Abstract
Background
Therapeutic plasma exchange (TPE) with albumin replacement has emerged as a potential treatment for Alzheimer's disease (AD). The AMBAR trial showed that TPE could slow cognitive and functional decline, along with changes in core and inflammatory biomarkers in cerebrospinal fluid.
Objective
To evaluate the safety and effectiveness of TPE in a real-world setting in Argentina.
Methods
From 2022 to 2024, 32 patients with mild-to-moderate AD received TPE and were compared to a historical control group (2008–2018, n = 194) matched for inclusion criteria and cognitive assessments. The protocol included six weekly intensive sessions followed by at least 10 monthly maintenance sessions. Outcomes were measured using the Mini-Mental State Examination (MMSE), and tests of memory, language, executive function, and attention. Linear models were used for analysis.
Results
Patients had a mean age of 72.1 years; 42.4% were female. Baseline MMSE scores ranged from 15 to 26. A total of 514 procedures were performed; 81.5% were uneventful. Mild-to-moderate adverse events occurred in 18.5% of sessions, mainly related to venipuncture; no severe events were reported. Mean plasma exchange volumes were 88.2% and 49.8% of estimated plasma volume during the intensive and maintenance phases, respectively. TPE significantly slowed MMSE decline (45% less than controls, p < 0.001) and reduced memory deterioration (88% less in immediate recall, p < 0.001; 74% in delayed recall, p = 0.04). Other domains were also better preserved.
Conclusions
TPE appears to be a safe and effective intervention for slowing cognitive decline in AD, supporting the AMBAR findings.
Keywords
Introduction
Alzheimer's disease (AD) is a progressive and incurable neurodegenerative disorder associated with the accumulation of amyloid plaques in the brain, formed from extracellular aggregates of amyloid-β protein (Aβ), the formation of intracellular neurofibrillary tangles of phosphorylated tau (p-tau) proteins, and chronic inflammation. 1 AD is the most common form of dementia, accounting for approximately 70% of all cases. The global number of people with AD dementia, prodromal AD, and preclinical AD were estimated at 32, 69, and 315 million, respectively. Together they constituted 416 million across the AD continuum, or 22% of all people aged 50 and above. 2
Therapeutic plasma exchange (TPE) is a well-established immunomodulatory extracorporeal technique that can rapidly reduce circulating disease mediators through removal of patient plasma. 3 TPE with albumin replacement (TPE-Alb) has emerged as a novel therapeutic approach for AD.4–8
The rationale for TPE in AD is based on its potential to remove pathological factors such as Aβ, p-tau, and other circulating molecules that may contribute to disease progression 4 TPE is associated with a reduction in plasma Aβ levels and changes in cerebrospinal fluid (CSF), suggesting a peripheral sink effect. Cognitive benefits were observed, particularly in the moderate AD subgroup, indicating a potential therapeutic role of TPE in AD. 4 TPE not only eliminates these mediators but also exerts immunomodulatory effects that can influence AD pathophysiology and long-term inflammatory changes have been described.9,10 Thus, removing Aβ from plasma through TPE may facilitate its clearance from the brain, potentially slowing disease progression.11–13 However, reducing peripheral Aβ alone may not yield significant cognitive benefits. 14 Beyond Aβ removal, TPE also eliminates inflammatory mediators implicated in AD. 15 Furthermore, albumin, used as a replacement fluid in TPE, enhances its therapeutic potential by acting as an Aβ transporter, possessing antioxidant properties, aiding toxin elimination, and modulating immune and inflammatory responses.16,17
The most significant clinical evidence supporting TPE-Alb in AD stems from the Alzheimer's Management by Albumin Replacement (AMBAR) study, a phase 2b/3 clinical trial. The AMBAR study indicates that TPE with albumin exchange may slow cognitive and functional decline in patients with AD dementia. 18
In this context, we studied a real-world cohort in Argentina, evaluating the clinical effect of TPE-Alb in AD patients outside the controlled environment of a clinical trial. Importantly, this study provides independent data from a population distinct from the AMBAR cohort, aiming to reinforce the external validity of TPE as a therapeutic approach.
Methods
Study design
We conducted a real-world cohort study between 2022 and 2024, involving 32 patients from the Outpatient Neurogeriatric Clinic and Residence, Nuestra Señora de las Nieves, Buenos Aires, Argentina. We selected patients based on the inclusion criteria described below. Once a patient was identified, we invited them and their caregiver to participate. We provided detailed explanations about the study and obtained signed informed consent. The study adhered to the ethical principles outlined in the XVIII World Medical Assembly Declaration of Helsinki and its subsequent revisions.
Patients
Inclusion criteria
We included male and female patients aged 50 to 85 years with a diagnosis of mild to moderate probable AD according to the 2011 National Institute on Aging–Alzheimer's Association. Eligible patients scored between 15 and 26 on the Mini-Mental State Examination (MMSE). They must have structural brain imaging computed tomography or magnetic resonance imaging within the previous 6 months showing a Fazekas score below 3, no cortico-subcortical infarctions ≥1 cm³, no macrohemorrhages and positron emission tomography showing evidence of cortical amyloid plaques.
Amyloid PET scans were performed at the Centro de Imágenes Moleculares, FLENI Escobar, Argentina, using a Discovery 690 PET/CT scanner (General Electric) to assess diffuse cortical amyloid deposition. Patients received an intravenous injection of 10 mCi (±10%) of [¹¹C] Pittsburgh Compound B, and image acquisition began 50 min post-injection. All images were visually assessed by board-certified nuclear medicine physicians. Although quantitative analysis using the centiloid scale was not available at the time, visual interpretation has been shown to correlate strongly with high amyloid burden.19,20
Patients on stable doses of acetylcholinesterase inhibitors and/or memantine for at least three months prior to selection were eligible and they had a caregiver available to assist during study visits.
Exclusion criteria
We excluded patients with contraindications for TPE or conditions rendering TPE unfeasible, such as difficult venous access, or abnormal coagulation parameters.
Additional exclusions included: contraindications to replacement products, including hypersensitivity to albumin or frequent adverse reactions, active or treated oncological disease, IgA deficiency, hemoglobin <10 g/dL, fibrinogen <1.5 g/L, or plasma creatinine >2 mg/dL, uncontrolled hypertension (systolic BP ≥ 160 mmHg or diastolic BP ≥ 100 mmHg despite treatment), liver disease (GPT > 2.5× upper limit of normal or bilirubin >2 mg/dL), acute coronary syndrome or cerebrovascular events within six months, New York Heart Association classification class III or IV heart failure, conditions with a life expectancy <1 year, substance or alcohol abuse.
Historical control group selection
To establish a comparison group, we retrospectively identified a subsample of 194 patients from a longitudinal clinical database maintained at our memory clinic, which includes over 1000 individuals evaluated between 2008 and 2018. From this cohort, we selected patients who met the same inclusion and exclusion criteria applied to the TPE-treated group. Specifically, controls were required to have a diagnosis of mild-to-moderate AD, similar age range, baseline cognitive status (MMSE), and follow-up data using the same neurocognitive assessment tool.21–24
Although these patients were not screened during the same period as the TPE-treated group (2022–2024), all were evaluated and followed using standardized institutional protocols for cognitive testing and clinical management. This selection strategy aimed to minimize baseline differences and ensure comparability across groups.16,18–20
All patients included in this study had signed informed consent.
Intervention
The intervention consisted of two phases.
The intensive phase
We performed six TPE procedures over a period of up to 8 weeks. Each session took place within 7 to 10 days of the previous one. During each procedure, one plasma volume (1VL, calculated as 35–45 mL/kg based on patient characteristics) was exchanged, using 5% albumin as the replacement solution.
The maintenance phase
During the maintenance phase, patients received at least 10 TPE sessions, performed approximately every 30 to 45 days. Each session involved exchanging ∼50% of the estimated plasma volume using 5% human albumin. This protocol modified the AMBAR study design by replacing low-volume exchanges with a fixed 50% volume, aiming to maintain procedural consistency in a real-world setting. Although not tailored to individual tolerance at each session, this standardized approach allowed for structured assessment of feasibility and safety in a clinically heterogeneous population. It also helped reduce variability associated with comorbidities and baseline differences, enhancing treatment uniformity across a broader patient cohort.
All procedures were performed through peripheral venous access in an external medical office, without the need for hospitalization or central venous catheterization. TPE procedures were performed using a centrifugation-based apheresis system and the albumin used was produced by the Universidad Nacional de Córdoba (Argentina).
Safety and monitoring
Before each procedure, the following clinical evaluations were performed; complete blood count: white blood cell count, platelet count, red blood cell count, hematocrit, hemoglobin concentration, mean corpuscular hemoglobin, mean corpuscular hemoglobin concentration, mean corpuscular volume, red cell distribution width. Leukocyte differential counts; neutrophils, lymphocytes, monocytes, eosinophils, basophils. Basic coagulation profile: prothrombin time, thrombin time, activated partial thromboplastin time, fibrinogen, platelet count. Ionogram: chloride, sodium, potassium, ionized calcium, magnesium. Proteinogram: total proteins, albumin, Alpha-1 globulin, Alpha-2 globulin, Gamma globulin.
Vital signs (blood pressure, heart rate, respiratory rate, and body temperature) were monitored throughout each session.
We recorded all adverse events (AEs) during and between procedures. Caregivers were encouraged to be present to reduce patient anxiety and facilitate communication.
AEs were categorized as mild, moderate, or severe according to World Health Organization MeDRA version 17.1. 25 We further classified AEs based on their likelihood of being related to the procedure as unlikely or probable. The probability of each AE was expressed as the proportion of each AE type judged to be treatment related.
Follow-up and clinical efficacy assessment
Follow-up visits were conducted between 10 and 12 (T2) months after baseline (T1) assessment. We assessed clinical efficacy at two timepoints: baseline (Time 1, T1), before starting treatment and end of visit, after the maintenance phase (Time2, T2).
Cognitive test MMSE: is assessed for dementia signs and changes over time, with scores ranging from 0 to 30. Scores classified patients as mild (20–26), moderate (10–19), or severe (<10).26,27
The Functional Assessment Questionnaire (FAQ) outcomes: commonly used in clinical settings to evaluate a person's ability to perform instrumental activities of daily living. 28
Memory test
Rey Auditory Verbal Learning Test (RAVLT) is a standardized neuropsychological tool used to assess episodic memory, learning, and susceptibility to interference. It involves tasks such as memorizing and recalling a list of 15 words. 29 We assessed five components: (a) Immediate Free Recall List A (RAVLT IFR List A), (b) Delayed Recall 20 min (RAVLT DR 20 min); (c) Recognition Memory (RAVLT RM), (d) Intrusions (RAVLT Intrusions), (e) False Positives (RAVLT FP).
Language tests
Boston Naming Test (BNT): assessed language abilities through object naming tasks. It consists of naming 60 figures presented sequentially to the patient. 30
Category Semantic Verbal Fluency (SVF): measured semantic verbal fluency under specific conditions. 31
Executive function and attention tests
Digit Span Test (DST) Forward and Backward: evaluated memory and attention by repeating number sequences in direct and reverse order. 32
Category Phonemic Verbal Fluency (PVF): measured phonological verbal fluency under specific conditions. It involves naming as many words as possible that start with a predetermined letter within one minute. 31
Trail Making Test B (TMT-B): assessed processing speed and cognitive flexibility in sequential and alternating tasks. 33
Statistical analysis
We compared demographic and baseline characteristics between the TPE group and controls using appropriate statistical tests (ANOVA for continuous variables and Fisher's exact test for proportions).
To analyze the effect of TPE treatment versus the natural history control group, we calculated the change in each outcome from baseline (T1) to post-intervention (T2) as T2-T1, and modeled these changes using linear models (LMs), with treatment group as the primary predictor. This allowed us to assess whether the observed changes differed significantly between groups. The LMs were adjusted by the explanatory variables included age, sex, education level in years, treatment period (in days), and Global Clinical Dementia Rating Scale (CDR). The CDR score measures cognitive impairment across key functional areas to track disease progression. Patients are classified based on their scores as follows: 0 (Normal), 0.5 (Very Mild Dementia), 1 (Mild Dementia), 2 (Moderate Dementia), and 3 (Severe Dementia). 34 It is important to note that the CDR may lack sensitivity to short-term changes, particularly over treatment periods shorter than one year. For this reason, the CDR score was used to describe the groups at baseline and served solely as an explanatory variable rather than a response variable.
Model selection was conducted using a stepwise approach guided by the Akaike Information Criterion (AIC), guided by the ΔAIC > 2 threshold as pre-specified in the statistical analysis plan.35,36
For the following outcomes, higher scores indicate better cognitive function, and a positive change (T2–T1) favors TPE: MMSE, RAVLT IFR List A, RAVLT DR 20 min, RAVLT RM, BNT, SVF, DST, PVF.
For the following outcomes, lower scores indicate better cognitive function, and a negative change (T2–T1) favors TPE, FAQ, RAVLT Intrusions, RAVLT FP and TMT-B.
Although our study was retrospective in nature, and it was not powered to detect a specific effect size, power calculations were conducted using the pwr package 37 to ensure a significance level of 0.05 and 90% power, considering the effect size detected in each case. All LMs were carried out using the free software R. 38
Results
A total of 32 patients diagnosed with AD were treated with TPE between 2022 and 2024.
The mean treatment period in days was 332 days (SD = 104) TPE group was compared with a control group of 194 patients who met the inclusion criteria of this study. Baseline characteristics of the control and TPE groups are shown in Table 1.
Baseline demographic and clinical characteristics of Alzheimer's disease patients by control and therapeutic plasma exchange groups.
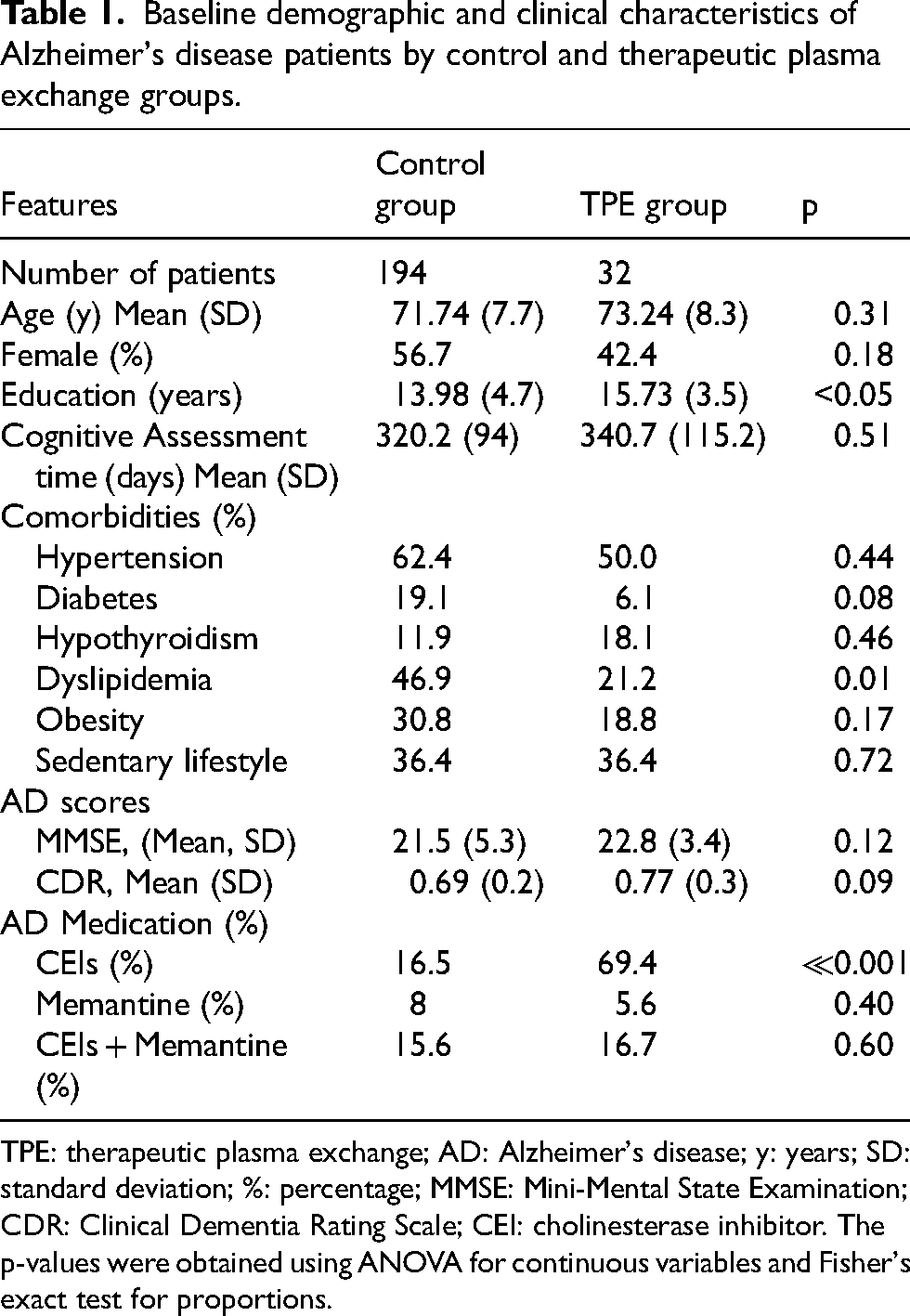
TPE: therapeutic plasma exchange; AD: Alzheimer's disease; y: years; SD: standard deviation; %: percentage; MMSE: Mini-Mental State Examination; CDR: Clinical Dementia Rating Scale; CEI: cholinesterase inhibitor. The p-values were obtained using ANOVA for continuous variables and Fisher's exact test for proportions.
Intervention
Intensive Phase: during the intensive phase, patients underwent a total of six TPE sessions. The mean volume exchanged per procedure was 2429.27 mL (SD: 725.73), representing an average of 88.4% (SD: 17.96) of the estimated total plasma volume.
Maintenance Phase: in the maintenance phase, patients received between 10 and 12 TPE sessions. The mean exchanged volume per session was 1294.40 mL (SD: 414.89), corresponding to an average of 49.8% (SD: 17.46) of the estimated total plasma volume.
Safety profile
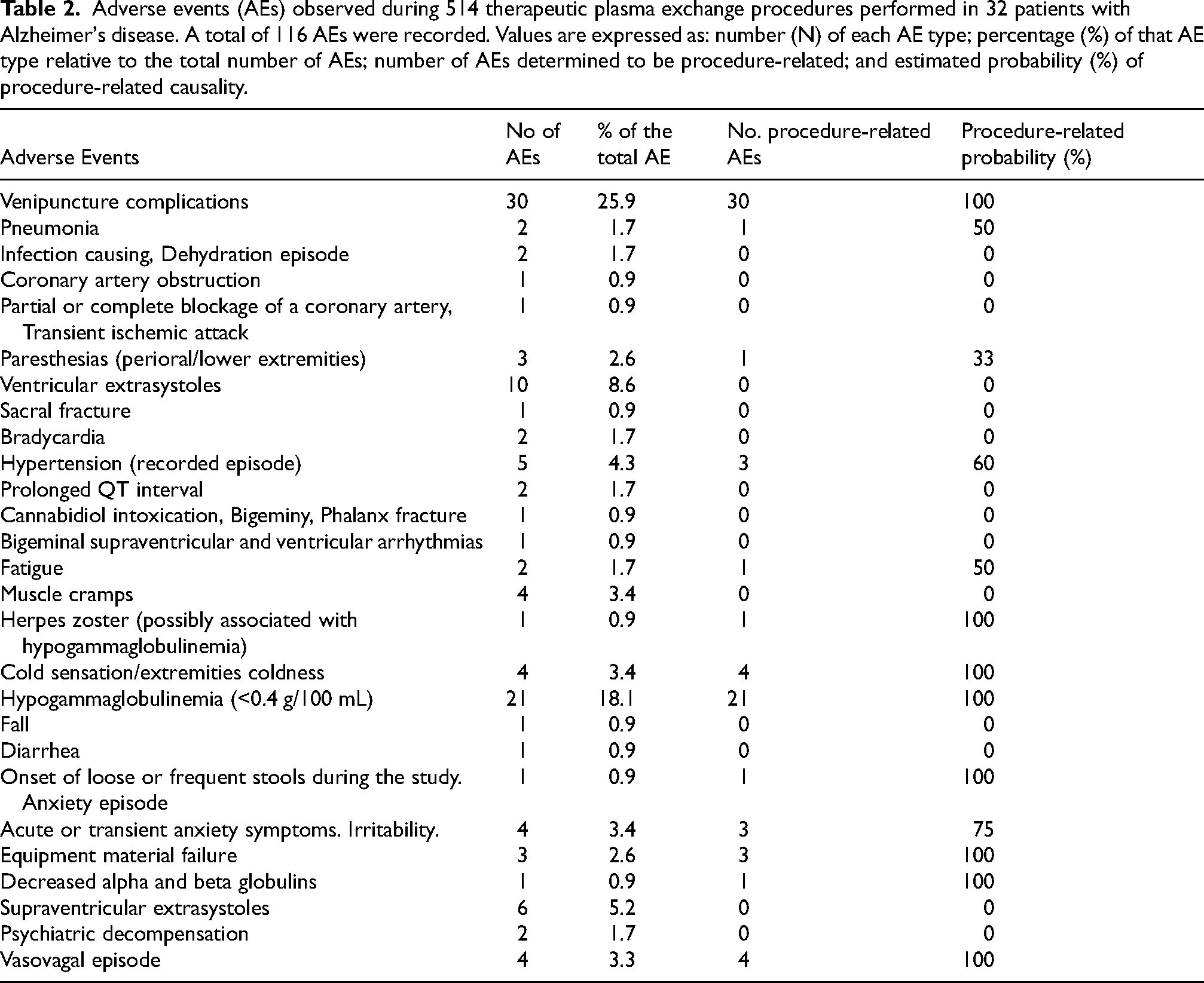
A total of 514 procedures were performed, 81.7% of which were uneventful. The remaining 18.3% (n = 94) were associated with at least one AE. Among these 94 procedures, 8 patients experienced two AEs, and two patients experienced three AEs in the same procedure, resulting in a total of 116 documented AEs. Of these 116 AEs, 88.8% (n = 103) were mild and, the remining 11.2% (n = 13) were moderate. No serious or severe AEs were registered (Figure 1). The assessments revealed that 36.2% (n = 42) of AEs were unlikely to be related to the treatment, while 63.8% (n = 74) were considered probably related to the treatment. The most frequent AE was venipuncture related (25.9%) followed by hypogammaglobulinemia (18.0%, <0.4 gr/100 ml). All documented AEs are in Table 2.
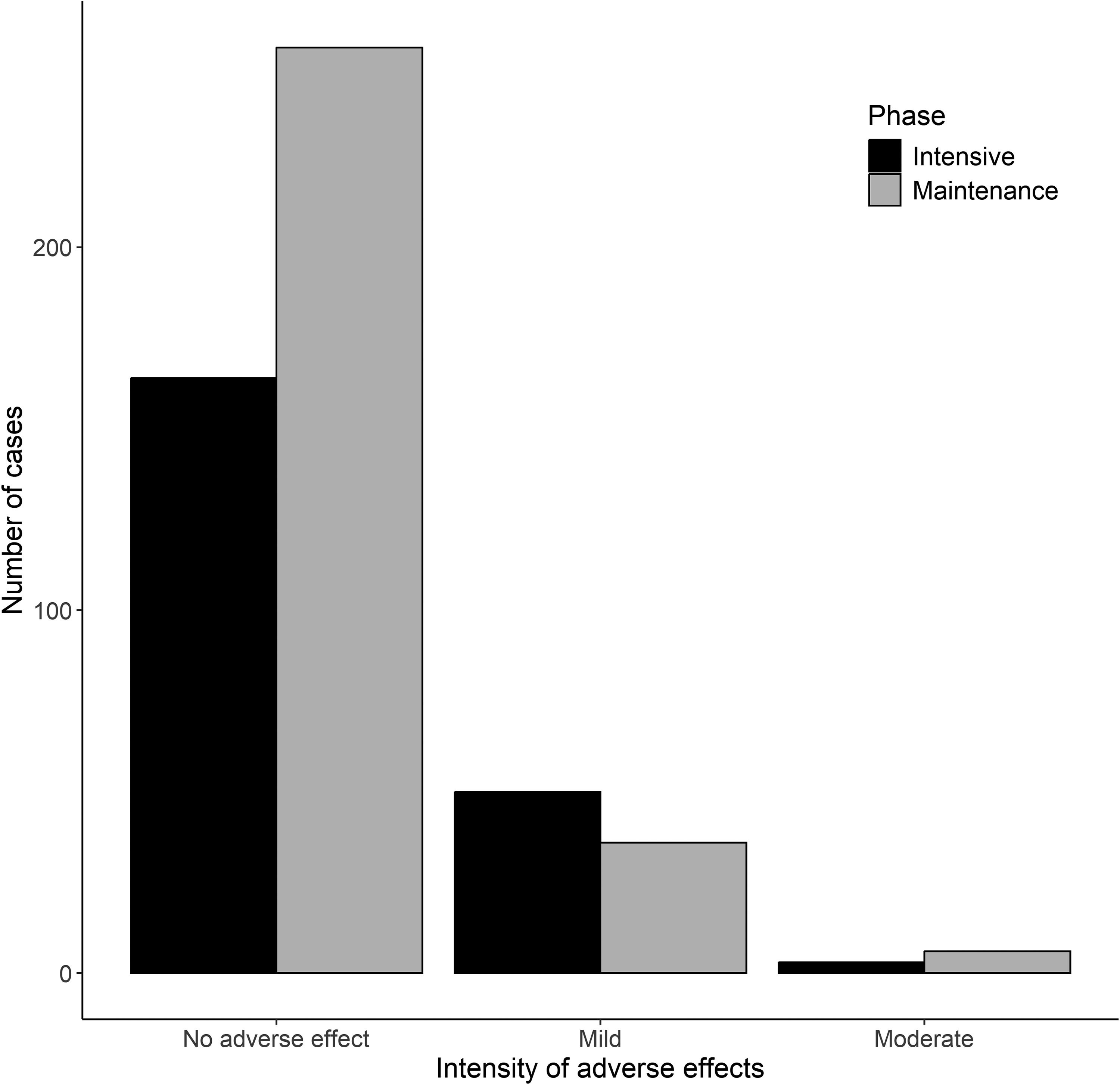
Number of adverse events observed during 514 therapeutic plasma exchange procedures conducted on 32 Alzheimer's disease patients during the intensive and maintenance phases.
Adverse events (AEs) observed during 514 therapeutic plasma exchange procedures performed in 32 patients with Alzheimer's disease. A total of 116 AEs were recorded. Values are expressed as: number (N) of each AE type; percentage (%) of that AE type relative to the total number of AEs; number of AEs determined to be procedure-related; and estimated probability (%) of procedure-related causality.
Efficacy
The deterioration in MMSE scores from baseline to the end of the treatment period was 2.2 points lower in the TPE group compared with the control group. This difference reflects a significantly reduced rate of cognitive decline in the TPE group (45% less decline, p ≪ 0.001, Figure 2).
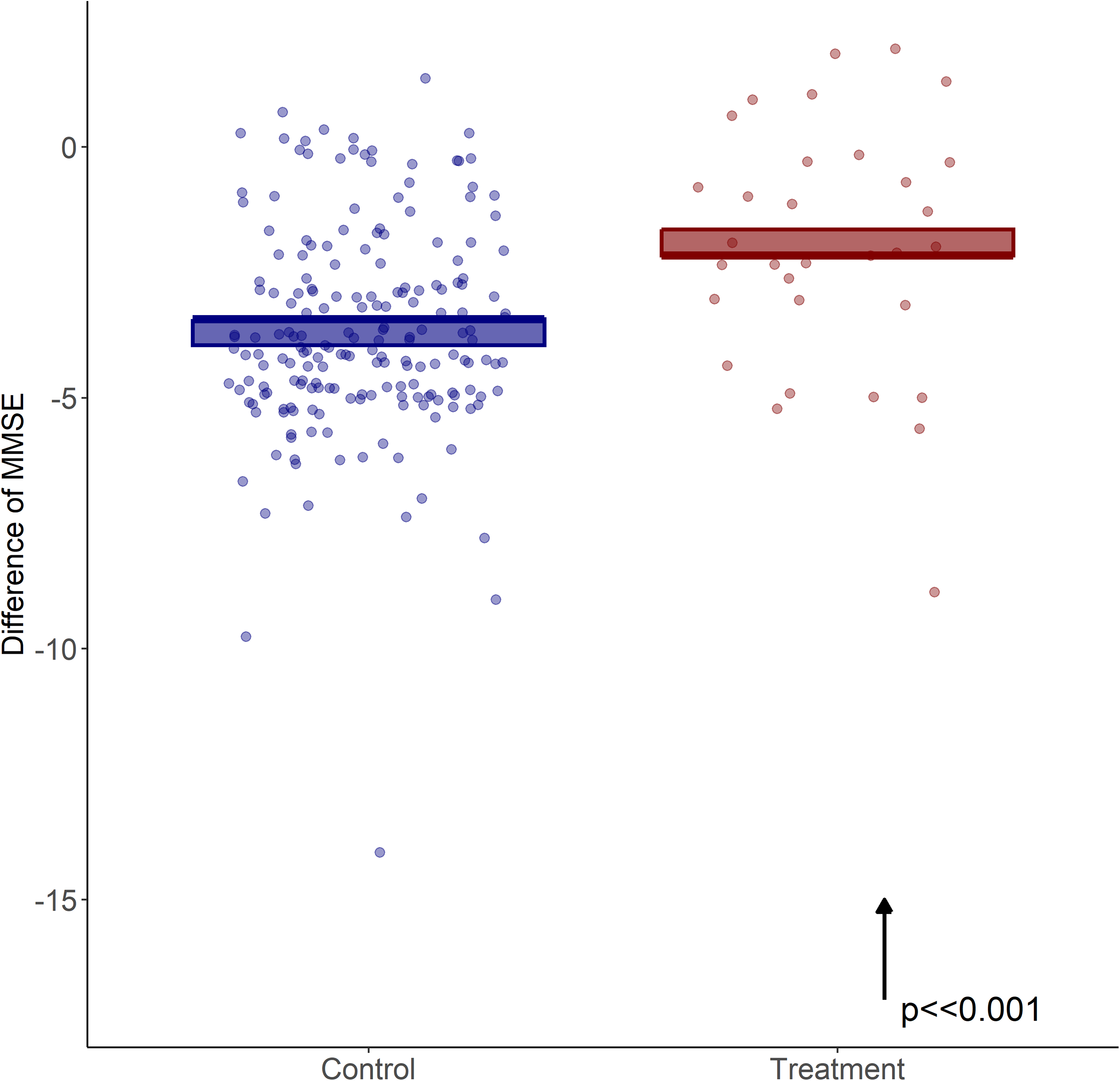
Mini-Mental state examination (MMSE) outcomes. Model-derived estimates adjusted for group and relevant covariates. Mean change (± standard error of the mean) in MMSE scores from baseline to post-intervention in patients with mild to moderate Alzheimer's Disease. Individual data points (dots) represent each patient. The Treatment group received therapeutic plasma exchange (TPE), while the Control group represents a historical cohort. Arrow reflects the direction of clinical interpretation: an upward arrow (↑) denotes less cognitive decline. On average, the TPE group showed a 45% reduction in cognitive decline compared to the historical control group.
The FAQ result showed a slightly less numerical decline in the functional abilities in the TPE group, and was not statistically significant (p = 0.51)
Memory test outcomes
All five components of the RAVLT showed significantly less decline in the TPE group compared to control (Figure 3). The RAVLT IFR List A showed 88% less decline (p ≪ 0.001). The RAVLT DR 20 min showed 74% less decline (p = 0.04). The RAVLT RM showed 69% less decline (p ≪ 0.001). The RAVLT Intrusions showed 74% less decline (p = 0.01). The RAVLT FP showed 59.5% less decline (p = 0.006).
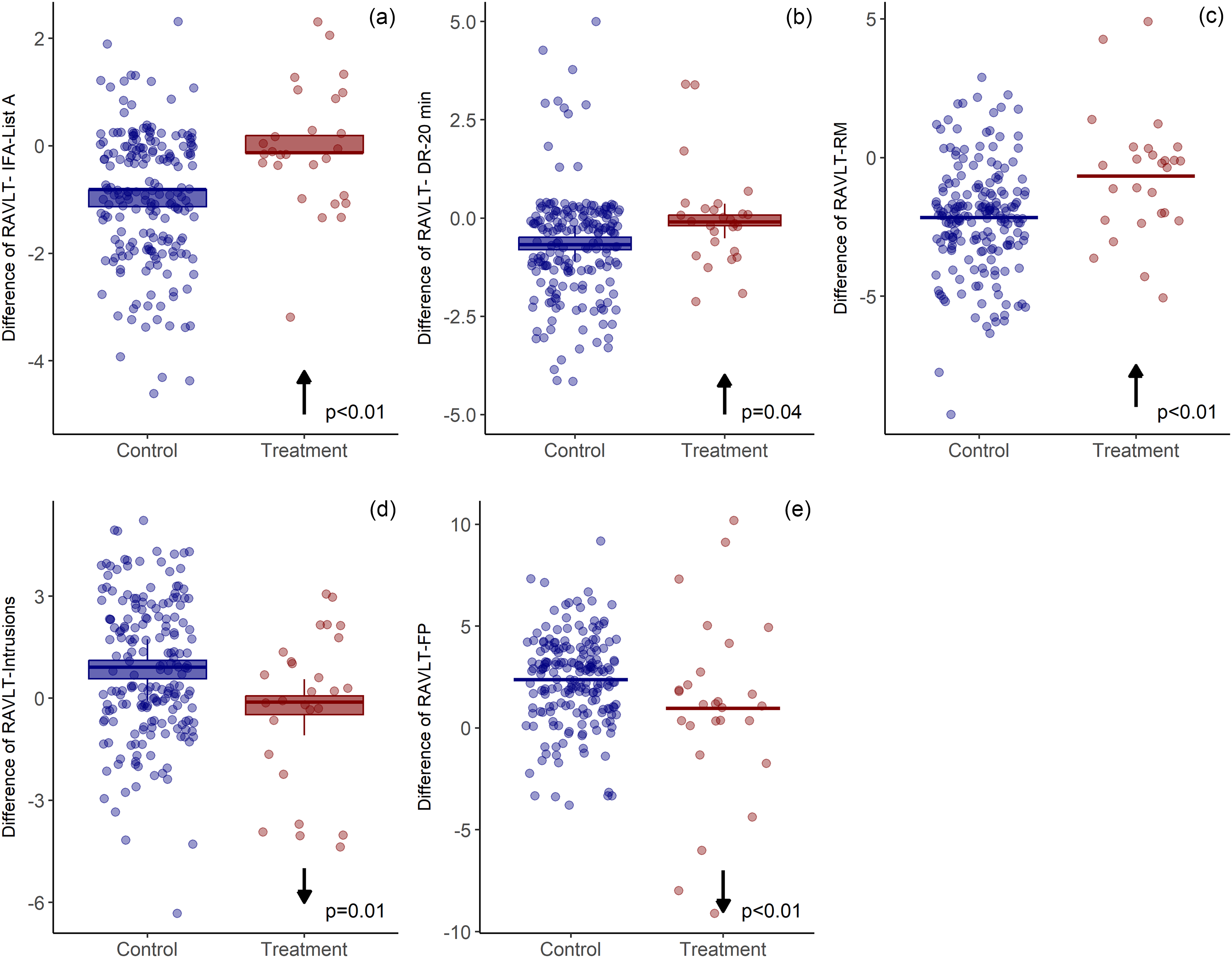
Memory test outcomes. Model-derived estimates adjusted for group and relevant covariates. Mean change (± standard error of the mean) in five components of the Rey Auditory Verbal Learning Test (RAVLT) from baseline to post-intervention in patients with mild to moderate Alzheimer's Disease. Each dot represents an individual patient. The Treatment group received therapeutic plasma exchange (TPE), and outcomes were compared to the historical control group. Arrows indicate the direction of clinical interpretation related to the test. Both, an upward arrow (↑) and downward arrow (↓) reflect less cognitive decline. Compared to the control group, the TPE-treated group exhibited significantly less decline across all RAVLT components: 88% less decline in RAVLT Immediate Free Recall (List A) ↑, 74% less decline in RAVLT Delayed Recall (20 min) ↑, 69% less decline in RAVLT Recognition Memory ↑, 74% fewer Intrusions ↓, 59.5% less decline in RAVLT Forgetting Percentage ↓.
Language test outcomes
The SVF and BNT showed significantly less decline in the TPE group compared to control group, Figure 4. The BNT showed 35% less decline (p = 0.03), while the SVF showed 37% less decline (p ≪ 0.001). Patients with CDR 1 showed less decline compared with patients with 0.5 (p = 0.017. This result is not shown in the Figure 4.
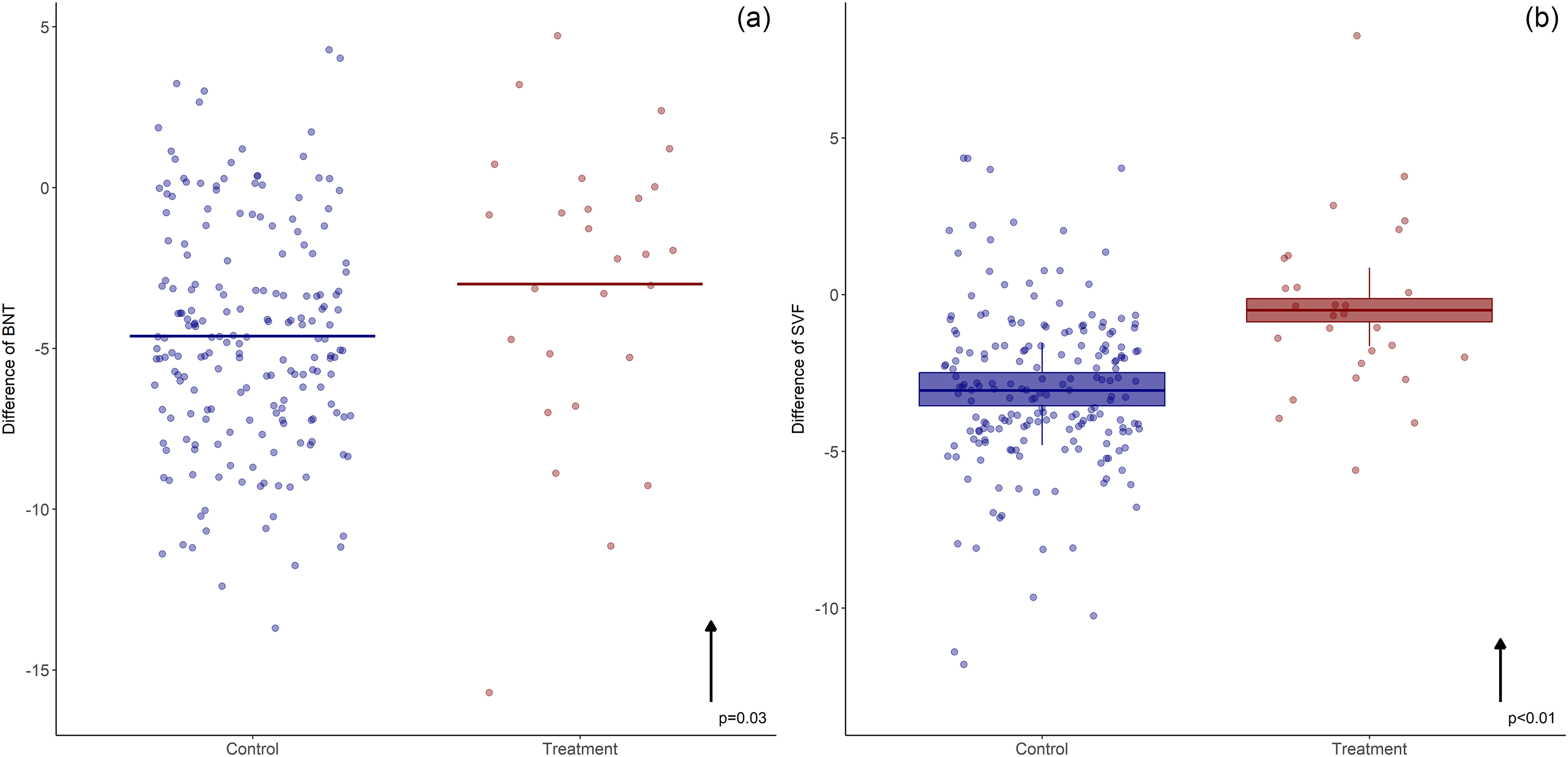
Language test outcomes. Model-derived estimates adjusted for group and relevant covariates. Mean change from baseline scores (± standard error of the mean, represented by bars) from baseline to post-intervention in (a) Boston Naming Test (BNT), (b) Semantic Verbal Fluency (SVF) of mild to moderate Alzheimer's Disease patients treated with therapeutic plasma exchange (Treatment) compared to controls. Patients are represented by dots. Arrows indicate the direction of clinical interpretation related to the test. Upward arrow (↑) reflects less cognitive decline. The Treatment group experienced 35% less cognitive decline in the BNT (P = 0.03) and, 37% less decline in SVF (P ≪ 0.001) compared to the historical control group.
Executive function and attention outcomes
PVF showed 49% less decline (p ≪ 0.001) and the TMT-B also showed less decline in the TPE group, but the difference was not statistically significant (p = 0.12) (Figure 5).
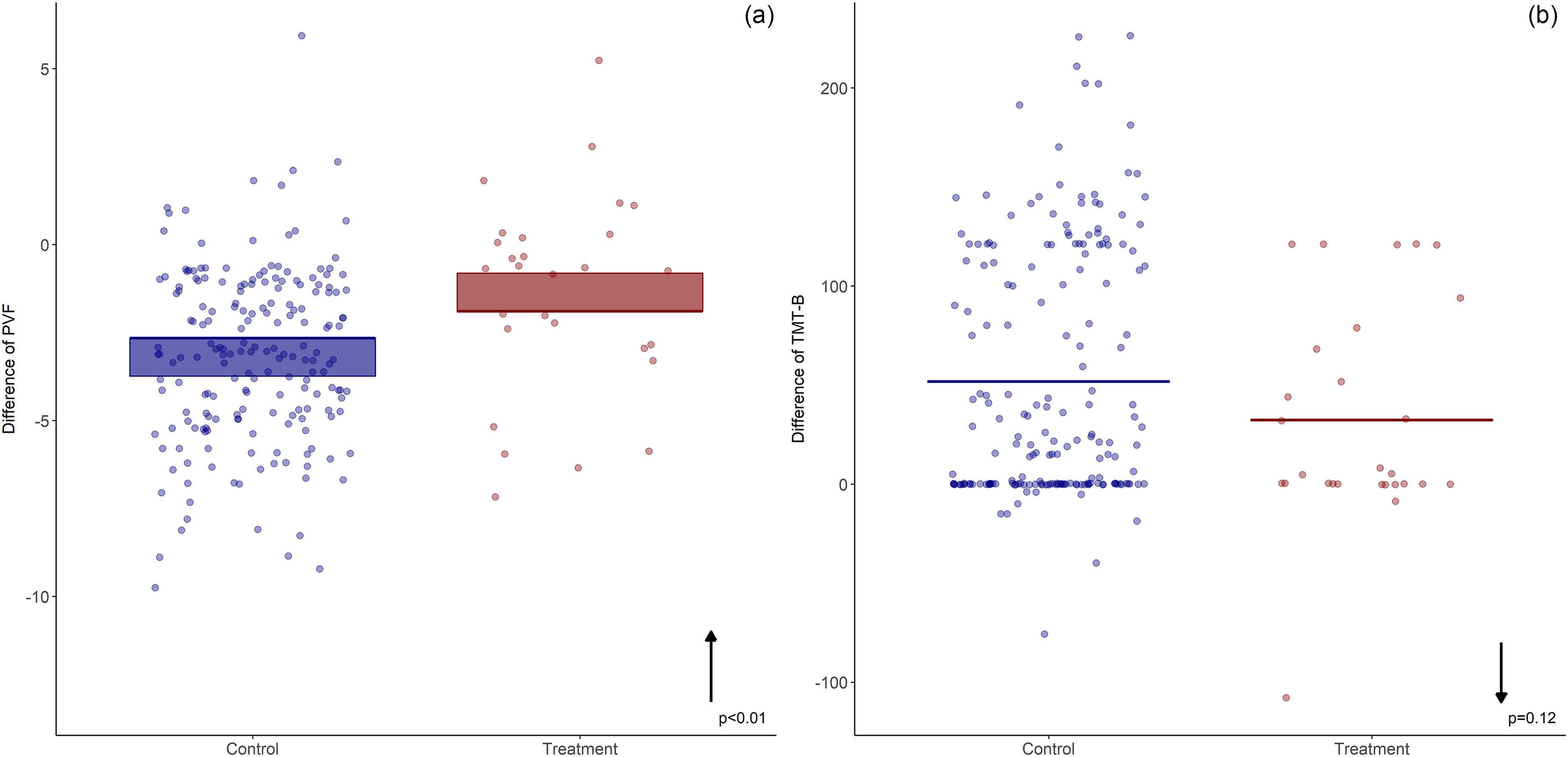
Executive function and attention outcomes. Model-derived estimates adjusted for group and relevant covariates. Mean change from baseline scores (± standard error of the mean, represented by bars) to post-intervention in (a) Category Phonemic Verbal Fluency (PVF), (b) Trail Making Test-Part B (TMT-B) of mild to moderate Alzheimer's Disease patients treated with therapeutic plasma exchange (Treatment) compared to controls. Patients are represented by dots. Both, an upward arrow (↑) and downward arrow (↓) reflect less cognitive decline. Compared to the control group, the TPE-treated group exhibited significantly less. The Treatment group experienced 49% less cognitive decline in the PVF ↑ and 37.5% less decline in the TMT-B ↓, but was not statistically significative.
The DST, both forward and backward, shows numerically less decline in the TPE group compared to controls. DST Forward showed less decline (19.1%), though not significant (p = 0.55), while DST Backward showed 80% less decline, marginally significative (p = 0.06).
All average values for Clinical Efficacy Outcomes at T1 and T2, along with the respective mean differences between the control TPE groups, are provided in Supplemental Table 1. Estimates and p-values from linear models are presented in Supplemental Table 2.
Discussion
Our findings further support the cognitive benefits of TPE in real-world AD patients, particularly in MMSE scores, memory and verbal fluency and naming ability. Our results provide additional evidence for the potential of TPE in slowing cognitive decline.
Safety
The safety profile observed in our study reinforces the overall safety of TPE in AD patients.18,39 No serious or severe AEs were reported AEs were reported, and the mild-to-moderate AEs observed were manageable.
A total of 81.5% of the procedures were uneventful. Among the 18.5% of procedures with reported AEs, 88.8% were mild and resolved within 24 h. These findings align with historical data on TPE-related AEs, which report mild AEs in 8–30% of cases and severe AEs in 0–3% of procedures. 40
The AMBAR study similarly reported that nearly 90% of procedures were uneventful, with AEs related to central venous access accounting for 20.1% compared to 13.1% for peripheral access. In our study, all procedures were performed via peripheral access, eliminating any risk associated with central venous catheters. Regarding severe AEs, the AMBAR study noted higher rates in groups receiving immunoglobulins (1.9% and 1.7%) compared to groups receiving low-albumin plasma or placebo; 0.7% and 0.9%, respectively. 18 Since immunoglobulins were not used in our study, the 0.7% rate serves as the reference for comparison. Although we recorded a higher rate of overall AEs (8.5% more than the AMBAR study) compared to AMBAR, no severe AEs were reported in our cohort. Our findings support the safety of TPE in AD patients and highlight the potential for reducing risks by exclusively utilizing peripheral venous access.
TPE–Alb is not known to be associated with amyloid-related imaging abnormalities (ARIA), such as vasogenic cerebral edema or microhemorrhages, adverse events commonly observed with anti-amyloid immunotherapies. Given the mechanism of action of TPE–Alb, a causal relationship with ARIA is considered biologically implausible. In our cohort, no patients developed clinical signs suggestive of ARIA; however, it is important to note that our study was not specifically designed to detect ARIA, as most cases are asymptomatic and require MRI monitoring for diagnosis. 41 While the absence of ARIA in our study does not confirm its non-occurrence, it nonetheless supports the favorable safety profile of TPE–Alb.
Clinical efficacy
Our findings indicate that TPE has a protective effect on cognitive function in real-world AD patients, as evidenced by the MMSE. In less than one year, the TPE group showed 2.2 points less decline compared to controls, corresponding to a 45% reduction in cognitive decline.
This is a clinically meaningful outcome, as even smaller changes in MMSE scores are associated with significant differences in disease progression and patient quality of life.26,27 Observational studies documented the natural progression of cognitive decline in AD patients. A meta-analysis of 37 studies, involving 3492 patients, found that the annual rate of change in MMSE scores over an average of 2 years was a decline of 3.3 points. 42 This highlights the potential of TPE as an intervention that may modify the course of cognitive deterioration in AD, offering a significant advantage over the typical worsening seen in observational studies.
Although FAQ outcome showed numerically less decline in the TPE group, that difference did not reach statistical significance. Several factors may help explain this. First, the relatively small sample size may have limited statistical power to detect subtle changes in functional outcomes. Second, FAQ scales may lack sensitivity to short-term changes, particularly over a treatment period of less than one year.
In this context, it is also important to consider that the outcome measures used in this real-world study differ from those employed in the AMBAR trial. AMBAR reported significant treatment effects on the Clinical Dementia Rating–Sum of Boxes (CDR-SB), Clinical Global Impression of Change (CGIC), and the Alzheimer's Disease Cooperative Study–Activities of Daily Living (ADCS-ADL). These instruments are generally more sensitive to clinical changes than FAQ, which may explain the absence of significant findings in our study despite favorable trends.
Our findings indicate that TPE has a protective effect on cognitive-memory function in AD patients, as shown by the reduced decline in all five components of the RAVLT. The observed 88% reduction in decline RAVLT IFR highlights TPE's potential in preserving short-term memory and learning capacity, which are critical for daily functioning.43–46 Similarly, the 74% less decline in RAVLT DR suggests a benefit in long-term memory retention. These findings are particularly relevant as memory decline is a hallmark of AD progression. The 69% reduction in RAVLT RM decline further supports the hypothesis that TPE may reduce susceptibility to interference and improve recognition accuracy, a cognitive domain often impaired in ADs. Additionally, the significant reductions in RAVLT Intrusions and RAVLT FP point to improved inhibitory control and reduced vulnerability to memory errors, factors that contribute to overall cognitive stability.43,47,48
The 37% less decline in SVF reflects preserved access to semantic memory, which involves the retrieval of information stored in categorical structures. SVF is particularly relevant for AD, as semantic memory is often compromised in early stages. TPE's ability to slow this decline may indicate its role in maintaining the integrity of semantic networks or mitigating neural damage in regions associated with language, such as the temporal lobes.49–52
The 49% less decline in PVF suggests preserved phonological processing and executive functions, as this task requires both lexical access and cognitive flexibility to generate words under phonological constraints. The stronger effect seen in PVF compared to SVF could imply a more pronounced impact of TPE on fronto-parietal networks responsible for executive control.50,52
The 35% reduction in decline on the BNT indicates that TPE may help maintain naming abilities, a task dependent on both lexical retrieval and visual recognition. This outcome highlights TPE's potential role in preserving cortical regions involved in language production and object recognition, such as the left inferior temporal gyrus. 53
Our study has several limitations that should be acknowledged, many of which are inherent to real-world clinical research. The relatively small sample size may limit the generalizability of our findings to the broader population of individuals with AD. Additionally, selection bias is a potential concern, as the patients included in the study may not fully represent the heterogeneity of the AD population. One factor contributing to this potential bias is that patients or their families were responsible for covering the direct costs of the TPE procedures. We recognize that this out-of-pocket expense may have influenced participation.
A further limitation is the use of a historical control group, which, while necessary for comparison, introduces potential variability in data collection methods and reporting practices. These differences could affect the consistency and reliability of our findings. Moreover, variations in patient adherence to treatment protocols in a real-world setting may have contributed to an underestimation of treatment outcomes.
Another limitation of this study is the marked imbalance in the use of acetylcholinesterase inhibitors (iACEs) between the TPE and control groups, with a significantly higher proportion of TPE patients receiving these medications. This discrepancy likely reflects real-world prescribing patterns: many patients in the control group were treated more than a decade ago, during a period when iACEs may have been prescribed more conservatively due to regulatory, clinical, or economic considerations. As this is a real-world study employing a historical control cohort, such imbalances are inherent and expected. Importantly, all patients receiving iACEs in the TPE group were stable on these medications at least three months prior to initiating plasma exchange, and there are no reports in the literature indicating that iACEs alone can produce outcomes like those observed in our study. Nonetheless, we acknowledge that concomitant iACEs use may have contributed, at least in part, to the observed differences. Future studies should aim to control baseline medication use either through matching or stratification, to more clearly isolate the specific effects of TPE. Another limitation of this study is the difference in educational level between groups, with patients in the TPE group having on average a higher level of formal education than those in the control group. Given the well-established association between educational attainment and cognitive reserve, this imbalance may have influenced both baseline cognitive performance and the observed trajectory of cognitive decline. Although changes were assessed longitudinally within individuals, the higher educational level in the TPE group could have contributed to a greater capacity to compensate for underlying neuropathology, potentially affecting responsiveness to intervention.
To mitigate these limitations, we employed a control group six times larger than the treated cohort to enhance statistical power and reduce the risk of false-positive findings. Despite these efforts, cautious interpretation of the results is warranted, and further prospective studies are necessary to validate our findings.
In conclusion, our result aligns with results from the AMBAR study, where TPE showed a positive impact on cognitive outcomes, suggesting that TPE-Alb may mitigate the progression of neurodegeneration associated with AD. When compared to the AMBAR study, our real-world results reinforce the potential of TPE in AD treatment. However, it is noteworthy that while AMBAR primarily focused on a controlled trial population, our study demonstrates similar cognitive benefits in a real-world cohort, underscoring the applicability of TPE in broader clinical practice. These findings have the potential to offer AD patients with overt dementia a new modality of treatment. However, additional studies are needed to further explore current areas of uncertainty, including the optimal number of TPE procedures, treatment duration, and TPE volumes.
Supplemental Material
sj-docx-1-alz-10.1177_13872877251375430 - Supplemental material for A real-world study on the safety and efficacy of therapeutic plasma exchange in patients with Alzheimer's disease
Supplemental material, sj-docx-1-alz-10.1177_13872877251375430 for A real-world study on the safety and efficacy of therapeutic plasma exchange in patients with Alzheimer's disease by Fernando Taragano, Daniel Seinhart, Patricia Epstein, Vanina Sylvestre, Cecilia Barañano, Victoria Otero Castro, Viviana Sánchez, Ariadna Kilstein, Ruben González, Valentina Franco-Trecu and Paula Costa-Urrutia in Journal of Alzheimer's Disease
Footnotes
Acknowledgements
We would like to thank the Research Unit from Instituto Nuestra Señora de las Nieves, for providing research facilities. We also would like to thank Mickael Beraud, Patricia Jarabo, and Khaddouj Benmoussa for their valuable contributions, which significantly improved the manuscript. We thank Dr Mónica Martini and Dr Martín Fontana for their valuable support with data entry. Part of literature search was using Scispace AI ![]()
Ethical considerations
Ethical approval was not required
Consent to participate
All participants provided written informed consent prior to participating.
Consent for publication
Not applicable
Author contributions
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by the GARCIA PUMARINO PUENTE-MP, unrestricted grant #B0010-00008790.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Paula Costa-Urrutia is a full-time employee in the Medical Affairs department at Terumo BCT. The remaining authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
Data sharing is not applicable to this article as data generation is still ongoing, but it is available upon request.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
