Abstract
Background
Understanding how sociodemographic characteristics and medical history are associated with progression (or regression) of Alzheimer's disease and related dementias could inform intervention strategies, personalized prognoses, and projections of population-level burden.
Objective
We estimated transition rates for progression and reversion between normal cognition, cognitive impairment, dementia, and death in a longitudinal cohort, as well as associations with sociodemographic characteristics and medical history.
Methods
We applied a multistate transition model to a cohort of 960 participants (with 2–16 (median 3) annual visits; 2006–24). Covariate hazard ratios (HRs) were estimated in models adjusted for age group.
Results
Several covariates were associated with faster progression from normal cognition to cognitive impairment but slower progression from cognitive impairment to dementia. For example, non-Hispanic Black participants transitioned from normal to cognitive impairment at higher rates (HR: 2.29, 95% CI: 1.63, 3.21) and to dementia at lower rates (HR: 0.12, 95% CI: 0.06, 0.23) than non-Hispanic White participants. Additionally, amnestic versus non-amnestic impairment emerged as a strong predictor of transitions from cognitive impairment by reducing reversion to normal cognition (HR: 0.51, 95% CI: 0.35, 0.74) and accelerating progression to dementia (HR: 2.51, 95% CI: 1.49, 4.22). History of traumatic brain injury was associated with reversion from cognitive impairment to normal cognition (HR: 2.43, 95% CI: 1.13, 5.23).
Conclusions
A better understanding and measurement of cognitive impairment is needed to explain and predict both reversion to normal cognition and why factors associated with faster onset of impairment may be associated with delayed onset of dementia.
Introduction
Dementia has a progressive and prolonged disease course with gradually deteriorating cognitive functions. 1 As the disease progresses, individuals experience reduced quality of life, including cognitive decline, physical disability such as reduced mobility and difficulty in swallowing, reduced capacity for independent living, and higher risk of mortality. 2 The trajectory of dementia is heterogeneous, with individuals experiencing varying rates of cognitive decline in different periods.
The disease is divided into three broad stages: preclinical, mild cognitive impairment (MCI), and dementia. 3 The transition from preclinical to MCI is often marked by subtle symptoms of cognitive decline such as memory deficiency and language decline. Though the pathological changes do not revert, the disease course can vary greatly, with some individuals experiencing longer periods of cognitive stability or even temporarily reverting to apparently normal cognitive levels.3–6 Such reversion from MCI to apparent normal cognition is not uncommon, yet the factors that contribute to such transitions remain inadequately understood and require further research. Subsequently, when the disease progresses to the dementia stage, individuals face more functional difficulties in performing cognitive-related activities of daily living, including recognition difficulty, diminished communication, emotional and physical dysfunctions, which are related to neuronal damage in different parts of the brain.4,7 Patients with dementia also have an increased risk of death. There is a strong need for further study of the progression of the disease, and a better understanding of the associations of patient characteristics and medical history on the trajectory of dementia could offer potential pathways for prevention or mitigation.
The rate of cognitive decline is influenced by diverse factors. 8 Factors like age, genetics, sex, and family history are not modifiable, while other characteristics such as education level, living situation, and marital status are potentially modifiable factors that could be leveraged to delay or prevent disease progression.1,4,7 The progression of dementia may also be interlinked with health conditions, including the presence of cardiovascular diseases and diabetes. 5 A better understanding of how these characteristics are associated with the speed of dementia progression is crucial for developing a comprehensive picture of dementia.
To bridge the gap in our understanding of the nature of transitions among various states of dementia, we used a Markov multistate transition model to estimate the propensities (transition hazard rates) of disease progression or reversion in the University of Michigan Memory and Aging Project (UM-MAP) longitudinal cohort. Multistate transition models have been widely used to explore disease progression by providing a quantitative framework for analyzing how individuals move through risk or disease states over time, including Alzheimer's disease.5,9–12 We characterized the transition patterns between different states by estimating the associations between sociodemographic and medical history covariates and transition rates to better understand the predictors of transitions in cognitive impairment and dementia. This approach allowed us to identify possible risk and protective factors and quantify their associations with the transition propensities. The study provides insights that can be used to make more effective clinical care management decisions, develop personalized estimates of possible future trajectories and treatment plans, and enable more precise large-scale predictions for future disease burdens.
Methods
Data
We used data from the University of Michigan Memory and Aging Project (UM-MAP), a longitudinal cohort study of adults ages 50 years or older run by the Michigan Alzheimer's Disease Research Center (MADRC). The UM-MAP recruited participants from the Michigan Medicine Neurology Cognitive Disorders Clinic, Neuropsychology Clinic, and health research website, as well as from the local community (southeast Michigan). We excluded participants with only one visit, as they were uninformative for the transition model, along with observations without clinical disease status information. Our analysis included 960 participants with 3484 total observations, who were surveyed at various intervals between 2006 and 2024 (approximately yearly). The number of observations per participant ranged from 2 to 16, with a median of 3. Participants completed a standardized evaluation (National Alzheimer's Coordinating Center 13 Uniform Data Set 2.0 or 3.0) guided by trained clinicians during each visit. Observation dates were truncated to year to ensure participants were not identifiable. Participant deaths were recorded through electronic health records (for participants in the Michigan Medicine health system) or reported by next-of-kin. This cohort study is ongoing, and participants may be between visits at the time of the data pull. So as to not attribute normal follow-up times to loss to follow-up, we defined participants as pending follow-up if their most recent observation was within 2 years of the data pull and if they were not recorded as having died. We defined participants as lost-to-follow-up if their most recent observation was more than 2 years earlier and if they were not recorded as having died. For the purposes of visualization, once a participant is classified as pending, they remain pending.
All the participants gave written informed consent. This secondary data analysis was determined to be not regulated as human subjects research by the University of Michigan Institutional Review Board (HUM00240110).
Based on a clinical consensus diagnosis (determined by three clinicians) and informed by neuropsychiatric tests, participants were categorized into one of three clinical disease states at each visit (normal cognition, cognitive impairment, dementia) or were recorded as dead. Cognitive impairment referred to diagnosis of ‘cognitively impaired but not MCI’ (8.8% of observations classified as cognitive impairment), ‘non-amnestic MCI, single domain’ (19.2%), ‘non-amnestic MCI, multiple domains’ (13.3%), ‘amnestic MCI, single domain’ (21.9%), or ‘amnestic MCI, multiple domains’ (36.6%). We assumed that once a participant was diagnosed with dementia, any subsequent non-dementia state was a temporary masking of neurodegeneration, and so such states were reclassified as dementia (8 participants), effectively meaning that any subsequent transition back to dementia would not be included in the model to bias the results for participants with no prior dementia diagnosis.
In this analysis, we considered sociodemographic characteristics, medical history, and dementia symptom covariates, each updated at each observation, estimating their associations with the progression or regression of cognitive impairment and dementia. The sociodemographic variables included age group (50–64, 65–69, 70–74, 75–79, 80 + years), sex (female, male), BMI (<25, 25–29.9, 30+), race and ethnicity (non-Hispanic (NH) White, NH Black, Other; please note that race and ethnicity are social concepts and are included as proxies for experiences of racialization and systemic racism), education level (high school or GED (General Educational Development Test), bachelor's degree, master's degree, doctorate), and marital status (married/partnered, never married). We also investigated cigarette smoking (never, current, former; defined by lifetime use of 100 cigarettes and past-30-day use). We considered information on whether participants reported ever experiencing the following comorbid conditions: alcohol abuse, heart attack, atrial fibrillation, congestive heart failure, stroke, B12 deficiency, thyroid disease, hypertension, hypercholesterolemia, traumatic brain injury, and diabetes. We also considered conditions that may be symptoms of dementia, including recent depression (past 2 years), urinary incontinence, and bowel incontinence. For participants with cognitive impairment, we also included whether that impairment was amnestic or non-amnestic. Participants originally classified as “Impaired, not MCI” were considered non-amnestic. We described the baseline characteristics of the study sample using mean and range for continuous variables and count and frequency for categorical variables.
We assessed the potential for confounding of the association between transition rates and race by education, reading level, and cognitive assessment by comparing educational attainment, reading level (as measured by the Wechsler Test of Adult Reading), and cognitive assessment (measured by Mini-Mental State Examination or Montreal Cognitive Assessment) by race and ethnicity.
Transition modeling
We used a continuous-time Markov multistate transition model to estimate the transition hazard rates underlying longitudinal data of individuals’ cognitive impairment and dementia trajectories and to understand how transitions between the four states were associated with individual sociodemographic and medical history characteristics. Multistate transition models are finite-state models that assume that the underlying transitions evolve according to a stochastic process defined by transition hazard rates for each possible transition. The stochastic process is Markov, which means the transition rates depend only on the individual's current state, independent of their longer trajectory. 14 Though we only observed participants’ states annually, the continuous-time model accounts for the fact that transitions can occur at any time between visits. We have previously used these models to investigate changes in disease status or disease-relevant risk factors.15,16
Our multistate transition model estimates transition rates, which are the instantaneous risks of transitioning between two states; a rate can be thought of as a propensity to transition. These transition rates, all taken together, determine the future transition probabilities of being in each state, thereby connecting the rates to longitudinal data of the observed states. In brief, the transition matrix Q is the matrix whose entries
The transitions and states considered in this analysis are illustrated in Figure 1. Based on participant states and time between observations (in years), we estimated transition rates for the whole sample and converted the rates to transition probabilities, which we visualized with heatmaps. As a sensitivity analysis, we also estimated transition rates excluding participants’ visit numbers 7 or greater.
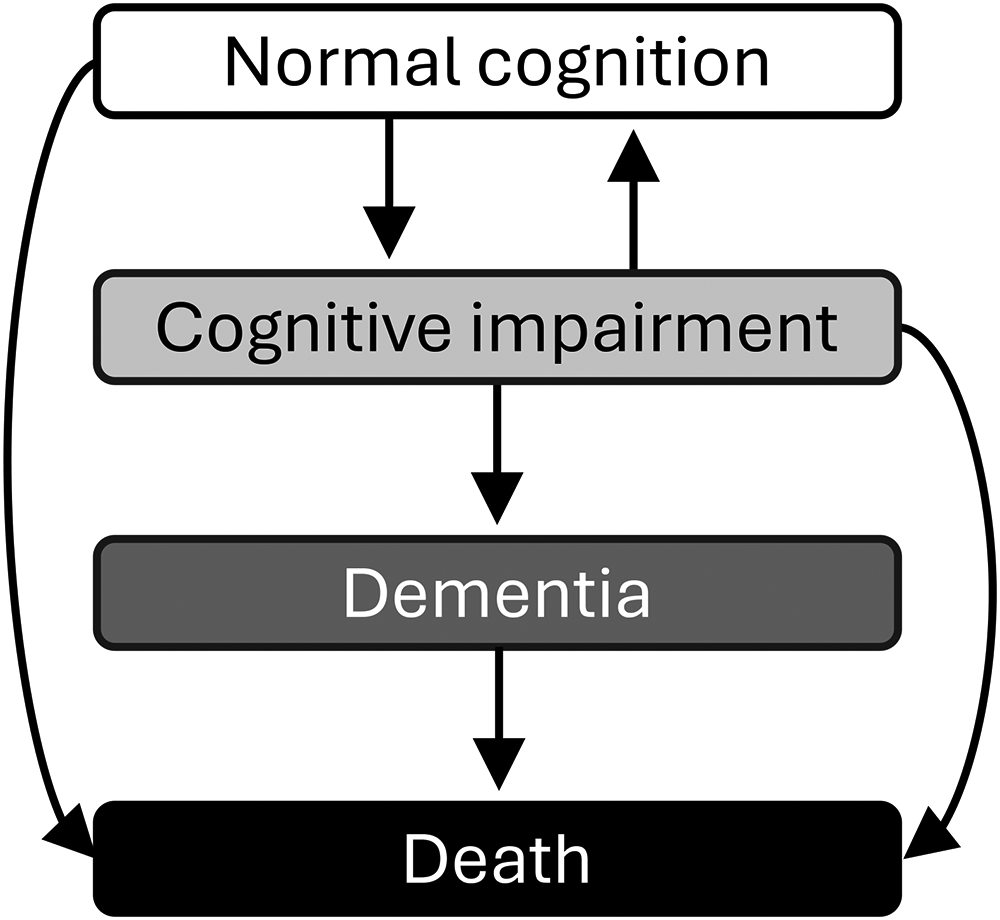
The transitions between clinical cognitive states in the multistate transition model of cognitive impairment and dementia.
We focused our covariate analysis on the four transitions that had sufficient observations: normal cognition to cognitive impairment, cognitive impairment to normal cognition, cognitive impairment to dementia, and dementia to death. We estimated covariate hazard ratios for each covariate separately, adjusting for age group (for variables other than age group), for these four transitions. Because of small numbers, we did not include covariates other than age group when estimating transitions rates from normal cognition or cognitive impairment to death. Missing values were coded as a separate “missing” category and not removed, so that the dataset was consistent across all analyses. To visualize the magnitude of the effect of covariates, we created heatmaps for transition probabilities for selected covariate levels (of age group, race and ethnicity, education, marital status, hypertension, diabetes, traumatic brain injury, and impairment type; for covariates other than age group, age group was set to the most common age group (70–74 years) for the purposes of visualization). As a sensitivity analysis, we also created a final multivariable model including age, sex, and race and ethnicity, as well as any variable that was statistically significant for at least one transition in a model adjusted for age group. As two additional, separate sensitivity analyses, we created age-group-adjusted models with 1) “lost-to-follow-up” as a separate state and different rates of loss-to-follow-up by cognitive state and 2) redefining the first instance of cognitive impairment to normal cognition to reduce the possibility that cognitive status misclassification caused by “reverse masking” is biasing the results. Transition rates were estimated using the MSM package (v1.7) 17 in R (v4.3.1).
Results
Descriptive statistics
The transition analysis included 960 participants who had been surveyed at least two times, with a total of 3484 observations. An alluvial plot of the observations is given in Supplemental Figure 1; 95% of observations were from visit numbers 6 or less. An abbreviated alluvial plot distinguishing between amnestic and non-amnestic impairment is given in Supplemental Figure 2, demonstrating that amnestic impairment was not always consistent over time. Table 1 shows the descriptive characteristics of the cohort at their baseline visit. The cohort was 59.8% female. The cohort was approximately three-quarters (72.0%) NH White and one-quarter (24.7%) NH Black. Education levels among participants varied widely, ranging from high school or GED to a doctorate degree. Most participants were married or partnered (69.1%). The prevalence of comorbid medical conditions and dementia symptoms ranged from more than 40% (hypercholesteremia, hypertension) to less than 3% (congestive heart failure, stroke). Because these are baseline descriptors and participants could develop conditions later in the study, the total number of observations for individuals with each condition is higher than the numbers in Table 1.
Characteristics of the study sample during their first visit.
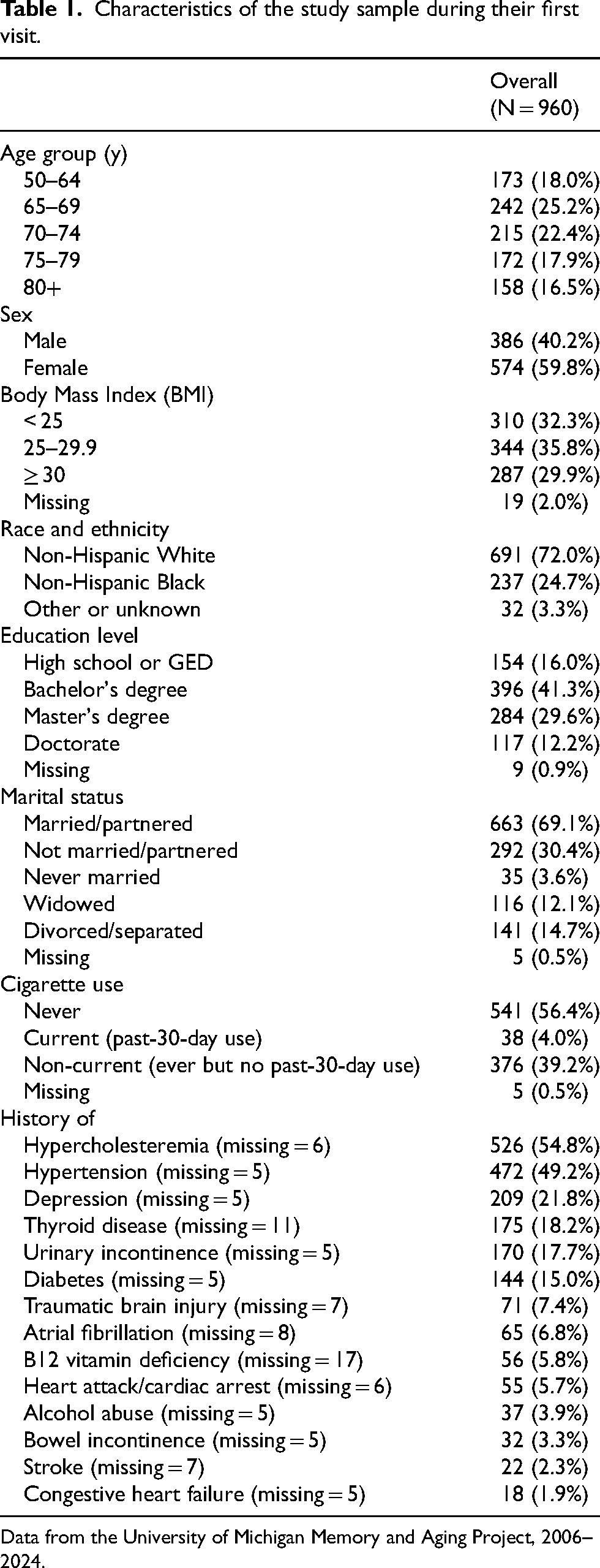
Data from the University of Michigan Memory and Aging Project, 2006–2024.
Transition probabilities and hazard ratios
Among pairs of observations where a participant started with normal cognition, we observed 1093 instances of participants remaining in the normal cognition state, 152 transitions to cognitive impairment, 4 transitions to dementia, and 8 deaths. Among pairs of observations where a participant started with cognitive impairment, we observed 446 instances of participants remaining in that state, 120 transitions to normal cognition, 87 transitions to dementia, and 13 deaths. Among pairs of observations where a participant started with dementia, we observed 525 instances of participants remaining in that state, and 76 deaths. These numbers do not account for the specific number of years between observations, and participants were counted multiple times if they have more than 2 observations.
The one-year transition probabilities—which do account for time between observations—between the four states are visualized in Figure 2A. We found high persistence within the states, as 89.0% (95%CI: 87.1, 90.5%) of participants with normal cognition would remain in their current state, as would 73.2% (95%CI: 69.6, 76.1%) of participants with cognitive impairment and 89.8% (95%CI: 87.4, 91.7%) of those with dementia. These probabilities correspond to mean residence times of 7.8 years, 3.1 years, and 9.3 years, respectively. The model also estimated that 10.1% (95%CI: 8.6, 11.6%) of participants with normal cognition would progress to cognitive impairment in one year, while 10.0% (95%CI: 8.3, 12.2%) of participants with cognitive impairment would develop dementia. Furthermore, there was also a potential of reversion, with 15.5% (95%CI: 13.0, 18.5%) of participants improving from cognitive impairment back to normal cognition in one year. We discounted the possibility of reverting from dementia (see Methods) and estimated 10.2% (95%CI: 8.3, 12.6%) of participants with dementia would die in a one-year period. Removing participant visits number 7 or greater did not change the transition probabilities substantially (Supplemental Figure 3), but relabeling each participant's first cognitive impairment status to normal cognition reduced the probability of transitioning from cognitive impairment to normal cognition from 15.5% to 10.8% (95%CI: 7.3, 16.1%) (Supplemental Figure 4).
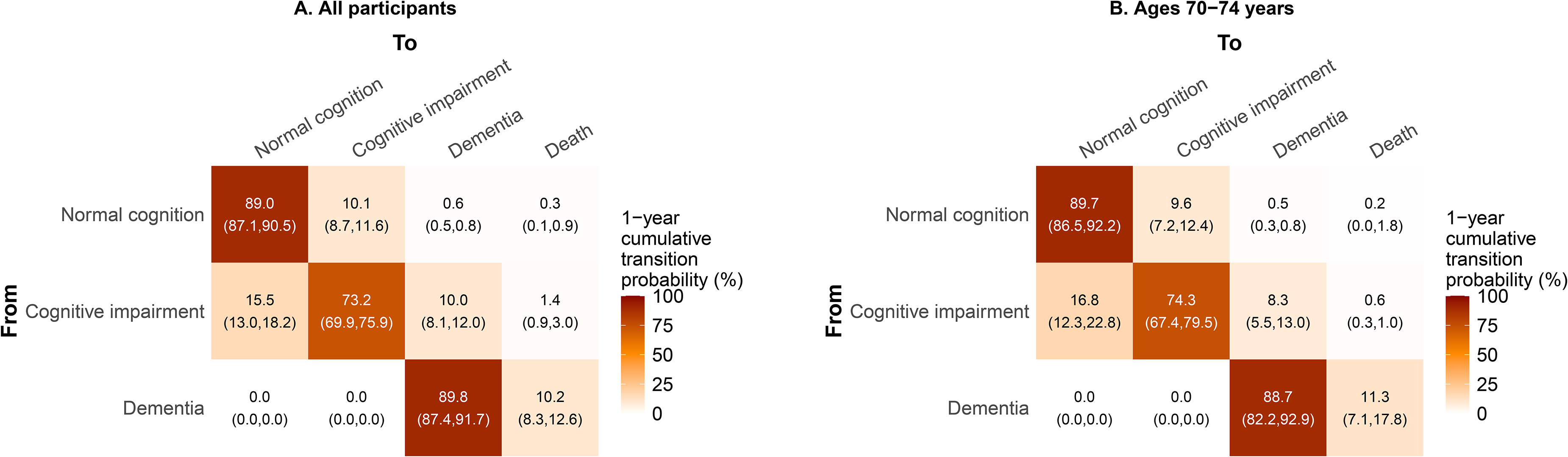
Modeled 1-year cumulative transition probabilities for transitions in cognitive state in a longitudinal cohort of 960 individuals in the University of Michigan Memory and Aging Project (2006–2024) for (A) all participants and (B) participants ages 70–74 years.
Estimates of covariate hazard ratios (HRs) for each sociodemographic and medical history covariate in age-group-adjusted and multivariable models are given in Table 2. In the age-group-only model, younger participants had a lower rate of transitioning from normal cognition to cognitive impairment compared to those ages 80+ (e.g., HR for ages 50–64 versus 80+: 0.24, 95% CI: 0.12, 0.49). We additionally estimated that younger participants also had a lower rate of transitioning from cognitive impairment to dementia, although these associations were not statistically significant. To visualize how these HRs translated to different transition probabilities, we evaluated the one-year transition probabilities for those ages 70–74 in Figure 2B (as the referent for all subsequent visualizations) and for 50–64 and 80 + in Figure 3A and 3B. Participants ages 50–64 had one-year transition probabilities of 4.7% (95%CI: 2.5, 8.1%) for the normal to cognitive impairment transition, lower than the 17.5% (95%CI: 12.7, 22.7%) for participants ages 80 + . Additionally, those ages 50–64 had lower transition probabilities (8.1%; 95%CI: 4.3, 14.3%) from cognitive impairment to dementia within one year compared to those ages 80+ (11.7%; 95%CI: 8.1, 17.0%).
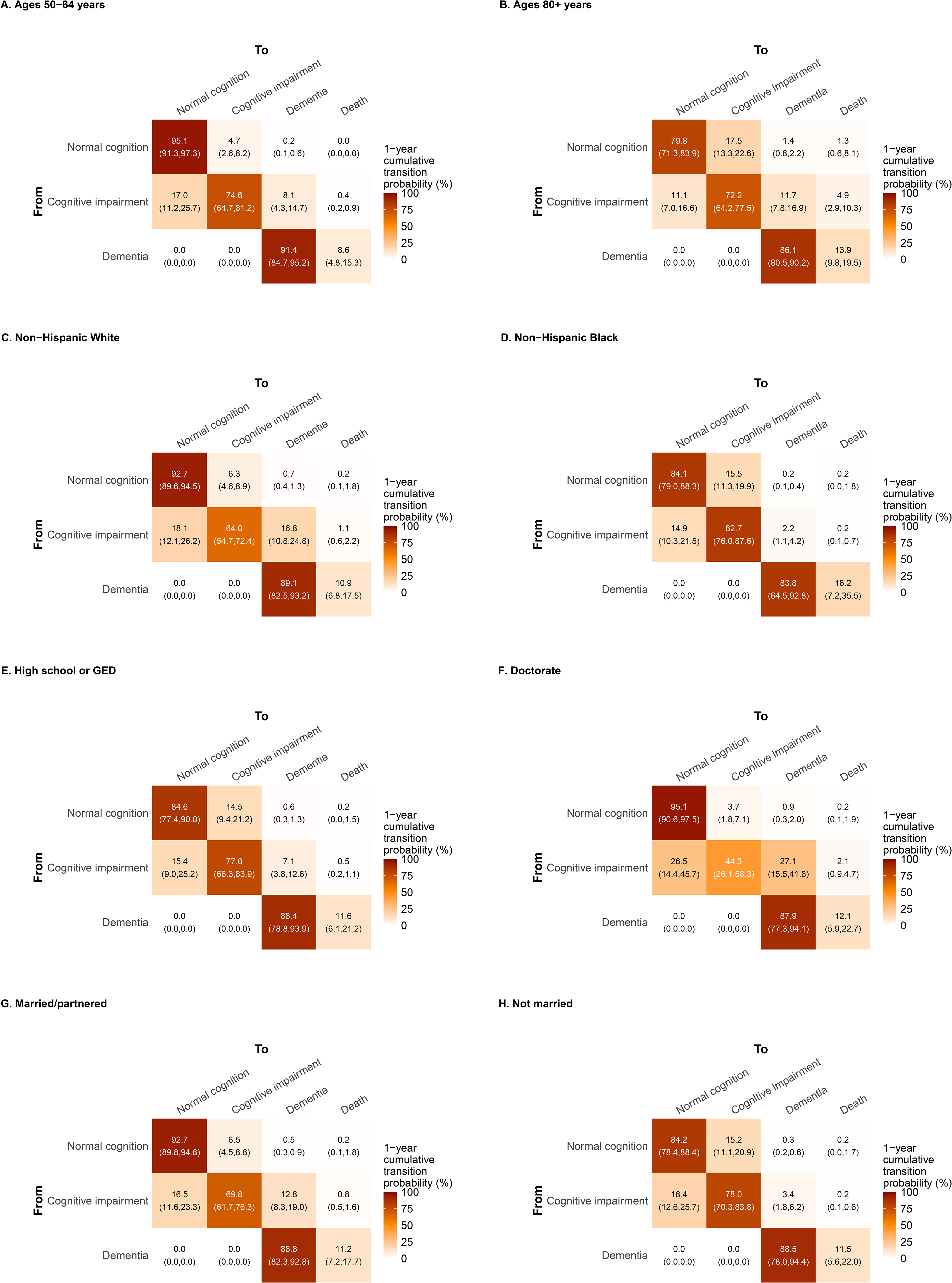
Modeled 1-year transition probabilities for cognitive state in a longitudinal cohort of 960 individuals in the university of Michigan memory and aging project (2006–2024) by selected groups of (A and B) age group, (C and D) race and ethnicity, (E and F) education level, and marital status (G and H). For the covariates other than age group, these probabilities are calculated for the 70–74 years age group, and so the plots can be compared to Figure 2B.
Covariate hazard ratios (HR) and 95% confidence intervals (CI) for transitions between cognitive states in age-adjusted and multivariable models.
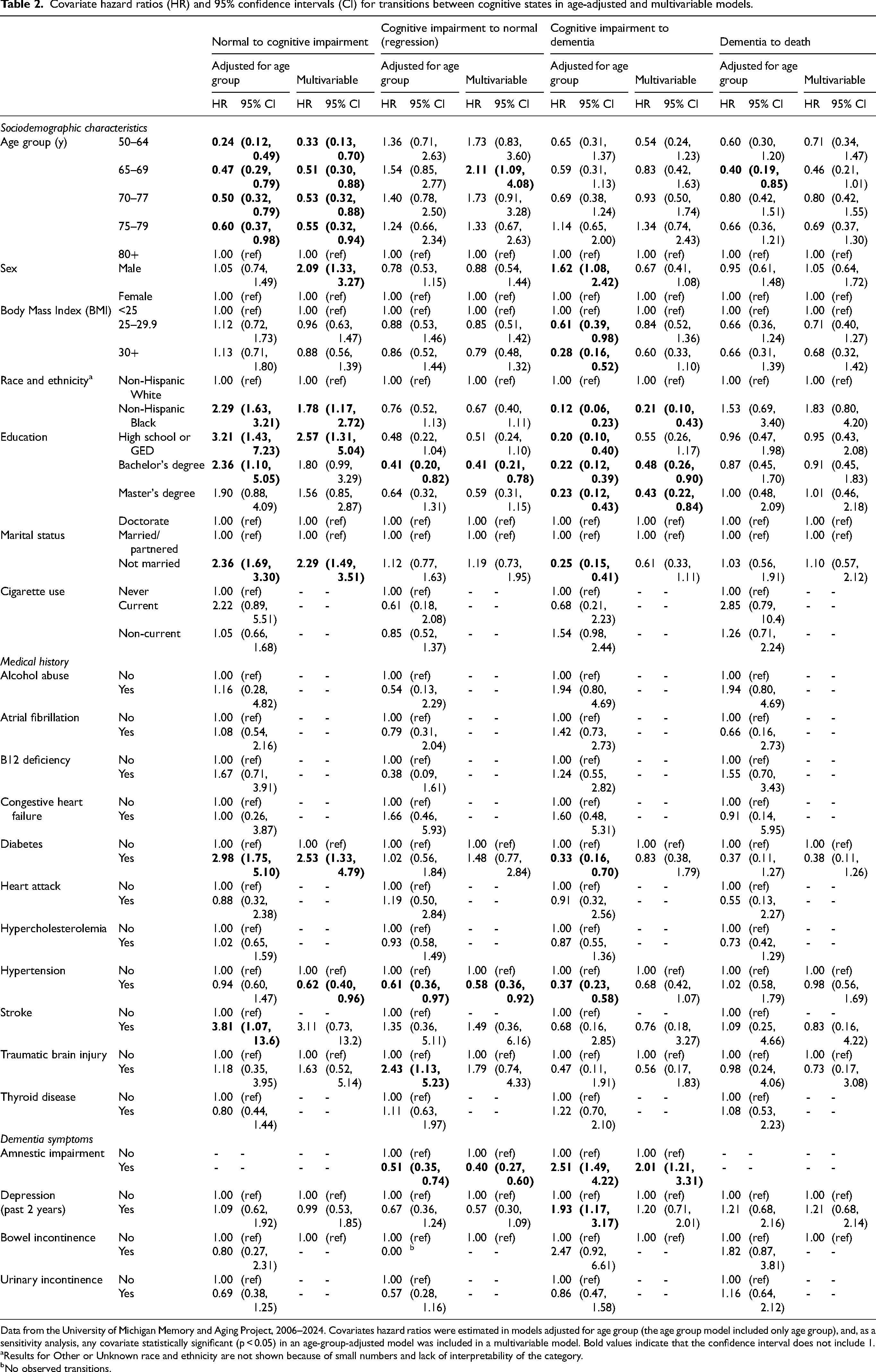
Data from the University of Michigan Memory and Aging Project, 2006–2024. Covariates hazard ratios were estimated in models adjusted for age group (the age group model included only age group), and, as a sensitivity analysis, any covariate statistically significant (p < 0.05) in an age-group-adjusted model was included in a multivariable model. Bold values indicate that the confidence interval does not include 1.
Results for Other or Unknown race and ethnicity are not shown because of small numbers and lack of interpretability of the category.
No observed transitions.
NH Black participants had a higher rate (HR: 2.29, 95% CI: 1.63, 3.21) of transitioning from normal cognition to cognitive impairment and a lower rate (HR: 0.12, 95% CI: 0.06, 0.23) of transitioning from cognitive impairment to dementia than NH White participants. Correspondingly, the one-year transition probabilities for NH Black individuals were 15.5% (95%CI: 11.4, 21.0%) and 2.2% (95%CI: 1.1, 4.6%), compared to 6.3% (95%CI: 4.6, 8.8%) and 16.8% (95%CI: 11.2, 24.6%) for NH White individuals for these transitions, respectively (Figure 3C and D; values are model estimates for 70–74-year-olds). NH White and Black participants did differ statistically significantly in education and reading level, but not in cognitive assessment (Supplemental Table 1).
Compared to those with a doctorate degree, those with a high school degree or GED holders had a higher rate of progressing from normal cognition to cognitive impairment (HR: 3.21, 95% CI: 1.43, 7.23) but a lower rate of transitioning from cognitive impairment to dementia (HR: 0.20, 95% CI: 0.10, 0.40). A similar pattern was seen for those with a Bachelor's degree compared to a doctorate degree. The one-year transition probabilities for normal cognition to cognitive impairment was 14.5% (95%CI: 9.5, 21.4%) and 3.7% (95%CI: 1.9, 7.3%) for high school degree or GED and doctorate degree holders respectively, while the one-year transition probabilities for cognitive impairment to dementia were 7.1% (95%CI: 3.8, 13.2%) and 27.1% (95%CI: 15.8, 44.3%) (Figure 3E and F; values are model estimates for 70–74-year-olds).
Marital/partner status was also significantly associated with the progression transitions. The HR for the transition from normal cognition to cognitive impairment was 2.36 (95% CI: 1.69, 3.30) for participants who were not married/partnered compared to those that were. However, this group also had a reduced rate of progressing from cognitive impairment to dementia in comparison with married/partnered participants (0.25; 95% CI: 0.15, 0.41). These differences are also reflected in the one-year transition probabilities matrix (Figure 3G and H; values are model estimates for 70–74-year-olds).
Only a few of the medical history variables were statistically significantly associated with one or more transitions in the bivariable models. Participants with a history of hypertension had a lower rate (HR: 0.61, 95% CI: 0.36, 0.97) of regressing from impaired to normal cognition and a lower rate (HR: 0.37, 95% CI: 0.23, 0.58) of progressing from cognitive impairment to dementia, compared to those without a history of hypertension. Individuals with a history of diabetes also had a higher rate (HR: 2.98, 95%CI: 1.75, 5.10) of progressing from normal cognition to cognitive impairment and a reduced rate (HR: 0.33, 95% CI: 0.16, 0.70) of progressing from cognitive impairment to dementia, compared to those without diabetes. Participants with a history of stroke also had a higher rate of transitioning from normal cognition to cognitive impairment (HR: 3.81, 95%CI: 1.07, 13.6) compared to those without.
Participants with a history of traumatic brain injury had a higher rate (HR: 2.43, 95% CI: 1.13, 5.23) the rate of regression to normal cognition than people without a prior traumatic brain injury. Those with recent depression had a higher rate of transitioning from cognitive impairment to dementia (HR: 1.93, 95%CI: 1.17, 3.17). Finally, amnestic impairment was associated with a reduced rate (HR: 0.51, 95% CI: 0.35, 0.74) of regression to normal cognition and a greater rate (HR: 2.51, 95% CI: 1.49, 4.22) of progression to dementia compared to non-amnestic impairment. The transition probabilities associated with selected conditions are given in Figure 4 (values are model estimates for 70–74-year-olds).
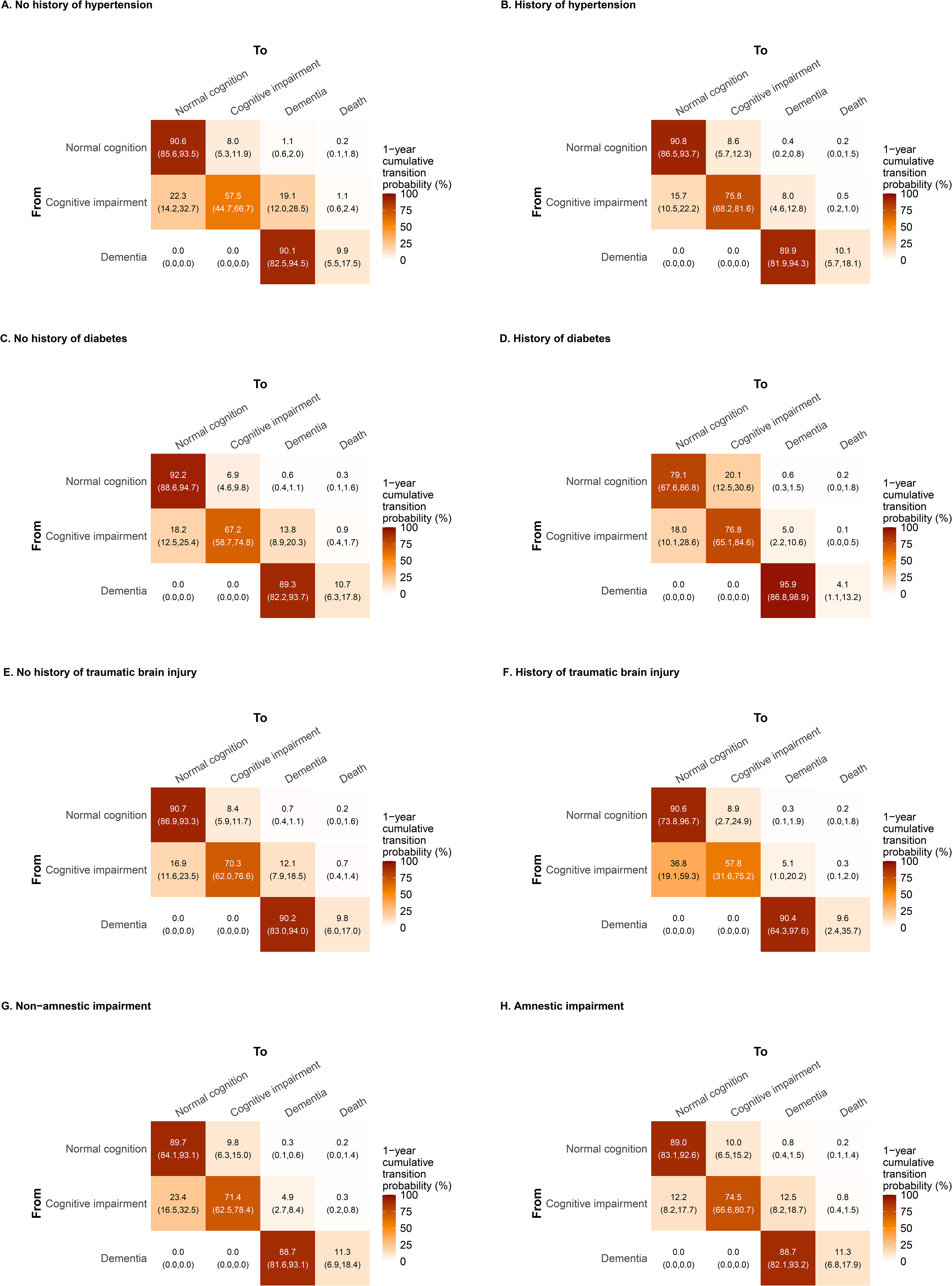
Modeled 1-year cumulative transition probabilities for cognitive state in a longitudinal cohort of 960 individuals in the University of Michigan Memory and Aging Project (2006–2024) by history of hypertension (A and B), diabetes (C and D), traumatic brain injury (E and F), and amnestic impairment (G and H). These probabilities are calculated for the 70–74 years age group, and so the plots can be compared to Figure 2B.
In the multivariable sensitivity analysis, many of the sociodemographic variables, e.g., age group, race and ethnicity, and education, retained similar, if sometimes attenuated, associations. Interestingly, sex, which was not associated with the normal cognition to cognitive impairment transition in the age-adjusted model, became a statistically significant predictor in the multivariable model (HR 2.09, 95%CI: 1.33, 3.27, for men versus women). For the medical comorbidities, diabetes and hypertension remained significant for at least one transition while stroke and traumatic brain injury did not (although the point estimates remained in the same direction as before). In the multivariable model, recent depression was no longer significant for any transitions. The association between amnestic type impairment and transitions from cognitive impairment to normal cognition and dementia remained similar.
For the other sensitivity analyses, the transition age-adjusted models relabeling the first cognitive impairment status as normal cognition are given in Supplemental Table 2; the hazard ratios remained similar, though there was a loss of statistical power for transitions from cognitive impairment. The overall transition probabilities and age-adjusted models accounting for differential loss-to-follow-up by cognitive status are given in Supplemental Figure 5 and Supplemental Table 3; the results again remain broadly similar, with few covariates statistically significantly associated with loss to follow up.
Discussion
In a clinically well-characterized longitudinal cohort of older adults in Southeast Michigan, we estimated transitions rates and probabilities for progression from normal cognition to cognitive impairment and dementia, as well as regression from cognitive impairment to normal cognition. We also estimated associations between sociodemographic characteristics, medical history, and dementia symptoms and these transitions. As expected,1,4 age was a dominant factor in the onset of cognitive impairment. Among the other covariates, a common pattern emerged: characteristics associated with a faster progression from normal cognition to cognitive impairment were often also associated with a slower progression from cognitive impairment to dementia. In the age-group-adjusted models, this pattern was seen for race and ethnicity, marital status, and education, as well as by history of diabetes. As discussed below, we hypothesize that this pattern could result from the cognitive reserve hypothesis, systematic misclassification of cognitive status, or from the difficulty in distinguishing which people with cognitive impairment have a progressive, neurodegenerative condition. These findings may inform clinical practice and help to develop population-level projections.
The cognitive reserve hypothesis, cognitive status misclassification, and the cognitive impairment umbrella
Our primary finding was that multiple characteristics, including education, were associated with faster transitions to impairment paired with slower transitions to dementia (or vice versa). These results could be potentially consistent with the cognitive reserve hypothesis, which suggests that some individuals have a greater adaptability of neural networks, offering resistance to cognitive decline and impairment.18–20 A higher cognitive reserve is associated with higher later-in-life cognitive function, delaying the onset of symptoms of neurodegenerative decline and influencing the clinical diagnosis. Although a higher cognitive reserve might be expected to reduce progression to both cognitive impairment and to dementia, it has also been hypothesized that once the disease progresses to a certain extent, it may seem to advance more rapidly, as early brain changes or damage may have simply been masked rather than prevented (Figure 5A).
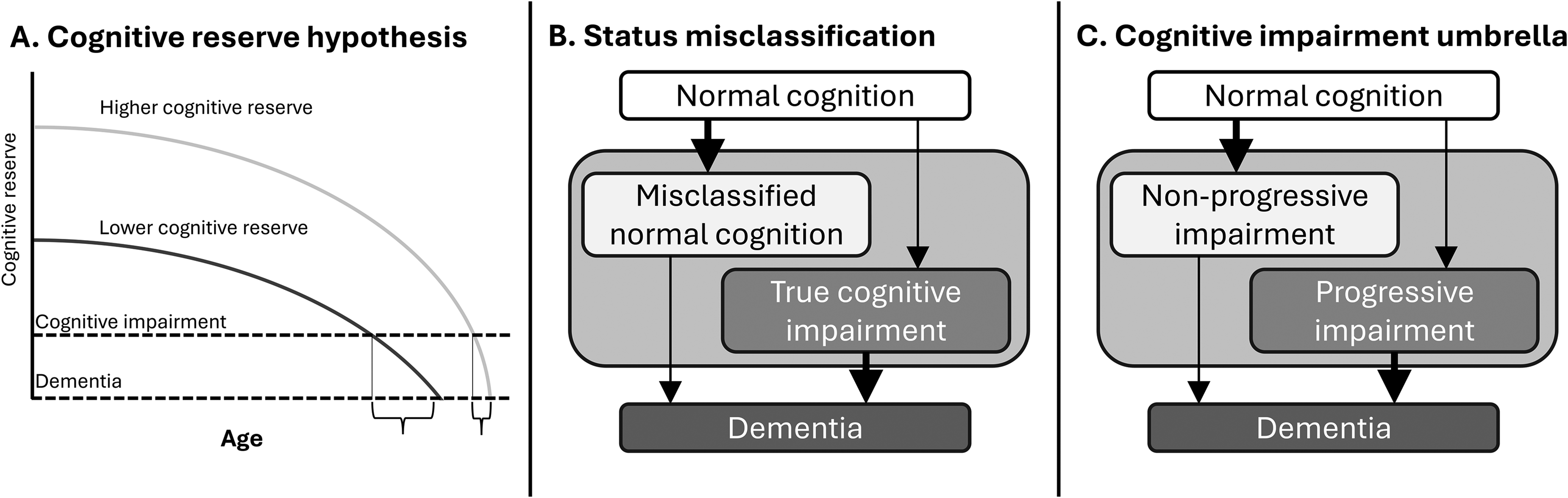
Three models of how faster transitions to cognitive impairment may be associated with slower transitions to dementia. (A)
We propose that our results for education level and marital status may be related to the cognitive reserve hypothesis. Previous work has strongly associated higher educational attainment with higher cognitive reserve, through the tolerance of the disease-related pathology and enhanced neural networks.5,18,21 Although the protective effect of higher educational attainment on the transition to cognitive impairment was not statistically significant, there was a gradient in the expected direction, and we did find that participants who had obtained a doctorate degree progressed from cognitive impairment to dementia at a greater rate than participants with lower educational attainment. We also found that married/partnered participants were less likely to transition to cognitive impairment but more likely to progress to dementia than those who were either never married or widowed, divorced, or separated. These results are also consistent with the prior literature.22,23 The idea that marital status may be associated with the cognitive reserve hypothesis is grounded in the understanding that people with partners may benefit from higher access to social interaction, leisure activities, and emotional support. 24
Alternatively, the results by educational attainment could be a function of measurement error in the form of systematic misclassification of cognitive status. Cognitive status is largely measured by tests, a form of evaluation that people with higher education are more practiced at. So, some participants with a high school or equivalent education and normal cognition may be misclassified as cognitively impaired, while some participants with a doctorate degree and dementia may be misclassified as cognitively impaired. Covariates associated with a higher likelihood of being misclassified as cognitively impaired when having normal cognition would have higher rates of transition to cognitive impairment and lower rates of transitioning from cognitive impairment to dementia (Figure 5B).
Systematic misclassification may also explain the results by race and ethnicity. We observed that NH Black participants had a higher rate of transitioning to cognitive impairment (HR 2.29, 95%CI: 1.63, 3.21) compared to NH White participants, yet they progressed to dementia much more slowly (HR 0.12, 95%CI: 0.06, 0.23). Slower progression from impairment to dementia for NH Black versus White participants was similarly reported by Gleason et al. 25 One possible explanation for our results is that NH Black participants in this study with cognitive impairment had higher kinship and community support compared to NH White participants, resulting in delayed progression to dementia. Alternatively, the misclassification explanation is that the cognitive impairment status assigned to the NH Black participants may not represent a meaningful cognitive decline caused by underlying neurodegenerative disease but rather represent an overall lower cognitive test scores caused by cumulative minority stress and systemic disparities in socioeconomic status and educational quality.26–28 Indeed, in this study NH Black participants had overall lower reading level and educational attainment (though the fractions of those with a Bachelor's and Master's degrees were nearly the same) compared to NH White participants in this study (Supplemental Table 1). Both educational attainment and race and ethnicity remained significant predictors of transitions in the multivariable model, but future work could examine whether educational attainment or reading level are effect modifiers of the associations between race and ethnicity and transitions in cognitive status. We also note that the result about slower transitions from cognitive impairment to dementia may appear at first to conflict with studies indicating that NH Black people have poorer late-in-life cognition and a higher prevalence of Alzheimer's disease and related dementias, 29 but they are not necessarily contradictory; further simulation work would be needed to determine how these two transitions combine to predict prevalence of dementia at any given age.
The pattern of covariates with opposite associations with transitions from normal to cognitive impairment and transitions to dementia could also be consistent with a situation where true cognitive impairment has a variety of causes, some of which are associated with progressive neurodegeneration and others are not. For brevity, we will refer to this clinical classification problem as the “cognitive impairment umbrella.” Some individuals may present with mild cognitive decline because of other systemic health issues; because the underlying etiology of the cognitive impairment is different, these cognitively impaired individuals would be less likely to develop dementia than individuals whose cognitive impairment is more likely to have progressive neurodegenerative causes (Figure 5C).
The results for diabetes may be explained by this cognitive impairment umbrella. That is, diabetes may be associated with real, mild but not necessarily progressive cognitive impairment, but, among people with cognitive impairment, people with diabetes progress to dementia slower than those without, as those without diabetes are more likely to have cognitive impairment because of degenerative decline. Although diabetes has been associated with increased risk of developing dementia, other studies have suggested that the diabetes-related cognitive impairment is not necessarily progressive and may be reversable if the underlying pathophysiology is addressed.30–32
The results by marital status could also be related in part to a combination of misclassification and the cognitive impairment umbrella. Partner-reported symptoms have been shown to be more predictive of progressive cognitive decline than self-reported symptoms,33,34 so that cognitively impaired individuals without partners may be less likely to have progressive neurodegenerative disease than those with partners.
Our results for traumatic brain injury are qualitatively different from the other characteristics in that we observed a significant positive association with reversion from cognitive impairment to normal cognition. These results are also likely a result of the cognitive impairment umbrella. Individuals with traumatic brain injury may experience cognitive impairment and symptoms such as memory deficits.35,36 However, the recovery potential we found in this analysis suggests the impairment observed after the injury may be temporary and may not progress to dementia. Other studies have suggested that traumatic brain injury may contribute to pathological processes and facilitate the development of dementia,37–39 so further work in this area is needed.
Other predictors
If other medical conditions may cause cognitive impairment that does not necessarily progress to dementia, it may be possible to distinguish between types of cognitive impairment that are progressive and those that are not. Our results regarding amnestic and non-amnestic impairment are a step in the right direction. We found that amnestic impairment, within either a single domain or multiple domains, was predictive for a greater progression from impairment to dementia (HR 2.51, 95%CI: 1.49, 4.22) compared to non-amnestic impairment and a reduced reversion from impairment to normal cognition (HR 0.51, 95%CI: 0.35, 0.74), consistent with previous studies. 40 The presentation of memory deficits that exceed what is normally expected for one's age often indicates underlying neuropathological changes. Advanced imaging techniques and biomarker studies have enabled the identification of these changes in individuals at the amnestic stage, further supporting their predictive significance for dementia.41,42 We did not separate amnestic and non-amnestic cognitive impairment into separate classes in our model because the categories were not stable over time (Supplemental Figure 2), likely a limitation of current clinical diagnostic tools. Nevertheless, our results suggest that it may be possible to distinguish progressive and non-progressive cognitive impairment.
Hypertension, which can cause cerebrovascular pathology, is an established risk factor for vascular dementia but has been linked in some studies to higher risk of Alzheimer's disease as well.43,44 However, some meta-analyses have found no link between hypertension and Alzheimer's disease45,46 and other studies have suggested the relationship may be modified by age. 47 In our study, we found that a history of hypertension was protective against the transition from cognitive impairment to dementia (and, in the multivariable model, protective against developing cognitive impairment). One important limitation is that we do not know whether the participants used antihypertensives, which have been suggested to reduce Alzheimer's disease risk. 48 Our work further underscores the complicated relationship between hypertension and dementia.
Finally, our results indicating that recent depression was associated with a faster progression to dementia should be interpreted with caution. In addition to being symptoms of neurodegeneration, dementia symptoms may also directly influence the clinical assessment. We included them in this analysis because, regardless of whether they influence the measurement of cognitive status, accounting for them could be useful for predicting changes in an individual's clinical cognitive status.
Strengths and limitations
One strength of our analysis is in the moderately large, longitudinal dataset, with nearly 1000 participants with about 3500 total clinical diagnostic observations, providing a robust foundation for our results. Additionally, the multistate transition model implemented here bridges the gap between separately observed statuses, translating them holistically into continuous transition rates and estimating interpretable hazard ratios for potential risk and protective factors. Additionally, our results were largely robust to multiple sensitivity analyses. Two limitations of our analysis include the focus only on sociodemographic and medical history covariates, as well as the use of clinical diagnosis based primarily on cognitive testing rather than biomarker or neuroimaging information that could provide information about the actual underlying neurodegeneration. We also did not have temporal information about the timing of the medical history conditions, such as TBI, which reduces their interpretability. The sample size was also such that there were relatively few cases of the less common medical conditions that may be associated with cognitive decline. Accordingly, some results may be subject to sparse data bias, and estimates with large uncertainty should be interpreted with care, even when statistically significant. Additionally, our data, which includes participants from Michigan only, are not nationally representative. This regional limitation may restrict the generalization of our findings to a larger context. In future research, we plan to expand our work to larger-scale studies that are representative of broader populations, thereby enhancing the external validity of the findings. Finally, there was differential loss-to-follow by cognitive status, which may have led to attrition bias, although our sensitivity analysis suggests such affects are likely minimal.
Conclusion
Our analysis highlights the transition patterns in cognitive decline and progression to dementia. These findings underscore the predictive value of certain characteristics and medical history for cognitive decline, but they also highlight multiple challenges in interpreting the association between transitions, particularly when working with clinical interpretations of cognitive impairment and dementia that are based primarily on cognitive tests (as opposed to biomarkers or imaging). We identified a common pattern of characteristics associated with faster transitions to cognitive impairment also being associated with slower transitions from cognitive impairment to dementia, and we discussed how this pattern could be related to cognitive reserve, cognitive status misclassification, or non-progressive cognitive impairment. Our work emphasizes the need to better understand and predict when cognitive impairment is likely to progress to dementia, and our results regarding amnestic versus non-amnestic impairment are an important step in this direction. Ultimately, this study helps to provide insights that can enhance the understanding of dementia, will contribute to the development of personalized estimates of future cognitive trajectories, and will enable more accurate, large-scale predictions for future disease burdens.
Supplemental Material
sj-docx-1-alz-10.1177_13872877251324236 - Supplemental material for Predictors of transitions between normal cognition, cognitive impairment, and dementia in a longitudinal cohort
Supplemental material, sj-docx-1-alz-10.1177_13872877251324236 for Predictors of transitions between normal cognition, cognitive impairment, and dementia in a longitudinal cohort by Nan Shi, Kelly M Bakulski, James F Burke and Andrew F Brouwer in Journal of Alzheimer's Disease
Footnotes
Acknowledgements
We thank Jonathan Reader and Allyson Gregoire of the Michigan Alzheimer's Disease Research Center's Data Management and Statistical Core for their assistance with data preparation.
Ethical considerations
This secondary data analysis was determined to be not regulated as human subjects research by the University of Michigan Institutional Review Board (HUM00240110).
Consent to participate
All the participants gave written informed consent.
Author contribution(s)
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was partially supported by the National Institute of Aging of the National Institutes of Health through the Michigan Alzheimer's Disease Research Center (P30AG072931) and Michigan Center for the Acceleration of Population Research in Alzheimer's (P30AG066582), as well as the University of Michigan Alzheimer's Disease Center, Berger Endowment (G102865/321033).
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability
The data are available to researchers with Institutional Review Board approval through the Michigan Alzheimer's Disease Research Center (https://medresearch.umich.edu/labs-departments/centers/madc/research/investigator-researcher-support) or through the National Alzheimer's Coordinating Center (![]() ).
).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
