Abstract
Background
Plasma p-tau217 shows exceptional diagnostic performance for Alzheimer's disease (AD) in cohort studies. Recent real-world studies support its validity in more diverse populations.
Objective
Test the performance of plasma p-tau217 in representative real-world early-onset cognitive disorders patients.
Methods
118 adult patients with cognitive complaints or potential neurodegenerative diseases (ND) from a single healthcare network in Melbourne, Australia, were categorized into three groups based on clinical diagnosis: non-ND (n = 52), early-onset AD (EOAD, n = 39), and non-AD ND (other-ND, n = 27). Plasma p-tau217 was measured using the Simoa® ALZpath p-Tau 217 Assay.
Results
Plasma p-tau217 was elevated in EOAD (mean ± SD: 1.13 ± 0.51) compared with non-ND (0.31 ± 0.27, p = 5.43e-23) and other-ND (0.23 ± 0.10, p = 9.65e-21). ROC analysis revealed excellent diagnostic performance, with AUCs of 0.944 [95% CI:0.893–0.996] for EOAD versus non-ND and 0.984 [0.962–1.000] for EOAD versus other-ND. Using a binary cut-off (p-tau217 > 0.42 pg/mL), 17% non-ND, 95% EOAD and 7% other-ND were A + . Using a two-tiered cut-off approach (0.40 pg/mL > p-tau217 > 0.63 pg/mL), 4% non-ND, 94% EOAD and 0% other-ND were A + . 10% of participants (n = 12) were within the intermediate range. Using the binary cut-off of p-tau217 > 0.64 pg/mL for T+, 100% EOAD and 0% other-ND A + participants, defined by the two-tiered cut-off approach, were T + . No associations of p-tau217 with age, sex, or ethnicity were found within ND groups. P-tau217 was elevated in individuals with severely impaired renal function.
Conclusions
The findings support the clinical utility of plasma p-tau217 in the real-world diagnostic evaluation of EOAD. However, careful interpretation of false positive results in patients with severe renal impairment is required.
Introduction
Alzheimer's disease (AD) with symptom onset before the age of 65 years, known as early-onset AD (EOAD), presents unique diagnostic challenges due to its heterogeneous clinical presentations. Individuals with EOAD more commonly present with atypical or variant AD, such as posterior cortical atrophy (PCA), logopenic variant primary progressive aphasia or frontal/dysexecutive AD.1–4
The cerebrospinal fluid (CSF) amyloid-β (Aβ) and tau or Aβ-PET5,6 gold standard biomarkers that enable a biologically informed diagnosis of AD are invasive or expensive. Blood-based biomarkers are emerging as promising cost-effective and accessible tools for biologically informed diagnosis of AD.7–9 Among these, plasma tau phosphorylated at the threonine-217 residue (p-tau217) has demonstrated exceptional concordance with core AD pathology, assessed using CSF Aβ and tau or Aβ-PET.10–14 Plasma p-tau217 may represent a superior biomarker compared with the other putative plasma biomarkers for AD in differentiating individuals with AD from those who are cognitively unimpaired15–18 and those with non-AD neurodegenerative disorders.19–24 However, previous studies evaluating the diagnostic value of plasma p-tau217 have largely had strict inclusion and exclusion criteria, using fasted blood samples, which can be difficult for individuals with cognitive disorders and medical comorbidities, limiting the generalizability to real-world clinical settings. New real-world data on plasma p-tau217 highlight the potential for translation of this diagnostic,12,25,26 but the application to EOAD remains less explored.
Relatively recent studies on plasma p-tau217 have determined cut-offs for Aβ and tau positivity measured using the ALZpath p-Tau217 assay.10,12 Additionally, to address the challenge of intermediate plasma p-tau217 values near the Aβ positivity threshold, a ‘two-cutoff’ approach has also been determined. 10 To explore the generalizability of these cut-offs, and to evaluate their real-world diagnostic performance in clinical settings and whether their application extends to EOAD, is paramount for clinical deployment.
Therefore, in this study, we aimed to (1) Compare plasma p-tau217 levels in individuals clinically diagnosed with EOAD to those with non-neurodegenerative (ND) conditions (non-ND) and those with other-ND (e.g., frontotemporal dementia, dementia with Lewy bodies) in patients presenting with cognitive complaints to an Australian healthcare network, and evaluate the diagnostic performance of plasma p-tau217 in differentiating clinically diagnosed EOAD from non-ND and other-ND; (2) Evaluate the application of pre-published cut-offs (binary and two-tiered) to determine Aβ positivity and tau positivity status in clinically diagnosed EOAD; and (3) Evaluate the association of p-tau217 with age, sex, ethnicity and renal function within different diagnostic groups. Group classifications were made based on multidisciplinary clinical diagnosis using established consensus criteria, with CSF or Aβ-PET testing available for only a subset of participants. We aim to inform the clinical implementation of plasma p-tau217 as a scalable and accessible biomarker for EOAD diagnosis and patient management.
Methods
Participants and clinical setting
Study participants comprised adult patients undergoing diagnostic assessment for cognitive disorders at Eastern Health in Melbourne, Australia. 27 Eastern Health is a metropolitan healthcare network comprising over 40 clinical locations and 5 hospitals. Eastern Cognitive Disorders Clinic is located at Box Hill Hospital, a tertiary care university setting. Eastern Health has over 1.5 million episodes of patient care annually including >325,000 outpatient specialist presentations. The Eastern Health network serves a large geographical area and comprises a diverse and multiracial population, of which 26% have a non–English-speaking background. Clinical diagnosis is determined by a multidisciplinary panel, including cognitive neurologists, neuropsychologists, speech pathologists, an occupational therapist, and a cognitive nurse consultant. Case discussions include a presentation of the history and informant report; viewing and rating of magnetic resonance imaging (MRI) and fluorodeoxyglucose positron emission tomography (FDG-PET) images; neuropsychological, occupational therapy, and speech therapy, assessments. 28
Participant inclusion criteria for this study comprised presenting to any Eastern Health care service with a cognitive complaint or potential neurodegenerative disorder with symptom onset at less than 65 years of age. Most participants were referred to the Eastern Cognitive Disorders Clinic, a tertiary referral service for people with atypical dementia.
Exclusion criteria included patients with no cognitive complaints or a life expectancy of less than 12 months. Eight participants (7 EOAD, 1 frontotemporal dementia) had Aβ status confirmed by Aβ-PET or CSF Aβ and p-tau181 gold standard biomarkers. Following a consensus meeting of three neurologists, based on the consultation report and multiple follow up letters per patient, study participants were categorized into one of three groups based on their individual diagnostic assessments: non-neurodegenerative diseases (non-ND, n = 52), early-onset Alzheimer's disease (EOAD, n = 39), or non-AD neurodegenerative diseases and other dementia (other-ND, n = 27) (Supplemental Figure 1, Supplemental Table 1). Clinical diagnoses assigned by clinicians were based on established diagnostic criteria: NIA-AA criteria for probable AD,29,30 Rascovsky criteria for behavioral variant frontotemporal dementia, 31 Gorno-Tempini criteria for primary progressive aphasia variants, 32 Movement Disorder Society Criteria for Progressive Supranuclear Palsy, 33 Limbic-predominant age-related TDP-43 encephalopathy criteria, 34 McKeith criteria for dementia with Lewy bodies, 35 and DSM-5 criteria for non-neurodegenerative psychiatric or functional diagnoses. 36 Clinical diagnosis served as the ground truth for evaluating plasma p-tau217. CSF or Aβ-PET results were not available to inform or revise the clinical diagnosis for most participants except for a subset of participants (7/39 in the EOAD group). Clinical decision-making was informed by CSF or Aβ-PET biomarkers in patients for whom these data were available.
Sex assigned at birth was used, and all participants in this study were cisgender. Patient ethnicities were not recorded as part of clinical examination. For this study, ethnicity was determined based on place of birth, first language, and where available, parental place of birth. Participants were then grouped according to the Australian Standard Classification of Cultural and Ethnic Groups; Oceanian (broad group 1), European (broad groups 2 and 3), Asian (broad groups 5–7), Peoples of the Americas (broad group 8), Sub-Saharan African (broad group 9). 37 Estimated glomerular filtration rate (eGFR) and Mini-Mental State Examination (MMSE) data were included if taken within 3 years of date of research blood sample collection.
All participants provided written informed consent before participation. Participants were referred to the research study following clinic visit or as an inpatient. Blood samples were taken at a time convenient for the participant and their care team, and were not fasting, to reduce participant burden.
This study was approved by the Eastern Health Human Research Ethics Committee (Approval number E21-006-72840).
Blood collection, processing and storage and measurement of plasma p-tau217
After receiving consent, blood was collected in a K3 EDTA tube (Greiner Vacuette 9 mL, catalogue number 455036) via venepuncture and centrifuged at 3000 rpm (≈1420 g) at 4°C, for 10 min. Plasma collected from the centrifuged tube was divided into 500 µL aliquots and stored at −80°C until analysis. One aliquot was thawed to measure plasma p-tau217 utilizing the ALZpath pTau217 Advantage PLUS Assay kit (Product number 104570, Lot number 504180, Quanterix, Billerica, MA USA) on the ultra-sensitive single molecule array (Simoa) HD-X Analyzer platform (located at the BioAnalytics Australia Facility, Florey Institute of Neuroscience and Mental Health, Heidelberg, Melbourne Australia), as per the manufacturer's instructions. Calibrators were run in triplicate, quality controls (Control 1 and Control 2) and samples were run in duplicate. The average inter-assay %CV for Control 1 and Control 2 was 1.4% and 1.9%, respectively, and the average intra-assay %CV for Control 1 and Control 2 was 2.5% and 1.3%, respectively. The lower limit for quantification (LLOQ) for this assay was 0.00978 pg/mL.
Statistical analysis
Descriptive statistics, including means and standard deviations, were calculated for each group with comparisons employing Kruskal-Wallis tests for continuous variables with non-parametric distributions, general linear models (GLM) for continuous variables with parametric distributions; and Chi-square tests for categorical variables. GLMs employed to compare plasma p-tau217 between groups were also adjusted for covariates age, sex, ethnicity and eGFR (n = 98). Plasma p-tau217 was natural log transformed to better approximate normality and variance homogeneity. Following transformation, residuals from GLMs, before and after adjustment for age, sex, ethnicity, and eGFR, were normally distributed, as assessed by the Shapiro-Wilk test. Logistic regression with clinical diagnosis as the response was used to evaluate predictive models and receiver-operating characteristic (ROC) curves were constructed from the logistic scores to determine area under the curves (AUCs). AUCs and their 95% confidence interval were calculated for EOAD versus non-ND and EOAD versus other-ND using p-tau217 data available for groups of interest to characterize the biomarker's discriminative ability. AUC 95% confidence intervals were calculated using the DeLong method. The diagnostic performance of plasma p-tau217 was evaluated using pre-published cut-offs for A + and T+, and cut-offs derived from Youden's index based on clinical diagnosis. For each cut-off, sensitivity, specificity, positive predictive value (PPV), and negative predictive value (NPV) were calculated using confusion matrices. Spearman's correlations were used to assess the association of p-tau217 with age in all participants and in each diagnostic group. Mann-Whitney U tests or Kruskal-Wallis tests were used to compare plasma p-tau217 concentrations between women and men, ethnicities, and eGFR ranges within diagnostic groups, as required. All analyses and data visualization were carried out using IBM SPSS (v30), GraphPad Prism (v10.4.1) and R (v4.4.2). p < 0.05 was considered as statistically significant and all statistical tests were two-tailed.
Results
Cohort characteristics
Participant characteristics are presented in Table 1. There were more women in the EOAD group compared with the other-ND group (67% versus 30%, p = 0.005) but not compared with the non-ND group (48%, p > 0.05). There was no significant difference in age, age at symptom onset, ethnicity and eGFR categories between diagnostic groups (p > 0.05). MMSE scores were significantly lower in the EOAD group (mean ± SD: 20.94 ± 5.81) compared with the other groups (non-ND: 27.62 ± 2.85, p = 6.08e-4; other-ND: 25.12 ± 4.14, p = 0.047). MMSE scores were not significantly different between the non-ND group and other-ND group (p > 0.05).
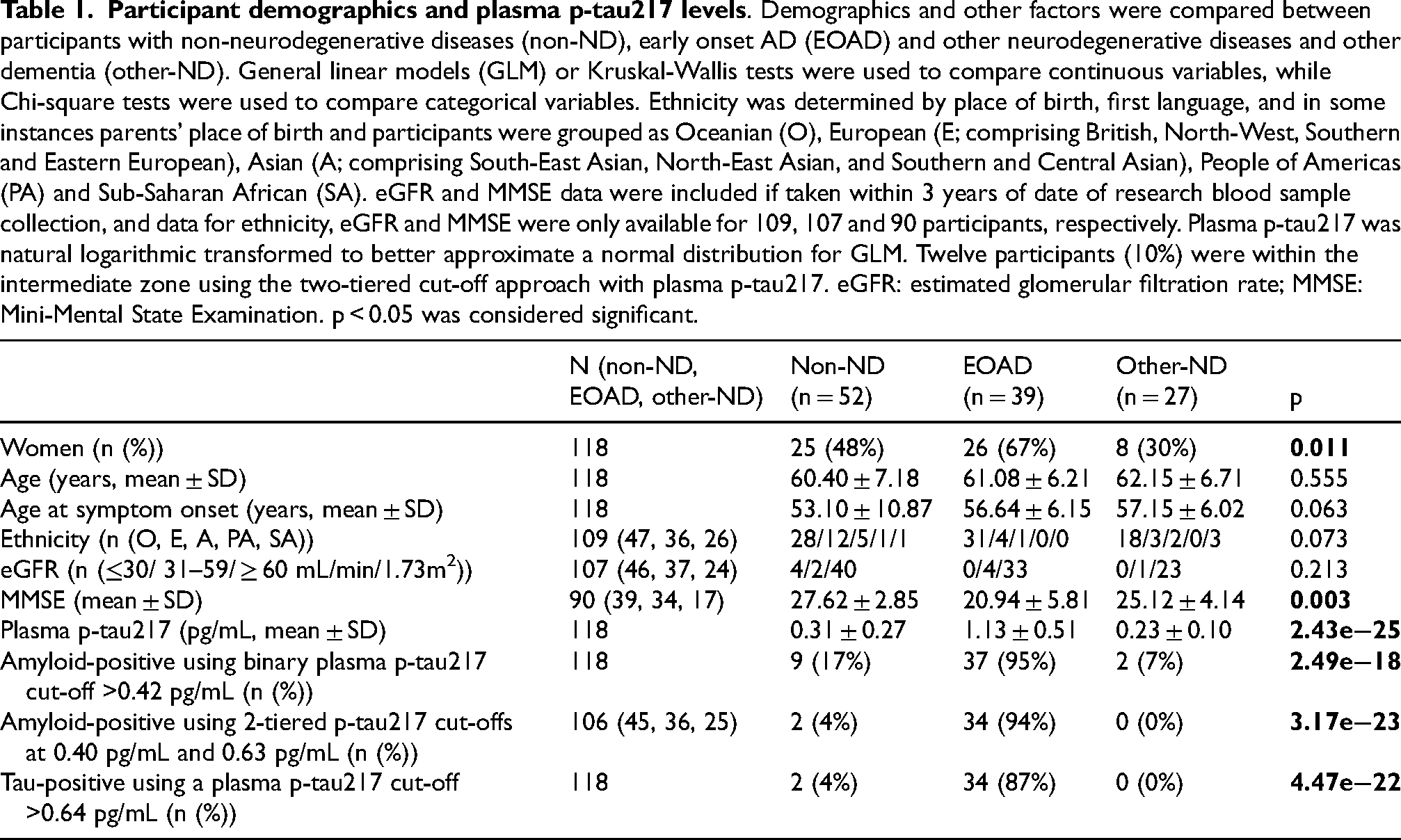
Comparison of plasma p-tau217 in EOAD with non-ND and other-ND
Plasma p-tau217 was elevated in EOAD (mean ± SD: 1.13 ± 0.51) compared with non-ND (0.31 ± 0.27, p = 5.43e-23) and other-ND (0.23 ± 0.10, p = 9.65e-21, Figure 1A). These observations remained consistent after adjusting for age, sex, ethnicity and eGFR (n = 98; 41 non-ND, 34 EOAD, 23 other-ND). Using ROC curves, plasma p-tau217 distinguished EOAD from non-ND (AUC [95% CI]: 0.944 [0.893–0.996]) and other-ND (0.984 [0.962–1.000], Figure 1B).
Plasma p-tau217 in clinically diagnosed non-neurodegenerative diseases (non-ND), early onset Alzheimer's disease (EOAD) and other neurodegenerative diseases and other dementia (other-ND). (A) Plasma p-tau217 is elevated in EOAD (N = 39), compared with participants with non-ND (N = 52) and other-ND (N = 27) using general linear models, before and after adjusting for age, sex, ethnicity and eGFR. Plasma p-tau217 values were natural logarithm transformed to ensure that the model residuals approximated normal distribution. The error bars represent 95% CI, and the line segment represents the median. The dotted lines represent a previously published plasma p-tau217 intermediate range (0.4 pg/mL – 0.63 pg/mL) for amyloid-β pathology. 10 Plasma p-tau217 concentrations of 12 individuals were within this intermediate range (non-ND = 7, AD = 3, other-ND = 2). NS represents non-significant, and p < 0.05 was considered significant. (B) represents receiver operating characteristic (ROC) curves distinguishing EOAD from non-ND and other-ND.
Application of predetermined binary and two-tiered cut-offs
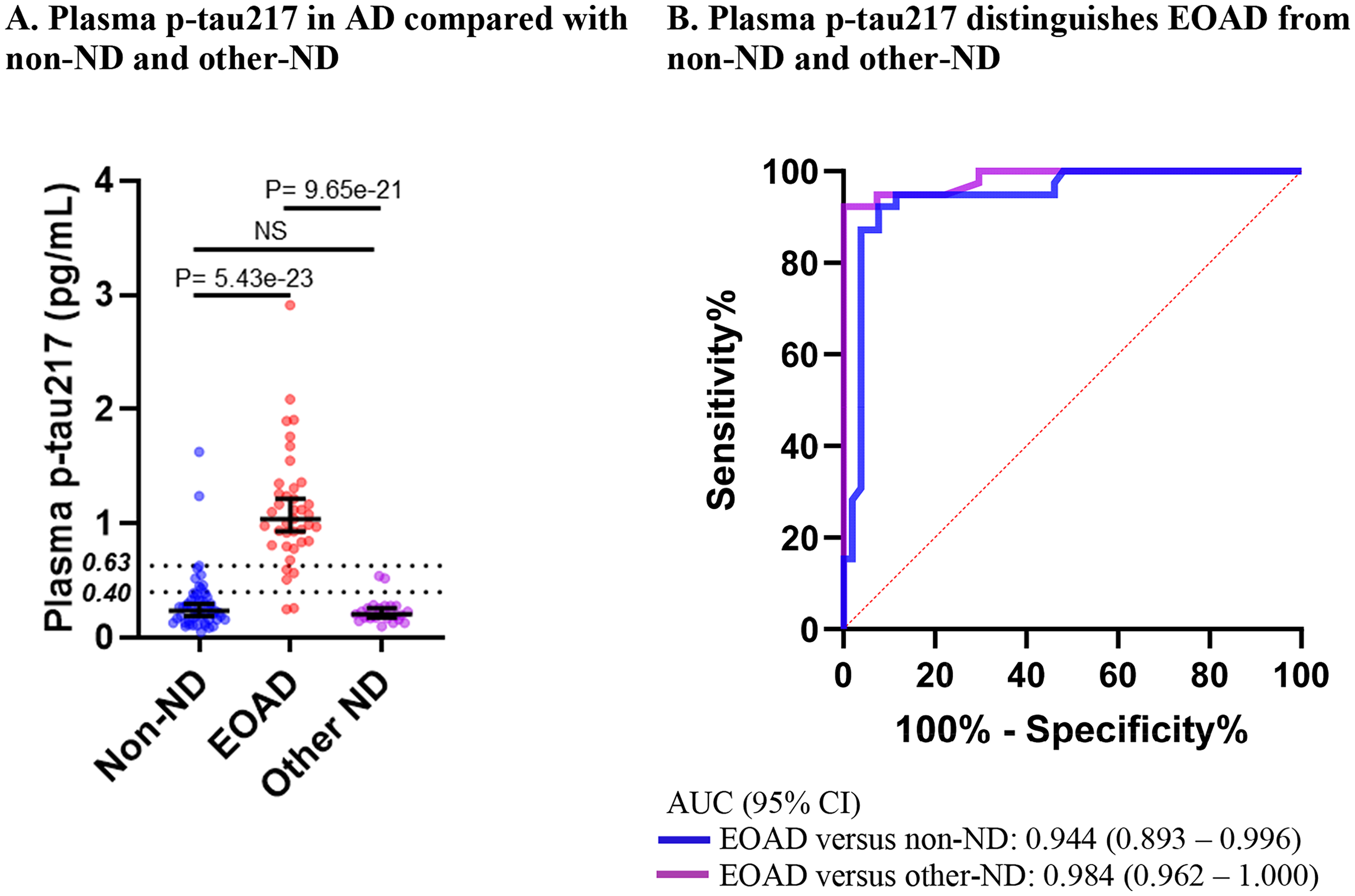
Utilizing a previously published plasma p-tau217 binary cut-off of > 0.42 pg/mL assessed from Aβ-PET positivity reflecting a centiloid value >24, 10 17% (9 of 52), 95% (37 of 39) and 7% (2 of 27) participants from the non-ND, EOAD and other-ND groups, respectively, were classified as Aβ positive (A+, Supplemental Figure 2, Supplemental Table 2).
On applying a previously published two-tiered cut-off approach for an A + status, comprising lower (<0.4 pg/mL) and upper (>0.63 pg/mL) reference points, 10 excluding cases in the intermediate zone, 4% (2 of 45), 94% (34 of 36) and 0% (0 of 25) were classified as A + in the non-ND, EOAD and other-ND groups, respectively, appearing to increase the specificity of p-tau217 for EOAD. Sensitivity, specificity, PPV and NPV for the binary cut-off and two-tiered cut-off approach for A+, along with the cut-off derived at Youden's index from the current data based on EOAD clinical diagnosis are presented in Table 2. Using the two-tiered cut-off approach resulted in 10% of study participants (n = 12) falling within the intermediate range (0.40 ≤ ptau217 ≤ 0.63 pg/mL; non-ND = 7, AD = 3, other-ND = 2, Supplemental Table 2).
Diagnostic performance metrics of plasma p-tau217 using cut-off derived at Youden's index and the previously published cut-offs in distinguishing clinically diagnosed EOAD from non-ND and other-ND.
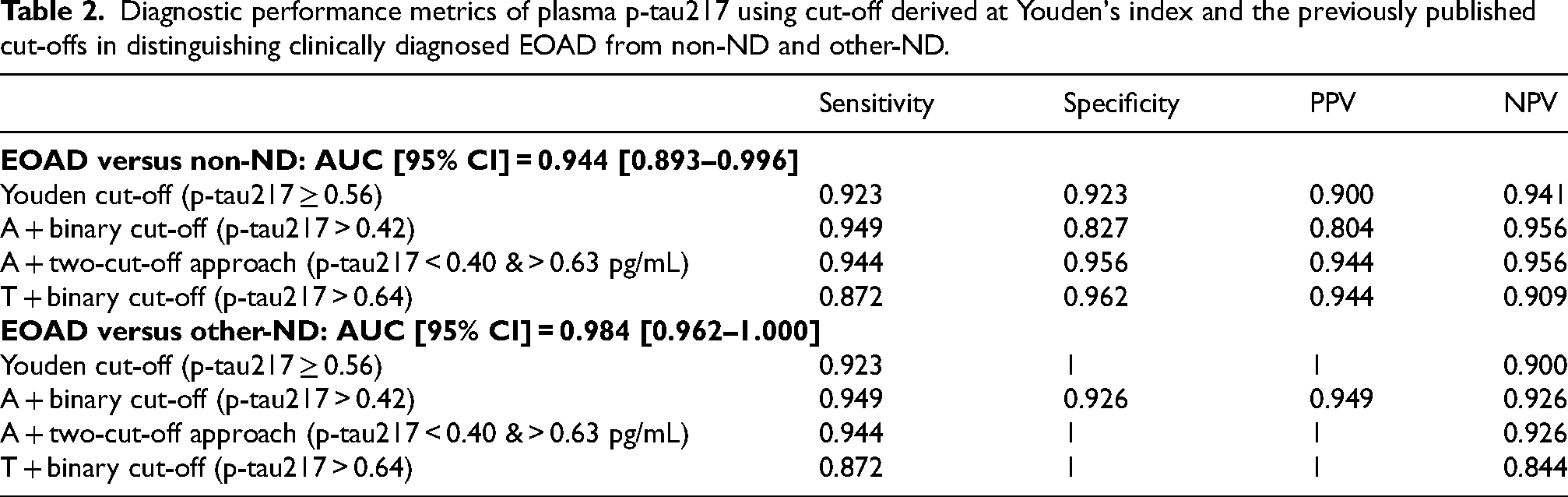
Additionally, utilizing a previously published binary cut-off reflecting AD related tau pathology (T+, plasma p-tau217 > 0.64 pg/mL), assessed using tau PET, 10 4% (2 of 52), 87% (34 of 39) and 0% (0 of 27) of non-ND, EOAD and other-ND groups were observed to be T+, respectively (Figure 2). In the A + participants, defined by the binary cut-off, 22% (2 of 9), 92% (34 of 37), and 0% (0 of 2) of non-ND, EOAD and other-ND, respectively, were observed to be T + . In the A + participants, defined by the two-tiered cut-off approach, 100% (2 of 2) of non-ND, 100% (34 of 34) of EOAD and 0 (0 of 0) other-ND, were observed to be T + (Supplemental Table 2). Sensitivity, specificity, PPV and NPV for the binary cut-off for T + are presented in Table 2.
Association of p-tau217 with age, sex, eGFR, and ethnicity
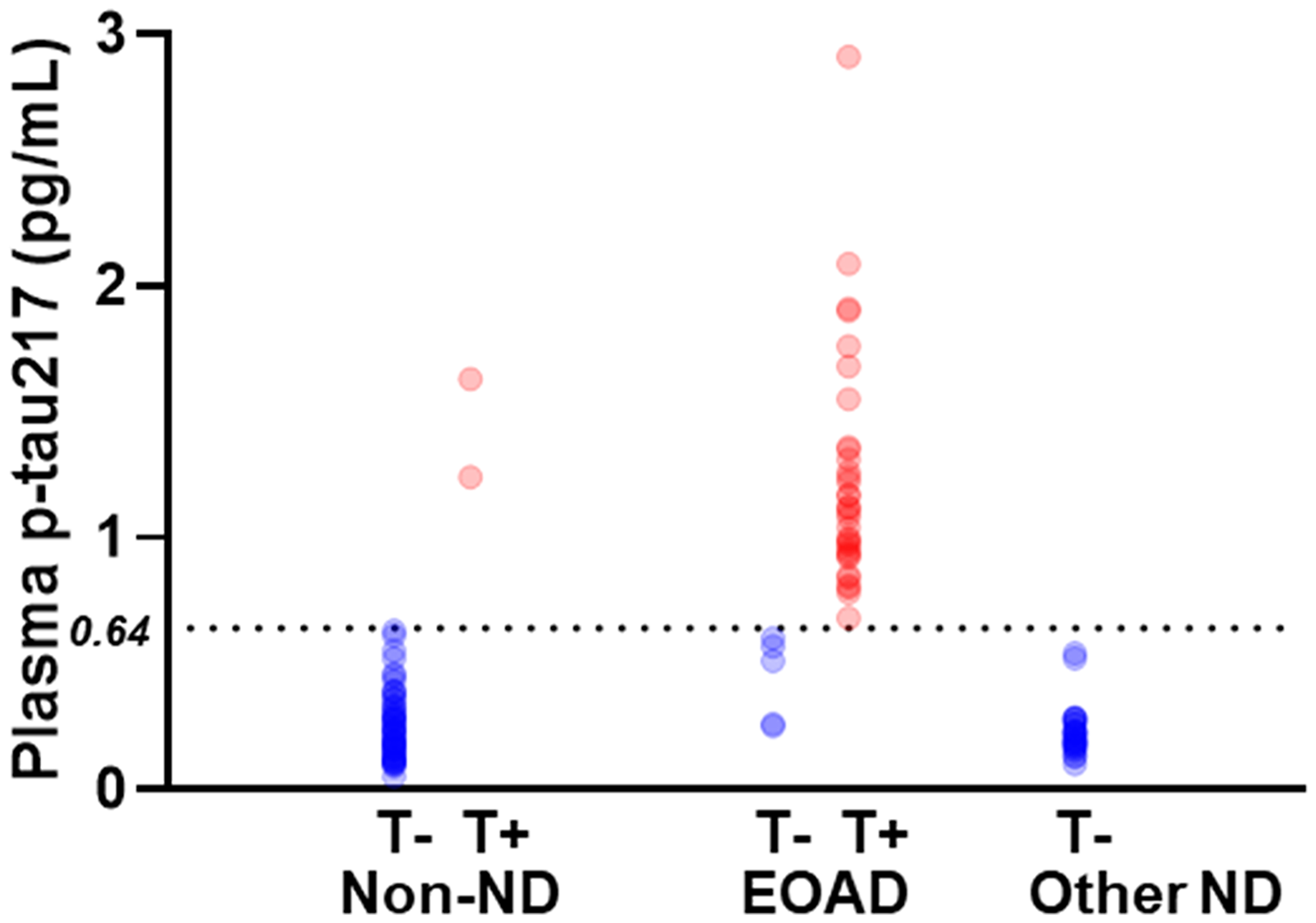
There was no significant association of plasma p-tau217 with age in all participants (R = 0.120, p = 0.197); however, after stratifying by clinical diagnosis, p-tau217 correlated with age in the non-ND group (R = 0.302, p = 0.030) but not in the EOAD group (R = 0.066, p = 0.689) or other-ND group (R = 0.215, p = 0.282). No significant difference in p-tau217 was observed between women and men or between ethnicities within each diagnostic group (p > 0.05). P-tau217 was significantly elevated in individuals with severely impaired renal function (eGFR ≤ 30) in the non-ND group (Figure 3). There were no individuals with an eGFR ≤ 30 in the EOAD and other-ND groups.
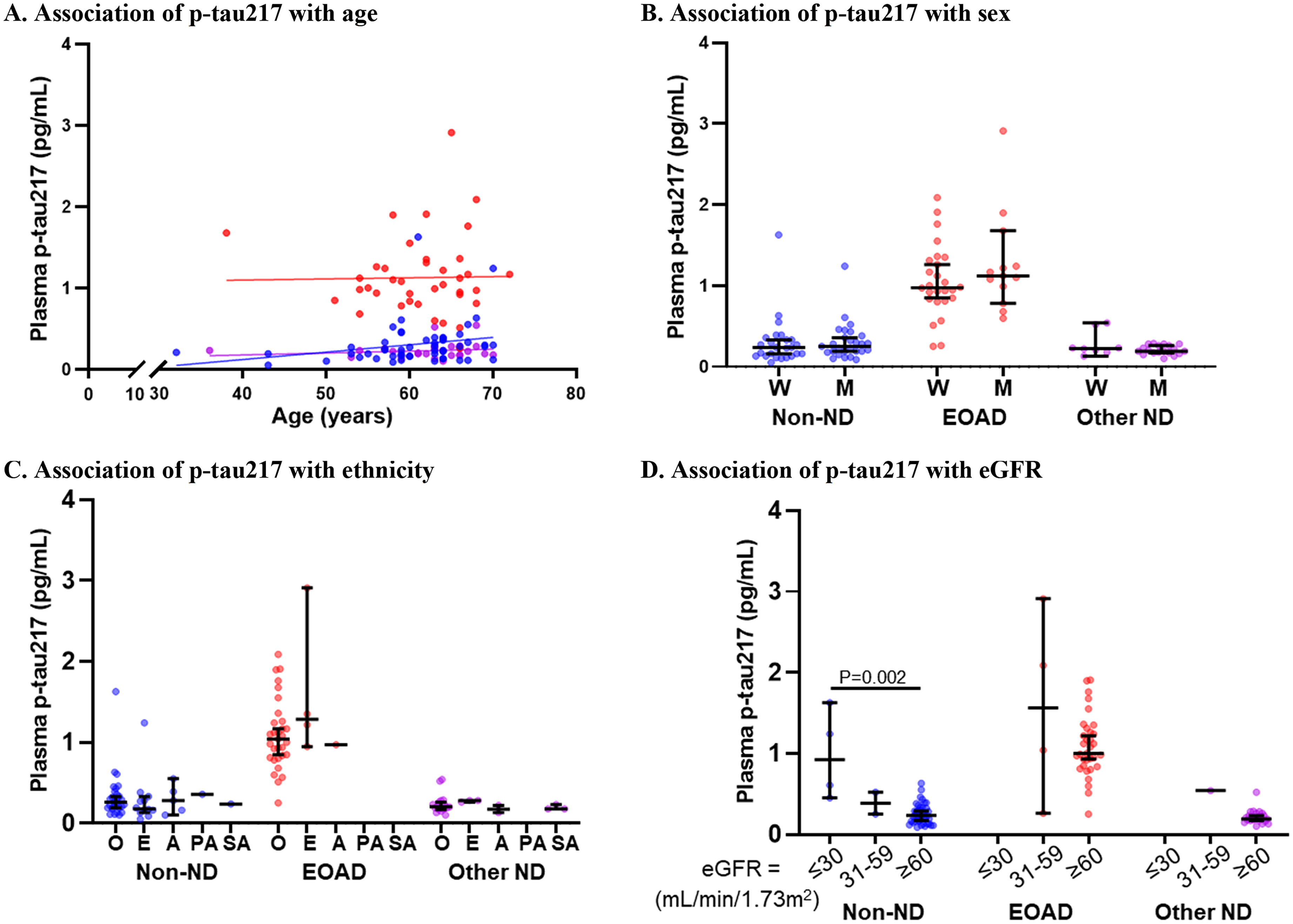
Discussion
Blood biomarkers or diagnostics are presently reshaping dementia clinical practice, including ‘real-world’ clinical studies demonstrating their utility for late onset AD.12,25,26 Here we show that plasma p-tau217 is elevated in individuals clinically diagnosed with EOAD compared to those diagnosed with non-ND and other-ND in a real-world clinical setting, further validating this biomarker. Providing clarity for EOAD diagnosis is especially important, given the inherent challenges in formulating a diagnosis for this disease with such variable presentation. We also show that plasma p-tau217 performed well in clinically diagnosed EOAD when using a previously published binary cut-off to determine A+, determining 95% (37 of 39) EOAD participants as A + .
In our study, there were just two EOAD participants that did not meet the A + cutoff. One of the two had a clinical diagnosis of AD PCA. While the majority of early-onset PCA are associated with underlying AD pathology, 38 around 10% are due to non-AD pathology. In this case, low p-tau217 levels may suggest that this participant's clinical phenotype of PCA could be attributed to non-AD pathology. The binary A + cut-off also identified 17% (9 of 52) of non-ND participants and 7% (2 of 27) of other-ND participants as A + . While it is possible that these non-ND and other-ND participants may harbor AD co-pathology, utilization of the two-tiered cut-off approach for plasma p-tau217, resulted in higher diagnostic specificity of p-tau217, such that only 4% (2 of 45) non-ND participants and 0 other-ND participants remained A + .
Both non-ND participants with high p-tau217 (reflecting A+) had renal disease: one with end stage renal disease (eGFR = 5 mL/min/1.73m2) and the other had severe loss of renal function (eGFR = 22 mL/min/1.73m2). High plasma p-tau217 levels in these participants most likely reflect reduced renal clearance of peripheral p-tau217, rather than the presence of AD pathology (A+). High plasma p-tau217 levels have been reported in individuals with eGFR < 45 mL/min/1.73m2 reflecting severe renal impairment. 39
Further, using a previously published binary cut-off to determine T +, 10 87% (34 of 39) of all EOAD participants were classified as T + . Interestingly, using the binary cut-off approach for A+, 92% (34 of 37) of all A + EOAD cases were T + . In contrast, using the two-tiered cut-off approach for A + resulted in 100% (34 of 34) of A + EOAD cases being T + . The amyloid hypothesis posits that Aβ pathology precedes and induces tau pathology in the AD continuum, and this sequence is strongly associated with symptom manifestation.40–43 Hence, we would have expected that 100% of A + EOAD participants should be T+, suggesting the two-tiered cut-off approach for A + may be more accurate. Nonetheless, the three A + EOAD participants defined by the A + binary cut-off who did not meet the T + threshold (p-tau217 > 0.64 pg/mL) had p-tau217 levels that were relatively close to this threshold (0.60 pg/mL, 0.57 pg/mL, and 0.51 pg/mL). It is also possible that these cases were clinically misdiagnosed. More specific clinical details for these three participants are available in Supplemental Table 3.
EOAD is the most common diagnosis of early onset dementia, but there are many causes, including frontotemporal dementia, dementia with Lewy bodies, vascular dementia, and progressive supranuclear palsy. Individuals with EOAD commonly do not present with the classical amnestic presentation of late onset AD. They often present with atypical forms such as PCA, logopenic aphasia or frontal/dysexecutive AD. Autosomal dominant AD caused by mutations in genes PSEN1, PSEN2 or APP is strongly associated with EOAD. Only one of our EOAD participants was a known PSEN1 mutation carrier (p-tau217 = 1.68 pg/mL).
In a tertiary care memory clinic setting, Quispialaya et al. demonstrated that plasma p-tau217 has strong discriminative accuracy for A + (versus A–) in individuals with early-onset dementia or in those aged 65 and older with behavioral or non-amnestic cognitive symptoms as the predominant clinical presentation. 44 The current study extends these findings by demonstrating the diagnostic value of p-tau217 for EOAD in a broader and more representative sample of young onset tertiary care patients, utilizing both a published binary and a two-tiered cut-off to determine A + status, further supporting its diagnostic utility in this population.
This study was performed in an Australian tertiary care setting. In this setting, consultation is covered by universal healthcare (Medicare), and diagnostic workup tools typically include brain imaging with CT or MRI and FDG-PET, and neuropsychological assessments,45,46 as these investigations are covered by Medicare. However, biomarkers that enable a biologically informed diagnosis of AD, such as CSF Aβ and tau or Aβ-PET,5,6 are not part of standard care and not covered by universal health care. These biomarkers are typically accessible only to patients enrolled in relevant clinical trials or research studies at centers where such testing is available, unless individuals cover the cost themselves, highlighting financial, geographic and sociodemographic barriers. Plasma p-tau217 has the potential to fill this critical gap by offering a scalable, accessible, and minimally invasive blood biomarker that can support earlier and more accurate diagnosis of AD in clinical settings.
A key strength of this study is its focus on a real-world patient population drawn from clinical settings. Most participants had coexisting comorbidities, reflecting the clinical complexity encountered in routine dementia care. We examined the performance of these biomarkers in early-onset dementia patients, including cases of EOAD, subgroups often underrepresented in biomarker research. Furthermore, participants were non-fasted, spanned multiple ethnic backgrounds, and had varying blood sampling times, enhancing the generalizability of the findings to diverse clinical settings. To ensure robust diagnostic classification, the initial clinical diagnosis was made by a single neurologist from a pool of over five experienced cognitive neurologists, along with a multidisciplinary team, and review of MRI and FDG-PET imaging, followed by a consensus diagnosis involving three neurologists, providing high diagnostic confidence.
Despite its strengths, this study has some limitations. Notably, Aβ and tau PET or CSF protein were not available, which limits the ability to confirm underlying AD pathology with gold-standard biomarkers. However, the use of expert clinical diagnosis, including a consensus approach among neurologists, strengthens diagnostic accuracy. Future studies incorporating Aβ and tau PET or CSF biomarkers will be important to further validate these findings. But plasma p-tau217 is already demonstrating concordance with PET at a similar or superior degree to CSF biomarkers.10,14 Additionally, although the number of participants with severe renal impairment in this study was limited, this remains a relevant clinical subgroup. Our findings offer preliminary insights into the interpretation of plasma p-tau217 levels in adults with comorbidities, an important consideration for future clinical implementation.
In summary, plasma p-tau217 measured using the ALZpath assay effectively distinguished clinically diagnosed EOAD from non-ND and other-ND in a real-world clinical setting. The implementation of a two-tiered cut-off approach enhanced specificity, allowing for more precise identification of AD cases while reducing the likelihood of false positives. This strategy also offers a cost-effective solution by reserving additional confirmatory testing for cases in the intermediate zone in a clinical setting context, thereby optimizing resource allocation. Furthermore, while PCA in EOAD is predominantly driven by AD pathology, plasma p-tau217 may aid in identifying non-EOAD early onset dementia PCA cases without underlying AD pathology, refining diagnostic accuracy in atypical presentations. Notably, in this study population, plasma p-tau217 levels were not significantly influenced by age, sex, or ethnicity in EOAD and other-ND groups. However, severe renal dysfunction in participants with non-ND did cause false positive results, highlighting the need for careful interpretation of biomarker levels in this context. Overall, these findings support the clinical utility of plasma p-tau217 in the diagnostic evaluation of EOAD, with potential implications for both standard and atypical AD presentations.
Supplemental Material
sj-docx-1-alz-10.1177_13872877251376694 - Supplemental material for Real world diagnostic performance of plasma p-tau217 for early-onset Alzheimer's disease in a cognitive disorders tertiary care setting
Supplemental material, sj-docx-1-alz-10.1177_13872877251376694 for Real world diagnostic performance of plasma p-tau217 for early-onset Alzheimer's disease in a cognitive disorders tertiary care setting by Pratishtha Chatterjee, Svetlana Ivanic, Adam Southon, Claire McCarthy, Sheila K Patel, Maja Christensen, David Darby, Ashley I Bush, Scott Ayton, Emilio Werden and Amy Brodtmann in Journal of Alzheimer's Disease
Footnotes
Acknowledgements
The authors would like to acknowledge all clinician referrers who supported the study including Dr Chris Kyndt, Dr James Bice, Dr En Ye Ong, Dr Susanna Hii, Dr Will Lee, Dr Lin Zhang and Dr Nicola Hogan. We would like to thank the dementia trials team at the Eastern Clinical Research Unit for their assistance with recruitment activities as well as the wider Cognitive Dementia and Memory Service (CDAMS) team for their participant referrals. Special thanks to Malcolm Chung, Cynthia Zupan and Georgia Papagiannopoulos for creating translation videos and to all the administration staff at Box Hill Hospital and Wantirna Health hospital for their guidance with clinic lists and overall support. The team at the Florey Institute are acknowledged for their efforts in processing blood samples. We also acknowledge Ms Harmilaap Kaur for her assistance with data curation for this study.
ORCID iDs
Ethical considerations
This study was approved by the Eastern Health Human Research Ethics Committee (Approval number E21-006-72840).
Consent to participate
All participants provided written informed consent before participation.
Consent for publication
Not applicable
Author contributions
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was funded in part by grants from the Dementia Australia Research Foundation, and the National Health and Medical Research Council Medical Research Future Fund (MRF2022896).
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
The data supporting the findings of this study are available on request from the corresponding author. The data are not publicly available due to privacy or ethical restrictions.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
