Abstract
Background
Use of claims data allows to analyze health service characteristics of dementia, which is one of the most frequent cognitive disorders in Germany and worldwide.
Objective
The study aimed at describing the variability in dementia diagnoses and in antidementia drug prescription pattern.
Methods
We analyzed data from a population-based sample of one of the largest German statutory health insurances. The cohort included 30,403 patients with incident dementia diagnosis from 2006–2016. We described frequencies, patterns, and interrelations of diagnoses (Alzheimer's disease (AD), vascular dementia, other specific dementia, unspecified dementia (UD), antidementia drugs (ADD), and professional groups. We described switches in diagnostic and medication patterns between index quarter and following quarters, and evaluated the prescriptions in relation to national guidelines.
Results
A total of 87% of patients received a diagnosis of UD in at least one quarter of insurance. In the quarter of incident diagnosis, 14% of patients received more than one diagnostic code of dementia, whereas over the course of observation, the majority of patients received more than one diagnostic code (61%). Most patients were diagnosed by a general practitioner without involving a specialist. All professional groups primarily made UD diagnoses except specialists who mainly diagnosed AD. Thirty-five percent of all patients and 67% of AD patients were prescribed an ADD at least once.
Conclusions
Specialists made the most specific diagnoses and prescribed most ADDs. A specialist consultation may be advisable, but only 34% of patients visited one. Many AD patients might be left untreated due to underdiagnosis or -treatment.
Introduction
The number of patients with dementia living in Germany is expected to rise from 1.8 million today 1 to 2.7 to 3 millions by 2050.2,3 There is no cure for dementia, but treatment with antidementia drugs (ADDs) may delay its progression.4,5
Health claims data has been used to study dementia in a large number of patients. Previous studies from Germany showed that health claims data contains multiple diagnostic codes of dementia for one patient in a quarter to a third of patients within one insurance year, and the diagnoses are often unspecified.4,6 This ambiguous information poses a special challenge for determining the diagnostic code of dementia compared to other analyses which have only one diagnosis per patient.4,5,7–9
To our knowledge, the present study is the first study with German claims data that investigates dementia diagnoses, prescriptions of ADDs, and physician groups involved in diagnostics and therapy longitudinally over a ten-year observation period. It is based on a large data sample from one of the largest German statutory health insurances and is one of a few health claims analyses which take all dementia diagnoses of a patient into account. It includes data from up to 11 uninterrupted years of insurance, which allows characterizing switches in diagnoses and medication pattern between index quarter and the following quarters. Previous studies looked only at patients treated with ADD, 10 which did not allow to show how many dementia patients received treatment. Other studies analyzed diagnoses from the outpatient sector6,10,11 and the ADD groups cholinesterase inhibitors and memantine.5,7,12–15 This study additionally takes inpatient diagnoses into account as well as the whole spectrum of anti-dementia medication according to the Anatomical Therapeutic Chemical Classification System. We investigated the variability in dementia diagnosis, i.e., which codes and how many different diagnostic codes a patient received and whether the diagnosis changed compared to the incident one. Furthermore, we explored how many patients received an ADD and of what kind, whether the prescriptions were in line with the applicable guidelines, and whether there was a change in medication after the initial prescription. We investigated if diagnostic and prescription patterns were related to the type of professional group. On the methodological side, this study also illustrates both the challenges researchers face when utilizing secondary health claims data and the possibilities which such real-world data sources offer.
Methods
Study population
This study is based on the analysis of a random longitudinal 5% sample (2004–2016) of people 50 years (in 2004) and above of one of the largest German statutory health insurances, Allgemeine Ortskrankenkasse (AOK), which covers about one third of Germany's population aged 50 years and above. A prior comparison of the 5% sample in 2004 limited to persons born before 1945 (in our study: before 1946) to the German population in 2004 showed a comparable gender and age distribution. 16 Details on the sample and the German health system have been published previously. 17 Inclusion criteria were: minimum age of 58 at the beginning of 2004; continuous insurance from 2004 until 2016, death, or withdrawal from the AOK insurance, whichever occurred first; minimum of 7 quarters survival after first quarter with dementia diagnosis; minimum of 75% of quarters in observation period with a “valid” dementia diagnosis, i.e., a verified diagnosis from the outpatient sector or inpatient diagnosis flagged as discharge or secondary diagnosis to exclude false negative diagnoses. Excluded were patients with a diagnosis in the first 8 quarters of the study (2004, 2005) in order to ensure incident dementia diagnoses, rendering a minimum age at first dementia diagnosis of 60 years.
A subgroup analysis was carried out with patients surviving for a minimum of 15 quarters after the quarter of initial dementia diagnosis. The subgroup analysis was carried out to determine if patients who survive longer have a different diagnostic and medication experience.
Anonymized data only without direct involvement of human subjects was used in this study, so no informed consent was possible or necessary.
Variables and analyses
All analyses were descriptive and involved the three key variables diagnosis, professional group, and ADD defined in Table 1. The three variables were described in relation to each other. Comparisons are based on the magnitude of differences without any statistical testing. Analyses were carried out both on patient level and diagnosis or prescription level.
Variables included in descriptive health claims data analysis of dementia. a
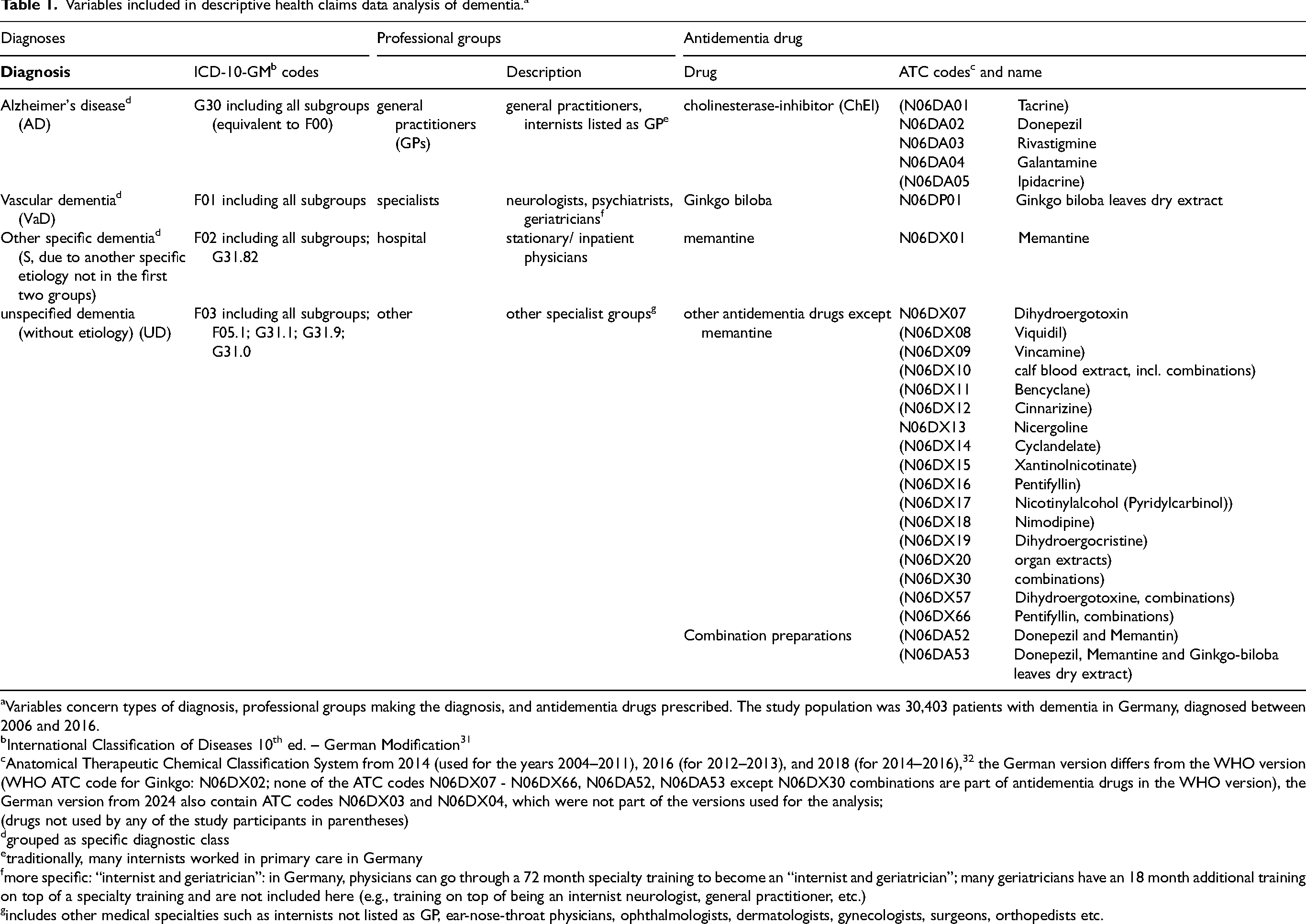
Variables concern types of diagnosis, professional groups making the diagnosis, and antidementia drugs prescribed. The study population was 30,403 patients with dementia in Germany, diagnosed between 2006 and 2016.
International Classification of Diseases 10th ed. – German Modification 31
Anatomical Therapeutic Chemical Classification System from 2014 (used for the years 2004–2011), 2016 (for 2012–2013), and 2018 (for 2014–2016), 32 the German version differs from the WHO version (WHO ATC code for Ginkgo: N06DX02; none of the ATC codes N06DX07 - N06DX66, N06DA52, N06DA53 except N06DX30 combinations are part of antidementia drugs in the WHO version), the German version from 2024 also contain ATC codes N06DX03 and N06DX04, which were not part of the versions used for the analysis;
(drugs not used by any of the study participants in parentheses)
grouped as specific diagnostic class
traditionally, many internists worked in primary care in Germany
more specific: “internist and geriatrician”: in Germany, physicians can go through a 72 month specialty training to become an “internist and geriatrician”; many geriatricians have an 18 month additional training on top of a specialty training and are not included here (e.g., training on top of being an internist neurologist, general practitioner, etc.)
includes other medical specialties such as internists not listed as GP, ear-nose-throat physicians, ophthalmologists, dermatologists, gynecologists, surgeons, orthopedists etc.
The quarter with the initial dementia diagnosis in a patient is called index quarter. Medication data was only available for filled prescriptions.
A patient may have received more than one diagnostic code of dementia and a prescription of more than one type of ADDs. For the purpose of investigating switches compared to the initial diagnosis or drug use, the diagnosis reported at most doctors’ visits was called the main diagnosis for that patient and the most frequent ADD prescribed was defined as the main drug. If two diagnoses were reported with the same frequency, both diagnoses were simultaneously reported (double diagnosis); an analogous method was applied to ADDs (double medications).
In Germany, the S3 guidelines “Demenzen” 18 give recommendations for the diagnostics and treatment of dementia. Alzheimer's disease (AD) is the only diagnostic code of dementia with recommended ADD treatment, namely with cholinesterase-inhibitors (ChEIs) and memantine. For vascular dementia (VaD) as well as two codes of other specific dementias (Morbus Parkinson, F02.3, and Lewy-body dementia, G31.82), an off-label use of ChEIs and/or memantine may be considered. Unspecified dementia (UD) was not explicitly described in the guidelines. Due to its unknown etiology, we refer to UD as a diagnostic code rather than a type of dementia; a type infers a known etiology like with AD, VaD, or other specific dementia.
All analyses were carried out using SAS version 9.4.
Results
Baseline characteristics
The 5% sample contained data of 591,815 patients aged 50 years or older in 2004 and 471,462 patients aged 58 years or older. Of these, 95,783 had a “valid” dementia diagnosis in 2006 with no prior diagnosis in 2004 or 2005; 30,403 fulfilled all inclusion criteria. The majority of these patients were women (68.1%). Mean age at incident diagnosis was 80.5 years. Almost all patients had comorbidities and a prescription of medication other than ADDs (Table 2). Dementia patients were observed for an average of 17.3 quarters, i.e., for almost 4.5 years, and up to 11 years.
Baseline characteristics of a health claims data analysis of 30,403 patients with dementia in Germany, diagnosed between 2006 and 2016.
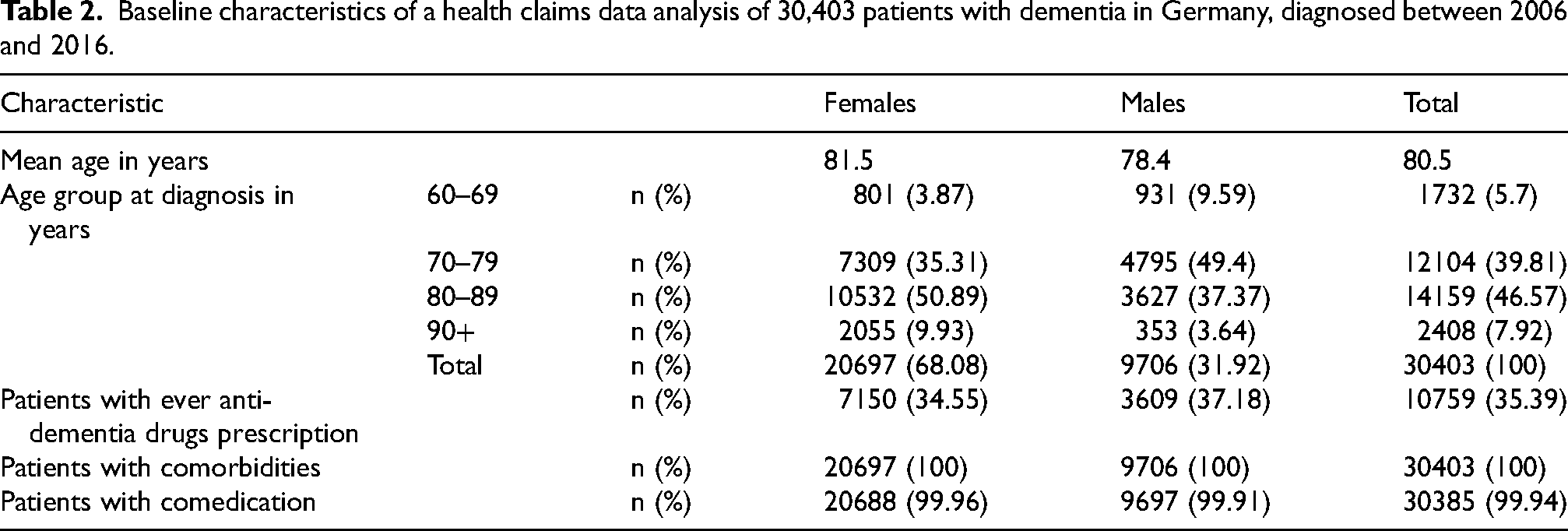
Only half the patients from the main analysis fulfilled the 4-year-survival criterion of the subgroup analysis (n = 15,199). The percentage of women was higher (70.2%) and mean age of 79.3 years was slightly lower.
Type of diagnostic code of dementia
The majority of patients had at least one UD diagnosis (87.1%) and almost half of patients had at least one diagnosis of AD and VaD each (44.0% and 48.1%, respectively, Supplemental Table 1). The main diagnosis was UD in the majority of patients (56.4%), followed by AD (22.9%), and VaD (17.9%) (Table 3). Only 1.6% had a double diagnosis as main diagnosis.
Frequency of diagnoses, by professional group.a
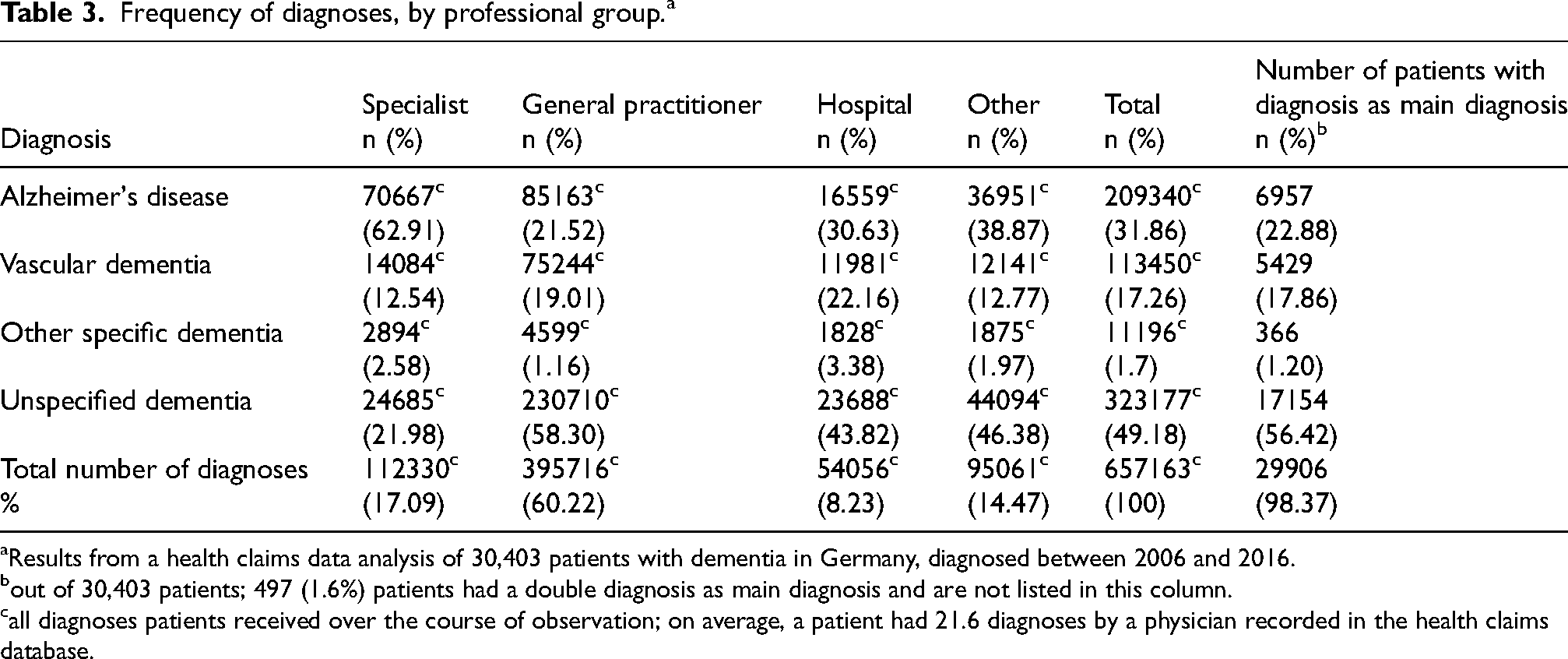
Results from a health claims data analysis of 30,403 patients with dementia in Germany, diagnosed between 2006 and 2016.
out of 30,403 patients; 497 (1.6%) patients had a double diagnosis as main diagnosis and are not listed in this column.
all diagnoses patients received over the course of observation; on average, a patient had 21.6 diagnoses by a physician recorded in the health claims database.
General practitioners (GPs) made the majority of diagnoses (60.2%). The most common diagnosis by specialists was AD (62.9%, Table 3). The most common diagnosis by the other professional groups was UD (Table 3).
Number of distinct diagnoses in a patient
Most patients received two, three, or even four diagnostic codes of dementia over the course of observation (61.3%, see “Total” in Table 1, Supplemental Table 2), and most patients (71.6%, subgroup analysis: 75.8%) visited more than one professional group (Supplemental Table 3). In the index quarter, 86.1% of patients had only one distinct diagnosis, and 81.2% visited only one professional group (Supplemental Tables 2 and 3).
Almost all patients (93.7%) had at least one diagnosis by a general practitioner, and 25.7% had diagnoses exclusively from one; 34.1% had at least one diagnosis from a specialist, 55.0% from a hospital physician (Supplemental Table 4). Nevertheless, only 8.2% of all diagnoses were made by a hospital physician (Table 3). Almost all patients with a specialist diagnosis also had a diagnosis by a general practitioner (30.2%, n = 9173) (data not shown). Three quarters of specialists and two thirds of GPs gave only one diagnostic code to a patient, whereas only 56.7% of hospital physicians stuck to one code. Only 38.7% of patients got only one diagnostic code over the course of observation across all physicians they saw (Figure 1, Supplemental Table 2).
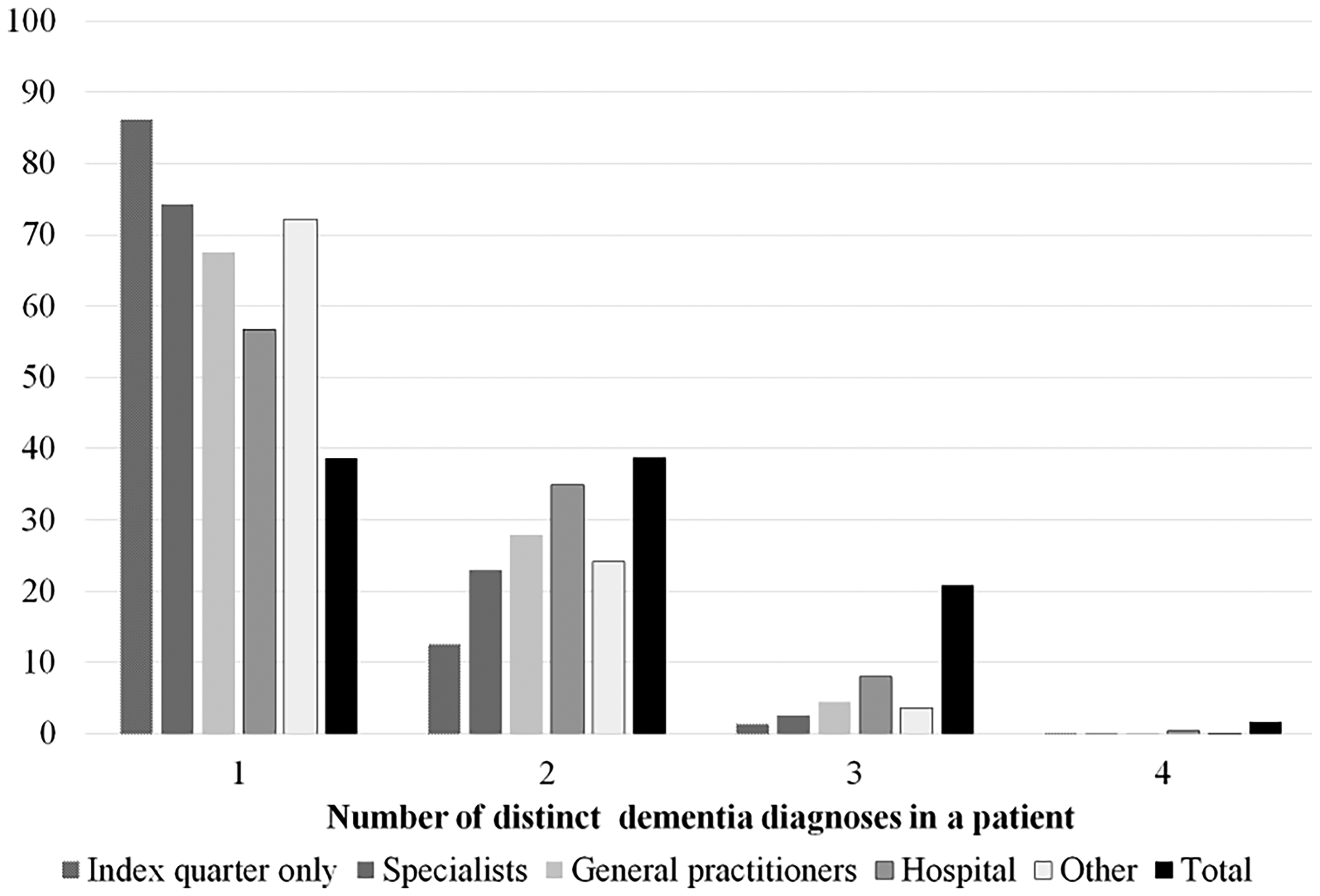
Frequency of the number of distinct dementia diagnoses in a patient out of the four diagnoses Alzheimer's disease, vascular dementia, specific dementia, UD. Stratified by: all diagnoses a patients received, diagnoses from the index quarter only, and diagnoses within each professional group. Health claims data analysis of 30,403 patients with dementia in Germany, diagnosed between 2006 and 2016.
Switches in diagnosis
In the following, we compared diagnoses in the index quarter to all other diagnoses a patient received. In the index quarter, much fewer patients had more than one diagnostic code of dementia (13.9% versus 61.3%, Supplemental Table 2), and much fewer visited more than one professional group (18.8% versus 71.6, Supplemental Table 3). Moreover, fewer patients had a diagnosis of AD and VaD (37.1% and 27.2%, respectively, Supplemental Table 1).
The initial diagnosis was identical to the main diagnosis in 70.5% of patients (Figure 2, subgroup analysis: 67.9%). For an additional 12.4%, the main diagnosis was already one of several diagnoses in the index quarter (data not shown). The index diagnosis was completely different from the main diagnosis in 17.2% (n = 5137) of patients (subgroup analysis: 19.4%).
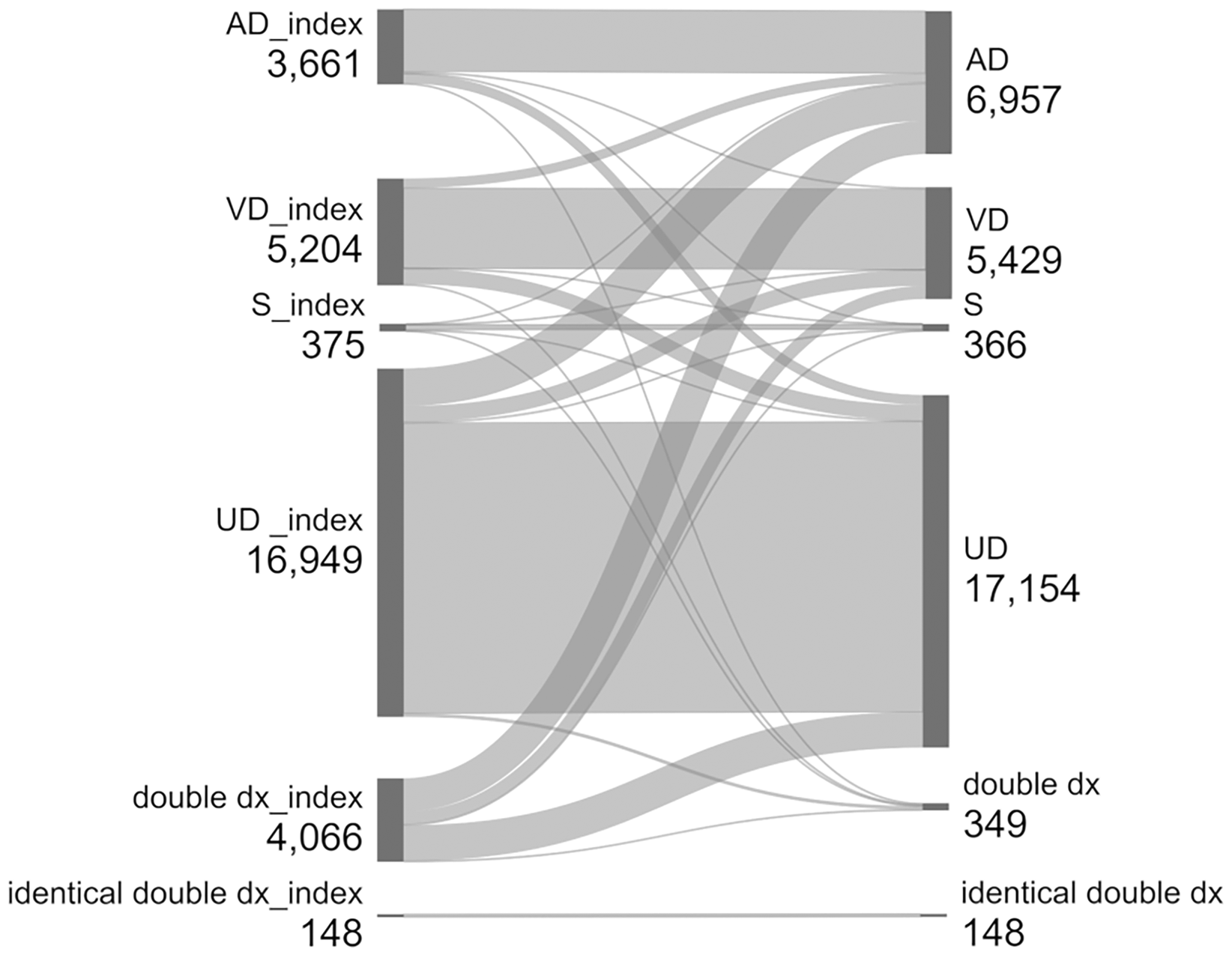
Flow between number of patients with incidence diagnosis in index quarter (left) and number of patients with main diagnosis, i.e., most frequent diagnosis in study period (right). Health claims data analysis of 30,403 patients with dementia in Germany, diagnosed between 2006 and 2016. AD: Alzheimer's disease; VD: vascular dementia; S: other specific dementia; UD: unspecified dementia; double dx: two, three, or four of the diagnoses AD, VD, S, or UD. Created with https://sankeymatic.com, modified.
Overall, the greatest switches were in patients with two diagnoses in the index quarter, the majority of whom had only AD or UD as a main diagnosis, and in patients with UD, 10.8% of which received AD as main diagnosis (Figure 2).
Medication
A total of 35.4% (n = 10,759) of patients ever received an ADD (subgroup analysis: 43.8% (n = 6658), Table 2). Fewer women than men had prescriptions (34.6% versus 37.2%, Table 2). The proportion of patients receiving ADDs was much lower in older patients than in younger ones (60–69-year-olds: 42.6% versus 90 + years: 16.9%, Supplemental Table 5).
Of all patients with ADD prescriptions, 73.2% were prescribed a drug within the first year of initial diagnosis (subgroup analysis: 69.7%), 2.2% of patients were not prescribed ADDs until 5 years or longer after initial diagnosis.
On average, patients who received ADDs were prescribed the drugs for 51% (subgroup analysis: 56%) of quarters since their initial diagnosis (range 2–100%), 29.9% (subgroup analysis: 27.5%) received the drug for 75–100% of quarters.
Of all patients with ADD prescription, 66.9% were prescribed ChEIs, 45.8% memantine, 7.5% Ginkgo, and 1.9% other anti-dementia drugs at least once (Figure 3, Supplemental Table 6). Patients may have had drugs from more than one drug class prescribed, but 70.5% had a prescription of only one drug group, 27.1% had prescriptions from two groups.
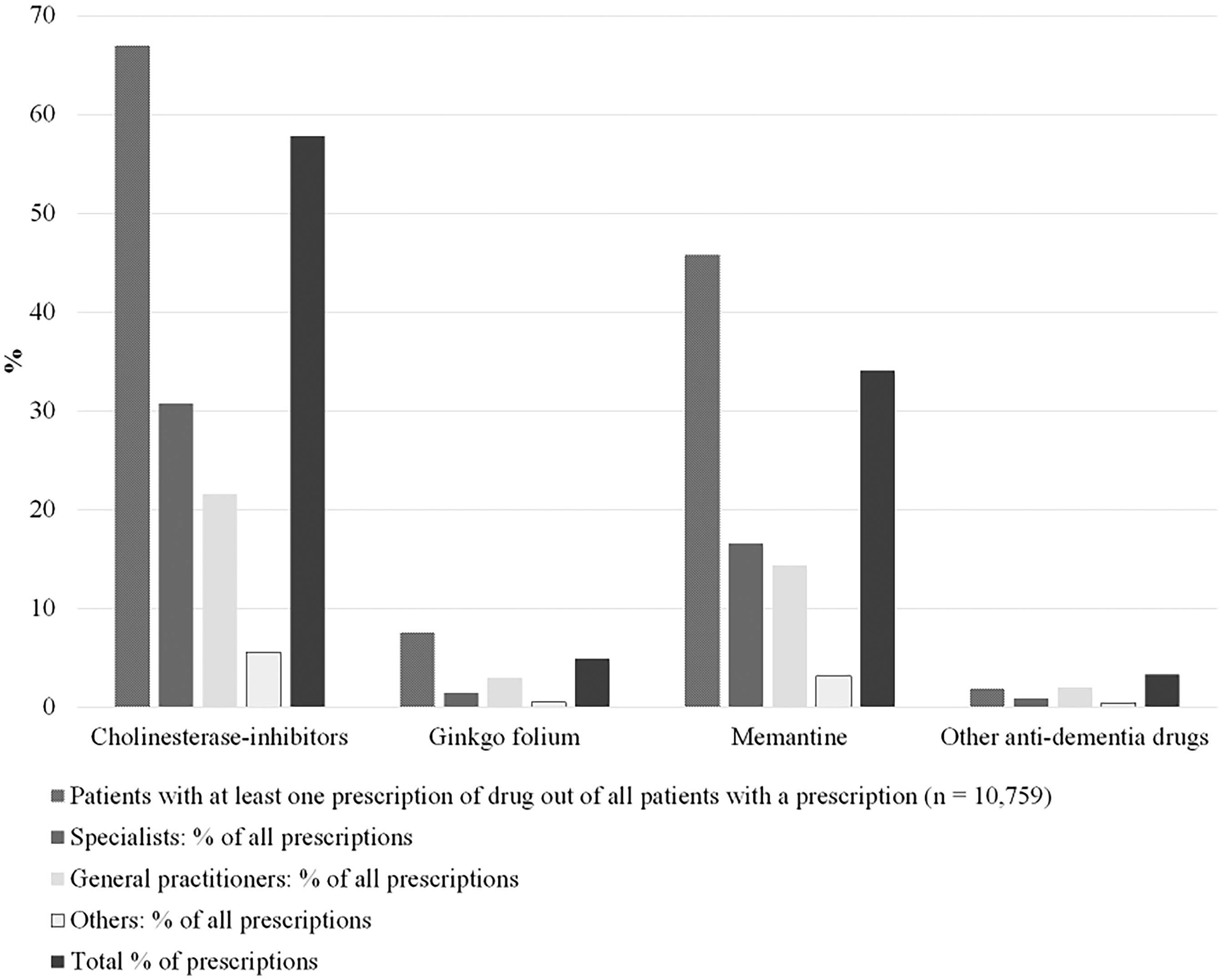
Antidementia drug type in patients with antidementia drug prescription (n = 10,759), stratified by professional group making the prescription. Health claims data analysis of 30,403 patients with dementia in Germany, diagnosed between 2006 and 2016.
The majority of ADDs were prescribed by specialists and GPs (49.6% and 40.9% respectively). Within each professional group, GPs prescribed 52.7% ChEIs and 35.2% memantine, specialists prescribed 61.9% ChEIs and 33.4% memantine, and other groups prescribed 57.8% ChEIs and 32.8% memantine. Medication given by hospital physicians was not available (Figure 3 and Supplemental Table 6).
Of all Ginkgo prescriptions, GPs had the greatest share (60.6%) albeit the total number of Ginkgo prescriptions was low (n = 7777, 4.9%, Supplemental Table 6).
Two thirds of patients with AD as main diagnosis were treated with an ADD (n = 6957, 67.4%, Supplemental Table 7). However, older patients received an ADDs less often than younger ones (60–69-year-olds: 73.6% versus 90 + years: 38.3%). The majority of AD patients with ADD treatment received cholinesterase inhibitors (n = 2915, 62.1%) or memantine (n = 1616, 34.5%) as main drug (Supplemental Figure 1, Supplemental Table 7). Moreover, 39.6% (n = 366) of patients with specific dementia, 26.8% (n = 17,154) of patients with UD, 20.6% (n = 5429) with vascular dementia as main diagnosis, and 43.9% (n = 497) with a double diagnosis were treated with an ADD. A total of 10.9% (n = 122) of patients with the main diagnosis VaD were treated with Gingko as main drug (Supplemental Table 7). For patients with UD as main diagnosis and an ADD prescription, 54.2% had a ChEI and 36.7% a memantine prescription.
Overall, 90.1% of patients with ADD use had already received the main drug in the first quarter of ADD prescription; there was no difference between men and women. A switch was more frequent in the younger age groups than in the older ones (60–69 years: 12.9%, 90 years and older: 5.9% (subgroup analysis: 13.9% versus 7.2%)).
Summary
Most patients had at least one diagnosis of UD (87.1%), and most received more than one diagnostic code of dementia (61.3%), e.g., UD and AD. Despite having several diagnostic codes of dementia, the initial diagnosis was identical to the main diagnosis in 70.5% of patients. Most diagnoses were made by general practitioners (60.2%). The most common diagnosis by specialists was AD (62.9%).
About a third of patients (35.4%) received an ADD, two thirds of which were prescribed ChEIs (66.9%) and almost half memantine (45.8%). There were few switches between the initial type of ADD prescribed and the main ADD prescribed over the course of treatment observation (9.9%). Two thirds of patients with AD as main diagnosis were treated with ADD (67.7%), and most of these received an ChEI (62.1%) or memantine (34.5%). Specialists made almost half the ADD prescriptions (49.6%), followed by GPs (40.9%).
Discussion
Diagnosis and professional groups
There seems to be uncertainty in the diagnostics of dementia. First, this becomes obvious because the majority of diagnoses were unspecified. This observation was also made in other studies on claims data and data from practices as well.4,6,9,13,19 Similarly, in Denmark, where patients with dementia are registered in two well-established national registries, 55.3% of a random sample of 200 dementia patients had a dementia diagnosis without specification (ICD-10 F03.9). 20 In our study, GPs disproportionately often diagnosed UD, whereas specialists made the most specific diagnoses. This could also be observed in other studies.21,22
The results of several surveys of GPs on challenges and problems with diagnosing dementia may give a partial explanation for the high number of UD diagnoses by this group. Among other reasons, some GPs seem to perceive differential diagnosis as an obstacle; they feel to have a lack of geriatric education.23–27 They have relatively small numbers of dementia patients and little diagnostic and therapeutic routine. 21 The majority of GPs find the S3 treatment guidelines only partially or somewhat helpful. 28 Moreover, many GPs delegate their patients to specialists.23–27 Thus, they limit their role to basic diagnostics. Alternatively, GPs can refer patients with suspected dementia to specialists without making a verified diagnosis themselves. Similarly, in the Netherlands, the GP is responsible for referring dementia patients for diagnostic follow-up. 29 According to a Dutch study on patients with a dementia diagnosis at age 70 or younger, GPs used the International Classification of Primary Care coding P70, which stands for “dementia”. 30 In our analysis, 62.9% of all diagnoses by specialists, who may have better possibilities for a more specific diagnosis, were AD. Specialists’ diagnoses may be the closest diagnoses according to clinical criteria: Different sources estimate that prevalent dementia cases are made up of 50–80% AD patients and 10–25% VaD patients.18,31,32 Many UD diagnoses may be AD, like in a Danish validity study on data of 200 dementia patients identified in two national registries whose diagnoses were compared to diagnoses validated through different steps like medical chart review. A total of 36 of 109 patients with unspecified dementia (F03.9) were found to have AD after the validation process. 20 Older patients had more unspecified diagnoses as main diagnosis, which may be due to several possible reasons: As the prevalence of frailty rises with old age,33,34 visiting a specialist and undergoing procedures like an MRI-scan or a lumbar puncture can represent a considerable expenditure and might therefore be refused by a patient or their relatives. Also, the consequences of the testings might be considered not very relevant to the therapeutic regime, e.g., because the patient refuses a therapy anyways, because of other diseases or circumstances, which limit the patient's therapeutic options.
Second, this analysis showed that more than half of patients received between two and four different diagnostic codes over the course of observation. To our knowledge, this has not been similarly investigated in other studies. Two studies covering the incidence year only observed 27–32% patients with multiple diagnostic codes.4,6 The lower percentage compared to our study is most likely due to the shorter time span. This is in line with the observation in our study, which showed that only 14% of patients had multiple diagnostic codes in an even shorter time span, the index quarter. Most studies did not include multiple diagnostic codes; they either restricted the diagnosis to the first one,4,9 restricted their analyses to AD,5,7,8,35 or had only one type of dementia recorded. 21
For a large part, multiple diagnoses arose when patients visited different professional groups. This is in line with a study by van den Bussche et al. 22 on diagnoses in the index year, in which 11.6% of GPs and 13.3% of specialists assigned multiple diagnostic codes, but in patients with diagnoses by both GPs and specialists, 69.9% had multiple diagnostic codes. It is possible that the initial dementia diagnosis was made as an additional/secondary diagnosis when patients were admitted to the hospital. This may explain the high proportion of hospital diagnoses in the index quarter (20.9%) compared to all quarters (8.3%).
Within a professional group, the disagreements concerning diagnoses are relatively low. The least ones are observed within specialist diagnoses, the greatest within hospital diagnoses. Multiple diagnoses within one professional group may arise when the same physician changes their diagnosis over the course of observation or assigns a second diagnostic code of dementia, or if a patient visits more than one physician with differing diagnoses of the same professional group. Multiple types of dementia may indeed arise in the same patient. The Alzheimer's Association 31 suggests that more than half of older dementia patients have more than one pathology underlying their dementia.
There is a high concordance between the index diagnosis and the main diagnosis.
Under the assumption that the main diagnosis is the true one, this observation is beneficial as different types of dementia must be treated in different ways. However, UD remained the most frequent diagnosis over the course of observation, and it is not defined in the S3 treatment guidelines 18 and thus lacks treatment recommendations. Anyhow, 16.7% of UD diagnoses from the index quarter were specified later on. Although this is still a low proportion, this specification is encouraging.
The specification may be due to several reasons. First, the unspecified diagnosis may have been specified at a later point in time or by a specialist. This may be obvious because the process of diagnosing dementia is a two-step-procedure according to national guidelines, 36 with the first step diagnosing dementia in general (according to ICD-10 (Table 1a) criteria for dementia via collecting medical history, making cognitive tests, and various assessments) 36 and the second one doing differential diagnostics, unless the patients objects this step. Upon a patient's first doctor's visit with symptoms of dementia, only the first step may be carried out, and it is GPs who usually make the diagnosis in the index quarter. A specification of an unspecified dementia would be best practice. Second, fewer diagnoses in the index quarter than over the entire course of observation were made by neurologists/psychiatrists who are specialists for dementia diagnoses.
To our knowledge, only two other studies investigated switches in dementia diagnoses in secondary data to date, both from Germany: in Kaduszkiewicz et al., 6 the analysis period was one year. This limits comparability to our results because the shorter the analysis period, the fewer multiple diagnoses are seen. Moreover, a main diagnosis was not defined. Similar to our results, only 9% out of all patients with multiple diagnoses saw a specification by the end of the analysis year. Bohlken et al. 21 investigated changes in diagnostic patterns between 2005 and 2015 by GPs and specialists, however, not within the same patient, which limits comparability as well. While there were little diagnostic changes by GPs, specialists diagnosed 16% more AD and 9% less UD in 2015 compared to 2005.
Medication
ADD treatment according to the S3 guidelines 18 may help to slow down the development of the disease. Overall, there are indications for an adherence to the guidelines, but there are also indications of an under-treatment.
The ADD treatment rates for dementia patients in Germany generally seem not to exceed 50%. Thirty-five percent of patients had a prescription in the current analysis, 24.6% in a nationwide health claims analysis of outpatient services on all patients statutorily insured in Germany in 2011. 4 In the US, a third of patients with dementia or dementia-related symptoms received ChEI or memantine in a study on Medicare data (2008–2016). 37 Similarly, 38% of patients with dementia (age ≥65) received ADD in a cross-sectional study of the French national health insurance database in 2013. 38 A total of 50.5% (n = 283) of patients treated within a dementia network in Germany, a model of integrated care aiming at providing adequate treatment, 14 had a prescription.
Specialists made the majority of prescriptions in our study although only 34.1% of patients visited a specialist. This is similar to a nationwide study from Germany, in which patients treated by a specialist only or together with a GP received an ADD prescription more often (48.6% and 48.4%, respectively) than those treated exclusively by a GP (24.5%). 4 One reason might be that patients treated by specialists have more advanced disease stages than those not treated by specialists.
According to the guidelines, an ADD treatment is clearly recommended only for AD patients, 18 two thirds of whom did receive an ADD, and most of them within the first year of diagnosis. This agrees largely with international observations: 70.4% (n = 2105) of AD patients in a registry-based cohort study in Spain were prescribed an ADD at diagnosis 5 ; 66.1% (n = 6358) of patients from the Swedish Dementia Registry received ChEI and 12.0% received memantine (n = 1154) 8 ; only 3% of AD patients did not use any ADD during a 4-year follow-up period in a nationwide cohort study from Finland, 7 and 68% AD patients received an ADD in a cross-sectional study from Russia with 12,051 dementia outpatients. 39 In a German dementia network, 72.0% of AD patients were treated. 14 On the other hand, in the German nationwide study by Bohlken et al., 4 only 42.4% of AD patients had at least one ADD prescription in 2011. This may partially be explained by the shorter time frame in the study.
There was a decreased rate of treatment with ADD in patients with a late onset of the disease for all types of dementia. This was also observed in other studies. In the nationwide analysis with prevalent dementia patients from Germany in 2011, old patients, but also young ones had fewer drug prescriptions compared to 75–79-year-olds (31.9%, 13.0% in patients 90 and above, 18% in 60–64-year old). 4 Similarly, old age of 85 years and above was significantly associated with a lower ChEI use in AD patients compared to 65–74-year-olds in the Spanish cohort study. 5 This might be due to the fact that comedication because of other diseases may cause interactions. 40 Another reason may be the fear of adverse effects of the drug which may outweigh the drug benefit. One more reason may be the on average worsening health status with age. A poor health status was associated with receiving fewer ADD in the French cross-sectional study. 38 In contrast, the prevalence of ADD use peaked in the age group 80–89 years in an analysis of a cohort including 98% of all insured persons in Austria. 41
The majority of patients with VaD were not treated with an ADD. It is possible that they were treated for the underlying disease and vascular risk factors, which is the only drug use recommendation in the guidelines.
About three quarter of UD patients were not treated with ADDs. UD is not mentioned in the S3 guidelines. 18 Nevertheless, of those treated, most received ChEIs. ChEIs can be used to treat dementia in patients with Parkinson's disease. Another study on dementia networks found that 41% of patients with UD were treated with ADD. 14 Few patients were treated with other anti-dementia drugs. Although these drugs are listed as ADD in the Anatomical Therapeutic Chemical Classification System (compare Table 1), these drugs have indications such as circulatory disorders or vertigo.
Different sources report that 50–80% of all prevalent dementia cases have AD.18,31 Therefore, a large share of the patients classified as having UD in the data analyzed are supposedly AD patients, and they are not treated for the larger part.
Patients treated with ADDs were treated rather consistently with one type of drug. Also, there were few switches between the drug initially prescribed and the main drug prescribed over the course of observation. A more detailed analysis 10 on the continuity of ADD treatment in Germany found that nearly 60% of patients continued treatment after one year, i.e., they neither changed drug type (one of three ChEI or memantine) nor had more than 90 days treatment gap with the initial doctor. Continuation was associated with treatment by a specialist compared to a GP.
Subgroup analysis
There was a slightly higher percentage of women in the subgroup analysis most likely because women both with and without dementia survive longer on average42,43 and could fulfill the 4-year-survival criterion to a higher degree. Similarly, patients in the subgroup analysis were slightly younger on average because the oldest old from the main analysis could not fulfill the 4-year-survival criterion.
Overall, the medication rate of the subgroup of patients who survived longer was noticeably higher than that of the entire study population. Younger average age may have contributed to having more drug prescriptions, which is in line with both our observation that patients from the oldest age group received fewer ADDs than younger age groups (as discussed above) and another study on ADD drug prescription in Germany, which showed that every year of age reduced the likelihood of receiving appropriate prescription. 44
Other than that, differences in the diagnostic experience between the two groups were minor and might be explained by the fact that patients who survived longer had more time and thus more opportunities, e.g., to visit more different types of professional groups.
Strengths and limitations
This study has several strengths. It was a longitudinal analysis of a large population-based sample over a long insurance period. Therefore, the results can be generalized to the population of 60 years and above in Germany. The data included in- and outpatient diagnoses as well as medication from four groups. There was no recall bias or nonresponse bias. Continuous data from 2004 until end of study, death or exit from insurance company was available. A subgroup analysis addressed some of the selection bias due to the inclusion criterion of minimum survival time. The study results can be generalized with respect to health claims data in Germany. The international generalizability depends on the data structure of health claims data and on the structures of care for dementia patients, e.g., whether there are many specialized centers or programs.
On the other hand, there are also some limitations. The diagnostic certainty for dementia diagnoses in the claims data used is a challenge. Unspecified and multiple types of diagnoses and their issues were discussed in this paper. They constituted a large part of the diagnoses although only inpatient and verified outpatient diagnoses were used. Claims data are in this way different from registry data from Spain, 5 Sweden, 8 or Finland 35 where only clinically verified AD patients were registered. The temporal resolution of the data does not allow to detect whether two diagnoses were made on the same day. Moreover, information on stage of disease, which might be related to diagnosis, ADD use, and professional groups, was not available.
An underdiagnosis of dementia is possible due to the criterion that 75% of insured quarters since initial diagnosis had to have a diagnosis. On the other hand, this avoided the inclusion of false positive dementia diagnoses.
The available data on filled prescriptions cannot necessarily be equated with the true ADD intake. Data on medication intake by inpatient patients was not available.
This study informs future analysts of dementia subtypes in German claims data on possibilities and limitations of their data sources, such as defining a main diagnosis, investigating time trends, and the potential challenge of detecting under-diagnosis and -treatment.
The majority of dementia diagnoses were unspecified. Specialists made more specific diagnoses; 63% of their diagnoses were AD. This agrees with estimates from different sources, which quantified 50–80% of dementia cases to have AD.18,31 Thus, there may be an underdiagnosis of specific dementia types, which might be alleviated by more specialist diagnoses or better diagnostic training of GPs. However, two thirds of patients never visited a specialist.
A more targeted diagnosis may also improve dementia treatment. ADD treatment is recommended only for AD patients, 18 and two thirds of AD patients were prescribed some ADD, at least. Overall, however, only 35% of all dementia patients had an ADD prescription. Assuming that a portion of patients with UD truly have AD, there is a clear undertreatment of a portion of patients.
Specialists do not only make more specific diagnoses but also made the majority of ADD prescriptions. Medical care by specialists therefore seems to be advisable for patients with dementia. However, since a relatively low number of specialists are facing a growing number of dementia cases, this is a challenge: Dementia networks may be expanded, as the treatment rate in a German dementia network 14 was closer to what seemed advisable according to guideline recommendations. Furthermore, geriatric education of GPs may be strengthened to foster diagnostics.
Supplemental Material
sj-docx-1-alz-10.1177_13872877251319468 - Supplemental material for Dementia diagnosis and prescription of antidementia drugs: An analysis of German claims data (2006–2016)
Supplemental material, sj-docx-1-alz-10.1177_13872877251319468 for Dementia diagnosis and prescription of antidementia drugs: An analysis of German claims data (2006–2016) by Cornelia Becker, Lucas Herschung, Willy Gomm and Britta Haenisch in Journal of Alzheimer's Disease
Footnotes
Acknowledgments
We are grateful to the Scientific Research Institute of the AOK, WIdO, for providing the data.
Author contributions
Cornelia Becker (Data curation; Formal analysis; Methodology; Visualization; Writing – original draft); Lucas Herschung (Conceptualization; Data curation; Formal analysis; Methodology; Visualization; Writing – review & editing); Willy Gomm (Conceptualization; Methodology; Supervision; Writing – review & editing); Britta Haenisch (Conceptualization; Funding acquisition; Supervision; Writing – review & editing).
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability
Data cannot be shared publicly or be transmitted to a third party due to legal restrictions regarding claims data.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
