Abstract
Background
Dementia is a global public health challenge, with Alzheimer's disease (AD) and vascular dementia (VaD) being the most prevalent subtypes. Air pollution is a potential risk factor for dementia, but few studies separate AD and VaD.
Objective
This study aims to investigate whether exposure to air pollution is associated with the incidence of AD and VaD.
Methods
This study involved 30,247 participants from the Malmö Diet and Cancer Study (1991–2014), with 1216 AD cases (including mixed type) and 531 VaD cases, clinically validated. Long-term exposure to PM2.5 and NOx was modeled using the past 5-years or 10-year mean concentration with 50m × 50 m spatial resolution at residential addresses. The association between AD and VaD and air pollution was assessed using adjusted Cox-proportional hazards.
Results
For AD the hazard ratios (HRs (95% CIs) were 1.06 (0.89–1.27) for PM2.5 and 1.08 (0.96–1.22) for NOx per interquartile range (IQR) increase in past 10-year mean pollution-levels. Nonsignificant associations were observed in two-pollutant models. For past 5-year exposure, we observed a statistically significant association between NOx and AD 1.14 (1.00–1.30). For VaD, we found no evidence for associations in any models.
Conclusions
We found that while both PM2.5 and NOx seemed linked to increased AD risk in single-pollutant models, only the associations with NOx remained in two-pollutant models, particularly for exposures in the past five years. No associations were observed for VaD. The results underscore the importance of distinguishing dementia subtypes, identifying critical exposure windows, and applying multi-pollutant models in future studies.
Introduction
Dementia, an umbrella term for a range of cognitive impairments, is a pressing public health issue. In 2019, it was estimated that around 55 million people worldwide were living with dementia, a figure projected to rise to 78 million by 2030 and 139 million by 2050. 1 Dementia affects individuals by progressively impairing cognitive functions such as memory, reasoning, and language, leading to reduced autonomy and eventual reliance on caregivers. This cognitive and functional decline places substantial emotional, psychological, and economic stress on family members. The societal monetary costs are furthermore substantial, with the global cost of dementia estimated at US$ 1.3 trillion in 2019, potentially rising to US$ 2.8 trillion by 2030. 2 As life expectancy rises, the number of people living with dementia continues to grow, even as age-specific incidence decreases in high-income countries, underscoring the importance of identifying and implementing effective prevention strategies.
The 2024 update of the Lancet Commission on dementia offers encouraging new evidence on dementia prevention, intervention, and care. 3 According to the report, low education, hearing loss, hypertension, smoking, obesity, depression, physical inactivity, diabetes, excessive alcohol use, traumatic brain injury, social isolation, untreated vision impairment, high LDL cholesterol, and air pollution, are all associated with increased dementia risk. A comprehensive review for the WHO, Forastiere and colleagues, concluded that the association between fine particulate air pollution (PM2.5) and dementia is sufficiently probable to warrant including in health risk assessments. However, they noted greater uncertainty regarding the reliability of the concentration-response function (CRF) for dementia compared to those for more established health outcomes. 4
Alzheimer's disease (AD) and vascular dementia (VaD) are the most common forms of dementia, with AD accounting for 60–70% of global dementia cases, 1 and VaD representing about 15–30%. 5 While a growing body of literature suggests that air pollution is a risk factor for dementia,6–9 significant heterogeneity exists across studies, with some reviews pointing to the need for improved methodology especially regarding case identification to strengthen the evidence and draw more definitive conclusions. 10 For example, a systematic review and meta-analysis explored the association between air pollutants and dementia risk, adjusting for study-specific factors. 11 The review included 51 longitudinal studies of adults aged 18+, assessing ambient pollution exposure averaged over a year or more and its link to clinical dementia. Among the studies, those with active case ascertainment showed a higher hazard ratio (HR) for PM2.5 at 1.42 (95% CI: 1.00–2.02), while passive ascertainment yielded an HR of 1.03 (0.98–1.07) per 10 μg/m³ increment. Additionally, there is considerable uncertainty regarding the role of cardiovascular disease as a mediator in this relationship. Some studies indicate substantial mediation, 12 while others find less evidence of this effect. 13
It is likely that air pollution's impact on AD and VaD differs due to the distinct pathological mechanisms of each subtype. AD is primarily associated with the buildup of amyloid-beta plaques and neurofibrillary tangles in the brain, leading to neuron degeneration and cognitive decline. VaD, however, results from restricted blood flow due to cardiovascular issues like stroke or atherosclerosis, damaging neurons via oxygen deprivation. While both dementia types share common risk factors (e.g., age, lifestyle factors), the divergent pathology suggests that specific risk factors may have different impacts on each. AD risk may be more closely related to neurodegeneration and genetic susceptibility, whereas VaD risk is more associated with cardiovascular health. Air pollution may exacerbate these pathways by promoting neuroinflammation and oxidative stress in AD and by inflammatory responses impairing vascular function in VaD. This distinction highlights the need to stratify dementia by subtype when studying air pollution's effects, as it allows for a clearer understanding of its role in each disease.
The objective of this study was to examine whether exposure to air pollution is associated with dementia incidence, separately analyzing clinically validated cases of AD and VaD to account for subtype-specific risks.
Methods
Population and setting
The Malmö Diet and Cancer (MDC) cohort is a population-based study recruited in Malmö, a city in southern Sweden. The study recruited individuals who lived in Malmö born between 1923 and 1950.14–16 A total of 30,446 individuals, aged 44 to 74, were recruited during a five-year period between 1991 and 1996. A questionnaire was administered to gather information on socio-demographic factors, including educational level and occupation, as well as lifestyle factors such as physical activity, smoking, diet, and medical history. Participants also underwent a comprehensive health screening, which included blood sampling and measurements of blood pressure, height, and weight.14,17
The follow-up period began at recruitment and ended at the time of an event, censoring, or on December 31, 2014. The AD dataset included a total of 30,247 participants and 30,246 for VaD, with 199 individuals excluded for AD and 200 for VaD (Figure 1). Exclusions were due to one participant's death on the day of enrollment, one with all data missing, eight missing age data, five lacking all exposures data, and 184 for AD and 185 for VaD who were under the age of 55 at the time of event or censoring. There were no prevalent cases of AD or VaD by the time of recruitment.
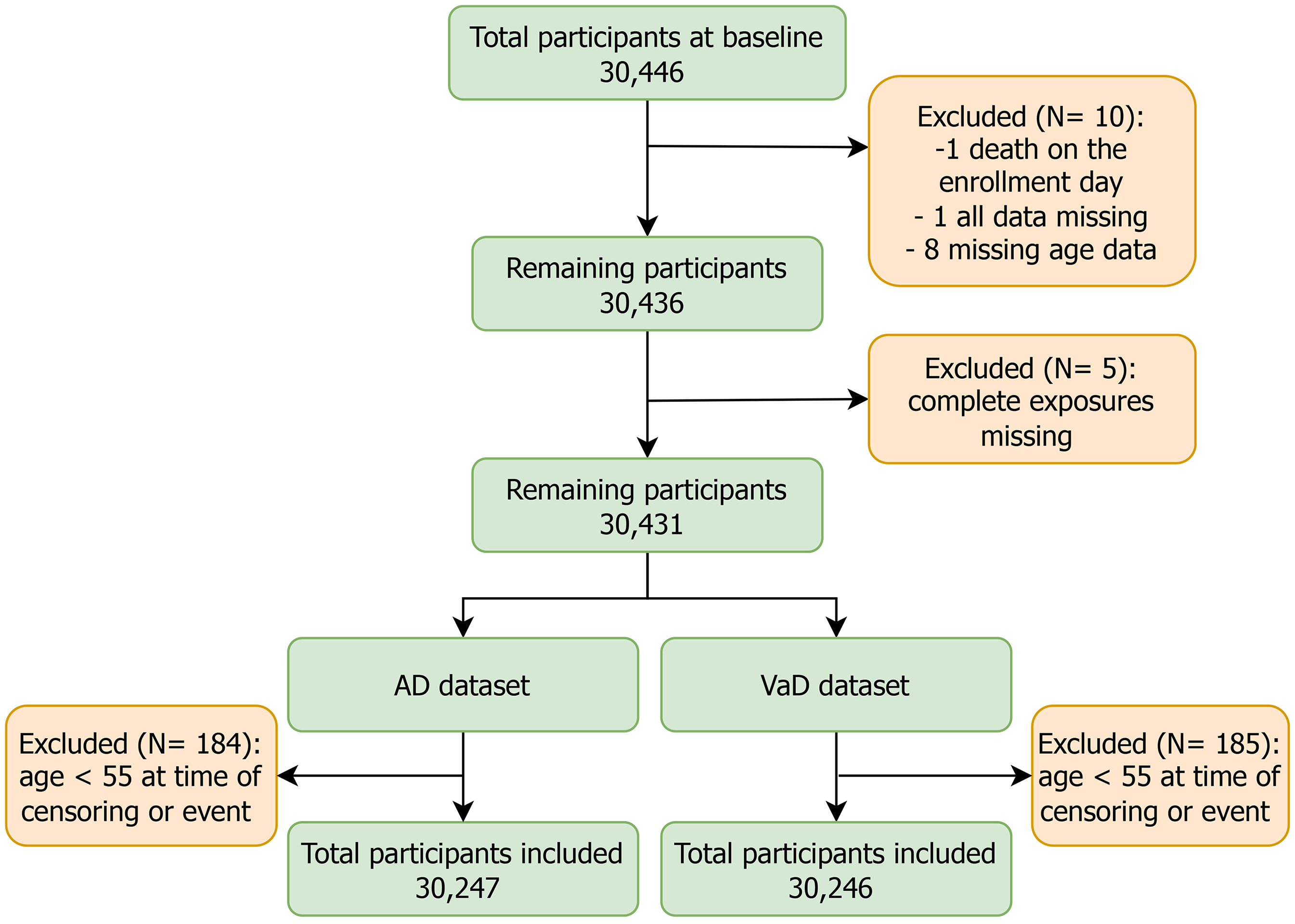
Flowchart of the study: Two separate datasets were created for the analysis of AD and VaD, respectively. AD dataset was created by combining AD and mixed dementia, and other dementia subtypes were coded as missing. For VaD dataset, VaD was used, with other dementia subtypes coded as missing.
Air pollution
To estimate the yearly exposure levels of individuals in the MDC cohort, their annual residential addresses from 1990 to 2014 (or at time of censoring) were obtained from Statistics Sweden. Each address was geocoded at the entrance of the buildings, using Geographic Information Systems (GIS), and annual mean exposures assigned to each address.
Ambient source-specific air pollution concentrations were modeled by the Environmental Department of Malmö utilizing the EnviMan software package (Opsis AB, Sweden). The modeling area covered Malmö, the third largest city in Sweden, and had a size of 18 km × 18 km. The modeling process involved separate emission databases for the years 1992, 2000, and 2011, compiled using existing inventories provided by the municipality. The resulting yearly mean modeled concentrations were stored as grids with a spatial resolution of 50 m × 50 m. The modeled years between the three-time points were interpolated linearly and adjusted for year-to-year variations in local meteorology using a ventilation factor estimated from calculations over the entire period. 17
We included PM2.5 and NOx and applied a 10-year moving average (lag1–10) as a marker for long-term exposure, and we also stratified the exposure by two lag periods; 1–5 years (lag1–5) and 6–10 years (lag6–10). Participants with more than 40% missing observations (yearly means) were excluded.
Outcome
Dementia cases were retrieved from the National Patient Registry up to 31st of December 2014. Established in the 1960s, this register has included 99% of all inpatient medical diagnoses since 1987 and hospital-based outpatient diagnoses since 2001. 18 Both primary and secondary diagnoses were considered, enabling the identification of additional dementia cases beyond primary diagnoses. The dementia types that were retrieved for this study were AD (ICD-10 F00, G30; ICD-9 331A/331.0) and VaD (ICD-10 F01; ICD-9 290E/290.4). Then, all diagnoses were clinically validated by experienced physicians using patient files and the DSM-5 criteria, the validation procedures were outlined by (Nägga et al., 2022). 18 We included two dementia subtypes in this study: AD (including mixed dementia) and VaD.
Covariates
We included a range of baseline covariates from the time of enrollment to control for potential confounding factors. Among these covariates are well-established risk factors for dementia, such as age, education level, and the presence of the apolipoprotein E ε4 (APOE ε4) allele.18,19 The age at baseline was retrieved from the personal identification number, 16 and was categorized into five-year intervals: 55–60, 61–65, 66–70, and 71–75. Socio-economic and lifestyle variables were obtained from a self-administered questionnaire completed at baseline, with occupation recorded based on the most recent at that time.16,20 Occupation was divided into five groups based on Statistics Sweden's socioeconomic index (SEI) as follow: High-rank white-collar employers/self-employed/academics (HR white-collar), Mid-rank white-collar (MR white-collar), Low-rank white-collar (LR white-collar), High-rank blue-collar (HR blue-collar), and Low-rank blue-collar/farmers (LR blue-collar). This system classifies occupations based on the educational level required for the job, the level of responsibility within the organization, and the actual tasks performed. 20 Education was classified into three categories: more than 12 years, 9–12 years, and less than 9 years. 20 Smoking status and alcohol consumption were obtained through the questionnaire. Smoking status was categorized as Never, Former, and Current. Alcohol consumption was categorized into sex-specific tertiles based on its distribution with groups labeled as Low, Medium and High. Physical activity was assessed using a total activity score derived from participants’ reported effort and time spent on activities in the questionnaire, and categorized as High, Medium, and Low. Major pre-dementia health conditions, including coronary events, stroke, and diabetes mellitus, were recorded as binary variables (Yes/No). Prevalent conditions existing before enrollment were identified through participants’ self-reported disease history and medication use, while incident cases occurred during the follow-up period but before dementia diagnosis were obtained from registers. Body mass index (BMI) used as continuous variable, was calculated by dividing each participant's weight (kg) by the square of their height (m). Systolic and diastolic blood pressure were measured once at enrollment and treated as continuous variables. The APOE ε4 genotype was categorized as a binary variable (Non-carrier or Carrier). Furthermore, area-level income was assigned based on SAMS (Small Area Market Statistics) areas from Statistics Sweden and categorized into three levels Low, Medium and High. 21
Statistical analysis
We used Cox proportional hazards models to examine the association between air pollution exposures (PM2.5 and NOx) and the risk of AD and VaD. The models were applied separately for each pollutant and outcome, with one model incorporating a two-pollutant (TP) approach. We used the mean lag1–10 for both PM2.5 and NOx, with exposure effects modeled per interquartile range (IQR) increase in pollutant concentration across the population for the study period. Time in study was treated as the underlying time scale in all analyses. Participants were censored at the earliest occurrence of a dementia event, death, emigration, loss to follow-up, or the end of the follow-up period, which was defined as December 31, 2014. Missing data was handled automatically by excluding rows with missing values. Proportional hazards assumptions were tested using the Schoenfeld residuals test. All covariates were treated as constants except exposures which were modeled as time-varying variables. We identified covariate-adjusted models for each single-pollutant analysis, chosen based on experience from previous studies. Model 1 was a crude analysis without any covariate adjustment to provide baseline estimates. Model 2 was adjusted for age, sex, and education to account for key demographic factors. Model 3 was further adjusted for smoking status, physical activity, alcohol consumption, occupation, and APOE ε4 genotype. Model 4, which incorporated all covariates from Model 3 along with pre-dementia coronary events, stroke, and diabetes mellitus to account for the impact of major health conditions on dementia risk. As an extra analysis, model 5 was adjusted for all covariates included in Model 4, with the addition of adjusting for area-level income to account for area-level socioeconomic disparities.
A Pearson correlation coefficient was used to assess the correlation between PM2.5 and NOx. We also stratified analyses by APOE ε4 and by sex using model 4. Penalized spline plots were used to assess potential non-linear associations between the pollutants and dementia outcomes in model 4. In a sensitivity analysis, we adjusted model 4 to BMI, systolic and diastolic blood-pressure to investigate potential residual confounding from cardiovascular indicators. We furthermore examined exposures during 1–5 years and 6–10 years before diagnosis to explore potential temporal variation in associations. All analysis was conducted using R software version 4.4.1, with survival, survminer and splines packages up to date.
Results
Population characteristics
Table 1 (full table in Supplemental Table 1) summarizes key characteristics of all study participants as well as for individuals with AD; (n = 1216), VaD; (n = 531). Additionally, a low-to-moderate Pearson correlation coefficient of 0.193 between Lag1–10 PM2.5 and Lag1–10 NOx indicated a limited association between the two pollutants.
Characteristics of the participants, for the total study cohort as well as for the cases of Alzheimer’s Disease (AD) and Vascular Dementia (VaD) separately where N denotes the number of cases.
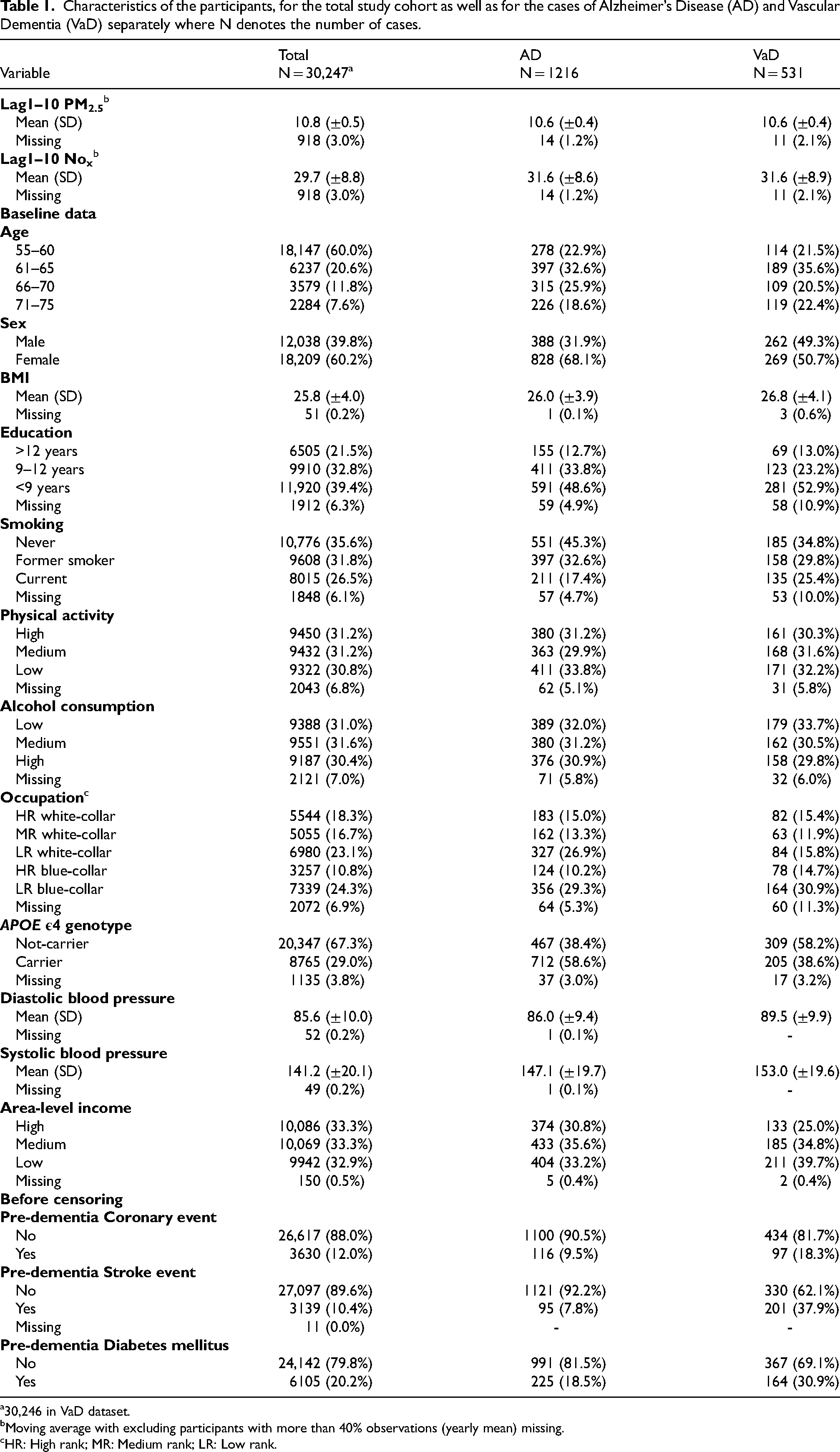
30,246 in VaD dataset.
Moving average with excluding participants with more than 40% observations (yearly mean) missing.
HR: High rank; MR: Medium rank; LR: Low rank.
Air pollution and risk of dementia
The interquartile range (IQR) over the study period for PM2.5 was 1.22 µg/m³, while the IQR for NOx varied slightly: 11.36 µg/m³ for AD and 11.34 µg/m³ for VaD. The HRs for AD in association with PM2.5 in Models 1 and 2 were close to 1, whereas the HRs were somewhat elevated when adjusting for potential confounders in Model 3–5 (Figure 2, Table 2). In the two-pollutant models, the HR for AD in association with PM2.5 was again very close to 1 (Figure 2, Table 2). For NOx, there was a statistically significant HR in the crude model 1, whereas the estimate was attenuated and no longer statistically significant after adjustment for confounders in Model 2–5. In the two-pollutant models, the HR for AD in association with NOx was very similar to the single-pollutant HRs in Model 2–5 (somewhat elevated, but not statistically significant).
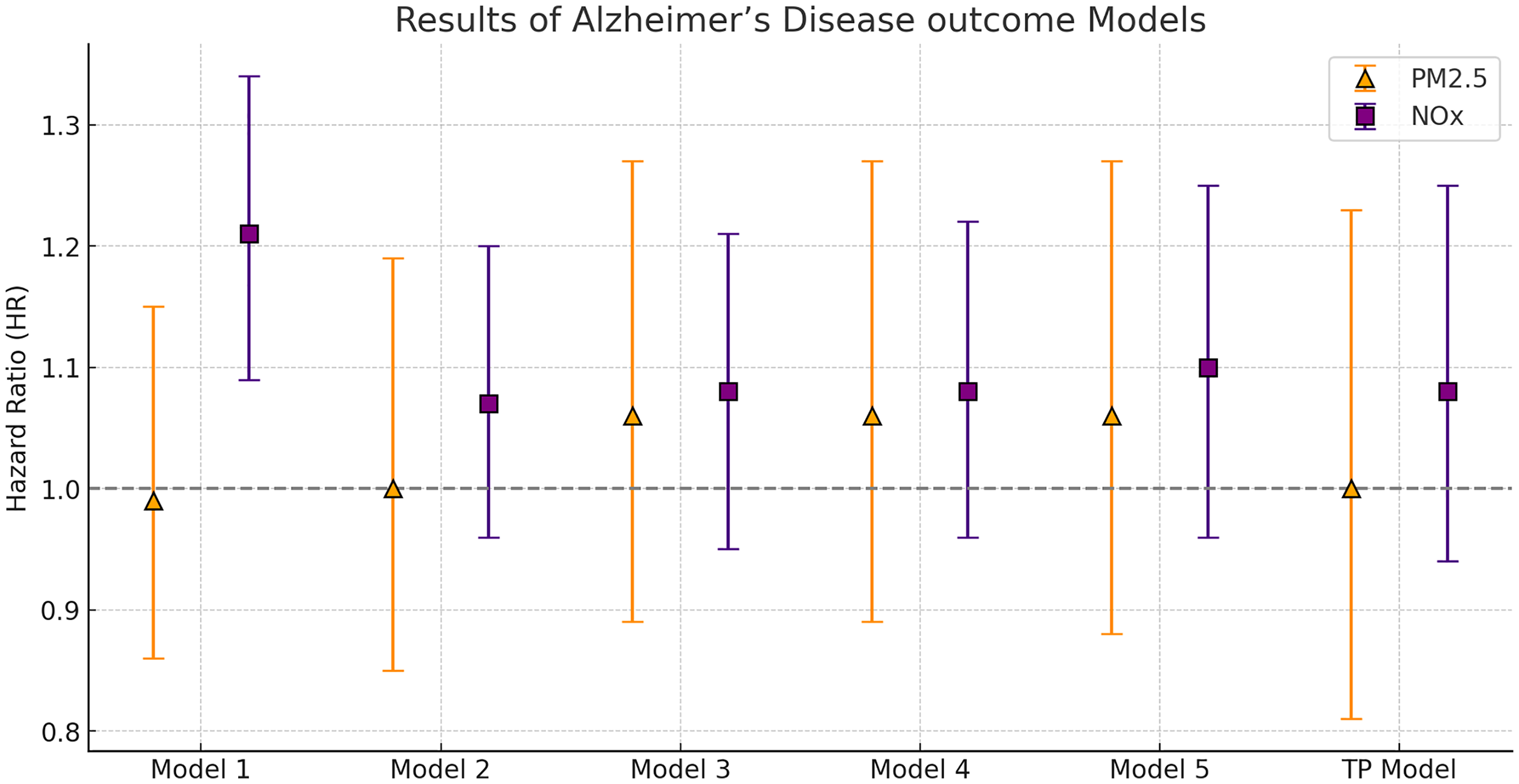
Hazard ratios (HR) for Alzheimer’s disease per intra-quartile (IQR) range increase in Lag 1–10 NOx and PM2.5. Model 1: Crude analysis. Model 2: Adjusted for age, sex, and education. Model 3: Adjusted for age, sex, education, smoking status, physical activity, alcohol consumption, occupation, and APOE ε4 genotype. Model 4: Adjusted for all covariates in Model 3, plus pre-dementia coronary events, stroke, and diabetes mellitus. Model 5: Adjusted for all covariates in Model 4, plus area-level income. Single-pollutant models were used, except in the two-pollutant (TP) model, where NOx and PM2.5. were included simultaneously, based on Model 4 adjustments.
Hazard Ratios (HRs) with 95% confidence intervals (CIs) and p-values for Alzheimer’s disease in association with an intra-quartile (IQR) increase in Lag1–10 PM2.5 and NOx.
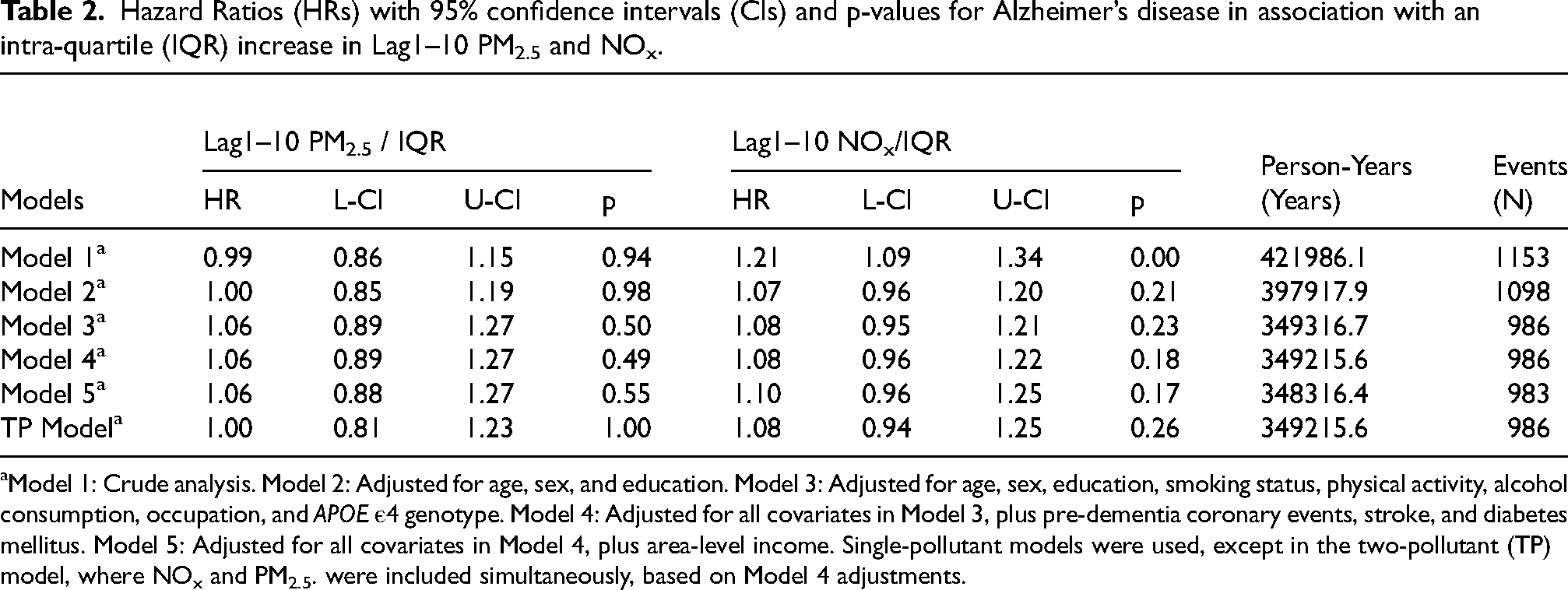
Model 1: Crude analysis. Model 2: Adjusted for age, sex, and education. Model 3: Adjusted for age, sex, education, smoking status, physical activity, alcohol consumption, occupation, and APOE ε4 genotype. Model 4: Adjusted for all covariates in Model 3, plus pre-dementia coronary events, stroke, and diabetes mellitus. Model 5: Adjusted for all covariates in Model 4, plus area-level income. Single-pollutant models were used, except in the two-pollutant (TP) model, where NOx and PM2.5. were included simultaneously, based on Model 4 adjustments.
For VaD (Figure 3, Table 3), HRs for both PM2.5 and NOx were generally close to 1 after adjustment for confounders, indicating no association between pollutant exposure and VaD. There were some indications of mediation by cardiovascular comorbidities in VaD, where the HR for an IQR increase in NOx was 1.05 (95% CI: 0.87–1.27) without adjustment for cardiovascular comorbidities and 1.01 (95% CI: 0.83–1.22) with adjustment. For AD, the estimates seemed unaffected by adjustment for cardiovascular comorbidities.
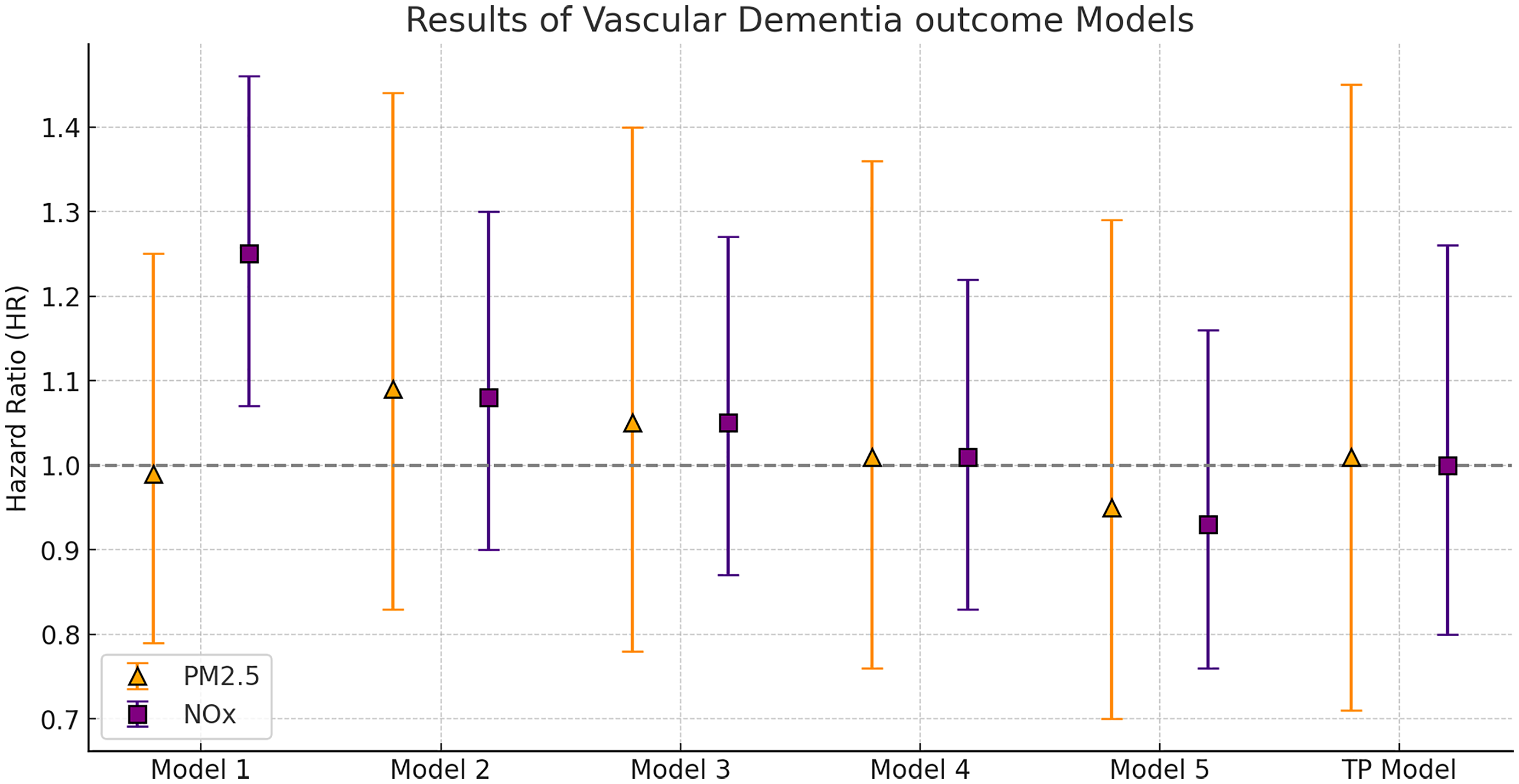
Hazard ratios (HR) for vascular dementia per intra-quartile (IQR) range increase in Lag1–10 NOx and PM2.5. Model 1: Crude analysis. Model 2: Adjusted for age, sex, and education. Model 3: Adjusted for age, sex, education, smoking status, physical activity, alcohol consumption, occupation, and APOE ε4 genotype. Model 4: Adjusted for all covariates in Model 3, plus pre-dementia coronary events, stroke, and diabetes mellitus. Model 5: Adjusted for all covariates in Model 4, plus area-level income. Single-pollutant models were used, except in the two-pollutant (TP) model, where NOx and PM2.5. were included simultaneously, based on Model 4 adjustments.
Hazard ratios (HRs) with 95% confidence intervals (CIs) and p-values for vascular dementia in association with an intra-quartile (IQR) increase in Lag1–10 PM2.5 and NOx.
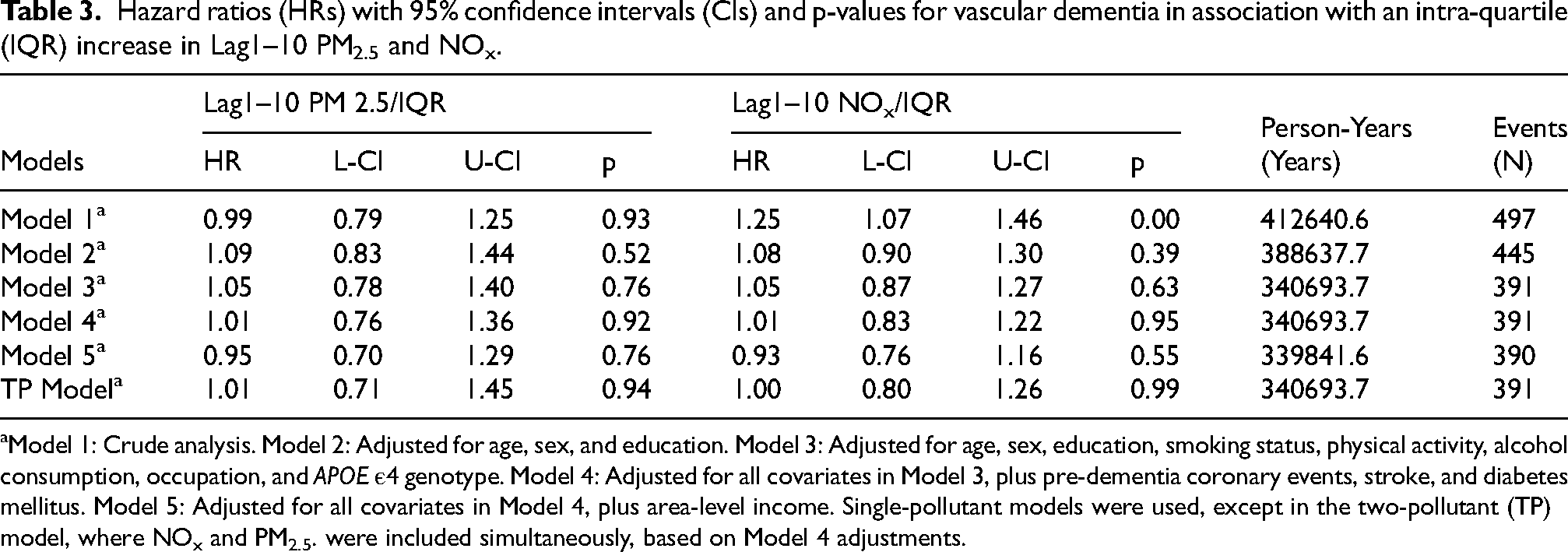
Model 1: Crude analysis. Model 2: Adjusted for age, sex, and education. Model 3: Adjusted for age, sex, education, smoking status, physical activity, alcohol consumption, occupation, and APOE ε4 genotype. Model 4: Adjusted for all covariates in Model 3, plus pre-dementia coronary events, stroke, and diabetes mellitus. Model 5: Adjusted for all covariates in Model 4, plus area-level income. Single-pollutant models were used, except in the two-pollutant (TP) model, where NOx and PM2.5. were included simultaneously, based on Model 4 adjustments.
Furthermore, we performed a sensitivity analysis for BMI, as well as systolic and diastolic blood pressure, which revealed that including them had negligible effects on the results. The HRs for 1 unit increment are presented in Supplemental Material 1. The penalized spline plots (Supplemental Material 2) indicated uncertainty in the estimates particularly at higher concentrations. The Schoenfeld residuals are shown in Supplemental Material 3.
There were some indications that NOx exposure may be more strongly associated with AD among APOE ε4 carriers (HR: 1.10, 95% CI: 0.95–1.28) than non-carriers (HR: 1.06, 95% CI: 0.89–1.27) in Model 4, but the p-values for effect modification did not suggest effect modification (p > 0.3) (see Supplemental Material 4). For PM2.5 in Model 4 we observed potential effect modification, with substantially higher hazard ratios for AD among APOE ε4 non-carriers and for VaD among APOE ε4 carriers. The p-values for interaction were 0.07 for AD and 0.08 for VaD. However, these tendencies were somewhat reduced after adjusting for NOx and the confidence intervals were too wide to be conclusive (Supplemental Material 4). In sex-stratified analyses, no clear differences were observed between men and women in the associations between air pollution and dementia (all p-values > 0.3) (see Supplemental Material 4).
When stratifying the analysis by exposure time windows, we found that the association between NOx and AD was primarily evident for lag 1–5 years (HR: 1.14, 95% CI: 1.00–1.30), whereas the association for lag 6–10 years was less pronounced (HR: 1.04, 95% CI: 0.92–1.17; see Supplemental Material 5 and 6). Regarding VaD, there seemed to be tendencies for decreasing risks, especially for lag 6–10 years, with a HR of 0.87 (95% CI: 0.67–1.12).
Discussion
Key results
We investigated the association between long-term exposure to ambient PM2.5 and NOx and the risk of AD and VaD. We found evidence for an association between NOx exposure AD over the past 10 years (lag 1–10), with the association appearing stronger for exposures in the most recent 5 years (lag 1–5) compared to the earlier period (lag 6–10). For PM2.5, there were tendencies for associations with AD in single-pollutant models, but in two-pollutant models, only the association with NOx remained. In contrast, no clear associations were observed between either NOx or PM2.5 and VaD across any of the models.
Strengths and limitations
This study has several strengths, including its large cohort with comprehensive individual-level data on lifestyle-related confounders and relevant comorbidities, as well as air pollution data with high spatial resolution (50 m × 50 m). Clinically validated dementia subtypes using national registries and DSM-5 criteria minimize diagnostic uncertainty, strengthening validity over studies relying solely on passive registry data. The meticulous assessment of exposure relied on high-quality longitudinal data spanning up to 25 years of recorded residential addresses. However, exposure misclassification remains a challenge in studies of long-term air pollution exposure and health effects. Exposure estimates typically rely on modeled air pollution levels at residential addresses over extended periods, as direct measures are not feasible. This approach often lacks information on occupational exposure or exposure during commuting. Nonetheless, for populations similar to those studied in comparable research, the bias introduced by focusing solely on residential address is likely minimal. 22 Including both single- and two-exposure models (PM2.5 and NOx) addresses a key gap in air pollution research, which often relies on single-exposure analyses. We used a ten-year lag as a proxy for long-term exposure, and we lacked data on life course exposures—which is a critical aspect for studies on air pollution and brain health. 23 The 10-year moving average may obscure distinct critical periods of exposure, such as midlife and late-life, which neuropathological evidence suggests could differentially affect AD development. 24 Our stratified analysis suggested that the association with AD was primarily driven by exposure during the most recent five years, while no clear association was observed for exposures occurring in the five years prior to that, implying the importance of exploring sensitive exposure windows. Another limitation is that we were not able to adjust for traffic noise or lack of greenness. Both these exposures have been suggested to be potential environmental risk factors to cognitive function, but evidence on dementia is still much more limited than for air pollution. In a study conducted in Greater London where multi-pollutant models were feasible, researchers examined air pollutants and noise as separate factors. When both were included in the models, only the association with air pollutants persisted, suggesting that the link between pollutants and health outcomes was more robust than that of noise. 25 This highlights the need for research that investigates these exposures independently as well as in combination to understand their potential contributions to cognitive disorders.
Overall interpretation
While the overall published evidence increasingly supports the link between particulate air pollution, particularly PM2.5, and elevated dementia risk, significant uncertainties remain regarding various dementia subtypes, other pollutants, sensitive time windows, and the mediation effect of cardiovascular disease.10,11,26–28 In the present study, the crude association with VaD was completely explained by potential confounding factors, including cardiovascular pre-existing disease, indicating some mediation and confounding by cardiovascular co-morbidities. Also, for AD, the associations decreased from Model 1, indicating confounding, but seemingly to a lesser extent. The confidence intervals were quite wide and an even bigger cohort with more individuals and events would have been ideal; however, this could be accomplished in meta-analyses, which are increasingly necessary to achieve precise enough results in air pollution epidemiology. Cheng et al. conducted a meta-analysis showing an increase in dementia risk per 10 μg/m³ increase in PM2.5 exposure, 27 where several of the individual studies were inconclusive in themselves. The pooled hazard ratio for a 10 μg/m³ increase in nitrogen dioxide was 1.02 (95% CI: 0.98–1.06) based on nine studies, and for nitrogen oxide, it was 1.05 (95% CI: 0.98–1.13) based on five studies. 11 Furthermore, most previous studies have employed single-pollutant approaches, which could not distinguish the effects from different air pollutants. Our multi-pollutant model with both PM2.5 and NOx suggested that NOx may have stronger associations with AD than PM2.5. However, due to the high correlation between PM2.5 and NOx, these models should generally be interpreted carefully. It may not be possible to disentangle the independent effects of the two pollutants in this dataset, and the results are more appropriately interpreted as reflecting potential effects of traffic-related pollution on AD risk, rather than effects of NOx specifically. Taken together with prior studies on air pollution and mortality within this cohort, also showing associations with NOx rather than PM2.5, 29 may suggest however that NOx may serve as a reliable marker of health-hazardous air pollution in this population, perhaps indicating that local traffic-related air pollutants are more health-hazardous than overall particle mass in this setting.
The mechanisms by which air pollution may contribute to dementia are not entirely understood, but potential pathways include the entry of particulate air pollution into the brain via the bloodstream or the olfactory system. 30 Inhalation of pollutants can trigger systemic inflammation, oxidative stress, and neuronal damage.6,30,31 For VaD, the underlying mechanism may also relate to impaired cardiovascular system. 32 In our study, there were some indications of mediation by cardiovascular comorbidities in VaD as the effect estimates attenuated after including cardiovascular related variables, but the attenuation was small and the confidence intervals were wide. For AD, however, the estimates seemed unaffected by adjustment for cardiovascular comorbidities. Adjusting for area-level socioeconomic factors appeared to have no impact on the estimates for AD. However, the estimates for VaD were somewhat affected, suggesting that area-level socioeconomic factors may capture certain cardiovascular risk factors not accounted for by the individual-level variables included in this study. The findings possibly support the hypothesis that air pollution may influence different types of dementia differently. For AD, it may be through pathways, at least partly, independent of cardiovascular mechanisms.
We observed that the association between NOx exposure and AD was evident only for the most recent five years, with no clear association during the preceding five-year period. This association may reflect an acceleration of pre-existing cognitive decline rather than the initiation of disease. 33 Our findings align with previous studies that have linked traffic-related air pollution to increased dementia risk over similar exposure time windows.34,35 At the same time, others highlighted the importance studies have midlife exposures for dementia onset, 36 emphasizing the need for analyses that account for multiple temporal windows. Further research is needed to clarify these temporal dynamics and their implications for prevention.
It is worth noting that in the shorter lags analyses (lag 1–5 and especially lag 6–10), we observed a tendency toward decreased risks of both AD and VaD in relation to PM2.5 exposure. In two-pollutant models (with possible collinearity issues), the association even reached statistical significance (HR: 0.83; 95% CI: 0.70–0.98), since we consider it unlikely that PM2.5 or any air pollutant would have a protective effect on dementia risk, we report this unexpected finding with caution.
Generalizability
Air pollution consists of a complex mixture of gases, particulate matter, and biological molecules originating from both human and natural sources, and its composition varies significantly across locations and over time. This variation poses challenges to the generalizability of findings from studies on long-term exposure to air pollution and its health impacts. PM2.5, used in this study as a marker for particulate pollution, represents all particles smaller than 2.5 µm in aerodynamic diameter, measured by density. However, PM2.5 does not specifically account for ultrafine particles (UFPs), which could have unique relevance for brain health due to their ability to penetrate biological barriers; their weight is so minimal that they contribute very little to overall PM2.5 mass. In this context, NOx may serve as a more relevant marker for local emissions and ultrafine particle (UFP) exposure, given that NOx in this region is predominantly related to traffic. Future studies should investigate the role of UFP exposure on cognitive health. Despite some limitations, the generalizability of this study aligns with that of similar research in this field. To improve our understanding of air pollution's impact on dementia, it remains crucial to conduct studies across diverse populations and exposure levels. For instance, more research is needed to capture effects at the upper end of the exposure-response curve 4 and to address the lack of data from low- and middle-income countries 37 and it is not evident that the results from the present study can be generalized to such settings. The cohort's older age and relatively low pollution exposure may limit the generalizability of our findings to younger populations or those living in areas with higher pollution levels.
Conclusion
In this study, we examined the association between long-term exposure to ambient PM2.5 and NOx and the risk of AD and VaD. We found consistent evidence linking NOx exposure to increased AD risk over the past 10 years (lag 1–10), with the association being more pronounced for exposures in the most recent 5 years (lag 1–5). While PM2.5 showed a potential association with AD in single-pollutant models, this did not persist in two-pollutant models, where only the association with NOx remained. No clear associations were found between either pollutant and VaD. These results highlight the need for improved exposure assessment, dementia subtype-specific analyses, and further investigation into critical exposure windows in air pollution and cognitive health research.
Supplemental Material
sj-docx-1-alz-10.1177_13872877251360225 - Supplemental material for Long-term exposure to air pollution and validated cases of Alzheimer's disease and vascular dementia in the Malmö diet and cancer cohort
Supplemental material, sj-docx-1-alz-10.1177_13872877251360225 for Long-term exposure to air pollution and validated cases of Alzheimer's disease and vascular dementia in the Malmö diet and cancer cohort by Abdullah-J Alzhrani, Leo Stockfelt, Yiyi Xu, Florencia Harari, Susanna Gustafsson, Gunnar Engström, Oskar Hansson and Anna Oudin in Journal of Alzheimer's Disease
Supplemental Material
sj-docx-2-alz-10.1177_13872877251360225 - Supplemental material for Long-term exposure to air pollution and validated cases of Alzheimer's disease and vascular dementia in the Malmö diet and cancer cohort
Supplemental material, sj-docx-2-alz-10.1177_13872877251360225 for Long-term exposure to air pollution and validated cases of Alzheimer's disease and vascular dementia in the Malmö diet and cancer cohort by Abdullah-J Alzhrani, Leo Stockfelt, Yiyi Xu, Florencia Harari, Susanna Gustafsson, Gunnar Engström, Oskar Hansson and Anna Oudin in Journal of Alzheimer's Disease
Supplemental Material
sj-docx-3-alz-10.1177_13872877251360225 - Supplemental material for Long-term exposure to air pollution and validated cases of Alzheimer's disease and vascular dementia in the Malmö diet and cancer cohort
Supplemental material, sj-docx-3-alz-10.1177_13872877251360225 for Long-term exposure to air pollution and validated cases of Alzheimer's disease and vascular dementia in the Malmö diet and cancer cohort by Abdullah-J Alzhrani, Leo Stockfelt, Yiyi Xu, Florencia Harari, Susanna Gustafsson, Gunnar Engström, Oskar Hansson and Anna Oudin in Journal of Alzheimer's Disease
Supplemental Material
sj-docx-4-alz-10.1177_13872877251360225 - Supplemental material for Long-term exposure to air pollution and validated cases of Alzheimer's disease and vascular dementia in the Malmö diet and cancer cohort
Supplemental material, sj-docx-4-alz-10.1177_13872877251360225 for Long-term exposure to air pollution and validated cases of Alzheimer's disease and vascular dementia in the Malmö diet and cancer cohort by Abdullah-J Alzhrani, Leo Stockfelt, Yiyi Xu, Florencia Harari, Susanna Gustafsson, Gunnar Engström, Oskar Hansson and Anna Oudin in Journal of Alzheimer's Disease
Supplemental Material
sj-docx-5-alz-10.1177_13872877251360225 - Supplemental material for Long-term exposure to air pollution and validated cases of Alzheimer's disease and vascular dementia in the Malmö diet and cancer cohort
Supplemental material, sj-docx-5-alz-10.1177_13872877251360225 for Long-term exposure to air pollution and validated cases of Alzheimer's disease and vascular dementia in the Malmö diet and cancer cohort by Abdullah-J Alzhrani, Leo Stockfelt, Yiyi Xu, Florencia Harari, Susanna Gustafsson, Gunnar Engström, Oskar Hansson and Anna Oudin in Journal of Alzheimer's Disease
Supplemental Material
sj-docx-6-alz-10.1177_13872877251360225 - Supplemental material for Long-term exposure to air pollution and validated cases of Alzheimer's disease and vascular dementia in the Malmö diet and cancer cohort
Supplemental material, sj-docx-6-alz-10.1177_13872877251360225 for Long-term exposure to air pollution and validated cases of Alzheimer's disease and vascular dementia in the Malmö diet and cancer cohort by Abdullah-J Alzhrani, Leo Stockfelt, Yiyi Xu, Florencia Harari, Susanna Gustafsson, Gunnar Engström, Oskar Hansson and Anna Oudin in Journal of Alzheimer's Disease
Footnotes
Acknowledgements
We sincerely thank Mr Hussain Jaafari and Mr Daniel Oudin Åström for their valuable support and assistance with R software coding which has been instrumental in this work.
Ethical considerations
The study was approved by the Swedish Ethical Review Authority (dnr 2021–05548-01), with the project name “Urban brain” (Studies of environmental exposures in relation to mental and cognitive health).
Consent to participate
All participants provided written consent.
Author contributions
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Umm Al-Qura University provided financial support for Abdullah Alzhrani's PhD study, covering tuition fees and salary. The sponsor had no role in the study design, data collection, analysis, interpretation, or manuscript preparation.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: OH is an employee of Lund University and Eli Lilly, and he has previously acquired research support (for Lund University) from AVID Radiopharmaceuticals, Biogen, C2N Diagnostics, Eli Lilly, Eisai, Fujirebio, GE Healthcare, and Roche. In the past 2 years, he has received consultancy/speaker fees from Alzpath, BioArctic, Biogen, Bristol Meyer Squibb, Eisai, Eli Lilly, Fujirebio, Merck, Novartis, Novo Nordisk, Roche, Sanofi and Siemens. The remaining authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
The data supporting the findings of this study are available on request from the corresponding author, provided the steering group of MDC's approval. The data are not publicly available due to privacy or ethical restrictions.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
