Abstract
Background
KLOTHO-VS heterozygosity (KL-VSHET) and soluble α-Klotho (sαKl) protein interfere with Alzheimer's disease (AD) pathophysiology, but the specific relationships remain unclear. This study explored these associations across the AD continuum, focusing on core AD biomarkers and markers of neurodegeneration, neuroinflammation, and synaptic dysfunction.
Objective
We investigated whether 1) KL-VSHET is associated with lower AD biomarker burden (Aβ42, Aβ42/40 ratio, P-tau181, T-tau) and neurodegeneration (NfL); 2) sαKl relates to AD biomarkers, neurodegeneration (NfL), neuroinflammation (GFAP), and synaptic dysfunction (Ng); 3) associations vary by APOE ε4 status and clinical subgroup.
Methods
Participants (n = 223) were categorized as cognitively healthy (n = 38), aMCI-AD (n = 94), and AD dementia (n = 91). KLOTHO genotyping was available for 128 participants; 138 had cerebrospinal fluid (CSF) and serum sαKl measurements; and 42 had both. Multiple linear regression evaluated associations between KL-VSHET, sαKl levels, and biomarkers, stratified by APOE ε4 status and clinical subgroup.
Results
Overall, the associations between KL-VSHET and higher CSF Aβ42 and Aβ42/40 ratio were non-significant (ps ≥ 0.059) except when restricted to APOE ε4 carriers only (β = 0.11, p = 0.008 and β = 0.16, p = 0.033, respectively). Within clinical subgroups, KL-VSHET was positively associated with Aβ42/40 ratio only in aMCI-AD (β = 0.23, p = 0.034). No significant associations were observed between KL-VSHET and tau biomarkers or NfL. For sαKl, associations with biomarkers were non-significant except for a negative association of serum sαKl with P-tau181 in aMCI-AD (β = −0.25, p = 0.036) and a positive association with Aβ42/40 ratio in APOE ε4 non-carriers (β = 0.24 p = 0.047).
Conclusions
KL-VSHET may help protect against amyloid pathology, particularly in the presence of APOE ε4, and regardless of APOE status in aMCI-AD.
Introduction
Alzheimer's disease (AD) remains a significant challenge in the field of neuroscience due to its intricate pathophysiology and the complex interplay of various genetic, lifestyle, and biochemical factors. Among the genetic factors, the apolipoprotein allele 4 (APOE ε4) is recognized as the strongest genetic risk factor for sporadic AD. 1 However, certain genetic variants may offer protective effects against AD pathology.
Recent research suggests that heterozygosity for the KLOTHO-VS (KL-VS) haplotype may be associated with a reduced burden of key AD biomarkers, such as cerebrospinal fluid (CSF) amyloid-β (Aβ) 42, total tau (T-tau), and phosphorylated tau (P-tau). This positive association has been observed in both cognitively normal individuals and those diagnosed with AD.2–6 Specifically, KL-VS heterozygosity (KL-VSHET) appears to mitigate age- and APOE ε4-related accumulation of Aβ42, T-tau, and P-tau, suggesting a potential protective role against AD pathology in APOE ε4 carriers.6–12 In addition, recent findings indicate that KL-VSHET may also be associated with reduced age-related neuroinflammatory and neurodegenerative profiles in cognitively unimpaired adults at risk for AD. 13
KL-VSHET has further been linked to higher levels of soluble CSF and/or serum based soluble α-Klotho (sαKl) protein.14–16 Predominantly expressed in the kidneys and choroid plexus in the brain, α-Klotho exists in membrane-bound, secreted and soluble forms.17,18 This protein has garnered attention for its neuroprotective properties, which include mitigation of oxidative stress, reducing neuroinflammation, modulation of signaling pathways and enhancement of synaptic plasticity.19,20 Among its mechanisms, α-Klotho enhances the expression of antioxidant enzymes, 20 while also decreasing pro-inflammatory markers, thereby reducing inflammation triggered by Aβ exposure. 19 This antioxidative and anti-inflammatory action is particularly relevant in the context of AD, where oxidative damage and chronic inflammation are hallmarks of the disease Additionally, α-Klotho enhances synaptic plasticity and cognitive function by modulating N-methyl-D-aspartate receptors, critical for long-term potentiation.16,21,22 Its overexpression in the hippocampus improves synaptic function and cognitive performance in animal models,21,22 suggesting its role in promoting cognitive resilience alongside neuroprotection.
Cognitively unimpaired older adults show higher CSF sαKl levels compared to those with AD. 4 However, prior research investigating the relationship between sαKl levels (serum/plasma and CSF) and AD biomarkers across the AD continuum has yielded mixed results. Driscoll and colleagues found no association between serum sαKl and AD biomarkers (CSF Aβ42/40, Aβ42, T-tau, P-tau) in pre-symptomatic individuals. 23 Conversely, Ren and colleagues reported a negative correlation between plasma sαKl and CSF Aβ42, but not T-tau or P-tau181. 24 Grøndvedt and colleagues presented a more complex picture: CSF sαKl positively correlated with CSF Aβ42 and negatively with CSF T-tau and P-tau181, supporting a potential modulatory role in AD pathology. Plasma sαKl, however, only linked negatively to CSF T-tau and P-tau181, highlighting potential differences between serum and CSF sαKl dynamics. 4 These discrepancies underscore the need for further research to elucidate sαKl's role in AD and understand the potential distinctions between serum and CSF sαKl dynamics.
In light of these findings, our study aims to build on previous work by examining the relationship between KL-VSHET, CSF and serum sαKl levels, core AD markers and additional biomarkers of non-specific processes involved in AD pathophysiology, including glial fibrillary acidic protein (GFAP; for neuroinflammation), neurofilament light chain (NfL; for neurodegeneration), and neurogranin (Ng; for synaptic dysfunction). By incorporating these non-specific biomarkers alongside the core AD markers, this study offers a novel approach to understanding the interaction between KL-VSHET, sαKl, and AD-related pathology. Specifically, we set out to test the following hypotheses in a patient sample from the Czech Brain Aging Study:
KL-VSHET is associated with reduced burden of AD pathology (as measured by CSF Aβ42, Aβ42/40 ratio, T-tau and P-tau181) and neurodegeneration (as measured by NfL); Higher CSF sαKl protein levels are independently associated with lower AD pathology burden and improved profiles for biomarkers of non-specific processes, regardless of KL-VS haplotype.
We hypothesized that the protective association of KL-VSHET and higher sαKl protein levels would be present only in individuals who are also carriers of the APOE ε4 risk variant. By addressing these hypotheses, our study aims to contribute to the understanding of genetic and biochemical factors that may mitigate the risk of AD, potentially guiding future therapeutic strategies.
Methods
Participants
In total, 223 participants from the Czech Brain Aging Study, a prospective multicenter study focusing on the neuroepidemiological characteristics of brain aging in the Czech Republic, 25 met inclusion criteria for this study. All participants underwent standard neurological and laboratory evaluations within three months from the initial visit. All participants involved in this study signed written informed consent approved by the Motol University Hospital ethics committee.
Sample 1 (participants with available genotyping) included 128 participants. Of them, 55 met the clinical criteria for AD dementia, 26 55 met the clinical criteria for aMCI due to AD (aMCI-AD), 27 and 18 were cognitively healthy participants. Demographic data are shown in Table 1.
Characteristics of study participants in Sample 1 (participants with available genotyping).
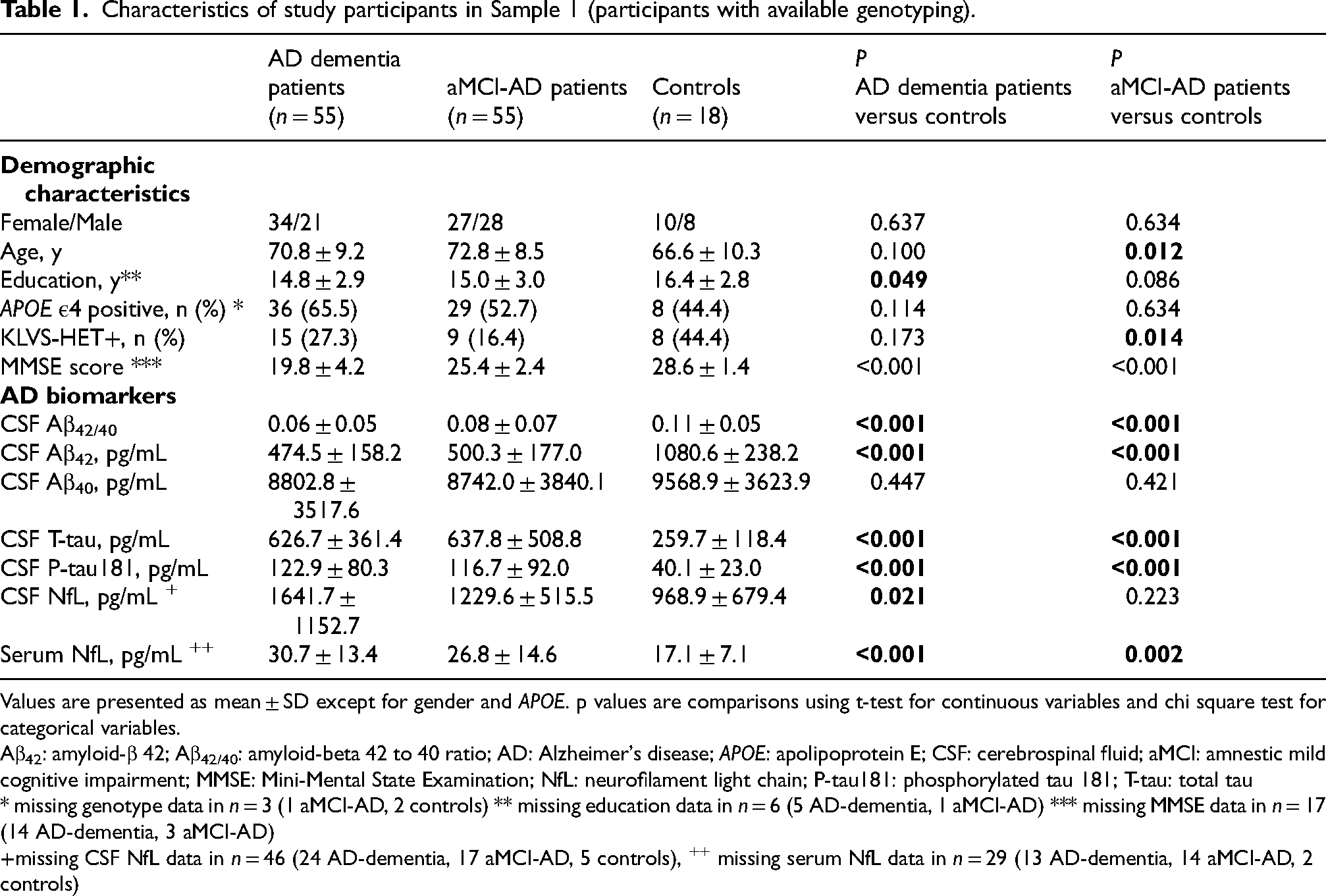
Values are presented as mean ± SD except for gender and APOE. p values are comparisons using t-test for continuous variables and chi square test for categorical variables.
Aβ42: amyloid-β 42; Aβ42/40: amyloid-beta 42 to 40 ratio; AD: Alzheimer's disease; APOE: apolipoprotein E; CSF: cerebrospinal fluid; aMCI: amnestic mild cognitive impairment; MMSE: Mini-Mental State Examination; NfL: neurofilament light chain; P-tau181: phosphorylated tau 181; T-tau: total tau
* missing genotype data in n = 3 (1 aMCI-AD, 2 controls) ** missing education data in n = 6 (5 AD-dementia, 1 aMCI-AD) *** missing MMSE data in n = 17 (14 AD-dementia, 3 aMCI-AD)
missing CSF NfL data in n = 46 (24 AD-dementia, 17 aMCI-AD, 5 controls), ++ missing serum NfL data in n = 29 (13 AD-dementia, 14 aMCI-AD, 2 controls)
Sample 2 (participants with available CSF or serum-based sαKl protein) included 138 participants. Of them, 56 met the clinical criteria for AD dementia, 26 56 met the clinical criteria for aMCI-AD, 27 and 26 were cognitively healthy participants. Demographic data are shown in Table 2.
Characteristics of study participants in Sample 2 (participants with available CSF and serum-based sαKl protein).
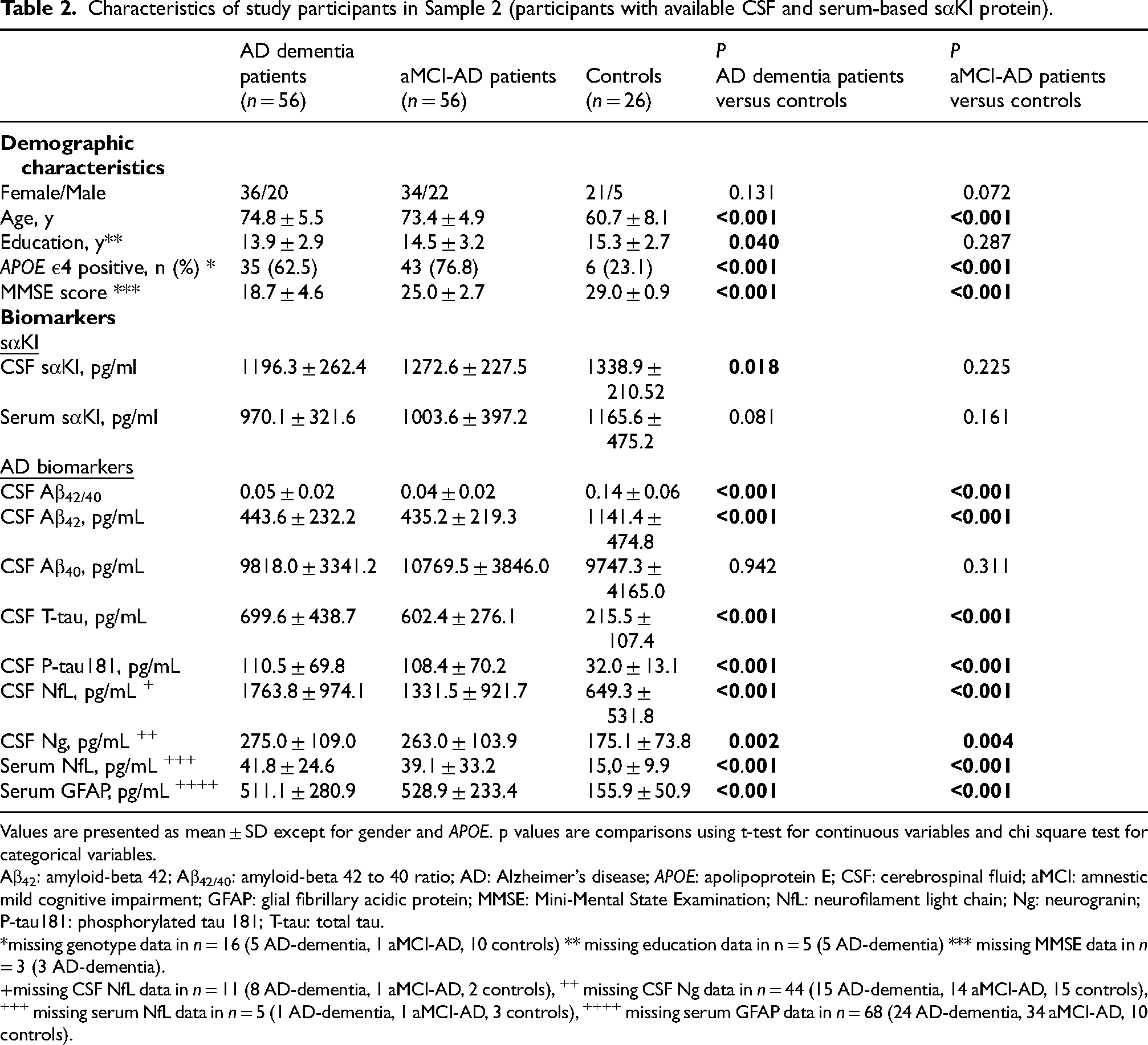
Values are presented as mean ± SD except for gender and APOE. p values are comparisons using t-test for continuous variables and chi square test for categorical variables.
Aβ42: amyloid-beta 42; Aβ42/40: amyloid-beta 42 to 40 ratio; AD: Alzheimer's disease; APOE: apolipoprotein E; CSF: cerebrospinal fluid; aMCI: amnestic mild cognitive impairment; GFAP: glial fibrillary acidic protein; MMSE: Mini-Mental State Examination; NfL: neurofilament light chain; Ng: neurogranin; P-tau181: phosphorylated tau 181; T-tau: total tau.
*missing genotype data in n = 16 (5 AD-dementia, 1 aMCI-AD, 10 controls) ** missing education data in n = 5 (5 AD-dementia) *** missing MMSE data in n = 3 (3 AD-dementia).
missing CSF NfL data in n = 11 (8 AD-dementia, 1 aMCI-AD, 2 controls), ++ missing CSF Ng data in n = 44 (15 AD-dementia, 14 aMCI-AD, 15 controls), +++ missing serum NfL data in n = 5 (1 AD-dementia, 1 aMCI-AD, 3 controls), ++++ missing serum GFAP data in n = 68 (24 AD-dementia, 34 aMCI-AD, 10 controls).
There was an overlap of 42 participants who had both KLOTHO genotyping and CSF/serum-based sαKl measurements. This overlap consisted of 20 participants with AD dementia, 16 participants with aMCI-AD, and 6 cognitively healthy participants.
All participants included in the study underwent lumbar puncture to determine the status of AD biomarkers. Participants diagnosed with AD dementia or aMCI-AD had a positive AD biomarker status based on CSF (reduced Aβ42 and elevated P-tau181 [<620 pg/mL and >61 pg/mL, respectively]). Cognitively unimpaired controls had normal levels of AD biomarkers in CSF.
Participants diagnosed with AD dementia met the NIA-AA 2011 criteria for dementia due to AD, 26 based on progressive decline in at least two cognitive domains (i.e., ≥ 1.5 standard deviations (SD) lower memory test score than the age- and education-adjusted norms as well as similarly low score in at least one non-memory cognitive test), alongside evidence of AD pathophysiology, and significant impairment in the ability to perform daily activities.
Participants diagnosed with aMCI-AD met the NIA-AA 2011 criteria for MCI 27 based on subjective reports of memory decline compared to their prior level, objective evidence of memory impairment (i.e., ≥ 1.5 SDs lower score than the age- and education-adjusted norms in any memory test), preserved independence in daily activities, and the absence of dementia.
Cognitively unimpaired controls (n = 38) were recruited from:
Exclusion criteria
The study excluded participants with pre-existing neurological or psychiatric conditions, including depression (≥6 points on the 15-item Geriatric Depression Scale), 29 and patients with severe white matter vascular lesions on MRI (Fazekas score > 2 points). 30 Participants suffering from renal failure were excluded since kidneys are a major site of α-Klotho production. Participants carrying the ε2/ε4 (n = 6) or ε2/ε3 (n = 5) allele combination were also excluded since the ε2 allele has been shown to be protective against AD. Four participants were excluded from the study sample because of KL-VS homozygosity.
Genotyping
APOE and KLOTHO genotypes were determined at the Department of Clinical Biochemistry, Hematology and Immunology, Homolka Hospital, Prague, Czech Republic.
DNA isolation was performed by Zybio Nucleic extraction kit WB-B from whole blood samples according to manufacturer's protocol (Zybio, Chongqing, China).
APOE genotyping was performed according to IdahoTech protocol (Luna Probes Genotyping Apolipoprotein [ApoE] Multiplexed Assay) for high-resolution melting analysis (HRM).31,32 The APOE ε4 carrier group included APOE ε4 homozygotes (ε4/ε4) and heterozygotes (ε3/ε4).
The analysis of KL-VS haplotype was done also by HRM analysis of single nucleotide polymorphism rs9536314 (G/T). The reaction was performed with LightScanner Master mix (BioFire Diagnostics, SLC., USA) according to manufacturer's PCR reaction conditions with forward and reverse primers: KL1F 5´- ATAACCTTTCATCTATTCTGC-3´; KL1R 5´- AAGTCAGCAGTTCCTTTG-3´; Temperature profile was: 95°C for 2 min followed by 40 cycles of 95°C/30 s; 63°C/30 s; 72°C/30 s; Melting 60°C–90°C. HRM analysis was performed on LightScanner (IdahoTech).
CSF and blood collection and processing
Blood samples were drawn by venipuncture, allowed to clot at room temperature for 15 min, and centrifuged at 1700 × g at 20°C for 5 min within 30 min of collection. CSF was obtained by lumbar puncture in 8-mL polypropylene tubes, gently mixed, and centrifuged at 1700 × g at 20°C for 5 min within 30 min of collection. CSF and serum supernatant were aliquoted in polypropylene tubes of 0.5 ml and stored at −80°C. Before analysis, serum and CSF samples stored at −80°C were thawed at room temperature and vortexed for 15 s.
Immunological assays
CSF levels of Aβ42, Aβ40, T-tau, P-tau181, and Ng were measured using a LUMIPULSE® G600II instrument (Fujirebio, Ghent, Belgium). 33 CSF Aβ42/40 ratio was calculated and used as an additional dependent variable. Commercial enzyme-linked immunosorbent assay (ELISA) was used to measure levels of NfL in CSF and serum (UmanDiagnostics, 10-7001CE and 20-8002RUO). Serum GFAP concentration was measured using the Quanterix Simoa Neurology 4-Plex B following the manufacturer's protocol (Quanterix, Billerica, MA).
Protein levels of sαKl in serum and CSF were quantified using commercially available enzyme-linked immunosorbent assay (ELISA) kit (Immuno-Biological Laboratories Co Ltd, Japan; cat. no. JP27998) following the manufacturer's instructions. Serum and CSF samples were measured in duplicate and were analyzed undiluted. At the end of the assay, absorbances were read at 450 nm using a microplate reader (Dynex Technologies, Virginia, USA), and the protein concentration was calculated by comparison with a standard curve. The intra-assay coefficient of variance (CV%) was <3% and the inter-assay CV was <8%.
Statistical analyses
We used the independent samples t-test to evaluate between-group differences in age, years of education, and global cognitive functioning as assessed by Mini-Mental State Examination (MMSE), and the χ2 test to evaluate differences in frequencies for sex, APOE ε4, and KL-VSHET. Independent samples t-tests were used to compare mean sαKl levels between KL-VSHET and wild-type individuals and to compare MMSE and biomarker levels between APOE ε4 carriers and non-carriers.
Normality was assessed through the inspection of histograms, skewness, and the Shapiro-Wilks test of normality. Serum sαKl, CSF core AD biomarkers (CSF Aβ42, Aβ40, T-tau, P-tau181) and biomarkers of non-specific processes involved in AD pathophysiology (GFAP, Ng, NfL) had nonnormal distribution; therefore, log transformation was applied.
To assess the association between KL-VSHET, core AD biomarkers (Aβ42, Aβ40, T-tau, P-tau181) and biomarker of neurodegeneration (NfL), we used multiple linear regression models with each biomarker as the dependent variable and KL-VS haplotype as the independent variable. Age and sex were included as covariates in all models.
Similarly, we examined the relationship between CSF and serum sαKl protein levels, core AD biomarkers (Aβ42, Aβ40, T-tau, P-tau181) and biomarkers of non-specific processes involved in AD pathophysiology (GFAP, Ng, NfL) using separate multiple linear regression models for each biomarker. Each model included the respective AD biomarker as the dependent variable, CSF or serum sαKl level as the independent variable, and age and sex as covariates.
Results
Sample characteristics
Group demographic and clinical characteristics of Sample 1 (participants with available genotyping) are presented in Table 1. There was no significant difference in sex distribution among the study groups. Controls were significantly younger compared to aMCI-AD patients (p = 0.012). Participants diagnosed with AD dementia had fewer years of education than the controls (p = 0.049). The proportion of KL-VSHET individuals was significantly lower in the aMCI-AD group compared to the control group (p = 0.014), but not in the AD dementia group compared to the control group (p = 0.173). Finally, individuals with aMCI-AD and AD dementia had significantly lower MMSE score compared to controls (both ps < 0.001).
Group demographic and clinical characteristics of Sample 2 (participants with available CSF or serum based sαKl protein) are presented in Table 2. Both the aMCI-AD and AD dementia groups were significantly older than the controls (both ps < 0.001). The AD dementia group had significantly fewer years of education than the controls (p = 0.040). The percentage of APOE ε4 positive individuals was significantly higher in the AD dementia and aMCI-AD groups compared to controls (both ps < 0.001). Finally, MMSE score was significantly lower in the aMCI-AD and AD dementia groups compared to controls (both ps < 0.001).
Group differences by APOE ε4 status in Sample 1
MMSE scores did not differ between the APOE ε4 non-carriers and APOE ε4 carriers (p = 0.269). CSF Aβ42 levels were significantly higher in the APOE non-carriers compared to the APOE ε4 carriers (p = 0.016). However, no significant differences between the groups were found for the CSF Aβ42/40 ratio (p = 0.203), CSF T-tau (p = 0.738), or CSF P-tau181 (p = 0.801), CSF NfL (p = 0.183) or serum NfL levels (p = 0.202) (Figure 1).
Group differences by APOE ε4 status in Sample 2
No significant differences were observed in MMSE scores between the APOE ε4 non-carriers and APOE ε4 carriers (p = 0.532). Similarly, sαKl levels in both CSF and serum did not differ significantly between the APOE groups (p = 0.183 and p = 0.516, respectively). Additionally, no significant group differences were observed in CSF P-tau 181 (p = 0.091), CSF NfL (p = 0.341), CSF Ng (p = 0.237), or serum NfL (p = 0.776). However, significant group differences were found for CSF Aβ42 and the Aβ42/40 ratio, which were higher in the APOE ε4 non-carriers compared to the APOE ε4 carriers (p = 0.002, p = 0.007, respectively). CSF T-tau and serum GFAP levels showed a trend toward being higher in the APOE ε4 carriers, although this difference did not reach statistical significance (p = 0.064 and p = 0.083, respectively) (Figure 2).
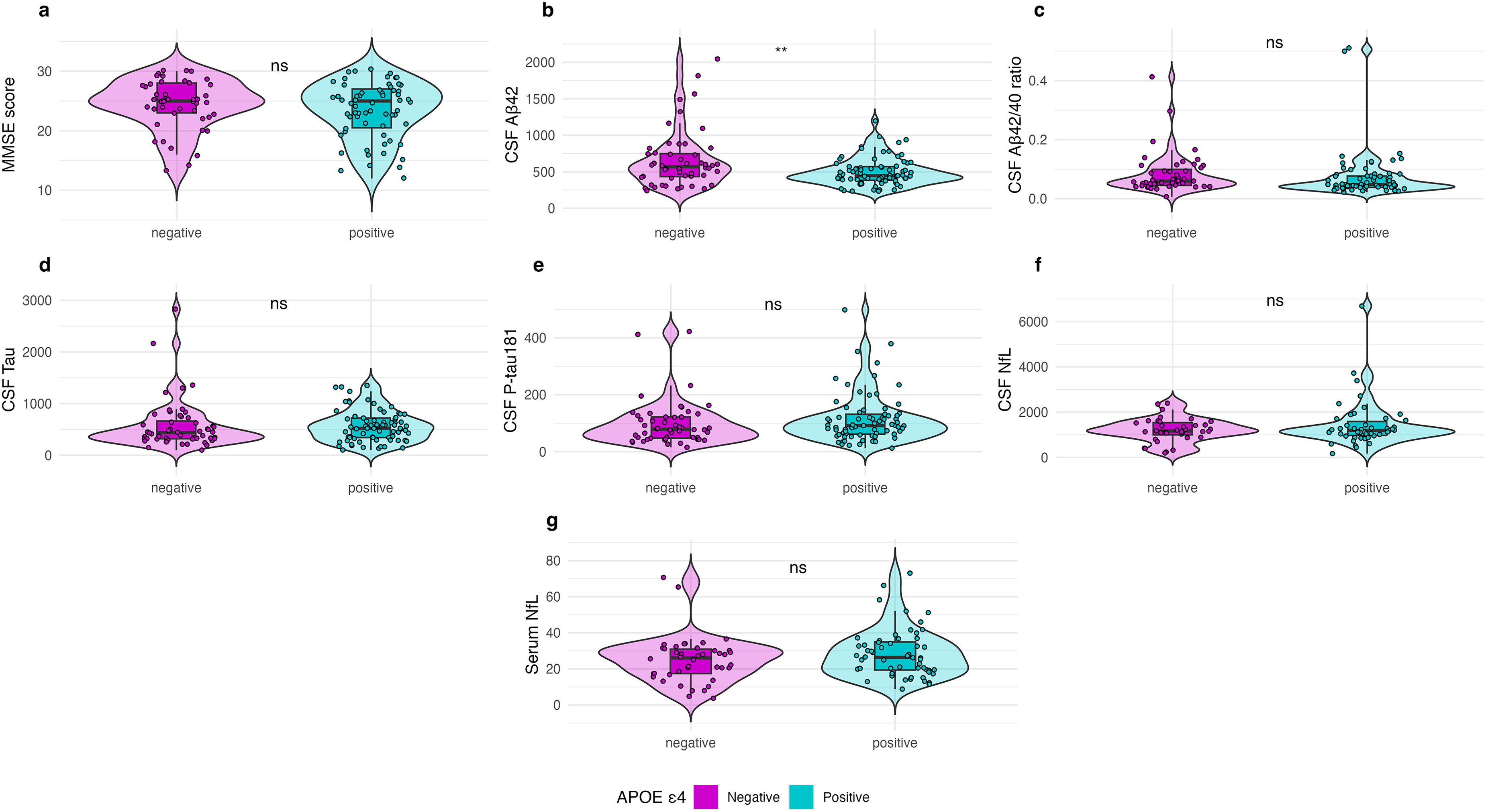
Group Differences by APOE ε4 Status in Sample 1 (participants with available genotyping).
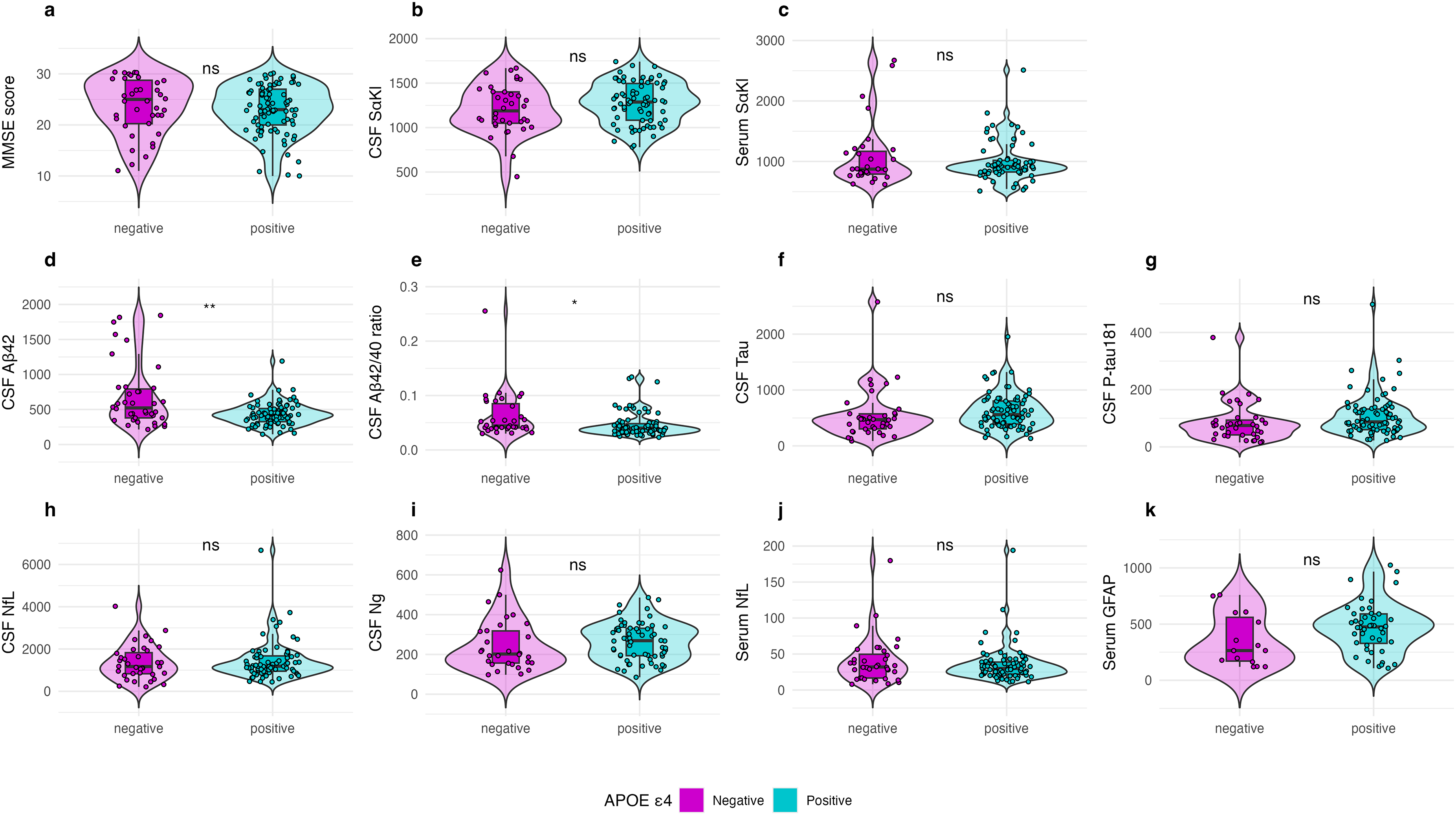
Group Differences by APOE ε4 Status in Sample 2 (participants with available CSF and serum-based sαKl protein).
Association of KL-VSHET with core AD biomarkers and NfL
Controlling for age and sex, KL-VSHET showed a non-significant trend towards a positive association with Aβ42/40 ratio when using the entire sample (β = 0.10, p = 0.091). Stratifying by APOE ε4 revealed this association was significant in APOE ε4 carriers (β = 0.16, p = 0.033), which included both ε4/ε4 homozygotes and ε3/ε4 heterozygotes, but not in APOE ε4 non-carriers (β = 0.03, p = 0.786). When analyzing by clinical subgroups, KL-VSHET was significantly associated with the Aβ42/40 ratio, but only in the aMCI-AD group (β = 0.23, p = 0.034).
A similar pattern emerged for Aβ42, with a non-significant trend towards positive association between carrying the KL-VSHET haplotype and higher Aβ42 levels in the entire sample (β = 0.08, p = 0.059), a significant association in APOE ε4 carriers (β = 0.11, p = 0.008), and a non-significant association in APOE ε4 non-carriers (β = 0.03, p = 0.683).
No significant associations were detected for KL-VSHET with T-tau, P-tau181 or NfL levels in the entire sample or in the APOE ε4 or clinical subgroups (all ps > 0.05). The regression coefficients of each of the linear regression models showing KL-VSHET in relation to the levels of biomarkers are presented in Table 3.
Linear regression models showing the association of KL-VS heterozygosity with levels of biomarkers in the entire sample and by APOE ε4 and clinical subgroups.
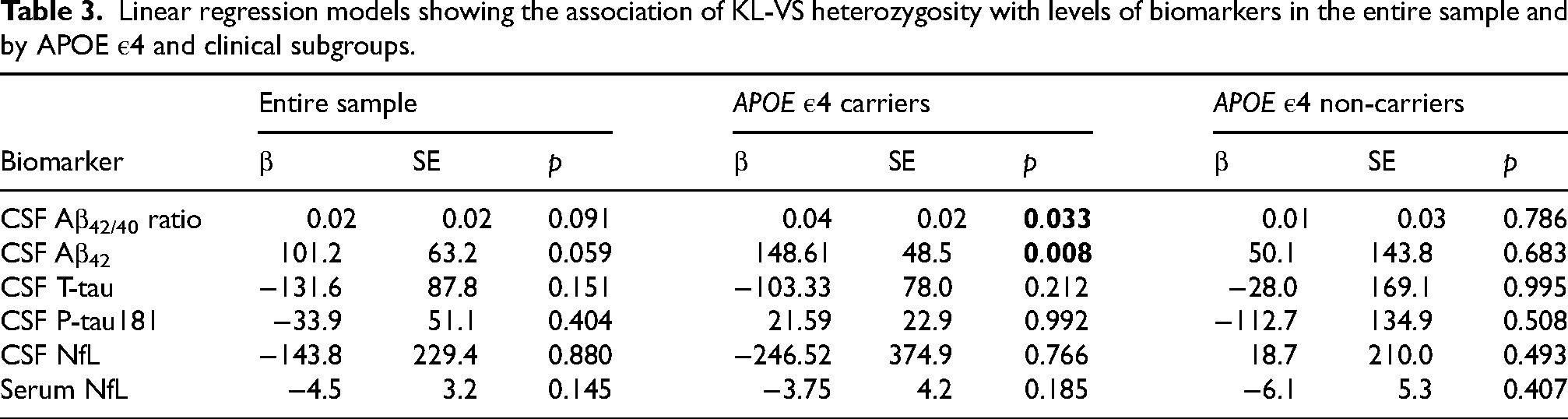
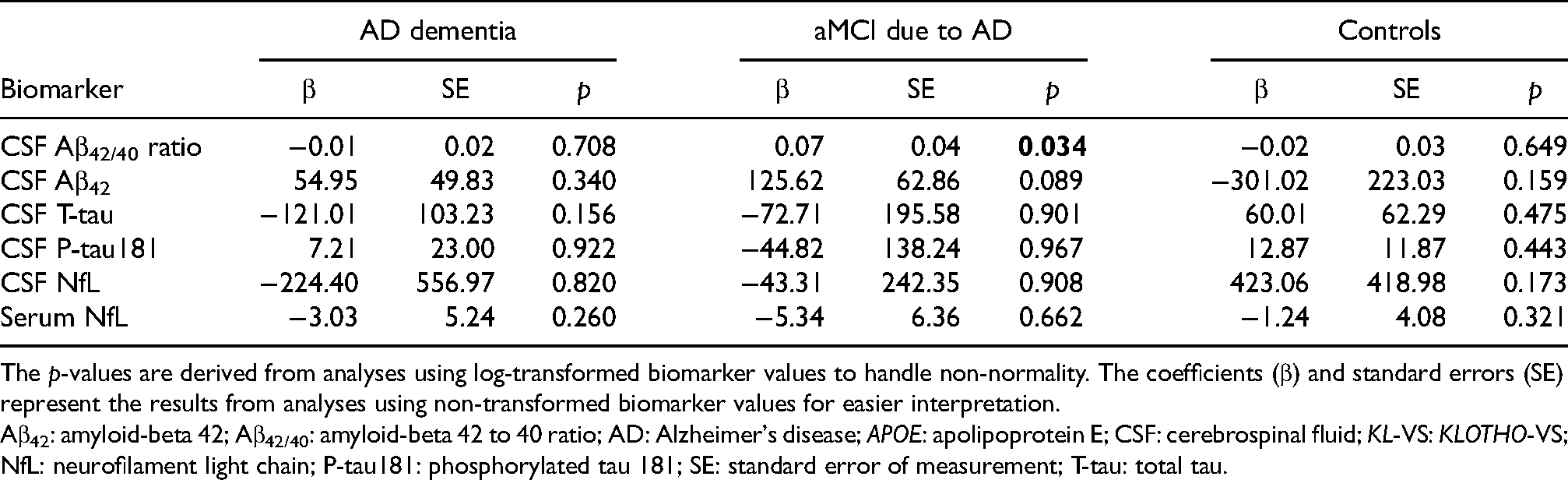
The p-values are derived from analyses using log-transformed biomarker values to handle non-normality. The coefficients (β) and standard errors (SE) represent the results from analyses using non-transformed biomarker values for easier interpretation.
Aβ42: amyloid-beta 42; Aβ42/40: amyloid-beta 42 to 40 ratio; AD: Alzheimer's disease; APOE: apolipoprotein E; CSF: cerebrospinal fluid; KL-VS: KLOTHO-VS; NfL: neurofilament light chain; P-tau181: phosphorylated tau 181; SE: standard error of measurement; T-tau: total tau.
Association of sαKl protein levels with core AD biomarkers and biomarkers of non-specific processes involved in AD pathophysiology
No significant associations were identified between CSF sαKl levels and any of the core AD biomarkers or biomarkers of neurodegeneration (NfL), neuroinflammation (GFAP) and synaptic dysfunction (Ng) in either the entire sample or within the APOE ε4 or clinical subgroups (Table 4).
Linear regression models showing the association of
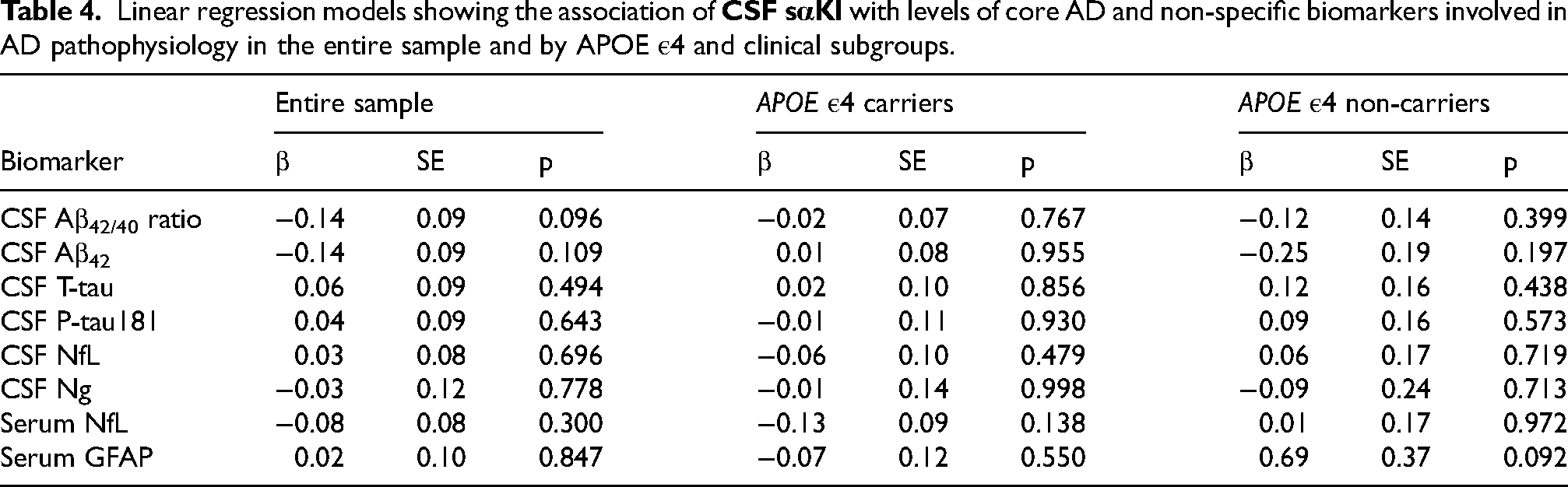
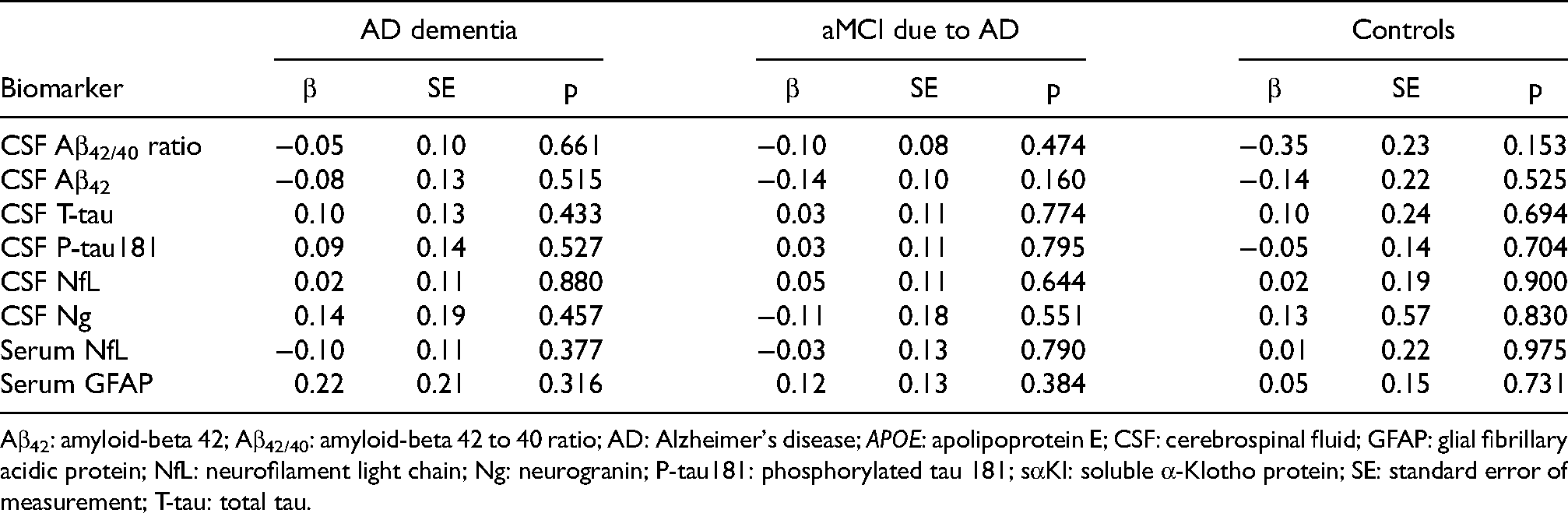
Aβ42: amyloid-beta 42; Aβ42/40: amyloid-beta 42 to 40 ratio; AD: Alzheimer's disease; APOE: apolipoprotein E; CSF: cerebrospinal fluid; GFAP: glial fibrillary acidic protein; NfL: neurofilament light chain; Ng: neurogranin; P-tau181: phosphorylated tau 181; sαKl: soluble α-Klotho protein; SE: standard error of measurement; T-tau: total tau.
Similarly, serum sαKl levels did not show a significant association with any of the core AD biomarkers or biomarkers of non-specific processes involved in AD pathophysiology when analyzing the entire sample. When stratifying the analysis based on APOE ε4 carriership, serum sαKl levels were positively associated with Aβ42/40 ratio, but only in APOE ε4 non-carriers (β = 0.24, p = 0.047) (Table 5). When stratifying the analysis based on clinical subgroups, serum sαKl levels were negatively related to P-tau181 in the aMCI-AD group (β = −0.25, p = 0.036). No significant associations were observed for any of the other biomarkers in either APOE ε4 or clinical subgroups.
Linear regression models showing the association of
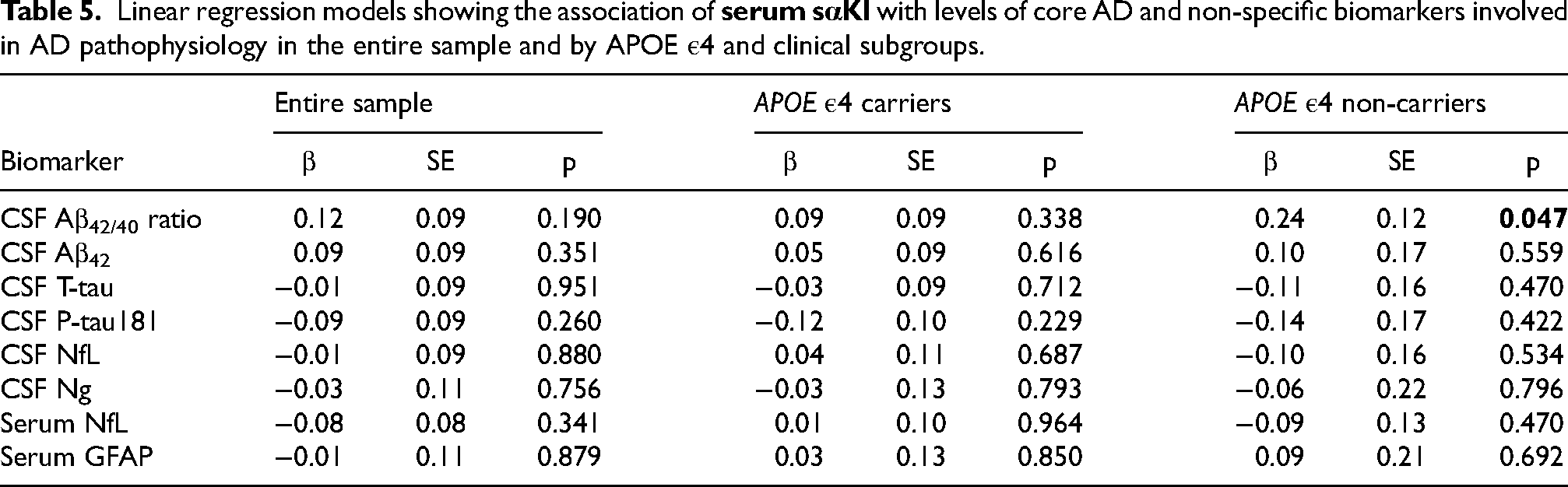
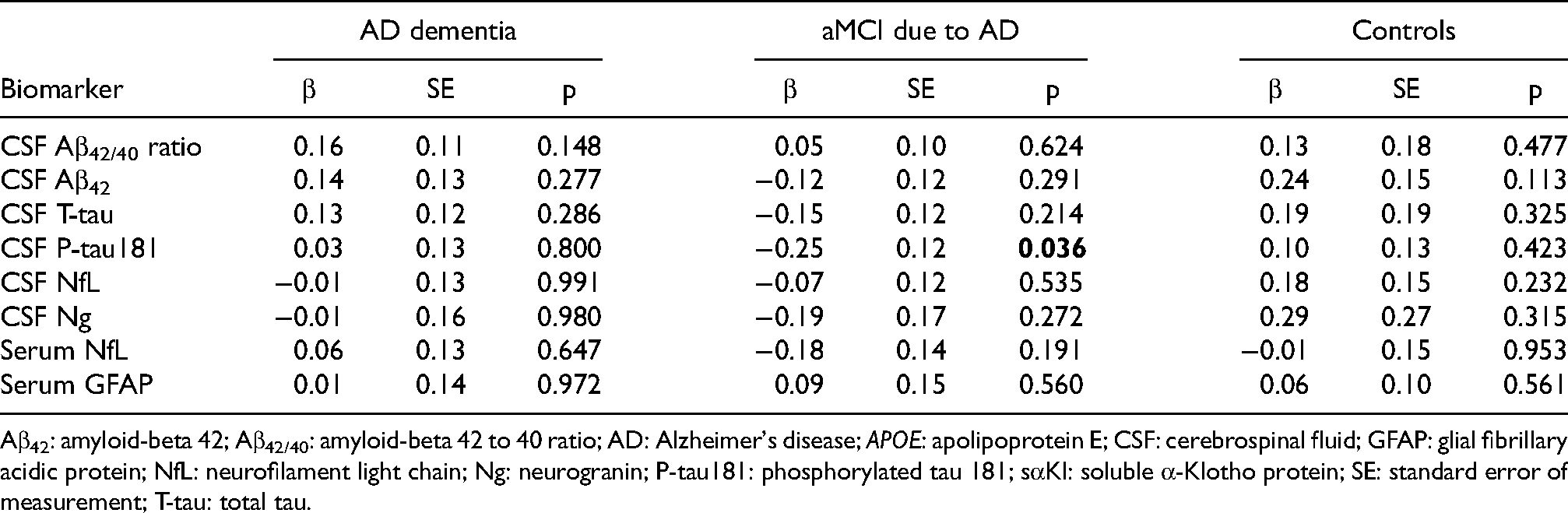
Aβ42: amyloid-beta 42; Aβ42/40: amyloid-beta 42 to 40 ratio; AD: Alzheimer's disease; APOE: apolipoprotein E; CSF: cerebrospinal fluid; GFAP: glial fibrillary acidic protein; NfL: neurofilament light chain; Ng: neurogranin; P-tau181: phosphorylated tau 181; sαKl: soluble α-Klotho protein; SE: standard error of measurement; T-tau: total tau.
Association of KL-VS haplotype with sαKl protein levels
Additionally, we explored the relationship between KL-VS haplotype and sαKl levels in both CSF and serum. In the subset of participants with both KL-VS haplotyping and sαKl level measurements, we found a significant difference in CSF sαKl levels between KL-VSHET individuals and those with the wild-type genotype. Specifically, KL-VSHET individuals exhibited higher CSF sαKl levels compared to wild-type individuals (1473.2 ± 183.0 pg/mL versus 1237.9 ± 254.9 pg/mL, p = 0.025). In contrast, sαKl levels in serum showed no significant difference between the two groups (p = 0.37) (Figure 3).
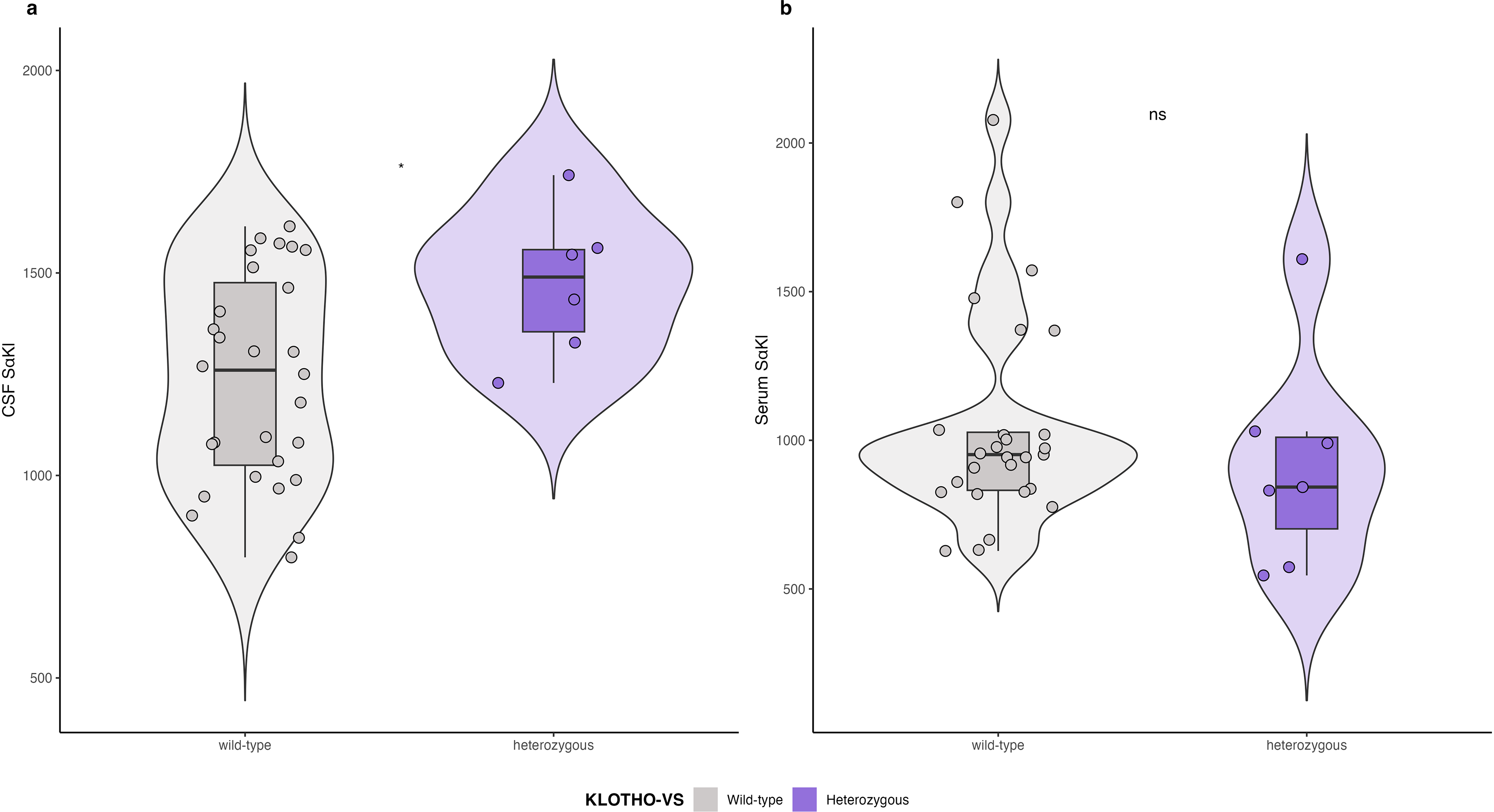
CSF and serum sαKl protein levels by KL-VS haplotype.
Discussion
This study aimed to investigate the protective role of KL-VSHET and sαKl protein levels on AD biomarker burden across the AD continuum, including both core AD biomarkers and biomarkers of non-specific processes like neuroinflammation, neurodegeneration and synaptic dysfunction. We found that KL-VSHET showed a non-significant trend towards a positive association with CSF Aβ42/40 ratio and Aβ42 levels in the whole sample. However, significant associations were only observed in APOE ε4 carriers, whereas the association was essentially non-existent among APOE ε4 non-carriers, suggesting that any potential protective effect of KL-VSHET on Aβ pathology may operate as a function of APOE ε4 status. These findings align with previous studies demonstrating that KL-VSHET is associated with lower Aβ burden specifically in cognitively normal individuals carrying the APOE ε4 risk allele.5–10 Notably, our results suggest that this protection may extend to individuals already diagnosed with clinically symptomatic AD, as we observed a significant positive association between KL-VSHET and Aβ42/40 ratio specifically in the aMCI due to AD group. This aligns with Cook and colleagues, who found KL-VSHET carriers exhibited consistent patterns of reduced Aβ burden across the AD continuum, 2 suggesting a broader effect beyond cognitively unimpaired individuals.
Recent evidence highlights a regionally specific relationship between α-Klotho and apoE protein levels in the brain, particularly under environmental stress. Using irradiated Rhesus macaques, Kundu et al. 34 demonstrated a strong positive correlation between α-Klotho and apoE protein levels in the amygdala, suggesting a potential adaptive response to stress. However, no such relationship was observed in the prefrontal cortex, and α-Klotho levels did not predict regional brain volumes or change with age in this sample. While the results warrant replication in human studies, these findings indicate that the interplay between α-Klotho and apoE may be region-specific and context-dependent. Although our study did not examine this relationship directly, the results underscore the importance of investigating potential synergistic effects of α-Klotho and apoE on brain structure and function, particularly in regions implicated in AD, such as the limbic system.
While we observed a positive association between KL-VSHET and Aβ biomarkers, we did not detect significant associations between KL-VSHET and tau biomarkers (T-tau or P-tau181) or NfL, a neurodegeneration marker, in our study sample. Driscoll et al. 13 found that KL-VSHET attenuated age-related neurodegeneration as measured by CSF NfL levels in cognitively unimpaired individuals at risk for AD. However, the absence of an association with NfL in our sample, which included individuals across various stages of AD, may suggest that the neuroprotective effects of KL-VSHET on markers of neurodegeneration are more prominent at earlier, preclinical stages. This finding may indicate that KL-VSHET selectively modulates Aβ processing or clearance pathways, with its effects on neurodegeneration markers like NfL diminishing as the disease progresses or as AD pathology becomes more advanced.
Recent findings indicate that both Aβ and tau biomarkers in CSF reach plateau levels at different stages of the disease, reducing their utility in monitoring AD pathology as the disease progresses. 35 In our sample, however, we observed significant differences in the levels of all CSF AD biomarkers between AD dementia and aMCI-AD. Specifically, both Aβ42 and Aβ42/40 ratio, as well as T-tau and P-tau181 levels, changed from aMCI to dementia. This suggests that while both types of biomarkers are dynamic over the disease course in our sample, their associations with KL-VSHET might differ due to underlying mechanisms. One possible explanation for this discrepancy is that KL-VSHET may specifically modulate pathways related to Aβ processing or clearance, rather than tau phosphorylation and aggregation. Similarly, Cook et al. observed a non-significant trend for reduced tau burden in cognitively normal KL-VSHET carriers but no effect in individuals with aMCI or dementia due to AD. 2 Conversely, Neitzel and colleagues reported a protective effect of KL-VSHET against amyloid-dependent tau accumulation specifically in patients with aMCI-AD, particularly APOE ε4 carriers. 6 Finally, some studies report no significant associations between KL-VSHET and both Aβ and tau biomarkers (T-tau, P-tau181) among AD patients.4,5 These discrepancies highlight the need for further research with larger and more diverse cohorts, focusing on longitudinal changes and stratifying by AD stage to definitively elucidate the complex role of KL-VSHET in both Aβ and tau pathology across the AD continuum.
Contrary to our hypothesis, we did not observe any significant associations between CSF sαKl and any of the core AD or non-specific biomarkers involved in AD pathophysiology, including NfL, GFAP, and Ng. Serum sαKl similarly exhibited no significant associations in the overall sample but showed a positive association with CSF Aβ42/40 ratio only in APOE ε4 non-carriers and a negative association with CSF P-tau181 only in aMCI due to AD patients. Our findings contrast with Grøndvedt et al., who reported a positive association of CSF sαKl with CSF Aβ42 and a negative association with CSF T-tau and P-tau, while plasma sαKl only correlated negatively with CSF T-tau and P-tau. 4 Ren et al. observed a negative correlation between plasma Klotho and CSF Aβ42, but not any of the other investigated CSF biomarkers (T-tau, P-tau181, NfL) in a mixed sample of cognitively unimpaired individuals and AD patients, 24 suggesting that higher sαKl levels are associated with greater Aβ accumulation and potentially more advanced AD pathology. Driscoll et al. reported no differences in serum sαKl between pre-symptomatic individuals who were positive vs. negative for Aβ or tau, 23 suggesting no clear association between sαKl levels and early AD pathology. This aligns with our lack of significant findings in the entire sample. However, unlike the pre-symptomatic cohort studied by Driscoll and colleagues, 23 our participants exhibited advanced AD pathology.
While no previous studies have examined the association between sαKl protein levels and biomarkers like NfL, GFAP, or Ng, Driscoll et al. investigated whether the KL-VSHET genotype influences these markers in cognitively unimpaired individuals at risk for AD. 13 Their findings demonstrated that KL-VSHET attenuated the deleterious effects of age on CSF levels of Ng and GFAP, suggesting that KL-VSHET may play a protective role in synaptic function and neuroinflammation during early disease stages. 13 Our lack of significant associations between sαKl levels and these biomarkers could imply that sαKl protein, unlike KL-VSHET genotype, does not modulate these non-specific AD pathophysiology markers, or that its influence may differ based on disease stage and cohort characteristics. The absence of associations in our sample, which included a range of AD stages, could also suggest that any influence of sαKl on non-specific markers like NfL, GFAP, and Ng is more detectable in preclinical or earlier stages of AD pathology, aligning with Driscoll et al.'s findings in cognitively unimpaired adults. These seemingly divergent findings highlight the complexity of sαKl ‘s involvement in AD and the potential influence of factors like sample characteristics and disease stage. However, without longitudinal data and larger sample sizes, drawing definitive conclusions remains challenging.
Finally, we explored the association between KL-VSHET and sαKl levels in a subset of participants with available data. Our results revealed significantly higher CSF sαKl levels in KL-VSHET carriers compared to those with the wild-type haplotype, consistent with prior studies reporting a relationship between KL-VSHET and increased CSF sαKl protein levels.4,14 However, consistently with Gaitán et al., 14 we did not find a significant difference in serum sαKl levels between the haplotype groups, which may be attributable to the limited sample size and potentially distinct dynamics of sαKl across different biological compartments. These findings suggest a potential haplotype-specific modulation of sαKl levels in the brain (as reflected by CSF measurements), which may not be captured by serum measurements.
Some study limitations should be noted. Our study was limited by sample size, which could have obscured potential associations. Future studies with larger samples are needed. Due to the small sample size, the APOE ε4 carrier group combined ε3/ε4 heterozygotes and ε4/ε4 homozygotes. This restricts our ability to determine if the observed associations between KL-VSHET and AD biomarker levels differ by APOE ε4 dose (one versus two ε4 alleles). Additionally, the cross-sectional design prevents definitive conclusions about directional relationships. Furthermore, α-Klotho protein analysis was not performed across both cohorts due to the limited availability of biological samples and data at the time of analysis. This prevented us from maximizing the sample size for these analyses. Finally, while we conducted a preliminary genotype-phenotype analysis on the subset of participants with both KL-VS haplotype and sαKl protein data, the limited sample size limits the strength and generalizability of these findings.
In summary, our key finding is that KL-VSHET carriers showed higher CSF Aβ42 and Aβ42/40 ratio levels that was attributable to differences in APOE ε4 carriers only. This suggests a potential protective effect of KL-VSHET against amyloid pathology, but only in individuals carrying the APOE ε4 allele. KL-VSHET haplotype did not show an association with measures of tau burden or neurodegeneration as measured by NfL, indicating that its protective effects may be more specific to amyloid pathways rather than neurodegenerative processes generally. In addition, there was no association observed between sαKl protein levels and AD biomarkers, nor with non-specific biomarkers of AD pathophysiology, including NfL, GFAP, and Ng. This lack of association suggests that sαKl protein may not directly influence these biomarkers or that its potential effects on neuroinflammation, neurodegeneration, and synaptic dysfunction markers may vary by disease stage and may be less detectable in advanced AD. Furthermore, the link between KL-VSHET and Aβ levels may not play a role in the context of sαKl levels, as the observed effects of KL-VSHET on Aβ burden do not seem to be mediated by sαKl concentration alone. While KL-VSHET generally correlates with sαKl protein levels,14–16 it might not fully capture the functional protein abundance or its specific activity within different compartments like CSF and plasma or serum, potentially leading to divergent results. Conclusive evidence remains elusive and further research using larger, stage-specific cohorts and longitudinal data will be crucial to disentangle the complexities surrounding KL-VSHET's and sαKl's involvement in AD and its potential as a therapeutic target or prognostic marker.
Footnotes
Acknowledgments
We would like to thank the Alzheimer Foundation Czech Republic for their funding support and the Czech Brain Aging Study individuals for their participation.
Ethical considerations
Not applicable.
Consent to participate
All participants involved in this study signed written informed consent approved by the Motol University Hospital ethics committee.
Author contributions
Alzbeta Katonova (Conceptualization; Data curation; Formal analysis; Investigation; Methodology; Writing – original draft; Writing – review & editing); Ross Andel (Conceptualization; Data curation; Formal analysis; Methodology; Supervision; Writing – original draft; Writing – review & editing); Vanesa Jurasova (Writing – review & editing); Katerina Veverova (Data curation; Writing – review & editing); Francesco Angelucci (Data curation; Writing – review & editing); Vaclav Matoska (Data curation; Writing – review & editing); Jakub Hort (Conceptualization; Writing – original draft; Writing – review & editing).
Consent for publication
Not applicable.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the project National Institute for Neurological Research (Programme EXCELES, ID Project No. LX22NPO5107); by the Institutional Support of Excellence 2 2. LF UK (Grant No. 6980382); and by the Czech Alzheimer's Foundation.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
