Abstract
Background
Skeletal muscle weakness and mild cognitive impairment (MCI) commonly occur with aging.
Objective
We examined whether presence of chronic morbidities in MCI is associated with specific alterations in muscle health, functional capacity, and whole body amino acid kinetics.
Methods
A group of 247 older adults were stratified into MCI/non-MCI (Montreal Cognitive Assessment) and presence/absence of chronic diseases. We measured lean mass by dual-energy x-ray absorptiometry, strength by dynamometry, and functional capacity by 6-min walk test. Postabsorptive whole body production (WBP) of amino acids were assessed by pulse administration of a mixture of 18 amino acid stable isotopes.
Results
MCI was associated with lower lean mass, functional capacity (p < 0.003), and WBP of arginine, glycine, leucine, and phenylalanine to tyrosine conversion (reflecting net protein breakdown (net PB)) but higher WBP of taurine (all p < 0.05). Presence of chronic morbidities was associated with lower muscle strength, WBP of glycine, and net PB (p < 0.0001), but higher WBP of phenylalanine, glutamate, taurine, tryptophan, and leucine (all p < 0.05). MCI*chronic morbidity interactions were found for muscle strength and net PB (p < 0.0001), with the lowest values in MCI with chronic morbidities.
Conclusions
Presence of MCI and chronic morbidities in the older population affect different markers of muscle health and functional decline. Individuals with both MCI and chronic morbidities are at increased risk for severe muscle weakness likely related to a severe downregulation of glycine production and net protein breakdown. Therefore, it is important to consider the presence of chronic morbidities when investigating muscle health and functional capacity in MCI.
Keywords
Introduction
Approximately 49% of individuals with mild cognitive impairment (MCI) have been diagnosed with at least one chronic morbidity, including but not limited to chronic obstructive pulmonary disease (COPD) and diabetes.1–8 It is also known that older adults with one or more of these chronic morbidities are at an elevated risk for MCI. 9 More research in needed in this group of older adults as presence of MCI and chronic morbidities are independently associated with an increased risk of frailty and poor muscle health (muscle loss and weakness), due to factors including malnutrition, physical inactivity, and low social activity.1,6,10,11 We and others have found that poor muscle health in these groups of older adults is indicative of disturbances in protein and amino acid metabolism.6,10,12–14
Older adults with dementia were previously found to have lower valine concentrations compared to those without dementia.15,16 Interestingly, plasma concentrations of all branched-chain amino acids (BCAAs: valine, leucine, and isoleucine) were decreased in MCI patients whose cognitive decline progressed to Alzheimer's disease (AD). 17 Previously, we found lower plasma concentrations of leucine and isoleucine in COPD patients with depression compared to those without depression. 18 As the BCAA also play an important role in muscle building and repair, particularly MCI patients with chronic morbidities are at high risk for perturbations in BCAA metabolism and poor muscle health. Lower plasma glutamate to glutamine concentration ratio were also found in MCI as compared to individuals with normal cognitive function. 19 Imbalances in this ratio have been related to neuronal death. 19 We previously observed increased glutamine concentrations and conversion rate from glutamate to glutamine in COPD patients, 20 and elevated whole body glutamine production rates in chronic morbidities with presence of or at risk for cognitive dysfunction.6,14,18,21,22 We hypothesize that presence of chronic morbidities in MCI patients is associated with a further deregulation of their glutamate and glutamine metabolism.
We and others also have found decreased plasma concentration of tryptophan, a precursor of serotonin, in older adults with MCI 23 and in those with chronic morbidities, such as COPD. 6 Low plasma tryptophan concentration has been associated with poor cognitive and skeletal muscle function. 6 Both tryptophan and the BCAAs are essential amino acids that are primarily responsible for upregulating protein synthesis in both healthy and diseased populations. 24 This suggests that particularly MCI patients with chronic morbidities are at risk for low tryptophan concentrations, negatively affecting both muscle and brain health. The amino acid glycine is also known to be an important amino acid for cognitive and skeletal muscle health.25–27 Recent clinical trials have shown better cognitive function in AD patients with the highest plasma glycine concentration. 28 Furthermore, we have found lower glycine production to be associated with increased skeletal muscle strength in COPD. 29 More research is needed whether MCI patients with chronic morbidities have low plasma glycine concentrations due to an inadequate upregulation of its production rate, negatively affecting their muscle and brain health.
As the impact of chronic morbidities on skeletal muscle health, functional capacity, and amino acid metabolism remains unclear in older adults with MCI, the aim of the present study was to address this knowledge gap by determining if the presence of chronic morbidities in MCI is associated with a further dysregulation in metabolism and function or whether specific (MCI by chronic morbidity) interaction effects are present. We hypothesize that presence of chronic morbidities in MCI results in more severe amino acid metabolic perturbations, particularly of the BCAA, glutamate/glutamine, tryptophan and glycine, contributing to a further decline in skeletal muscle health and quality of life in older adults with MCI. We characterized a large group of MCI individuals with and without chronic morbidities for their nutritional, functional, and amino acid metabolic phenotype as compared to their non-MCI counterparts. We utilized a multi amino acid stable isotope pulse technique20,30 to assess the specific perturbations in plasma concentrations as well as whole body metabolic production rates of amino acids. This information is important to further guide targeted nutritional approaches to attenuate cognitive decline and improve nutritional and functional status by restoring specific amino acid perturbations in older adults with MCI with and without chronic morbidities.
Methods
Study population
We analyzed the data of 247 older adults (age ≥50y) with and without MCI who participated in the Metabolism of Disease with Isotope Tracers (MEDIT) trial between 2013 and 2022 (Supplemental Figure 1). MEDIT is a large and still ongoing controlled trial enrolling subjects with and without various chronic morbidities. All subjects in MEDIT are characterized by in-depth analysis of skeletal muscle mass and function, body composition, and comprehensive metabolic phenotyping using novel stable isotope pulse techniques. In the current study, baseline values of multiple clinical trials (NCT02082418, NCT02770092, NCT02780206, NCT03327181, NCT03796455, NCT04461236) were included using as primary selector the previous completion of a Montreal Cognitive Assessment (MoCA), 31 and as secondary selector the previous assessment of Charlson Comorbidity Index Score (CCIS). 32 CCIS and medical history was assessed by physical examination, medical interview, and review of medical charts (including medication). Subjects were excluded if they had a diagnosis of cancer, acute illness, metabolically unstable chronic disease (e.g., renal or hepatic disease), and used oral steroids or antibiotics within 1 or 3 months, respectively, before enrolling in the clinical trial. Blood pressure was measured using a sphygmomanometer while at rest. The selected 247 individuals were stratified into 4 groups based on their MCI status (MCI: 18 ≤ MoCA Score ≥ 25, non-MCI: 26 ≤MoCA Score ≥30)31,33 and presence of chronic morbidities (CCIS > 0) or absence (CCIS = 0)) into: 54 MCI subjects with chronic morbidities, 60 MCI subjects without chronic morbidities, 73 without MCI but with chronic morbidities, and 60 without MCI nor chronic morbidities (healthy controls).
All clinical trials included in this study were approved by the Texas A&M University Institutional Review Board and all participants provided written informed consent.
Wellbeing, cognitive function, and lifestyle
The prevalence of anxiety and depression was measured by the Hospital Anxiety and Depression Scale (HADS). 34 Visual-motor tracking skills and psychomotor speed were measured by the Trail-Making Test (TMT). 35 The presence of MCI was measured by the MoCA (version 1 or 3) administered by certified study personnel who completed the mandatory training on the online MoCA platform. 31 Executive functioning and cognitive flexibility were assessed using the interference score of the Stroop Color-Word Test (SCWT). 36 Executive function and processing speed were measured with the Phonemic Fluency test.37,38 Lastly, domains of episodic memory including learning and delayed recall were measured using the Rey-Auditory Verbal Recall Test.39,40 Lifestyle was assessed by measuring habitual dietary intake using 24 h dietary recall, 41 and daily physical activity level using the Physical Activity Scale for the Elderly questionnaire (PASE). 42
Assessment of body composition, muscle function, and functional performance
Body weight and height were measured by a digital beam scale and stadiometer to calculate BMI. Lean and fat mass on both the whole body level and in the extremities, as well as visceral adipose tissue (VAT) were measured using dual-energy x-ray absorptiometry (Hologic QDR 4500/Version 12.7.3.1, Hologic, Bedford, MA, USA). 43 Handgrip and single leg (lower extremity) strength were assessed by handgrip (Vernier, Beaverton, OR, USA) and isokinetic dynamometry (Kincom, Chattanooga, TN, USA), respectively, and respiratory muscle strength (maximal inspiratory pressure) by mouth pressure device (Micro Respiratory Pressure Meter).6,44 Functional performance was measured by the 6 Minute Walk Test 45 and balance sway area, a marker of postural balance function and neuromuscular control, using a force platform (Advanced Mechanical Technology, Inc, Watertown, MA). 46
Whole-body amino acid metabolism
The metabolic study day began in the early morning following a > 10-h overnight fast. A catheter was inserted into an antecubital vein of the lower arm or hand for the administration of the stable isotope tracer pulse containing a mixture of 18 amino acid tracers (see Supplemental Table 1), and subsequent blood sampling. Participants placed their hands in a temperature-controlled “hot box”, which allowed the hand to remain at a temperature of approximately 40°C throughout the study to obtain arterialized-venous blood. 47 A baseline blood sample was taken before tracer infusion (t = 0) to measure background tracer enrichments and plasma amino acid concentrations. Additional blood samples were collected at t = 10, 20, 30, 60, and 120 min after amino acid tracer pulse administration for tracer enrichment analysis.
Biochemical analysis and calculations
Arterialized-venous blood was sampled into Li-heparinized tubes (SARSTEDT, Numbrecht, Germany) and immediately stored on ice to minimize enzymatic reactions. Blood was centrifuged at 4°C, 8000 × g for 5 min. For deproteinization, Li-heparinized plasma was added to 0.1 vol of 33% (w/w) trichloroacetic acid and vortexed. The deproteinized Li-heparinized plasma was instantly frozen and stored at −80°C until further analyses. Tracer enrichments [tracer:tracee ratios (TTRs)] and amino acid concentrations were analyzed batch-wise by LC-MS/MS by isotope dilution as previously described. 48 TTRs were corrected for the background TTR. We natural-log (ln)-transformed the TTR and performed ANCOVA linear multi-regression analysis with the covariates: MCI, sex, age, LN(time of blood draw), chronic morbidities, lean mass, and MCI*chronic morbidities interaction. The estimated ln(TTR) was converted back by taking its exponential. This TTR decay curve was fitted with a 2 exponential formula (TTR = a*EXP(−k1*t) + b*EXP(−k2*t)) in Graphpad Prism for 4 groups: non-MCI with/without chronic morbidities and MCI with/without chronic morbidities. 49 The curve fitting parameters a, b, k1, and k2 were used to calculate the predicted whole body production, compartmental analysis, and 95% Confidence Intervals of the measured parameters. 49 The formulas used to estimate the compartment size and fluxes between the compartments are shown in Supplemental Table 2.
We performed compartmental analysis, as previously described, allowing the calculation of the tracee fluxes between compartments using information gained from the tracer pulse method.49,50 The conversion from an amino acid to a metabolite was calculated by multiplying the WBP of the product amino acid by the estimated ratio between the TTR of the product and of the substrate. 49 The compartmental framework visualizes the distribution of a tracee in two principal cellular spaces: the accessible (extracellular) domain, labeled as Q1, and the inaccessible (intracellular) domain, marked as Q2. Q2 (and any of the pools) are the space where the small molecules live. In this model, the appearance of tracee in the accessible plasma pool is known as Whole Body Production (WBP). The flow of molecules from Q1 to Q2 is denoted as F2,1, which equals the flow from Q2 to Q1. Intracellular amino acid production is represented by F0,2. Fractional loss (Fracloss) is the loss in the “intracellular” Q2 pool and is the ratio between what is leaving irreversibly (F02), divided by what has entered. Fractional loss represents the proportion of the intracellular amino acid production (F0,2) that is lost and not retained within the system. Input into Q2 is from U (which equals F0,2) and F2,1 (which equals F1,2). The formula for fractional loss is F0,2 / (F0,2 + F2,1), and irreversible loss is calculated as F0,2 multiplied by fractional loss. Intracellular protein breakdown will thus deliver the amino acids via the U2 entry and will be disposed of via the F0,2 exit. Any recycling will indeed add to the turnover (as it does with continuous infusion protocols). The used formulas are provided in Supplemental Table 1 and described previously (Cobelli 2002). 51
Whole body production of tau-methylhistidine was used as a marker of myofibrillar protein breakdown. 52 Whole body production of phenylalanine was used as a marker of whole body protein breakdown 53 and the conversion rate of phenylalanine to tyrosine was used as a marker of net-whole body protein breakdown. Plasma concentration of hsCRP and glucose were measured on a Cobas C111 Analyzer with standard kits (Roche Diagnostics, Mannheim, Germany).
Statistical analysis
Mean, standard error and confidence intervals were calculated. Our primary outcomes include the differences in amino acid plasma concentrations and whole body production and conversion rates between MCI and non-MCI subjects, subjects with and without chronic morbidities, determined by the Charlson Comorbidity Index, as well as the MCI*chronic morbidity interactions. Secondary outcomes include group differences in body composition, muscle function, functional performance, and well-being. Data were tested for normality and, if log-normally distributed, log-transformed before further statistical analysis. We expressed our data as mean [95% CI]. Group differences for categorical variables were determined using Fisher's exact test. The effects of MCI, the presence of chronic morbidities, as well as the (MCI×chronic morbidities) interactions, were studied by analysis of covariance (ANCOVA) using the covariates sex, age, and centered (BMI). Post-hoc analysis was performed to examine differences between the 4 subgroups. The significance was set at p < 0.05. Statistical analysis was completed using Graphpad Prism (Version 10.0, GraphPad Software Inc., San Diego, CA, USA).
Results
Population characteristics
Based on the MoCA score stratification (Table 1), 114 participants had evidence of MCI and 133 were non-MCI. One or more chronic morbidities were found in 127 of the subjects whereas 120 subjects had no chronic morbidities. Although no MCI or chronic morbidity effects were observed for age, the MCI individuals were older than the non-MCI when chronic morbidities were present (p = 0.0168).
General and clinical characteristics.
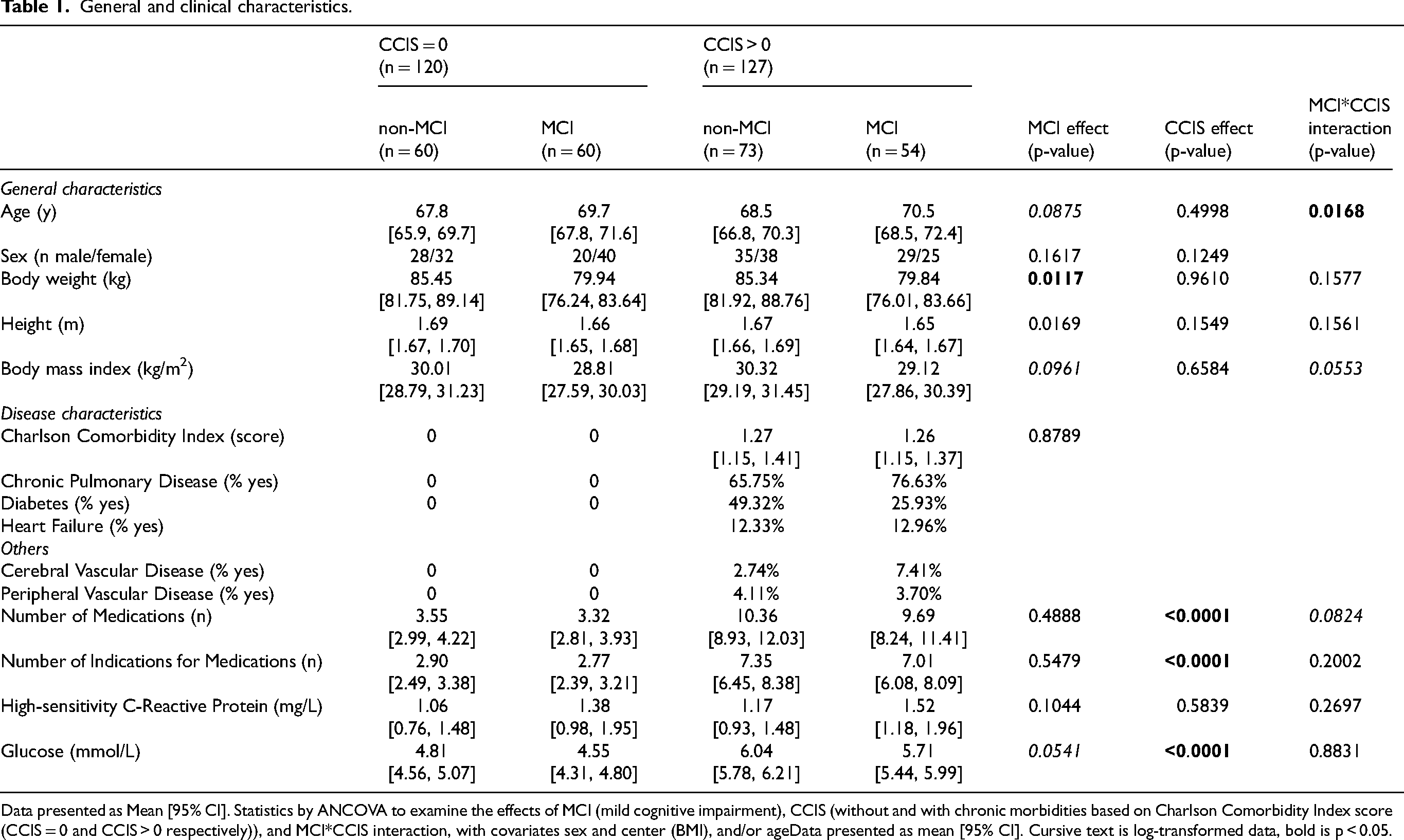
Data presented as Mean [95% CI]. Statistics by ANCOVA to examine the effects of MCI (mild cognitive impairment), CCIS (without and with chronic morbidities based on Charlson Comorbidity Index score (CCIS = 0 and CCIS > 0 respectively)), and MCI*CCIS interaction, with covariates sex and center (BMI), and/or ageData presented as mean [95% CI]. Cursive text is log-transformed data, bold is p < 0.05.
Body weight was lower in those with MCI (p = 0.0117) but no chronic morbidity effect was observed. Also, no MCI or chronic morbidity effects were found for plasma concentration of hsCRP, but glucose was higher in those with chronic morbidities (p < 0.0001). The most prevalent morbidities in the chronic morbidity groups included moderate to severe COPD (non-MCI: 65.75%, MCI: 79.63%), diabetes (non-MCI: 49.32%, MCI: 25.93%), and heart failure (non-MCI: 12.33%, MCI: 12.96%). Other morbidities that were present to a lower degree included peripheral (non-MCI: 4.11%, MCI: 3.70%) and cerebrovascular disease (non-MCI: 2.74, MCI: 7.41%). The presence of chronic morbidities but not MCI was associated with a higher number of medications (p < 0.0001) and indications for medications (p < 0.0001). Common medications taken more frequently by MCI individuals within the chronic morbidity group were anti-emetic (non-MCI: 0.00% versus MCI: 5.56%), anti-fungal (non-MCI: 0.00% versus MCI: 11.11%), and sedative-hypnotic (non-MCI: 4.11% versus MCI: 18.52%) medications (data not shown).
Wellbeing and cognitive function
MCI effect
The MCI individuals (Table 2) had more severe anxiety (p = 0.0174), but no differences in depression compared to those without MCI. The average MoCA score in MCI patients was 23 and 27 in non-MCI. MCI individuals performed worse on the Trail Making Test (p = 0.0004), Stroop Test (p = 0.0422), and Phonemic fluency (p = 0.0022) test.
Wellbeing and cognitive function.

Data presented as mean [95% CI]. Statistics by ANCOVA to examine effects of MCI (mild cognitive impairment), CCIS (without and with chronic morbidities based on Charlsen Comorbidity Index score (CCIS = 0 and CCIS > 0 respectively)), and MCI*CCIS interaction, with covariates sex, (centered) BMI, and age. Cursive text is log-transformed data, bold is p < 0.05.
Chronic morbidity effect
The presence of chronic morbidities was associated with more anxiety (p < 0.0001) and depression (p = 0.0072), and lower performance on the MoCA (p = 0.0263) and Trail-Making (p = 0.0244) tests than those without chronic morbidities.
MCI*chronic morbidity interaction was found for MoCA score (p = 0.0023) indicating that the MCI group with no chronic morbidity had worse cognitive performance than those with chronic morbidities as compared to their non-MCI counterparts.
Body composition, muscle function, functional performance, and habitual dietary intake
MCI effect
The MCI individuals (Table 3) had lower values for total lean mass (p = 0.002), extremity lean mass, (p = 0.0008), and 6-min walk distance (p = 0.0003) (Figure 1) than those without MCI. The MCI subjects consumed less fiber (p = 0.0306) than those without MCI (Supplemental Table 3).
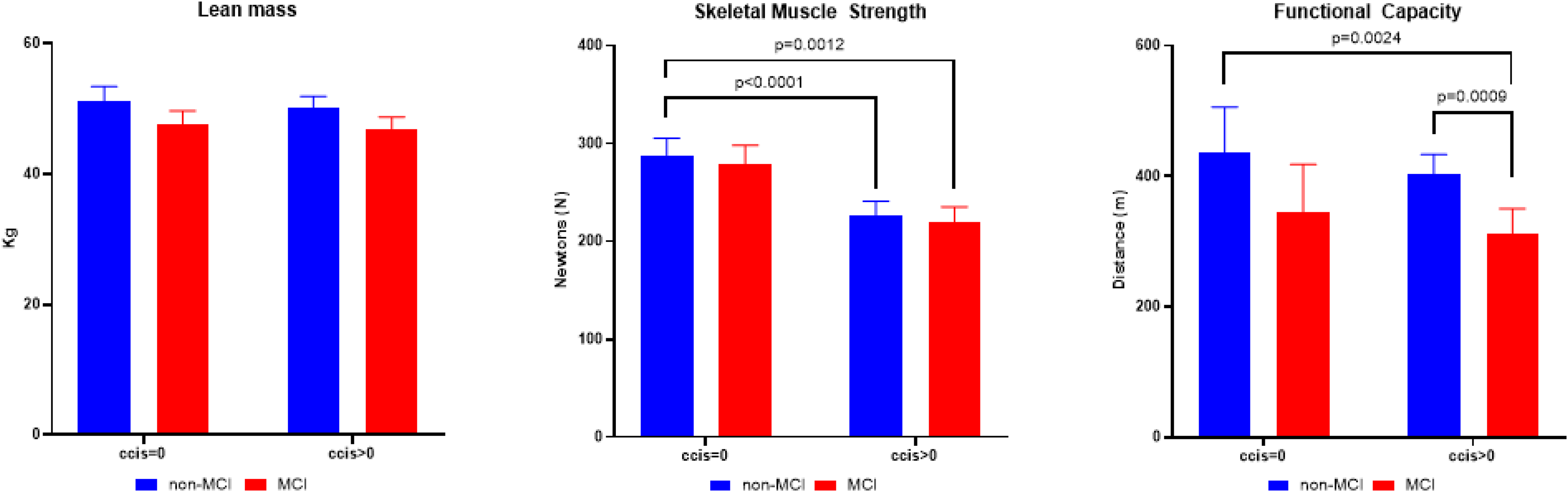
Lean mass (kg, left panel)), skeletal muscle strength (N, middle panel), and functional capacity (6 meter walk distance in m, right panel) of older adults with mild cognitive impairment (MCI) (red bars), non-MCI (blue bars), and those without and with chronic morbidities based on Charlsen Comorbidity Index score (CCIS = 0 and CCIS > 0 respectively). Data are presented as mean [95% CI]. Statistics by Analysis of Covariance adjusted for age and sex, and BMI in case of skeletal muscle strength. The p-values above the middle and right panel refer to post-hoc analysis when statistical significance was reached between groups (p < 0.05).
Body composition, muscle function, and functional performance.
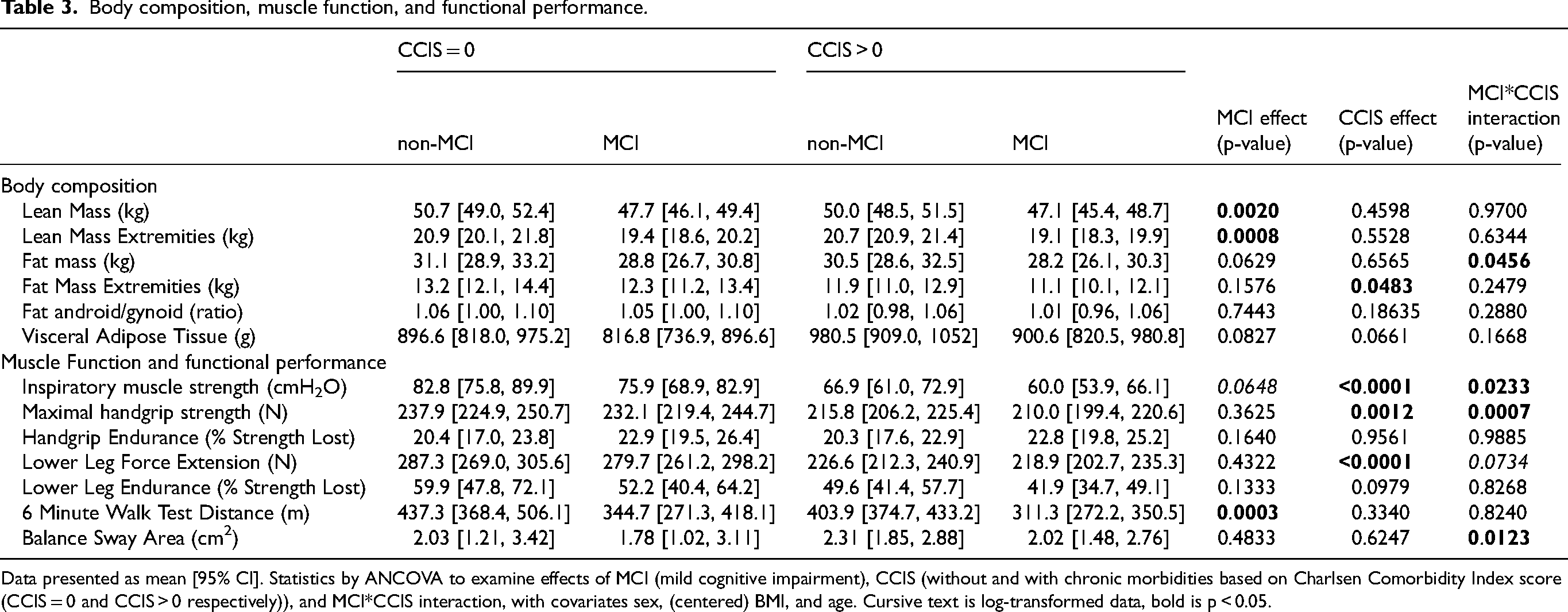
Data presented as mean [95% CI]. Statistics by ANCOVA to examine effects of MCI (mild cognitive impairment), CCIS (without and with chronic morbidities based on Charlsen Comorbidity Index score (CCIS = 0 and CCIS > 0 respectively)), and MCI*CCIS interaction, with covariates sex, (centered) BMI, and age. Cursive text is log-transformed data, bold is p < 0.05.
Chronic morbidity effect
Individuals with chronic morbidities had lower values for fat mass in the extremities (p = 0.0483), inspiratory muscle strength (p < 0.0001), handgrip strength (p = 0.0012), and lower leg strength (p < 0.0001). Subjects with chronic morbidities consumed more calories (p = 0.0218), protein (p = 0.0248), and fat (p = 0.0378) than those without chronic morbidities.
The MCI*chronic morbidity interactions observed indicated that MCI subjects with chronic morbidities had worse inspiratory muscle strength (p < 0.0233), and handgrip strength (p = 0.0007) than those without chronic morbidities as compared to their healthy counterparts. Post hoc analysis (Figure 1) showed lower values for skeletal muscle strength in the chronic morbidities group independent of the presence of MCI (p < 0.002). Functional capacity (6-min walk distance) was lower in the MCI group with comorbidities compared to both non-MCI subgroups (p < 0.003).
Amino acid plasma concentrations
MCI effect
Subjects with MCI (Table 4) had lower plasma concentrations of alanine (p = 0.0360), proline (p = 0.0157), leucine (p = 0.0210), sum of all BCAAs (p = 0.0434), and sum of all EAA (p = 0.0170) than those without MCI.
Plasma amino acid concentrations (µmol/L).
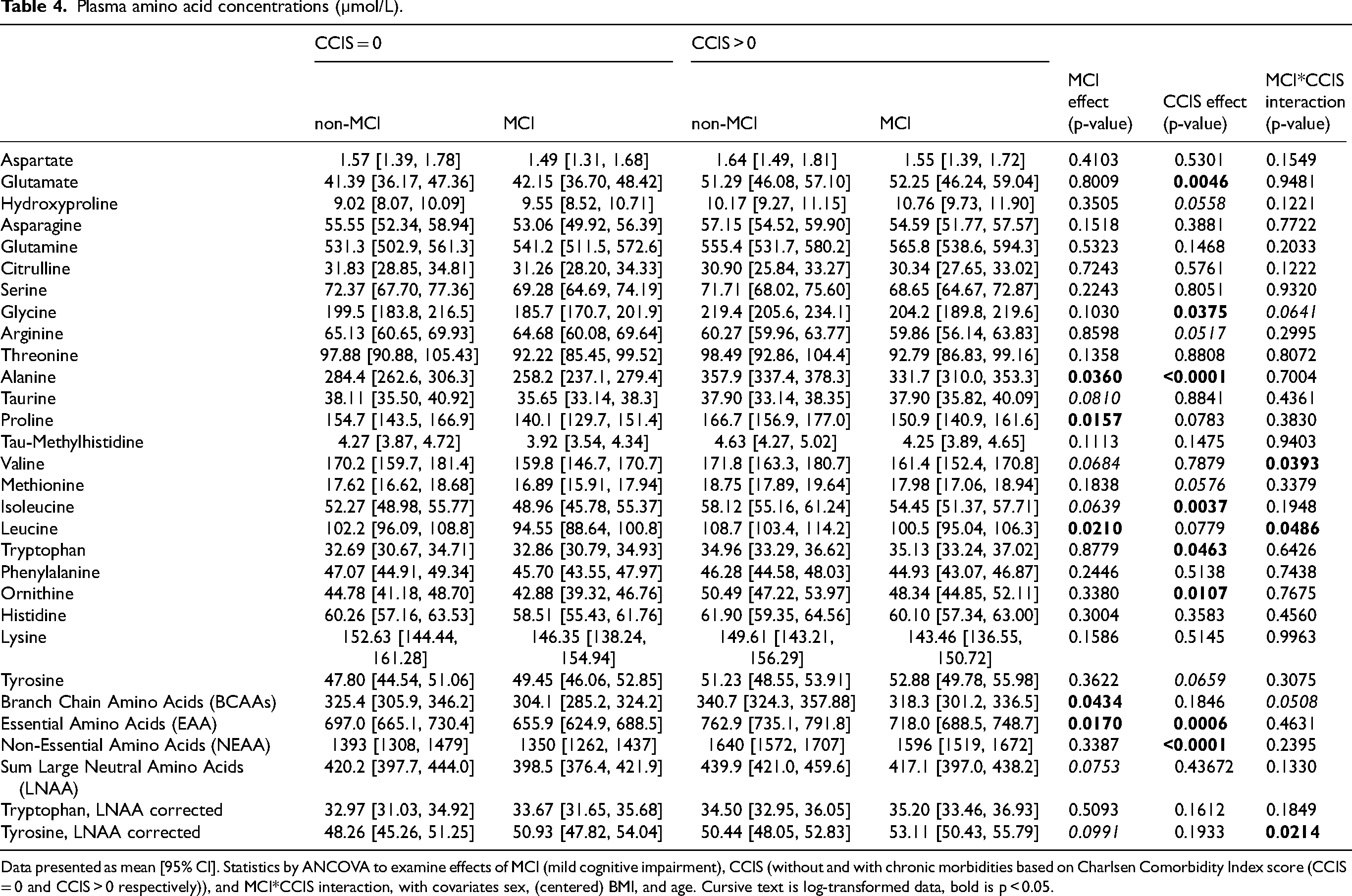
Data presented as mean [95% CI]. Statistics by ANCOVA to examine effects of MCI (mild cognitive impairment), CCIS (without and with chronic morbidities based on Charlsen Comorbidity Index score (CCIS = 0 and CCIS > 0 respectively)), and MCI*CCIS interaction, with covariates sex, (centered) BMI, and age. Cursive text is log-transformed data, bold is p < 0.05.
Chronic morbidity effect
Subjects with chronic morbidities had higher values for plasma glutamate (p = 0.0046), glycine (p = 0.0375), alanine (p < 0.0001), isoleucine (p = 0.0037), tryptophan (p = 0.0463), ornithine (p = 0.0107), sum of all EAA (p = 0.0006), and sum of all non-essential amino acids (NEAA) (p < 0.0001) than those without chronic morbidities.
Whole body amino acid kinetics
MCI effect
Subjects with MCI (Table 5, Figures 2 and 3) had lower WBP of arginine (p < 0.0001), glycine (p = 0.0013), leucine (p = 0.0215), and the conversion of phenylalanine to tyrosine (marker of net protein breakdown) (p < 0.0001) compared to non-MCI subjects, but higher WBP of taurine (p = 0.0463).
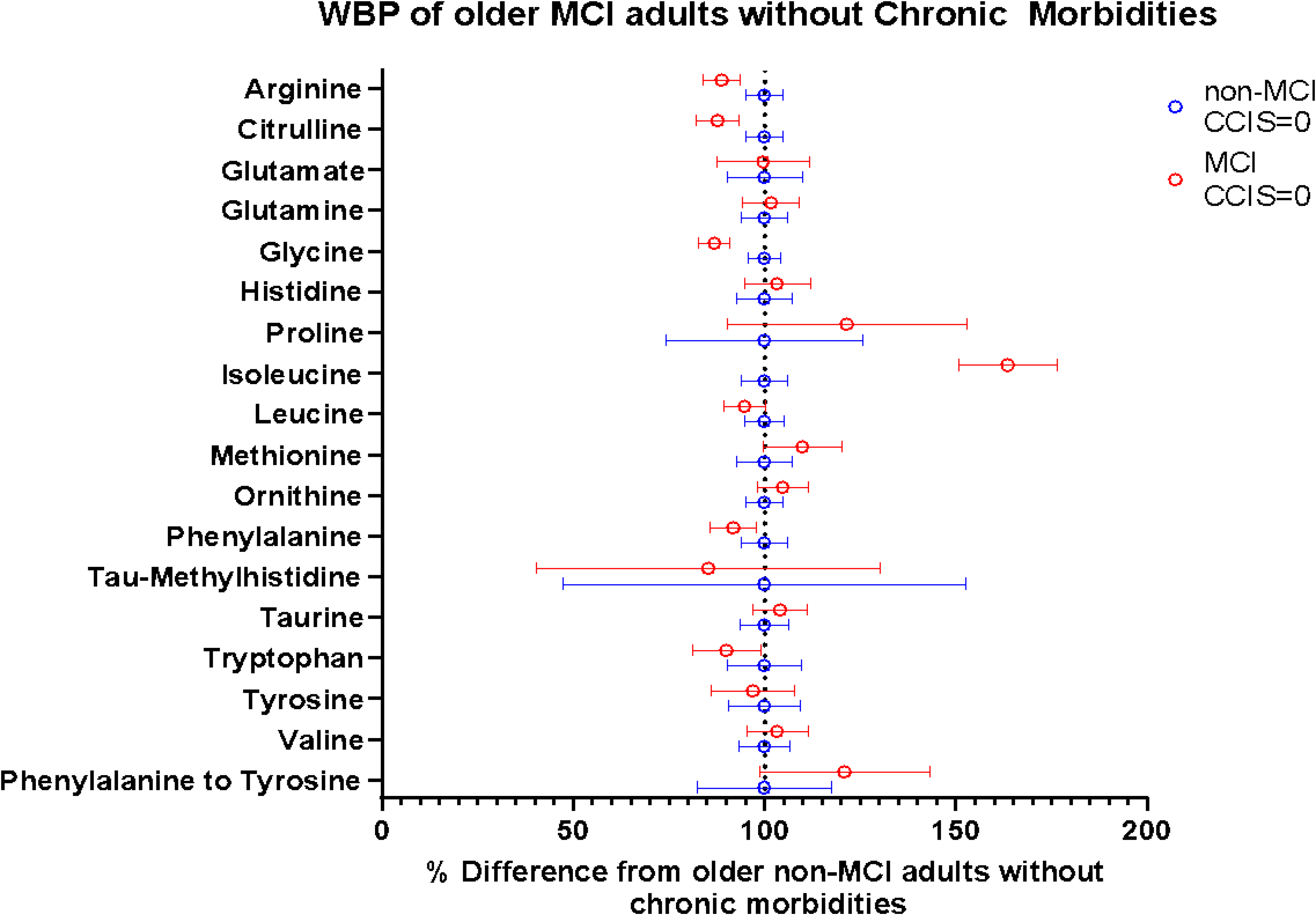
The % difference of whole body amino acid production (WBP) rates in older adults without chronic morbidities (based on Charlsen Comorbidity Index score (CCIS)) and with mild cognitive impairment (MCI) (red circles) as compared to those without MCI (blue circles). The blue circles for the non-MCI and the corresponding confidence intervals all align at “100%” because this is the group MCI is being compared to. The red circles for MCI and the corresponding confidence intervals depict how much different (in %) the whole body production rates of MCI is from non-MCI. If the confidence interval for a specific amino acid does not overlap the dotted line at 100%, the whole body production of this amino acid is significantly different (p < 0.05) in MCI compared to non-MCI when chronic morbidities are not present.
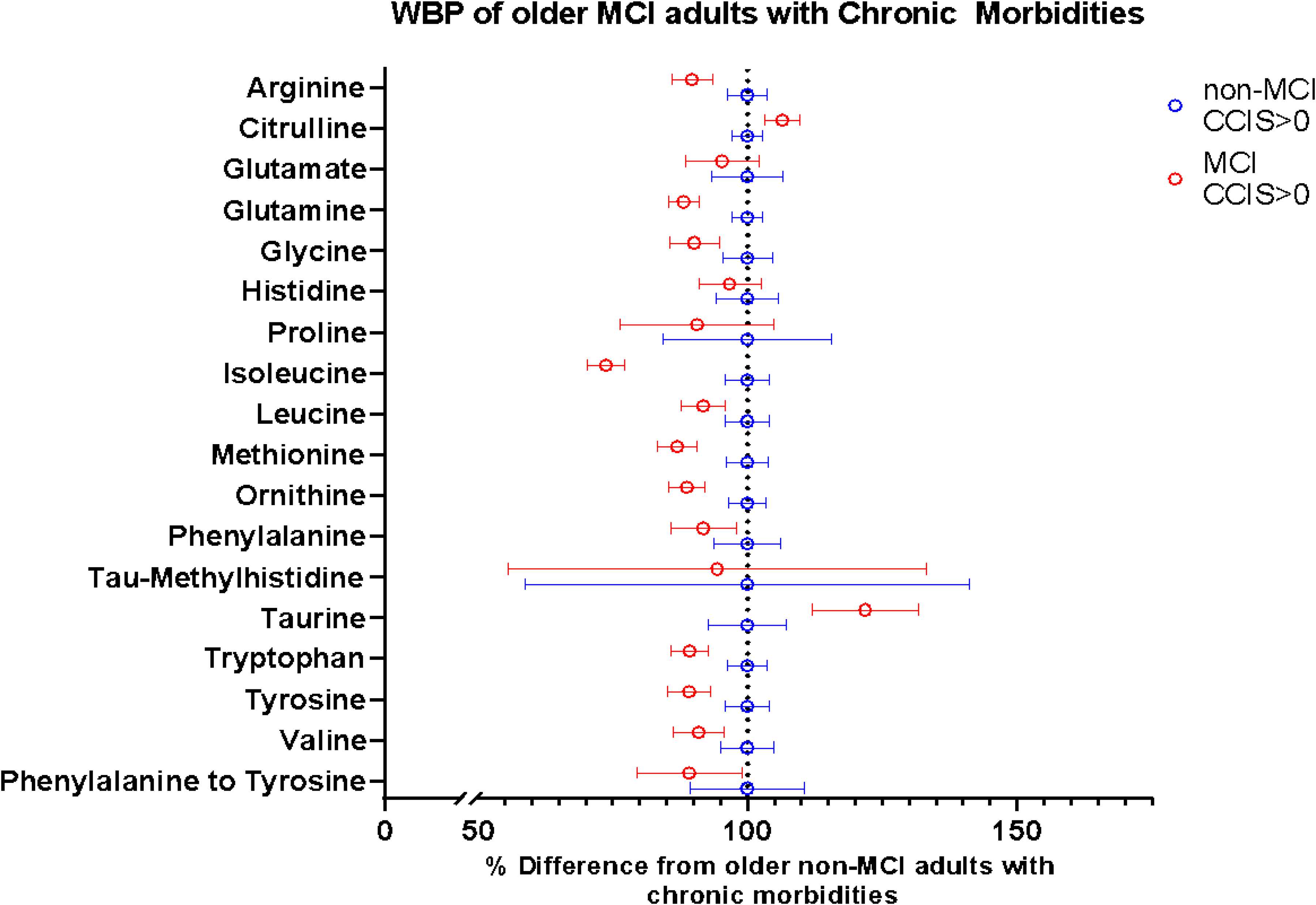
The % difference in whole body amino acid production rates (WBP) in older adults with chronic morbidities (based on Charlsen Comorbidity Index score (CCIS)) and with mild cognitive impairment (MCI) (red circles) as compared to those without MCI (blue circles). The blue circles for the non-MCI and the corresponding confidence intervals align at “100%” because this is the group MCI is being compared to. The red circles for MCI and the corresponding confidence intervals depict how much different (in %) the whole body production rates of MCI is from non-MCI. If the confidence interval for a specific amino acid does not overlap the dotted line at 100%, the whole body production is significantly different (p < 0.05) in MCI compared to non-MCI when chronic morbidities are present.
Whole body amino acid production and conversion rates (µmol/min).
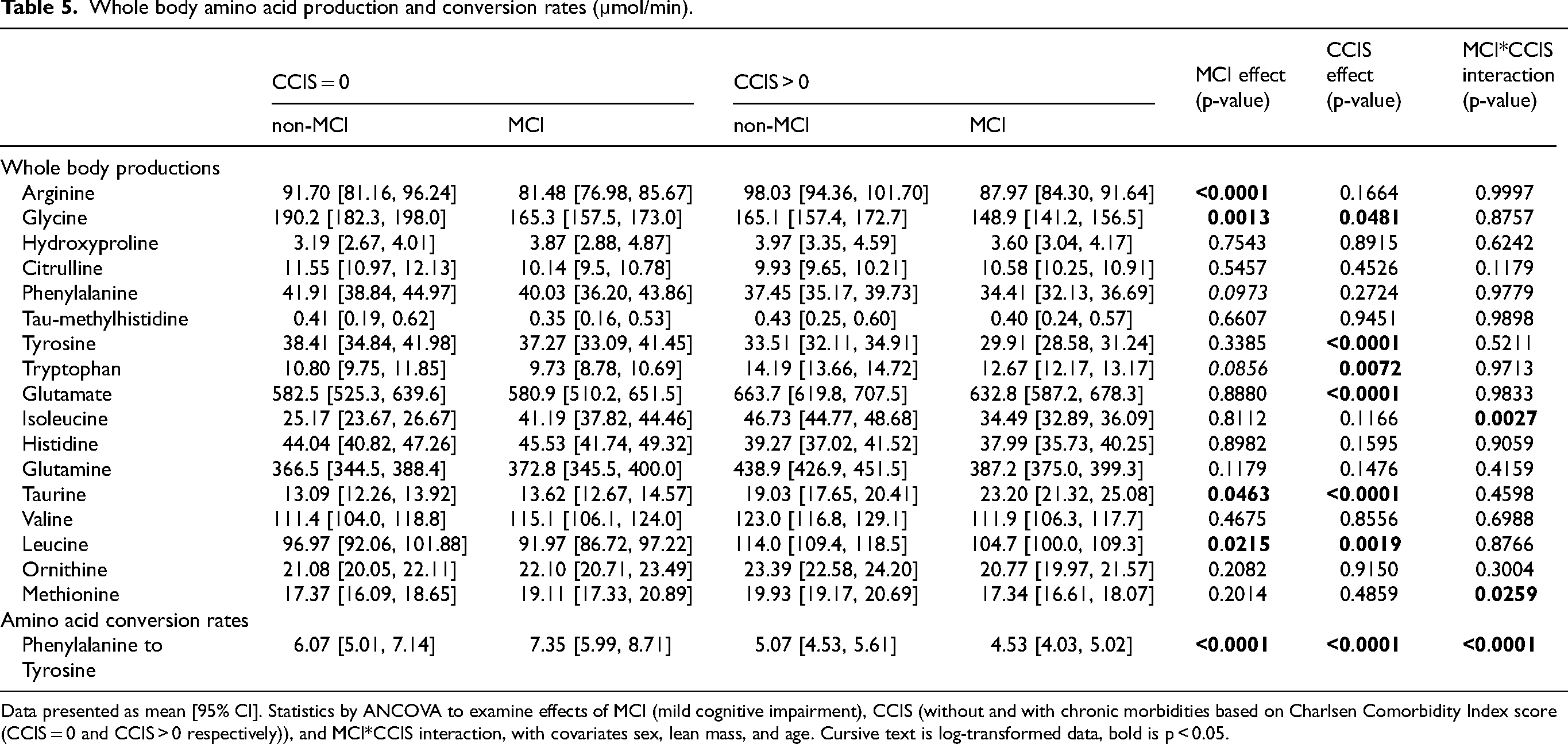
Data presented as mean [95% CI]. Statistics by ANCOVA to examine effects of MCI (mild cognitive impairment), CCIS (without and with chronic morbidities based on Charlsen Comorbidity Index score (CCIS = 0 and CCIS > 0 respectively)), and MCI*CCIS interaction, with covariates sex, lean mass, and age. Cursive text is log-transformed data, bold is p < 0.05.
Chronic morbidity effect
Lower values for WBP were found for many amino acids in those with chronic morbidities (Table 5, Figures 2 and 3). Subjects with chronic morbidities had lower WBP of glycine (p = 0.0481) and tyrosine (p < 0.0001), and conversion of phenylalanine to tyrosine (p < 0.0001) than those without chronic morbidities. In addition, subjects with chronic morbidities had higher WBP of tryptophan (p = 0.0072), glutamate (p < 0.0001), taurine (p < 0.0001), and leucine (p = 0.0019) when compared to those without chronic morbidities.
MCI*chronic morbidity interactions were observed for WBP of isoleucine (p = 0.0027) and methionine (p = 0.0259), as well as for the conversion rate of phenylalanine to tyrosine (p < 0.0001) (Table 5). Posthoc analysis (Figure 4) showed the highest values for WBP of taurine (p < 0.03) and lowest values for net protein breakdown (p < 0.0001) in the MCI group with comorbidities compared to the other subgroups. Presence of MCI was associated with an increase in net protein breakdown in the group without chronic morbidities (p < 0.0001) and a reduction in net protein breakdown (p < 0.0001) in those with chronic morbidities.
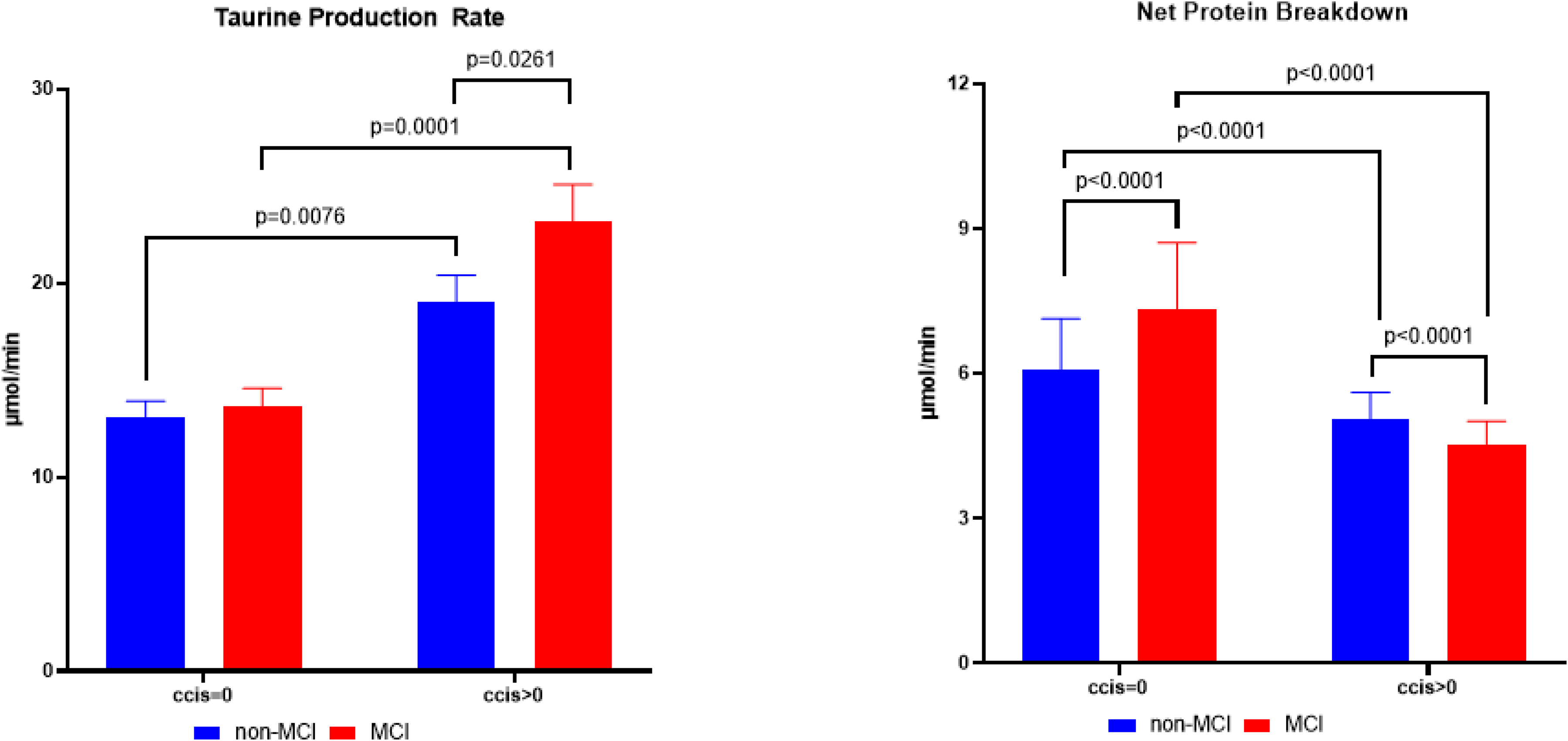
Whole body production rate of taurine (μmol/min, left panel) and net protein breakdown (phenylalanine to tyrosine conversion in μmol/min, right panel) of older adults with mild cognitive impairment (MCI) (red bars), non-MCI (blue bars), and those without and with chronic morbidities (CCIS = 0 and CCIS > 0 respectively). Data are presented as mean [95% CI]. Statistics by Analysis of Covariance adjusted for age, sex, and lean mass. The p-values above the panels refers to post-hoc analysis when statistical significance was reached between the groups (p < 0.05).
Differences between the groups for WBP of the various amino acid production rates are summarized in Figure 5, showing the % differences as compared to older adults without MCI and without chronic morbidities.
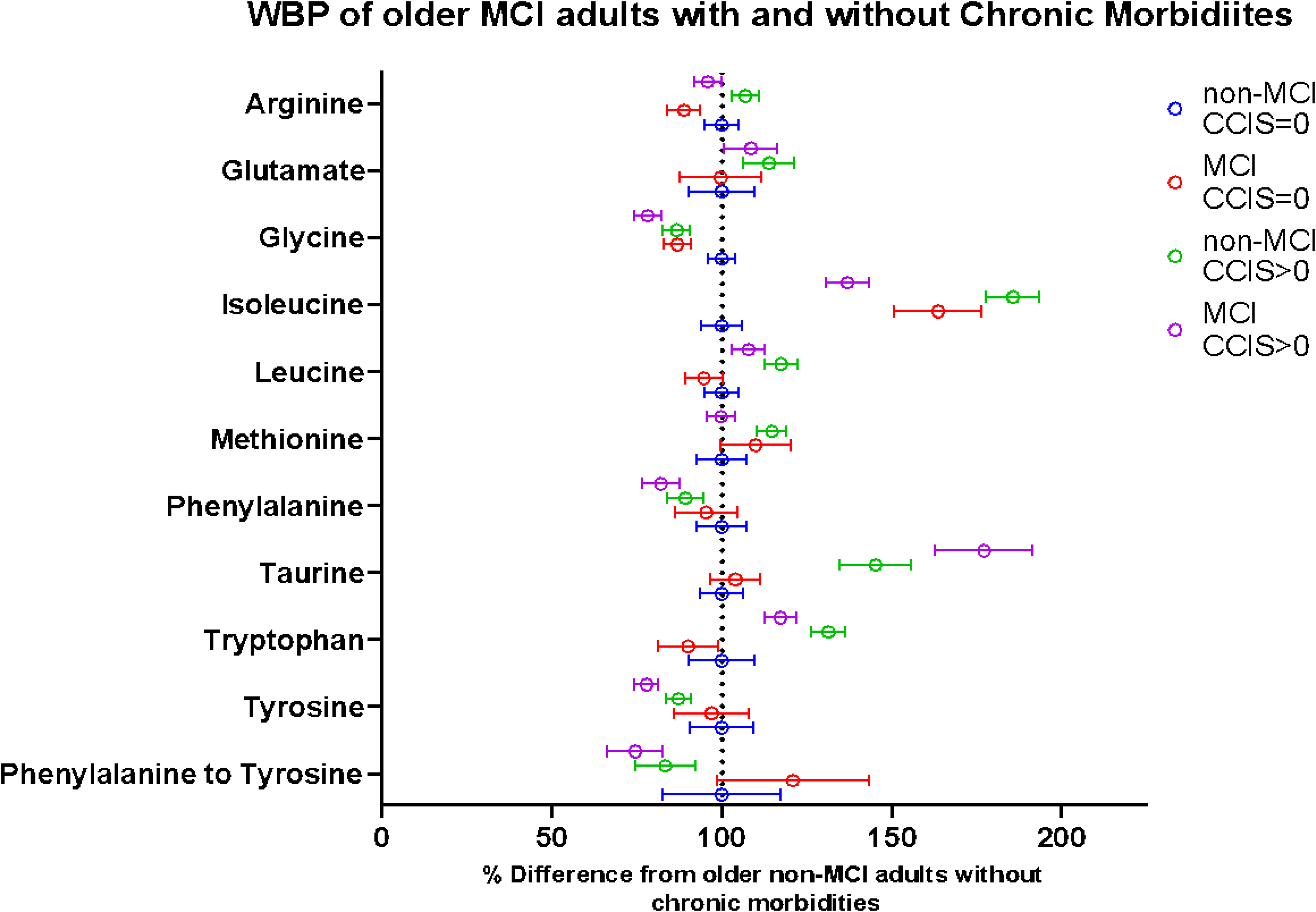
The % difference in whole body amino acid production rates (WBP) in older adults with mild cognitive impairment (MCI) without chronic morbidities (red), non-MCI with chronic morbidities (green), and MCI with chronic morbidities (purple) as compared to non-MCI older adults without chronic morbidities (blue). The non-MCI without chronic morbidities (blue) with the corresponding confidence intervals align at “100%” because this is the group all other groups are being compared to. The red, green, and purple circles with confidence intervals depict how much different (in %) the WBP of MCI without chronic morbidities, non-MCI with chronic morbidities, and MCI with chronic morbidities are from the older adults without both MCI and chronic morbidities, respectively. Presence of chronic morbidities is based on Charlsen Comorbidity Index score (CCIS). If the confidence interval for a specific amino acid does not overlap the dotted line at 100%, the whole body production rate of this amino acid is significantly different (p < 0.05) compared to that of older non-MCI adults without chronic morbidities.
Discussion
Our data suggests that the presence of chronic morbidities and MCI in the older population affects skeletal muscle function and metabolic pathways differently in older adults. Presence of MCI was associated with lower (extremity) lean mass and functional capacity, and chronic morbidities with lower respiratory and skeletal muscle strength. Also, differences in metabolism were observed as shown by a downregulation in metabolism in MCI of multiple amino acids (i.e., arginine, glycine, and leucine), except for the higher taurine production, but an upregulation of metabolism was found in those with chronic morbidities of phenylalanine, glutamate, taurine, tryptophan, leucine). The older adults with both MCI and chronic morbidities had the lowest values for muscle strength and functional performance. We believe this is likely related to the severe downregulation of glycine production and net protein breakdown, and upregulation of taurine.
Cognitive function and mood: role of MCI and chronic morbidities?
The presence of MCI as assessed by MoCA was associated with poor scores on several of the cognitive tests. Our results support the notion that the studied MCI individuals showed signs of cognitive decline related to decreased verbal fluency, 33 but did not show declines in learning or delayed recall abilities. Because deficits in language abilities occur particularly in the beginning of cognitive decline 54 and the subjects in the MCI group had an average MoCA score of 23, it is possible that our subjects were still in the early phase of cognitive decline.
The MCI individuals and those with chronic morbidities performed worse on the trail-making test, suggesting decreased executive function and visuomotor control. This is in line with previous studies showing an association between a decline from cognitively normal to dementia with a loss of complex and fine motor control. 55 We did not observe differences in performance for the Stroop test, phonemic fluency tests, or the AVLT between individuals with and without chronic morbidities. This, along with previous research, 56 suggests that the presence of chronic morbidities mainly affects certain domains of cognitive function, which may occur particularly in those with preexisting muscle dysfunction and the presence of abdominal obesity as previously observed in COPD. 44 Therefore, we believe it is critical to delineate the effects of the presence of chronic morbidities on several cognitive domains in older adults in MCI patients by using a comprehensive battery of cognitive assessments. Furthermore, because the MoCA test is an overall assessment of multiple cognitive domains, the test may not be adequate for detecting domain-specific changes in cognitive function present in older adults with chronic morbidities.
Intake of more than 3 prescription medications, as present in some of our individuals with chronic morbidities to treat diabetes and diastolic blood pressure, has previously been associated with a reduced risk of MCI progression to dementia and an increased probability of MCI remaining stable or reverting back to normal cognitive function. 57 This seems to be supported by the observation that MCI is reversible when existing chronic morbidities are treated through the performance of lifestyle activities. 58 Whether our studied MCI group with chronic morbidities may exhibit reversible MCI remains a critical gap and deserves further investigation.
Detecting mood disorders in MCI is especially important when chronic morbidities are present due to their severe negative impact on quality of life.59,60 While MCI has previously been associated with increased levels of depression, 61 our data indicate an association between MCI and anxiety. The presence of chronic morbidities in our older adults was associated with higher levels of both depression and anxiety, in line with our previous findings that chronic morbidities are associated with mood disorders independent of the presence of MCI. 18 Previously, specific disturbances in BCAA metabolism (lower plasma concentrations and increased clearance of leucine) have been observed in COPD patients with mild depression. 18 Whether the observed anxiety particularly in our study population with MCI and chronic morbidities is also linked to specific changes in BCAA kinetics or other metabolic alterations including that of TRP requires further investigation.
Different markers of muscle health impairment in MCI and chronic morbidities
In the present study, we observed (extremity) lean mass loss in MCI compared to non-MCI individuals. This is in line with previous research showing that sarcopenia is associated with MCI and cognitive decline.62,63 The lower lean mass was accompanied by reduced functional performance as reflected by a shorter 6-min walking distance. Previous studies64,65 reported that the presence of MCI is associated with a decline in motor control and the ability to control force production of skeletal muscle. However, we did not observe a significant reduction in muscle strength without the presence of chronic morbidities. We and others 29 also observed that the presence of chronic morbidities was associated with respiratory and skeletal weakness but with preservation of muscle mass. Interestingly, those with MCI and chronic morbidities had the lowest values for muscle weakness, which could not be explained by higher systemic inflammation or lower physical activity and habitual protein intake levels as these measures were comparable between the groups. Research is needed to understand the specific metabolic mechanisms underlying poor muscle health in this group of individuals.
Reduced (net) whole body protein breakdown in chronic morbidities
Our data showed that the presence of chronic morbidities is independently associated with a downregulation of net protein breakdown (protein breakdown minus synthesis). Particularly the older adults with both MCI and chronic morbidities had besides poor muscle strength and functional performance also the lowest values for net protein breakdown. In line, our previous study also showed a reduced net whole-body protein breakdown in a large group of moderate to severe COPD patients, and a strong association with presence of muscle weakness and exercise intolerance (6-min walk test). 44 A lower net protein breakdown may be related to a lower rate of autophagy as previously suggested. 44 Autophagy is responsible for the lysosome-mediated degradation of damaged proteins and organelles and therefore is an essential process to maintain cellular homeostasis and function. Whether increasing dietary protein intake in older adults with both MCI and chronic morbidities upregulates net protein breakdown and autophagy warrants further investigation. 44 In contrast, WBP of several amino acids were elevated in those with chronic diseases, in line with previous studies 20 suggesting enhanced protein turnover which is known to be an energy consuming process.
Reduced whole body glycine production in MCI and chronic morbidities
Glycine functions as an inhibitory neurotransmitter66,67 within the central nervous system. Glycine's role as a neurotransmitter includes roles in voluntary motor control, processing sensory information, generating reflex responses, auditory functions, cardiovascular functions, and respiratory functions. 66 Increased plasma glycine concentrations have previously been associated with decreased motor control. 68 While we did not observe differences in glycine concentrations in MCI, we did observe higher glycine concentrations in the presence of chronic morbidities. Whether the decreased trail-making test performance, indicative of poor motor control, in those with chronic morbidities could be explained by elevated plasma glycine concentrations deserves further investigation.
On the other side, glycine is utilized in several pathways including the formation of creatine, 67 which has roles in energy metabolism in both skeletal muscle and nervous tissue. Deficiencies in creatine, particularly concerning the brain, can result in cognitive disorders. 69 Glycine is also utilized in the formation of glutathione, 67 a key cellular antioxidant. Research has shown that dietary glycine and related compounds such as glutathione, might act as potential therapeutic agents in the fight against acute whole-body inflammatory responses and to improve cognitive function in aging populations with and without chronic morbidities.27,70 Besides muscle weakness, those participants with both MCI and chronic morbidities were characterized by a severe downregulation of whole body glycine production. Whether glycine supplementation may serve as a therapeutic intervention to improve skeletal muscle and cognitive health in MCI individuals and especially in those without chronic morbidities, warrants further investigation.
Altered response in leucine and isoleucine kinetics in MCI when chronic morbidities are present
Plasma leucine concentrations were lower in MCI compared to non-MCI, in line with previous research where decreased plasma concentrations of leucine were associated with cognitive decline. 71 Evidence exists that lower leucine concentrations may result from a combination of risk factors associated with aging including obesity, hypertension, diabetes, and dyslipidemia. 71 Although WBP of leucine was lower in MCI than in non-MCI, it was elevated when chronic morbidities were present, which is in line with our previous work showing increased leucine production in COPD patients compared to healthy controls. 20 Leucine specifically has the potential to activate signaling pathways to upregulate muscle protein synthesis. 72 The relatively maintained leucine production in MCI with chronic morbidities therefore could be explained by the contradictory effects of both conditions on leucine production.
Besides leucine, also plasma isoleucine was elevated when chronic morbidities were present, in line with previous studies in chronic morbidities with poor metabolic health (cardiovascular disease, insulin resistance, and diabetes).44,73 Some researchers suspect a link between increased isoleucine level and AD pathogenesis, 74 suggesting that elevated isoleucine concentration may be a risk factor for the progression of cognitive decline particularly in those with chronic morbidities. Moreover, increased levels of isoleucine have been related to poor muscle health. 75 Because isoleucine is primarily taken up and metabolized in the skeletal muscle for ATP production, 75 future studies should be conducted to understand the possible mechanisms underlying the changes in isoleucine metabolism as well as the consequences on brain and muscle health.
Increased taurine production rates in MCI and chronic morbidities
Taurine is suggested to play a role in preserving neurogenesis 76 and can act as a neuroprotectant against glutamate-induced excitotoxicity. 77 Evidence exists that increased plasma taurine concentrations are a sign of the beginning stages of cognitive impairment, 76 while later stages of cognitive impairment are characterized by a decrease in plasma taurine concentrations. While we did not find a difference in plasma taurine concentration between older adults with MCI and non-MCI, MCI individuals had a higher WBP of taurine, indicative of an elevated taurine clearance rate. Additionally, individuals with chronic morbidities were observed to have higher taurine production rates in line with previous research.29,78 Consequently, those with both MCI and chronic morbidities had the highest levels for WBP of taurine despite comparable data for plasma taurine. Evidence exists for taurine being beneficial for skeletal muscle health 76 as it has a regulatory effect on calcium homeostasis, ion channels, oxidative stress and control of membrane excitability. Taurine supplementation has been used to ameliorate performance and muscle strength during aging and to reduce atrophy due to disuse 79 and is important in the defense against free radical-mediated damage after exercise. 80 The mechanism to explain the large upregulation of WBP of taurine in those with MCI and chronic morbidities remains unclear as well as whether increasing dietary taurine for older adults with both MCI and chronic morbidities may provide beneficial effects for their cognitive and skeletal muscle health.
Increased tryptophan production rates in chronic morbidities
We observed higher tryptophan concentration and production rates when chronic morbidities were present. Tryptophan is the sole precursor for serotonin, melatonin, and kynurenine production.23,81 Tryptophan can pass through the blood-brain barrier 82 where its metabolism shifts in favor of kynurenine over serotonin when glucocorticoids are released following stress and inflammation. 83 We therefore anticipate a shift to more kynurenine production when comorbidities are present, which has been associated with presence of skeletal muscle dysfunction, decreased cognitive function, and mood disorders.83,84 Tryptophan also competes with large neutral amino acids to bind to transporters providing entry into skeletal muscle fibers. 83 Interestingly, tryptophan and its metabolite serotonin have been shown to increase protein synthesis through upregulating growth hormone secretion. 83 In contrast, the accumulation of kynurenine has a catabolic effect on protein and skeletal muscle fibers.83,84 Further research is needed whether the elevated plasma and WBP of tryptophan observed in individuals with chronic morbidities reflects the alterations in tryptophan metabolites that could contribute to the observed disturbances in cognitive and muscle health.
Altered glutamate and glutamine metabolism with chronic morbidities
We did not observe a difference in plasma glutamate concentration between MCI and non-MCI in contrast to previous research suggesting that cognitive decline is associated with decreased plasma glutamate concentrations 85 and showing a lower ratio of glutamate to glutamine in subjects with MCI as compared to non-MCI. 19 Glutamate functions as the primary excitatory neurotransmitter and plays an important role in learning and memory. 86 Our subjects with chronic morbidities had higher values for concentration and WBP of glutamate. The observed increase in glutamate/glutamine WBP ratio may reflect the cerebral glutamate/glutamine cycle which has been associated with neuronal cell death and Alzheimer's disease pathogenesis. 86 As plasma concentrations of amino acids are generally higher than those in cerebrospinal fluid, 87 further research is needed to test if the changes in amino acid kinetics on whole body level reflect those in cerebrospinal fluid. This is particularly of importance for glutamate as glutamate does not cross the blood-brain barrier. 86
Limitations
Due to the reporting bias associated with self-reported measures of physical activity and dietary intake, there is a potential of recall bias in the data for our subjects’ lifestyle characteristics. Furthermore, as there is a heterogeneity of participants with different chronic morbidities (i.e., COPD, HF, and diabetes), it remains unclear whether specific differences exist in metabolic phenotypes between these chronic diseases that may contribute to the presence of MCI. Future research is needed to conduct a post hoc analysis between larger subgroups of chronic diseases with MCI.
Conclusion
The present study revealed specific differences in functional, nutritional, and metabolic phenotypes in older adults with MCI with and without the presence of chronic morbidities. However, those with both presence of MCI and chronic morbidities are at increased risk for severe muscle weakness likely related to a severe downregulation of glycine production and net protein breakdown.
Future (longitudinal) research is needed to investigate whether the metabolic and physiological alterations can be used as biomarkers to predict MCI development.5,15 Longitudinal observational studies using multiple amino acid stable tracer approaches are also important to examine potential differences in trajectories between MCI patients with and without chronic morbidities. Moreover, how interventions—whether nutritional (increased intake of dietary protein, and modulation of amino acids such as glycine, taurine, leucine, taurine, tryptophan, glutamine), pharmacological, or exercise-based (endurance versus resistance)—could modulate the observed amino acid “signatures” in MCI participants with/without chronic morbidities and improve skeletal muscle health and attenuate cognitive decline in these groups of older adults warrants further investigation as it will show the clinical impact of the findings.
Supplemental Material
sj-docx-1-alz-10.1177_13872877251336618 - Supplemental material for Presence of chronic morbidities alters skeletal muscle health and amino acid kinetics in mild cognitive impairment
Supplemental material, sj-docx-1-alz-10.1177_13872877251336618 for Presence of chronic morbidities alters skeletal muscle health and amino acid kinetics in mild cognitive impairment by Sofie M De Wandel, Nicolaas EP Deutz, Sarah K Kirschner, Elise EN Deutz, Laura E Ruebush and Mariëlle PKJ Engelen in Journal of Alzheimer's Disease
Footnotes
Acknowledgments
We thank the participants for their willingness to participate in this research study and for making this work possible. Furthermore, we thank the CTRAL research personnel for assisting in the data collection.
Ethical considerations
All clinical trials (NCT02082418, NCT02770092, NCT02780206, NCT03327181, NCT03796455, NCT04461236) included in this study were approved by the Texas A&M University Institutional Review Board and all participants provided written informed consent.
Consent to participate
All participants provided written informed consent before study procedures took place.
Author contributions
Sofie M De Wandel (Data curation; Formal analysis; Investigation; Methodology; Writing—original draft; Writing—review & editing); Nicolaas EP Deutz (Conceptualization; Data curation; Formal analysis; Investigation; Methodology; Resources; Software; Supervision; Writing—review & editing); Sarah K Kirschner (Data curation; Investigation; Writing—review & editing); Elise EN Deutz (Formal analysis; Investigation; Methodology; Writing—review & editing); Laura E Ruebush (Investigation; Project administration; Resources; Supervision; Writing—review & editing); Marielle PKJ Engelen (Conceptualization; Formal analysis; Funding acquisition; Investigation; Methodology; Project administration; Resources; Supervision; Writing—original draft; Writing—review & editing).
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study is funded by internal grants from Texas A&M University.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability
Data described in the manuscript, code book, and analytic code will be made available upon request, pending approval of the principal investigator.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
