Abstract
Background
Despite treatment guidelines for Alzheimer's disease (AD), access to AD medications varies significantly within the same health care setting.
Objective
Predictors influencing access to AD medication were assessed in a retrospective cohort study comprising most individuals diagnosed with AD in Stockholm 2013–2021.
Methods
14,884 individuals were included, with an incident AD diagnosis between 2013–2021 in Stockholm, Sweden. The primary outcome was the dispensation of AD-specific drugs (cholinesterase inhibitors or memantine) within 90 days before to 180 days after AD diagnosis. We used a generalized linear mixed-effects model (GLMM). Variables included patient-centered characteristics such as age and comorbidities, and characteristics of the primary care centers (PCC) where the individuals were listed, including Care Need Index (CNI).
Results
Overall, 77% of diagnosed patients were dispensed AD medications. Individuals listed at PCCs with high CNI score, indicative of lower socio-economic resources of the area, were less likely to receive a dispensation of any AD medication compared with low CNI score (76.2% versus 84.8%; OR 0.65, 95% CI 0.50–0.86). The association between CNI score and likelihood of receiving AD medication was attenuated in subgroup analysis of individuals living in nursing homes. Also associated with lower likelihood of receiving AD medications were age ≥85 years (OR 0.25, 95% CI 0.21–0.28), nursing home residency (OR 0.37, 95% CI 0.34–0.41), and comorbidity (OR 0.63, 95% CI 0.57–0.70).
Conclusions
Socio-economic factors strongly influenced the likelihood of receiving AD medication, in addition to more established factors such as age and comorbidities. Interventions are needed to eliminate barriers to equal drug treatment in AD, particularly those related to socio-economic disadvantages.
Keywords
Introduction
Currently available treatments of Alzheimer's disease (AD), while not curative, have shown some effectiveness in managing the symptoms of Alzheimer's disease. 1 The three acetylcholinesterase inhibitors (donepezil, galantamine, rivastigmine) and one NMDA (N-methyl-D-aspartate) receptor antagonist (memantine) are currently approved. These pharmaceuticals have been included in the reimbursement system for many years in Sweden, in several other countries within the European Union (EU)2,3 and within Medicare in the US (United States). 4 Newer disease-modifying biological treatments are currently being approved and introduced to the market. 5 These drugs face considerable thresholds to broader use, such as considerable costs, uncertain risk-benefit profile and limited indication (early-stage disease). 5 Hence, traditional AD medications remain the mainstay treatment option for most individuals with AD.
Treatment guidelines vary internationally. While countries like Canada and the UK6,7 recommend AD medication use, others, such as France, have delisted these drugs from state reimbursement, 8 Swedish guidelines recommend the initiation of AD medication among individuals diagnosed with AD, 9 with a target set by the National Board of Health and Welfare (NBHW) stating that at least 75% of primary care patients should receive treatment options. 9
The Swedish healthcare system is bound by law to provide equitable care. 10 Nonetheless, significant disparities persist, which have received attention in reports issued by Swedish authorities.11,12 For example, The NBHW reports that individuals with limited educational backgrounds, as well as those with non-Nordic background, are less frequently prescribed AD medications. 13 Furthermore, a report commissioned by the Swedish Association of Local Authorities and Regions, highlights that patients with higher education levels more frequently receive newer medications and experience fewer polypharmacy complications. 11
There are regional differences across Sweden as to the percentage of individuals receiving cholinesterase inhibitors or memantine following an incident AD diagnosis. 14 Such disparities are also evident between primary care centers (PCC). For example, the percentages of individuals with AD receiving treatment with AD medications vary substantially across PCC:s in the Stockholm County. 15 This is confirmed in our data where there is a distribution between 48 percent and 100 percent (Figure 1). To ensure an equal healthcare system, it is important to identify factors promoting such differences. To our knowledge, socio-economic factors remain largely unexplored as determinators for unequal drug treatment in dementia. A systematic review from 2018 reported that RCTs on AD often fail to account for socio-economic factors, leaving a knowledge gap regarding their influence on health outcomes. 16 We aimed to investigate disparities in the dispensation of AD medications within the Stockholm County, focusing on socio-economic factors as well as individual characteristics which may explain differences in prescription patterns.
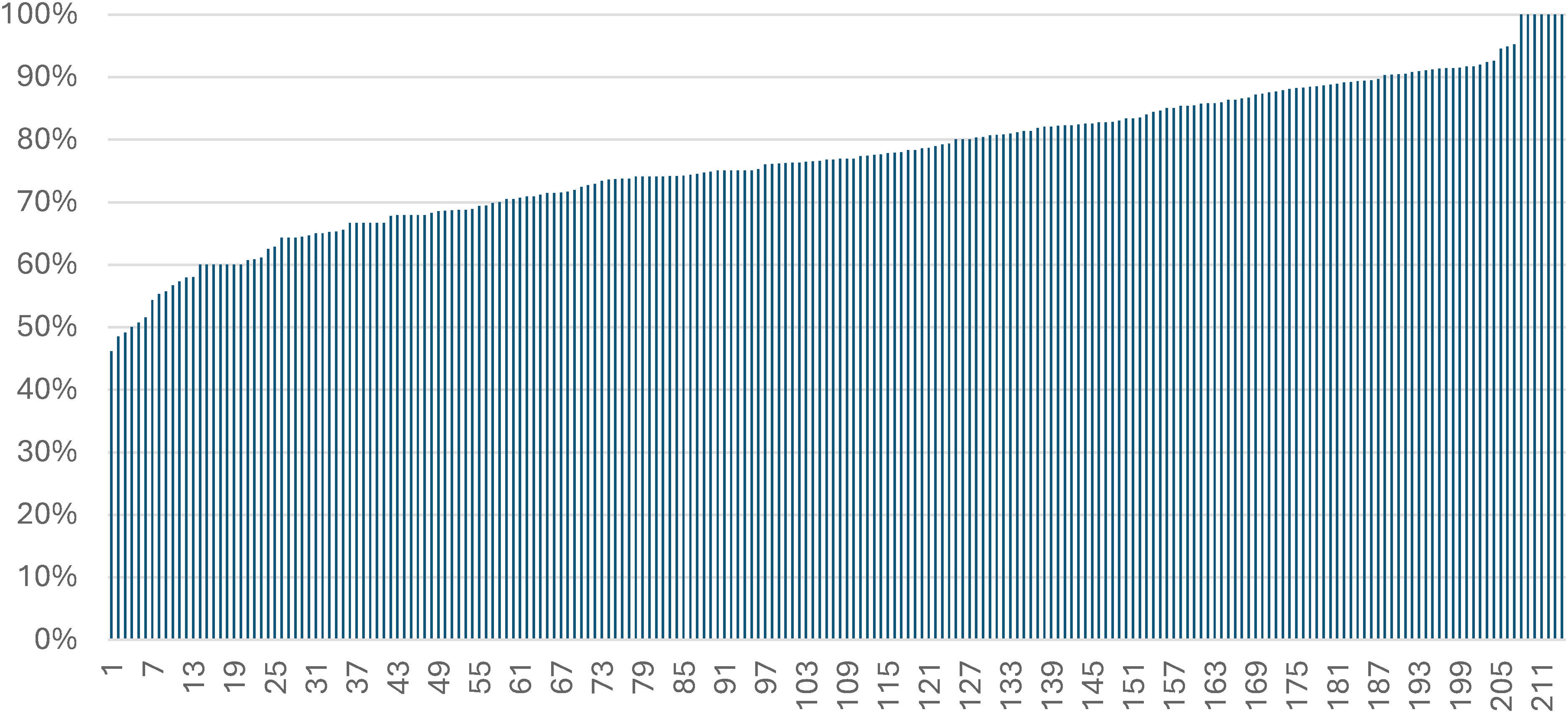
The proportion of patients in each PCC receiving AD-treatment within 180 days from the diagnosis of AD, among 214 PCCs in the Stockholm County. X-axis displays PCC number 1-214.
Methods
Study design
Retrospective registry-based cohort study on individuals diagnosed with Alzheimer's disease in the Stockholm County.
Study sample and follow-up
The dispensation of AD medications was studied among individuals who received a first incident diagnosis of AD between January 1, 2013 and May 31, 2021 at a PCC, geriatric clinic, outpatient clinic specialized in cognitive disorders, nursing homes (NH) or geropsychiatric clinic in the Stockholm County. Covariates other than AD diagnosis were obtained in the register data during follow up between January 1, 2006 and December 31, 2021. The study population included individuals with a diagnosis of other forms of dementia or with a mild cognitive impairment (MCI) diagnosis prior to first incident diagnosis with AD. The following ICD-10 codes 17 were used to identify individuals with AD (proportion of each diagnosis in our cohort): F000 Dementia in Alzheimer disease with early onset (4%), F001 Dementia in Alzheimer disease with late onset (43%), F002 Dementia in Alzheimer disease, atypical or mixed type (46%), F009 Dementia in Alzheimer disease, unspecified (7%).
Data sources
Data were obtained from Stockholm Regional Healthcare Data Warehouse (VAL), which provide pseudo-anonymized individual-level data on all healthcare consumed by citizens in the county in both primary and secondary healthcare. 18 VAL mirrors the National Prescribed Drug Register (PDR) maintained by the NBHW which contains data on all prescription drugs dispensed at any pharmacy in Sweden. 19 Hence, in our study “dispensed medication” or “received medication” or “access to medication” refers to medications that have been prescribed by a healthcare provider and subsequently dispensed by a pharmacy.
Stockholm County, the largest of Sweden's 21 counties, has a population of approximately 2.4 million, the whole population of Sweden being 10.5 million in 2024. The Swedish healthcare system is fully tax-funded, ensuring that access to healthcare services is largely independent of an individual's economic status or insurance coverage.
Definition of outcomes
We defined our primary outcome variable as the dispensation of any AD medication within a period of 90 days before until 180 days after the AD diagnosis (binary variable, received or not). The following ATC codes were used to define “AD medication”: N06DA02 (donepezil), N06DA03 (rivastigmine), N06DA04 (galantamine) and N06DX01 (memantine). This outcome was analyzed in the entire cohort, and in two sub analyses among individuals who were community-dwelling and those residing in a NH. We did not distinguish between cholinesterase inhibitors and memantine when analyzing medication dispensation. This approach was chosen to examine broader trends in AD drug dispensation rather than potential variations between drug classes.
We performed the same analyses stratified for individuals living in NH with the hypothesis that potential driving factors for unequal drug treatment would be less prominent in this setting.
As additional outcome we assessed the likelihood for the 214 PCCs where the 14884 individuals with AD were listed, to reach a treatment target (binary variable, reached or not) set by the NBHW. This target requires that a minimum of 75% of individuals with an incident AD diagnosis should receive AD medication. 20
Predictor variables and definitions
Individual characteristics included age at AD diagnosis, sex, Charlson Comorbidity Index (CCI) (12 months prior to diagnosis of AD until 6 months after), 21 total number of prescribed medications (number of ATC codes; 12 months prior to diagnosis of AD until 6 months after, “as needed” medications included), ever use of interpreter services during a healthcare consultation (ever during follow-up), community-dwelling versus NH-residency (defined as ‘nursing home’ if the individual lived in or moved to a NH within 180 days after receiving an AD diagnosis), presence of MCI diagnosis prior to AD diagnosis (ever during follow-up), year of AD diagnosis, presence of behavioral and psychological symptoms of dementia (BPSD) and Mosaic™ index. 22
A proxy for BPSD was defined as the presence of at least one of the following ICD-10 codes within 12 months before and up to 6 months after AD diagnosis: visual hallucinations (R44.1), auditory hallucinations (R44.0), unspecified hallucinations (R44.3), other hallucinations (R44.2), insomnia (G47.0), depressive episodes (F32), anxiety disorder (F41.9), conduct disorder (F91.9), delirium superimposed on dementia (F05.1), unspecified delirium (F05.9), and disorientation (R41.0). Mosaic™ social index is a commercially available database (InsightOne Nordic, Stockholm, Sweden), 22 used to classify neighborhoods into three socio-economic groups (low, medium, high) based on geographic characteristics such as education, income, and living arrangements. Details are provided in the supplement section.
PCC characteristics included socio-economic index (Care Need Index, CNI) of the PCC, PCC ownership (public or private), number of patients listed at the PCC and annual number of new AD diagnosis at the PCC. A PCC's Care Need Index (CNI) indicates the expected care burden in the group of patients listed at the PCC and is calculated by a prediction model including five socio-economic factors. 23 It is used to determine economic compensation from the authorities to the PCCs. A high CNI (>1) indicates a population with a higher need for care, while a low CNI (<1) suggests a lower need. Details are provided in the supplement section (SM section 1). CNI, PCC number of listings and PCC annual number of AD diagnoses were ascertained as a point estimate for each PCC in 2020.
Statistics
All analyses were conducted in R statistical programming software version 4.2.1 24 with added libraries gtsummary, 25 lme4, 26 ggplot2, 27 DHARMa 28 performance 29 and finalfit. 30 To assess which variables predicted the outcome (dispensation of AD drug) we fitted a Generalized Linear Mixed-effects Model (GLMM), using the command ‘glmer’ from the R package ‘lme4’. This allowed us to estimate the relative effect of both the variables for which we had measurements per individual, and the variables pertaining to individual PCCs which had the same value for many patients. The GLMM included fixed effects of age, sex, CCI (categories), number of prescribed medications (categories), BPSD (y/n), Diagnosis of MCI prior to AD diagnosis (y/n), year of AD diagnosis (categories), ever use of interpreter (y/n) residing in nursing home (y/n), mosaic socio-economic index (categories), PCC Care need index (categories), PCC management (public/private), PCC number of listed patient (categories), PCC Number of AD diagnoses per year (categories). A random intercept effect of PCC was added to take into account the hierarchical structure of the data (individuals clustered within PCCs). Two sub analyses were performed using two GLMMs equivalent to the main analysis, with the same variables, but each using one subset of the main dataset: i) only individuals residing in a nursing home and ii) only community-dwelling individuals.
For the sensitivity analysis we performed a standard logistic regression model at the level of the PCC with the outcome PCC target reached (yes/ no; yes if at least 75% of individuals with incident AD receiving AD treatment). As predictors the model included the same 4 PCC-related variables also present in the main GLMM, i.e., PCC management (public/private), PCC number of listed patients, PCC Number of AD diagnoses per year, PCC CNI. adjusted for characteristics pertaining to the individuals diagnosed with AD at the PCC.
All models adhered to model assumptions (i.e., normality, homoscedasticity and influential outliers) using residual diagnostics 28 and multicollinearity. 29
Results
From an initial population of 16,225 individuals with AD our final cohort comprised 14,884 individuals after applying exclusion criteria (Figure 2). These individuals were distributed across 214 PCCs, out of a total of 245 assessed PCCs throughout the Stockholm County.
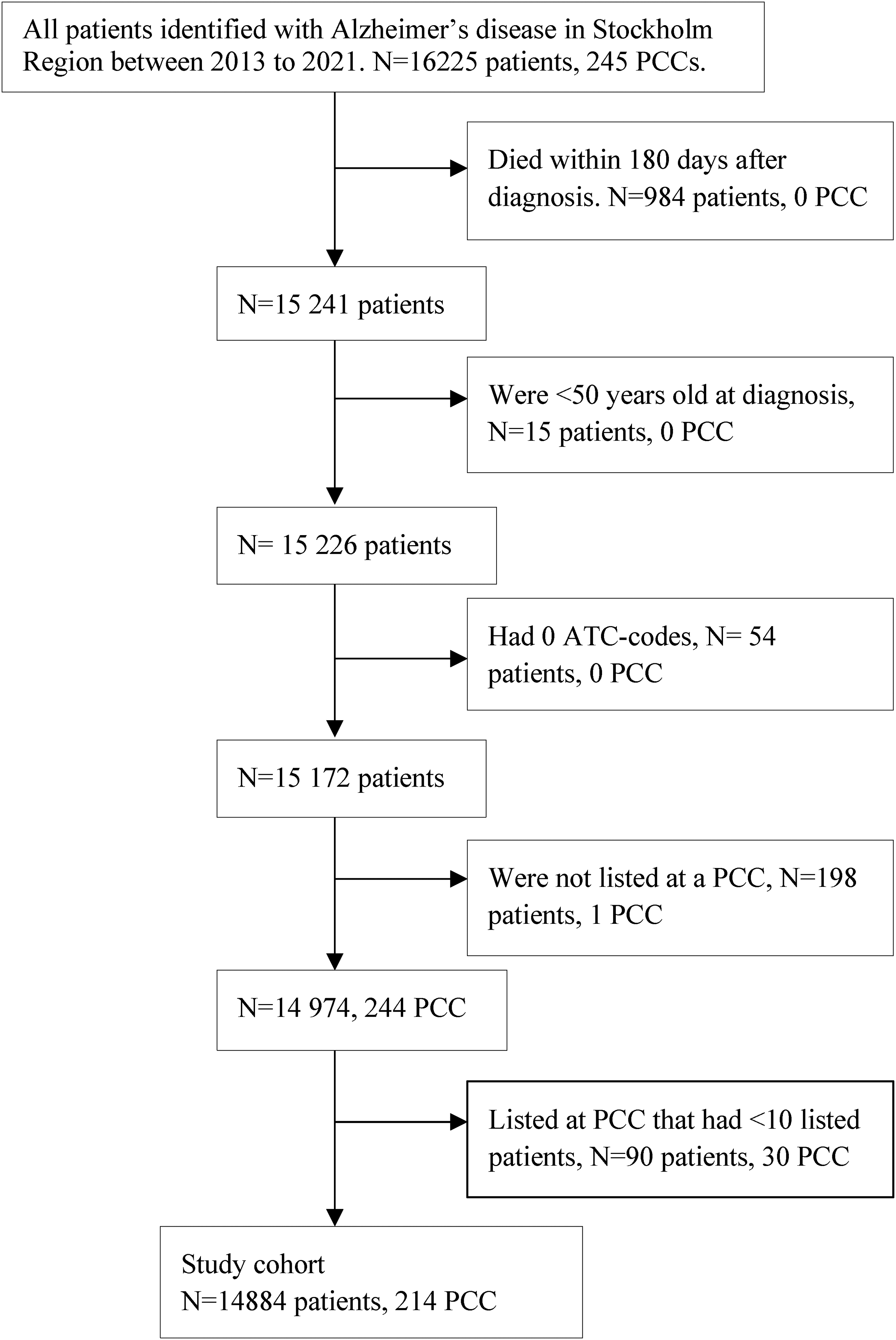
Flow diagram showing selection criteria for the study.
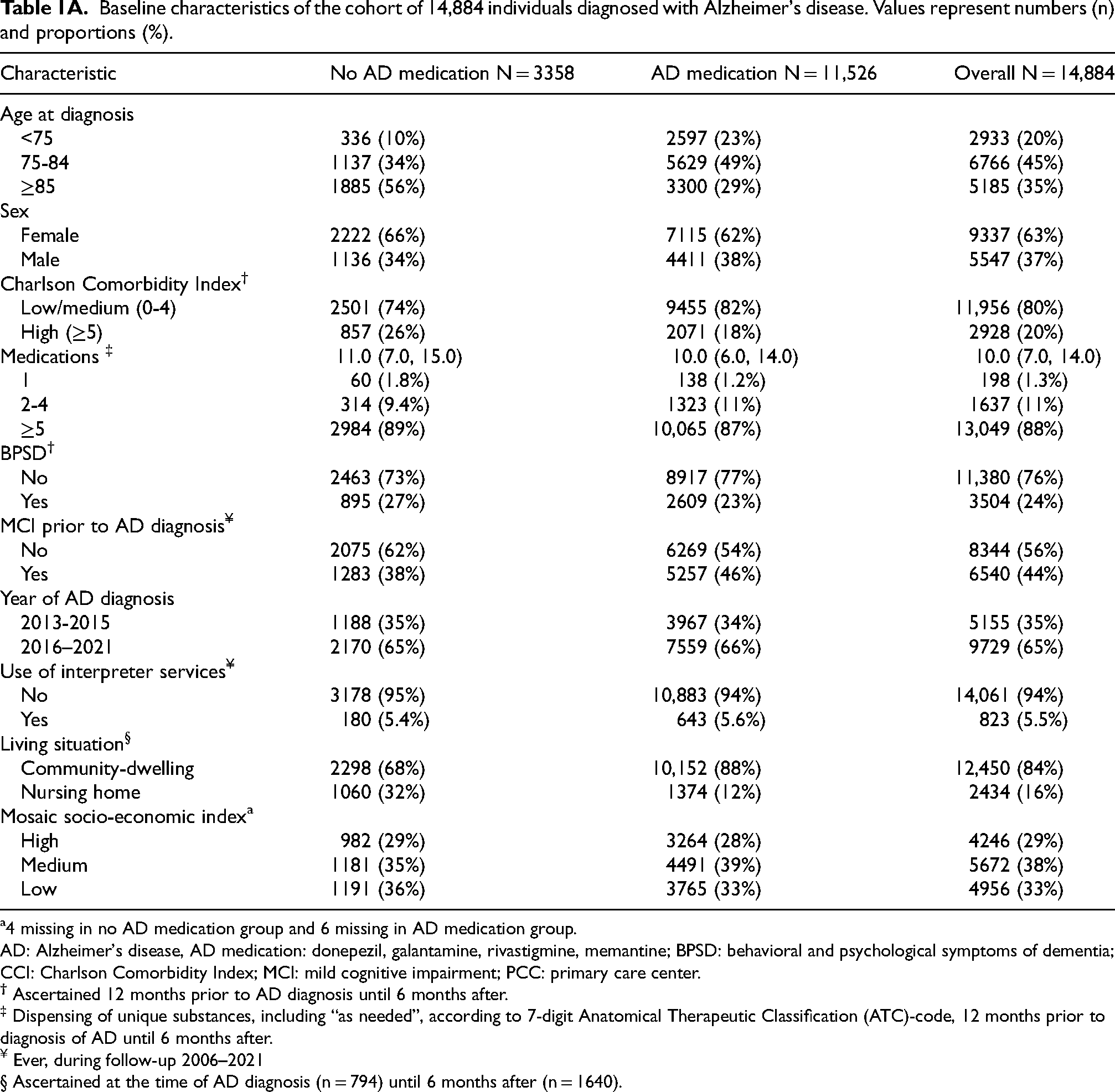
77% of individuals (11,526 / 14,884) received AD medication during follow-up (Table 1A). Sixty three percent were women. The mean age was 82 years and 35% of the cohort was 85 years or older. 80% had a low to medium comorbidity burden as measured by the CCI and the population showed substantial medication use, with a median of 10 prescribed medications per individual (Table 1A).
Baseline characteristics of the cohort of 14,884 individuals diagnosed with Alzheimer's disease. Values represent numbers (n) and proportions (%).
4 missing in no AD medication group and 6 missing in AD medication group.
AD: Alzheimer's disease, AD medication: donepezil, galantamine, rivastigmine, memantine; BPSD: behavioral and psychological symptoms of dementia; CCI: Charlson Comorbidity Index; MCI: mild cognitive impairment; PCC: primary care center.
‡ Dispensing of unique substances, including “as needed”, according to 7-digit Anatomical Therapeutic Classification (ATC)-code, 12 months prior to diagnosis of AD until 6 months after.
¥ Ever, during follow-up 2006–2021
§ Ascertained at the time of AD diagnosis (n = 794) until 6 months after (n = 1640).
44% had received a diagnosis of mild cognitive impairment prior to their AD diagnosis. BPSD were documented among 24%. Sixteen percent of individuals resided in NH either at the time of receiving the AD diagnosis or within 180 days thereafter (Table 1A). The majority of the PCC's had a medium CNI score and were privately owned (Table 1B).
Baseline characteristics of 214 primary care centers (PCCs) where the 14,884 individuals with Alzheimer's disease were listed. Values represent numbers (n) and proportions (%).

1 missing.
CNI: Care Need Index; PCC: primary care center.
Predictors of AD drug dispensation
Individual characteristics
Our analysis identified three major factors at the individual level that reduced the likelihood to receive AD medication (Table 2A, Figure 3A). First, older individuals were less likely to receive AD medication, in particular individuals 85 years or older (OR 0.25, 95% CI 0.21–0.28 (Table 2A). Second, NH-residency strongly reduced the likelihood of receiving AD medication (OR 0.37, 95% CI 0.34–0.41). Third, a high comorbidity burden reduced the likelihood of dispensation (OR 0.63, 95% CI 0.57–0.70) (Table 2A).

Forest plot with predictors for receiving AD-medication following an incident diagnosis with AD among 14488 individuals in the Stockholm region 2013-2021. The association between the predictors and the outcome are presented with odds ratios with corresponding 95% confidence intervals (CI).
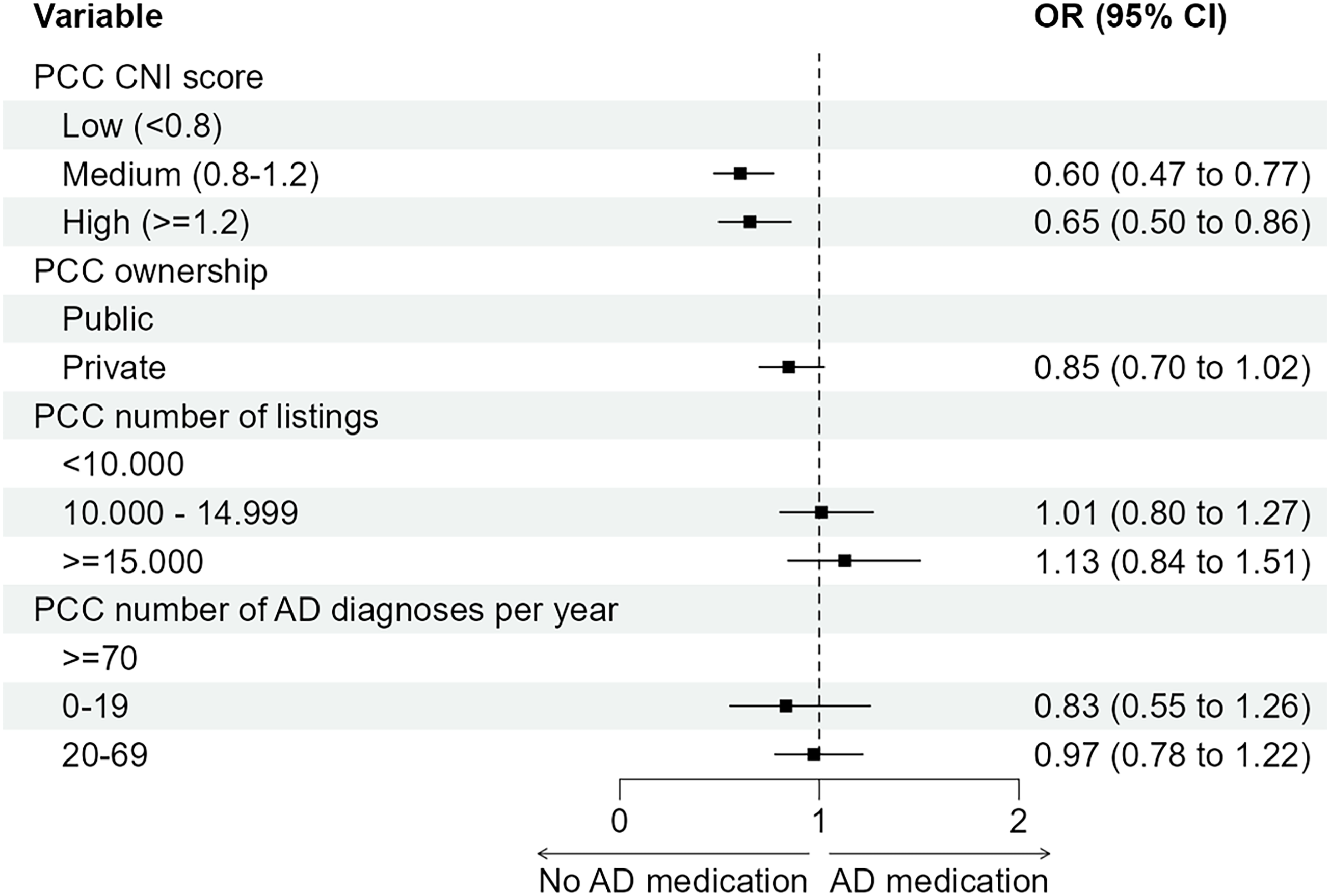
Forest plot for predictors for receiving any Alzheimer’s disease medication among 14884 individuals, following an incident diagnosis with Alzheimer’s disease. Predictors pertain to the 214 primary care center where the individual was listed. The association between the predictors and the outcome are presented with odds ratios with corresponding 95% confidence intervals (CI).
Predictors for receiving AD medication following an incident diagnosis with AD among 14,488 individuals in the Stockholm region 2013–2021. Values represent numbers (n) and proportions (%). The association between the predictors and the outcome are presented with odds ratios with corresponding 95% confidence intervals (CI).

AD: Alzheimeŕs disease; AD medication: donepezil, galantamine, rivastigmine, memantine; BPSD: behavioral and psychological symptoms of dementia; CCI: Charlson Comorbidity Index; MCI: mild cognitive impairment; PCC: primary care center.
*Additional covariates for adjustments pertaining to each individual: year of AD diagnosis (2013–2015 versus 2016–2021), Mosaic socio-economic index (high/medium/low). Variables pertaining to PCC where each individual was listed: PCC CNI score (low/medium/high), PCC ownership (public; private), PCC number of listings, PCC annual number of AD diagnoses.
‡ Dispensing of unique substances, including “as needed”, according to 7-digit Anatomical Therapeutic Classification (ATC)-code, 12 months prior to diagnosis of AD until 6 months after.
¥ Ever, during follow-up 2006–2021
§ Ascertained at the time of AD diagnosis (n = 764) until 6 months after (n = 1640).
Conversely, individuals who had been dispensed five or more medications were more likely to receive an AD medication (OR 2.31, 95% CI 1.65–3.25) as compared to those with no other prescriptions). Having a prior MCI diagnosis increased the likelihood of receiving AD medication (OR 1.26, 95% CI 1.15–1.38).
Primary care center characteristics
Individuals who were listed at PCCs with a higher Care Need Index (≥ 0.8), indicative of a stronger socio-economic disadvantage, were less likely to receive AD medications (Table 2B; for CNI 0.8–1.2 OR 0.60, 95% CI 0.47–0.77, and for CNI ≥1.2 OR 0.65, 95% CI 0.50–0.86). There was a tendency for lower likelihood to receive AD medication for individuals listed at privately owned PCCs, although this result did not reach statistical significance (OR 0.85, 95% CI 0.70–1.02) (Table 2B).
Predictors for receiving any Alzheimer's disease medication among 14,884 individuals, following an incident diagnosis with Alzheimer's disease. Predictors pertain to the 214 primary care centers where the individual was listed. Values represent numbers (n) and proportions (%). The association between the predictors and the outcome are presented with odds ratios with corresponding 95% confidence intervals (CI).
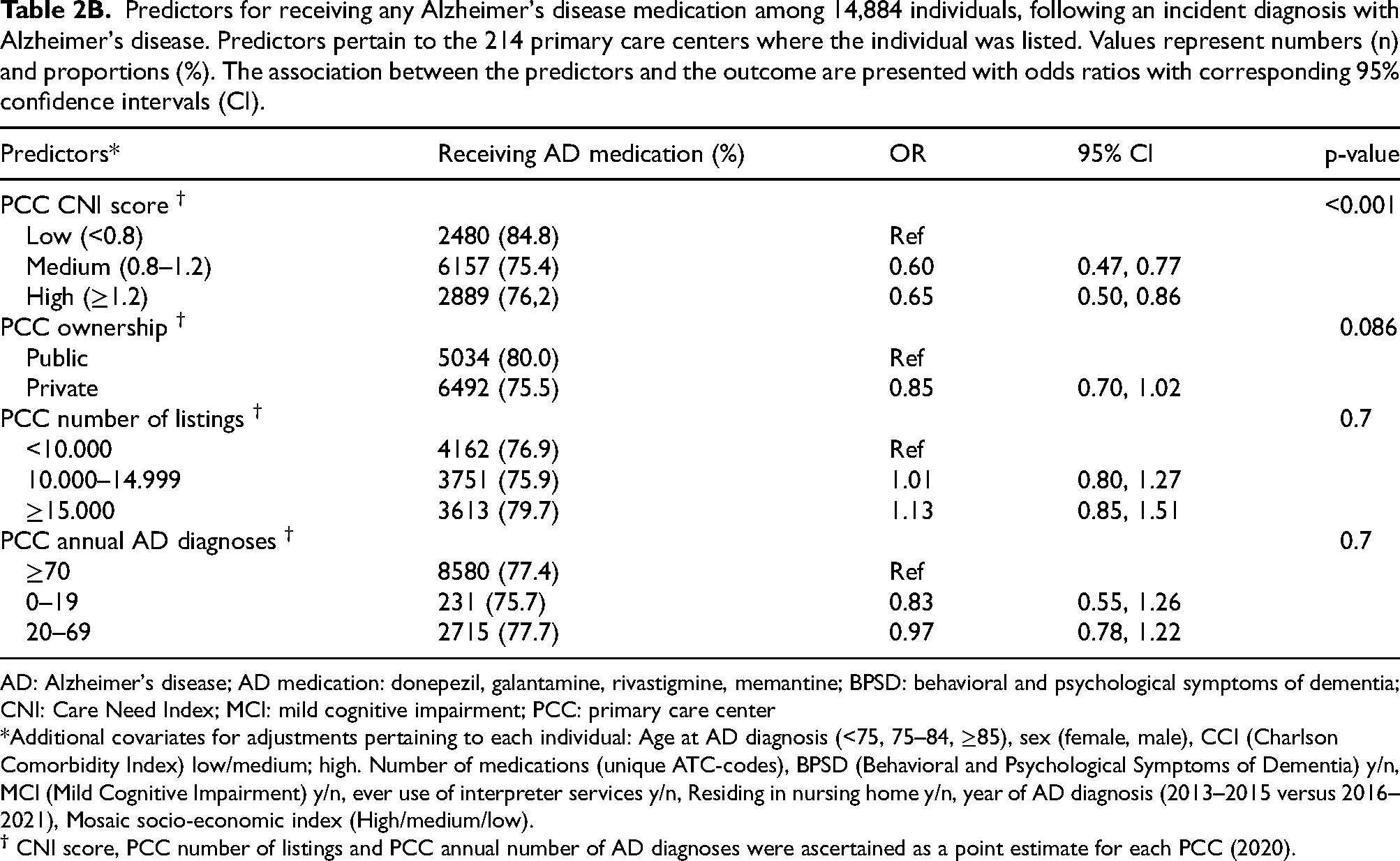
AD: Alzheimer's disease; AD medication: donepezil, galantamine, rivastigmine, memantine; BPSD: behavioral and psychological symptoms of dementia; CNI: Care Need Index; MCI: mild cognitive impairment; PCC: primary care center
*Additional covariates for adjustments pertaining to each individual: Age at AD diagnosis (<75, 75–84, ≥85), sex (female, male), CCI (Charlson Comorbidity Index) low/medium; high. Number of medications (unique ATC-codes), BPSD (Behavioral and Psychological Symptoms of Dementia) y/n, MCI (Mild Cognitive Impairment) y/n, ever use of interpreter services y/n, Residing in nursing home y/n, year of AD diagnosis (2013–2015 versus 2016–2021), Mosaic socio-economic index (High/medium/low).
Subgroup-analysis for living situation
As in the main analysis, community-dwelling individuals with greater socio-economic disadvantages (CNI score above 0.8) had lower likelihood receiving AD medication (OR 0.54, 95% CI 0.41–0.72). Among NH residents, however, greater socio-economic disadvantage was not significantly associated with lower likelihood of receiving AD medication (highest versus lowest CNI score: OR 0.80, 95% CI 0.58–1.11). Among NH residents, the occurrence of behavioral and psychological symptoms increased the likelihood of receiving AD medication (OR 1.36, 95% CI 1.14–1.63), while the opposite pattern emerged for community-dwelling individuals (OR 0.82, 95% CI 0.73–0.92) (Table 3). Ever use of interpreter service was not a predictor of receiving AD medication among NH residents but was associated with lower probability of receiving medication among community-dwelling individuals (OR 0.75, 95% CI 0.60–0.93; (Table 3).
Predictors with odds ratios for receiving AD medication following an incident diagnosis with AD among 2434 individuals residing in nursing homes and 12,450 community dwelling individuals the Stockholm region 2013–2021.
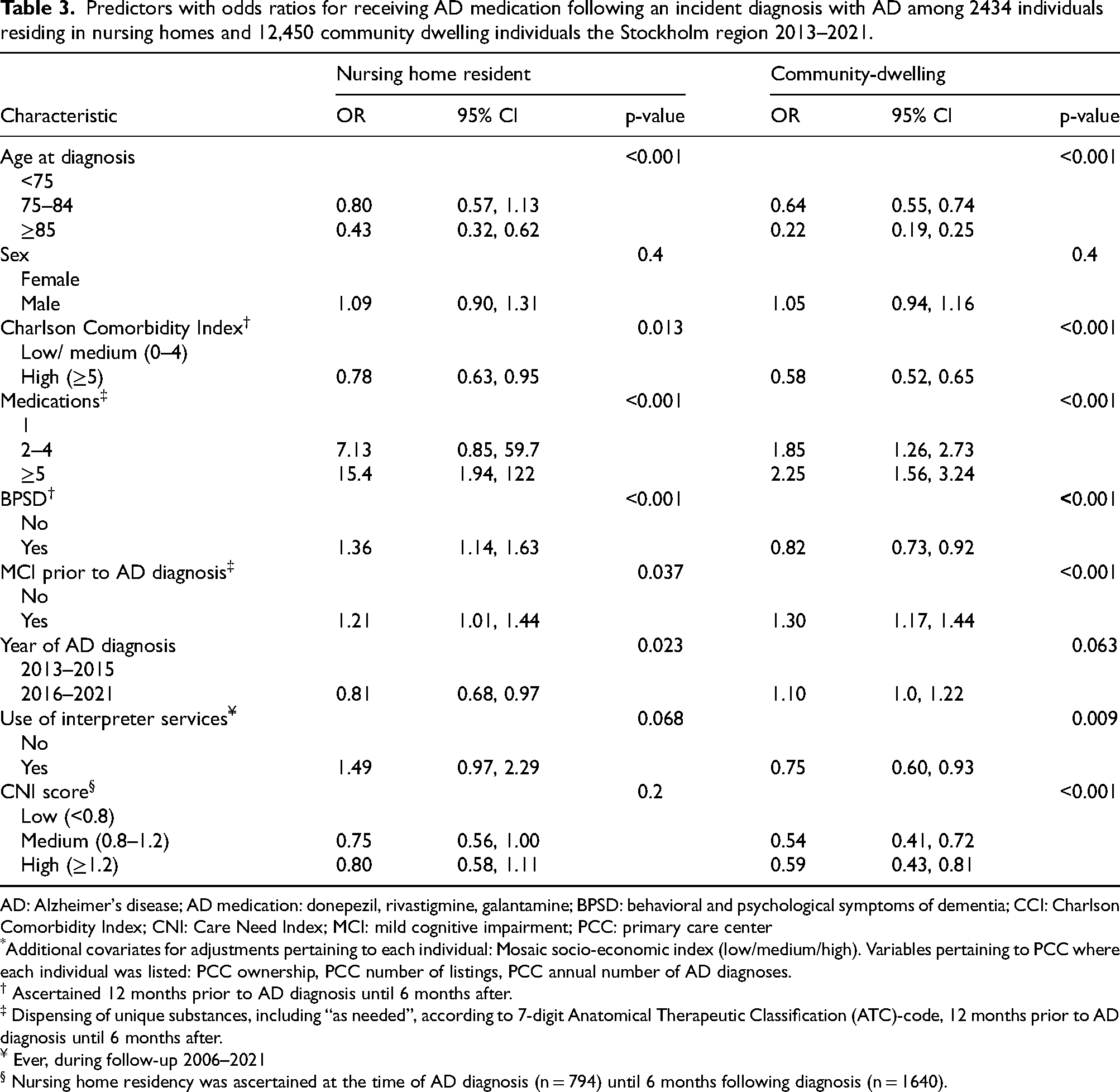
AD: Alzheimer's disease; AD medication: donepezil, rivastigmine, galantamine; BPSD: behavioral and psychological symptoms of dementia; CCI: Charlson Comorbidity Index; CNI: Care Need Index; MCI: mild cognitive impairment; PCC: primary care center
*Additional covariates for adjustments pertaining to each individual: Mosaic socio-economic index (low/medium/high). Variables pertaining to PCC where each individual was listed: PCC ownership, PCC number of listings, PCC annual number of AD diagnoses.
† Ascertained 12 months prior to AD diagnosis until 6 months after.
‡ Dispensing of unique substances, including “as needed”, according to 7-digit Anatomical Therapeutic Classification (ATC)-code, 12 months prior to AD diagnosis until 6 months after.
¥ Ever, during follow-up 2006–2021
§ Nursing home residency was ascertained at the time of AD diagnosis (n = 794) until 6 months following diagnosis (n = 1640).
Sensitivity analysis
Predictors of PCC target achievement
Among the PCC within the Stockholm County, the proportion of patients receiving AD treatment within 180 days from the diagnosis of AD varies between 48 percent and 100 percent (Figure 1). Higher CNI scores (greater socio-economic disadvantage) were associated with lower likelihood for individual PCCs to achieve the 75% treatment target (OR 0.31, 95% CI 0.12–0.69; Table 4 and Supplement Table 1). PCC ownership (public or private), the number of listed patients and the number of annual AD diagnoses were not associated with target achievement
Predictors with odds ratios for 214 PCCs to reach the target* that a minimum of 75% of listed patients with newly diagnosed AD receive AD medications. Values are number (percentages) unless otherwise stated.
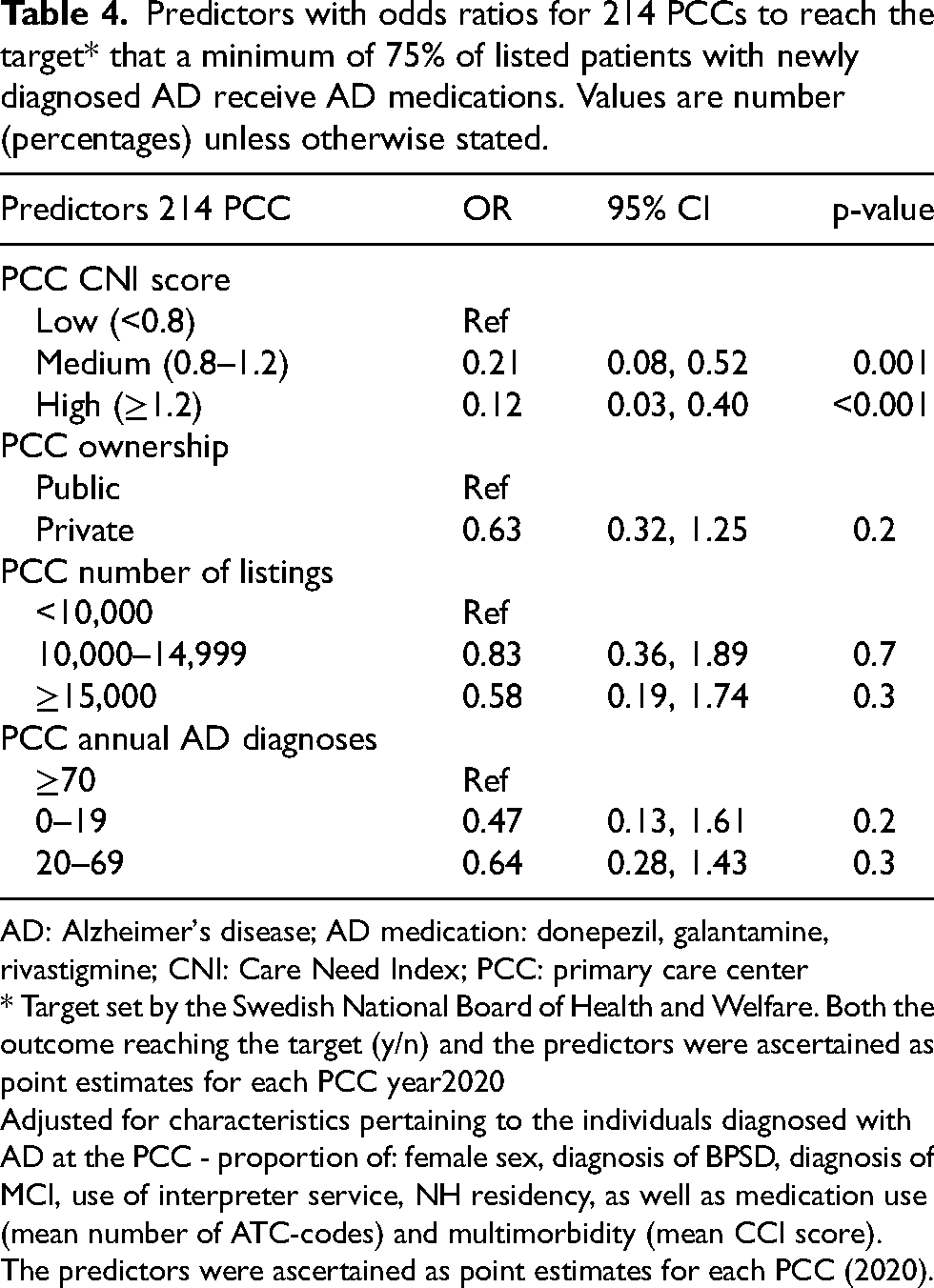
AD: Alzheimer's disease; AD medication: donepezil, galantamine, rivastigmine; CNI: Care Need Index; PCC: primary care center
* Target set by the Swedish National Board of Health and Welfare. Both the outcome reaching the target (y/n) and the predictors were ascertained as point estimates for each PCC year2020
Adjusted for characteristics pertaining to the individuals diagnosed with AD at the PCC - proportion of: female sex, diagnosis of BPSD, diagnosis of MCI, use of interpreter service, NH residency, as well as medication use (mean number of ATC-codes) and multimorbidity (mean CCI score).
The predictors were ascertained as point estimates for each PCC (2020).
Discussion
In this retrospective cohort study including 14,884 individuals with AD in Stockholm, Sweden, we found that factors associated with lower socio-economic status decreased the likelihood of receiving AD medication following an incident diagnosis with AD. A lower likelihood was also found among individuals with advanced age or multimorbidity, and among those residing in a NH. Having other prescriptions or a diagnosis of MCI prior to the AD diagnosis increased the likelihood of receiving AD medication. Our cohort was mainly constituted by late onset AD and mixed AD.
As a proxy for socio-economic status of the individual study participant we used the CNI score for the PCC where the individual was listed. The CNI score determines the level of financial support that the PCC receives from the authorities. 31 It is calculated based on indicators like income, education, employment, and living conditions of the individuals listed at the PCC and essentially aims to estimate the healthcare needs of the PCC's population. CNI has been used in other studies as a proxy for socio-economic status.32,33 In two Swedish register-based cohort studies social deprivation was associated with a higher prevalence of the diagnosis of interest. Children listed at a PCC with a high CNI score (10th percentile) were more likely to have three or more caries lesions (OR 3.55, 95% CI 3.39–3.73) 32 and heart failure was more likely to occur in the most deprived CNI percentile (10th percentile) compared with the most affluent area. 33
There are several plausible explanations to our findings. PCCs with a high healthcare need (high CNI score) are typically located in socio-economically deprived areas. PCCs in such areas may have challenges to recruit, which could impair medical competence at the PCC and lower the adherence to guidelines. Alternatively, the physician may consciously or unconsciously be biased during medical decision-making 34 and prescribe recommended treatments less often to individuals with lower socio-economic status or less often refer them to secondary care memory clinics. In the Stockholm County, the primary care physician either prescribes AD medications at the PCC following AD diagnosis or, when further work-up is needed (approximately 50 percent of cases) refers the patient to a memory clinic for assessment, diagnosis and prescription by a geriatrician specialized in dementia. 13
Our data did not distinguish between non-prescription (physiciańs decision) and non-dispensing (patient´s decision). Hence, it is plausible that the impact of a high CNI score may reflect the patient´s readiness to comply with the physician's order. A non-Swedish country of origin, low income and/or fewer years of formal education could lower the likelihood to actually withdraw the prescription at the pharmacy. Finally, medication cost (annual self-cost ceiling €260 in 2025) may be unaffordable for some socio-economic groups, affecting the likelihood of withdrawing medication. In a recent Swedish study, socio-economic factors influenced both the initiation and persistence to drug treatment. 35
We assessed use of interpreter service as a proxy for language barriers and non-Swedish background. For individuals living independently, but not for those residing in at NH, the use of interpreter services was associated with a lower likelihood of AD medication dispensation. A Swedish nationwide register-based study included 57,982 individuals with dementia showed that foreign-born individuals had lower use of cholinesterase inhibitors (p > 0.001). 36
The strong inverse relationship between advanced age and medication dispensation represents a partly expected but still possibly concerning finding. Individuals aged 85 and older showed low odds (0.25) of receiving AD medication compared to their younger counterparts, even after accounting for comorbidity burden and other factors. While increased susceptibility to adverse effects and more complex medical conditions may partially explain this pattern, the magnitude of the difference suggests potential undertreatment of this vulnerable population. Age-related treatment disparities have been similarly observed in studies examining equity in the use of cholinesterase inhibitors and other AD medications. 37
Likewise, the low odds ratio (0.37) of receiving AD medication among NH residents compared to independently living individuals with AD is noteworthy. The median age of the population residing in NHs in Sweden is 87 years 38 and a large proportion are defined as frail, which entails an accumulation of several comorbidities and medications and loss of functional capacity. 39 The reduced chance of receiving AD medication could be a signal of systematic undertreatment of AD in this setting, which calls for vigilance. On the other hand, any new medication strategy must be carefully evaluated to ensure a positive risk-benefit balance in this population where the risks of adverse events and drug interactions are significant. To that end it may be the effect of a rational adjustment of standard treatment guidelines among these individuals with a high comorbidity burden and a limited life expectancy.
The association between prior MCI diagnosis and medication dispensation could be explained by a longer time period of prevalent cognitive symptoms eventually leading up to treatment. It also raises concerns about disparities in access to early cognitive assessments, which may favor individuals with higher socio-economic status or health literacy.
We performed analyses stratified for individuals residing in NH and found that neither CNI score nor use of interpreter services were predictors for AD treatment in this setting. This was expected since potential driving factors for unequal drug treatment such as prior geographic area of living, apparel, attributes and conduct are most likely less prominent in the NH setting. The physician may therefore be more likely to standardize treatment approaches irrespective of the patients’ background. A further explanation could be the standardized dispensation process where the medication is sent to the NH from the pharmacy thereby reducing compliance/cost issues raised above.
Similar to CNI score, language barriers, indicated by interpreter use, emerged as a significant predictor of lower medication dispensation among community-dwelling individuals but not among NH residents. Effective patient-provider communication is vital in the outpatient setting, amplifying the impact of language barriers on treatment decisions. In contrast, the NH environment, where patients are observed in a more uniform manner by healthcare providers, might neutralize the impact of such barriers. In the stratified analysis we used the CNI score pertaining to the PCC where the patient was listed prior to moving to a NH. At the NHs in the Stockholm County, for the most part, the physician is affiliated to one of a few large organizations providing the medical service.
We performed a sensitivity analysis assessing the likelihood of the 214 PCCs to reach the national treatment target where at least 75% of newly diagnosed individuals with AD receive AD medication. We found the same strong association between socio-economic disadvantage (higher CNI-score) and lower rates of AD medication. This PCC-level analysis may facilitate the dissemination of our findings and stimulate appropriate measures at PCCs with a high CNI score.
A strength of the study is the use of a large, population-based cohort with individual-level data. VAL includes prospectively collected data on residency and all healthcare consumed by all citizens in the county, with low missingness and high coverage 18 which limits selection bias and misclassification. We did not have access to socio-economic factors at an individual level. However, by using the CNI score pertaining to each individual PCC we could still incorporate and adjust for the impact of socio-economic factors such as income, education level, marital status and country of origin.
There are some limitations. We lacked information on smoking and alcohol use. These factors are associated with lower socio-economic status, but except for in individuals with a history of seizures (memantine), they do not constitute an absolute contraindication to AD medication and therefore we do not consider them strong confounders. The variable interpreter service (yes/no) may partially be non-differentially misclassified since many patients are accompanied to the health care visit by family or friends who may act as translators without this being entered into the patient record. BPSD (yes/ no) may partially be misclassified in our data due to lack of formal ICD-10 code. The unexpected negative association between that variable and the outcome could be explained by such misclassification, or it may be a chance finding. Finally, the study population is from an urban, relatively homogenous and high-income catchment area, hence generalizations to other parts of Sweden, or populations outside Sweden, should be made with some caution.
Our findings suggest several important implications for clinical practice and policy. The strong influence of PCC socio-economic context on treatment patterns calls for system-level interventions to address healthcare disparities in AD treatment. These might include additional resources for PCCs serving disadvantaged populations, and closer monitoring of their adherence to recommendations. Authorities need to strengthen patient education and provide structured programs to overcome language and cultural barriers to treatment. 40 We also identify the need for targeted interventions to address age-related treatment disparities, particularly among those with more advanced age. These could include decision support tools to ensure treatment decisions reflecting individual patient factors rather than age alone. 12 The impacts of language barriers should encourage healthcare systems to increase access to interpreters and to provide culturally competent care. Such barriers seem pronounced for individuals with AD, particularly in outpatient care.
Conclusion
Our population-based study of nearly 15,000 individuals with Alzheimer's disease reveals significant inequalities in AD medication access, despite Sweden's universal healthcare system and established treatment guidelines. These patterns are consistent with previous research highlighting inequalities in dementia care. 36 Addressing these disparities represents an important opportunity to improve the quality and equity of care for individuals with AD.
Footnotes
Acknowledgements
The geriatric pharmacotherapy expert group within the Stockholm County Council Drug Committee, consisting of Alexandre Bonnard, Sofia Eriksson, Yvonne Freund Levi, Linda Furuäng, Katarina Holmgren, Maria Ljungdahl, Patrik Mattsson, Christian Molnár, Kristina Persson, Sarantos Stasinakis.
Ethical considerations
The study was conducted in accordance with the Declaration of Helsinki and was approved by the Swedish Ethics Authority (no. 2002/209-31) on March 05, 2009 and 2022-01406-02 on March 21, 2022.
Consent to participate
The need for written informed consent waived which was approved by the Swedish Ethics Authority.
Author contributions
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by The Stockholm County Council Drug Committee (Region Stockholms Läkemedelskommitté).
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
The data supporting the findings of this study are available within the article and/or its supplemental material.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
