Abstract
Background
An Alzheimer's disease (AD) diagnosis made in the earliest symptomatic stages substantially benefits patients and their care partners. However, little is known regarding the clinical, healthcare system-level, and patient-specific barriers that hinder timely diagnosis and treatment.
Objective
To explore real-world practices surrounding the diagnostic journey and management of mild cognitive impairment (MCI)/AD dementia patients.
Methods
Data were drawn from Adelphi Real World Dementia Disease Specific Programme™, a cross-sectional survey of physicians treating MCI/AD dementia patients in France, Germany, Italy, Spain, the United Kingdom, the United States, and Japan between 2022 and 2024.
Results
Overall, 779 physicians reported data on 5551 patients. Physicians indicated current disease severity for 5421 patients; 37.2% had MCI (87.3% with suspected prodromal AD and 12.7% undetermined etiology), 17.2% AD with mild dementia, 31.1% AD with moderate dementia, and 14.5% AD with severe dementia. When not immediately diagnosed
Conclusions
Timely MCI and AD diagnosis is impeded by referral delays and limited use of biomarker testing. Addressing these critical care gaps requires enhanced physician training, reduced wait times and increased biomarker utilization for early management.
Keywords
Introduction
More than 51 million people globally are living with Alzheimer's disease (AD) or a related dementia. The prevalence of dementia is predicted to rise to 152.8 million cases worldwide by 2050. 1 AD is a progressive, neurodegenerative disorder and the most common form of dementia, contributing to 60–80% of all dementia cases. 2
AD is seen as a continuum, which follows a progressive disease course, extending from an asymptomatic (preclinical) phase, to mild cognitive impairment (MCI), and ultimately culminating in AD with mild, moderate or severe dementia. 3 The primary pathophysiological mechanisms of AD are the accumulation of amyloid-β (Aβ) plaques followed by formation of tau-neurofibrillary tangles (NFTs), which result in neuronal dysfunction, neuronal cell loss and cognitive dysfunction. 4 This process is referred to as the amyloid cascade, a hypothesis which has been supported and debated in recent studies.5–11
Making a diagnosis of AD has historically relied solely on clinical assessment, including patient medical history and informant report, alongside cognitive, functional, and behavioral assessments.12–15 However, the clinical diagnosis of MCI or AD with mild dementia may be inaccurate due to the subtlety of early cognitive symptoms that may not be detected by clinical assessment, as well as the overlap in clinical presentation with other comorbidities that may cause cognitive and functional decline16–21; both under-diagnosis and incorrect diagnoses have been reported.22,23 Furthermore, features of the healthcare system may hinder early AD detection. These include differences in education and training of professionals involved in diagnosis 24 as well as the rapid evolution, unequitable availability and inconsistent reimbursement of diagnostic and biomarker assessments required to confirm AD pathology 25 alongside the variable availability of dementia physicians. 26 Furthermore, although routine cognitive assessment of older adults is recommended,27,28 specific frameworks for the implementation of this are lacking. Finally, psychological barriers still exist for both patients and providers, including AD stigma and a fear of a loss of autonomy, which affect patients’ willingness to be referred for formal assessment when concerns are raised,29–31 and lack of confidence among PCPs in diagnosing and caring for patients with AD. 32
Nonetheless, timely diagnosis is crucial as it offers multiple advantages, including time for patients and their families to plan and the opportunity to initiate interventions such as diet and lifestyle changes,33–36 assess interest and eligibility for new and anticipated disease modifying therapies 28 and provide care partner support and education. 37
Evidence from in vivo and in vitro biomarker studies demonstrates the potential to improve the accuracy of AD diagnosis. The pathological hallmarks of AD, such as Aβ and tau, can be detected via amyloid- and tau-positron emission tomography (PET) imaging scans and cerebrospinal fluid (CSF) biomarkers. 38 Blood-based biomarker assays for Aβ and tau are also becoming clinically available to assess the presence of Aβ and tau pathology in adults with symptoms of cognitive impairment.39–41
Current evidence-based management of AD includes a multi-factorial and tailored approach between the physician, patient and care partner. At a minimum, this includes pharmacological and non-pharmacological treatment and management of psychological and behavioral symptoms of dementia, 42 care navigation and access to home and community based services, 43 and care partner support and education. 37 In the past few years, amyloid-targeting therapies (ATTs) such as lecanemab and donanemab have demonstrated the ability to slow cognitive decline in participants with early symptomatic AD and evidence of AD neuropathology.6,7 This has increased the relevance of identifying patients with clinical symptoms and AD pathology who may be eligible for ATTs in addition to evidence-based care management. However, identifying patients who will benefit most from ATTs might be hampered by a lack of timely detection of AD.
There is a need for a deeper understanding around the current diagnostic journey and management of patients with MCI and early AD dementia, to inform care guidelines and health policies to optimize outcomes for patients. A multinational perspective is also crucial, as factors affecting timely diagnosis may differ across locations and healthcare systems. Therefore, this analysis aimed to describe the real-world journey from clinical presentation to treatment in the United States (US), Europe (France, Italy, Spain, Germany, and the United Kingdom [UK]), and Japan. The results of a large-scale physician survey conducted using well-established methodology were evaluated to shed light on real-world practices and to explore challenges and barriers to timely MCI and AD dementia diagnosis.
Methods
Adelphi Real World Disease Specific Programmes (DSPs)™ are large, multinational, cross-sectional surveys conducted in clinical practice to collect information on disease management, including diagnosis and treatment history. The DSP methodology has been previously published, validated44–47 and found to be consistent over time. 46 Briefly, eligible physicians complete a questionnaire relating to patient(s) under their care, using information available to them at the time of consultation including retrospective data from medical records. Patients and/or care partners may also complete a separate questionnaire.
Data source
Data were drawn from the Adelphi Real World Dementia DSP survey of physicians in Europe (France, Germany, Italy, Spain, the UK), the US, and Japan conducted from December 2022 to March 2024.
Participant selection
Primary care physicians (PCPs) including General Practitioners (GPs) and internists (US and Japan only) as well as specialists (neurologists, psychiatrists, geriatricians, psychogeriatricians, neuropsychiatrists and neurosurgeons) were eligible to participate in the survey if they were personally responsible for the management and treatment of patients with a physician-confirmed diagnosis of MCI or dementia. Physicians had to have a minimum clinical workload of at least three patients (for PCPs) or at least five patients (for specialists) in a typical week, excluding patients who had previously been admitted to a nursing home/hospital for an extended stay (defined as ≥3 months).
Patients were eligible for inclusion in the survey if they were aged 50 years or older and had a physician-confirmed diagnosis of MCI or dementia. Patients were excluded if they had vascular dementia only, had dementia caused by environmental factors (e.g., traumatic head injury, alcoholism), or were enrolled in a clinical trial at the time of data collection.
This paper focuses on patients with a current diagnosis of MCI (encompassing prodromal AD, predementia, and amnestic/non-amnestic MCI due to either suspected AD or undetermined etiology) or AD dementia (AD, early onset AD, mixed dementia).
Data collection
Physicians completed a survey that measured their attitudes and beliefs about dementia diagnosis, treatment, and management alongside a patient record form for approximately nine consecutively consulting patients who they saw for routine care and who met the patient eligibility criteria noted above. The number of patient record forms completed varied between physicians, but the total number of forms completed overall was sufficient to meet recruitment aims.
Physician survey items included specific questions about timely identification of patients with MCI and AD, reasons for delays to diagnosis, and the role of biomarker testing in diagnosing patients with AD.
Patient record forms included patient demographics, clinical characteristics, behavioral and physical symptoms, physician first consulted, diagnosing physician, time from first consultation to diagnosis, diagnostic tests, disease severity, and current treatment and management. Physicians were asked to report whether their patients currently (i.e., at the time of survey completion) had ‘MCI’ or ‘AD dementia’. For patients reported as having ‘AD dementia’, physicians were then asked to determine current disease severity as ‘mild’, ‘moderate’, or ‘severe’, using information collected during the consultation, patient/family feedback and their own judgement and diagnostic skills.
Physicians provided disease severity data at patients’ initial consultation and initial diagnosis, using data extracted from patients’ medical records. Disease severity at initial consultation was reported via the most common test to measure cognitive performance in the survey, namely Mini-Mental State Examination (MMSE) scores, where available. The MMSE is a 30-question assessment of cognitive function that evaluates attention and orientation, memory, registration, recall, calculation, language, and ability to draw a complex polygon. 48
Severity at initial diagnosis was reported via physician-determined severity and/or the MMSE score where available. Not all patients had their initial diagnosis confirmed using MMSE, possibly due to a lack of time in clinic and/or use of alternative assessments to aid diagnosis and measure severity.
MMSE bandings used to classify clinical severity in this study are geography-specific and in accordance with a relevant technology appraisal guidance. 49 For the UK, MMSE scores of 27–28 were used to categorize patients as having mild cognitive impairment, MMSE 21–26 for AD with mild dementia, MMSE 10–20 for AD with moderate dementia, and MMSE 0–9 for AD with severe dementia. 49 For all other geographies, bandings are as follows: MMSE 27–28 for mild cognitive impairment, MMSE 20–26 for AD with mild dementia, MMSE 10–19 for AD with moderate dementia, and MMSE 0–9 for AD with severe dementia.
In summary, this paper presents disease severity primarily based on physician-reported severity (e.g., clinical judgement), and MMSE scores where possible and available.
Data analysis
Means and standard deviations (SD) and medians and interquartile ranges [IQR] were calculated for continuous variables, and frequency and percentages were calculated for categorical variables. Missing data were not imputed, and therefore denominator sizes vary for some variables.
A post-hoc subgroup analysis was conducted on all patients with a current diagnosis of ‘MCI’ and ‘AD with mild dementia’. This subgroup represents patients with a similar clinical severity as participants in recent clinical trials of ATTs for the treatment of early symptomatic AD,6,7,50 although the current survey also included MCI patients for whom it is uncertain whether they will develop AD as explained in the Participant Selection section.
All analyses were conducted using STATA® Version 17 (StataCorp 2021, College Station, TX: USA StataCorp LLC).
Ethics and consent
Data collection was undertaken in line with European Pharmaceutical Marketing Research Association guidelines 51 and as such did not require ethics committee approval. Each survey was performed in full accordance with relevant legislation at the time of data collection, including the US Health Insurance Portability and Accountability Act 1996 52 and Health Information Technology for Economic and Clinical Health Act legislation. 53 No patients or physicians can be identified directly in the data. Ethnicity was not recorded in France or Japan in accordance with country-specific guidelines. Data were aggregated before being shared with the subscriber and/or for publication.
This study was granted ethical exemption by the Pearl Institutional Review Board, approval number (IRB number: #22-ADRW-172/3).
Results
Physician characteristics
Physician characteristics are summarized in Supplemental Table 1.
A total of 765 physicians completed an attitudinal survey and 779 physicians provided at least one patient record form. Across all countries, 44.1% of physicians were PCPs, including GPs and internists (US and Japan only), and 55.9% were specialists, including neurologists (32.7%), psychiatrists (10.8%), geriatricians (5.2%), psychogeriatricians (5.0%), neurosurgeons (1.6%), and neuropsychiatrists (0.7%).
Where reported (n = 250), neurologists’ specialties were mostly general neurology (62.4%), memory disorders/dementia (54.0%), and/or neurodegenerative diseases (48.0%). A high proportion of neurologists specialized in memory disorders/dementia and/or neurodegenerative diseases in Japan (≥80%) and Europe (around 60%). In the US, general neurology was the most common neurology specialty (69.1%).
Overall, 33.7% and 30.8% of physicians’ patients were seen in a public and private non-hospital medical facility, respectively, and 21.2% and 12.5% in public hospitals and private hospitals. Most patients in the US and France were seen in a private, non-hospital facility, and most patients in Germany, Spain, the UK, and Italy were seen in a public, non-hospital facility (Supplemental Table 1). In Japan, most patients were seen in a private hospital (53.1%). A total of 29.7% of physicians were working in a memory clinic, representing 44.4% of physicians in Germany and 20.6% of physicians in Japan. Physician characteristics across countries are shown in Supplemental Table 1.
Patient characteristics
Physicians reported data for 5551 patients. Overall, 23.0% of patients were from the US (n = 1275), 12.9% from France (n = 714), 12.3% from Germany (n = 680), 12.4% from Italy (n = 686), 13.0% from Spain (n = 723), 12.9% from the UK (n = 717), and 13.6% from Japan (n = 756). Patient demographics are shown in Table 1. Additionally, Figure 1 shows sample sizes for overall cohort and subgroup analyses.

Physician and patient sample size for overall cohort and subgroup analysis. AD, Alzheimer's disease; MCI, mild cognitive impairment; UK, United Kingdom; US, United States. *14 physicians in the UK did not complete the physician survey.
Physician-reported baseline demographics and clinical characteristics of patients with a current MCI or AD dementia diagnosis.
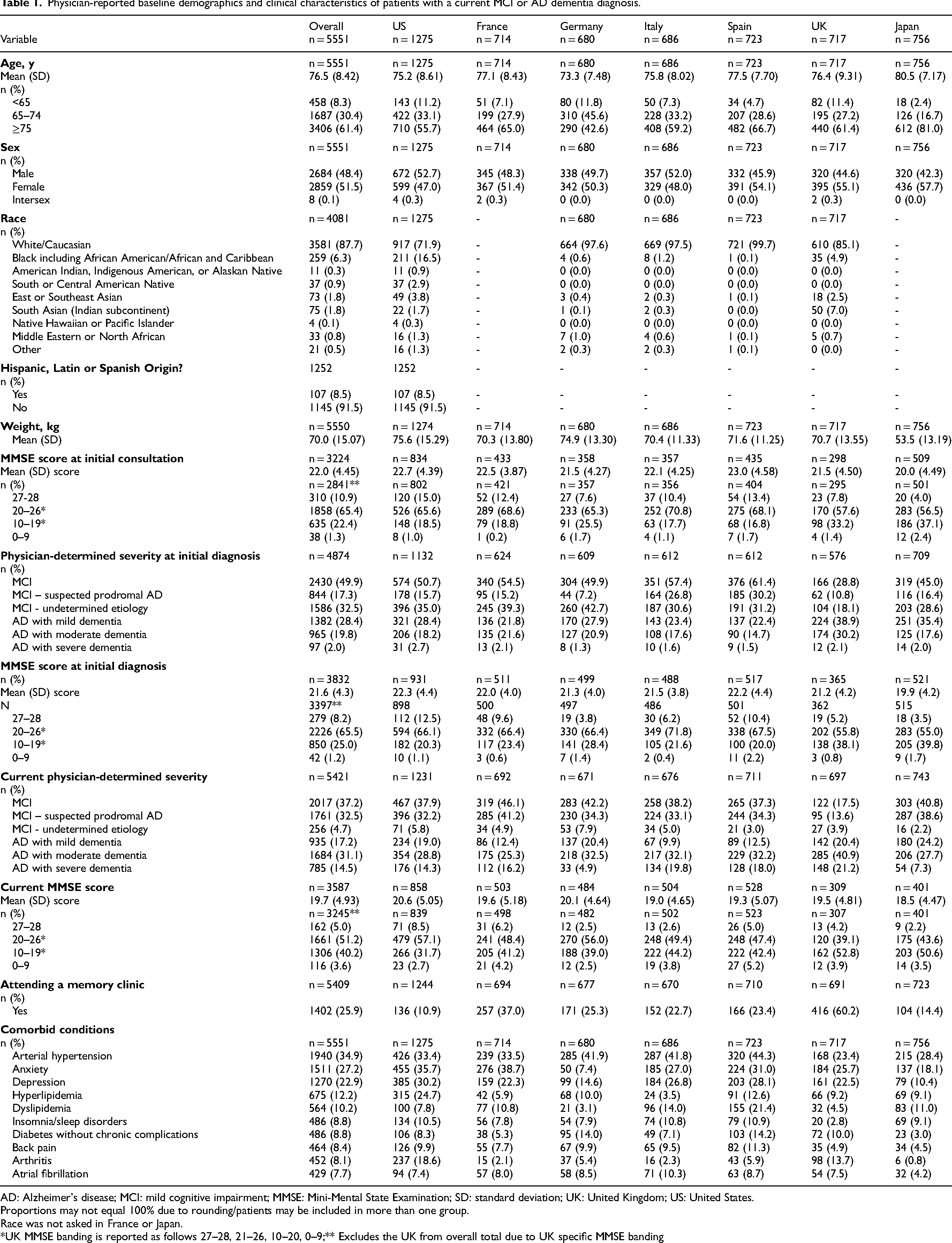
AD: Alzheimer's disease; MCI: mild cognitive impairment; MMSE: Mini-Mental State Examination; SD: standard deviation; UK: United Kingdom; US: United States.
Proportions may not equal 100% due to rounding/patients may be included in more than one group.
Race was not asked in France or Japan.
*UK MMSE banding is reported as follows 27–28, 21–26, 10–20, 0–9;** Excludes the UK from overall total due to UK specific MMSE banding
Patient mean (SD) age was 76.5 (8.42) years, 51.5% of patients were female and 87.7% of patients were White. Overall, 34.9% of patients had comorbid hypertension, 27.2% had anxiety and 22.9% had depression. The mean (SD) body weight was 70.0 kg (15.1 kg), although in Japan it was 53.5 kg (13.2 kg); Table 1. Physicians provided current disease severity data for 5421 (97.7%) patients. Physicians reported that 37.2% of patients had MCI (87.3% due to suspected AD and 12.7% undetermined cause), 17.2% had AD with mild dementia, 31.1% had AD with moderate dementia, and 14.5% had AD with severe dementia (Table 1). The mean (SD) current MMSE score was 19.7 (4.93) for patients with available MMSE current score data (n = 3587) (Table 1).
Diagnostic journey
Initial consultation
The most frequently reported reasons prompting patients to first consult their physician were short-term memory loss (81.9%), difficulties in concentration or attention (37.8%), and difficulties in recall of names or words (36.1%). The patient's partner or spouse was the most common person to first notice symptoms and prompt the initial consultation with a physician (43.5%) followed by the patient's child (27.9%). A total of 17.8% of patients were the first to notice their cognitive symptoms (Supplemental Table 2).
Physician-determined severity was not available for patients at initial consultation. Of the patients for whom an MMSE score at the time of initial consultation was reported in the survey (58.1%), the overall mean (SD) MMSE score was 22.0 (4.45), indicating the majority had AD with mild dementia. Country-specific MMSE scores are available in Table 1.
Initial diagnosis
At the time of the patient's initial MCI or AD diagnosis, behavioral and cognitive assessments were conducted for 91.3% of all patients. The most common test used was the MMSE (80.3%), followed by the Clock Draw Test only (24.8%), and Montreal Cognitive Assessment (12.4%). Data from informant reports were included in 89.0% of the diagnostic evaluations and a family history was obtained in 68.0% of the cases (Table 2).
Diagnostic testing in patients with a current MCI or AD dementia diagnosis.
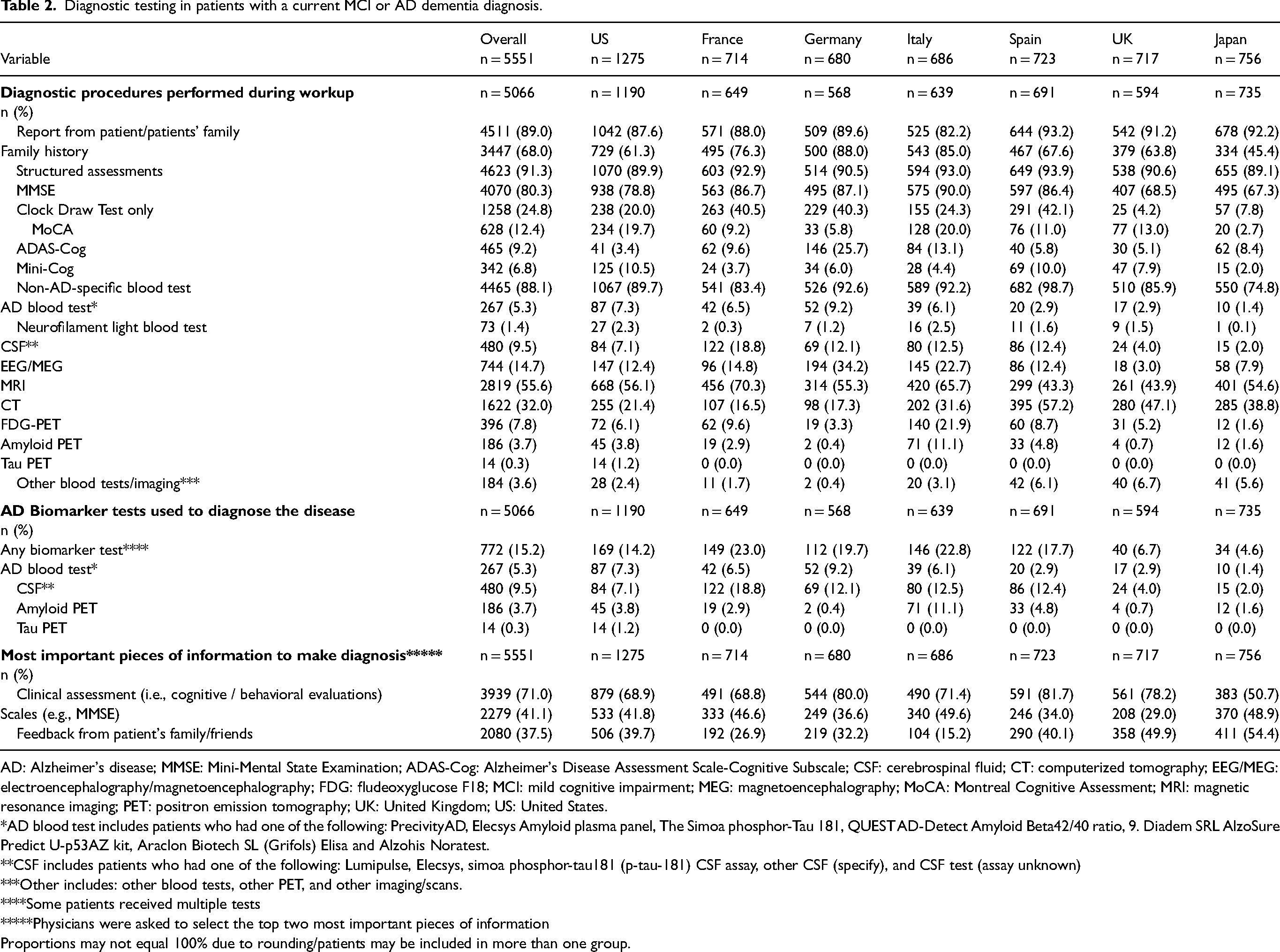
AD: Alzheimer's disease; MMSE: Mini-Mental State Examination; ADAS-Cog: Alzheimer's Disease Assessment Scale-Cognitive Subscale; CSF: cerebrospinal fluid; CT: computerized tomography; EEG/MEG: electroencephalography/magnetoencephalography; FDG: fludeoxyglucose F18; MCI: mild cognitive impairment; MEG: magnetoencephalography; MoCA: Montreal Cognitive Assessment; MRI: magnetic resonance imaging; PET: positron emission tomography; UK: United Kingdom; US: United States.
*AD blood test includes patients who had one of the following: PrecivityAD, Elecsys Amyloid plasma panel, The Simoa phosphor-Tau 181, QUEST AD-Detect Amyloid Beta42/40 ratio, 9. Diadem SRL AlzoSure Predict U-p53AZ kit, Araclon Biotech SL (Grifols) Elisa and Alzohis Noratest.
**CSF includes patients who had one of the following: Lumipulse, Elecsys, simoa phosphor-tau181 (p-tau-181) CSF assay, other CSF (specify), and CSF test (assay unknown)
***Other includes: other blood tests, other PET, and other imaging/scans.
****Some patients received multiple tests
*****Physicians were asked to select the top two most important pieces of information
Proportions may not equal 100% due to rounding/patients may be included in more than one group.
At initial diagnosis, physicians reported that 49.9% of patients had MCI, 28.4% had AD with mild dementia, 19.8% had AD with moderate dementia and 2.0% had AD with severe dementia. The overall mean (SD) MMSE score at initial diagnosis was 21.6 (4.3); in the US this was 22.3 (4.4), France 22.0 (4.0), Germany 21.3 (4.0), Italy 21.5 (3.8), Spain 22.2 (4.4), the UK 21.2 (4.2), and Japan 19.9 (4.2). Split by MMSE banding, 8.2% of patients had a score of 27–28, 65.5% were 20–26, 25.0% were 10–19 and 1.2% were 0–9. MMSE scores for the UK showed 5.2% of patients had a score of 27–28, 55.8% had a score of 21–26, 38.1% had a score of 10–20, and 0.8% had a score of 0–9. Physician-reported severity and MMSE score-based severity were aligned for 37.4% of patients (Supplemental Table 3).
Laboratory tests, such as thyroid stimulating hormone (TSH), B12, and complete blood count, were reported for 88.1% of patients at the initial diagnosis (Table 2). Few patients received at least one AD biomarker test to confirm pathology at diagnosis; 9.5% had CSF testing, 3.7% had amyloid PET, and 5.3% had AD-specific blood tests. The use of CSF in patient diagnosis ranged from 2.0% in Japan to 18.8% in France, and amyloid PET ranged from 0.4% in Germany to 11.1% in Italy. Physicians reported that the most important pieces of information used to make an initial diagnosis were cognitive and behavioral evaluations (71.0%), clinical scales (e.g., MMSE; 41.1%) and reports from informants, such as family and friends (37.5%; Table 2).
The patient pathway was split into three different routes from first consultation to initial diagnosis and is shown in Supplemental Tables 4 and 5 and Figure 2.
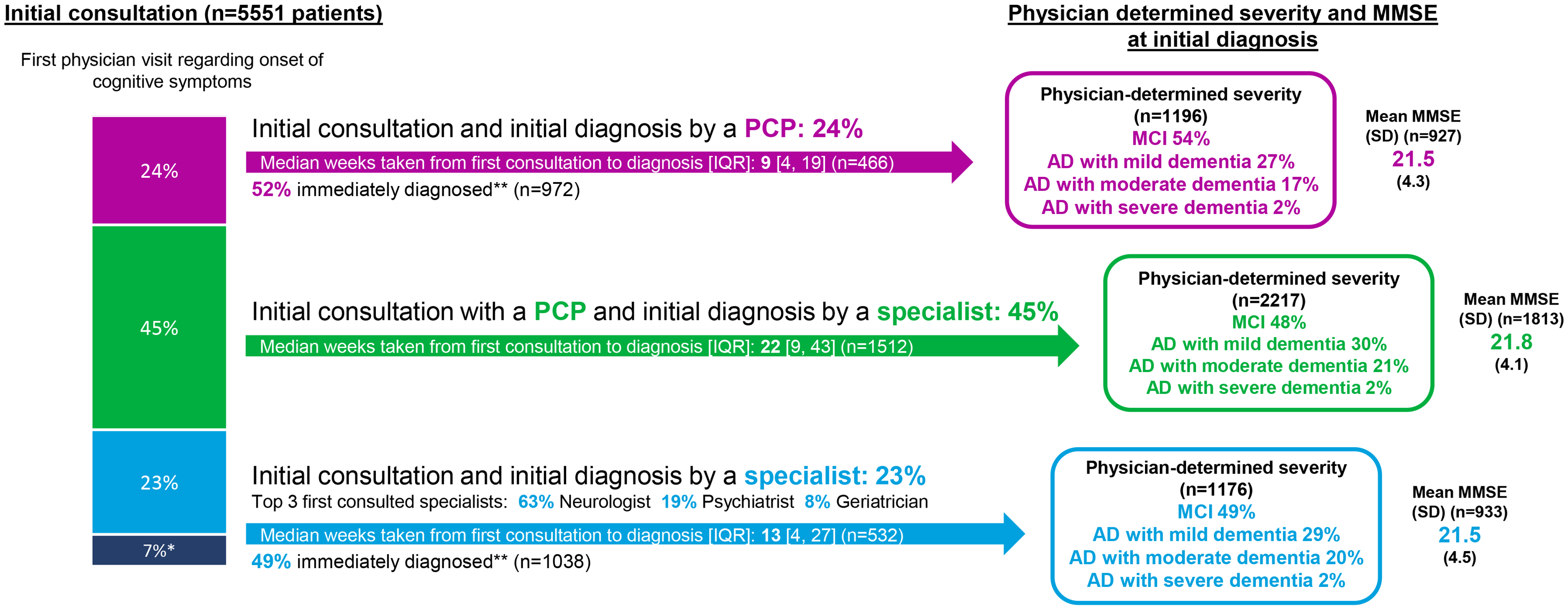
Diagnostic journey of patients with a current MCI or AD dementia diagnosis. AD: Alzheimer's disease; MCI: mild cognitive impairment; MMSE: Mini-Mental State Examination; PCP: primary care physician; SD: standard deviation; IQR: interquartile range. *‘Other’ includes alternative carers such as nurses and physician assistants and unknown pathways. **Patients immediately diagnosed defined as those whom physicians indicated were diagnosed ‘immediately’ or specified the same day as the patient's first consultation.
Patient pathway: initial consultation and initial diagnosis by a PCP
Overall, 1333 (24.0%) patients first consulted a PCP and were initially diagnosed by a PCP. This varied by country, ranging from 7.7% in Italy to 36.9% in the US (Supplemental Table 4).
The most frequently used behavioral/cognitive assessments used in the initial diagnosis among PCPs were MMSE (74.3%), Clock Draw Test only (19.8%), and Hasegawa Dementia Scale-Revised (HDS-R; 10.3%). Few patients received AD biomarker tests to confirm pathology at the time of diagnosis, with 4.0% receiving CSF testing, 1.6% receiving amyloid PET, and 3.7% receiving AD blood tests. The use of CSF in patient diagnosis ranged from 0.4% in Japan to 15.4% in Spain, and amyloid PET ranged from 0.0% in the UK to 10.4% in Italy.
Overall, 52.1% of patients diagnosed by PCPs were diagnosed at initial consultation. At initial diagnosis, PCPs determined that 53.8% of patients had MCI, 27.3% had AD with mild dementia, 16.6% had AD with moderate dementia, and 2.3% had AD with severe dementia. For patients who had a recorded MMSE score at initial diagnosis, mean (SD) MMSE score was 21.5 (4.3).
For patients who were diagnosed by a PCP, but not at initial consultation, the median [IQR] time from initial consultation to initial diagnosis was 8.9 [4.3, 18.6] weeks. This varied by country, ranging from 4.4 [4.3, 13.1] in Germany and 4.4 [3.0, 12.0] in Japan to 17.1 [6.0, 26.1] in Italy. Among these patients, for those diagnosed with MCI, median [IQR] time to initial diagnosis was 17.6 [9.4, 93.6] weeks. For those with AD with mild dementia this was 8.0 [7.0, 19.7] weeks, and for those with AD with moderate dementia this was 4.9 [2.0, 47.7] weeks.
Patient pathway: initial consultation with a PCP and initial diagnosis by a specialist
A total of 2501 (45.1%) patients first consulted a PCP and were initially diagnosed by a specialist. This ranged from 15.3% in Japan to 65.1% in the UK (Supplemental Table 5).
The diagnosing physician was typically a neurologist (67.7%–79.3%), except for the UK where 63.8% of patients were diagnosed by a psychogeriatrician, and Japan where 42.2% and 39.7% of patients were diagnosed by a neurologist and psychiatrist, respectively.
When patients first consulted a PCP and were diagnosed by a specialist, the most frequently used behavioral/cognitive assessments by either provider was the MMSE (85.5%), Clock Draw Test only (28.2%), and Montreal Cognitive Assessment (13.4%). Few patients received biomarker tests to confirm pathology, with only 11.6% of patients receiving CSF testing, 3.9% receiving amyloid PET, and 4.9% receiving AD-specific blood tests. The use of CSF in patient diagnosis ranged from 2.7% in Japan to 24.9% in France, and amyloid PET ranged from 0.0% in Germany to 7.1% in Italy.
At initial diagnosis, physicians reported that 47.5% of patients had MCI, 30.0% had AD with mild dementia, 21.1% had AD with moderate dementia, and 1.5% had AD with severe dementia. For those patients who had an available MMSE score reported at initial diagnosis, mean (SD) MMSE score was 21.8 (4.1). For patients who first consulted a PCP and had a diagnosis by a specialist, the median [IQR] time from initial consultation to initial diagnosis was 21.6 [9.4, 43.4] weeks, ranging from 8.9 [3.8, 30.6] weeks in Japan to 28.9 [17.1, 52.1] weeks in Spain. Median [IQR] time for those with MCI at initial diagnosis was 21.9 [9.8, 45.8] weeks. This was 20.8 [8.7, 40.3] weeks for those with an initial diagnosis of AD with mild dementia, 19.8 [10.6, 42.1] for AD with moderate dementia and 21.1 [9.7, 36.9] for AD with severe dementia. The time varied between initial PCP consultation to diagnosis, based on the specialist. For example, median [IQR] time to initial diagnosis was 21.8 [9.4, 46.9] weeks for neurologists, 17.4 [8.4, 39.3] for psychiatrists, 23.6 [12.9, 40.7] for geriatric psychiatrists, and 21.1 [9.4, 41.1] for geriatricians.
Patient pathway: initial consultation with a specialist and initial diagnosis by a specialist
A total of 1302 (23.5%) patients first consulted a specialist and were subsequently diagnosed by a specialist. Specialists first consulted were typically neurologists (60.4%), especially in Italy (82.6%) and Spain (78.1%). However, in the UK, first consultation was usually with a psychogeriatrician (37.5%) or a geriatrician (23.4%), and 36.8% of patients in Japan first consulted a psychiatrist (Supplemental Table 5).
Almost half (48.7%) of patients were diagnosed at initial specialist consultation. However, specialists referred 18.7% of patients to another physician, most commonly a neurologist (53.3%). At initial diagnosis, physicians reported that 48.5% of patients had MCI, 28.9% had AD with mild dementia, 20.2% had AD with moderate dementia, and 2.4% had AD with severe dementia. For patients with an MMSE score available for initial diagnosis, mean (SD) MMSE score was 21.5 (4.5).
When patients first consulted and were initially diagnosed by a specialist, the most frequently used cognitive assessments were MMSE (79.2%), Clock Draw Test only (23.8%) and HDS-R (13.9%). Few patients received biomarker tests to confirm pathology, with 10.1% receiving CSF testing, 5.3% receiving amyloid PET, and 6.6% receiving AD-specific blood tests. The use of CSF in patient diagnosis ranged from 2.6% in Japan to 20.0% in France, and amyloid PET ranged from 0.0% in the UK to 17.8% in Italy.
For patients who first consulted a specialist and had a diagnosis by a specialist, the median [IQR] time from initial consultation to initial diagnosis was 12.6 [4.4, 26.7] weeks. Median [IQR] time for those with MCI at initial diagnosis was 13.1 [5.0, 30.3] weeks. This was 13.1 [4.4, 34.7] weeks for those with an initial diagnosis of AD with mild dementia, 5.1 [1.7, 18.3] weeks for AD with moderate dementia and 10.7 [6.6, 19.5] weeks for AD with severe dementia. Delineated by diagnosing physician, median time to diagnosis was 13.1 [4.4, 31.0] weeks for neurologists, 4.4 [2.0, 11.9] weeks for psychiatrists, 25.3 [11.1, 52.1] weeks for geriatric psychiatrists, and 21.9 [11.0, 31.3] weeks for geriatricians.
Physician-reported barriers to timely diagnosis of MCI and AD
Specialists reported that the main barriers for timely detection of patients with MCI were delays in patients seeking help due to lack of awareness or stigma (reported by 59.8% of specialists), the lack of understanding about differences between normal aging and cognitive impairment caused by AD dementia (reported by 43.0% of specialists), and the variation in how patients typically first present (reported by 40.7% of specialists) (Table 3).
Specialist-reported barriers to timely AD identification and diagnosis, and the importance of biomarker assessment for identifying patients with AD in the future.
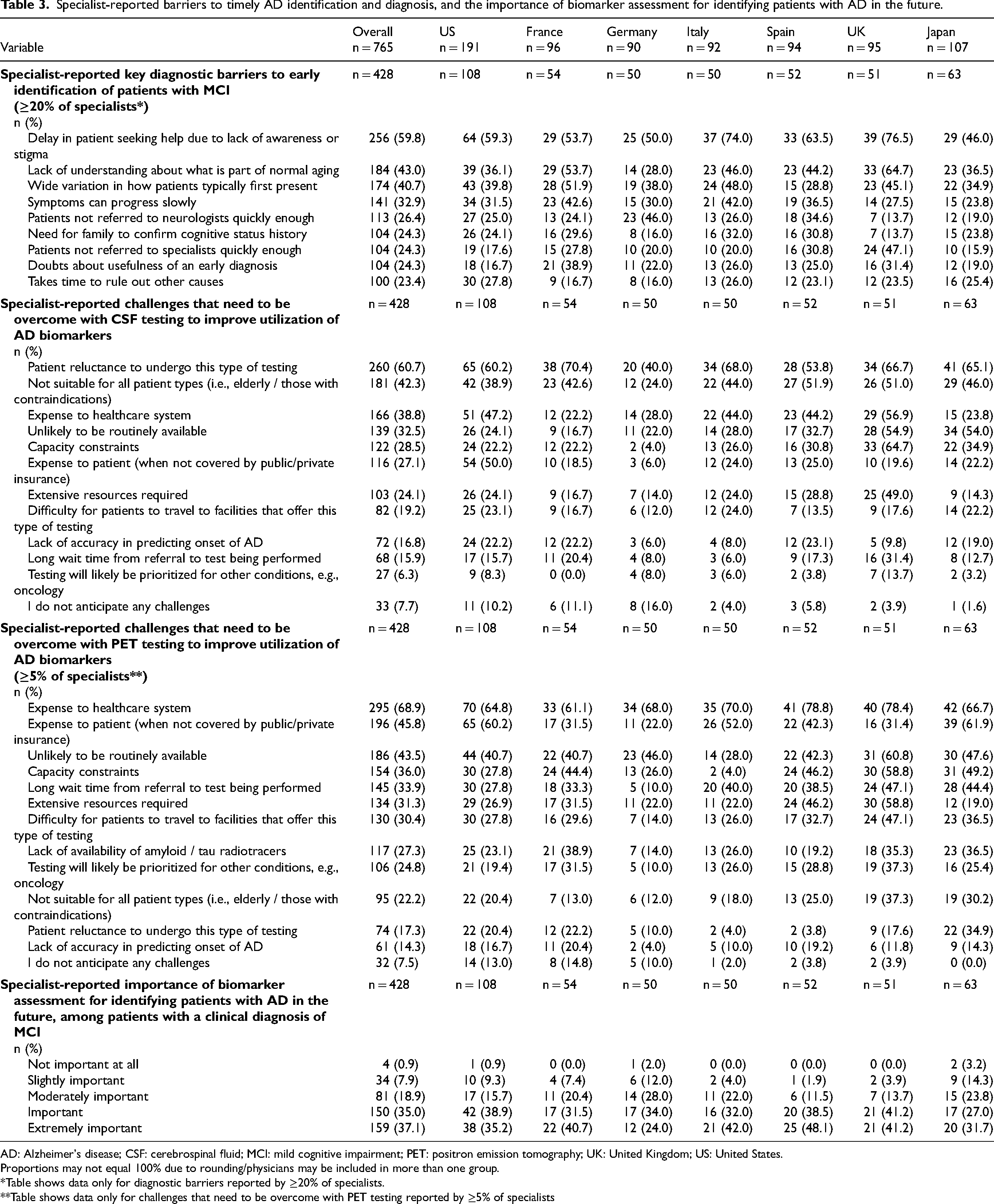
AD: Alzheimer's disease; CSF: cerebrospinal fluid; MCI: mild cognitive impairment; PET: positron emission tomography; UK: United Kingdom; US: United States.
Proportions may not equal 100% due to rounding/physicians may be included in more than one group.
*Table shows data only for diagnostic barriers reported by ≥20% of specialists.
**Table shows data only for challenges that need to be overcome with PET testing reported by ≥5% of specialists
Physician-reported delays to patients receiving a diagnosis are summarized in Table 4. According to physicians, patients raised cognitive concerns to a healthcare professional, on average, 1.5 (1.4) times before diagnostic testing was initiated. The most common reasons reported for delayed diagnosis were time delays to see a specialist (43.5% of patients), time before next consultation (31.7% of patients), and/or ‘time taken to schedule diagnostic tests (i.e., a lack of access to specialized diagnostic services’; 24.1% of patients; Table 4). In Japan, physicians also cited ‘time taken to get test results and a lack of availability of a specialized healthcare professional to interpret the results’ (17.6% of patients) and ‘difficulty for the patient to attend appointments’ (15.8% of patients) as prevalent reasons (Table 4). ‘Time taken to get test results/lack of availability of a specialized healthcare professional to interpret the results’ was also reported for 25.4% of patients in Spain.
Delays to diagnosis in patients with a current MCI or AD dementia diagnosis.
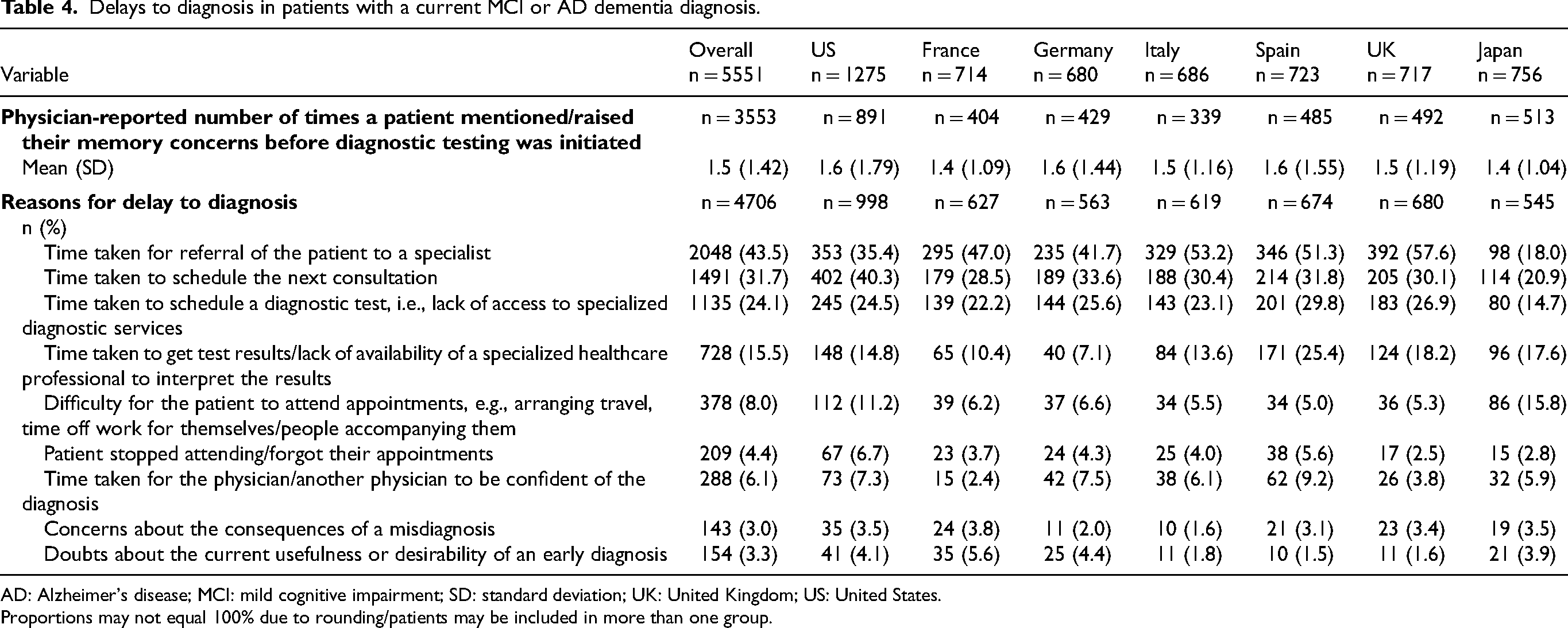
AD: Alzheimer's disease; MCI: mild cognitive impairment; SD: standard deviation; UK: United Kingdom; US: United States.
Proportions may not equal 100% due to rounding/patients may be included in more than one group.
The main specialist-reported barriers regarding the use of CSF tests for AD biomarkers were the patient's reluctance to undergo this type of testing (60.7% of specialists), not being suitable for all patients (42.3% of specialists), and the expense to healthcare system (38.8% of specialists; Table 3). Specialists also reported barriers to the use of PET in diagnostic assessments, including the expense to the healthcare system (68.9%), expense to the patient (when not covered by public/private insurance; 45.8%), that PET was unlikely to be routinely available (43.5%) and capacity constraints (36.0%), although this was low amongst specialists in Italy (4.0%; Table 3).
The importance of biomarker assessment for identifying AD in patients with a clinical diagnosis of MCI in the future was rated as important or extremely important by 72.2% of specialists, with 18.9% of specialists believing it to be moderately important (Table 3).
Patient treatment and management
Pharmacologic treatment was initiated in half (49.0%) of all patients by neurologists and in 22.4% of patients by PCPs following a diagnosis (Table 5). The median [IQR] time from diagnosis to physician-reported start of first line treatment was 2.4 [0.0, 21.9] weeks. Most patients were treated at first line with acetylcholinesterase inhibitors (79.0%), including donepezil (52.7%), rivastigmine (7.4% oral; 14.1% patch) and galantamine (5.7%). As part of first-line treatment, NMDA antagonists were prescribed to 29.4% of patients (29.2% memantine, 0.3% ifenprodil) and antidepressants to 27.8% of patients. First-line use of NMDA antagonists was particularly high in patients in the US (47.2%) and first-line use of antidepressants was high in France (45.4%).
Treatment initiation in patients with a current MCI or AD dementia diagnosis.
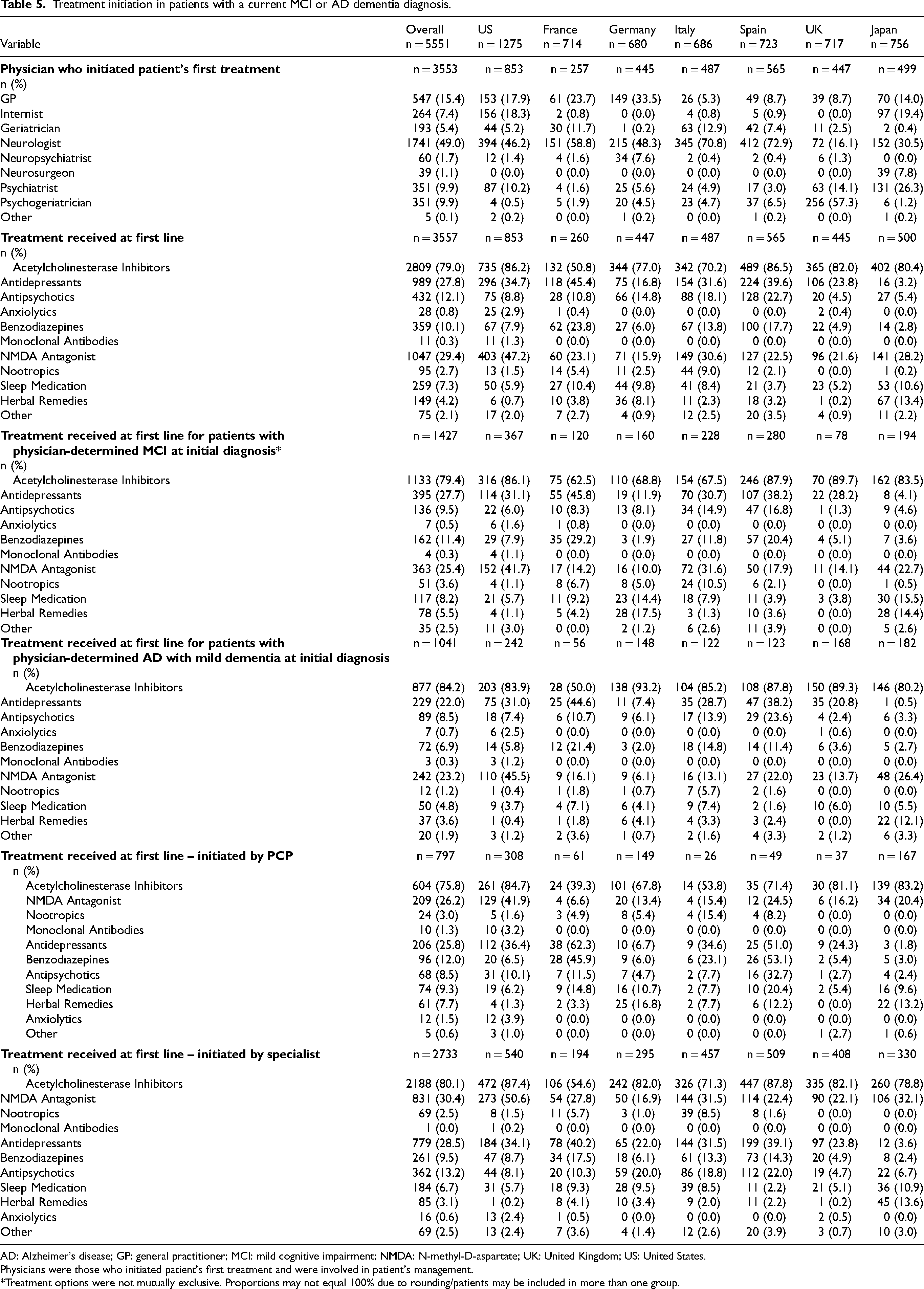
AD: Alzheimer's disease; GP: general practitioner; MCI: mild cognitive impairment; NMDA: N-methyl-D-aspartate; UK: United Kingdom; US: United States.
Physicians were those who initiated patient's first treatment and were involved in patient's management.
*Treatment options were not mutually exclusive. Proportions may not equal 100% due to rounding/patients may be included in more than one group.
Across disease severities, 79.4% of patients with MCI at initial diagnosis received acetylcholinesterase inhibitors as part of their first line treatment, this was 84.2% for AD with mild dementia, 72.6% for AD with moderate dementia and 71.7% for AD with severe dementia. For NMDA antagonists, this was 25.4%, 23.2%, 43.3% and 43.3% of patients, respectively.
Aducanumab was initiated as a first line treatment for 1.3% of all US patients included in the survey.
Subgroup analysis
A subgroup analysis was performed on the 2952 patients with a MCI or AD with mild dementia diagnosis, as this is the group most likely appropriate and eligible for ATTs.
Patient clinical and demographic characteristics for this subgroup are presented in Supplemental Table 6. Symptoms prompting initial consultation are shown in Supplemental Table 7 and the patient journey is presented in Supplemental Tables 8–10. Diagnostic tests were predominantly behavioral/cognitive assessments, informant reports, or non-AD specific blood tests. AD biomarker testing was uncommon in the diagnostic assessment (Supplemental Table 11). Patients were predominantly prescribed acetylcholinesterase inhibitors (80.9%) or anti-depressants (23.7%) as first-line treatment (Supplemental Table 12). Physician-reported delays to diagnosis in patients with a current MCI or AD with mild dementia diagnosis are reported in Supplemental Table 13.
Discussion
This analysis of real-world survey data from physicians and their patients examined the diagnostic journey from initial clinical presentation of cognitive symptoms to diagnosis and treatment for patients with MCI and AD dementia. This study was conducted using a validated survey methodology across a large, multi-national cohort of physicians in the US, Europe, and Japan. Given this large sample size, the current study provides insights from a varied and representative physician and patient population on diagnostic pathways, diagnostic assessments, and treatment and management of AD in clinical practice.
Diagnostic journey
Time from first consultation to initial diagnosis was notably long when a PCP made a referral to a specialist for a diagnosis. A shorter time to diagnosis allows for the prompt formulation of a care plan, incorporating interventions such as management of psychosocial and behavioral symptoms, lifestyle changes, better comorbidity management, and care partner support.33–36 However, the potential for delay is affected by many factors, including number of particular specialists per capita, caseload, physician experience and education, availability of diagnostic tests and country-specific variations in diagnostic guidelines.54,55
Although the need for prompt or timely diagnosis has been acknowledged, 28 precise target time frames have not been specified. Furthermore, research has previously considered the whole interval from the initial onset of symptoms to diagnosis, and to our knowledge no recent studies have quantified the time between first consultation and diagnosis or described the details of the diagnostic journey during this particular period.
Our findings regarding the short time to diagnosis when patients initially consulted and were diagnosed by a PCP demonstrates the importance of PCPs for both detection, assessment, and management of patients with MCI and AD dementia, a role which is becoming increasingly important given the widespread shortage of specialists,26,56–58 and may highlight a need to focus on collaborative dementia care between PCPs and specialists. 28 However, prior research suggests that PCPs are not always comfortable making the diagnosis of AD, perhaps due to lack of experience, adequate training, reimbursement of appropriate tests, or due to demands on their time that limit these assessments and follow-up conversations with patients and families.58,59 Previous studies have also reported that nearly 80% of PCPs lacked confidence in their ability to correctly identify a patient with a neurocognitive disorder, 59 and that 39% of PCPs were not comfortable making a diagnosis of AD dementia. 60 This aligns with the fact that 54% of physicians referred over half of patients to a specialist. 59 This adds to the delays in diagnosis and treatment when there is a secondary referral, such as in this study when 18.7% of specialists referred patients to another specialist. Future studies should examine patient characteristics such as age, ethnicity, the presence of relevant comorbidities, and functional status to determine whether some patient groups are more likely to be referred to specialists and experience a longer time to diagnosis.
Regarding disease severity, although some patients did not have an available MMSE score conducted to aid diagnosis of MCI or AD dementia, physicians determined patient severity using their own judgement and diagnostic skills, as well as information collected during the consultation. This is consistent with routine clinical practice, and therefore representative of the treatment and management landscape of MCI and AD dementia. 61 Physicians can select from a wide variety of available assessment tools; the use of MMSE in this study reflects its widespread use and does not imply superiority to other diagnostic tools such as MiniCog, Montreal Cognitive Assessment (MoCA) and the Clock Draw Test. Physician-reported severity and MMSE score-based severity were aligned for 37.4% of patients, though physicians tended to rate patients as less severe than their MMSE score would indicate and concordance was less aligned at either limit of the scale, i.e., those with either MCI or AD with severe dementia. Despite its widespread use in the clinical setting and in research, it is acknowledged that MMSE alone should not be used for diagnosis of dementia, in part due to factors such as educational and literacy levels, favored language use, or sensory disability, such as hearing or vision loss, that could affect scores.62,63 Furthermore, MCI can be heterogeneous and difficult to diagnose as it includes features which overlap with those of mild dementia. 64 Therefore, physicians may be hesitant to assign a diagnosis of the severity of AD before the condition has progressed to a stage where they are more confident in the AD designation. The discordance between MMSE and diagnosed MCI may also signify that patients frequently delay physician consultations until a more progressed stage of cognitive decline. Consequently, physicians might perceive patients with MMSE outcomes in the lower twenty-point range as being at the preliminary phase of cognitive impairment, despite the likelihood that they have been manifesting symptoms for an extended duration.
To aid diagnosis, physicians in the current study commonly used behavioral/cognitive assessments, informant reports, and laboratory blood tests such as TSH and B12. However, AD-specific biomarker testing was relatively uncommon, with amyloid PET used in only 3.7% of cases (range 0.4–11.1%), and CSF biomarker testing used in 9.5% of cases (2.0–18.8%). The use of biomarker confirmation of AD diagnosis in clinical practice is expected to evolve and increase over coming years, due at least in part to the emergence of ATTs and their availability in more countries. 65 The under-utilization of AD biomarker tests observed in the current study could be due to the invasiveness of obtaining CSF, the access and cost for amyloid PET, 66 the lack of data on how AD blood biomarkers perform in real world clinical populations, 67 and the perception among some physicians that the results will have a limited impact on treatment decisions. 66
The predominant use of structured cognitive assessments is unsurprising given the historical reliance on clinical assessment to identify AD dementia.12–16 However, supporting those tests with biomarker identification is integral to pathologically confirm an AD dementia diagnosis. Biomarker testing, especially low cost, noninvasive testing such as blood biomarkers, may be especially important in the earlier stages of disease.68–72 Indeed, the current study reported that most physicians deem biomarker testing to be important in future AD dementia diagnoses. However, further data are required on the generalizability of diagnostic biomarker research to wider clinical practice before this becomes routine. 68
Challenges of timely diagnosis
Many patients in the current study were diagnosed at a stage beyond MCI, reflected by a mean MMSE score of 22.0 at initial consultation and a score of 21.6 at initial diagnosis. This indicates a crucial need for reducing the time to diagnosis and increasing rates of timely detection. Yet, physicians face challenges to making a timely diagnosis, including delays to initial consultation, the variation in initial symptom presentation by patients, resource and capacity constraints, time pressures on routine appointments, and healthcare financing policies.54,73 In the current study, over half of specialists reported that patients delayed seeking help due to lack of awareness and/or stigma, and just under half reported that a key barrier is a lack of understanding about what is part of normal aging. There is a need to address the negative impacts of AD stigma, and interventions to tackle this at the individual, societal, and institutional level have been proposed including interactive contact-based approaches, targeted education, and person-centered care. 74 Variation in symptoms and lack of routinely available testing, the availability of HCPs to interpret results or simply patients’ reluctance to undergo diagnostic tests were also reported as key barriers to timely diagnosis. Taken collectively, these results show that significant barriers exist to timely diagnosis and treatment of patients with MCI and AD with mild dementia.
Importance of timely diagnosis
In the context of continuous disease progression, a delay to diagnosis may limit a timely intervention; it is in the earliest stages of symptomatic AD that treatments such as ATTs may be considered, 50 as reflected in the FDA approvals75,76 and the approvals in the UK,77,78 Japan 79 and the United Arab Emirates80,81 of donanemab and lecanemab for MCI or AD with mild dementia as well as the approval of donanemab in Qatar and of lecanemab in China, 82 South Korea, 83 Hong Kong, 84 Israel, 85 and Europe (initial approval by the Committee for Medicinal Products for Human Use with anticipation of full approval). 86 Indeed, in 2021 it was estimated that 1800 out of 2 million Americans with mild dementia due to AD and 2300 out of 2.5 million Europeans with AD with mild dementia would progress to moderate dementia each day. 87 Resolving barriers to consultation and delays to diagnosis is, therefore, necessary to ensure patients can benefit from interventions that slow disease progression.
Due to recent research utilizing biomarker-related endpoints in clinical trials for efficacy and safety of ATTs, clinical-neuropathological diagnosis of AD is expected to increase.42,67,68,88,89 Currently, however, this study demonstrated that real world use of biomarkers remains low. Barriers to the implementation of this approach to diagnosis should be identified, as well as ways in which they could be overcome.
The results reported here describe the patient journey and quantify the time to diagnosis across different clinical scenarios. Future research could investigate this further, to determine whether the differences observed have clinical significance and to assess the potential impact of delayed versus prompt diagnosis in this population.
Strengths and limitations
DSPs provide a crucial source of real-world data using consistent and validated methodology across countries and disease areas. Compounded by their depth and detail, DSPs generate real-world evidence that adds a unique perspective to current traditional research and influences future clinical practice. This study analyzed data from a large, robust cohort of both physicians and patients globally, and as such, results can be considered generalizable to the consulting MCI or AD dementia population, as well as the clinical landscape and current practices of treating physicians. The sample does likely under report minorities as they are less likely to engage with the healthcare system and have insurance coverage.90,91 In the US, Black individuals made up 16.5% of the sample and 8% were Hispanic in this study. This is broadly consistent with the characteristics of the general US Medicare population, 92 including those newly diagnosed with or treated for AD. 90 In the UK, 85.1% of individuals were White, 4.9% were Black, 7.0% were South Asian and 2.5% were East or Southeast Asian. This is consistent with the UK population 93 and indicates slightly higher representation of ethnic minorities compared to the diagnosed AD population. 94 Thus, the DSP may broadly represent the racial and ethnic diversity of the broader consulting population with MCI or AD in these countries.
The current descriptive findings should be considered with regards to limitations of the survey. The cross-sectional design of the DSP prevented any conclusions about causal relationships. Recall bias, a common limitation of surveys, might also have affected responses of physicians. However, physicians had the ability to refer to the patients’ records while completing the survey, thus minimizing the possibility of recall bias. Given that missing data was not imputed, data for variables with a low base size should be interpreted with caution. The DSP is based on a pseudo-random, convenience sample of physicians; while minimal inclusion criteria governed the selection of physicians, participation was influenced by their willingness to complete the survey. The sample does not necessarily represent the proportion of specialists diagnosing or treating patients with AD within each country.
Furthermore, it is expected that system-level differences between countries and regions will have an impact on some of the findings of the current study. For example, involvement of different physician types at different stages of the diagnostic process may be influenced by their relative availability in that region, or by the standard physician training pathways in a particular country. In the case of AD-specific biomarker testing, factors such as socioeconomic inequality, variable access to biomarkers, or challenges associated with a mixed public/private health care delivery system (as is the case in Japan 95 ) may play a role. 55 There is, however, the potential for countries to instigate measures that improve the diagnostic process, for example by increasing availability of and access to biomarker testing (including at the primary care level), facilitating the tailoring of diagnostic strategy to individual patients, refining the referral system, and disseminating education about dementia. Examples include the Dutch ‘ABOARD’ project, a national collaboration between public and private sectors, and the ‘US National Plan to Address Alzheimer's Disease’.96,97
Diagnosis of MCI and AD dementia was determined by the responding physician's observations and, as such, relied on the physicians’ ability to recognize the early cognitive symptoms of AD dementia and distinguish them from normal aging. Similarly, patient severity was subjectively defined by physicians, with no uniform criteria implemented, potentially resulting in variation between physicians and countries, with over- or under-estimation dependent on physicians’ opinions. Furthermore, when surveyed physicians were asked to select the assessments used to aid diagnosis, clinical assessments of behavioral and psychological symptoms (such as the Neuropsychiatric Inventory) were not included as possible options. Therefore, the use of such assessments may be under-reported. Future surveys could consider collecting the results of other assessments such as the Clock Draw Test (used in only 24.8% of patients in our study, most commonly in Spain, France and Germany) or the MoCA (used in only 12.4% of patients in our study, most commonly in Italy and the US). Results of such assessments could also be used to stratify patients by clinical severity, as a preferable alternative to the MMSE bandings that were used in this study (where these data were available). It should be noted, however, that there is no single assessment universally utilized by all physicians. As discussed above (in the ‘Discussion: Diagnostic journey’ section), this accurately reflects the complexity of diagnosis and severity rating that exists in clinical practice, and the ongoing debate around how best to determine the boundary between MCI and AD with mild dementia. The assessment selected may have known limitations, such as a ceiling effect or the patient's educational attainment, language and culture affecting the result, but this must be balanced against factors such as access to the resources and/or specialist knowledge required to administer and interpret this assessment or any alternatives. 64
Finally, the survey focused on initiation of pharmacological treatment and not on other evidence-based treatments such as counselling and referrals to community resources. Participating patients may not reflect the general AD dementia population since the DSP only includes patients who are actively consulting with their physician. Patients with less severe AD dementia, or those at the start of their disease journey, are more likely to consult more frequently with physicians and, therefore, have a higher likelihood of being included. Additionally, the study population did not include patients who consulted a physician about cognitive symptoms but were subsequently determined to have a different diagnosis to MCI/AD.
Conclusion
The results of this large, real-world study elucidate the complex journey from clinical presentation to treatment made by patients in the US, Europe and Japan, whilst also highlighting barriers to timely diagnosis of MCI and AD dementia.
The care gaps identified from the data are significant and multifaceted. Firstly, specialists reported that barriers to timely diagnosis resulted mostly from patients delaying seeking help due to lack of awareness or stigma. Improved recognition of the early indicators of cognitive impairment among older people and their families is therefore required, and stigma must be addressed at an individual, societal, and institutional level. Secondly, diagnosis delays after initial consultation were predominantly due to time taken for patient referrals to specialists as well as scheduling of diagnostic testing. Enhanced training and support for PCPs and specialists, as well as education on the importance and the application of biomarkers, can equip them with the knowledge to recognize early MCI/AD symptoms. This would facilitate timely diagnosis, which is vital for effective disease management. Finally, the study demonstrated under-utilization of biomarkers. It is important to encourage improved access to and increased availability of biomarker testing as well as the identification of patients for whom AD-specific biomarker testing may be most appropriate. This would allow the initiation of innovative treatments at a time when they can be more effective.
Overall, tackling these care gaps will not only enhance patient care but also might contribute to the broader goals of scientific progress in the field of AD. Implementation of these changes within healthcare systems is imperative to improve the diagnostic processes and enable the timely implementation of disease-modifying treatments, ultimately improving the outcomes for patients and their care partners.
Supplemental Material
sj-docx-1-alz-10.1177_13872877251322978 - Supplemental material for Diagnostic journey and management of patients with mild cognitive impairment and Alzheimer's disease dementia: A multinational, real-world survey
Supplemental material, sj-docx-1-alz-10.1177_13872877251322978 for Diagnostic journey and management of patients with mild cognitive impairment and Alzheimer's disease dementia: A multinational, real-world survey by Simona Z Vasileva-Metodiev, Drew Spargo, Eric G Klein, Frances-Catherine Quevenco, Sarah Cotton, Pascual Sanchez-Juan, Yoshiki Niimi and Nicole R. Fowler in Journal of Alzheimer's Disease
Footnotes
Acknowledgments
This work was presented first as a poster at: AMCP Nexus 2023; October 16–19, 2023; Orlando, FL, USA: Vasileva SZ, Wheeler A, Cotton S, Klein EG. The diagnostic journey of patients with mild cognitive impairment or Alzheimer's disease dementia, and challenges associated with timely diagnosis: results from a real-world survey in the United States; ISPOR Europe 2023; November 12–13, 2023; Copenhagen, Denmark. Abstract 130137: Simona Z, Vasileva-Metodiev SZ, Quevenco F-C, Sanchez-Juan P, Chloe Walker, Klein EG. The diagnostic journey of patients with mild cognitive impairment and Alzheimer's disease dementia, and the importance of biomarker testing for timely diagnosis: A real-world survey in Europe; and at the Japanese Society for Dementia Research 2023; November 24–26, 2023; Nara, Japan: Kaname Ueda, Taka Matsumura, Sarah Cotton, Chloe Walker, Eric G. Klein, Simona Z. Vasileva. Physician-reported detection and challenges of timely diagnosis of MCI and AD dementia in Japan.
ORCID iDs
Ethical considerations
This study was granted ethical exemption by the Pearl Institutional Review Board, approval number [IRB number: #22-ADRW-172/3]. A survey number was assigned to all participating physicians and patients to enable anonymous data collection and data linkage during data collection and analysis, enabling physician-patient data matching. Using a checkbox, patients provided informed consent to take part in the survey. Data were collected in such a way that patients and physicians could not be identified directly. Data were aggregated before being shared with the subscriber and/or for publication. Data collection was undertaken in line with European Pharmaceutical Marketing Research Association guidelines 51 and as such did not require ethics committee approval. Each survey was performed in full accordance with relevant legislation at the time of data collection, including the US Health Insurance Portability and Accountability Act 1996 52 and Health Information Technology for Economic and Clinical Health Act legislation. 53 Fieldwork partners adhered to the national data collection regulations in the USA, France, Germany, Italy, Spain, the UK, and Japan.
Consent to participate
Patients provided informed consent to take part in the survey.
Consent for publication
Not applicable
Author contributions
Simona Z Vasileva-Metodiev (Conceptualization; Methodology; Supervision; Writing – original draft; Writing – review & editing); Drew Spargo (Conceptualization; Writing – original draft; Writing – review & editing); Eric G Klein (Conceptualization; Writing – review & editing); Frances-Catherine Quevenco (Conceptualization; Writing – review & editing); Sarah Cotton (Conceptualization; Data curation; Methodology; Writing – original draft; Writing – review & editing); Pascual Sanchez-Juan (Writing – review & editing); Yoshiki Niimi (Writing – review & editing); Nicole R. Fowler (Writing – review & editing).
Funding
Data collection was undertaken by Adelphi Real World as part of an independent survey, entitled the Adelphi Real World AD DSP. The DSP is a wholly owned Adelphi Real World product. Eli Lilly & Company is one of multiple subscribers to the DSP. Publication of survey results was not contingent on the subscriber’s approval or censorship of the publication. No funding or sponsorship was received for this study or publication of this article.
Declaration of conflicting interests
Simona Z Vasileva-Metodiev, Drew Spargo, Eric G Klein, and Frances-Catherine Quevenco are employees and shareholders of Eli Lilly & Company. Dr Pascual Sanchez-Juan is currently employed by Research Foundation for Neurological Diseases Fundacion CIEN and has received payments from Lilly for consulting during the conduct of the study. Sarah Cotton is an employee of Adelphi Real World.
Medical writing support under the guidance of the authors was provided by Sue Libretto, PhD, of Sue Libretto Publications Consultant Ltd, and Bethan Hahn, of Bethan Hahn Communications Ltd, on behalf of Adelphi Real World in accordance with Good Publication Practice (GPP3) guidelines. 98
Editorial support was provided by Rebecca Charlton, an employee of Adelphi Real World.
Data availability
All data, i.e., methodology, materials, data, and data analysis, that support the findings of this survey are the intellectual property of Adelphi Real World. All requests for access should be addressed directly to Sarah Cotton at sarah.cotton@adelphigroup.com. Sarah Cotton is an employee of Adelphi Real World.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
