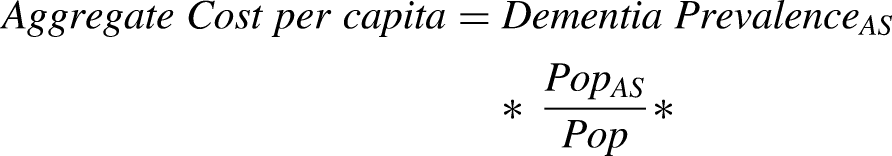Abstract
Background
There are 5.5 million people living with dementia in the United States (US), with the cost of unpaid care making up a significant portion of the care costs.
Objective
Summarize variation in the cost of dementia care across the US and examine the association between medical spending and costs of unpaid care at the state level.
Methods
We estimated total cost for dementia by combining recent medical spending estimates from the Disease Expenditure project and unpaid care cost estimates from Lastuka and colleagues. Hours of unpaid care were valued as the hourly wage of a home health aide. We used linear regression to measure the association between the cost of unpaid care and medical spending. The spending that would have occurred if unpaid care had been provided by professional home health care workers was used to measure the cost of unpaid care.
Results
The annual cost of care attributable to dementia in 2019 was $53,502 (95% uncertainty interval [UI] 46,135–60,594) per case. The contribution of unpaid care to total costs varied by state, ranging from 70.2% (95% UI 64.3–75.4) in the District of Columbia to 89.9% (95% UI 87.8–91.5) in Arizona. We found that higher costs of unpaid care were associated with lower medical spending on nursing facility care.
Conclusions
The large variation in total costs of dementia shows that the economic burden of dementia care is distributed unevenly throughout the US.
Keywords
Introduction
There are currently 5.5 million people in the United States (US) who are living with Alzheimer's disease and other dementias, 1 and this number is expected to rise to 10.5 million by 2050. 2 A hallmark of the progression of dementia is loss of functional ability, requiring additional supervision and assistance for activities of daily living. These growing care needs and the gradual decrease in the fraction of population that is working age caused by population aging in the US are expected to put increasing strain on the health care system, as well as family and other unpaid caregivers. 3 Given that people living with dementia need a substantial amount of personal care as their condition progresses, both medical spending (which includes pharmaceuticals, ambulatory care, inpatient care, emergency department care, paid home health care, and nursing home care and is sometimes referred to as “direct” costs) and the costs of unpaid care (which are sometimes referred to as “indirect costs”) are high; however, little is known about the tradeoff between these two cost categories.
Several studies have measured medical spending and the cost of unpaid caregiving to arrive at a total cost of dementia,4–9 and they consistently find that the cost of unpaid care for dementia makes up a large proportion of costs, ranging from 48% 6 to 60% 8 of the total cost of dementia care in the US. Unpaid care can be valued in several ways; however, the large contribution of unpaid care to dementia costs is consistent across methodologies. A gap in this literature is the lack of subnational cost estimates. Most studies are conducted in one country at the national level and are therefore unable to capture variation across regions. 9However, variation in the type of care utilized has been captured at an individual level. Prior work on elder care that was not specific to dementia care did find evidence that unpaid care is a substitute for paid home health visits, nursing home stays, nights spent in the hospital, and number of physician visits. 10
We are building from estimates of medical spending and costs of caregiving attributable to dementia. In these estimates, comorbidities have been adjusted for, and what is being measured is spending that is caused by dementia. We quantify costs of unpaid care by measuring the cost that would be paid if all unpaid care hours were replaced with a paid home health aide. 11
We add medical spending and the cost of unpaid care to arrive at a “total cost of care” for each US state. While several previous studies have used the term societal cost to mean the sum of medical spending and costs of unpaid care,4–6 we avoid this term because other studies include additional nonmedical costs such as travel costs to doctor appointments, home modifications that were not covered by insurance, and health impacts to family caregivers. 12 We decompose this total cost of care in order to examine the effect of each state's age profile, age-standardized dementia prevalence, medical spending costs, unpaid caregiving time, and the home health aide wages that were used to estimate the value of unpaid caregiving time. We also examine the association between medical spending and costs of unpaid care to look for evidence of substitution between these cost categories. We considered medical spending all together, as well as specific components of medical spending such as nursing facility care, paid home health care, and emergency department visits. Paid home health care and nursing facility care provide some of the same services as family caregivers; therefore, those costs were the most likely to exhibit a relationship with the costs of unpaid care. Emergency department costs were considered based on a known association between dementia and increased emergency department visits.13,14 Several studies have examined the role that family caregivers play in emergency department use for people living with dementia, but no link has been drawn between the amount of care provided and emergency department visits.15–17 In this work, we add to the body of literature on dementia costs in two ways: First, we present novel estimates of total costs of these two types of costs dementia care at the US state level. Second, we characterize the relationship between medical spending and unpaid care costs.
Methods
Medical spending estimates
Medical spending estimates for people living with dementia from 2010–2019 were extracted from ongoing work by Dieleman and colleagues for the Institute for Health Metrics and Evaluation's Disease Expenditure project. The Disease Expenditure project created estimates for medical spending by US county, payer (Medicare, Medicaid, private insurance, and out-of-pocket spending), type of care (ambulatory, inpatient, emergency department, long-term care, pharmaceuticals, and dental), age and sex group for 148 conditions, including dementia. These estimates were created by combining and harmonizing data from Medicare, Medicaid, commercial claims, hospital administrative data, and the Medical Expenditure Panel Survey. The long-term care spending category includes both short-term and long-term nursing facility care. It includes spending by Medicare, Medicaid, private insurance, and out-out-pocket spending. After harmonization, claims data were aggregated to reflect health system encounters. Encounters were assigned a primary diagnosis, and spending was disaggregated across all comorbidities based on regression analysis. Estimates were aggregated by payer, type of care, health condition, age and sex group, and US county and state, and adjusted to align with estimates from the National and State Health Expenditure Accounts. A comprehensive list of data sources can be found in Supplemental Tables 1–7, while details are extensively outlined in previously published research. 18 Spending estimates were adjusted for comorbidities so that each dollar spent was assigned to only one condition and represents spending that is attributable to this health condition (rather than merely being spending that is on someone with this condition). All 147 non-dementia conditions are included in the adjustment model; however, some conditions never co-occur with dementia and have an adjustment factor of zero. Spending estimates were adjusted for inflation and reported in 2019 US dollars.
Cost of unpaid dementia care
For the cost of unpaid dementia care, we used recently published state-level estimates of the replacement cost of care from 2010–2019, created by Lastuka and colleagues. 11 The replacement cost estimates were based on the cost of replacing each hour of care with a home health aide. Home health aide cost was estimated using data from the Bureau of Labor and Statistics Occupational Employment and Wage Statistics tables and Genworth Cost of Care Surveys.19,20 Genworth defines home health aides as those who offer assistance with activities of daily living (e.g., bathing) but not medical care (e.g., injections). These cost estimates were adjusted for inflation and reported in 2019 US dollars. In Lastuka and colleagues, the home health aide wages used to indirectly quantify unpaid hours of caregiving were also adjusted for regional purchasing power based on price indices created by the Bureau of Economic Analysis; however, in this work, no regional adjustments were made. The hours of care were estimated using survey data from the Health and Retirement Study, the National Health and Aging Trends Study, and the Behavioral Risk Factor Surveillance System. Supplemental Table 8 provides a list of all data sources and the years available that were used to create the unpaid care cost estimates.
Care estimates from these three sources were harmonized to account for key differences in survey methodology. The Health and Retirement Survey and National Health and Aging Trends Study provides national estimates, while the Behavioral Risk Factors Surveillance System provides state-level estimates. However, these state-level caregiver estimates were not available for every state each year. We used spatiotemporal Gaussian process regression to interpolate missing state-years. 21 Spatiotemporal Gaussian process regression is a Bayesian modeling process in which covariates that are available for all states and years are used to form a prior estimate for caregiving hours. This estimate is then updated using the available survey data. We were not able to disaggregate by age or sex of the care recipient due to a lack of information about the care recipient's age or sex in the BRFSS survey.
The resulting estimates reflect all caregiving time, including time spent assisting with activities of daily living, instrumental activities of daily living, and supervision. The caregiving hours were then adjusted for comorbidities to reflect only the hours of care attributable to dementia (similar to the process used for medical spending described above). Data from the National Health and Aging Trends Study were used to model the impact of comorbidities on caregiving hours, and the comorbidities found to be significant in our model were depression, high blood pressure, and stroke. More detail on the methodology used for the replacement cost estimates can be found in the Supplemental methods and in Lastuka and colleagues. 11 For both medical spending and costs of unpaid caregiving, national cost estimates were created by aggregating per-case costs using dementia prevalence rates from the Global Burden of Disease Study. 2
Drivers of costs
Using population numbers from the Global Burden of Disease Study,
21
we also estimated per capita costs of dementia for each state. The per capita cost of dementia is a function of each state's dementia prevalence rate and age profile as well as medical spending and costs of unpaid care per case in that state. The per capita cost of dementia care can be written as follows to facilitate factor decomposition:
Using this equation, we performed decomposition of 2019 costs, based on methods developed by Das Gupta, 22 to quantify the contribution of each of these factors in explaining the difference in cost per capita relative to the national mean.
Statistical analysis
We assessed the relationship between per-case medical spending and the costs of unpaid dementia care using linear regression. We estimated a model in which medical spending on dementia was the outcome, as well as models with the cost of paid home health care, nursing facility care, and emergency department care as the outcomes. The outcome variable and the cost of unpaid care were both transformed using the natural log. Therefore, the regression coefficient represents the amount that the outcome variable changes when the independent variable (cost of unpaid care) changes by 1%.
We used Bayesian model selection 23 to select control variables that should be included in the regression from the following list: obesity prevalence, smoking prevalence, hearing loss, low exercise, high blood pressure, air pollution, hospital beds per capita, median income, and an index that reflects health care access and quality.24–27 Supplemental Table 9 lists the covariates that were selected for each regression. To control for systematic non-linear effects of time, we included individual year-specific time indicators. We used robust standard errors to account for the fact that the residual terms are likely grouped by state. We incorporated data uncertainty by sampling 50 draws from the distribution of medical spending estimates and 1000 draws from the distribution of unpaid care cost estimates, and we performed all calculations at the draw level.
We followed GATHER requirements for accurate and transparent health estimates reporting (Supplemental Table 10).
Results
Summary of costs by state
In 2019, the total cost of dementia care in the US was $277 billion (95% uncertainty interval [UI] 239–313), with $53 billion (95% UI 51–55) of medical spending. The per-case cost was $53,502 (95% UI 46,135–60,594). Medical spending on dementia was composed of $7519 (95% UI 7157–7843) for nursing home care, $560 (95% UI 509–615) for home health care, $172 (95% UI 143–198) for pharmaceuticals, $656 (95% UI 612–705) for ambulatory care, $62 (95% UI 56–66) for emergency department care, and $814 (95% UI 778–847) for inpatient care. The cost of unpaid dementia care was $43,719 (95% UI 36,498–50,671), making up 80.8% (95% UI 77.8–83.2) of the total cost. Medical spending on dementia by age and sex is shown in Supplemental Table 11. Costs of unpaid care were not estimated by age or sex due to data limitations.
Medical spending, cost of unpaid care, and total costs per case in 2019 (2019 USD).
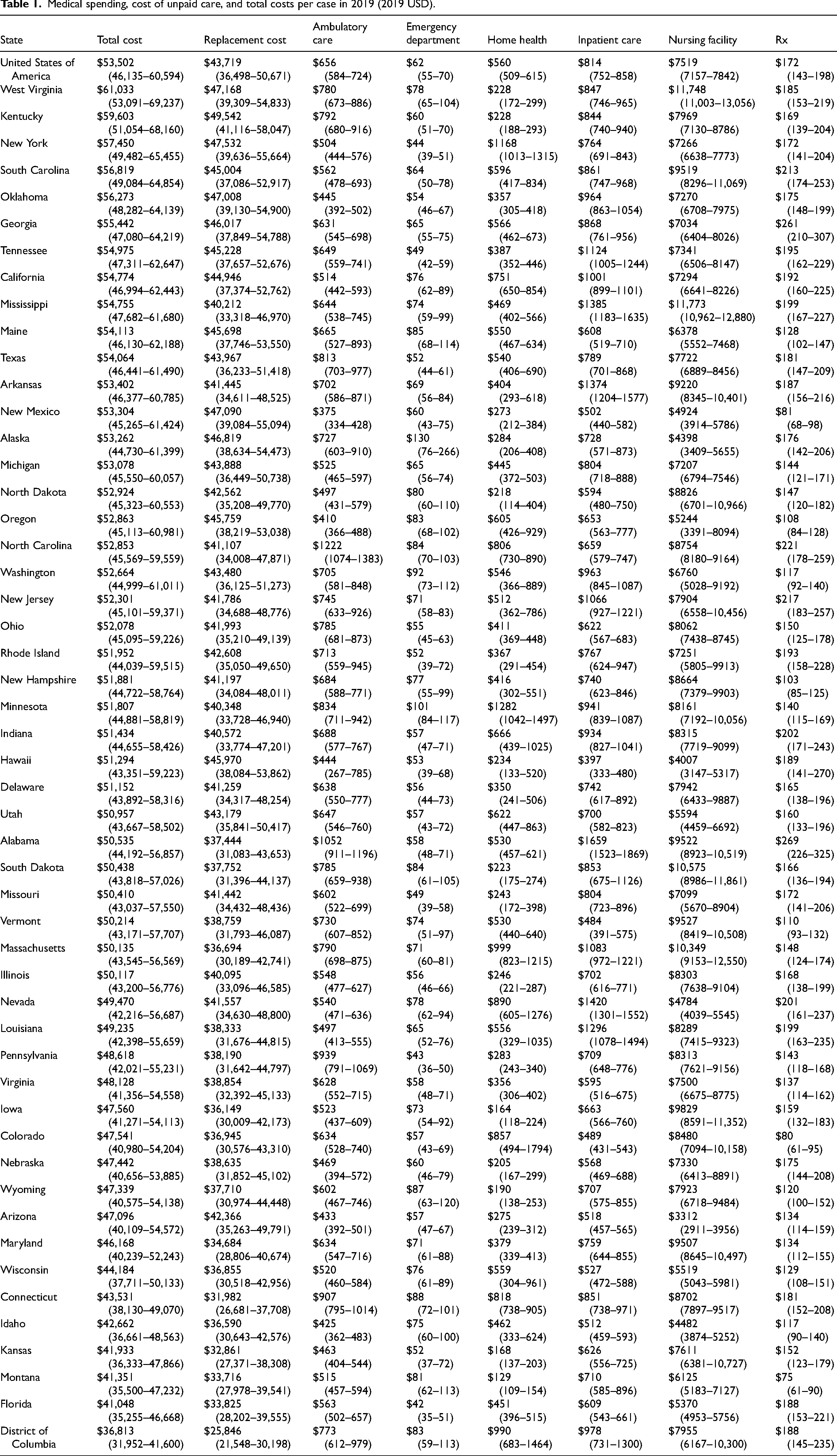
Figure 1 shows the total cost of dementia care for each state in 2019. This cost ranged from $36,813 (95% UI 31,952–41,600) per case in the District of Columbia to $61,033 (95% UI 53,091–69,237) per case in West Virginia. The contribution of the cost of unpaid care to the total cost ranged from 70.2% (95% UI 64.3–75.4) in the District of Columbia to 89.9% (95% UI 87.8–91.5) in Arizona.
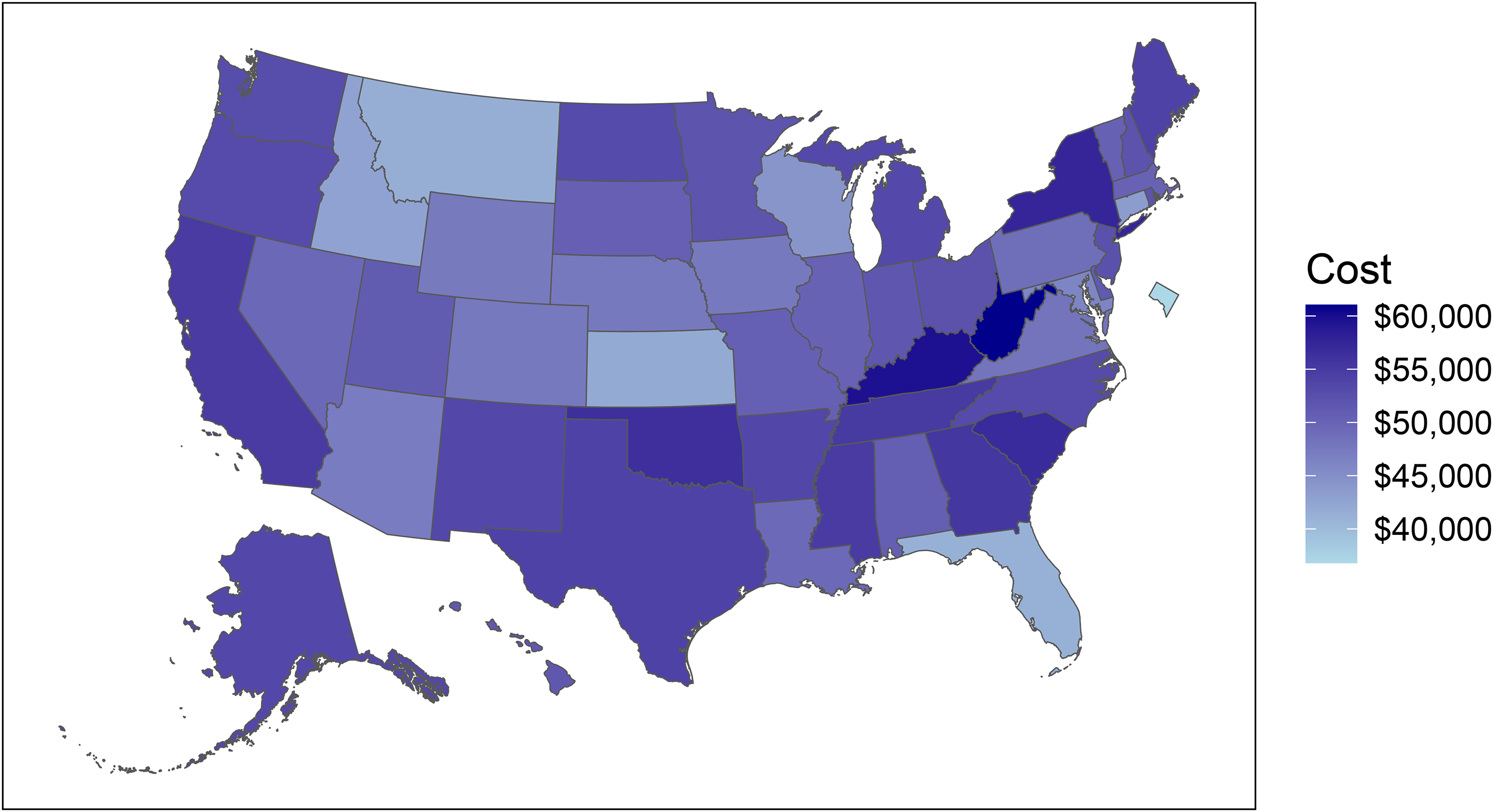
Total per-case cost (medical spending + unpaid care cost) in 2019 (2019 USD).
Table 1 shows the contribution of each cost component by state in 2019. It is ranked in decreasing order of total cost per case, with West Virginia, Kentucky, and New York having the highest total costs and Montana, Florida, and the District of Columbia having the lowest costs.
Medical spending on dementia was separated into six types of care: ambulatory care, emergency department care, paid home health care, inpatient care, nursing facility care, and pharmaceutical costs. Nursing facility costs are the main driver of dementia medical spending, making up 76.9% (95% UI 74.5–78.9) of all medical spending. Mississippi, West Virginia, and South Dakota have the highest nursing home costs, while Arizona, Hawaii, and Alaska have the lowest nursing home costs.
Cost drivers
Figure 2 shows how each state's age profile, age-standardized dementia prevalence, medical spending, caregiving time, and home health aide wages contribute to its total cost. The states are ordered from highest to lowest per capita cost. The ordering is similar but not identical to the per-case costs. For example, Kentucky has the second highest per-case costs but drops to seventh place in per capita terms due to its dementia prevalence rate being less than the national average. West Virginia has the highest per capita costs, which are $531 (95% UI 246–740) above the national mean. While the cost of a home health aide in West Virginia is less than the national average (shown by the yellow bar having a negative value), the other four cost components are higher than the national average. Figure 2 shows that the number of unpaid care hours plays a large role in the variation of per capita cost across the US, relative to the other cost components.
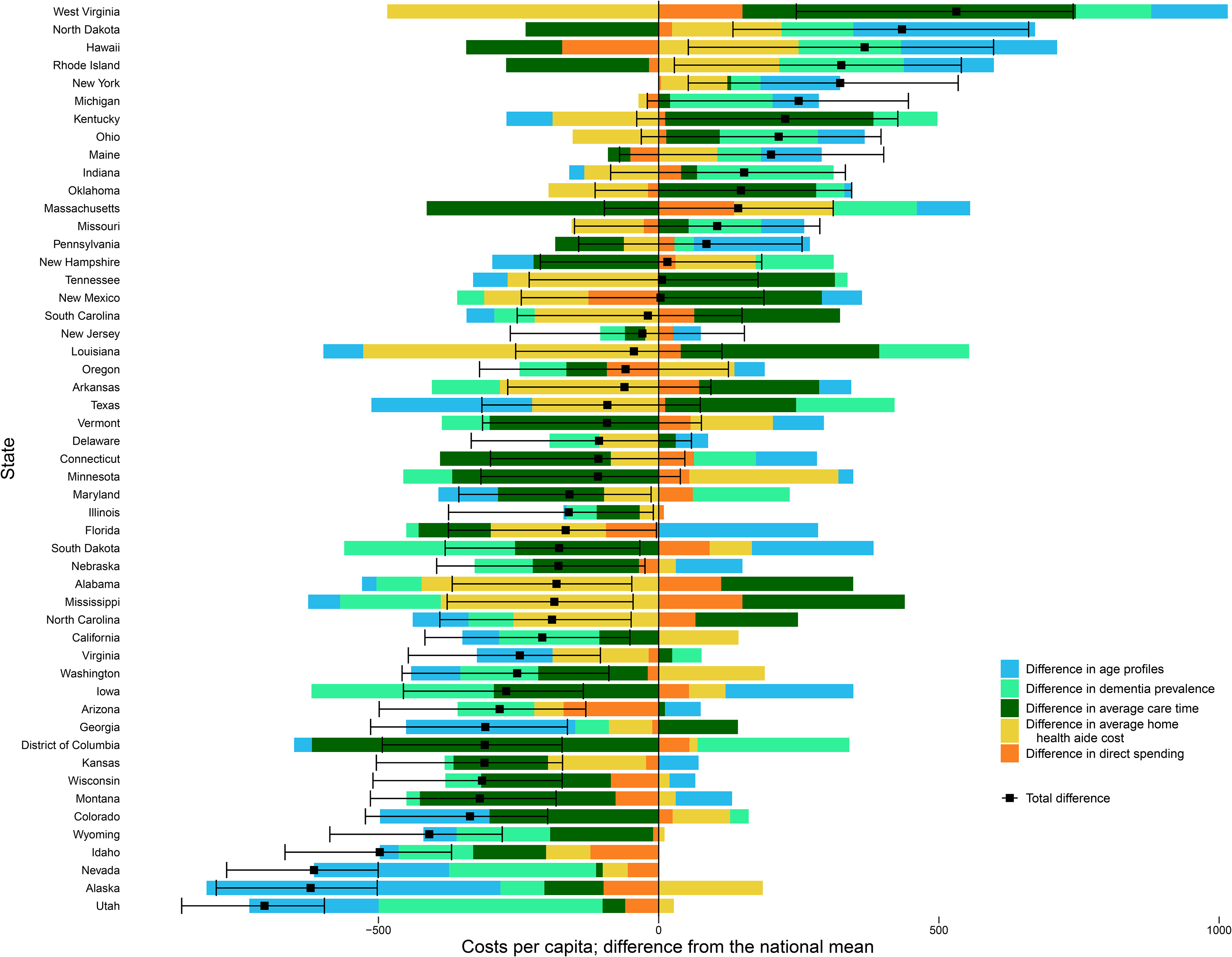
Per capita cost decomposition (2019 USD). Horizontal bars represent US dollars. The small square represents the total difference between a given state's per capita cost and the national mean per capita cost, and a 95% confidence interval is shown around the square.
Relationship between medical spending on dementia and costs of unpaid dementia care
We found an inverse relationship between unpaid care costs and nursing facility care costs (Figure 3). The regression coefficient on medical spending was −0.3, suggesting that a 10% increase in unpaid care costs is associated with a 3% (95% UI 1–5%) decrease in medical spending on dementia. Unpaid care costs did not have a significant association with paid home health care costs or emergency department costs. This decrease in medical spending appears to be driven by nursing home costs. We found that a 10% increase in unpaid care costs is associated with a 4% (95% UI 2–7%) decrease in nursing home costs.
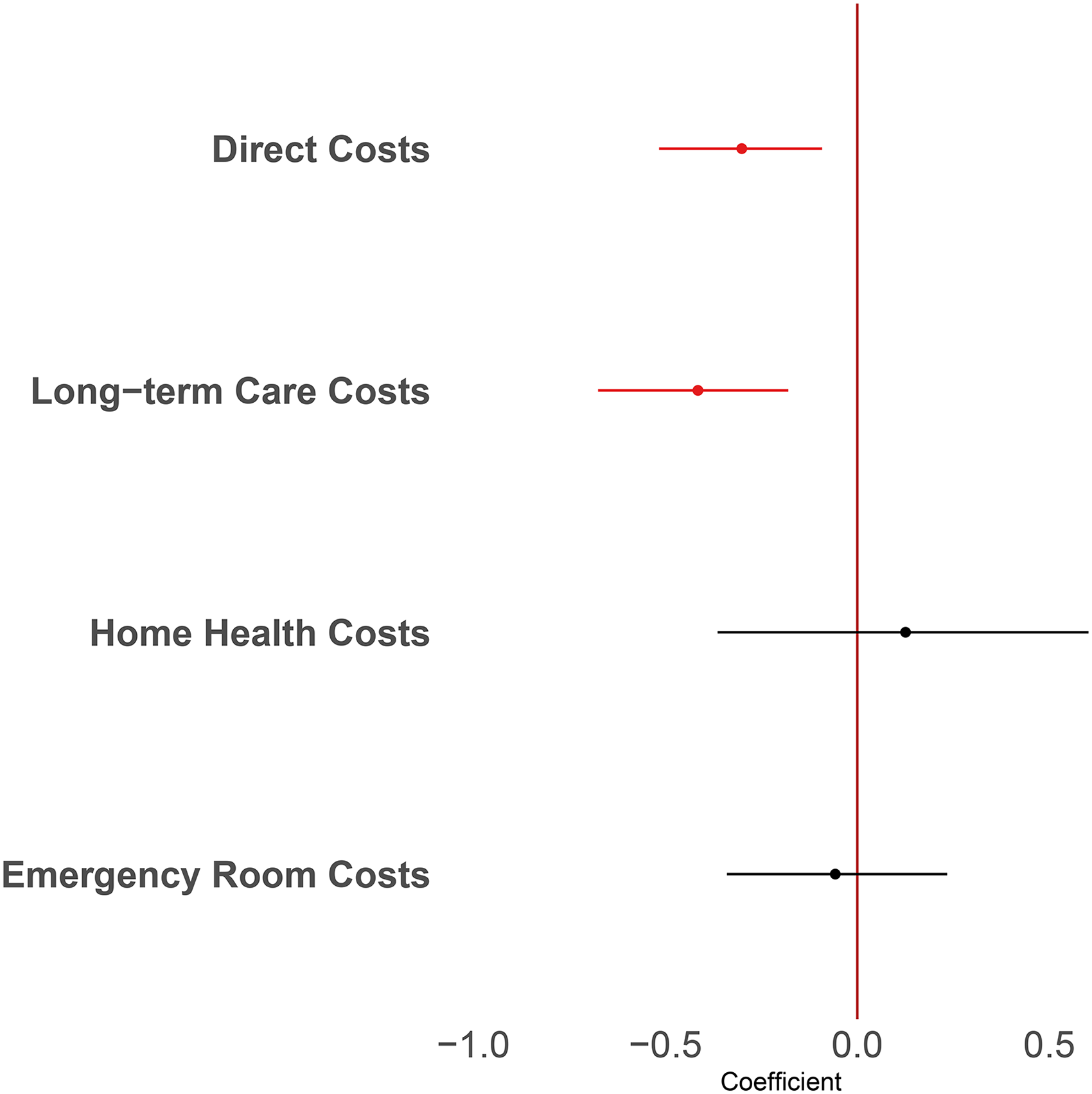
Regression coefficient for the replacement cost of care, with 95% confidence intervals. Coefficients for direct costs and long-term care costs are statistically significant.
Discussion
We combined comprehensive estimates of medical spending on dementia and costs of unpaid dementia care to arrive at a total cost of dementia care. The annual total cost of dementia care in 2019 was more than $250 billion, with over three-quarters of this figure representing the cost of unpaid care. We found that the cost of dementia care varies widely by state. The states with the lowest per-case costs are Florida, DC, and Montana. Low total costs are mainly driven by low unpaid care costs; however, three of the five least expensive states also have below-average costs for nursing home care.
Our national estimate of $277 billion for the total cost of dementia aligns closely with the forecasted estimate from Hurd and colleagues for the year 2020, which was $296 billion when adjusted to 2019 USD. 4 Relative to our estimates, Hurd and colleagues found that the cost of unpaid care made up a smaller proportion of the total cost but still accounted for approximately two-thirds of the cost of dementia care. This difference was driven by both our cost of unpaid care estimates being higher and our medical spending estimates being lower than Hurd's estimates. A key difference between our cost of unpaid care estimates and those from Hurd is that ours incorporate all caregiving activities, including supervision time, while Hurd's methodology limits caregiving time to support for activities of daily living and instrumental activities of daily living. The medical spending estimates differ for multiple reasons. First, our spending estimates were created in conjunction with cost estimates for 147 other health conditions, and the final, attributable cost estimates are therefore accounting for a much larger set of comorbidities. Second, a large source of difference between our estimates was the cost for paid home care, which was larger in Hurd's results. Their use of survey data rather than claims data may lead to a more accurate estimate of this type of care, which is often paid for in cash outside of the health care system. A recent study by Nandi and colleagues updated Hurd's estimates and found a national cost of $450 billion for dementia care in the US in 2020. Their estimate exceeds both Hurd's projections and our estimates due mainly to higher paid home care and nursing home costs. Similarly to Hurd and colleagues, Nandi uses survey data to estimate both paid home care hours and nursing home stays. While a survey-based approach may capture more utilization of these services, it is subject to recall bias and requires making assumptions about the cost of paid home health and nursing home services using outside sources.
We further explored cost drivers by decomposing the variation in per capita cost into the impact of medical spending, caregiving hours, home health aide wages, dementia prevalence, and the age profile of each state. The largest factor in the cost differences among states is the number of unpaid care hours. Almost all states in the South had above-average caregiving time, with the exceptions of Florida and the District of Columbia. This contributed to the high total costs in the southern states. These results imply that focusing on unpaid care, rather than dementia prevalence or medical spending on dementia, has the largest potential to bring down dementia care costs.
Little is known about potential drivers of variation in the amount of unpaid dementia care provided across the US. Examining the relationship between medical spending on dementia and costs of unpaid care across states, we found evidence of a negative association, with a 10% increase in the cost of unpaid care being associated with a 3% decrease in medical spending. This relationship was driven by nursing facility costs, which are the largest component of medical spending costs. We did not see a relationship between unpaid care costs and paid home health costs or emergency department costs. The lack of statistically significant association between unpaid care and home health costs is in line with the mixed findings in the literature. While some studies have found evidence that paid home health care and unpaid care are substitutes, there is also evidence that these two forms of care become complements at higher levels of disability.10,28
Characterizing this tradeoff between health systems costs (mainly nursing facility costs) and unpaid care is important, because several policies have been put in place to rebalance Medicaid long-term services and supports away from institutionalization and toward home and community-based services. 29 These policies have been implemented in part due to consumer preferences as well as a desire to save money from the perspective of state budgets.30,31 However, these cost savings do not account for additional unpaid care needed for older adults to age in place and the potential burden associated with that care. 32 Unpaid family care has been called “the invisible copayment” by Allison Hoffman, 33 and our results show that unpaid care is offsetting direct medical spending on nursing facilities. However, when a value is assigned to that unpaid care, that cost is typically larger than other spending on dementia care. States should consider all costs associated with older adults aging in place. Given that many of the states with the highest total costs of dementia care are also states with high poverty rates, 34 some states may need additional federal support to help alleviate the dual burden of high medical costs and high costs of unpaid care.
This study has several limitations. First, the cost of unpaid care estimates relied on smoothing across space and time, due to a limited amount of state-level data on caregiving for people living with dementia. While the smoothing does not bias our cost estimates in either direction, it limits our ability to examine changes over time within a given state. Second, the medical spending estimates do not include durable medical goods, costs of transportation, or spending by Veterans Affairs or Indian Health Services. Third, the methodology to control for comorbidities and thereby estimate attributable cost differed between our medical spending and cost of unpaid care estimates. While comorbidity adjustment could be refined with richer data, these adjustments achieve their goal of ensuring that costs are not double-counted, and they serve to make the cost estimates more conservative. Finally, measures of dementia severity are not included in either cost estimates. Prior research has shown that both medical spending and unpaid caregiving costs are impacted by dementia severity.7,9 If the average level of dementia severity is higher in some US states, then both costs will be higher in those states, thereby obscuring the relationship between medical spending and costs of informal care.
Our results highlight wide differences in the cost of dementia care across US states. Some states are experiencing both high medical spending and costs of unpaid care, which likely puts a large strain on many families affected by dementia. Ongoing discussions around policies to support people living with dementia and their family caregivers should acknowledge both medical spending and costs of unpaid caregiving and their uneven distribution across the US.
Supplemental Material
sj-docx-1-alz-10.1177_13872877251326231 - Supplemental material for The costs of dementia care by US state: Medical spending and the cost of unpaid caregiving
Supplemental material, sj-docx-1-alz-10.1177_13872877251326231 for The costs of dementia care by US state: Medical spending and the cost of unpaid caregiving by Amy Lastuka, Michael R Breshock, Kayla V Taylor and Joseph L Dieleman in Journal of Alzheimer's Disease
Footnotes
Author contributions
Amy Lastuka (Conceptualization; Data curation; Formal analysis; Funding acquisition; Investigation; Methodology; Supervision; Validation; Visualization; Writing – original draft; Writing – review & editing); Michael R Breshock (Data curation; Formal analysis; Investigation; Methodology; Visualization; Writing – original draft; Writing – review & editing); Kayla V Taylor (Project administration; Writing – review & editing); Joseph L Dieleman (Conceptualization; Formal analysis; Funding acquisition; Investigation; Methodology; Project administration; Supervision; Validation; Writing – original draft; Writing – review & editing).
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by Gates Ventures.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability
The data supporting the findings of this study are available within the article and/or its supplemental material.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.