (MCI) as diagnosed by criteria set out in Petersen et al. or Winblad et al.20,21 Participants who reported subjective cognitive complaints were considered to have NC for the purpose of these analyses.
Amyloid pathology
Presence or absence of amyloid pathology was determined based on Aβ42 levels or Aβ42/40 ratio in CSF (n = 20 centers) or an amyloid-PET scan (n = 13 centers); 9 centers had both CSF and PET data. Amyloid pathology was defined using methods described previously. 17 Briefly, we used either center-specific or data-driven cut-offs obtained from Gaussian Mixture Modelling (GMM) to dichotomize CSF data. Center-specific cutoffs were used to dichotomize all PET data (n = 6298 participants) and CSF data for 33% of participants (n = 1776). GMM-based cutoffs were used to dichotomize CSF data for 67% of participants (n = 3652). Measurement details of amyloid biomarkers and applied cutoffs have been specified elsewhere. 17
Lifestyle factors
For each individual included, data was available on at least one of the following lifestyle factors: alcohol consumption, smoking behavior, sleep quality, physical activity, cognitive activity, or social activity. The lifestyle assessment was performed within six months of the amyloid assessment in 41 centers and within 12 months of the amyloid assessment in one center. Supplemental Tables 2 and 3 present measurement information and the number of participants with available data for each center. Lifestyle data was dichotomized as detailed below. If multiple measures for a lifestyle factor were available in one center, we prioritized rating scale data and other continuous data over self-reported and/or categorical data.
Alcohol consumption
Participants were classified as drinkers (>= 1 alcohol unit per week) or abstainers based on (1) information on alcohol units in drinks consumed per day or week (18 centers, n = 6215) or (2) medical history (25 centers, n = 1444). We did not include participants in the analysis of alcohol consumption if information was limited to alcohol abuse status.
Smoking status
Participants were classified as ever-smokers (past or present) or never-smokers based on (1) information on smoking units in cigarettes per day or week (10 centers, n = 5954) or (2) medical history (32 centers, n = 4713).
Sleep quality
Participants were classified as being with or without sleep problems based on (1) rating scales for sleep quality and sleep problems (8 centers, n = 2555), (2) sleep hours (8 centers, n = 5168), (3) a positive answer to the screening question on the sleep domain of the neuropsychiatric inventory questionnaire (NPI) or NPI-Q (15 centers, n = 1293), or (4) self-report (9 centers, n = 801). Rating scales provided included the Pittsburgh Sleep Quality Index (PSQI 22 ; sleep problems if score > 5), Epworth Sleep Quality Index (ESS 23 ; sleep problems if score > 10). and Profile of Elderly Quality of Life (PEQOL 24 ) Sleep disturbance (tertile-based cut-off). Participants who reported sleeping less than 6 or over 9 h of sleep per night were considered to have sleep problems.25,26
Physical activity
Participants were classified as being physically inactive or active based on (1) rating scales for physical activity (3 centers, n = 456), (2) frequency and/or intensity of physical exercise (11 centers, n = 6876), or (3) self-report (8 centers, n = 375). Rating scales provided included the International Physical Activity Questionnaire (IPAQ 27 ) and the Physical Activity Scale for the Elderly (PASE 28 ). To account for variability in activities classified as physical activity, cohort-specific cutoffs based on the upper tertile were applied to rating scale and frequency and/or intensity data.
Cognitive activity
Participants were classified as being cognitively inactive or active based on (1) the Wilson cognitive activity questionnaire 29 (current life epoch; 4 centers, n = 412), (2) frequency of participation in cognitive activities (3 centers, n = 349), or (3) self-report (4 centers, n = 201). Data on frequency of participation in cognitive activities was recoded to match the rating outcomes of the Wilson cognitive activity questionnaire (frequency rated on a scale of 1–5). Participants with an average score > 3.57 on the Wilson cognitive activity questionnaire or a score of > 3.57 as averaged over activities were considered to be cognitively active. 30
Social activity
Participants were classified as being socially inactive or active based on (1) frequency of participation in social activities (3 centers, n = 330), (2) frequency of meeting with friends (1 center, n = 118), (3) self-report (3 centers, n = 203), or (4) information on social network size (2 centers, n = 83). Participants were considered socially active if they participated in a social activity or met with friends at least once a week. A center-specific median cut-off was applied to classify participants as socially active or inactive based on information on their social network size.
Other covariates
Age, sex, and education-years were based on participant self-report.17–19
Statistical analysis
Differences in demographic and clinical characteristics between those with and without amyloid pathology were tested using ANOVA (continuous variables) or chi-square tests (categorical variables). Generalized estimating equations were used to assess the associations between the individual lifestyle factors and amyloid pathology. We assumed a logit link function for binary outcomes with an exchangeable working correlation matrix and robust variance estimators to account for within-study correlation.
Analyses were performed separately in participants with NC and participants with MCI. Age, sex, and education were included as covariates in all models, with age and education years included as continuous measures and age centered at the mean. We first tested the overall association between the individual lifestyle factors and amyloid pathology. In secondary analyses, we used continuous lifestyle measures (alcohol units per week, number of cigarettes per week, average number of hours slept each night) instead of dichotomous lifestyle measures.
The significance level was set at p < 0.05 for unpaired, 2-sided tests. Analyses were conducted with IBM SPSS Statistics version 27 and images were created using R (version 4.0.0, R Foundation for Statistical Computing). We used Bonferroni adjustment to correct for multiple comparisons when evaluating the primary outcomes. We report uncorrected p-values and note if the association was no longer significant after correction for multiple comparisons.
Results
Participant characteristics
Baseline characteristics are presented in Table 1, stratified by cognitive and amyloid status. Of participants with NC, 58% were women, 34% were APOE ε4 carrier, and 27% had amyloid pathology. Of participants with MCI, 48% were women, 47% were APOE ε4 carrier, and 57% had amyloid pathology. The mean age was 69 years and participants had a mean of 15 years of education. 47% of participants did not consume alcohol, 72% never smoked, 70% did not have sleep problems, 40% were physically active, 48% were cognitively active and 42% were socially active. NC participants with amyloid pathology were more likely to be older, more likely to be APOE ε4 carrier, more likely to have fewer years of education, and less likely to be recruited from a research setting than NC participants without amyloid pathology. MCI participants with amyloid pathology were more likely to be older, more likely to be APOE ε4 carrier, more likely to have more years of education, more likely to be recruited from a clinical setting, and less likely to have sleep problems or be cognitively or socially active than MCI participants without amyloid pathology.
Participant characteristics.
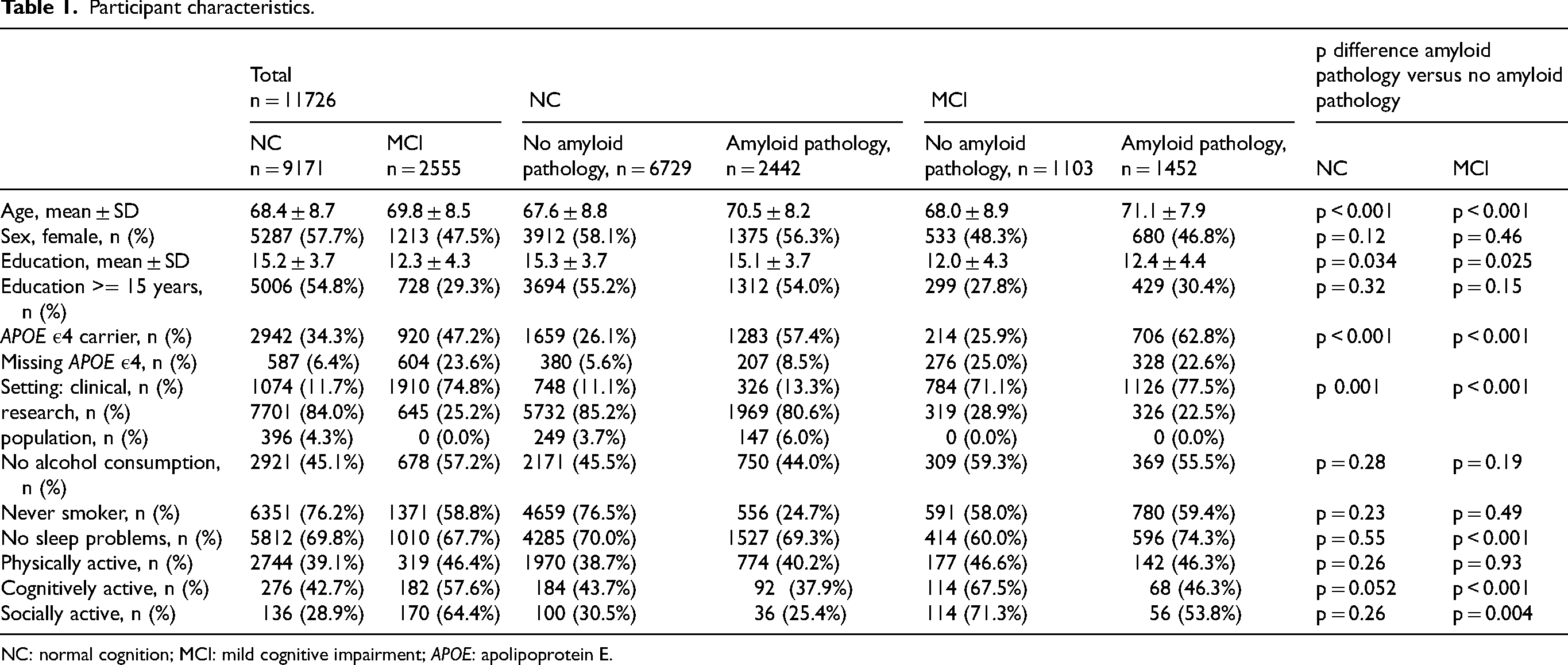
NC: normal cognition; MCI: mild cognitive impairment; APOE: apolipoprotein E.
Associations between dichotomous lifestyle factors and amyloid pathology
The odds ratios (ORs) for amyloid pathology corresponding to each lifestyle factor are presented in Figure 1.
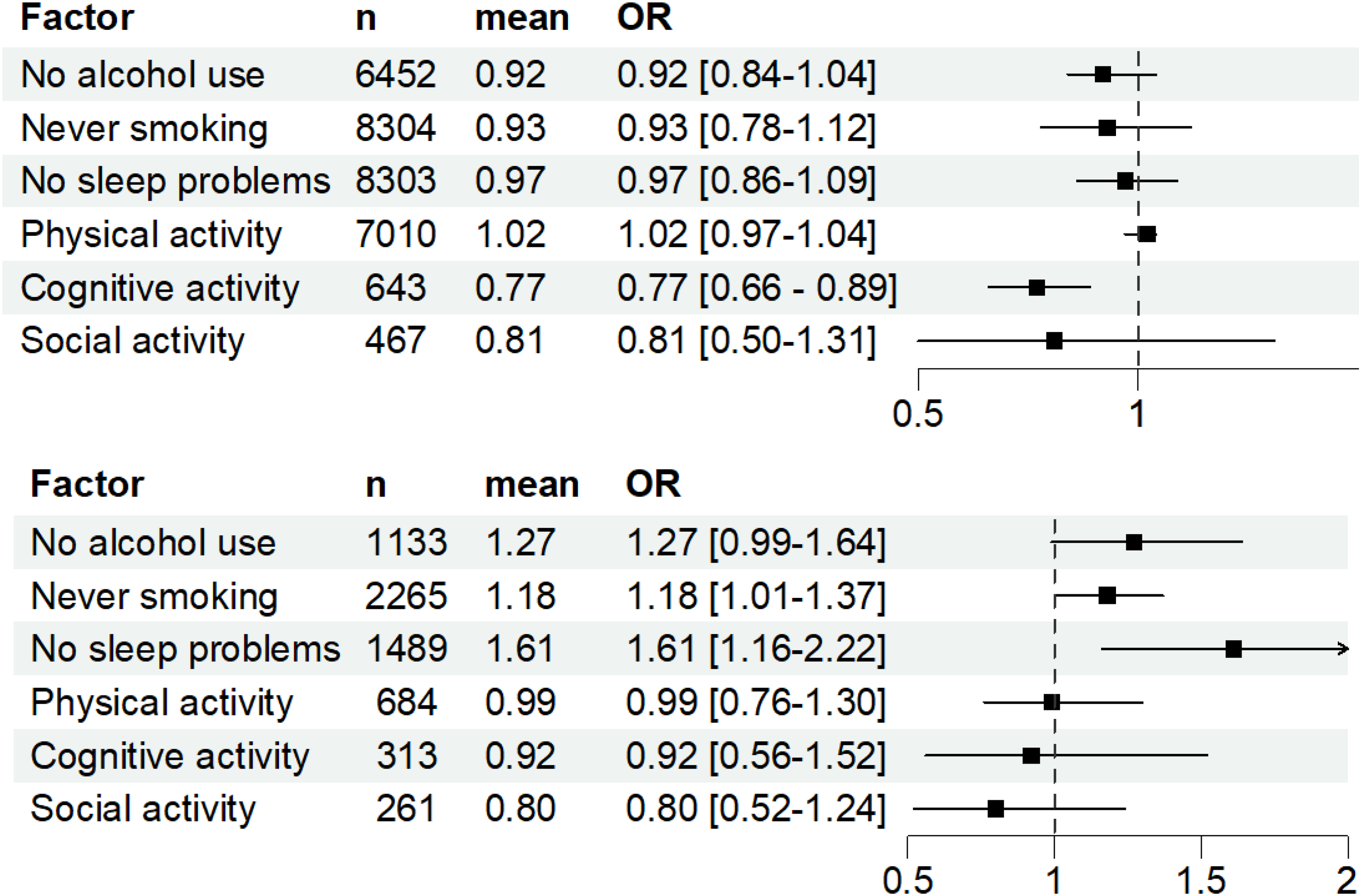
Odds ratios for amyloid pathology by cognitive group and lifestyle factor (top: NC, bottom: MCI).
Individuals with NC
In participants with NC, cognitive activity was associated with amyloid pathology. Cognitively active participants had amyloid pathology less often than cognitively inactive participants (OR = 0.77, 95% CI 0.66–0.89, p < 0.001). Alcohol consumption, ever smoking, sleep quality, physical activity, and social activity were not associated with amyloid pathology (no alcohol use: OR = 0.92, 95% CI 0.84–1.04, p = 0.060; never smoking: OR = 0.93, 95% CI 0.78–1.12, p = 0.45; no sleep problems: OR = 0.97, 95% CI 0.86–1.09, p = 0.59; physically active: OR = 1.02, 95% CI 0.97–1.04, p = 0.41; socially active: OR = 0.81, 95% CI 0.50–1.31, p = 0.39).
Individuals with MCI
In participants with MCI, those who had ever smoked and those who had sleep problems were less likely to have amyloid pathology (OR = 0.85, 95% CI 0.73–0.99, p = 0.029, not significant after correction for multiple comparisons; OR = 0.62, 95% CI 0.45–0.86, p = 0.004). Alcohol use, physical activity, cognitive activity, and social activity were not associated with amyloid pathology (no alcohol use: OR = 1.27, 95% CI 0.99–1.64, p = 0.062; physically active: OR = 0.99, CI 0.76–1.30, p = 0.95; cognitively active: OR = 0.92, CI 0.56– 1.52, p = 0.74; socially active: OR = 0.80, CI 0.52–1.24, p = 0.33).
Associations of continuous lifestyle factors with amyloid pathology
Individuals with NC
In participants with NC, we found that those who smoked a higher number of cigarettes per week were more likely to have amyloid pathology (b = 0.002, 95% CI 0.0–0.003, p = 0.018, Figure 2). Number of alcoholic drinks consumed and sleep duration were not associated with amyloid pathology (b = -0.001, 95%CI −0.005–0.004, p = 0.81; b = -0.048, 95%CI −0.198–0.11, p = 0.112, respectively).
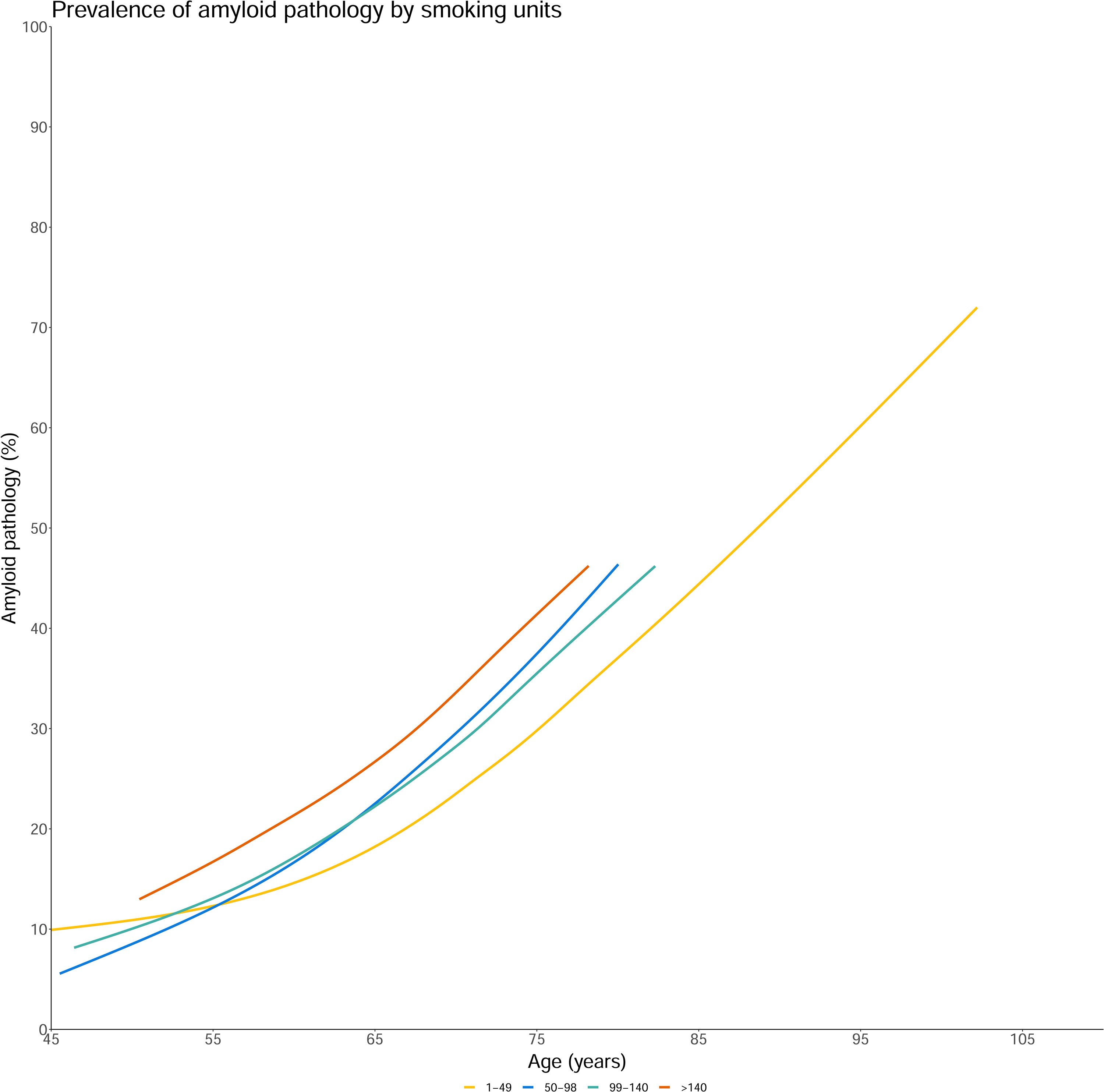
Association between smoking units per week and amyloid pathology in participants with NC.
Individuals with MCI
In participants with MCI, those who consumed a higher number of alcoholic drinks and smoked a higher number of cigarettes per week were less likely to have amyloid pathology (b = -0.23, 95% CI −0.040 – −0.006, p = 0.009; b = -0.004, 95% CI −0.005– −0.002, p < 0.001, respectively). Increased sleep duration was associated with higher odds of amyloid pathology (b = 0.61, 95%CI 0.54–0.68, p < 0.001).
Discussion
The aim of our study was to examine the associations of lifestyle factors with amyloid pathology in a large group of persons without dementia by harmonizing lifestyle and amyloid measures across 42 cohorts. We robustly harmonized amyloid measures and measures of alcohol consumption, smoking behavior, sleep quality and physical, cognitive, and social activity across cohorts. In participants with NC, we found that cognitively active persons were 23% less likely to have amyloid pathology. In NC, there was no association between the other lifestyle factors studied and amyloid pathology. In participants with MCI, we found that those who smoked and those who had sleep problems were less likely to have amyloid pathology (OR 0.62 and 0.85, respectively), though the association between smoking behavior and amyloid pathology was no longer significant after correction for multiple comparisons. In MCI, none of the favorable lifestyle behaviors studied were associated with a lower frequency of amyloid pathology.
Alcohol consumption and smoking behavior
Earlier work identified positive associations between alcohol consumption and p-tau/Aβ42 and t-tau/Aβ42 ratios in cognitively unimpaired individuals as well as positive associations between smoking behavior and amyloid pathology in cognitively unimpaired individuals and persons with AD dementia.15,16,32 While we did find an indication that NC participants who smoked a higher number of cigarettes per week were more likely to have amyloid pathology, we did not replicate the previously reported findings for alcohol consumption in this population. 31
Sleep duration and quality
In participants with NC, we did not find any association between sleep quality and amyloid pathology. In participants with MCI, worse sleep quality was associated with lower odds of amyloid pathology while increasing sleep duration was associated with higher odds of amyloid pathology. Effects of sleep on amyloid pathology generally seem difficult to disentangle due to potential disparities in the association of different aspects of sleep with amyloid pathology, though accumulating evidence suggests that sleep problems, more generally defined, are associated with increasing amyloid pathology.2–4,32–39
Our observation that unfavorable lifestyle behaviors were associated with a lower frequency of amyloid pathology in MCI, contrary to earlier observations, could have several explanations. Results may be reflective of underlying non-AD pathological mechanisms (e.g., vascular mechanisms, specifically for smoking) which resulted in participants presenting at the memory clinic with cognitive problems. Results may also be reflective of resilience mechanisms; indicating that the favorable lifestyle behaviors, i.e., no sleep problems, may mitigate or compensate for the effects of amyloid pathology on cognitive decline 40 . This would delay conversion to dementia and result in an increased frequency of amyloid pathology in those without sleep problems in the MCI phase when assessed cross-sectionally.
Past adherence to unfavorable lifestyle behaviors could also have biased results. For instance, differences in health status between participants who adhered to favorable versus unfavorable lifestyle behaviors previously may have affected study inclusion. Participants with MCI may moreover have recently modified their lifestyle patterns as results of the diagnosis. Longitudinal studies may help to overcome these biases.
Cognitive activity
We found that cognitively active NC participants were less likely to have amyloid pathology than cognitively inactive NC participants. This observed effect extends on earlier findings that more specifically identified early and midlife as well as overall lifetime cognitive activity to be associated with decreased Pittsburgh compound B (PiB) retention,10,12,41,42 supporting the idea that cognitive activity in NC may provide resistance (delaying the appearance of or retard pathological progression of) to amyloid pathology. 40
Physical activity
We did not find an association between physical activity and amyloid pathology in either the NC or MCI group. Earlier work mostly identified inverse associations or reported no effect.6,8–10,43–46 The measure of physical activity used, and more specifically which activities are considered to constitute physical activity, may strongly influence the observed associations, providing a potential explanation for the inconsistency in observations.
Social activity
We did not find an association of social activity with amyloid pathology in NC or MCI. The limited availability of data on social activity and the wide disparity in methods of measurement complicates interpretation. Relatively few studies up to date have focused on social activity in relation to amyloid pathology. Two identified an inverse association between social activity and amyloid pathology (PET and CSF, respectively), while two found no effects.5,6,13,14 To further study this association, detailed and fine-grained measures of social activity should ideally be used.
Harmonization of lifestyle data
In the current paper, we present a first approach to harmonizing lifestyle data across a large number of cohorts. To be able to integrate as much data as possible, we needed to create dichotomous outcome measures, which inevitably resulted in loss of granularity. However, it was the only way to utilize all lifestyle information that was currently, and is commonly, available across memory clinic and research cohorts. Given the existing heterogeneity in the literature and the sustained interest in the role of lifestyle in cognitive impairment and AD, the field may benefit from the creation of standard operating procedures for the collection and analysis of lifestyle data.
Strengths and limitations
The current study has several limitations. The inclusion of data from and harmonization across multiple cohorts may have introduced sources of variance including heterogeneity in the assessment of amyloid pathology and lifestyle. Lifestyle was measured cross-sectionally, by different and -most often- subjective instruments, which may have introduced additional bias through potential differences in underlying constructs measured and reporting and recall bias. We could not draw any conclusions regarding causality given the cross-sectional nature of the study. Lastly, we did not consider potential moderating effects of vascular risk factors, hearing loss, or interactions between lifestyle factors. Still, the collation of data and the resulting large sample size gave us the opportunity to study associations between lifestyle and amyloid pathology with sufficient power to detect small associations using lifestyle measures commonly available in memory clinic and research cohorts.
Conclusion
In this cross-sectional study, cognitive activity was associated with a lower frequency of amyloid pathology in those with NC, while other favorable lifestyle behaviors were not associated with a lower frequency of amyloid pathology. Generally, results suggest that cognitive activity may contribute to resistance to amyloid pathology in persons with NC, while in persons with MCI, identified associations may be reflective of non-AD pathological mechanisms or resilience mechanisms. The results of the current study contribute to the broader evidence base on lifestyle and Alzheimer's disease by further characterizing the role of lifestyle behaviors in AD pathology across different clinical stages. Longitudinal evaluation of associations between lifestyle factors and amyloid pathology, using detailed lifestyle measures, will be needed to fully establish the association between lifestyle and AD pathophysiology. The field may further benefit from the creation of standard operating procedures for the collection and analysis of lifestyle data.
Supplemental Material
sj-docx-1-alz-10.1177_13872877251379083 - Supplemental material for Associations of lifestyle factors with amyloid pathology in persons without dementia
Supplemental material, sj-docx-1-alz-10.1177_13872877251379083 for Associations of lifestyle factors with amyloid pathology in persons without dementia by Julie E Oomens, Stephanie JB Vos, Nancy N Maserejian, Merce Boada, Mira Didic, Sebastiaan Engelborghs, Tormod Fladby, Wiesje M van der Flier, Giovanni B Frisoni, Lutz Frohlich, Kiran Dip Gill, Timo Grimmer, Jakub Hort, Yoshiaki Itoh, Takeshi Iwatsubo, Aleksandra Klimkowicz-Mrowiec, Susan M Landau, Dong Young Lee, Alberto Lleo, Pablo Martinez-Lage, Alexandre de Mendonca, Philipp T Meyer, Piero Parchi, Matteo Pardini, Lucilla Parnetti, Julius Popp, Lorena Rami, Eric M Reiman, Juha O Rinne, Karen M Rodrigue, Pascual Sanchez-Juan, Isabel Santana, Nikolaos Scarmeas, Philip Scheltens, Ingmar Skoog, Reisa A Sperling, Yaakov Stern, Sylvia Villeneuve, Gunhild Waldemar, Jens Wiltfang, Henrik Zetterberg, Daniel Alcolea, Ricardo F Allegri, Daniele Altomare, Randall J Bateman, Simone Baiardi, Ines Baldeiras Kaj Blennow, Anouk den Braber, Mark A van Buchem, Min Soo Byun, Jiří Cerman, Kewei Chen, Elena Chipi, Gregory S Day, Alexander Drzezga, Laura L Ekblad, Stefan Forster, Juan Fortea, Yvonne Freund-Levi Lars Frings, Eric Guedj, Christian G Habeck, Ron Handels, Lucrezia Hausner, Sabine Hellwig, Julio F Jimenez-Bonilla, Ane Iriondo Juaristi, Ramesh Kandimalla Silke Kern, Bjorn-Eivind S Bordewick Kirsebom, Johannes Kornhuber, Nienke Legdeur, Johannes Levin Wolfgang Maier, Marta Marquie, Shinobu Minatani, Silvia Daniela Morbelli, Barbara Mroczko, Eva Ntanasi, Catarina Resende de Oliveira, Adelina Orellana, Oliver Peters, Sudesh Prabhakar, Inez H Ramakers, Eloy Rodriguez-Rodriguez, Agustin Ruiz, Eckart Ruther, Jayant Sakhardande, Per Selnes, Dina Silva, Hilkka Soininen, Luiza Spiru, Akitoshi Takeda, Charlotte E Teunissen, Betty M Tijms, Lisa Vermunt, Asa KWallin, Wietse Wiels, Mary Yannakoulia, Dahyun Yi, Anna Zettergren, for the Alzheimer's Disease Neuroimaging Initiative (ADNI), A4 Study group, Dominantly Inherited Alzheimer Network (DIAN), European Prevention of Alzheimer's Dementia (EPAD) consortium, Fundació ACE Healthy Brain Initiative (FACEHBI), Japan Alzheimer's Disease Neuroimaging Initiative (J-ADNI), Korean Brain Aging Study for the Early Diagnosis and Prediction of Alzheimer's Disease (KBASE), Presymptomatic Evaluation of Experimental or Novel Treatments for Alzheimer's Disease (PREVENT-AD) research group, Rik Ossenkoppele, Frans RJ Verhey, Pieter Jelle Visser, and Willemijn J Jansen in Journal of Alzheimer's Disease
Supplemental Material
sj-docx-2-alz-10.1177_13872877251379083 - Supplemental material for Associations of lifestyle factors with amyloid pathology in persons without dementia
Supplemental material, sj-docx-2-alz-10.1177_13872877251379083 for Associations of lifestyle factors with amyloid pathology in persons without dementia by Julie E Oomens, Stephanie JB Vos, Nancy N Maserejian, Merce Boada, Mira Didic, Sebastiaan Engelborghs, Tormod Fladby, Wiesje M van der Flier, Giovanni B Frisoni, Lutz Frohlich, Kiran Dip Gill, Timo Grimmer, Jakub Hort, Yoshiaki Itoh, Takeshi Iwatsubo, Aleksandra Klimkowicz-Mrowiec, Susan M Landau, Dong Young Lee, Alberto Lleo, Pablo Martinez-Lage, Alexandre de Mendonca, Philipp T Meyer, Piero Parchi, Matteo Pardini, Lucilla Parnetti, Julius Popp, Lorena Rami, Eric M Reiman, Juha O Rinne, Karen M Rodrigue, Pascual Sanchez-Juan, Isabel Santana, Nikolaos Scarmeas, Philip Scheltens, Ingmar Skoog, Reisa A Sperling, Yaakov Stern, Sylvia Villeneuve, Gunhild Waldemar, Jens Wiltfang, Henrik Zetterberg, Daniel Alcolea, Ricardo F Allegri, Daniele Altomare, Randall J Bateman, Simone Baiardi, Ines Baldeiras Kaj Blennow, Anouk den Braber, Mark A van Buchem, Min Soo Byun, Jiří Cerman, Kewei Chen, Elena Chipi, Gregory S Day, Alexander Drzezga, Laura L Ekblad, Stefan Forster, Juan Fortea, Yvonne Freund-Levi Lars Frings, Eric Guedj, Christian G Habeck, Ron Handels, Lucrezia Hausner, Sabine Hellwig, Julio F Jimenez-Bonilla, Ane Iriondo Juaristi, Ramesh Kandimalla Silke Kern, Bjorn-Eivind S Bordewick Kirsebom, Johannes Kornhuber, Nienke Legdeur, Johannes Levin Wolfgang Maier, Marta Marquie, Shinobu Minatani, Silvia Daniela Morbelli, Barbara Mroczko, Eva Ntanasi, Catarina Resende de Oliveira, Adelina Orellana, Oliver Peters, Sudesh Prabhakar, Inez H Ramakers, Eloy Rodriguez-Rodriguez, Agustin Ruiz, Eckart Ruther, Jayant Sakhardande, Per Selnes, Dina Silva, Hilkka Soininen, Luiza Spiru, Akitoshi Takeda, Charlotte E Teunissen, Betty M Tijms, Lisa Vermunt, Asa KWallin, Wietse Wiels, Mary Yannakoulia, Dahyun Yi, Anna Zettergren, for the Alzheimer's Disease Neuroimaging Initiative (ADNI), A4 Study group, Dominantly Inherited Alzheimer Network (DIAN), European Prevention of Alzheimer's Dementia (EPAD) consortium, Fundació ACE Healthy Brain Initiative (FACEHBI), Japan Alzheimer's Disease Neuroimaging Initiative (J-ADNI), Korean Brain Aging Study for the Early Diagnosis and Prediction of Alzheimer's Disease (KBASE), Presymptomatic Evaluation of Experimental or Novel Treatments for Alzheimer's Disease (PREVENT-AD) research group, Rik Ossenkoppele, Frans RJ Verhey, Pieter Jelle Visser, and Willemijn J Jansen in Journal of Alzheimer's Disease
Footnotes
Acknowledgements
Members of the following groups contributed to this article and are listed in online Appendix 1: Alzheimer's Disease Neuroimaging Initiative (ADNI; protocol in Supplemental Material 1), A4 Study group, Dominantly Inherited Alzheimer Network (DIAN), Fundació ACE Healthy Brain Initiative (FACEHBI), Korean Brain Aging Study for the Early Diagnosis and Prediction of Alzheimer's disease (KBASE), and the Presymptomatic Evaluation of Experimental or Novel Treatments for Alzheimer's Disease (PREVENT-AD) research group.
Data used in preparation of the present article were obtained from the ADNI database (adni.loni.usc.edu). As such, ADNI investigators provided and contributed to the design and implementation of the ADNI data but did not participate in the analysis or writing of this article. A complete listing of ADNI investigators can be found at http://adni.loni.usc.edu/wp-content/ uploads/how_to_apply/ADNI_Acknowledgement_ List.pdf. The ADNI was launched in 2003 as a public-private partnership, led by principal investigator Michael W. Weiner, MD. The primary goal of ADNI is to test whether serial magnetic resonance imaging, PET, other biological markers, and clinical and neuropsychological assessment can be combined to measure the progression of mild cognitive impairment and early Alzheimer disease. Data collection and sharing for this project was funded by grant U01 AG024904 from the NIH and award W81XWH-12–2-0012 from the US Department of Defense. Data collection and sharing for this project was funded by the Alzheimer's Disease Neuroimaging Initiative (ADNI) (National Institutes of Health Grant U01 AG024904) and DOD ADNI (Department of Defense award number W81XWH-12-2-0012). ADNI is funded by the National Institute on Aging, the National Institute of Biomedical Imaging and Bioengineering, and through generous contributions from the following: AbbVie, Alzheimer's Association; Alzheimer's Drug Discovery Foundation; Araclon Biotech; BioClinica, Inc.; Biogen; Bristol-Myers Squibb Company; CereSpir, Inc.; Cogstate; Eisai Inc.; Elan Pharmaceuticals, Inc.; Eli Lilly and Company; EuroImmun; F. Hoffmann-La Roche Ltd and its affiliated company Genentech, Inc.; Fujirebio; GE Healthcare; IXICO Ltd; Janssen Alzheimer Immunotherapy Research & Development, LLC.; Johnson & Johnson Pharmaceutical Research & Development LLC.; Lumosity; Lundbeck; Merck & Co., Inc.; Meso Scale Diagnostics, LLC.; NeuroRx Research; Neurotrack Technologies; Novartis Pharmaceuticals Corporation; Pfizer Inc.; Piramal Imaging; Servier; Takeda Pharmaceutical Company; and Transition Therapeutics. The Canadian Institutes of Health Research is providing funds to support ADNI clinical sites in Canada. Private sector contributions are facilitated by the Foundation for the National Institutes of Health (![]() ). The grantee organization is the Northern California Institute for Research and Education, and the study is coordinated by the Alzheimer's Therapeutic Research Institute at the University of Southern California. ADNI data are disseminated by the Laboratory for Neuro Imaging at the University of Southern California.
). The grantee organization is the Northern California Institute for Research and Education, and the study is coordinated by the Alzheimer's Therapeutic Research Institute at the University of Southern California. ADNI data are disseminated by the Laboratory for Neuro Imaging at the University of Southern California.
The A4 and Longitudinal Evaluation of Amyloid Risk and Neurodegeneration (LEARN) studies are led by Dr Sperling at Brigham and Women's Hospital, Harvard Medical School, and by Dr Paul Aisen at the ATRI. The A4 and LEARN studies are coordinated by the ATRI, and the data are made available through the Laboratory for Neuroimaging. The participants screening for the A4 Study provided permission to share their deidentified data to advance the objective to find a successful treatment for Alzheimer disease. The A4 Study is a secondary prevention trial in preclinical Alzheimer disease that aims to slow cognitive decline associated with brain amyloid accumulation in clinically normal older individuals.
A proportion of the data used in preparation of this article was obtained from the following: LEARN study, which was performed within the framework of the Center for Translational Molecular Medicine, a Dutch public-private partnership (project LEARN; grant 02 N-101 from Center for Translational Molecular Medicine); DIAN (grant UF01AG032438 from NIA), which was funded by the NIA, German Center for Neurodegenerative Diseases, Raul Carrea Institute for Neurological Research, and partially by the research and development grants for dementia from Japan Agency for Medical Research and Development and Korea Health Technology Research and Development Project through the Korea Health Industry Development Institute; and PREVENT-AD program (https://douglas.research. mcgill.ca/stop-ad-centre; data release 5.0, November 30, 2017), which provided and contributed to the design and implementation of PREVENT-AD data but did not participate in the analysis or writing of this article. A complete listing of PREVENT-AD research group investigators can be found at ![]() acknowledgements/acknowledgements.php?date = [2020 = 04–01].
acknowledgements/acknowledgements.php?date = [2020 = 04–01].
Data collection and sharing for this project was supported by The Dominantly Inherited Alzheimer Network (DIAN, U19AG032438) funded by the National Institute on Aging (NIA),the Alzheimer's Association (SG-20-690363-DIAN), the German Center for Neurodegenerative Diseases (DZNE), Raul Carrea Institute for Neurological Research (FLENI), Partial support by the Research and Development Grants for Dementia from Japan Agency for Medical Research and Development, AMED, and the Korea Health Technology R&D Project through the Korea Health Industry Development Institute (KHIDI), Spanish Institute of Health Carlos III (ISCIII), Canadian Institutes of Health Research (CIHR), Canadian Consortium of Neurodegeneration and Aging, Brain Canada Foundation, and Fonds de Recherche du Québec—Santé. This manuscript has been reviewed by DIAN Study investigators for scientific content and consistency of data interpretation with previous DIAN Study publications. We acknowledge the altruism of the participants and their families and contributions of the DIAN research and support staff at each of the participating sites for their contributions to this study.
The FACEHBI study was supported by funds from Fundació ACE Institut Català de Neurociències Aplicades, Grifols, Life Molecular Imaging, Araclon Biotech, Alkahest, Laboratorio de Análisis Echevarne and IrsiCaixa.
The KBASE cohort was funded by the Ministry of Health & Welfare, South Korea (HI18C0630 & HI19C0149), the Ministry of Science and ICT (NRF-2014M3C7A1046042), and the NIA, United States (U01AG072177). The overview of the study can be found at ![]() .
.
Part of the present study was supported by the European Medical Information Framework Alzheimer's Disease (EMIF- AD), which received support from the Innovative Medicines Initiative Joint Undertaking under EMIF-AD grant agreement 115372 that comprised financial contribution from the EU's Seventh Framework Program (FP7/2007-2013) and in kind contribution from EFPIA companies. Research of Alzheimer Centre Amsterdam was part of the neurodegeneration research program of Amsterdam Neuroscience. Alzheimer Centre Amsterdam was supported by Stichting Alzheimer Nederland and Stichting VUmc Fonds. The SCIENCe project was supported by research grants from Gieskes-Strijbis Fonds and Stichting Dioraphte. PET scans in the Amsterdam Dementia Cohort were obtained with research grants from GE Healthcare, Life Molecular Imaging, AVID, and ZonMW- Memorabel, the research and innovation program for dementia.
The Czech Brain Aging Study was supported by project No. LX22NPO5107)—Funded by the European Union—Next Generation EU. Research of Alzheimer center Amsterdam is part of the neurodegeneration research program of Amsterdam Neuroscience. Alzheimer Center Amsterdam is supported by Stichting Alzheimer Nederland and Stichting Steun Alzheimercentrum Amsterdam. The chair, Wiesje van der Flier, is supported by the Pasman stichting.
The Sant Pau Memory Unit received funding from CIBERNED, ISCIII, which is jointly funded by FEDER, EU, Una manera de hacer Europa; Generalitat de Catalunya; Fundació La Marató TV3 Fundació Bancària Obra Social La Caixa; Fundación BBVA; Fundación Española para el Fomento de la Investigación de la Esclerosis Lateral Amiotrófica; Global Brain Health Institute; Fundació Catalana Síndrome de Down; and Fundació Víctor Grífols i Lucas.
The Phoenix Arizona APOE Cohort was funded by grant R01 AG031581 from the NIA. The Imabio3 and Shatau7-Imataustudies (Sarazin,Paris)were supported by grants PHRC-0054-N 2010 and PHRC-2013-0919 from the French Health Ministry, the Institut Roche de Recherche et Medecine Translationelle (Imabio3), Service Hospitalier Frédéric Joliot, Fondation pour la Recherchesur Alzheimer, Institut de Recherches Internationales Servier, and France-Alzheimer (Shatau7-Imatau).
The Nijmegen cohort was supported by the BIONIC project (grant 733050822 from ZonMW- Memorabel as part of the Dutch National Deltaplan for Dementia [zonmw.nl/dementiaresearch]), the CAFÉ project (grant 5R01NS104147-02 from the NIH), and the Selfridges Group Foundation (grant NR170024). The BIONIC project is a consortium of Radboudumc, LUMC, ADX Neurosciences, and Rhode Island University.
A part data used for this research was originally obtained from the Japanese ADNI database. Japanese ADNI is led by Prof Iwatsubo and the databse is available at the website of the NBDC Human Database (![]() ) of the Japan Science and Technology Agency (JST).
) of the Japan Science and Technology Agency (JST).
This work is supported by the Indian Council of Medical Research (ICMR), 52/3/2003, New Delhi, India. Ramesh Kandimalla is a senior research fellow (SRF) from ICMR, New Delhi, India.
HZ is a Wallenberg Scholar and a Distinguished Professor at the Swedish Research Council supported by grants from the Swedish Research Council (#2023-00356; #2022-01018 and #2019-02397), the European Union's Horizon Europe research and innovation programme under grant agreement No 101053962, Swedish State Support for Clinical Research (#ALFGBG-71320), the Alzheimer Drug Discovery Foundation (ADDF), USA (#201809-2016862), the AD Strategic Fund and the Alzheimer's Association (#ADSF-21-831376-C, #ADSF-21-831381-C, #ADSF-21-831377-C, and #ADSF-24-1284328-C), the Bluefield Project, Cure Alzheimer's Fund, the Olav Thon Foundation, the Erling-Persson Family Foundation, Stiftelsen för Gamla Tjänarinnor, Hjärnfonden, Sweden (#FO2022-0270), the European Union's Horizon 2020 research and innovation programme under the Marie Skłodowska-Curie grant agreement No 860197 (MIRIADE), the European Union Joint Programme—Neurodegenerative Disease Research (JPND2021-00694), the National Institute for Health and Care Research University College London Hospitals Biomedical Research Centre, and the UK Dementia Research Institute at UCL (UKDRI-1003).
Ethical considerations
The study protocol for each cohort was approved by the local ethics committee at each site and study procedures were conducted in accordance with the Declaration of Helsinki. The present study was approved by the Medical Ethics Committee of the Maastricht University Medical Center, which declared that the Medical Research Involving Human Subjects Act (WMO) does not apply to the study and waived the informed consent requirement because deidentified data were used.
Consent to participate
Written informed consent was obtained from all participants in each study, and data were de-identified by the respective cohorts.
Author contribution(s)
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The Amyloid Biomarker Study is supported by Biogen.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Dr Alcolea reported participating in advisory boards from Fujirebio-Europe and Roche Diagnostics; receiving speaker honoraria from Fujirebio-Europe, Roche Diagnostics, Nutricia, Krka Farmacéutica SL, Zambon SAU, and Esteve Pharmaceuticals SA; and filing a patent application (WO2019175379 A1 Markers of Synaptopathy in Neurodegenerative Disease).
Dr Altomare received funding by the Fondation Recherche Alzheimer, and the Swiss National Science Foundation (project CRSK-3_196354 / 1).
Dr Boada reported funding from the Instituto de Salud Carlos III (ISCIII) Acción Estratégica en Salud, integrated in the Spanish National RCDCI Plan and financed by ISCIII-Subdirección General de Evaluación and the Fondo Europeo de Desarrollo Regional (FEDER—Una manera de hacer Europa) grant PI17/01474; consulting for for Araclon, Avid, Grifols, Lilly, Nutricia, Roche, Eisai and Servier, receiving fees from lectures and funds for research from Araclon, Biogen, Grifols, Nutricia, Roche and Servier and grants/research funding from Abbvie, Araclon, Biogen Research Limited, Bioiberica, Grifols, Lilly, S.A, Merck Sharp & Dohme, Kyowa Hakko Kirin, Laboratorios Servier, Nutricia SRL, Oryzon Genomics, Piramal Imaging Limited, Roche Pharma SA, and Schwabe Farma Iberica SLU.
Dr Engelborghs reported participating in consultancy or on advisory boards of Biogen, Danone, Eisai Inc, Icometrix, Pfizer, Novartis, and Roche, and receiving unrestricted research grants (paid to institution) from ADx Neurosciences and Janssen Pharmaceutica.
Dr van der Flier reports research programs have been funded by ZonMW, NWO, EU-JPND, Alzheimer Nederland, Hersenstichting CardioVascular Onderzoek Nederland, Health∼Holland, Topsector Life Sciences & Health, stichting Dioraphte, Gieskes-Strijbis fonds, stichting Equilibrio, Edwin Bouw fonds, Pasman stichting, stichting Alzheimer & Neuropsychiatrie Foundation, Philips, Biogen MA Inc, Novartis-NL, Life-MI, AVID, Roche BV, Fujifilm, Eisai, Combinostics. WF holds the Pasman chair. She is recipient of ABOARD, which is a public-private partnership receiving funding from ZonMW (#73305095007) and Health∼Holland, Topsector Life Sciences & Health (PPP-allowance; #LSHM20106). All funding is paid to her institution. She has been an invited speaker at Biogen MA Inc, Danone, Eisai, WebMD Neurology (Medscape), NovoNordisk, Springer Healthcare, NovoNordisk, European Brain Council. All funding is paid to her institution. She is consultant to Oxford Health Policy Forum CIC, Roche, Biogen MA Inc, and Eisai. All funding is paid to her institution. She participated in advisory boards of Biogen MA Inc, Roche, and Eli Lilly. All funding is paid to her institution. She is a member of the steering committee of PAVE, and Think Brain Health. She was associate editor of Alzheimer, Research & Therapy in 2020/2021. WF is associate editor at Brain.
Dr Grimmer reported receiving consulting fees from AbbVie, Alector, Anavex, Biogen, BMS; Cogthera, Eli Lilly, Functional Neuromodulation, Grifols, Iqvia, Janssen, Noselab, Novo Nordisk, NuiCare, Orphanzyme, Roche Diagnostics, Roche Pharma, UCB, and Vivoryon; lecture fees from Biogen, Eisai, Grifols, Medical Tribune, Novo Nordisk, Roche Pharma, Schwabe, and Synlab; and has received grants to his institution from Biogen, Eisai, and Roche Diagnostics.
Dr Guedj reported having a scientific collaboration on amyloid positron emission tomography (PET) imaging with Life Molecular Imaging before 2018, and honorarium from General Electric and Life Molecular Imaging for expertise and training in the last 3 years.
Dr Hort has consulted Eisai, Biogen, Eli Lilly, Roche and Neurona lab and holds stock options in Alzheon company.
Dr Jiménez-Bonilla reports receiving the Investigative IDIVAL grant: INNVAL-21-15.
Dr Kern reported honorarium for consulting for Geras Solutions and Biogen and Bioarctic.
Dr Levin reports speaker fees from Bayer Vital, Biogen, EISAI, TEVA, Zambon, Esteve, Merck and Roche, consulting fees from Axon Neuroscience, EISAI and Biogen, author fees from Thieme medical publishers and W. Kohlhammer GmbH medical publishers and is inventor in a patent “Oral Phenylbutyrate for Treatment of Human 4-Repeat Tauopathies” (PCT/EP2024/053388) filed by LMU Munich. In addition, he reports compensation for serving as chief medical officer for MODAG GmbH, is beneficiary of the phantom share program of MODAG GmbH and is inventor in a patent “Pharmaceutical Composition and Methods of Use” (EP 22 159 408.8) filed by MODAG GmbH, all activities outside the submitted work.
Dr Marquié reported funding from the Instituto de Salud Carlos III (ISCIII) Acción Estratégica en Salud, integrated in the Spanish National RCDCI Plan and financed by ISCIII-Subdirección General de Evaluación and the Fondo Europeo de Desarrollo Regional (FEDER—Una manera de hacer Europa) grant PI19/00335, consulting for F. Hoffmann-La Roche Ltd and participating in advisory boards from Araclon Biotech-Grifols.
Dr Maserejian is an employee and shareholder of Biogen, Inc.
Dr Mroczko has received consultation and/or lecture honoraria from Abbott, Wiener, Roche, Cormay, Biameditek, and TK Biotech companies.
Dr Nobili reported receiving fees for teaching courses from GE Healthcare and Biogen, for advisory board participation from Roche and Biogen, and for consultation from Bial.
Dr Ossenkoppele has received research funding from European Research Council, ZonMw, NWO, National Institute of Health, Alzheimer Association, Alzheimer Nederland, Stichting Dioraphte, Cure Alzheimer's fund, Health Holland, ERA PerMed, Alzheimerfonden, Hjarnfonden (all paid to the institutions).R.O. has received research support from Avid Radiopharmaceuticals, Janssen Research & Development, Roche, Quanterix and Optina Diagnostics, and has given lectures in symposia sponsored by GE Healthcare. He is an advisory board member for Asceneuron and Bristol Myers Squibb. All the aforementioned has been paid to the institutions. He is an editorial board member of Alzheimer's Research & Therapy and the European Journal of Nuclear Medicine and Molecular Imaging.
Dr Popp reported receiving consultation and speaker honoraria from Nestle Institute of Health Sciences, Innovation Campus, EPFL, Ono Pharma, OM Pharma Suisse, and Fujirebio Europe.
Dr Rami reported funding from the Instituto de Salud Carlos III (ISCIII) Acción Estratégica en Salud, integrated in the Spanish National RCDCI Plan and financed by ISCIII-Subdirección General de Evaluación and the Fondo Europeo de Desarrollo Regional (FEDER—Una manera de hacer Europa) grant PI19/00745.
Dr Ruiz reported receiving support from Centro de Investigación Biomédica en Red sobre Enfermedades Neurodegenerativas (CIBERNED), Instituto de Salud Carlos III (ISCIII); the EU/European Federation of Pharmaceutical Industries and Associations (EFPIA) Innovative Medicines Initiative Joint Undertaking; grants from the EXIT (Exosomes Isolation Tool with Nanofluidic Concentration Device) project, EU Euronanomed3 Program, and PREADAPT project; grant from the Joint Program for Neurodegenerative Diseases; and research funding from ISCIII Acción Estratégica en Salud, which was integrated in the Spanish National RCDCI Plan and financed by a grant from ISCIII-Subdirección General de Evaluación and the Fondo Europeo de Desarrollo Regional (FEDER; Una manera de hacer Europa) by Fundación Bancaria La Caixa and Grífols SA (GR@ACE project).
Dr Scarmeas reported being a local recruiting site PI for an industry (NovoNordisc) funded multinational, multicenter industry sponsored phase III treatment trial for Alzheimer's disease—funding to institution.
Dr Snyder reported being a consultant to Alzheon Inc, AlzeCure Pharma, and AlzPATH Inc outside the submitted work.
Dr Soininen reported receiving personal consultation fees from AC Immune and Novo Nordisk outside the submitted work.
Dr Sperling reported receiving honorarium for consulting from AC Immune, Acumen, Alnylam, Cytox, Genentech, Janssen, JOMDD, Oligomerix, Neuraly, Neurocentria, Renew, Prothena, and Shionogi; reported receiving research funding from the National Institute on Aging (NIA), Alzheimer's Association, Eisai Inc, Eli Lilly and Company, and Janssen; and reported the following financial relationships for her spouse (Dr Keith Johnson): Cerveau, Janssen, AC Immune, and Novartis.
Dr Teunissen reported receiving research support from the European Commission (Marie Curie International Training Network and Joint Program for Neurodegenerative Diseases grants), Health Holland, the Dutch Research Council (ZonMW), Alzheimer Drug Discovery Foundation, The Selfridges Group Foundation, Alzheimer Netherlands, Alzheimer Association, and ABOARD (A Personalized Medicine Approach for Alzheimer's Disease), which is a public-private partnership supported by ZonMW, Alzheimer Nederland, Health Holland, Gieskes-Strijbisfonds, and Edwin Bouw Fonds; having a collaboration contract with ADx Neurosciences, Quanterix, and Eli Lilly and Company; performing contract research or receiving grants from AC Immune, Axon Neurosciences, Biogen, Brainstorm Therapeutics, Celgene, EIP Pharma, Eisai Inc, PeopleBio, Roche, Toyama, and Vivoryon; serving on editorial boards of Medidact Neurologie/ Springer, Alzheimer Research and Therapy, and Neurology: Neuroimmunology & Neuroinflammation; and being editor of a neuromethods book from Springer.
Dr Zetterberg reported having served at scientific advisory boards and/or as a consultant for Abbvie, Acumen, Alector, Alzinova, ALZPath, Annexon, Apellis, Artery Therapeutics, AZTherapies, Cognito Therapeutics, CogRx, Denali, Eisai, Nervgen, Novo Nordisk, Optoceutics, Passage Bio, Pinteon Therapeutics, Prothena, Red Abbey Labs, reMYND, Roche, Samumed, Siemens Healthineers, Triplet Therapeutics, and Wave, has given lectures in symposia sponsored by Alzecure, Biogen, Cellectricon, Fujirebio, Lilly, and Roche, and is a co-founder of Brain Biomarker Solutions in Gothenburg AB (BBS), which is a part of the GU Ventures Incubator Program (outside submitted work).
Dr Scheltens is a full-time employee of EQT Life Sciences (formerly LSP) and Professor Emeritus at Amsterdam University Medical Centers. He has received consultancy fees (paid to the university) from Alzheon, Brainstorm Cell and Green Valley. Within his university affiliation he was global PI of the phase 1b study of AC Immune, Phase 2b study with FUJI-film/Toyama and phase 2 study of UCB. He is past chair of the EU steering committee of the phase 2b program of Vivoryon and the phase 2b study of Novartis Cardiology and presently co-chair of the phase 3 study with NOVO-Nordisk.
Sebastiaan Engelborghs, Matteo Pardini, Yaakov Stern, Henrik Zetterberg, and Ines Baldeiras are Editorial Board Members of this journal but were not involved in the peer-review process of this article nor had access to any information regarding its peer-review.
The remaining authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article. Dr Jansen reported funding from ZonMw, Alzheimer Nederland, St. Rinsum-Ponssen and research support from Biogen (paid to institution).
Data availability statement
Data access is granted upon approval of individual cohort owners. Researchers interested in working with Amyloid Biomarker Study data can send a research proposal to Dr Willemijn Jansen (willemijn.jansen@maastrichtunivesrity.nl).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
