Abstract
Background
There is still need for a better understanding of which specific follow-up medical assessments might offer greater predictive value for diagnostic conversion from mild cognitive impairment (MCI) to Alzheimer's disease (AD).
Objective
To examine the longitudinal predictive importance of follow-up medical assessments to detect diagnostic conversion from MCI to AD.
Methods
A sample of 572 participants from the ADNI database with valid data at baseline medical visit were included. Bayesian hierarchical models were employed to investigate longitudinal predictors of diagnostic conversion in a 36-month medical follow-up cohort, for measures of cognitive function, psychopathological symptoms, and demographical data. An additional 48-month medical follow-up cohort was considered to investigate the predictive importance of cerebrospinal fluid biomarkers (Aβ42/Aβ40 ratio) for diagnostic conversion.
Results
Mini-mental State Examination (MMSE) (β = −2.6; 95% HDI: [−3.6–−1.5]) and Clinical Dementia Rating scale Sum of Boxes (CDR-SB) (β = 5.6; 95% HDI: [4.3–7.0]) can predict diagnostic conversion from MCI to AD over a 36-month medical follow-up, with CDR-SB showing the greatest predictive importance in all Bayesian models. Higher scores on CDR-SB were associated with increased risk for a diagnosis conversion, approximately 30% greater probability at 24-month follow-up, and > 50% greater probability at 36-month follow-up.
Conclusions
The CDR-SB provides a reliable cognitive assessment to detect diagnostic conversion from MCI to AD over a period of 36 months, which is key to help clinicians screening for early diagnosis of AD using affordable non-invasive procedures.
Keywords
Introduction
Current evidence suggests mild cognitive impairment (MCI) in middle aged and older adults to be a potential prodromal stage of dementia, meaning that many individuals with MCI might see at some point their diagnosis being converted to Alzheimer's disease (AD).1–4 Medical and cognitive follow-up assessment of individuals with MCI is therefore key for anticipating and managing early stages of dementia, particularly AD.1–3 The typical follow-up assessment protocol for adult MCI patients includes cognitive function (neuropsychological assessment), brain structures assessment using neuroimaging, cerebrospinal fluid (CSF) biomarkers, and psychopathological symptoms.Click or tap here to enter text.5,6 The information given by these assessments is clinically relevant, but there is still need for a better understanding of which specific follow-up assessments might offer greater predictive value for diagnostic conversion from MCI to AD.4,7
Recent literature has drawn attention to biomarkers of diagnostic conversion from MCI to AD coming from structural neuroimaging measures (e.g., magnetic resonance imaging (MRI)). Reduced cortical volumes and thickness have been soundly associated with greater risk for further AD.8–11 In addition, frequent moderate to severe MRI-visible perivascular spaces in the centrum semiovale were found to significantly predict diagnostic conversion from MCI to AD (hazard ratios ranging from 2.0 to 2.7). 12 Shape diffeomorphometry patterns of subcortical and ventricular structures were recently proposed as a novel neuroimaging biomarker for the same diagnostic conversion. 13 Another recent study tested a novel multi-modality neuroimaging model, in which structural MRI was combined with fluorodeoxyglucose positron emission tomography (PET), with promising results showing a good performance when predicting further AD diagnosis in MCI patients. 14
Additionally, multiple studies have identified CSF biomarkers to be clinically relevant for early diagnosis of dementia, particularly high levels of amyloid-β (Aβ)42, Aβ40, total tau, and phosphorylated tau.15–17 Extracellular amyloid plaques comprising Aβ peptides are regarded as an important neuropathologic feature of AD, with their assessment being typically performed by lumbar puncture or amyloid PET scans. Recent literature has suggested a greater predictive value of the ratio Aβ42/Aβ40 for predicting early stages of dementia and AD, in comparison with other CSF biomarkers.17,18 Positive plasma Aβ42/Aβ40 has been highlighted as a potential independent predictor of current and further brain amyloidosis and for diagnosis conversion to AD, even when amyloid PET is negative.15,16
The available literature has highlighted the importance of measuring cognitive disfunction in individuals with MCI, as it is regarded as an important marker to identify early stages of dementia, including AD.1,2,5,6 Recent longitudinal studies have confirmed the predictive value of cognitive assessment for the early detection of AD,19–21 although the available evidence is still limited. Common instruments that have been used to assess cognitive disfunction in MCI patients include the Mini-Mental State Examination (MMSE), 22 the Alzheimer's Disease Assessment Scale—Cognitive Subscale (ADAS-Cog), 23 and the Clinical Dementia Rating scale Sum of Boxes (CDR-SB). 24 Previous research suggests that scores from ADAS-Cog and CDR-SB might offer greater assessment precision for severity of cognitive dysfunction in individuals with different cognitive profiles (healthy, MCI and AD). 25
The comorbidity of AD and depression is well-know, although the nature of this association remains unclear. 26 The literature is ambiguous on whether depression predicts diagnostic conversion from MCI to AD, and whether it is regarded as a prodrome to AD. 26 Nevertheless, there is a growing body of literature showing that depression itself can be a risk factor for further AD and other forms of dementia, even when depression occurs years before the onset of AD. 27 The literature has highlighted that depression associated with AD can also occur as a reaction to the disease and its related difficulties. 28 The evidence of pathological mechanisms underlying the link between depression and AD is still preliminary and mostly based on pre-clinical research, with the literature drawing attention to hypothalamic-pituitary-adrenal (HPA) dysregulation, neuroinflammation, and deficits in the 5-HT receptors. 26
Other potential sociodemographic and clinical factors of diagnosis conversion from MCI to AD have been identified. According to a large cohort study using a novel machine learning-based model, factors such as the age at onset of MCI, ongoing obstructive sleep apnea, long-term opioids use, spondylosis, brain damage, and viral infection diseases of the abdomen (e.g., hepatitis C), can have an interesting clinical predictive value for the progression from MCI to AD. 4
In this article, we want to contribute to the investigation of longitudinal predictors of diagnostic conversion from MCI to AD, using contemporary and robust advanced statistical methods for predictive modelling, particularly Bayesian hierarchical models, which have been regarded as powerful statistical tools in clinical research.29,30 Previous research has highlighted the high predictive value of neuroimaging biomarkers to detect diagnostic conversion to AD, but evidence is still limited on the actual predictive value of other non-invasive clinical measures such as cognitive and neuropsychiatric assessments. These assessments might offer a less expensive and faster route to screen patients for early detection of AD. Better evidence on predictors of further diagnostic conversion to AD is of paramount importance, as it can contribute to better clinical decision making when managing early stages of dementia and choosing patient best treatment options.
Methods
Study design
The current research is an observational retrospective longitudinal study of patient data from the Alzheimer's Disease Neuroimaging Initiative (ADNI) database (adni.loni.usc.edu). 31 ADNI is a longitudinal multi-center observational study, whose main goal is to validate biomarkers for AD clinical trials. The ADNI study addresses disease progression using follow-up assessments of biological markers, including neuroimaging and CSF biomarkers, and cognitive function assessed by cognitive and neuropsychological tests. In the ADNI study, patient follow-up assessments comprise periodic visits, in which patients can be assessed for cognitive function, blood biomarkers, CSF, changes in the brain structures using MRI and PET scans, and psychopathological symptoms. At each patient visit a diagnosis is made by clinicians, based on available medical data. Patients’ medical history and sociodemographic data is collected at baseline.
In our previous study with the ADNI patient data, the diagnostic conversion from MCI to AD was investigated via single-modality and multi-modality neuroimaging classification approaches using machine-learning based models. 14 Findings included a novel multi-modality approach combining structural MRI with fluorodeoxyglucose positron emission tomography, with a classification performance (balance accuracy) for diagnostic conversion of 78.4%. The current study intends to continue previous work by examining the potential classification performance of other clinical variables such as cognitive function, psychopathological symptoms, CSF biomarkers, and socio-demographic patient variables (age at baseline, sex, education), as predictors of diagnostic conversion from MCI to AD. The current study is focused on examining cases of first-time diagnosis of AD, i.e., no cases of diagnostic reconversion (from AD to MCI) were considered.
Participants
For the current study, a sample of 572 patients with valid data at baseline medical visit was extracted from the ADNI patient data repository. Participants from ADNI-1 to ADNI-3 and ADNI GO were eligible for this study. Eligibility criteria included: diagnosis of MCI at baseline; have completed cognitive, CSF, psychopathological, and sociodemographic assessments at baseline; and follow-up medical assessments, including a valid diagnosis (MCI or AD). At the time data was collected (June 2024) we identified patient assessment data for a total follow-up of 198 months from baseline, with multiple intermediate follow-up assessments included. Full patient data for all follow-up assessments as extracted from ADNI data repository is presented in the Supplemental Material (see Supplemental Figure 1). For the current study, two subsampled cohorts were extracted from the original ADNI patient data, to include 36-month and 48-month medical follow-up assessments (details in Data Preprocessing section).
Measures
The study outcome measure is the formal medical diagnosis, MCI or AD, which is made at the medical visit during the medical follow-up. Independent variables comprised measures of cognitive function / decline, psychopathological symptoms, CSF biomarkers, and patient socio-demographical data.
Measures for cognitive function/decline include the MMSE 22 and CDR-SB. 24 The MMSE and the CDR-SB are widely used standardized instruments to assess cognitive impairment for staging dementia due to Alzheimer's disease.6,25
The assessment of psychopathological symptoms included the Geriatric Depression Scale (GDS), 32 and the Neuropsychiatric Inventory Questionnaire (NPIQ). 33 The GDS is a standardized questionnaire to assess symptoms of depression in older adults. The instrument has been validated for different clinical settings, including in inpatients and outpatients with mild to moderate cognitive impairment. 34 The NPIQ assesses neuropsychiatric symptoms and psychopathology, including depression, anxiety, apathy, sleep disturbances, changes in appetite, disinhibition, irritability, delusions, hallucinations, agitation, euphoria, and aberrant motor behaviors. The instrument has been validated and widely used in patients with neurological disorders. 35
CSF biomarkers (obtained via lumbar puncture) were assessed using the ratio Aβ42/Aβ40 calculated for each medical visit (when available). Patient socio-demographical data collected at baseline included age at baseline, sex, and education.
Data preprocessing and statistical analysis
All data preprocessing and statistical analyses were performed using
The main subsampled cohort (baseline to 36 months) allowed us to include the greatest number of observations for all cognitive, neuropsychiatric and demographical variables throughout the medical visits. The variable CSF biomarkers (ratio Aβ42/Aβ40) was not included in the main cohort due to its high proportion of missing cases throughout the 36-month medical follow-up. In the primary data extracted from ADNI, most patients had available CSF biomarkers (ratio Aβ42/Aβ40) assessments at baseline and months 24 and 48, with a considerably reduced number CSF assessments for other medical visits (see Supplemental Figure 1). For this reason, two separate cohorts were subsampled from the original dataset extracted from the ADNI repository: Cohort 1 (main): patient data for medical visits at baseline, plus follow-up assessments at months 12, 24 and 36. This dataset contained all demographical variables plus cognitive (CDR-SB; MMSE) and neuropsychiatric measures (NPIQ; GDS); Cohort 2: patient data for medical visits at baseline, plus follow-up assessments at month 24 and 48. This dataset included all demographical, cognitive and neuropsychiatric measures plus the variable CSF biomarkers (ratio Aβ42/Aβ40). Multiple imputation was performed for both datasets, assuming that missing observations were at random, using fully conditional specification implemented through the
The full data analysis strategy is presented in Figure 1. Bayesian hierarchical models were computed to investigate predictors of diagnostic conversion from MCI to AD during the medical follow-up included in our two cohorts. Unlike the frequentist statistical approach, Bayesian estimation models allow us to quantify uncertainty based on prior information on the distributions of parameters of interest. Bayesian estimation has become popular in medical research,29,30,39 and is regarded as a powerful statistical method for handling relatively small samples entailing clustered or multilevel data at higher precision, in comparison with the frequentist statistical approach.40,41 In addition, Bayesian hierarchical models account for both fixed and random effects. This can be advantageous in longitudinal data containing individual trajectories for which a random effect is expected to be associated with.
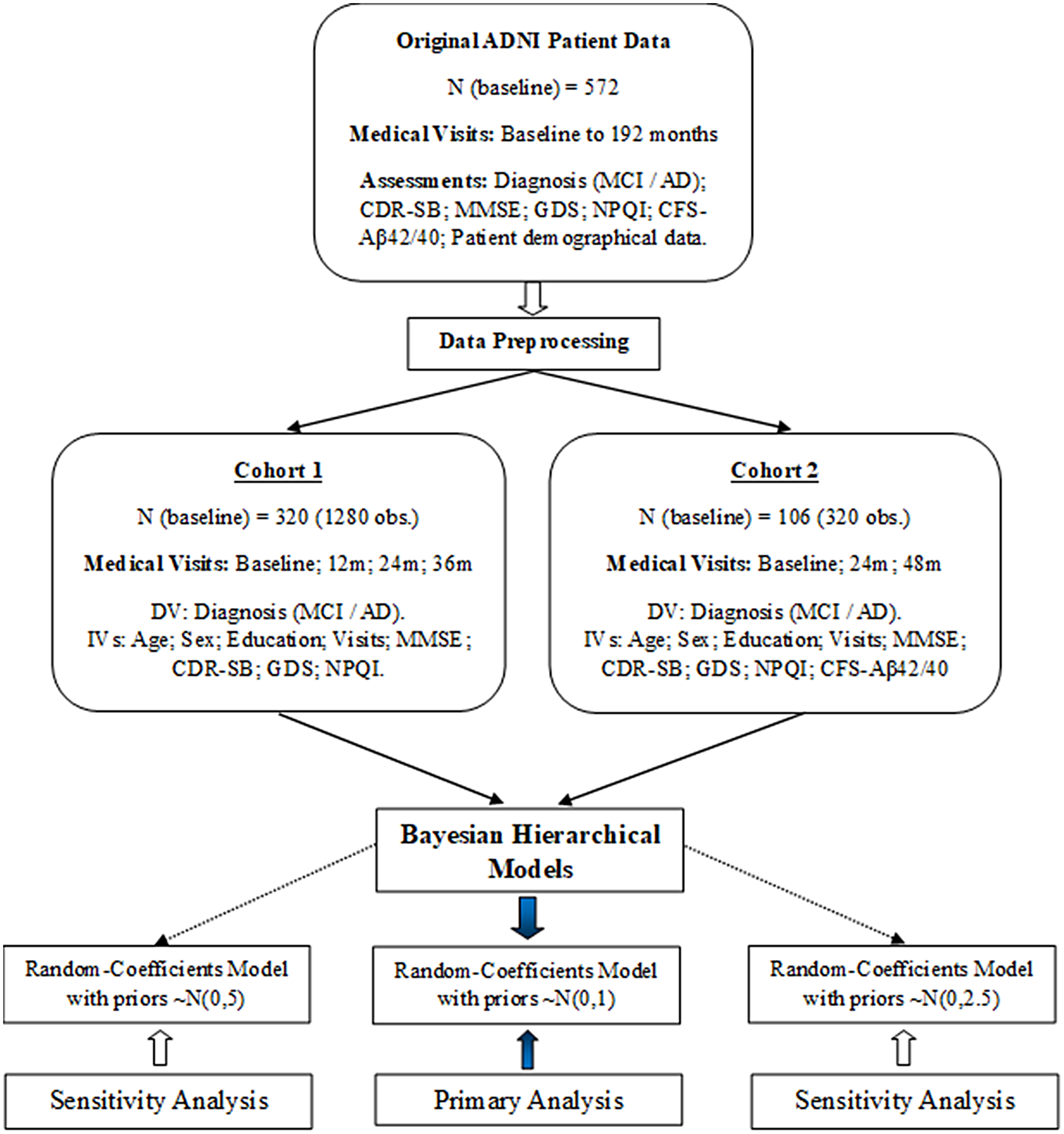
Data preprocessing and statistical analysis strategy.
Bayesian binomial generalized mixed-models were performed considering the longitudinal design of this study and the binary outcome measure (diagnosis: MCI or AD). Random-coefficients models were adopted as they allow us to model intra-individual variability for the intercept and for the slopes (individual trajectories over time), plus the fixed-effects and corresponding interactions.
In Bayesian estimation, informative priors are generally preferred over non-informative priors as they incorporate prior evidence, thus improving estimation precision.40,42,43 However, when reliable prior knowledge is lacking, as is the case for the predictive value of cognitive and neuropsychiatric measures in MCI-to-AD conversion, informative priors may introduce bias. At the same time, recent literature has shown that non-informative (flat) priors can lead to overestimated effect sizes, contradicting their supposedly neutral role.42–46 In light of this, weakly-informative priors have been recommended as a suitable alternative in contexts with limited prior knowledge, offering guidance to estimation without introducing substantial bias.44,45 For the main analysis, we adopted a weakly-informative prior of N(0,1) for fixed effects (β), and a half-normal prior for random effects (τ). Two additional models incorporating slightly vaguer weakly-informative priors were used in a sensitivity analysis. This strategy aligns with current Bayesian literature, especially in generalized models with limited prior evidence, where flat or overly vague priors (e.g., N(0100)) are generally discouraged.45–47
Bayesian generalized mixed-model (random-coefficients):
The outcome, diagnosis (MCI or AD), is modelled using a Binomial distribution
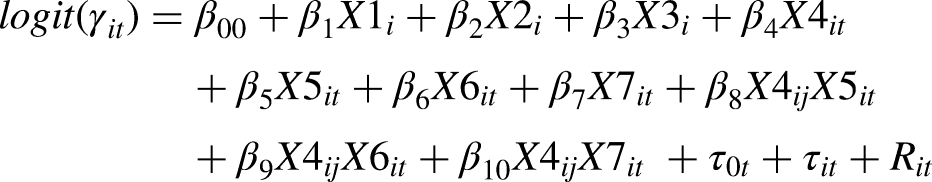
Model for the response binary variable γ (MCI or AD), with the covariates age at baseline, (X1), sex (X2), education (X3), assessment visit (X4), CDR-SB (X5), GDS (X6), CSF (Aβ42/Aβ40) (X7) and the temporal interactions X4:X5, X4:X6, and X4:X7. i = 1…n individuals, and t = 1 to 2 or 3 assessment visits (time). β represents the fixed-effects (level 1) and τ0t and τit represents the random-effects at individual level (intercept) and for the individual slopes respectively. Residuals are represented by Rit.
Model prior distributions:
All Bayesian models were run with the
Results
Participants’ characteristics at baseline are presented in Table 1. The distribution of the dependent variable “Diagnostic” throughout the 36-month follow-up is presented in Figure 2.
Distribution of the dependent Variable “diagnostic” throughout the medical visits. Variable “Diagnostic”: 0 = Mild Cognitive Impairment; 1 = Alzheimer's Disease; subID: subjects (total observations.
Sample characterization at baseline.
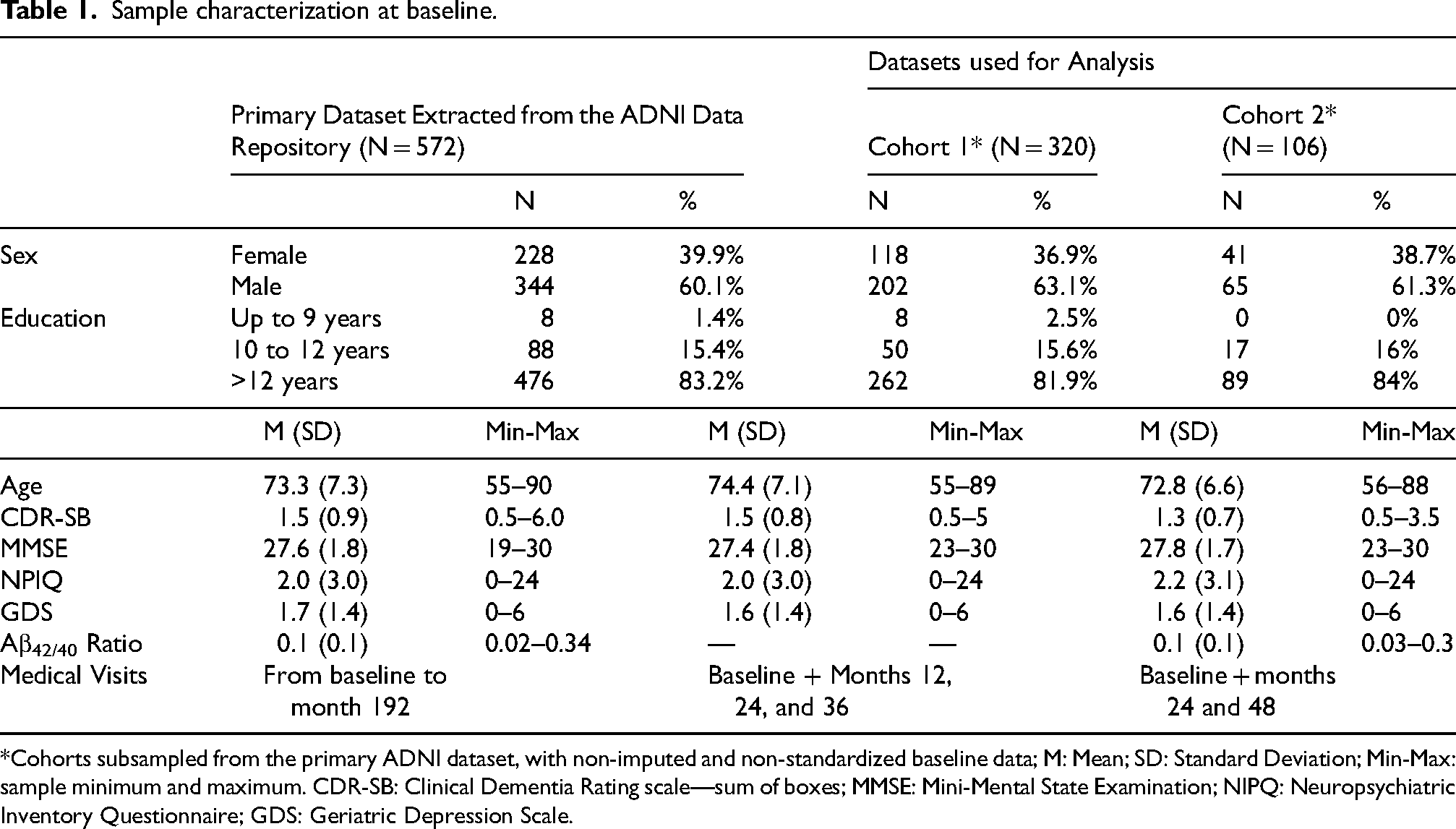
*Cohorts subsampled from the primary ADNI dataset, with non-imputed and non-standardized baseline data; M: Mean; SD: Standard Deviation; Min-Max: sample minimum and maximum. CDR-SB: Clinical Dementia Rating scale—sum of boxes; MMSE: Mini-Mental State Examination; NIPQ: Neuropsychiatric Inventory Questionnaire; GDS: Geriatric Depression Scale.
Predictors of diagnostic conversion—Bayesian hierarchical models
The results of Bayesian generalized mixed-models (random-coefficients) using different prior distributions are presented in Tables 2 and 3 for cohorts 1 and 2, respectively. All algorithms used in Bayesian models estimation converged, with all R-hat values (Gelman-Rubin convergence statistic for MCMC diagnostics) equal to 1 (Tables 2 and 3). Bayesian model 1 diagnostics, including trace-plots, posterior predictive distributions, posterior check plot, cumulative quantile plot, and effective sample sizes are presented in the Supplemental Material and confirmed model convergence and reliability of posterior estimates.
Bayesian generalized mixed models with the cohort 1 (random-coefficients).
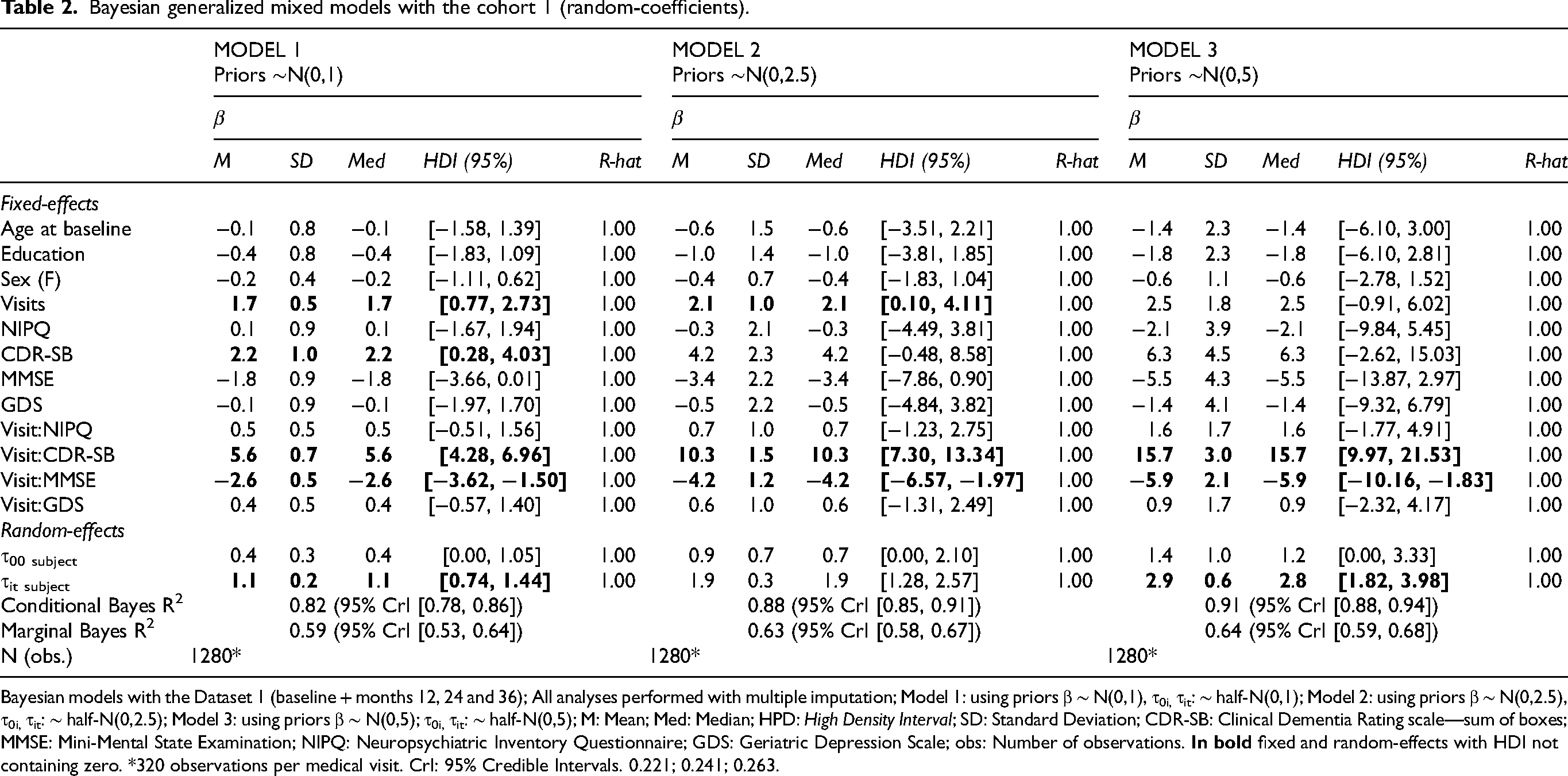
Bayesian models with the Dataset 1 (baseline + months 12, 24 and 36); All analyses performed with multiple imputation; Model 1: using priors β ∼ N(0,1), τ0i, τit: ∼ half-N(0,1); Model 2: using priors β ∼ N(0,2.5), τ0i, τit: ∼ half-N(0,2.5); Model 3: using priors β ∼ N(0,5); τ0i, τit: ∼ half-N(0,5); M: Mean; Med: Median; HPD:
Bayesian generalized mixed models with the cohort 2 (random-coefficients).
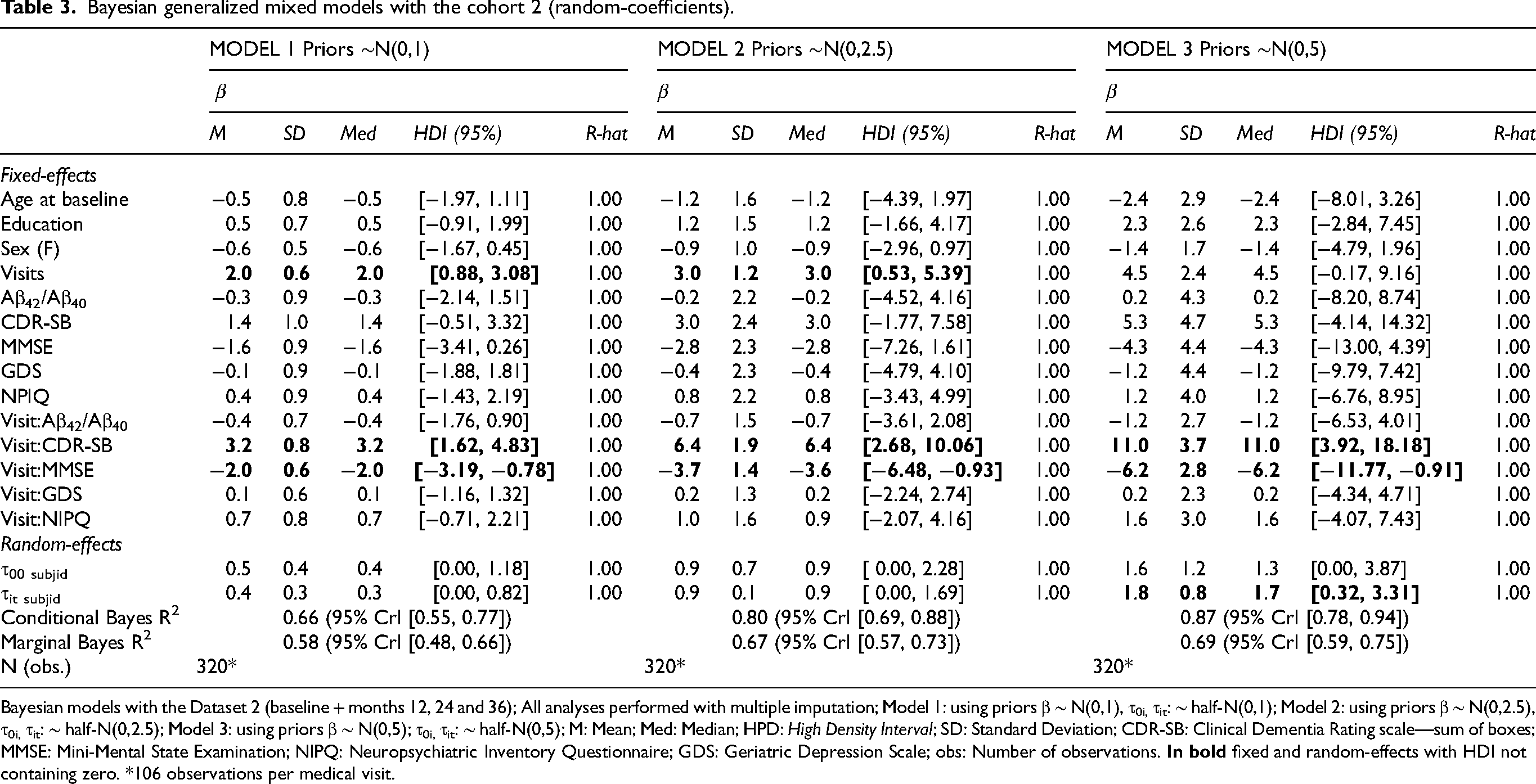
Bayesian models with the Dataset 2 (baseline + months 12, 24 and 36); All analyses performed with multiple imputation; Model 1: using priors β ∼ N(0,1), τ0i, τit: ∼ half-N(0,1); Model 2: using priors β ∼ N(0,2.5), τ0i, τit: ∼ half-N(0,2.5); Model 3: using priors β ∼ N(0,5); τ0i, τit: ∼ half-N(0,5); M: Mean; Med: Median; HPD:
Results using the cohort 1 (Table 2) suggested the interactions
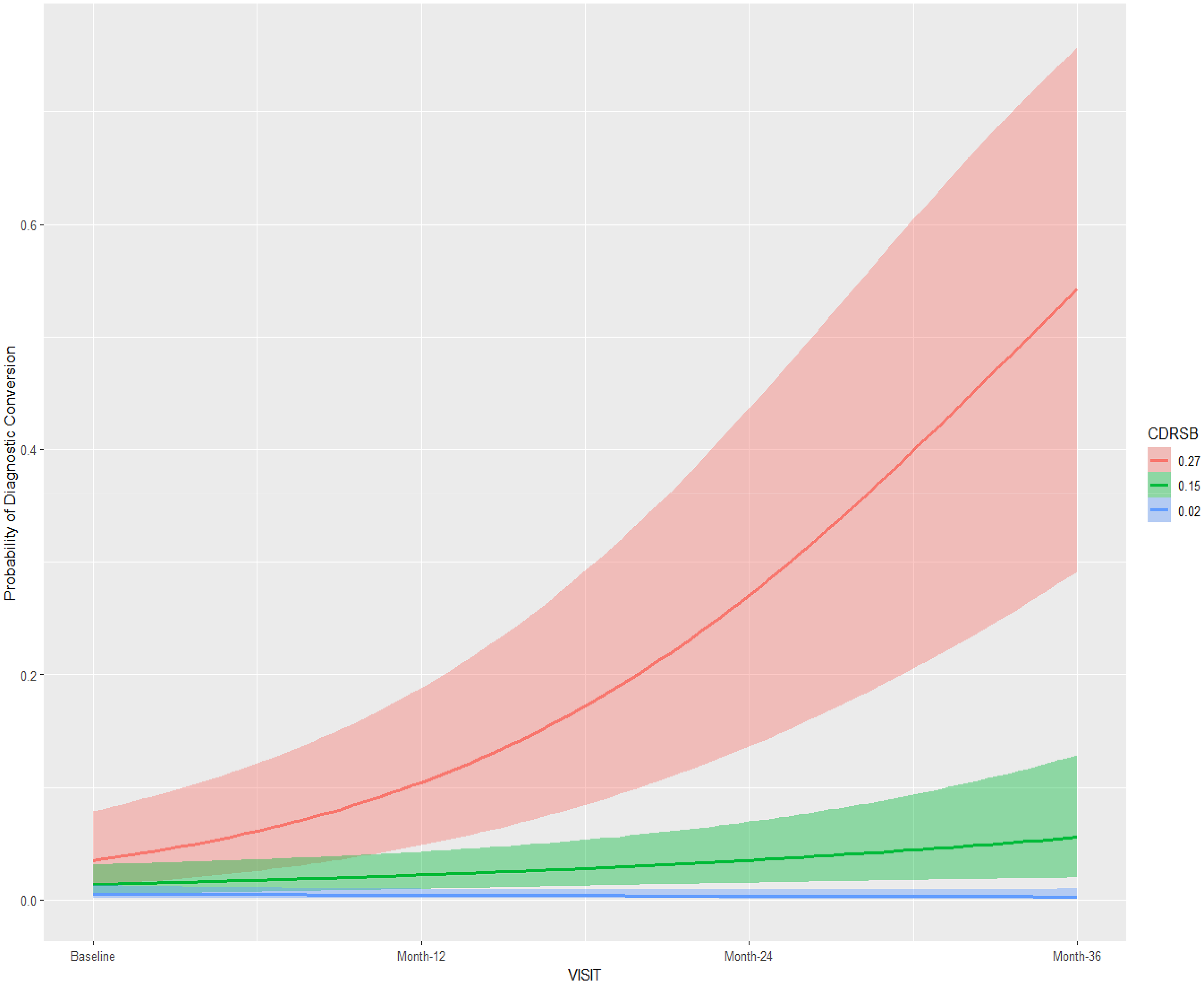
Posterior conditional effects for the interaction visit:CDR-SB on the diagnostic conversion from mild cognitive impairment to Alzheimer's disease. CDRSB: Cognitive Dementia Rating Scale; Standardized values: mean ±1 standard deviation.
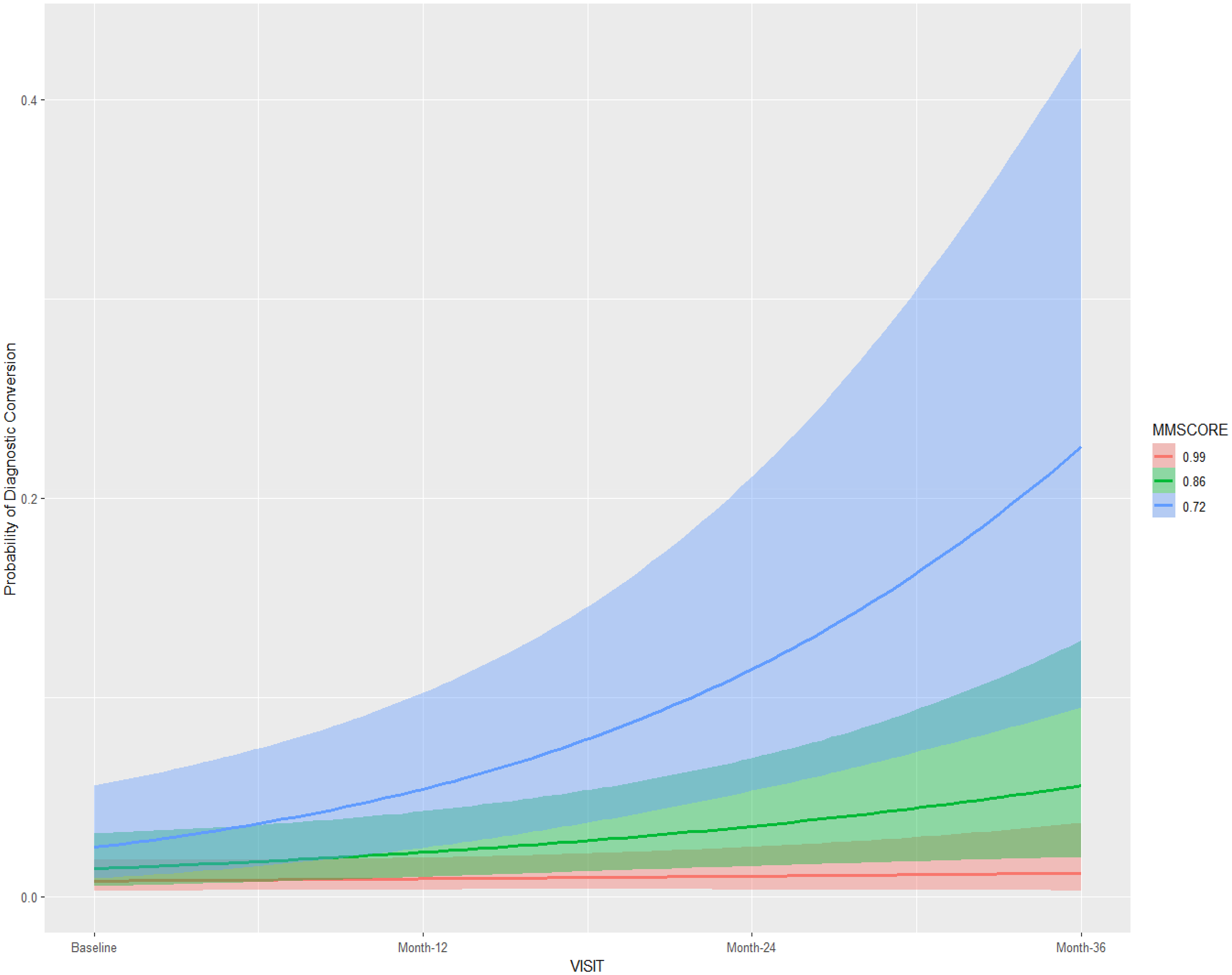
Posterior conditional effects for the interaction visit:MMSE on the diagnostic conversion from mild cognitive impairment to Alzheimer's disease. MMSE: Mini-Mental State Examination; Standardized values: mean ±1 standard deviation.
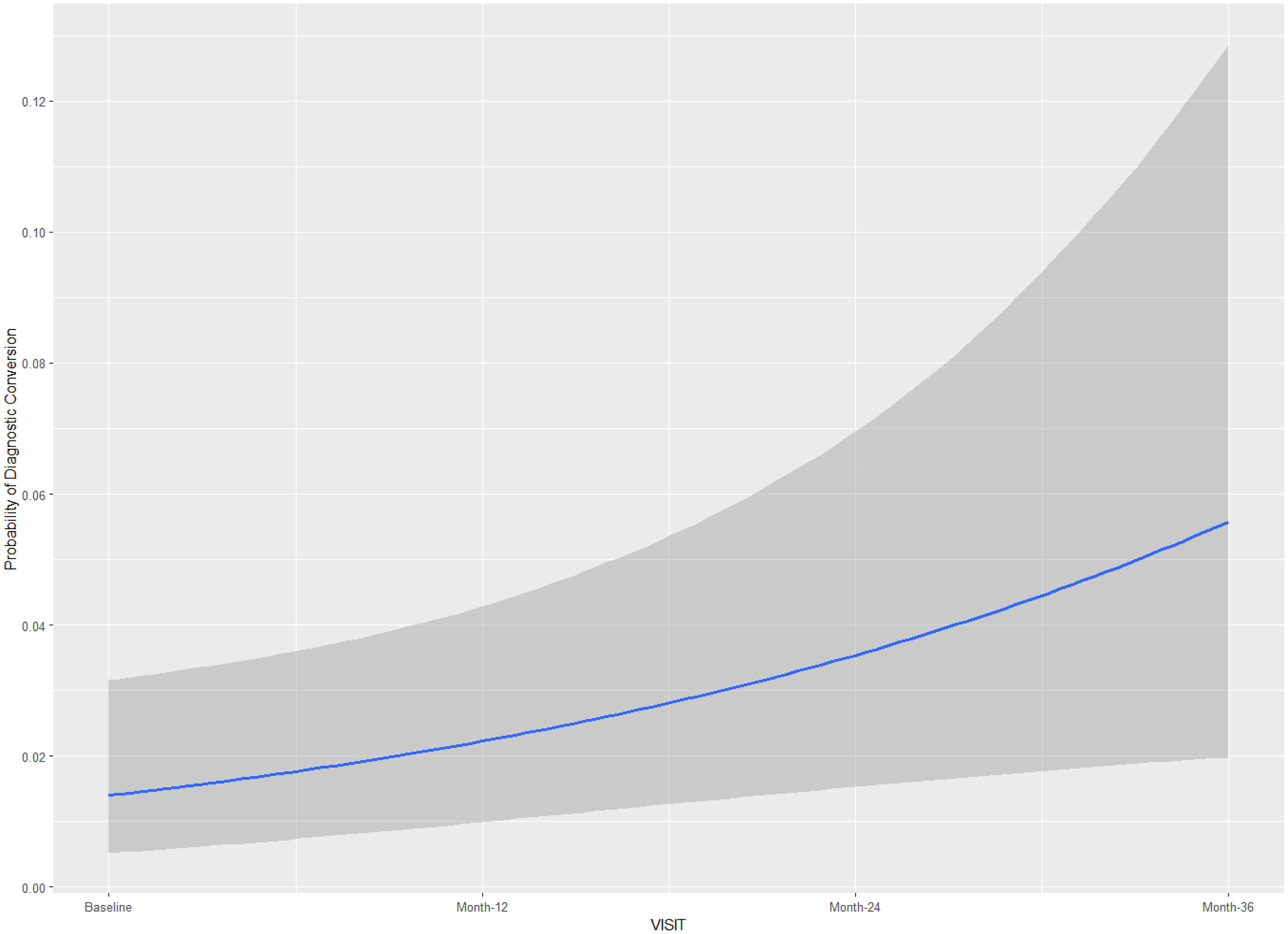
Posterior conditional effects for medical visits on the diagnostic conversion from mild cognitive impairment to Alzheimer's disease.
Results using the cohort 2 were very similar to those found with cohort 1, as the same fixed-effects were suggested to have an effect on diagnostic conversion to AD (Table 3). The CSF biomarker Aβ-42/40 ratio was not found to influence the diagnostic conversion to AD in all models, with the corresponding HDI not containing zero in all models. Random-effects for individual slopes (τit subject; medical follow-up) showed influence on the outcome measure only for model 3 (using vaguer weakly-informative priors). Conditional and Marginal R2 for all models are presented in Table 3, suggesting that 12% to 20% of the model variance (across models using different priors) is due to random-effects.
Discussion
A cornerstone of regular cognitive assessment in older adults with MCI is the early detection of AD, which is key to enhance treatment decision-making for AD patients. The current study contributes to increase the level of evidence regarding the predictive value of cognitive assessment as part of medical follow-up for detecting diagnostic conversion from MCI to AD. Bayesian hierarchical models suggested CDR-SB to be the cognitive assessment tool with the greatest predictive value for early detection of AD over a period of 36 months.
The CDR-SB is a widely validated cognitive measure that provides reliable input on patient cognitive status, which is crucial for medical follow-up of older adults with MCI. The instrument can also capture patient perceptions of cognitive performance, which has been identified as a good predictor of diagnostic conversion from MCI to AD.50,51 In particular, previous research has highlighted the potential predictive value of informant-related memory complaints for diagnostic conversion over a period of 4 years. 50 Our findings are consistent with a previous study suggesting that both CDR-SB and ADAS-Cog perform better for detecting diagnostic conversion from MCI to AD, in comparison with other cognitive measures. 25 In our study, the MMSE also showed a reliable performance to detect diagnostic conversion to AD over time, although with considerably poorer performance in comparison with the CDR-SB.
In our study, Bayesian hierarchical models suggested the GDS and the NPIQ instruments not to have a predictive value for diagnostic conversion. Previous research is unclear regarding the actual importance of depression and other neuropsychiatric symptomatology as a possible red flag for dementia in older adults with MCI, as well as the nature of the comorbidity between depression and AD.26,28 The lack of previous longitudinal studies addressing the role of depression symptomatology as a predictor of diagnostic conversion is one of the main reasons for the lack of evidence in this topic. The available literature from large-scale cohort studies suggests that depression, particularly the severity of depression, is associated with a greater risk for subsequent AD over a period of 20 years. 52 There is also preliminary evidence suggesting that depression might be a neuropsychiatric symptom of AD, with the onset triggered by the perception of functional limitations caused by the AD.28,53 Future larger-scale longitudinal studies should clarify the clinical value of GDS for early detection of AD, and whether the fact that the GDS is a self-report measure can affect its performance as a predictor of diagnostic conversion.
Recent literature has stressed the predictive importance of CSF biomarkers, particularly the CSF Aβ (Aβ42/Aβ40) ratio for early detection of AD in older adults with MCI.15–17 Studies have shown that the Aβ42/Aβ40 ratio can be the best CSF biomarker to predict diagnostic conversion overtime,17,18 together with neuroimaging biomarkers.12–14 Our Bayesian hierarchical models did not corroborate the predictive effect of Aβ42/Aβ40 ratio on diagnostic conversion over a period of 48 months. However, the results might have been affected by considerably high attrition rates over the assessment period (48 months), and the fact that for most patients the Aβ42/Aβ40 ratio data was only available at baseline, 24 and 48-month medical visits. The Aβ42/Aβ40 ratio is commonly obtained from lumbar puncture, which is an invasive medical procedure that is not usually done on a regular basis, which might, therefore, explain the limitations in the available Aβ42/Aβ40 ratio data. Future longitudinal studies with lower attrition rates should clarify the actual predictive value of this variable, as well as its importance in comparison with other predictors of diagnostic conversion.
The use of Bayesian hierarchical models in the context of medical research has proven to be of great value as they accommodate prior information on the parameters of interest and therefore offer more robust and precise predictions while being more flexible for sample size requirements, in comparison with the frequentist approach. 40 Recent literature has recommended to avoid the use of non-informative or flat prior distributions, in favor of more informative priors, even if they are weakly informative.42,43,54 Flat priors have been associated with increased risk of overestimation of the magnitude of effects leading to over-stated evidence on its sign, which in turn contradicts the “non-informative” assumption inherent to these priors. 45 Furthermore, Bayesian hierarchical models account for fixed and random-effects, which can be advantageous when dealing with longitudinal patient data, whose individual trajectories are of interest. This was corroborated by current results in the main cohort, with a considerable amount of the model variance being due to random-effects, and random-slopes showing a clear predictive effect on the outcome (diagnosis).
Main strengths of the current study include a cohort study design including a reasonable number of patient data collected from different clinical settings, and the use of contemporary and robust statistical methods such as Bayesian hierarchical models. Main study limitations include a considerably high attrition rate in cohort 2, with missing data between the included medical visits which might have limited the statistical models to capture the potential effect of predictors such as the CSF Aβ (Aβ42/Aβ40) ratio in relation to diagnosis conversion. Also, our predictive models would have strongly benefited from the inclusion of other relevant sociodemographic and clinical variables that have been previously identified as potentially important for diagnostic conversion such as age at onset of MCI, ongoing obstructive sleep apnea, long-term opioids use, spondylosis, brain damage, and hepatitis C. Our study did not include them in as model predictors which is a study limitation to be addressed in future longitudinal research. Finally, the fact that this study only accounted for pre-collected patient data limited our capacity to improve some aspects of the study design that could help reducing attrition rates and data integrity.
In conclusion, the current study highlighted the potential relevance of adopting the CDR-SB as one of the preferred clinical assessment tools in medical follow-up of MCI patients, to help screening for early diagnosis of AD. The use of CDR-SB in MCI patients offers an affordable, fast and non-invasive way to red-flag patients for an early AD diagnosis, which might warrant further medical investigation. Future large-scale cohort studies with lower attrition rates and including multimodal biomarkers will strongly contribute to generate more evidence on which assessment procedures better predict diagnostic conversion and help clinicians to improve care for patients with AD.
Supplemental Material
sj-docx-1-alz-10.1177_13872877251360228 - Supplemental material for Predicting diagnostic conversion from mild cognitive impairment to Alzheimer's disease: A Bayesian hierarchical model approach using ADNI patient data
Supplemental material, sj-docx-1-alz-10.1177_13872877251360228 for Predicting diagnostic conversion from mild cognitive impairment to Alzheimer's disease: A Bayesian hierarchical model approach using ADNI patient data by Hugo Senra, Maria Conceição Costa, Isabel Pereira, Daniel Agostinho, Miguel Castelo-Branco and for the Alzheimer’s Disease Neuroimaging Initiative in Journal of Alzheimer's Disease
Footnotes
Acknowledgments
Ethical considerations
All procedures involving human participants complied with the ethical standards of the institutional and/or national research committees, as well as the 1964 Declaration of Helsinki and its subsequent revisions or comparable ethical guidelines. The study was approved by the institutional review boards of all participating centers.
Author contributions
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
