Abstract
Background
Smoking, a modifiable risk factor for Alzheimer's disease (AD), is associated with impaired functional connectivity in resting-state networks (RSNs). This study investigates how smoking affects brain effective connectivity (EC).
Objective
Investigate smoking-associated EC alterations.
Methods
We identified 129 cognitively unimpaired (CU: 85 non-smokers, 44 smokers) and 84 mild cognitive impairment (MCI: 55 non-smokers, 29 smokers) participants. Granger causality analysis was used to calculate the directed interactions of information flows based on the seed areas of the default mode network, executive control network, and salience network. Mixed-effect analyses were performed to explore the interactive effects of smoking × cognitive status. Linear mixed-effects models evaluated correlations between EC values and longitudinal cognitive decline.
Results
Mixed-effect analyses revealed significant interactive EC differences among 4 groups: (1) Smoking MCI individuals showed reduced EC from the left putamen to the frontoinsular cortex (FIC) compared to the smoking CU and non-smoking MCI group; (2) Non-smoking MCI subjects had lower EC from the dorsolateral prefrontal cortex (DLPFC) to the right inferior occipital gyrus (IOG) than non-smoking CU; (3) Smoking CU subjects exhibited increased EC from the DLPFC to the left middle cingulate cortex compared to the with the non-smoking CU and smoking MCI individuals. Additionally, ECPosterior cingulate cortex−to−IOG and ECPutamen−to−FIC significantly predicted MMSE and ADNI_EF scores over time, respectively.
Conclusions
Smoking distinctly impacts EC within RSNs and overall brain function in both MCI and CU individuals, potentially reducing functional compensation in MCI. These results support smoking cessation as part of AD management strategies.
Keywords
Introduction
Alzheimer's disease (AD) is a neurodegenerative disorder characterized by cognitive dysfunction and is the main cause of dementia. In 2019, approximately 55 million people worldwide suffered from dementia, and this number will increase to 139 million by 2050 according to the World Health Organization. Caring for the patients with dementia is a huge burden on both families and society. 1 The high costs of treating and caring for AD patients, as well as the limited efficacy of drugs to reverse AD emphasize the need to reduce AD risk and delay onset through modifiable risk factors in the early stage of the disease. Mild cognitive impairment (MCI) is a condition characterized by noticeable declines in cognitive abilities that are not severe enough to interfere significantly with daily activities, serving as a potential early indicator of AD and other dementias. Implementing effective strategies to target these modifiable risk factors, such as smoking, could reduce the incidence of AD. 2 Current smokers were found to have a 1.79 higher risk of developing AD compared to non-smokers. 3 Structural imaging analysis has shown that smoking is associated with widespread cortical thinning, and recovery may take up to 25 years. 4 Thus, quitting smoking at any age can reduce the risk of dementia, even in individuals without cognitive impairments. 5
The resting-state networks (RSNs) refer to functional brain networks active at rest. Growing evidence indicates that RSNs like the default mode network (DMN), executive control network (ECN), and salience network (SN) significantly contribute to AD development.6,7 The DMN has been reported to be involved in self-related mental stimulations and memory-related processes. 8 The ECN is involved in target-directed activities, while the SN is involved in dynamic changes regulation in other networks. 9 These networks collaborate to support cognitive function and hold promise as potential sensitive markers for early disease diagnosis.10–13 Smoking has been shown to impact brain functional activity and functional connectivity (FC) within these networks.14,15 Research indicates that, compared to non-smokers, smokers exhibit decreased FC within both the DMN and ECN, alongside significantly reduced connectivity between key network hubs, including the dorsolateral prefrontal cortex (DLPFC) and parietal nodes within the ECN. 16 Individuals with nicotine (the primary component of tobacco) addiction exhibited reduced static functional network connectivity between the dorsal DMN and the right ECN. 17 Resting-state FC analyses showed reduced intrinsic connectivity within the DMN and decreased connectivity between the posterior cingulate cortex (PCC) and temporal cortex in smokers. 18 Network-of-interest-based analyses demonstrated decreased FC within the frontoparietal and SN in smokers compared to non-smokers. 19 Moreover, a multimodal neuroimaging study identified abnormal resting-state FC and structural integrity between the anterior cingulate cortex (ACC) and insula in the SN of young adult smokers. 20
While FC assesses temporal correlations between distant neurophysiological events in the brain, it lacks directional insights. In contrast, effective connectivity (EC) elucidates the directional influence between brain regions. 21 Granger causality analysis (GCA) is a method utilized in functional magnetic resonance imaging (fMRI) studies to estimate directed information flows from time series data, uncovering complex temporal and spatial dynamics underlying cognitive processes.21,22 GCA has been extensively used to investigate smoking cessation and addiction, as well as AD.23–25 Using the GCA method, previous studies have demonstrated that smoking affects the RSNs in cognitively unimpaired (CU) individuals23,24 and that extensive changes exist in the RSNs of AD and MCI patients.26,27 Therefore, we hypothesize that there may be directional differences in RSNs between smoking and non-smoking MCI individuals, which could be related to long-term cognitive function changes.
Thus, we examined the EC between core nodes of RSNs (PCC/anterior medial prefrontal cortex (aMPFC) of the DMN, frontoinsular cortex (FIC) of the SN, and DLPFC of the ECN) and the whole brain in a sample of 129 CU and 84 MCI patients using GCA. The primary objective was to investigate how smoking influences EC at early stage of cognitive impairment and whether these connectivity patterns are associated with cognitive performance. Our hypothesis posits that smoking impairs EC within brain networks, with the greatest impact observed in MCI patients.
Methods
Participants
We selected non-demented participants (including CU individuals and MCI patients) from the ADNI 3 database with permission to use data from the ADNI database. The ADNI is a project focused on developing clinical, neuropsychological, and neuroimaging biomarkers for early detection and monitoring of AD, launched by the National Institute on Aging (NIA), the National Institute of Biomedical Imaging and Bioengineering (NIBIB), the Food and Drug Administration (FDA), private pharmaceutical companies, and non-profit organizations. The ADNI database was approved by the Institutional Review Board at each participating center. Informed written consent was obtained from all participants at each institution.
Each participant underwent neuropsychological assessments, structural MRI, and resting-state fMRI scans. The diagnostic criteria for the cognitive status of the participants were as follows, following the ADNI protocol. For CU controls, individuals should have (1) a Mini-Mental State Examination (MMSE) score ranging from 24 to 30 (inclusive), (2) a Clinical Dementia Rating (CDR) score of 0, and (3) no signs of depression (Geriatric Depression Scale, GDS < 6) or dementia. For MCI patients, individuals should have (1) persistent subjective memory complaints, (2) objective cognitive scale changes without depression (GDS < 6), such as an MMSE score ranging from 24 to 30 (inclusive), (3) a CDR score of 0.5, and (4) scoring below an education-adjusted cut-off score on delayed recall of the Wechsler Memory Scale logical memory test. Participants with a history of significant head trauma, alcohol or drug abuse, other neurological or psychiatric disorders, or a significant risk of vascular disease, defined as Hachinski Ischemia Scale (HIS) > 4, were excluded from the study. Smoking status was determined based on self-reported smoking history in the medical records. Specifically, individuals with no history of smoking were classified as non-smokers, while those with a history of former or current smoking were classified as smokers in this study.
Ultimately, our study included 378 right-handed, cognitively healthy individuals—333 non-smokers and 45 smokers among CU, and 182 MCI patients, consisting of 149 non-smokers and 33 smokers. For a more comprehensive understanding of the inclusion process for the remaining participants, please refer to Supplemental Figure 1.
Neuropsychological assessments
All participants underwent neuropsychological assessments at baseline and the after 1, 2, and 4 years, using the CDR and MMSE to evaluate overall cognitive status. Additionally, various cognitive domains were evaluated, including memory (ADNI memory composite score), executive function (ADNI executive function composite score), language function (ADNI language function composite score), and visuospatial function (ADNI visuospatial function composite score). More detailed information can be found at: http://www.adni-info.org.
MRI acquisition
Based on 3.0-Tesla MRI scanners, T1 weighted structural images were acquired following parameters: repetition time (TR) = 2300 ms; echo time (TE) = 2.98 ms; 170 sagittal slices; within plane, field of view (FOV) = 256 × 240 mm2; voxel size = 1.1 × 1.1 × 1.2 mm3; flip angle = 9°; and bandwidth = 240 Hz/pix. The resting-state fMRI data were acquired using an echo-planar imaging (EPI) sequence with parameters: TR = 3000 ms; TE = 30 ms; the number of slices = 48; slice thickness = 3.3 mm; spatial resolution = 3.31 mm × 3.31 mm × 3.31 mm. Following the ADNI protocol, participants underwent scans with eyes open, focusing on a cross.
Imaging preprocessing
Data pre-processing was performed using the Data Processing Assistant for Resting-state fMRI (DPARSF, http://rfmri.org/DPASFA 28 ) and Resting-State fMRI Data Analysis Toolkit (REST; http://restfmri.net 29 ) based on Statistical Parametric Mapping 12 (SPM12; http://www.fil.ion.ucl.ac.uk/spm) package. The first 10 time points of resting-state fMRI data were removed due to the instability of the initial MRI signal and the inadaptation of the subjects to the scan noise. The remaining images were corrected for timing differences between each slice and head motion (6-parameter rigid body). Data with more than 3 mm maximum displacement in any x, y, or z directions or 3° of any angular motion were discarded, resulting in the removal of 49 subjects due to head motion (29 non-smoking CU, one smoking CU, and 19 non-smoking MCI). Eventually, we identified 304 non-smoking CU, 44 smoking CU, 130 non-smoking MCI, and 33 smoking MCI (Supplemental Figure 1). Subsequently, the resting-state fMRI images were normalized to the EPI template and resampled into 3 × 3 × 3 mm3. Nuisance covariate regression was performed to minimize the residual effects of motion and other non-neuronal factors, using the Friston 24 head motion parameters (6 head motion parameters, 6 head motion parameters from the previous time point, and 12 corresponding squared items), white matter signals, and cerebrospinal fluid signals. Given the controversy of removing global signals in resting-state fMRI data preprocessing, we omitted regressions on global signals. Spatial smoothing was performed with an isotropic 6 mm full-width at a half-maximum (FWHM) Gaussian kernel. Linear detrending and temporal band-pass filtering (0.01–0.08 Hz) were performed to remove bias from high-frequency physiological noise and low-frequency drift.
Propensity score matching
We conducted propensity score matching (PSM) using SPSS 26 to mitigate selection bias in our study. Propensity scores were estimated to match subjects between the non-smoking and smoking subgroups within the CU and MCI groups. This approach helped reduce the impact of selection bias on our results. Multivariable linear regression was employed to analyze the propensity scores, with age, sex, and years of education included as covariates. Non-smokers were matched with smokers in a 2:1 ratio for each cognitive status (CU and MCI) using nearest neighbor matching with replacement, with a caliper of 0.1 standard deviations. As a result, we identified 85 non-smoking CU participants, 44 smoking CU participants, 55 non-smoking MCI patients, and 29 smoking MCI patients from the initial population (Supplemental Figure 1). All findings presented in our study are based on the population after propensity score matching (PSM).
Granger causality analysis
We utilized the REST-GCA toolkit, based on Geweke's feedback model, to generate individual GCA maps. 29 The purpose of GCA is to analyze causal relationships among specific brain regions by making time-directed predictions between blood oxygenation level-dependent (BOLD)-fMRI time series. 30 GCA patterns provide information about the strength and direction of the relationship between seed regions (X) and all other voxels in the brain (Y). A positive coefficient from X to Y indicates that region X causally influences the activity in region Y, while a negative coefficient from X to Y suggests that region Y causally affects the activity in region X. The voxel-level GCA patterns capture the linear direct effect of X on Y (information flow from X to Y) and the linear direct effect of Y on X (information flow from Y to X) across the entire brain. Signed-path coefficients were calculated using the REST-GCA toolkit to generate the GCA maps, including X to Y and Y to X.
Seed areas of the PCC (MNI coordinates: 2, −51, 27), 31 aMPFC (MNI coordinates: −6, 52, −2), 32 FIC (MNI coordinates: 38, 26, −10), 33 DLPFC (MNI coordinates: 44, 36, 20) 33 were defined as previously described, anchoring the DMN (PCC and aMPFC), SN (FIC), and ECN (DLPFC) respectively. Seed regions were represented by 5-mm radius spheres centered on these coordinates.
Statistical analysis
Statistical analyses were conducted using IBM SPSS 26 for Windows, setting significance at p < 0.05. Linear mixed-effects (LME) models were performed using R version 4.4.1. Continuous variables were presented as mean ± standard deviation, and categorical variables as percentages. Between-group differences in demographic and neuropsychological characteristics of the four groups were analyzed using ANOVA, and post-hoc two-sample t-tests were performed with a significance level of p < 0.05, Bonferroni corrected. The Chi-square test was used to analyze sex distribution.
The REST-GCA toolkit was used for statistical analysis of GCA maps, employing a time lag of one TR (3000 ms) to estimate predictions between seed regions’ resting-state fMRI time series and the rest of the brain. The GCA maps (from X to Y and from Y to X) underwent a 2 × 2 mixed-effect analysis to explore the main effects of condition (smoking status: non-smoking versus smoking), group (cognitive status: CU versus MCI), and the interactive effects of smoking × cognitive status. Covariates included age, sex, years of education, and grey matter volume were applied. We also adjusted for APOE ε4 allele status in some populations. The significance level was set at p < 0.001 at the voxel level and p < 0.05 at the cluster level after Gaussian random field (GRF) correction. Furthermore, EC values from regions showing significant differences among the four groups were extracted, with post-hoc analysis at p < 0.01, Bonferroni corrected.
LME regression model was applied to identify whether baseline EC values could predict cognitive decline in CDR, MMSE, ADNI_MEM, ADNI_EF, ADNI_LAN, and ADNI_VS using the ‘lme4’ package in R software with a significance level of p < 0.05. Age, sex, years of education, and follow-up time were included in the model as covariates.
Results
Demographics and neuropsychological assessments
Table 1 presents the demographic and neuropsychological assessments for four groups, with no significant differences in sex, age, and education among non-smoking CU, smoking CU, non-smoking MCI, and smoking MCI (p > 0.05). Significant differences in global cognitive function (MMSE, p < 0.001) and cognitive domains, including memory (ADNI_MEM, p < 0.001), executive function (ADNI_EF, p = 0.001), and language function (ADNI_LAN, p < 0.001) were observed among the four groups. Subsequent post-hoc analysis revealed that the two CN groups demonstrated significantly superior performance in memory, executive functions, and language compared to the two MCI groups (p < 0.05). No significant differences were found in visuospatial function (ADNI_VS, p = 0.061) among the four groups.
Demographic information and neuropsychological scales for the study population.
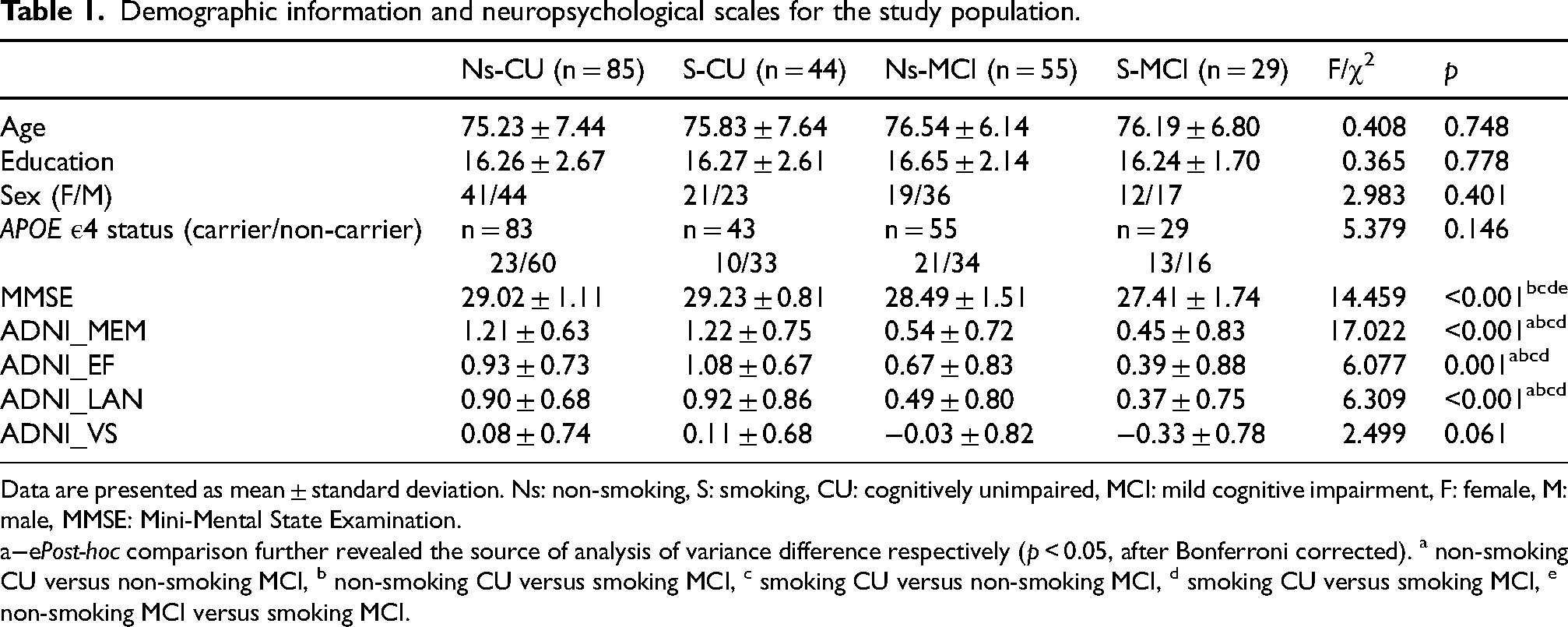
Data are presented as mean ± standard deviation. Ns: non-smoking, S: smoking, CU: cognitively unimpaired, MCI: mild cognitive impairment, F: female, M: male, MMSE: Mini-Mental State Examination.
Post-hoc comparison further revealed the source of analysis of variance difference respectively (p < 0.05, after Bonferroni corrected). a non-smoking CU versus non-smoking MCI, b non-smoking CU versus smoking MCI, c smoking CU versus non-smoking MCI, d smoking CU versus smoking MCI, e non-smoking MCI versus smoking MCI.
Interactive effects between smoking × cognitive status on EC
Interaction analyses were conducted among the four groups. The mixed-effects analysis revealed no significant difference in the interactive effect on the EC of DMN networks, specifically the PCC and aMPFC. However, in the non-smoking CU, smoking CU, non-smoking MCI, and smoking MCI groups, we observed significant alterations in the interactive effects of EC from the whole brain to the FIC and from the DLPFC to the whole brain. These findings are illustrated in Figure 1 (strength), Figure 2 (direction), and summarized in Table 2.
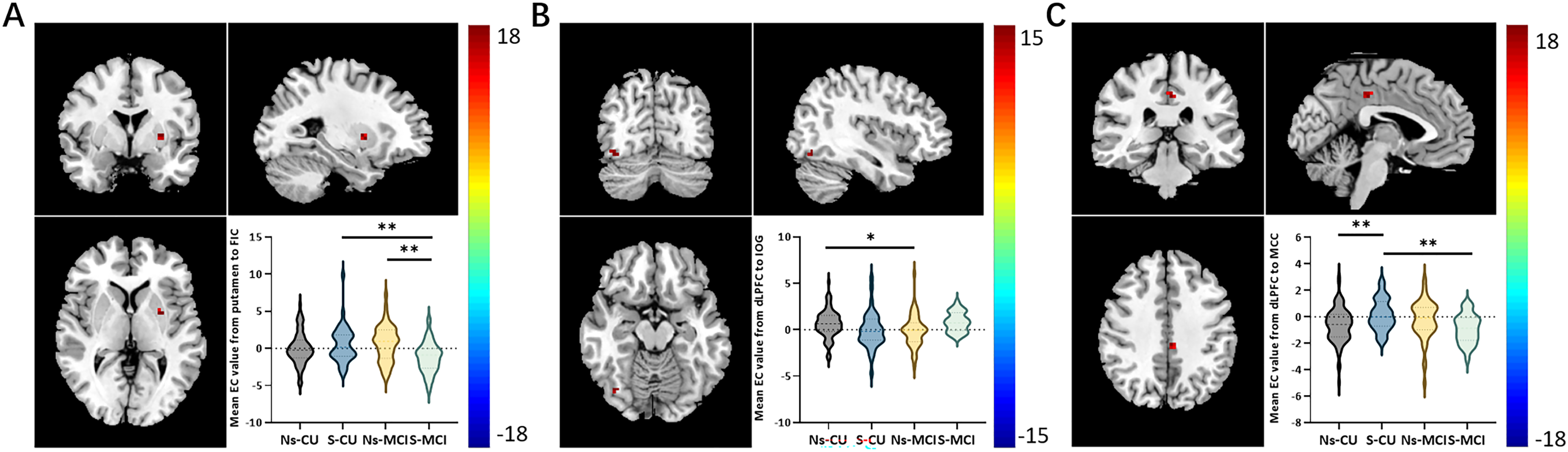
Representation of brain regions with interaction effects (smoking × cognitive state) on EC among non-smoking CU, smoking CU, non-smoking MCI, and smoking MCI. Brain regions showing significant differences of interaction effects in the causal effect from the left putamen to the FIC (A), from the DLPFC to the right IOG (B), and from the DLPFC to the left MCC (C) adjusting for age, sex, years of education, and grey matter volume (Gaussian random field corrected, p < 0.001 at the voxel level and p < 0.05 at the cluster level). EC: effective connectivity, CU: cognitively unimpaired, MCI: mild cognitive impairment, Ns: non-smoking, S: smoking, FIC: frontoinsular cortex, DLPFC: dorsolateral prefrontal cortex, IOG: inferior occipital gyrus, MCC: middle cingulate cortex.
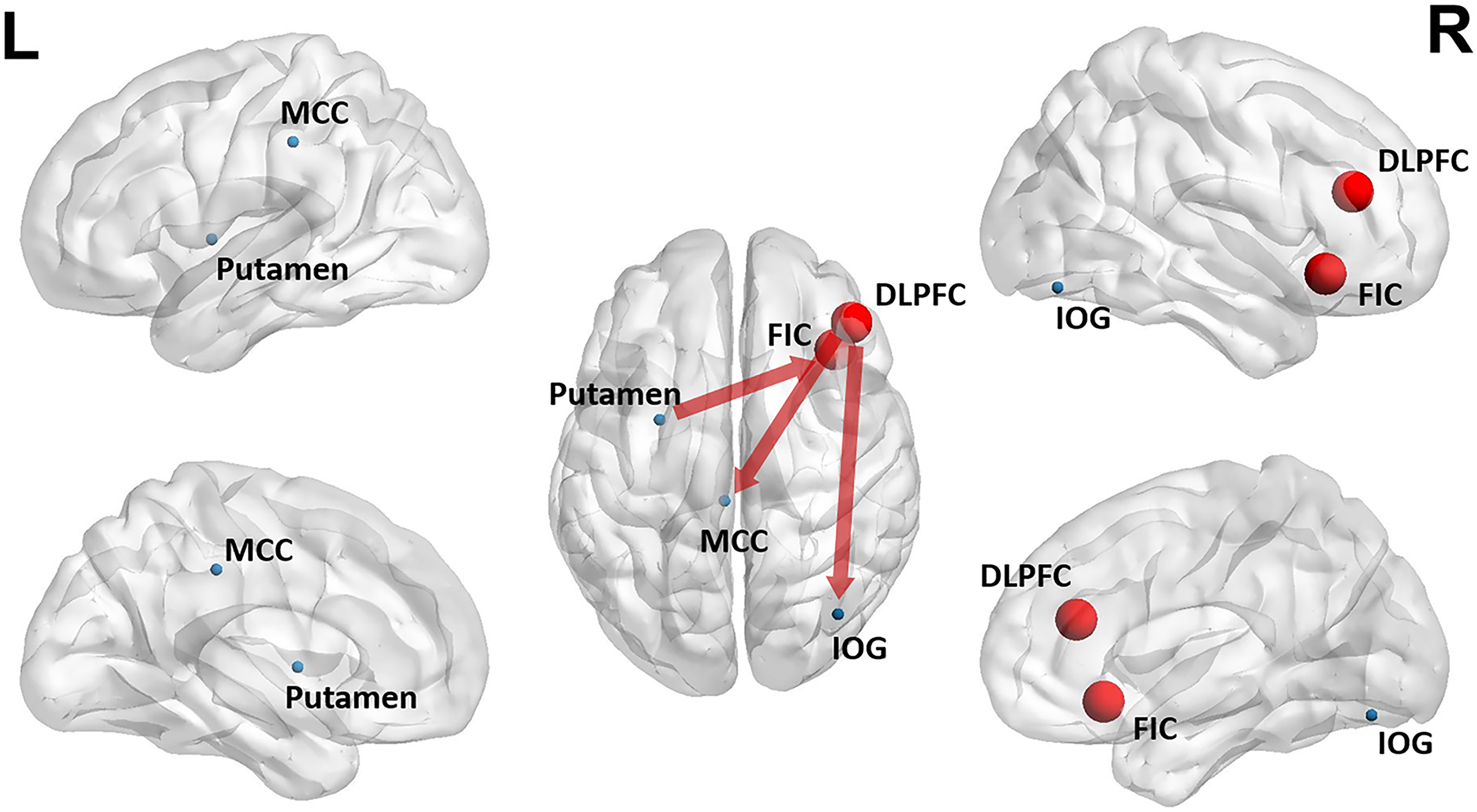
Sketch map of the differences in causal connectivity among non-smoking CU, smoking CU, non-smoking MCI, and smoking MCI.
Brain areas with significant group differences in Granger causality analysis among non-smoking CU, smoking CU, non-smoking MCI, and smoking MCI.
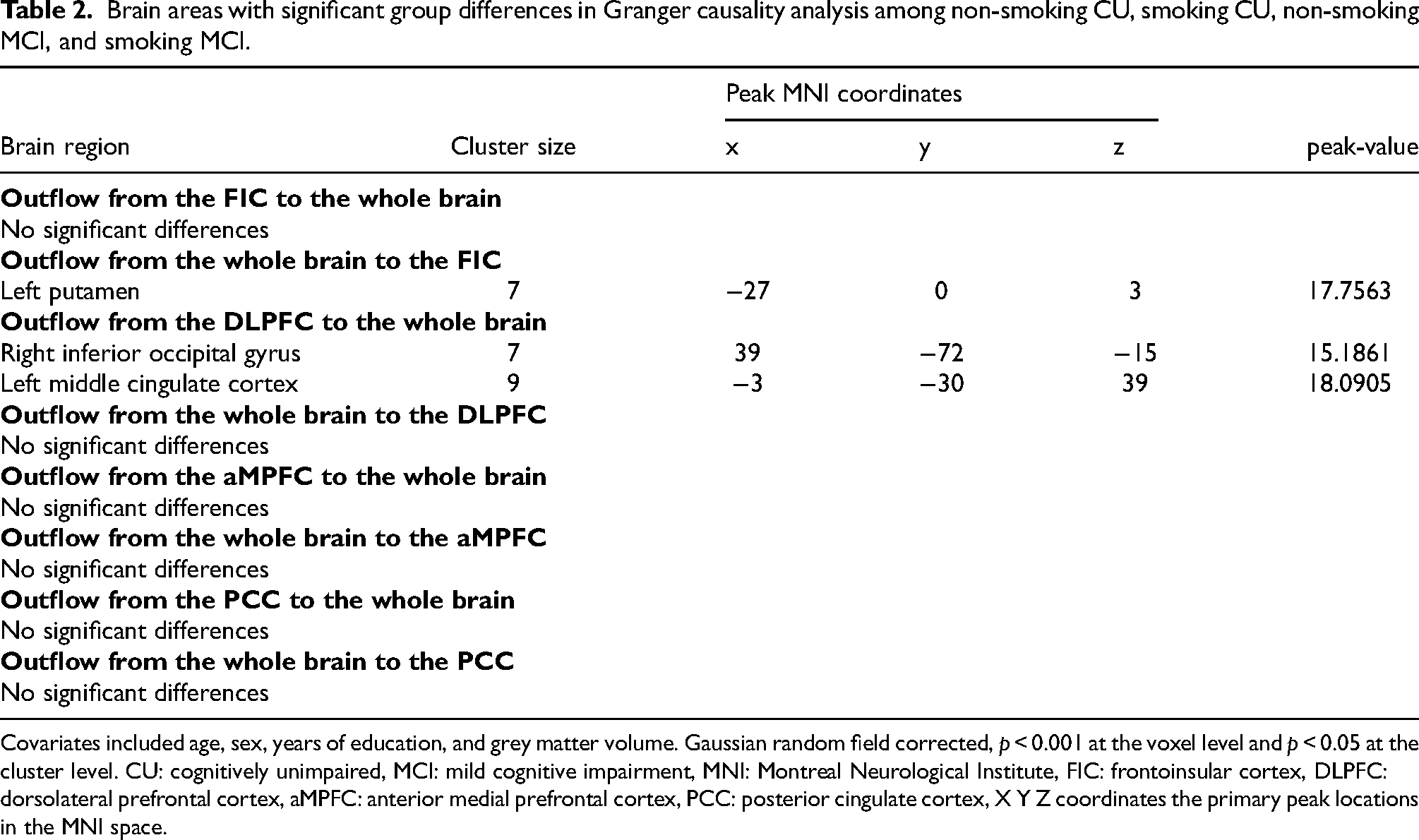
Covariates included age, sex, years of education, and grey matter volume. Gaussian random field corrected, p < 0.001 at the voxel level and p < 0.05 at the cluster level. CU: cognitively unimpaired, MCI: mild cognitive impairment, MNI: Montreal Neurological Institute, FIC: frontoinsular cortex, DLPFC: dorsolateral prefrontal cortex, aMPFC: anterior medial prefrontal cortex, PCC: posterior cingulate cortex, X Y Z coordinates the primary peak locations in the MNI space.
EC between the SN network and the whole brain
The mixed-effects analysis among the four groups revealed a significant difference in the EC from the whole brain to the FIC in the left putamen. Post-hoc analysis indicated that the smoking MCI group exhibited decreased EC from the left putamen to the FIC compared to the smoking CU group (p < 0.01, Bonferroni corrected, Figure 1A). Additionally, the smoking MCI group showed lower EC from the left putamen to the FIC compared to the non-smoking MCI group (p < 0.01, Bonferroni corrected, Figure 1A). This result remained significant after adjustment for APOE ε4 allele status (Supplemental Table 1 and Supplemental Figure 2). However, no significant differences were found in EC from the FIC to the whole brain among the four groups (Table 2).
EC between the ECN network and the whole brain
The mixed-effects analysis among the four groups showed significant differences from the DLPFC to the whole brain in the right inferior occipital gyrus (IOG) and left middle cingulate cortex (MCC). Post-hoc results showed that compared with the non-smoking CU, the non-smoking MCI displayed decreased EC from the DLPFC to the right IOG (p < 0.05, Bonferroni corrected, Figure 1B). Compared with the non-smoking CU, the smoking CU showed increased EC from the DLPFC to the left MCC (p < 0.01, Bonferroni corrected, Figure 1C). Compared with the smoking CU, the smoking MCI displayed decreased EC from the DLPFC to the left MCC (p < 0.01, Bonferroni corrected, Figure 1C). No significant difference was found in EC from the whole brain to the DLPFC among the four groups (Table 2).
Group effects of EC
Group effects (cognitive state) were analyzed among the four groups. Among non-smoking participants, the non-smoking MCI group displayed increased EC from the PCC to the whole brain in the left IOG compared to the non-smoking CU group (p < 0.01, Bonferroni corrected, Supplemental Table 2, Supplemental Figures 3 and 4). Additionally, the non-smoking MCI group showed decreased EC from the FIC to the whole brain in the left middle frontal gyrus (MFG) compared to the non-smoking CU group (p < 0.01, Bonferroni corrected). This result remained significant after adjustment for APOE ε4 allele status (Supplemental Table 3, Supplemental Figure 5). However, no statistically significant difference in group effect was found on the EC of the aMPFC and DLPFC (Supplemental Table 2).
Association and predictive ability of EC of cognitive decline
LME regression model was performed to analyze the ability of EC values to predict cognitive decline (Table 3). Following adjustment for age, sex, years of education, and follow-up time, the LME analysis reflected that ECPCC−to−IOG_L was associated with CDR (estimate 0.039; 95% CI [0.017, 0.061]; p = 0.001), ADNI_MEM (estimate −0.104; 95% CI [-0.175, −0.033]; p = 0.004), and ADNI_EF (estimate −0.081; 95% CI [-0.152, −0.010]; p = 0.026). ECFIC−to−MFG_L was associated with CDR (estimate −0.033; 95% CI [-0.058, −0.007]; p = 0.012). In addition, ECPutamen_L−to−FIC was related to MMSE (estimate 0.021; 95% CI [0.048, 0.205]; p = 0.002). In addition, the LME regression revealed the ECPCC−to−IOG_L can predict MMSE changes (estimate 0.091; 95% CI [-0.181, −0.001]; p = 0.049). ECPutamen_L−to−FIC show a significant prediction for ADNI_EF changes longitudinally (estimate 0.016; 95% CI [0.002, 0.030]; p = 0.026).
Statistical analysis of linear mixed-effect regression for EC values and cognitive function.
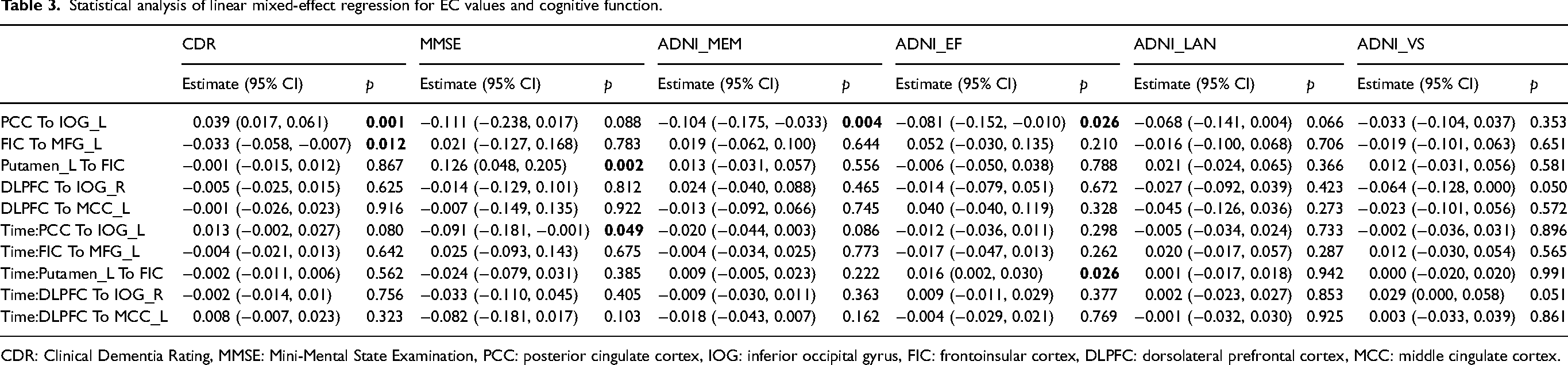
CDR: Clinical Dementia Rating, MMSE: Mini-Mental State Examination, PCC: posterior cingulate cortex, IOG: inferior occipital gyrus, FIC: frontoinsular cortex, DLPFC: dorsolateral prefrontal cortex, MCC: middle cingulate cortex.
Discussion
Our study, using the GCA method, explored how smoking affects the causal relationship between RSNs and the whole brain in CU and MCI patients. Specifically, the key findings of our study are as follows: (1) Smoking MCI patients exhibited decreased EC from the putamen to the FIC compared to both smoking CU and non-smoking MCI individuals. (2) Among non-smokers, MCI patients showed lower EC from the DLPFC to the IOG compared to CU individuals. (3) Smoking CU individuals displayed significantly higher EC from the DLPFC to the MCC than non-smoking CU individuals and smoking MCI patients. In addition, ECPCC−to−IOG_L was associated with cognitive status, memory, executive function, and longitudinal global cognitive function, while ECPutamen_L−to−FIC was associated with global cognitive function and longitudinal executive function.
Our study revealed a significant interaction effect between smoking and cognitive status on the EC between the SN/ECN and the whole brain, but not between the DMN and the whole brain. Consistent with previous findings, smoking primarily affects the FC and EC of the SN and ECN.19,20,23 In our study, smoking individuals with MCI exhibited significantly lower EC from the putamen to the FIC compared to both smoking CU individuals and non-smoking individuals with MCI. Smoking MCI patients showed decreased EC from ECN to the whole brain relative to smoking CU. The putamen is an essential part of the dorsal striatum. Previous imaging studies and animal experiments demonstrated the crucial role of putamen in inhibitory control and addiction.34,35 Cortical-striatal-thalamic-cortical loop circuits, as part of the SN, play a central role in cognitive function. A magnetic resonance study reported a significant reduction in putamen volume in probable AD patients, which correlated linearly with impaired global cognitive performance. 36 Early-onset AD patients exhibited more pronounced β-amyloid deposition and increased glucose metabolism in the striatum, particularly the putamen, compared to late-onset AD patients. 37 The DLPFC is also susceptible to smoking. A voxel-based morphometry study demonstrated gray matter loss in several brain regions, including the MCC, associated with pack-years of smoking exposure. 38 The DLPFC plays a critical role in working memory, selective attention, and task management,39,40 and it possesses neuroplasticity that enables compensation for dysfunction in other brain regions.41,42 ECN was reported to act as a compensatory network for cognitive reserve in AD patients. In the early AD stage, DLPFC dysfunction was found. 43 Previous research reported that MCI patients converted to AD displayed significantly decreased FC in DLPFC compared to non-converters. 44
Smoking MCI patients showed significant decline in EC of the SN-related and DLPFC-related networks. Although smoking can lead to a temporary increase in brain functional connectivity in the short term due to the stimulating effect of nicotine, chronic smoking can impair brain network functional connectivity and cognitive function in the long term.16,45 The brain function and network connectivity is impaired in MCI patients. A functional MRI study demonstrated reduced ECN connectivity in the ACC and DLPFC in patients with amnestic MCI. 46 In addition, MCI patients are often complicated by vascular damage, and presented decreased cerebral blood flow.47,48 Smoking can reduce cerebral blood perfusion and deteriorate the vascular regulatory function, which further affects brain function.49,50 We speculate that when individuals are either smokers without cognitive impairment or in the stage of MCI without smoking, the compensatory capacity of the whole brain for SN function is intact. However, when smokers experience cognitive impairment, the damage to brain network function is more likely to exceed the compensatory capacity, leading to disease exacerbation. This provides a basis for smoking cessation strategies for AD patients.
In the analysis of group effect, we found that non-smoking MCI showed increased EC from PCC to IOG compared to non-smoking CU while decreased EC from FIC to MFG. The results of previous studies on the functional and EC pattern of DMN and SN in MCI patients were inconsistent.25,51,52 The MFG is known to be involved in attentional switching and control. 53 MCI patients tend to exhibit decreased FC between different brain networks and increased FC within the SN in the MFG when compared to CU individuals. 54 Our findings align with a recent meta-analysis, which indicated that the interactions of the SN with other networks primarily occur in the MFG in individuals with MCI. 52
APOE ε4 allele is associated with a higher risk of Alzheimer's disease and other brain health problems. After adjustment for APOE ε4 allele status, the difference in EC from DLPFC to the whole brain among the four groups disappeared. The APOE ε4 allele does have an impact on brain function. Carriers of the APOE ε4 allele showed greater activity during working memory in the right DLPFC. 55 APOE ε4 allele carriers exhibited decreased EC from the PCC to the whole brain. 56
We found a higher EC from the PCC to the IOG was associated with a higher CDR score and worse memory and executive function. The higher the EC from the FIC to the MFG, the lower the CDR score. A higher EC from the putamen to the FIC was associated with a higher MMSE score. Previous studies have demonstrated the critical role of DMN in cognitive processes. 57 Disruptions in DMN connectivity are associated with cognitive decline. 58 The SN plays a critical role in regulating the activity of the DMN, and impaired SN function can lead to inefficient cognitive control. 59 Functional MRI analyses showed differences in FC and EC patterns between stable and progressive MCI.44,60 Our findings from the LME model analysis suggested that EC values could serve as predictors of disease progression in AD. EC from the Putamen to the FIC, which was decreased in smoking MCI patients, could significantly predict longitudinal changes in executive function. It provides more research evidence that smoking exacerbates the progression of cognitive impairment.
This study still has several limitations. Firstly, the available smoking data in the ADNI database, such as smoking history and nicotine dependence scales like the Fagerström Test for Nicotine Dependence (FTND), were not detailed enough. In future studies, it would be beneficial to collect more comprehensive smoking history data, including smoking status and FTND scores, to better analyze the impact of smoking on the MCI population. Secondly, the lack of neuropathological data, such as cerebrospinal fluid or positron emission tomography, to confirm cognitive status based on the Amyloid/Tau/Neurodegeneration framework is another limitation. Reliance solely on symptom-based diagnosis may result in misclassification of participants. Thirdly, there are some unresolved issues, including the discrepancy between the sampling rate (which is equivalent to the MRI repetition time) and the neuronal response, as well as the potential confounding effects of hemodynamic delays. 61 There is currently no established standard for coefficient GCA applications in resting-state fMRI data processing. Future studies could benefit from the integration of electrophysiological data to address these limitations and reduce potential errors. Fourthly, given that the ADNI database includes individuals from different locations, this could potentially introduce confounding bias. Lastly, the present study was limited by its cross-sectional design and modest sample size. Prospective longitudinal investigations with expanded cohorts were needed to definitively establish the causal mechanisms underlying our observations.
Conclusion
We examined the impact of smoking on the EC between RSNs and the entire brain in a non-demented cohort. Our findings revealed that smoking MCI patients displayed decreased EC compared to non-smoking MCI patients. Specifically, we observed reduced EC from the left putamen to the FIC and from the DLPFC to the left MCC in smoking MCI patients. ECPCC−to−IOG and ECPutamen−to−FIC were effective predictors of MMSE and executive function, respectively, and thus could be potential predicting biomarkers for disease progression. Our results suggest that smoking may diminish the compensatory effects of brain function in individuals with early-stage AD. Hence, smoking cessation is crucial for the management of AD.
Supplemental Material
sj-docx-1-alz-10.1177_13872877251333152 - Supplemental material for Smoking alters effective connectivity of resting-state brain networks in mild cognitive impairment
Supplemental material, sj-docx-1-alz-10.1177_13872877251333152 for Smoking alters effective connectivity of resting-state brain networks in mild cognitive impairment by Tianyi Zhang, Xiao Luo, Qingze Zeng, Kaicheng Li, Yi Chen, Yan Sun, Lumin Leng, Guoping Peng, Minming Zhang, Zhirong Liu and in Journal of Alzheimer's Disease
Footnotes
Acknowledgments
The authors thank all the participants, researchers, and clinicians in the ADNI database.
Data collection and sharing for the Alzheimer's Disease Neuroimaging Initiative (ADNI) is funded by the National Institute on Aging (National Institutes of Health Grant U19AG024904). The grantee organization is the Northern California Institute for Research and Education. In the past, ADNI has also received funding from the National Institute of Biomedical Imaging and Bioengineering, the Canadian Institutes of Health Research, and private sector contributions through the Foundation for the National Institutes of Health (FNIH) including generous contributions from the following: AbbVie, Alzheimer's Association; Alzheimer's Drug Discovery Foundation; Araclon Biotech; BioClinica, Inc.; Biogen; Bristol-Myers Squibb Company; CereSpir, Inc.; Cogstate; Eisai Inc.; Elan Pharmaceuticals, Inc.; Eli Lilly and Company; EuroImmun; F. Hoffmann-La Roche Ltd and its affiliated company Genentech, Inc.; Fujirebio; GE Healthcare; IXICO Ltd; Janssen Alzheimer Immunotherapy Research & Development, LLC.; Johnson & Johnson Pharmaceutical Research & Development LLC.; Lumosity; Lundbeck; Merck & Co., Inc.; Meso Scale Diagnostics, LLC.; NeuroRx Research; Neurotrack Technologies; Novartis Pharmaceuticals Corporation; Pfizer Inc.; Piramal Imaging; Servier; Takeda Pharmaceutical Company; and Transition Therapeutics.
ORCID iDs
Ethical considerations
The ADNI database was approved by the Institutional Review Board at each participating center.
Consent to participate
Informed written consent was obtained from all participants at each institution.
Consent for publication
Not applicable
Author contributions
Tian Yi Zhang (Conceptualization; Data curation; Formal analysis; Investigation; Writing – original draft); Xiao Luo (Conceptualization; Funding acquisition; Methodology; Writing – review & editing); Qingze Zeng (Formal analysis; Writing – review & editing); Kaicheng Li (Writing – review & editing); Yi Chen (Writing – review & editing); Yan Sun (Writing – review & editing); Lumin Leng (Writing – review & editing); Guoping Peng (Funding acquisition; Writing – review & editing); Minming Zhang (Methodology; Supervision; Writing – review & editing); Zhirong Liu (Conceptualization; Project administration; Writing – review & editing).
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was financially supported by the Ministry of Science and Technology of the People's Republic of China [grant number 2022YFC3602604], the National Natural Science Foundation of China [grant numbers 82071182, and 82271936], Zhejiang Traditional Chinese Medicine Youth Talent project [grant number 2022ZQ057].
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability
The datasets generated and/or analyzed in our study are available in the ADNI database (adni.loni.usc.edu).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
