Abstract
Background
Cerebrospinal fluid biomarkers can be used to diagnose biological Alzheimer's disease (AD). The psychological safety of this approach is often questioned given the paucity of effective therapies for AD.
Objective
We wanted to evaluate the psychological impact of biomarker assisted diagnosis on patients and their caregivers.
Methods
Using a mixed method design, 10 patients and 16 caregivers were evaluated before and after receiving a diagnosis of AD with this technique. Interviews were conducted to explore their perspectives. Questionnaires were used to evaluate the effects on quality of life, depression and caregiver burden.
Results
Participants mentioned themes like having an objective explanation for their symptoms, being able to act on the information, finding lumbar puncture invasive, and receiving a difficult diagnosis with little possible action. Most participants said they would recommend the procedure. Measures of quality of life, depression and caregiver burden were similar before and after disclosure.
Conclusions
This study suggests that, from a psychological perspective, cerebrospinal fluid-based diagnosis of AD could be offered to patients after discussion of potential benefits and risks. Further research is needed in this field, especially as new diagnostic methods become more available.
Keywords
Introduction
Alzheimer's disease (AD) is the leading cause of dementia, a condition with often devastating impact on patients and their families. Currently, well over 50 million people worldwide live with dementia, a rapidly growing prevalence that is expected to keep increasing in the future. 1
Until recently, the diagnostic conclusions of AD based on clinical evaluation was either probable or possible. Definite diagnosis of AD was only made with histopathological evidence. 2 Technological and medical progress has led to the creation of tests that detect biomarkers of the disease, allowing for precise diagnosis. This is currently possible through cerebrospinal fluid (CSF) analysis obtained by lumbar puncture (LP), or by use of specific positron emission tomography (PET) markers to tau or amyloid protein deposition. These techniques were first used as research criteria, 3 but are now entering clinical practice with appropriate use criteria having been devised.4–6 There is also emerging research into using serum phosphorylated tau protein levels to make a diagnosis. However, the transition from research to clinical practice for this specific technique is even more recent. While some studies suggest that its performance can be similar to invasive biomarkers, there are nonetheless significant challenges to accessibility. 7
Many clinicians are still reluctant to use these techniques to aid in the diagnosis of their patients. While it is more widely adopted in some European countries, it remains seldom used by clinicians in many other regions, such as North America. 4 There can be challenges in obtaining access to these tests, especially with PET. As for LP, it can be seen as an invasive option. In addition, some ethical concerns have been raised. The right to know or not to know, associated uncertainty with prognosis, potential social consequences, and possible negative emotional reactions are all cited as concerns. 8
There is also doubt cast upon the utility of such a diagnosis. A possible use of CSF analysis is to classify the patient according to the AT(N) classification, 9 which could provide some information on diagnosis and prognosis. 10 Regarding treatment, at the time of writing, options are very limited and mostly symptomatic. 8 The only approved disease-modifying treatments for AD are aducanumab and lecanemab, for which the efficacies are limited and debated.11,12 Furthermore, these treatments are not accessible in many countries and can only be given to a limited number of patients who meet specific criteria. It is worth noting, however, that this may change in the future, with numerous clinical trials underway for other disease-modifying treatments. 13
Some studies have sought the perspective of patients, but the majority of these were done in research settings, and almost all were done using amyloid PET. These studies were done in populations varying from cognitively normal patients enrolled in clinical research to mild cognitive impairment or dementia patients. A 2016 systematic review found no changes in anxiety or depression levels, but did report increased test related distress and changes in behavior related to insurance and health. 14 The studies included in this review were limited to cognitively normal individuals, and most of them looked at disclosure of apolipoprotein E genotype. In 2017, a qualitative study was conducted to investigate the experience of amyloid imaging disclosure results. It included 38 patients, eight of them having a positive result, and found that participants cited advantages such as increased information about their health, the ability to make arrangements based on the result and to take medication, improved relationships and greater enjoyment of life. They cited disadvantages such as emotional difficulties, risk of patronizing attitudes by relatives, and worry over worsening of their symptoms. 15 Another review evaluated two prospective studies on the disclosure of PET amyloid results and found the disclosure to have a low risk of psychological harm. 16 This same review also examined a few non-prospective studies, including some of the ones cited above, and found similar results. A 2020 study conducted on cognitively normal individuals, with subjective cognitive decline, found no worsening of depression or anxiety scores after disclosure of amyloid SPECT results. 17 A prospective cohort study from 2018 in symptomatic individuals showed no worsening of anxiety and depression after disclosure of PET results. 18 A qualitative study with 26 patient-caregiver dyads used semi-structured interviews to inquire about their experience with amyloid imaging. They included participants who did not undergo imaging and those with a negative result. 19 Important themes that emerged included having more diagnostic information, the ability to make treatment and life decisions based on the results, barriers to obtaining the test such as lack of insurance coverage, and the lack of benefit or change to care it would provide. Reactions included relief, feeling upset/depressed, satisfaction, anxiety, validation and shock.
A general limitation of these studies is that most examined the effect of disclosure on asymptomatic, cognitively normal patients, 20 which has less clinical applicability. 4 In general, disclosure of the results for these forms of biomarkers were deemed safe and had low risk of psychological harm.16,21 However, there is little known about the psychological effects that diagnosing AD, specifically through CSF analysis, can have on patients and their surroundings. Furthermore, relatively few of the above studies included caregivers.
The concepts of psychological harm and safety have previously been used in the evaluation of the impact of diagnosis of AD. While there are a multitude of aspects that could be considered, frequently measured aspects included symptoms of depression or anxiety, and test related distress.16,20,22
There are advantages of CSF analysis over PET, such as greater accessibility and better cost-effectiveness. 21 Considering these factors, our study sought to explore the psychological effects and patient experience of receiving an AD diagnosis through analysis of CSF obtained by LP. Caregivers were also included in this study given their important role in caring for patients with AD.
Given previous research into the subject of using alternative methods, we hypothesized that participants would generally appreciate the opportunity for more precise diagnosis and would not suffer major adverse events from it.
Methods
Recruitment
Participants were recruited in two early onset dementia clinics, managed respectively by L.C.W. and S.A.-S. Patients had been referred to this clinic, usually from a primary care provider, because of a clinical concern in cognitive decline. Both patient and a designated caregiver were invited. For members of a dyad to be eligible, the patient had to have done, or have scheduled, a LP to help determine if they have AD. They also had to be over 18 years of age. To keep the scope of this investigation more focused, potential participants were excluded if the patient had received a diagnosis of dementia other than AD. Due to the difficult nature of recruiting patients who are already in a state of cognitive decline by the time they are referred to the dementia clinics, participants were included no matter the degree of cognitive impairment. However, in cases where patients were unable to complete the patient portion of the questionnaires and interview due to advanced dementia, their caregiver was still invited to participate. Participants signed informed consent forms, and this study was approved by a local research ethics committee, the Vitalité Health network Ethics Office. The approval number was 101119. Recruitment began in October 2020 and ended in July 2023.
Study design
This mixed methods study was done prospectively when possible, and retrospectively in other cases. When patients had not yet received a result from their LP, participants were recruited for two data collection sessions: one before the disclosure and another after the disclosure. When they had already received the results, they only completed the post-disclosure session. Both groups were included in the study to allow their point of view to be heard, which could provide important additional data, especially in the qualitative sections. In all cases, counselling was done by the attending physician (S.A.-S., L.C.W.) before patients made the decision to undergo LP. This also included information on the reason of the procedure and the procedure itself, including the potential risks, such as hematoma formation, headache, and infection. A consent was signed with the patient and their power of attorney if needed. Benefits discussed included the AD biomarker analysis, which was presented as an additional source of information that could solidify the suspected diagnosis, and in some cases, ruling out alternative diagnoses with the same sample. The diagnosis of AD was made clinically by the attending physician by considering the results of neuropsychological testing, patient history, physical examination findings, results of brain MRI and brain SPECT CT scan for some patients and the biomarkers findings (level of Aβ42, 181 phosphorylated tau protein, and total tau protein in the CSF). It was then communicated as such to the patients and caregivers.
The data collection sessions included a semi-structured interview (Table 1), as well as two questionnaires per participant. Patients completed the Quality of Life in Alzheimer's Disease (QoL-AD) scale 23 and the Geriatric Depression Scale: Short Form (GDS), 24 while caregivers completed the QoL-AD (by proxy) 23 and the Zarit Burden Interview (ZBI). 25 Together, these quantitative elements were used to measure some of the adverse psychological symptoms that could be considered as harm, helping to evaluate psychological safety. Participants would then also have the opportunity to report any other symptoms or feelings during the qualitative portion. All portions of the interview were conducted regardless of whether it was pre-disclosure or post-disclosure. Interviews were conducted by two investigators, A.L. and S.E. Interviews were conducted in French or English, according to patient preference.
Semi-structured interviews.
Participants
A total of 26 participants completed at least a part of the study (see Table 2). Except for one patient/caregiver dyad, everyone to which the study was proposed accepted to participate. There were also some dyads where only the caregiver accepted or was able to participate, which explains the comparatively higher amount of caregiver participants. One dyad chose to withdraw from the study before completing the post-disclosure interview. Another patient was excluded for having negative biomarkers. The post disclosure interviews were conducted at an average of 1.1 years and a median of 6 months after disclosure, and they were all at least 10 days after the disclosure.
Participants characteristics.

MMSE: Mini-Mental State Examination (scores range from 0 to 30); s: standard deviation.
Statistical power calculation
A power analysis was conducted to determine the necessary sample size to detect a statistically significant difference between pre-disclosure and post-disclosure QoL-AD scores. The power analysis was aimed towards the QoL-AD score as it was the only measure that included both groups of participants. Given the absence of previously published studies investigating the outcome of QoL-AD scores in this context, we conducted a power calculation based on the first participants of the study. A difference of 11% between pre-post QoL-AD scores was observed after data was collected from 4 participants. Though not statistically significant, a difference of this degree on the QoL-AD measurement has some clinical relevance. Using this 11% difference in QoL-AD score, a sample size of 14 total participants (combining patient and caregivers) having completed both interviews was found to be necessary to achieve a power of 80% to identify a statistical difference with two-sided 95% confidence intervals in this score.
Data analysis
Qualitative data
During the interviews, key ideas were written down. When possible, interviews were also recorded, which allowed completion of interview notes. We chose to use thematic analysis, as it allows to identify themes and how they relate to the research question. Similar ideas were gathered together, then they were combined into various themes. Through an iterative process, themes were merged together if the new theme encompassed all included ideas well. This was done until it was felt that each theme was truly distinct. This analysis was performed by two of the investigators (A.L. and S.E.). To increase validity, both investigators had to reach a consensus during each step of this process.
Quantitative data
The scoring method for each measure was followed as per questionnaire guidelines.24,26,27 As recommended, missing answers were replaced by the average from the other answers, so long as at least 75% of the questions had been answered. Wilcoxon signed-rank tests were used to compare within group difference pre and post LP disclosure. Only data from participants that had participated in both interviews was included in pre-post comparative analyses. Wilcoxon rank-sum tests were also used to compare between patient and caregiver reported quality of life post disclosure. All analyses were conducted using SAS 9.4 (SAS Institute, Inc., Cary, NC, USA).
Results
Qualitative results
Advantages
By a wide margin (Table 3), the most cited advantage was to simply know with more certainty that AD was the cause of their symptoms. One patient explained it by saying that “it wouldn’t have been a good idea to not know. Just to be there and wonder, . . . what do I have?” Another said, “It felt good, in a way, to have the answer, because I don’t know what I would have done if I hadn’t had it.” Additionally, some patients felt having a LP was useful to rule out other diseases, such as rapidly progressive dementias like Creutzfeldt-Jakob disease, or curable causes to their symptoms: “he was hoping to rule out CJD, […] he was hoping to find inflammation or an infection, something he could cure.” In general, participants liked the increased certainty that CSF analysis provided to the diagnosis of AD.
Frequency of themes in qualitative questionnaire responses.

Participants also mentioned the ability to act upon the diagnosis of AD. For some, this meant the ability to start certain treatments that are known to be beneficial in AD. Others mentioned the ability to better prepare for the future with this information, like starting to consider measures to have a place available in a care home in the future. One participant explained it by saying: “Once we knew what it was, we could try, as a family, to prepare for the future, to get things in place.” Some participants also saw benefit in that it would help them contribute to AD research, including, but not limited to, therapeutic research. Finally, some explained that it had pushed them to spend more quality time with their loved ones.
A few participants said that having a diagnosis of AD gave them the possibility of having genetic counselling to see if other family members might be affected in the future, especially in the case of early-onset Alzheimer's: “[…] then you could find out if it is genetic, if it was going to be affecting the rest of her family.”
For some participants, knowing that their symptoms were caused by AD also helped them gain a better understanding of their prognosis and their expected disease course. As mentioned above, this knowledge helped some individuals make important life decisions and care related plans based on this concrete information: “This helped her come to terms with the diagnosis, and then she could figure out what the next steps were.”
Disadvantages
Many of the disadvantages named by patients were related to the LP itself, and side effects/complications they had afterwards. While there were no major complications in the study group, it was felt by most to be an invasive procedure, with one summarizing it as “well it wasn’t pleasant.”
Some mentioned how this was a difficult diagnosis to receive, with few possible actions available to them as a result. A caregiver explained how “it doesn’t change anything about her disease, there is no cure […]. She knows she has the disease and wants to be reminded of it as little as possible. It's kind of like twisting the knife in the wound.” Some participants then also mentioned how they found there was a lack of resources to support them in living with AD, both for patients and caregivers: “After that it was really up to us to figure out what to do.”
Finally, some mentioned how it had changed their plans for retirement, and they had given up on certain projects upon receiving the diagnosis.
Quantitative results
Recommendation of the procedure
We asked participants if they would recommend the test to a friend or relative if they found themselves in a similar situation. All patients (n = 11) and 93.8% (15 of n = 16) of caregivers said yes.
Quality of life
Overall, we observed no statistically significant differences when comparing quality of life scores before and after disclosure, including within subgroup analysis for patients or caregivers, respectively (Table 4). Looking at post-disclosure results, there was a significant difference between patient and caregiver's report of patients’ quality of life, with patients reporting higher scores than caregivers (38.7 compared to 30.9, p = 0.01). No such difference was observed in pre-disclosure results (36.7 versus 33.5, p = 0.42) (Figure 1).
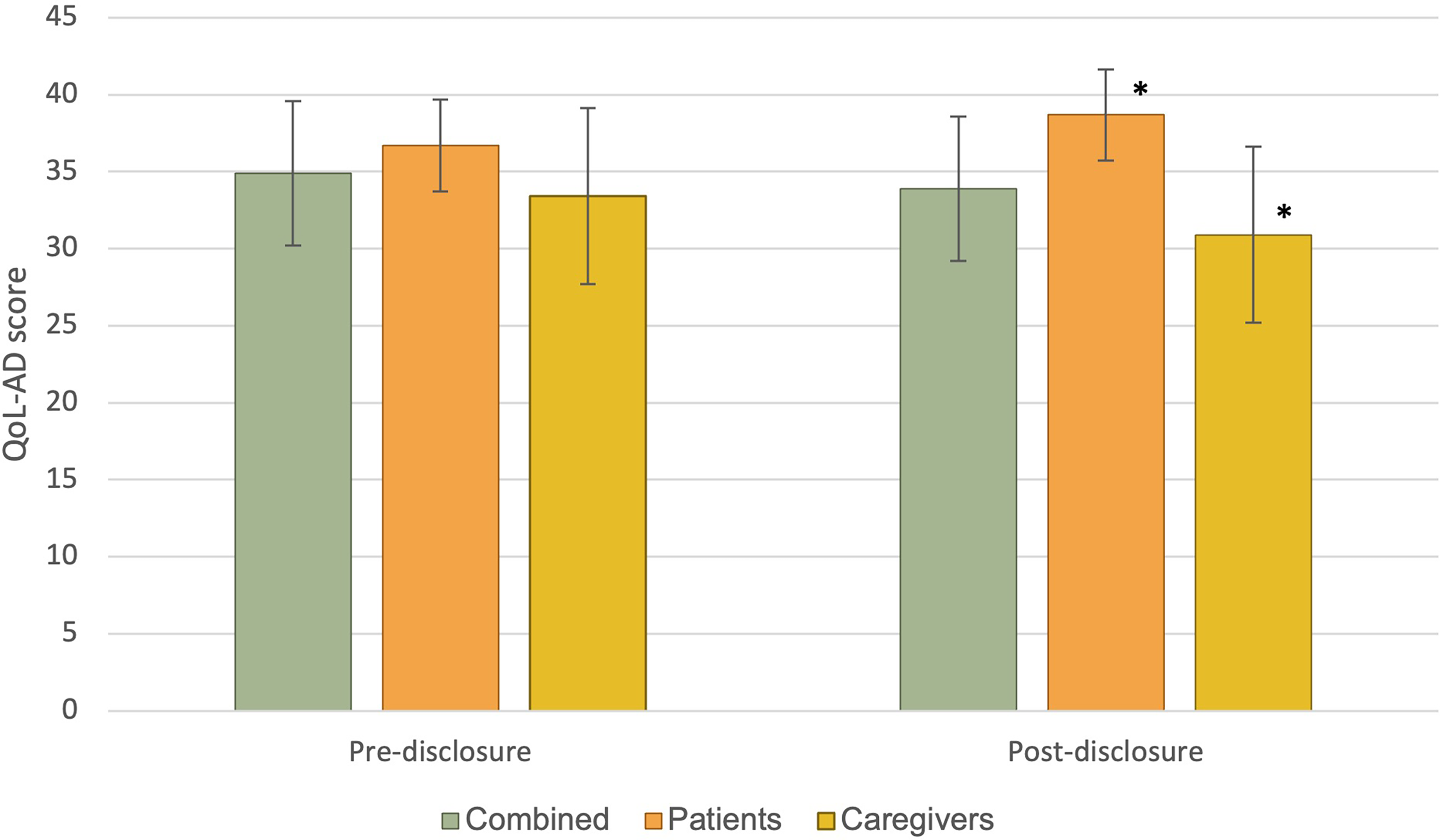
QOL-AD scores. Average scores of quality of life as measured by the Quality of Life in Alzheimer's Disease instrument, with possible scores ranging from 13 to 52. Error bars represent standard deviation. Caregivers scores represent the caregiver's estimation of the patient's quality of life. * Statistically significant difference between these two groups.
Questionnaire results.
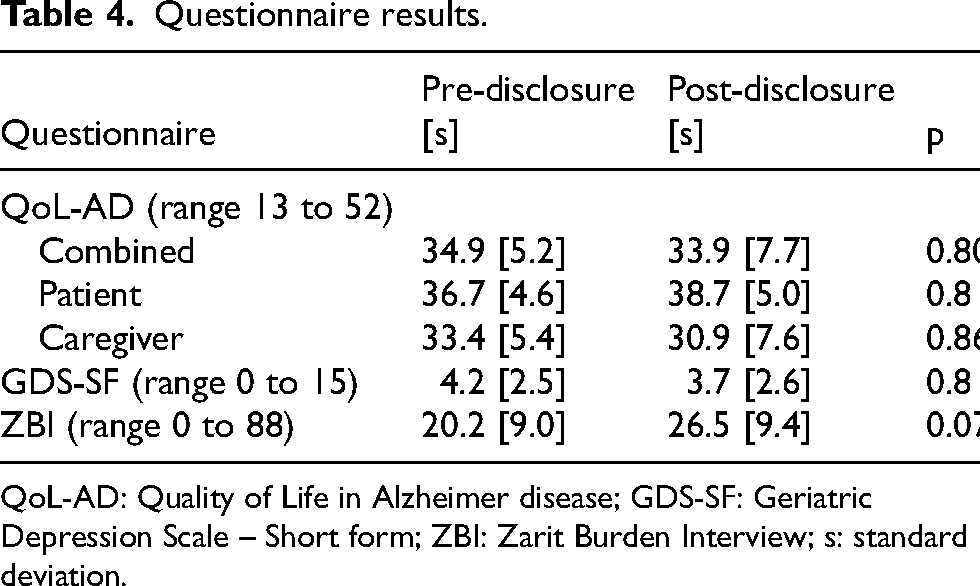
QoL-AD: Quality of Life in Alzheimer disease; GDS-SF: Geriatric Depression Scale – Short form; ZBI: Zarit Burden Interview; s: standard deviation.
Patient depression symptoms
There was no significant difference between Geriatric Depression Scale (short form) results when comparing before and after the disclosure (Table 4).
Caregiver burden
When comparing the scores of Zarit Burden Interview before and after disclosure, there was no statistically significant difference (Table 4). There was, however, a non-significant trend towards higher caregiver burden in the post-disclosure group.
Discussion
Our study found that participants appreciated LP for its ability to provide a more objective diagnosis, for the actions that could be taken with the results, and to obtain prognostic information. It tended to have high rates of recommendation among participants and caregivers, and we did not detect any statistically significant impact on quality of life, depression symptoms or caregiver burden, as will be discussed later. Conversely, participants had less appreciation for the procedure itself, how it did not change their disease course, and how it had caused them to not follow through on plans they would have otherwise.
Our qualitative findings on the impact of diagnosis disclosure on patients and caregivers are relatively similar to those described in the literature on the diagnosis by PET and in theoretical ethics papers.28,29 A notable difference from PET is the frequent mention of the invasiveness associated with LP. Though it was generally well tolerated, and has low rates of complications, 30 it was mentioned by many patients as unpleasant. This highlights the importance of increasing availability and affordability of already existing modalities such as PET, along with the development of less invasive technologies, such as blood-based testing.
There are significant limitations to the quantitative analyses we performed. The lack of pre-existing data on QoL-AD scores before and after diagnosis of AD made it impossible to perform a traditional power analysis to determine an adequate sample size. The method used, to base an analysis on the data observed after the first few patients of the study, is probably not as rigorous and reliable. Given the lack of observed difference, we cannot rigorously conclude that there is no effect, rather it is quite possible that our sample was too small to detect one. The limited ability to conclude a lack of effect also applies on our other analyses, such as the comparisons of depression scores and caregiver burden, as they did not have power analyses.
There were high rates of recommendation of LP for diagnosis of AD among our participants, but there is significant selection bias present with this finding. All the patients included in the study had initially made the choice to undergo LP. Nevertheless, it shows that people who made that choice are unlikely to regret it upon learning the results. Notably, all patients reported that they would recommend the procedure, with the only participant not recommending it being a caregiver. This gives some support to the idea that a CSF-based diagnosis of AD is appropriate to be offered to patients after a thorough discussion about its benefits and drawbacks.
We did not detect a statistically significant difference in scores of quality of life, depression, or caregiver burden when comparing the pre-disclosure and post-disclosure cohorts. However, as outlined below, it is difficult to draw significant conclusions from this.
There was an imbalance between the number of patients and caregivers that were recruited, with more of the latter having participated. In some cases, patients simply did not wish to participate, and in a few cases, patients had cognitive decline that was too advanced for them to be able to participate. While this difficulty is inherent to voluntary participation in research and to research in AD populations, it does limit our insight into the perspective of patients themselves. This is further highlighted by our finding of significantly higher quality of life score reported by patients compared to caregivers in the post-disclosure interviews. This may in part be related to anosognosia, causing patients to perceive less of an impact from their symptoms than their caregivers, who would then see the patients’ quality of life as poorer. However, interpretability of this finding is limited, as it was not our primary analysis. It does, however, allow us to hypothesize that it is important to seek the opinions of patients directly, not only caregivers, when conducting studies on quality of life in patients with AD.
There were some methodological limitations that are worth discussing. There was significant difficulty in recruitment, as this remained a relatively low-volume diagnostic tool in our setting. This became even more true near the end of our study period, as our center obtained increased availability to other diagnostic methods like 217-phosphorylated tau serum testing as part of some clinical trials. This prevented us from recruiting more patients than what was calculated to be strictly necessary by our power analysis. Given the limited reliability of this analysis, it prevents us from drawing significant conclusions from our quantitative analyses, as previously discussed. The difficulty in recruitment also prevented us from only accepting participants that would be able to participate in both pre-disclosure and post-disclosure interviews, as we had to recruit participant who had already had the disclosure in the past. We included such participants as we felt their perspective remained valuable, but it also created a significant variability as to the timing of the post-disclosure interview after the disclosure.
The strengths of this study include the use of a mixed methods approach. Questionnaire obtained data allowed for a quantitative comparison between key measures, while the qualitative data provided invaluable insights into the unique experiences and perspectives of both patients and caregivers seeking a definitive diagnosis of AD via the LP procedure. It also addresses a significant gap in literature, looking at the experience of participants specifically with LP assisted diagnosis. It also looked at participants who were symptomatic, contrarily to some past studies that looked at asymptomatic research participants. There are difficulties inherent to performing studies in patients symptomatic of AD, and their inclusion in this research is a strength.
While our quantitative data does not allow any definitive conclusions about the psychological safety of precise AD diagnosis using CSF analysis, the qualitative interview data along with the high rates of recommendation support the notion that it is reasonable to offer this to willing patients, after discussion of the relevant risks and benefits. This conclusion is largely consistent with the findings of other studies into the psychological impact of biomarker assisted diagnosis of AD, which have mostly evaluated other modalities. 16 While some of the participants’ comments were related to the LP itself, the majority were about the precise diagnosis of AD. Further, the LP was generally seen as a negative aspect of the process. Development into less invasive diagnostic modalities, including increasing the availability of existing modalities like PET, would be a potential way to reduce the negative aspects of precise diagnosis of AD. Serum-based biomarkers, as they are developed further, present an opportunity for accessible, accurate, and non-invasive diagnostic aid. This could have a significant impact on the experience of patients and caregivers and should be studied specifically, as this less invasive experience could significantly alter the psychological impact. Furthermore, as these become more available, the ease of collecting serum samples could make their use more widespread. They should be used cautiously as they also have the potential for significant psychological impact. They are probably best reserved for use in teams with specific training and expertise in neurodegenerative disease.
More research is needed on the psychological effects of diagnosing AD with biomarkers, especially as new modalities for diagnosis and treatment emerge. Larger, more diverse cohorts are needed. Future research should consider recruiting patients from multiple centers. Longer term longitudinal follow-up is also necessary to evaluate the ongoing impacts of living with a precise diagnosis of AD. Research should also be done about counselling patients and caregivers on biomarker assisted diagnosis of AD. Protocols could incorporate information from this and other similar studies.
Footnotes
Acknowledgements
We would like to thank Vitalité Health Network and the Centre de formation médicale du Nouveau-Brunswick for their support.
Ethical considerations
This research project has received approval from a research ethics committee, the Vitalité Health network Ethics Office. The approval number was 101119.
Consent to participate
All participant provided informed written consent.
Author contribution(s)
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: A grant from the Centre de Formation médicale du Nouveau-Brunswick was provided to one author (A.L.) as compensation for their work. They played no role in the conduct of the research or the preparation of the article.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
The data supporting the findings of this study can be made available upon request to the corresponding author. It has not been made publicly available due to privacy concerns.
